Abstract
Objectives
The aim of this study was to evaluate the effectiveness of ovariohysterectomy (OVH) on feline mammary fibroepithelial hyperplasia (FMFH) treatment, as well as the influence of previous injectable progestin on clinical signs and treatment of FMFH.
Methods
Seventy-nine female cats diagnosed with FMFH between 2014 and 2018 were included. The animals were distributed as follows: (1) treated only with OVH (TA) or OVH plus antiprogestin (TB); and (2) no administration (G1) or previous administration (G2) of injectable progestin before FMFH diagnosis. Data regarding clinical signs of FMFH, occurrence of persistent mammary growth after OVH, and time until complete remission of FMFH post-surgery were recorded and evaluated.
Results
Mastectomy was not performed in any cat during the study. TA was adopted in 71/79 (89.9%) cases, with 42/71 (59.2%) from G2 and 29/71 (40.8%) from G1. TB was adopted in 8/79 (10.1%) cats, all from G2. Total remission of FMFH was observed in 73/79 (92.4%) cats, with six animals from G2 having died (n = 6/79 [7.6%]). The time until complete remission of FMFH was higher (P <0.0001) in cats from G2 than from G1. Moreover, animals from G2 had a higher percentage of persistent mammary growth after OVH (P <0.001) and a tendency to higher mortality (P = 0.052) compared with G1.
Conclusions and relevance
Previous treatment with injectable progestin may result in a more complex clinical presentation of FMFH. Ovariohysterectomy is a good treatment option for FMFH when there is no interest in breeding the queen. Treatment with antiprogestin is recommended, especially if persistent mammary growth is detected after OVH.
Introduction
Feline mammary fibroepithelial hyperplasia (FMFH) was first characterized in 1973, and progesterone is considered an important trigger for the development of FMFH.1,2 Macroscopically, FMFH is characterized by enlargement of the mammary tissue, hemorrhages, ulceration and necrosis.3,4 The diagnosis is usually clinical and takes into account patient age, rapid growth of the mammary glands and medical history (particularly the use of progestins).1,4–6 Although progesterone is associated with FMFH pathogenesis, serum progesterone concentration is not a sensitive diagnostic marker. By the time of diagnosis, progesterone assay results do not allow clinicians to differentiate between endogenous or exogenous progesterone.2,3
During FMFH, epithelial and mesenchymal tissues present higher proliferative activity than normal mammary gland, and colocalization of progesterone receptors and growth hormone (GH) was detected in mammary gland cells of cats with FMFH.7,8 The mammary gland is considered an extra-pituitary source of GH, a hormone able to release insulin-like growth factors, which are active mitogens commonly expressed in mammary fibroblasts. 9 As progesterone induces GH release, the pathogenesis of FMFH might be related to the synergism between progesterone, GH and insulin-like growth factor in the mammary tissue.6,7,10,11 Because of its close relationship with progesterone, reports of FMFH are more common in female cats. However, FMFH has also been described in male cats and other species, such as cows and rats.12–17
Ovariohysterectomy (OVH) has been documented to treat FMFH, but other therapeutic approaches have been adopted, including antiprogestin drugs or even mastectomy.6,18,19 Nowadays, mastectomy should be avoided as antiprogestin drugs are often effective to treat FMFH, although treatment can take as long as 5 weeks and recurrence of FMFH after antiprogestin treatment has been reported.2,12,20,21
Therefore, the aim of this study was to evaluate the effectiveness of OVH, or its combination with antiprogestin, to treat FMFH in a large set of patients. The influence of injectable progestin on the clinical signs of FMFH and treatment effectiveness was also assessed.
Materials and methods
This study selected and reviewed the records of 79 privately owned female cats by a non-profit veterinary organization for pet population control. The diagnosis of FMFH was both clinical and cytopathological. 5 After diagnosis and wound care, all the animals were initially subjected to treatment A (TA, OVH). Patients with mammary tissue lesions, 48 h before OVH, were given amoxicillin + clavulanic acid (15.0 mg/kg PO q12h) and ketoprofen (1.0 mg/kg PO q24h), plus topical treatment with chlorhexidine 0.1% and bacitracin–neomycin ointment q24h.
On the day of surgery, hair in the surgical area was clipped and the skin was aseptically prepared. The anesthesia protocol was performed with tramadol (2.0 mg/kg IM), xylazine (1.1 mg/kg IM) and ketamine (22.0 mg/kg IM). The surgeries were performed with a spay hook after ventral midline celiotomy, but the flank approach was adopted in cases of excessive mammary enlargement.22,23 After surgery, animals were given amoxicillin + clavulanic acid (15.0 mg/kg PO q12h for 8 days) and ketoprofen (1.0 mg/kg PO q24h for 4 days), plus topical treatment as previously described. The owners were advised about the importance of keeping the cats in the recovery suit until healing was completed.
After OVH, the animals were clinically evaluated at 7–10-day intervals until complete remission of FMFH was noted. In cases of persistent mammary growth for up to 3 weeks after OVH (TA), cats were given injectable antiprogestin (10.0 mg/kg SC q24h for 4 days), moving them on to treatment B (TB).
Additionally, animals were independently placed into one of two groups based on a history of no oral or injectable progestins (G1), or when the owner informed us that only injectable progestin was given (G2) before the diagnosis of FMFH.
Statistical analysis
Data regarding animal age, previous treatment with injectable progestin, interval between last progestin administration and first clinical signs of FMFH, presence and severity of mammary lesion, mammary size after the onset of treatment (involution or persistent growth), time until complete involution of FMFH and treatment effectiveness were recorded.
As few cats were submitted to TB, the main clinical signs and therapeutic efficacy of TA and TB were evaluated using descriptive analysis. The influence of previous treatment with injectable progestin on mortality (%), presence of mammary ulceration/necrosis (%) and persistent mammary growth after OVH (%) were compared between groups using the χ2 test. The Mann–Whitney test was used to compare the animal’s age between groups, as well as to evaluate the FMFH total remission time between G1 and G2 and TA and TB. Age and remission time are presented as median (range). All analyses were performed with GraphPad Prism (version 6.0) software and differences were considered significant if P <0.05.
Results
Of the 79 cats included, neither oral nor injectable progestins were reported in 29 (36.7%) cats in G1, whereas 50 (63.3%) cats in G2 were previously given injectable progestin. Injectable progestin was mainly given by pet store assistants (n = 30/50 [60.0%]), followed by owners (n = 18/50 [36.0%]) and veterinarians (n = 2/50 [4.0%]). There was no significant difference (P >0.05) in the average age between cats in G1 (7 months [range 6–12 months]) and G2 (8 months [range 6–72 months]). The interval between progestin injection and the first clinical signs of FMFH in cats in G2 ranged between 7 and 30 days.
A shorter FMFH remission time (P < 0.05) was observed in the cats that received TA (49 days [range 28–180 days]) vs those that received TB (99 days [range 90–130 days]). After OVH, persistent mammary growth was observed in 19/79 cats (24.0%); all but one (n = 18/19 [94.7%]) were previously exposed to injectable progestin (Figure 1). The main clinical signs of the cats diagnosed with FMFH and the therapeutic effect are summarized in Table 1.

Case 42 – a 1-year-old cat that was given injectable progestin, and that then underwent ovariohysterectomy (OVH) and received antiprogestin to treat feline mammary fibroepithelial hyperplasia (FMFH). (a) Before OVH: an intense FMFH with an inflammatory aspect. (b) Fourteen days after OVH, with persistent mammary growth, cutaneous distension and necrosis. (c) Fifteen days after OVH, with cutaneous rupture in the first pair of mammary glands. (d) Remarkable mammary involution, 35 days after OVH and 2 weeks after antiprogestin treatment. (e) Sixty-three days after OVH, with discrete mammary hyperplasia and an absence of cutaneous lesions. (f) Total involution of FMFH, 92 days after OVH
Main clinical signs of 79 cats diagnosed with feline mammary fibroepithelial hyperplasia without (G1) or with (G2) previous exposure to injectable progestin, treated with ovariohysterectomy only (TA) or ovariohysterectomy and antiprogestin (TB)

Data are n (%)
PCN = presence of cutaneous necrosis; PMG = persistent mammary growth after ovariohysterectomy
Six cats from G2 died during the study (n = 6/79 [7.6%]) because of sudden death (n = 4/6 [66.7%]) or hemorrhage (n = 2/6 [33.3%]), according to the owners’ reports. All cats that died had persistent mammary growth after OVH. Total remission of FMFH was observed in the remaining 73 cats. The adoption of TA was effective in the majority of these cases (n = 67/71 [94.4%]; Figure 2) and the combination of OVH with antiprogestin was necessary only in five cats (n = 5/71 [5.6%]) that were previously given injectable progestin (G2).
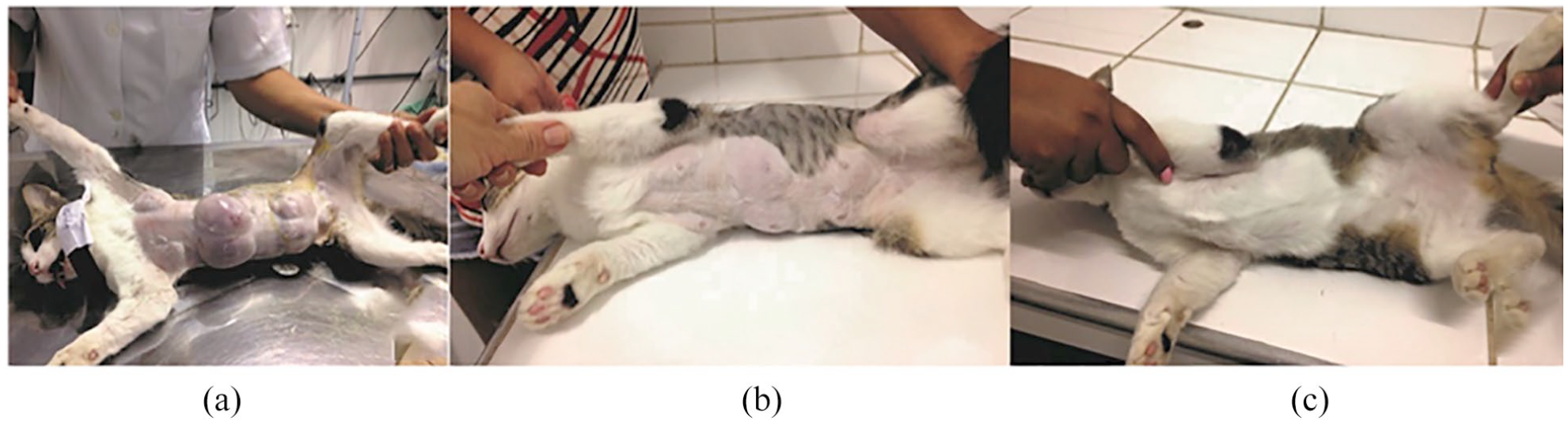
Case 20 – a 6-month-old cat with no previous exposure to injectable progestin treated exclusively with ovariohysterectomy (OVH). (a) Before OVH: mammary fibroepithelial hyperplasia in all mammary glands, with no cutaneous complications. (b) Eleven days after OVH, with remarkable mammary involution. (c) Total remission 30 days after OVH
The use of injectable progestin (G2; Figure 3) was associated with a higher percentage of persistent mammary growth (P = 0.001) and a longer interval between OVH and total remission of FMFH (P = 0.0001), as well as a tendency to higher mortality (P = 0.052), compared with animals with no report of injectable progestin (G1) (see Table 2).
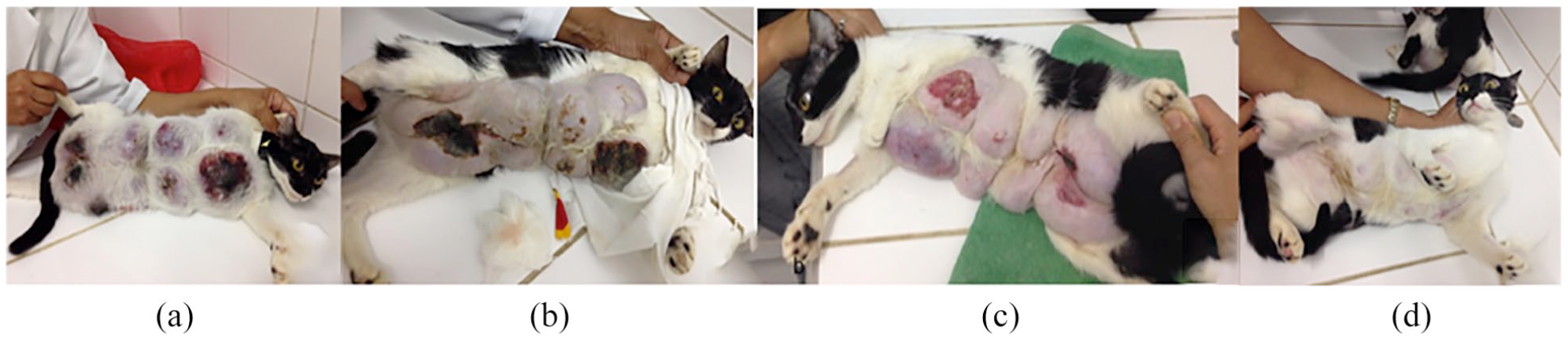
Case 21 – a 7-month-old cat, previously exposed to injectable progestin, that underwent ovariohysterectomy (OVH) to treat feline mammary fibroepithelial hyperplasia (FMFH). (a) Before OVH: generalized FMFH, with cutaneous distension and inflammation. (b) Ten days after OVH, with cutaneous ulcers and necrosis in the mammary glands. (c) Forty-two days after OVH, with mammary involution and granulation tissue in the mammary glands. (d) Total remission of FMFH, 98 days after OVH
Differences in clinical signs and therapeutic response of 79 cats diagnosed with feline mammary fibroepithelial hyperplasia, with or without previous administration of injectable progestin

For the calculation, cats that died were not included
PMG = persistent mammary growth after ovariohysterectomy; PCN = presence of cutaneous necrosis
Discussion
Synthetic progestin has been used for decades as a contraceptive.24,25 This study considered only the use of an injectable formulation of progestin (medroxyprogesterone acetate), as this is the most common progestin available in Brazil, has an affordable price (<US$1.00) and can be ordered without a prescription, facilitating inappropriate use, which probably increases the occurrence of FMFH.6,26 This problem is highlighted in the present study as injectable progestin was not given by veterinarians in 96.0% of the cats in G1. Reasons to choose OVH over antiprogestin as treatment for FMFH were: (1) that the owners had no further interest in breeding the cats; (2) most of the cats had access to an outdoor area and were at risk of an additional progestin injection in the future, to avoid undesired mating; and (3) the extra cost related to antiprogestin treatment.
Although the age of the cats with FMFH was statistically similar between G1 and G2, all the cats from G1 aged ⩽1 year and all cats with FMFH >1 year of age were given injectable progestin (G2). The occurrence of FMFH is most common in young cats,2,21 and the prevalence can be increased if progestin is adopted as a reversible method to prevent undesired pregnancy in early reproductive life.1,20,27–29 It is plausible that the occurrence of FMFH in adult and geriatric cats from G2 is associated with injectable progestin, as observed in other studies.1,12 The interval between progestin injection and the development of FMFH in the present study is in agreement with the ranges previously reported (between 2 and 90 days).20,28,29 The broad range observed is probably influenced by the reproductive status of the cat when progestin is given, as exogenous progestin acting together with endogenous progesterone can exacerbate the response of the mammary gland. 6
The use of injectable progestin was associated with more severe clinical signs of FMFH, such as persistent mammary growth, delayed remission time and a tendency to higher mortality. The differences observed between cats from G1 and G2 could be explained by the capability of progestins to stimulate cellular proliferation in mammary tissue,8,28 as mammary gland is commonly the first tissue to present adverse effects after exposure to progestin.25,30–33 Considering the rapid enlargement of the mammary gland during FMFH, 21 we inferred that the cat owners sought veterinary assistance shortly after clinical signs began, resulting in no significant difference regarding the presence of cutaneous necrosis.
In the present study, a longer time to total remission of FMFH was observed after TB than with TA. Previous studies reported similar remission times, comparing OVH only with antiprogestin treatment,12,31,33 and when exogenous progestin was the primary cause of FMFH, complete remission was observed approximately 4.7 weeks after starting antiprogestin treatment. 2 Most likely, our decision to start antiprogestin treatment only after 3 weeks of persistent mammary growth post-OVH delayed FMFH remission in the cats subjected to TB. The presence of residual connective tissue after hyperplasia collapse is common and edema reduction can take several weeks, resulting in a perimammary residual volume that can be misinterpreted as a persistent mammary growth. 12
All cats that died were given injectable progestin and presented persistent mammary growth after OVH. Four cases were reported as sudden death and might be a consequence of thromboembolism caused by cutaneous necrosis. 3 In the two cases of hemorrhage, trauma after scratching the mammary gland was reported by the owners. In the past, the adoption of mastectomy as a therapeutic method was indicated because of acute and severe mammary gland growth, occurrence of ulcer/necrosis and ambulation problems, despite the benign characteristics of FMFH.6,8,13,14,34 Bearing in mind the effectiveness of OVH in the present study, the current availability of antiprogestins and the fact that mastectomy is a very invasive surgery, we think that mastectomy is no longer necessary during treatment of FMFH, which is in agreement with previous reports.2,21,35 However, it is important to emphasize that proper wound management and permanently wearing the recovery suit are essential to avoid severe infection/sepsis and hemorrhage.28,33,35,36
OVH resulted in a total FMFH remission rate of 94%, which is similar to previous studies that adopted the early administration of antiprogestin.2,31,37,38 However, considering the complications of FMFH, and the fact that all cats that died were given injectable progestin and presented persistent mammary growth after OVH, we suggest the occurrence of two clinical presentations of the disease: non-complex (endogenous origin) and complex FMFH (exogenous origin). In the latter case, because of the delayed remission associated with a higher percentage of persistent mammary growth and mortality, we recommend using antiprogestin whenever no reduction of the mammary glands is observed after OVH (see the supplementary material).
Conclusions
Previous treatment with injectable progestin may result in a more complex clinical presentation of FMFH. OVH is a good treatment option for FMFH when there is no interest in breeding the queen. Treatment with antiprogestin is recommended, especially if persistent mammary growth is detected after ovariohysterectomy.
Supplemental Material
Supplemental Material
decision tree for FMFH treatment
Footnotes
Supplementary material
The following file is available online: Decision tree for FMFH treatment.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES), Finance Code 001.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognized high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animals described in this work for the procedures undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
