Abstract
Practical relevance:
Feline leukaemia virus (FeLV) is a retrovirus of domestic cats worldwide. Cats lacking strong FeLV-specific immunity and undergoing progressive infection commonly develop fatal FeLV-associated disease. Many aspects of FeLV infection pathogenesis have been elucidated, some during more recent years using molecular techniques. It is recommended that the FeLV status of every cat is known, since FeLV infection can influence the prognosis and clinical management of every sick cat. Moreover, knowledge of a cat’s FeLV status is of epidemiological importance to prevent further spread of the infection.
Clinical challenges:
Diagnosing FeLV infection remains challenging due to different outcomes of infection, which can vary over time depending on the balance between the virus and the host’s immune system. Furthermore, testing for FeLV infection has become more refined over the years and now includes diagnostic assays for different viral and immunological parameters. Knowledge of FeLV infection pathogenesis, as well as the particulars of FeLV detection methods, is an important prerequisite for correct interpretation of any test results and accurate determination of a cat’s FeLV status.
Aims:
The current review presents recent knowledge on FeLV pathogenesis, key features to be determined in FeLV infection, and frequently used FeLV detection methods, and their characteristics and interpretation. An algorithm for the diagnosis of FeLV infection in a single cat, developed by the European Advisory Board on Cat Diseases, is included, and FeLV testing in specific situations is addressed. As well as increasing awareness of this deadly infection in domestic cats, the aim is to contribute diagnostic expertise to allow veterinarians in practice to improve their recognition, and further reduce the prevalence, of FeLV infection.
Keywords
Feline leukaemia virus – still an enigma
Feline leukaemia virus (FeLV) is a well-known pathogen occurring worldwide in domestic and small wild cats.1,2 It is a gammaretrovirus that was first described as ‘a virus-like particle associated with leukaemia (lymphosarcoma)’ over 50 years ago by Jarrett et al. 3 FeLV infection can cause immunodeficiency, cytopenias and neoplasia in cats with the progressive form of the disease.4–10 The use of increasingly robust and accurate diagnostic assays to identify FeLV-infected cats and the development of efficacious FeLV vaccines has led to a reduction in the prevalence of FeLV infection in domestic cats in many geographic areas,7,11–13 although more recently stagnation in the decrease of FeLV prevalence has been reported.14–16 The risk remains that FeLV infection can spread quickly, particularly within naive multi-cat environments, if not recognised promptly. This review summarises recent developments that are of clinical relevance – notably in diagnostics as well as in the understanding of infection pathogenesis.
FeLV exposure and infection outcomes
Like all retroviruses, FeLV is an enveloped RNA virus. It carries an enzyme (reverse transcriptase) that reverse transcribes the viral RNA genome into a DNA form, which is then integrated into the host’s cell genome as provirus by another enzyme (integrase).1,2,17 In addition to exogenous FeLV, several endogenous retroviruses have been identified in domestic cats.1,18,19 They are present in every cat and are part of the host’s genome; thus, they are inherited by the cat’s offspring, but usually do not form infectious or pathogenic viruses by themselves. From a diagnostic point of view, it is important that FeLV tests which can discriminate exogenous FeLV from endogenous FeLV-like sequences are used; this is particularly a concern for molecular assays.20–22
JFMS Premier Reviews are invited state-of-the-art review papers on key issues in feline medicine and surgery. Written by expert international authors, these reviews are made freely available to maximise their impact.
FeLV is shed in large quantities in saliva,23–25 but it can also be found in faeces, urine and milk.25–28 FeLV is unstable in the environment, and therefore transmission is thought usually to require intimate friendly or aggressive contact between infected and naive cats.2,15,29 Indirect contact with saliva or, to a lesser extent, faeces from FeLV-infected cats can also be sufficient to transmit the infection (eg, via sharing of food bowls or litter boxes).28–30 In addition, FeLV can be transmitted vertically from an infected queen to the kittens.

FeLV infection usually starts in the mucosa of the oropharynx. Subsequently, viral replication takes place in the adjacent tonsils and local lymph nodes.31,32 The virus is spread throughout the body via infected lymphocytes and monocytes in the lymphoid tissue (primary viraemia). 33 Replication in the bone marrow, which involves infection of neutrophil and platelet precursors, leads to the initiation of secondary viraemia and systemic infection.31,32
Cats of all ages can become infected with FeLV. However, susceptibility to progressive FeLV infection is to some degree age-dependent, with kittens being more prone to develop progressive FeLV infection than adult cats.34,35Adult cats can nevertheless also become progressively FeLV infected. 36

Progressive FeLV infection
Progressively infected cats have bone marrow involvement leading to the establishment of a secondary (and persistent) viraemia, in which granulocytes and platelets (as well as lymphocytes and monocytes) in the peripheral blood are FeLV-infected (Figure 1).33,37 Progressive infection is characterised by persistent viraemia/antigenaemia and the absence of an efficient FeLV-specific immune response.34,35 In other words, the virus permanently gains the upper hand (Figure 2). Cats with progressive FeLV infection are clinically and epidemiologically the most important ones to identify. These cats shed high numbers of FeLV particles and pose an infection risk to other cats. They should be kept separated from FeLV-naive companions, regardless of the health status of the FeLV-infected cat.
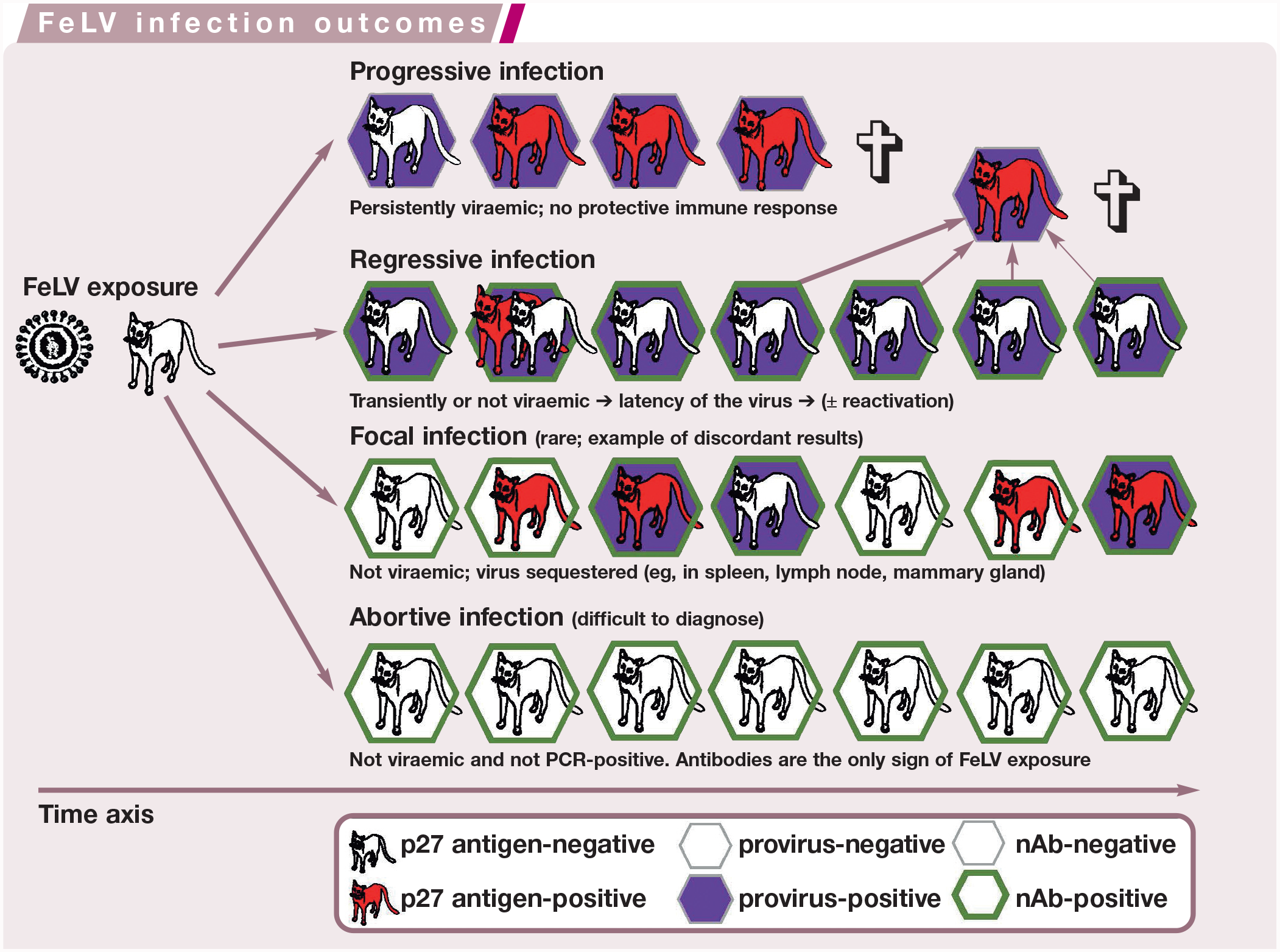

Schematic diagram showing the time course after feline leukaemia virus (FeLV) exposure of a cat and the four potential FeLV infection outcomes (progressive, regressive, focal [rare] and abortive infection). Cats are depicted according to their FeLV p27 antigen (red), FeLV provirus DNA (purple) and neutralising antibodies (nAb; green) status. For regressive infection, the potential for reactivation (recurrence of viraemia and virus shedding in previously FeLV p27 antigen-negative [aviraemic] cats) decreases with time. ✞ = death


It can be helpful to think of the outcomes of feline leukaemia virus (FeLV) infection in terms of a set of balance scales, with the cat’s immune response on one side and the virus on the other. In abortive FeLV infection, the cat has the upper hand (more weight on the balance scales); in progressive FeLV infection, the virus has the upper hand. In regressive infection, the cat’s immune system can keep the virus in check so that no, or only very limited, viral replication takes place, although reactivation (the recurrence of viraemia and virus shedding) can occur. In focal infection (rare), the cat’s immune system keeps viral replication sequestered in certain tissue(s). For each infection outcome (abortive, regressive, focal and progressive) the magnitude of the anti-FeLV immunity (pink), provirus integration (blue), virus replication (green) and the potential to induce FeLV-associated disease (grey) is shown. The three boxes with graduated colour indicate the possibility of either positive or negative status
Progressive infection is usually confirmed by repeated testing of the cat for antigenaemia several weeks or months apart;2,7,38 only repeated positive antigen test results verify the presence of a progressive infection (Figure 1). Notably, in a few cats, the progressive FeLV infection status can take several weeks to develop after initial FeLV contact (Figure 3; eg, cats 6 and 11) and/or cats can have p27 antigen test results that alternate between negative and positive (here-after referred to as ‘alternating’), particularly during early infection before the host–virus balance finds a steady-state (see Figure 2 and Figure 3 [cats 6, 7, 8, 11, 12, 13 and 15]).
Progressively infected cats have a poorer prognosis than cats with regressive FeLV infection (Table 1). They are at high risk of succumbing to potentially fatal FeLV-associated diseases, sometimes within just a few months (Figure 3 [cat 14]).4–8 ,15,41 Nonetheless, many progressively infected cats can continue to live a healthy and happy life for many years, if well cared for.
Characterisation of possible outcomes of feline leukaemia virus (FeLV) infection and test results defining them

Key to colours: pink = positive test results/status; green = negative test results/status; yellow = alternating/variable test results/status POC = point-of-care; IFA = immunofluorescence assay
Adapted from the ABCD FeLV diagnostic tool (see Figure 4), available at abcdcatsvets.org/wp-content/uploads/2017/12/Tool_ABCD_FeLV_diagnosis_2017.pdf

Examples of free p27 antigen and provirus results in 15 feline leukaemia virus (FeLV)-infected cats with different (in some cases exceptional) FeLV infection courses. All cats had been experimentally infected with the same virus strain (FeLV-A/Glasgow-1).20,36,39,40 Not all results are available at all time points: the early studies pre-dated the introduction of FeLV provirus PCR,36,40 and long-term follow-up was not available for all cats. Among the cats representing regressive FeLV infection, cats 2, 3, 8, 12, 13 and 15 had undergone transient viraemia. Some notable results are described in the box below. Repeated p27 antigen testing and quantification of proviral loads can help predict the final FeLV infection outcome in an individual cat. Reactivation refers to the recurrence of viraemia and virus shedding in previously FeLV p27 antigen-negative (aviraemic) cats

Regressive FeLV infection
Cats with regressive infection (Table 1) have developed a partially effective antiviral immune response31,38,39,42,43 and have recovered from the primary viraemia (Figure 2). Most regressively infected cats do not undergo bone marrow infection (with infection of neutrophil and platelet precursors), and so have only lymphocytes and occasional monocytes that are provirus-positive, and no viral RNA usually detectable in peripheral blood cells. 37
Clearance of antigenaemia is observed mostly within 1–12 weeks; 40 in rare cases, it can take many months (Figure 3, cat 4), 40 although the likelihood of clearance of viraemia decreases with time. Occasionally, outcomes can be observed with cats not following the defined FeLV infection courses; some of these cats can test transiently antigen-negative after being positive during the initial antigenaemia and can later become persistently positive as progressive infection establishes (Figure 3, cats 6, 7 and 11).39,40 Clearance of FeLV viraemia depends on the balance between the cat’s immune system and the virus (Figure 2) and can be influenced by many factors, such as the age and immune status of the cat, concurrent stressors, coinfections, the specific virus isolate and the exposure level. To determine whether a cat that initially tests positive for FeLV antigen undergoes regressive or progressive FeLV infection, repeated testing for FeLV antigen is necessary (regressively infected cats will eventually test antigen-negative, while progressively infected cats will continue to test antigen-positive). Cats with regressive infection usually do not develop FeLV-associated disease, although lymphoma or bone marrow suppression have been described in some cats with regressive infection.44,45
Early after FeLV infection, no difference is present in proviral and plasma viral RNA loads as determined by real-time PCR and real-time RT-PCR between cats with different infection outcomes (ie, regressive vs progressive infection). 6 Thus, at very early time points after FeLV exposure, FeLV proviral or plasma viral RNA loads cannot be used to differentiate regressively from progressively infected cats. However, a few weeks after FeLV exposure, cats with regressive infection have lower proviral blood and plasma viral RNA loads than progressively infected cats.4,20,38,46,47 Therefore, once FeLV infection is definitively established, proviral and viral RNA loads can be used to help distinguish progressive from regressive infection. In the field, given that it cannot usually be determined at what stage a naturally FeLV-infected cat is at, the proviral and plasma viral RNA loads alone at a single time point are not sufficient to determine whether the cat has progressive or regressive infection. Therefore, repeated testing 1–2 months later is recommended to clearly identify the course of infection.
Following recovery from antigenaemia during regressive infection, replicating virus may still be recovered for several months, and potentially up to a few years,36,48–50 by culturing bone marrow cells in the presence of high doses of glucocorticoids (testing for latency; note that since the introduction of FeLV provirus PCR, culturing of bone marrow is not usually performed anymore). The ability of the virus to reactivate (recurrence of viraemia and virus shedding) under immuno-suppressive doses of glucocorticoids50,51 clearly demonstrates that the virus is just kept in check by the cat’s immune system and is not completely eliminated. Using provirus PCR, FeLV provirus can be detected in the peripheral blood or bone marrow of regressively infected cats (FeLV provirus carriers)20,38 and viral plasma RNA might or might not be detectable by real-time RT-PCR.46,47,52
The potential for reactivation decreases as the timespan since exposure increases (Figure 1), and also depends on the balance between the host’s immune system and the virus (Figure 2). In cats with regressive infection, FeLV can reactivate in vivo if, for example, the cat becomes immunocompromised for any reason.50,51,53 The cat is no longer able to repress viral replication, antigenaemia/viraemia recurs, the cat sheds virus and can also develop FeLV-associated disease. 53 There is some evidence to show that regressively infected cats which are plasma viral RNA-positive have a higher probability of virus reactivation than those that are plasma viral RNA-negative. 6 Regressively infected cats probably never clear FeLV infection completely; 4 however, the proviral loads might be very low and might (at least temporarily) drop under the detection limit, depending on the sensitivity of the FeLV proviral PCR used.
Abortive FeLV infection
There is a group of cats that confines FeLV infection prior to provirus integration. All direct FeLV detection methods are negative (tests for FeLV antigen, provirus and virus), and antibody responses are the only sign of previous FeLV exposure (Table 1 and Figure 1). Only recently, the first routine anti-FeLV antibody test became commercially available; however, there is not yet sufficient data on whether this test reliably detects cats with abortive FeLV infection under field conditions. Abortively infected cats have strong anti-FeLV immunity (Figure 2) and will usually have experienced only a low level of FeLV exposure.28,30,38 Of the various infection outcomes, this is the most favourable for the cat – the balance is tilted in favour of the cat (Figure 2). Abortive infection can be experimentally induced in cats by exposure to only a very low amount of virus (eg, indirect transmission via faeces).28,30
Focal (localised or atypical) FeLV infection
In some cats, free viral antigen can be present in the blood (p27 antigen-positive) but no infectious virus (virus isolation-negative).54–56 These cats have been described in earlier studies as ‘discordant cats’.54–57 If antigenaemia in the absence of replicating virus in the peripheral blood persists for years, it can be caused by a so-called focal (localised or atypical) FeLV infection, in which the cat’s immune system keeps virus replication sequestered to certain tissues, such as the spleen, lymph nodes, small intestine, urinary tract or mammary glands.26,56,58 Production and release of free FeLV p27 antigen into the blood (but no or only minimal release of infected cells with provirus integration) in these cats can be intermittent or low-grade (Table 1).
Cats with focal FeLV infection are rare and probably not a major epidemiological concern, but this infection outcome can lead to perplexing appearances of FeLV infections and confusing FeLV test results. One case of focal infection is well documented in a queen, where the virus had been sequestered to the mammary glands; during a phase of negative free FeLV p27 antigen test results in the blood, the queen transmitted the virus to the kittens via the milk.26,59 Focal infections with discordant test results have been reported under experimental conditions,58,60,61 and have also been observed in up to 10% of naturally FeLV-infected cats.26,56,57,59,62 In one study, about one-third of the cats with discordant FeLV test results were provirus PCR-positive and it was assumed that in these cats the bone marrow was infected. 58
Finally, there are some cats with discordant or alternating test results in the early stage of FeLV infection (see box).

Key features in FeLV infection: antigenaemic cats, virus shedders and provirus carriers
From a clinical and epidemiological point of view, the priority is to determine whether a cat is viraemic/antigenaemic (ie, a virus shedder) and whether it has progressive or regressive infection (Table 1 and Figure 1). In recent years, the spectrum of methods available to achieve these goals has changed.
Antigenaemic cats
Antigenaemia (the presence of free p27 FeLV capsid antigen in blood, plasma or serum) has, for many years, been the most commonly applied marker of FeLV infection. In most cats, antigenaemia is a measure for viraemia (ie, replication-competent virus detectable in cell culture using virus isolation). However, some discordant antigenaemic but aviraemic cats have been described; as discussed, discordant results are mainly seen during the early phase of FeLV infection (see box on page 836), or with focal FeLV infection (see also Table 1 and above).26,54–60 ,62
Detection of viraemia (through virus isolation) is laborious and time-consuming and only available in specialised laboratories (Table 2). In contrast, antigenaemia can be easily detected by rapid point-of-care (POC) tests or by using quantitative plate-based ELISA in a laboratory. POC tests that detect free FeLV p27 antigen are based on ELISA, other immunochromatographic assays or rapid immunomigration assays and are available from several manufacturers.63–70 Some of these tests should preferentially be performed with serum or plasma, since whole blood has led to higher rates of false-positive results in some studies, particularly when the sample was haemolysed. 71 Some plate-based ELISAs can yield quantitative antigen loads or control for potentially confounding factors, such as cat anti-mouse antibodies.63,72 The diagnostic performance of most POC tests is generally good, although they can vary slightly in their sensitivities and specificities depending on the country of manufacture and application.64–70 None of the current p27 antigen tests should be used on saliva samples because this would miss some infected cats.73,74
Specifics of feline leukaemia virus (FeLV) detection methods

Antigenaemia is a measure for viraemia in most cats
Real-time PCR/RT-PCR in specialised laboratories should be used to determine quantitative results and to have a sufficiently high sensitivity IFA = immunofluorescence assay; POC = point-of-care

Some specialised laboratories offer RT-PCR for the detection of viral RNA; for example, in the saliva of infected cats. The detection of viral RNA by RT-PCR in the saliva generally correlates well with the detection of antigen in the blood of infected cats.23,24 While submitting a sample for RT-PCR to a specialised laboratory is more time-consuming and costly, it has the benefit that FeLV viral RNA in saliva, as well as in blood, can be detected as early as 1 week after FeLV exposure (ie, at least 2 weeks prior to detection of p27 antigen in the blood; Table 2). 27
FeLV shedders
FeLV shedders are of epidemiological importance because they pose an infection risk to FeLV-naive cats. Any cat with progressive FeLV infection and regressively infected cats that are antigen-positive (ie, in the early phase of regressive infection or after reactivation) should be considered an FeLV shedder (Figure 1).
It has been shown that there is excellent agreement between antigenaemia (free FeLV p27 antigen in blood) and the presence of viral RNA in saliva.23,24 Therefore, FeLV shedders can be detected by testing for FeLV p27 antigen in blood or by testing for viral RNA in saliva using RT-PCR; the latter test is positive earlier and might thus be useful diagnostically during a suspected very early infection (Table 2). 27
Saliva sampling and RT-PCR testing can also be used in multi-cat environments to confirm an absence of FeLV shedders in the population. It has been shown that a single positive cat among pooled saliva samples from up to 30 virus-negative cats would still yield a positive RT-PCR result, although a loss in assay sensitivity occurs due to a dilution effect. 23 Thus, under field conditions, pooling a maximum of 10 samples is recommended. Pooling of the samples can be performed in certain specialised laboratories (upfront enquiries are recommended). Note that testing of pooled samples is not appropriate for households with known FeLV-infected cats.
Provirus carriers
Provirus carriers are of epidemiological relevance since inadvertent FeLV transmission might occur if they are employed as blood donors. More generally, if the replication capacity of proviral DNA is no longer controlled by the immune system, virus replication recurs and these cats become FeLV shedders.
FeLV provirus is detected using DNA real-time PCR, which is highly sensitive and specific. The detection of FeLV provirus in peripheral blood using DNA real-time PCR was found to be more sensitive than the detection of free FeLV p27 antigen to demonstrate FeLV infection in a cat. 52 DNA PCR from whole blood detects all FeLV provirus carriers, which includes progressively and regressively infected cats (Figure 1). However, not all provirus carriers are FeLV p27 antigen-positive; regressively infected cats are usually FeLV provirus-positive and antigen-negative – the exceptions are during any initial antigenaemic phase or in the event of reactivation (Figure 1). In an early study in Switzerland, 10% of pet cats were found to have undergone regressive infection, as identified by FeLV provirus-positive and FeLV antigen-negative status. 20 Several studies have since confirmed the presence of regressively FeLV-infected cats (ie, provirus-positive, free p27 antigen-negative) in different cat populations worldwide.16,38,62,75 Provirus PCR on blood produces positive test results sooner after FeLV exposure than p27 antigen detection (Table 2). In experimental studies, cats were provirus-positive 1–2 weeks after FeLV exposure (vs 3–6 weeks for p27 antigen).20,39
FeLV detection methods
The following discussion focuses on the most frequently used methods for the detection of free FeLV p27 antigen, viral RNA and proviral DNA. The detection of FeLV antibodies is also discussed since, most recently, a POC antiFeLV antibody test has become available in Europe. Additional methods are available in specialised laboratories, such as virus isolation or immunofluorescence assays (Table 2), but are no longer commonly used and therefore are not discussed.
Detection of free FeLV p27 antigen in blood
When to test
The FeLV status of every cat should be known, and the p27 antigen test is the most common method of achieving this. In particular, testing should be conducted in the following cats or scenarios:
Cats suspected of having an FeLV infection for any reason;
Sick cats presented for veterinary examination;
Healthy cats prior to FeLV vaccination;
Cats with an unknown FeLV history;
For detection of FeLV shedders, such as in a multi-cat environment;
Prior to introducing a new cat into an environment.
How to interpret a single positive result
Any positive or questionable p27 antigen test result (eg, weakly positive or positive only after the test reading time indicated by the manufacturer has passed) should be confirmed immediately, particularly if FeLV prevalence is low or the expected FeLV exposure risk is low in the tested cat. With decreasing prevalence, the predictive value of a positive result becomes lower, meaning that even with the most accurate antigen tests the rate of false-positive test results increases. Confirmation can be performed by running a second, and preferably different, p27 antigen test (ie, POC test from a different manufacturer or quantitative ELISA in a laboratory). Alternatively, a saliva sample can be submitted for RT-PCR to detect viral RNA in the saliva or an EDTA blood sample can be submitted for provirus PCR testing. As discussed, antigen-positive cats are generally also provirus-positive (for rare exceptions refer to the focal infection section on page 836).
To be on the safe side, and since FeLV antigenaemia is associated with FeLV shedding, cats with a positive result for free p27 antigen (even if questionable or not yet confirmed positive) should be kept separated from FeLV-negative companions until lack of infection is confirmed.
How to interpret a confirmed antigen-positive result
If the positive result for free p27 antigen is confirmed, the cat is antigenaemic and an FeLV shedder at the point of testing. The animal should be retested after 6 weeks, and then if still positive tested again after another 6 weeks, to determine whether it is progressively infected with persistent antigenaemia/ viraemia, or has regressive infection with transient antigenaemia/viraemia. Antigenaemic cats present an infection risk and should always be kept separated from FeLV-negative companions, regardless of their health status and until retesting negative at a later time point.
A confirmed positive FeLV antigen test result (whether a single result or repeatedly positive result over time) should never be a death sentence for a cat, if circumstances allow keeping the cat separated from other cats.
How to interpret a negative result
A negative test result for free FeLV p27 antigen is highly reliable, because the predictive value of the negative result is much higher than that of a positive test result due to the low FeLV prevalence in most countries. This means that the cat is not antigenaemic at the time of testing. Thus, the cat variously was not exposed to FeLV (uninfected), is immune to FeLV (eg, has been vaccinated), has overcome antigenaemia (is regressively infected), has abortive FeLV infection or is not yet positive because it is still in a very early stage of FeLV infection. It usually takes at least 3–6 weeks (sometimes even longer) after FeLV exposure before FeLV antigen can be detected in the peripheral blood of an infected cat. If recent FeLV exposure cannot be absolutely excluded, the cat should be retested in approximately 6 weeks. Until the time of retesting, the cat should be kept separated from other cats (eg, quarantined in an animal shelter; no outdoor access for pet cats). This is so as not to pose a risk to others, and also so that it does not run the risk of becoming infected within the 6 weeks; the latter would lead to a requirement for further retesting, due to unknown FeLV exposure.
Detection of viral RNA by RT-PCR in saliva (single or multiple cats)
When to test
p27 antigen-positive cats are also typically positive for FeLV viral RNA in saliva.23,24,27 Therefore, detection of FeLV RNA by RT-PCR in saliva can be taken as a marker for antigenaemia and the indications to perform RT-PCR are the same as those discussed for free FeLV p27 antigen testing. However, in view of the relatively high costs of RT-PCR and the turnaround time for testing (typically 1–3 days), this test is not often used in individual cats. One exception might be where the collection of saliva circumvents problems associated with blood collection (eg, fractious cats or in shelters without on-site veterinary support). Another exception might be where RT-PCR is used as a confirmatory test following a positive or questionable FeLV p27 antigen test result on blood. In addition, RT-PCR on saliva can be useful during the very early phase of FeLV infection as viral RNA in saliva (and even more so in blood) is detectable approximately 2 weeks earlier than p27 antigen is detectable in the blood.
Another application of real-time RT-PCR testing is to test pooled saliva samples for the presence of FeLV viral RNA due to its extremely high sensitivity and the very high FeLV viral loads in saliva. Real-time RT-PCR of pooled saliva samples is a cost-effective and efficient screening assay to confirm the absence of FeLV shedders in multi-cat environments that are likely to be free of FeLV (see discussion of FeLV shedders on page 838).
How to interpret a positive RT-PCR result
If the FeLV RT-PCR result from saliva in a single cat is positive, the cat is antigenaemic and an FeLV shedder at the time it was tested. Thus, interpretation is the same as for a cat that tests positive for free FeLV p27 antigen (see page 839). The cat should be retested after 6 weeks, and then if still positive tested again after another 6 weeks, to determine whether it is undergoing progressive or regressive infection. FeLV-shedding cats should always be kept separated from FeLV-negative companions, regardless of their health status and until retesting negative.
Should a pooled saliva sample (population analysis) test positive, subsequent testing of individual cats is necessary to detect the FeLV-shedding cat(s) within the group of tested cats, either by using RT-PCR on single saliva swabs or FeLV p27 antigen testing of blood from individual cats.
False-positive RT-PCR results can be caused by laboratory contamination. It is important to only use reference laboratories where the sensitivity and specificity of the RT-PCR assay is known, and extraction controls and negative and positive PCR controls are performed.
How to interpret a negative result
If the FeLV RT-PCR result from saliva in a single cat is negative, the cat is not antigenaemic at the time of testing. Thus, the possibilities are that the cat has had no exposure to FeLV (uninfected), is immune to FeLV (eg, has been vaccinated), has overcome antigenaemia (is regressively infected), has abortive FeLV infection or is not yet positive because it is still in a very early stage of FeLV infection. The negative phase after exposure is significantly shorter for RT-PCR on saliva (and also shorter for RT-PCR on blood) than it is for p27 antigen tests on blood.27,39 It takes usually at least 3–6 weeks (sometimes even longer) after FeLV exposure before FeLV antigen can be detected in the peripheral blood of an infected cat, while viral RNA in the saliva (and blood) can be detected as early as 1 week after FeLV infection. 47
Detection of FeLV provirus in blood
When to test
FeLV provirus DNA PCR can be used in the following cats and scenarios:
As a confirmatory test for positive or questionable free p27 antigen test results;
To detect provirus carriers/regressively FeLV-infected cats;
To test at early time points after potential exposure (provirus PCR is positive earlier than p27 antigen tests);
To confirm the absence of FeLV provirus carriers in multi-cat populations;
To clarify obscure clinical cases with suspected FeLV infection but absence of FeLV antigenaemia;
To test blood donors and blood products prior to transfusion.
How to interpret a positive result
If the FeLV provirus PCR result is positive, the cat has been exposed to FeLV and has developed either progressive or regressive infection (Figure 1). Some laboratories also provide the provirus load. If the provirus load is high (low cycle threshold value), there is a good probability that the cat is antigenaemic at the time tested; if the provirus load stays persistently high, the cat is likely to be progressively infected. 20
If the FeLV provirus PCR result is positive, a test should be run to detect antigenaemia to distinguish between progressive and regressive infection. Several weeks into infection, blood proviral loads can also be used to differentiate cats with progressive and regressive infection. However, in naturally infected cats the time point of infection is usually unknown and during early infection proviral blood loads do not differ between regressively and progressively infected cats. 6
False-positive PCR results can be caused by laboratory contamination. It is important only to use reference laboratories where the sensitivity and specificity of the PCR assay is known, and extraction controls and negative and positive PCR controls are performed.
How to interpret a negative result
If the FeLV provirus PCR result is negative, the cat does not have provirus integrated into its genome and is neither progressively nor regressively infected. Thus, the cat variously has had no exposure to FeLV or has focal or abortive infection, or it is in the very early stage of infection. However, it usually takes only 1–2 weeks after FeLV exposure for a cat to become FeLV provirus-positive, and therefore it is highly unlikely that infection would be missed within this short window of time.
Detection of anti-FeLV antibodies
When to test
Cats exposed to FeLV can develop different degrees of immune response to the virus (Figure 2). While cellular immune response is very cumbersome to determine, even in specialised laboratories,43,76 there are several methods to determine anti-FeLV antibodies, including neutralisation assays and a novel POC test. To determine the true humoral immunity against FeLV, quantification of biologically active virus-neutralising antibodies would be necessary. However, virus neutralisation is performed only in specialised laboratories and requires time-consuming cell culture assays.
In cats with abortive FeLV infection, antibodies are the sole indicator of exposure to FeLV (Figures 1 and 2). Thus, testing for FeLV antibodies is the only method to detect abortively infected cats. However, as abortively infected cats will not shed the virus, will not develop clinical signs, and will not reactivate the infection, their clinical and epidemiological relevance is very low.
Testing for FeLV neutralising antibodies can be used to help characterise the disease outcome (ie, progressive vs regressive infection).6,20,54 Most cats with regressive infection exhibit a strong humoral immune response with high levels of neutralising antibodies, while progressively infected cats commonly have low levels of or no neutralising antibodies against FeLV (Figure 2). 36 None of the current FeLV vaccines induce a response manifesting in neutralising antibodies; these vaccines protect against challenge presumably because they stimulate cellular immunity.6,36,77,78
Examination of different FeLV antigens to assess their diagnostic utility for the development of a POC test that detects anti-FeLV antibodies has identified a recombinant preparation of FeLV p15E (envelope transmembrane protein) to be the most promising antigen. 79 In naturally infected cats, the p15E ELISA showed a diagnostic sensitivity of 77.1% and a specificity of 85.6% when compared with provirus PCR results. 79 Use of this antigen in an FeLV antibody test (in combination with FeLV p27 antigen testing) might offer most promise for recognising all FeLV-exposed cats. The antibody test is expected to be positive in cats with regressive or abortive infection and the antigen test will recognise all cats with progressive infection.
It is unknown how well the presence of anti-p15E antibodies correlates with protection from FeLV infection, and if antibodies in cats with abortive infection are present life-long. These are important questions that still have to be answered to determine if an abortively infected cat might be immune to new infection and, thus, not require vaccination. In FeLV-vaccinated cats the results of antibody testing with the p15E antigen depend on the vaccine used. While cats vaccinated with a whole virus vaccine might develop strong anti-p15E antibodies, this might not be the case when a recombinant protein or canarypox-based vaccine is used. 79
How to interpret a positive result
FeLV antibody testing is currently not used routinely (Table 2). A POC test detecting anti-bodies against FeLV p15E antigen 79 has been introduced recently onto the European market. However, insufficient data is currently available to assess the value of this test for the diagnosis of FeLV infection in the field. Controlled studies will be necessary to determine whether this test can reliably predict FeLV infection or immunity against FeLV, and if it could be used, for example, as a pre-vaccination test.
How to interpret a negative result
Most cats without antibodies to FeLV p15E likely will not have had exposure to FeLV previously; 79 but since not all cats that have been FeLV-exposed maintain anti-FeLV antibodies, a negative antibody test result in the absence of FeLV antigen does not rule out prior FeLV exposure.
Testing for FeLV under specific circumstances
Testing a single cat for FeLV
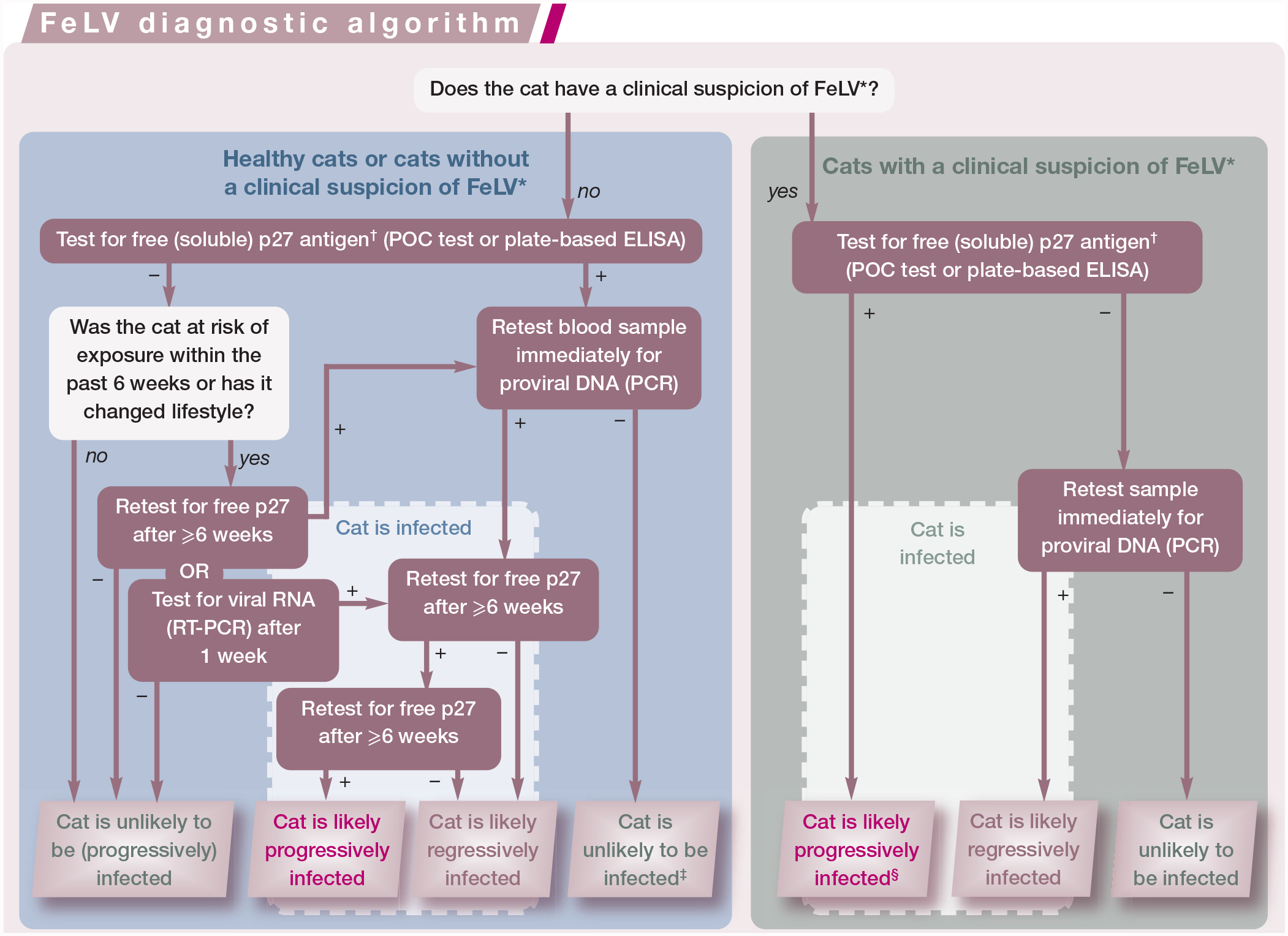
The European Advisory Board on Cat Diseases (ABCD) has created a diagnostic algorithm (‘ABCD FeLV diagnostic tool’) that is intended to lead veterinary practitioners through the steps to determine the probability of whether a cat has been exposed to FeLV and whether it has undergone progressive or regressive infection. These diagnostic steps are shown in the box below. This diagnostic tool, which is based on risk assessment as well as the cat’s clinical presentation, takes into account the different test characteristics, the timespan over which a test will produce positive results, and the positive and negative predictive value of tests. It also highlights the steps for confirmation of results as well as for repeated testing to determine the different courses of FeLV infection. Additional information on use of the ABCD FeLV diagnostic tool can be found at abcdcatsvets.org.
Testing of blood donors
It has been demonstrated that regressively infected cats can transmit FeLV to naive recipients via the transfusion of blood. 80 Some of the cats receiving blood from regressively infected cats have subsequently developed progressive FeLV infection and FeLV-associated disease (non-regenerative anaemia and lymphoma). The blood that had been transfused had tested negative for free FeLV p27 antigen and was negative in virus isolation but positive for FeLV proviral DNA. Thus, methods to detect free FeLV p27 antigen are not sufficient to prevent inadvertent transmission of FeLV infection. Sensitive realtime PCR is recommended to detect FeLV provirus in any cat serving as a blood donor to exclude inadvertent transmission of FeLV to recipients, and also more generally to further decrease the prevalence of FeLV infection within the cat population.
Testing to prevent introduction of virus into an FeLV-free cat population
If a cat from an unknown environment (eg, a rescue cat) or with an unknown history of FeLV exposure (eg, a cat with outdoor access or a cat from a facility where not all cats have been tested or some cats have outdoor access) is introduced to a population of FeLV-uninfected cats, such as a breeding premises or household with pet cats, the incoming cat should be tested for FeLV antigenaemia and/or shedding prior to introduction. The ABCD FeLV diagnostic algorithm can be followed for this purpose (Figure 4).

The European Advisory Board on Cat Diseases (ABCD) feline leukaemia virus (FeLV) diagnostic tool. This diagnostic algorithm should be followed to determine the probability of whether or not a cat is infected with FeLV and which infection outcome is most likely. *For risk factors or clinical disorders associated with FeLV, see the box below. †Whenever testing for free FeLV p27 antigen in blood samples is suggested (point-of-care [POC] test, plate-based ELISA) in any of the steps in the algorithm, testing for viral RNA in saliva samples (RT-PCR) can alternatively be used. ‡In very rare cases, a focal FeLV infection can be the reason for a positive free p27 antigen and negative provirus PCR result in blood samples. §In cats with a clinical suspicion of FeLV infection and a positive free p27 antigen test, a confirmatory test is not absolutely necessary as a false-positive test is less likely in these cats; the positive predictive value is high as the cats are already in the group with a high risk of FeLV infection. This diagnostic algorithm is based on the ‘ABCD FeLV diagnostic tool’, which is available at abcdcatsvets.org
It is important that during the entire testing period, including potential retesting, the cat to be introduced is quarantined without any contact with other cats in the facility/house-hold until it is confirmed that the cat is not shedding FeLV. The most frequent error that occurs is when cats test negative and it is not considered that the cats might have been infected very recently and it is too early for a positive test result. If the FeLV diagnostic algorithm (Figure 4) is followed closely, and cats are kept separated during this time, this pitfall can be avoided.
Testing cats in a multi-cat facility for freedom from FeLV infection
The best strategy in this scenario depends on the goal. Ideally, no FeLV carriers are present in a multi-cat facility. To confirm that this goal is achieved, all cats would have to be tested for FeLV provirus using DNA PCR to also detect cats that are only FeLV provirus carriers (regressive infection). Since FeLV provirus PCR tests can be positive approximately 2 weeks after FeLV exposure (Table 2), cats need to be kept isolated from any potential FeLV infection risk for 2–3 weeks.
Alternatively, the goal might be to have no active FeLV shedders in the facility in order to prevent infection of any FeLV-naive cats. Absence of FeLV shedders can be confirmed by FeLV antigen blood testing of all animals (antigen-positive cats are shedders) or, more cost-effectively, by using RT-PCR to test saliva samples from all cats for viral RNA. For the latter analysis, sample collection is straight-forward, and the saliva samples can be pooled for testing by specialised laboratories. As already discussed, it is recommended that no more than 10 saliva samples are pooled per test (enquire with the laboratory in advance). RT-PCR also has the advantage that infected cats can be detected as soon as approximately 1 week after exposure. If antigen testing is used and recent FeLV exposure cannot be excluded, cats need to be retested after 6 weeks. During this period, cats testing FeLV-positive should be kept separated. More information on the management of FeLV in multi-cat environments is available from the ABCD.81,82

Key Points
Testing of cats for FeLV infection is an important task for veterinarians in clinical practice. Interpretation of FeLV tests is not trivial, and requires a fundamental knowledge of disease pathogenesis, virus–host interactions, and different FeLV tests and their characteristics.
The FeLV status of every cat should be known; it influences therapeutic and epidemiological decisions that need to be discussed with the cat owner.
Further awareness of this deadly feline infection and its proper diagnosis is necessary to decrease FeLV infection prevalence and even potentially to eradicate it in certain geographic areas.

Footnotes
Acknowledgements
The diagnostic algorithm on which Figure 4 is based is provided by courtesy of the European Advisory Board on Cat Diseases (ABCD) (![]() ).
).
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work did not involve the use of animals and therefore ethical approval was not necessarily required.
Informed consent
This work did not involve the use of animals and therefore informed consent was not required. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
