Abstract
Objectives
The aim of this study was to assess the reliability of an EDTA-based method for measuring cat blood bioamines.
Methods
Test 1 involved collecting blood samples from seven university laboratory cats. The samples were transferred to EDTA, heparin and plain tubes to determine concentrations of four bioamines (serotonin, dopamine, epinephrine [adrenaline] and norepinephrine [noradrenaline]). Correlation of measurements performed on EDTA plasma, with those performed on heparinized plasma or serum were assessed by intraclass correlation coefficients (ICCs). In test 2, blood samples from five owned cats were collected and stored in EDTA tubes and divided between duplicate Eppendorf tubes labeled as different cats for blinding purposes and analyzed independently for the same four bioamines as in test 1. Reliability of concentration determination for these duplicates was assessed by ICCs and coefficients of variation (CVs).
Results
In test 1, there was no significant correlation between the EDTA plasma serotonin and serum serotonin concentrations. There was also no significant correlation between EDTA plasma and heparin plasma concentrations for either epinephrine or norepinephrine. There was a statistically significant but weak correlation between EDTA plasma and heparin plasma dopamine concentrations. In test 2, there was no correlation for repeat-analyzed serotonin and epinephrine concentrations. Although there were statistically significant correlations for dopamine and norepinephrine, CVs for each analyte were in excess of 30%.
Conclusions and relevance
Before any further attempt is made to measure and report on neurotransmitter concentrations in domestic cats, it is essential that the robustness of the methodology is carefully validated and the data presented.
Introduction
Historically, much research has focused on the use of glucocorticoids as physiological markers of stress in several biological animal matrices.1,2 Measurement of bioamines such as serotonin and norepinephrine (noradrenaline) to assist in the evaluation of animals’ emotional states is a more recent phenomenon, and the reliability of the available methods to evaluate them in non-human animals remains largely unknown; however, such methods have been routinely validated for human samples. As part of a larger study into feline house-soiling and emotional profile in which fecal glucocorticoids and bioamines were measured, 3 feline blood samples were collected into EDTA, heparin and plain blood tubes for potential cross-validation of blood bioamine levels in the samples.
Materials and methods
Ethical approval for the study was provided by the FMVZ-USP Ethical Committee.
Approximately 6 ml of blood was collected from the jugular vein of 12 cats that were fasted for 12 h (seven university laboratory cats were used in test 1 and five cats belonging to the research team were used in test 2 after informed owner consent was received). The cats were very friendly towards humans and thus there were no difficulties during blood collection. Some of the blood collected was used for hematology and serum biochemistry in order to confirm the general good health status of the participant cats. The remainder (approximately 4 ml) was transferred to EDTA, heparin and plain tubes for test 1, to determine blood bioamine levels, or to EDTA tubes only for test 2. Given that some bioamines are photosensitive, tubes were protected from light by aluminium foil. Within about 15 mins of collection, the tubes were centrifuged for 15 mins under refrigeration (4ºC), at 3000 rpm. The supernatant was then transferred into amber Eppendorf tubes (approximately 1 ml in each). The Eppendorf tubes were then stored in a freezer (–80ºC) for a few days until analysis. Two tests described below were then performed.
In test 1 (effect of blood tube type), the samples from the seven university laboratory cats were stored in both EDTA and heparin tubes (for measuring dopamine, epinephrine [adrenaline] and norepinephrine) or EDTA and plain tubes (for measuring serotonin). Correlation between EDTA samples and either heparin or plain tube samples were assessed using intraclass correlation coefficients (ICCs).
In test 2 (duplicate samples test), the samples from five cats belonging to the research team were stored in a similar way in EDTA tubes and divided between two duplicate Eppendorf tubes, which were then labeled as different cats for blinding purposes and analyzed independently for the same four bioamines as in test 1. Researchers responsible for the laboratory analyses were not aware of the duplicate procedure. In order to test for reliability, concentrations from these duplicates were compared using ICCs and calculation of their coefficients of variation (CVs).
Bioamine concentrations were determined by the quality-assured Nephrology Department of São Paulo Federal University, Brazil, using high performance liquid chromatography (HPLC). In the case of catecholamines, these were measured using ion-pair reverse phase chromatography coupled with electrochemical detection. Fast isocratic separation was obtained using an RP 18 Aquapore cation F micron, Brownlee Column (Applied Biosystems) (4.6–250 mm) eluted with the following mobile phase: 0.02 M sodium dibasic phosphate; 0.02 M citric acid; pH 2.64; 10% methanol; 0.12 mM Na2EDTA; and 566 mg/l heptanesulfonic acid. The total time for sample analysis was 30 mins. Plasma (500 μl) had already gone through the following purification steps: 50 mg Al2O5 was weighed out in centrifuge tubes and the samples were added to Tris-buffer, pH 8.8, plus 40 ml (8 ng) dihydroxybenzylamine (DHBA; internal standard). The suspension was vortex-mixed for 10 mins. The precipitated alumina was washed three times and vortex-mixed with 1 ml of water, and the catecholamines were eluted with 400 ml 0.1 M of perchloric acid after 3 mins of vortex mixing. After centrifugation for 3 mins at 1500 g, the supernatant was filtered and 100 ml was injected into the reverse phase column. Monoamine concentration was expressed as pg/mg cell protein.
Serotonin was also determined by using ion-pair reverse phase chromatography coupled with electrochemical detection. Separation was obtained using a Chromolith SpeedROD 50-4.6 mm reverse-phase column (Merck). The HPLC system consisted of a pump (model LC-10 ADVP-Shimadzu), electrochemical detector (model L-ECD-6A-Shimadzu) and automatic injector (SIL-20AC-Shimadzu). Serotonin was separated by isocratic elution at a flow of 1.0 ml/min for approximately 10 mins; the mobile phase was composed of 0.02 M sodium phosphate (Merck), 0.02 M sodium citrate (Merck), EDTA 40 mg (Merck), 5 mg (Sigma) heptane sulfonate and 10% methanol (Licrosolv), with the pH adjusted to 2.53 by the addition of 50% perchloric acid (Merck). Plasma (100 μl) was deproteinized with 50 μl of ice cold 0.34 M perchloric acid and vortexed for 1 min, followed by freezing (liquid nitrogen) for 5 mins and centrifuged at 10,000 rpm at 4ºC for 40 minutes. Next, 894 μl of sodium disulfite and 66 μl of internal standard (DHBA diluted 100-fold from the 1 mg/ml stock in perchloric acid) were then added to 40 μl of the supernatant. One hundred microliters of the sample was injected into the reverse-phase column. The serotonin concentration was expressed as pg/mg cell protein.
Both sample extraction and HPLC separation were performed on the same day, thus avoiding any interassay variation associated with differences in time to analysis. Furthermore, DHBA constitutes an internal standard for calculation and control purposes in relation to HPLC.
This method has been reported to be both reliable and valid for human blood samples.4,5
Results
Effect of blood tube type
Results of the effects of blood tube type test can be found in Table 1. There was no correlation between plasma EDTA and serum concentrations of serotonin (ICC = 0.351; P = 0.180). EDTA samples had a reported median concentration of serotonin of 731.93 ng/ml vs 549.93 ng/ml for samples stored in plain tubes. There was no correlation between EDTA and heparin plasma measurements of epinephrine (ICC = 0.147; P = 0.351) and norepinephrine (ICC = 0.039; P = 0.454). The median concentration of epinephrine was 303.23 pg/ml for EDTA samples and 393.69 pg/ml for heparin samples; and for norepinephrine median concentrations were 366.79 pg/ml and 350.89 pg/ml, respectively. Although there was a significant correlation between EDTA and heparin plasma measurements for dopamine (ICC = 0.781; P = 0.012), the correlation was considered weak. EDTA samples had a median reported concentration of dopamine of 235.03 pg/ml vs 193.29 pg/ml for heparinized samples.
Results of the effects of blood tube type test

Samples from the seven university laboratory cats were stored in both EDTA and heparin tubes (for measuring dopamine [Dopa], epinephrine [Epi], and norepinephrine [Nor]) or in both EDTA and plain tubes (for measuring serotonin [Sero])
Significant positive correlation between concentrations in the two samples
Undetermined concentration for this sample
Duplicate samples test
Serotonin (ICC = 0.00; P = 0.915) and epinephrine (ICC = 0.00; P = 0.664) showed no correlation, with CVs of around 55% and 67%, respectively (Table 2). Although there was a significant correlation for the concentrations of dopamine (ICC = 0.714; P = 0.038) and norepinephrine (ICC = 0.962; P <0.001), the CV for both was in excess of 30% (see Table 2).
Results of the duplicate samples test
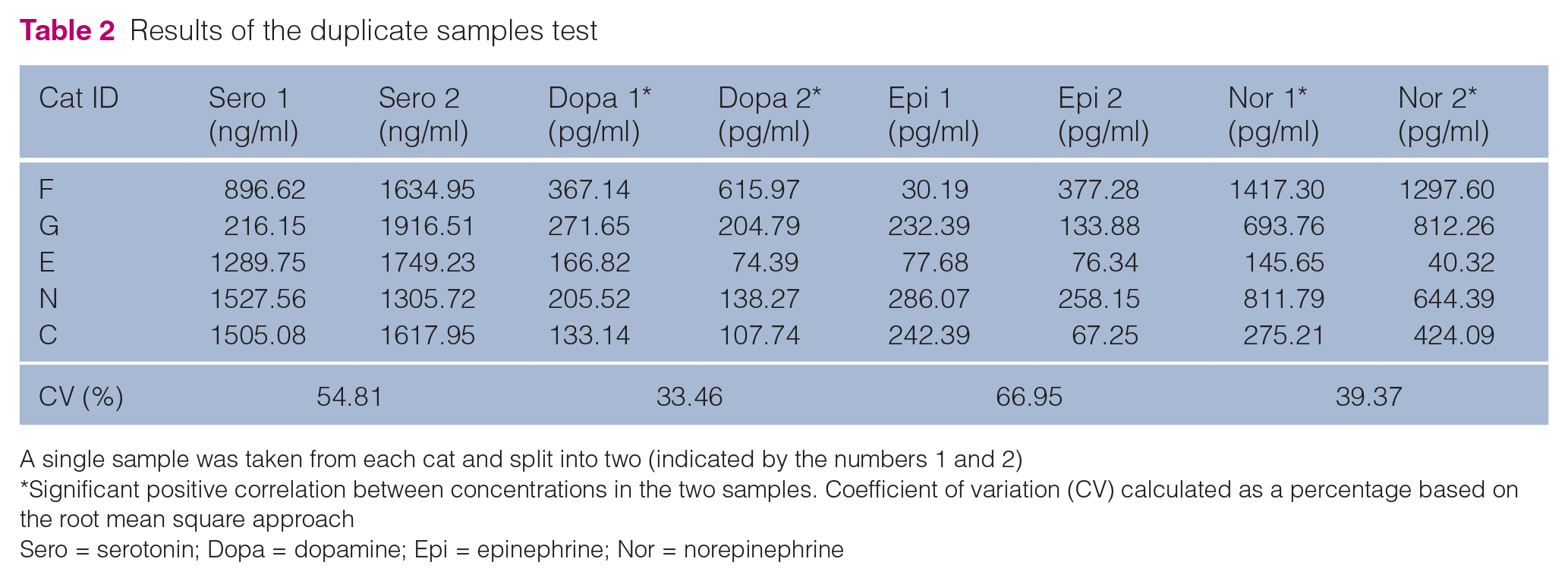
A single sample was taken from each cat and split into two (indicated by the numbers 1 and 2)
Significant positive correlation between concentrations in the two samples. Coefficient of variation (CV) calculated as a percentage based on the root mean square approach
Sero = serotonin; Dopa = dopamine; Epi = epinephrine; Nor = norepinephrine
Discussion
Using HPLC to measure feline bioamines in samples stored in EDTA tubes appears to be unreliable, with little correlation and a very large CV detected even in the same sample when analyzed twice. Thus, it should not be assumed that a method used to assay human samples will necessarily be valid for non-human animals. Although our results have focused on EDTA samples, without undertaking a similar duplicate test procedure, it cannot be assumed that the same problem is not present in either lithium heparinized plasma or serum. The poor correlation between samples cannot be explained by intra-cat variability as the values compared in both tests came from the same cat samples transferred to different tubes; however, great inter-cat variability, particularly for serotonin, is present.
We hypothesize that a problem just affecting the EDTA samples exists, but it is possible that it might be a wider problem than is currently recognized with sample storage and the HPLC method used. This is of concern, as previous studies correlating behavioral problems with the emotional profile in pet dogs have used this method for measuring serotonin and catecholamines.6,7 We also note a widespread absence of CVs in published studies examining the relationship between blood or other sources of bioamines and problem behavior. 8 This is a standard metric used to assess the reliability of the analysis and should be considered essential in any such report.
Although it might seem most likely that the source of the problem relates to the antioxidative properties of EDTA being insufficient to prevent oxidative degradation, 9 this should not be assumed. Several studies on the stability of catecholamines and serotonin (including one testing human and cat blood samples) recommend the use of heparinized tubes containing an antioxidant and immediate centrifugation;9–11 however, our results indicate that this should not be assumed to be a reliable method unless appropriate reliability assessments have been made together with a careful evaluation on the influence of pre-analytical procedures on measured bioamines. As noted by Alberghina et al 12 in their study of ELISA kits for measuring serum serotonin in dogs, several pre-analytical factors, such as handling, storage time and temperature, also need to be considered and standardized in reporting before any method is recommended for use in practice, or comparisons made between studies.
Conclusions
Before any further attempt is made to measure and report on neurotransmitter levels in species for which an established method has not been developed, it is essential that the reliability of the methods used are evaluated and reported, rather than assumed from human data.
Footnotes
Acknowledgements
We would like to thank Carine Savalli Redigolo for the statistical analysis. We also thank FAPESP Research Foundation for funding the project and giving a doctorate scholarship to D Ramos.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This article was funded by the FAPESP Research Foundation – 08/55385-6.
Ethical approval
This work involved the use of experimental animals and the study therefore had ethical approval from an established committee as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). For any animals or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
