Abstract
The noble gas xenon seems to have minimal cardiovascular side-effects and so may be an ideal anaesthetic agent when investigating cardiovascular physiology. In comparison with standard modern anaesthetics, we investigated the haemodynamic and hormonal effects of xenon in Beagle dogs. After a 30 min baseline period, anaesthesia was induced with propofol and maintained with either (1) 1.2% isoflurane/70% nitrous oxide (N2O), (2) 0.8% isoflurane/0.5 µg/kg/min remifentanil or (3) 63% xenon/0.5 µg/kg/min remifentanil (n = 6 per group). Haemodynamics were recorded and blood samples taken before and 60 min after induction. Mean arterial blood pressure (MAP) was higher in conscious dogs than during isoflurane/N2O (86 ± 2 vs. 65 ± 2 mmHg, mean ± SEM) and isoflurane/remifentanil anaesthesia (95 ± 2 vs. 67 ± 3 mmHg), whereas MAP did not decrease significantly in response to xenon/remifentanil anaesthesia (96 ± 4 vs. 85 ± 6 mmHg). Bradycardia was present during isoflurane/remifentanil (54 ± 2/min) and xenon/remifentanil (40 ± 3/min), but not during isoflurane/N2O anaesthesia (98 ± 3/min, P < 0.05). Xenon/remifentanil anaesthesia induced the highest reduction in cardiac output (CO) (–61%), and the highest increase in systemic vascular resistance (+120%) among all treatment groups (P < 0.05). A simultaneous increase in endogenous adrenaline and noradrenaline concentrations could only be observed in the xenon/remifentanil group, whereas angiotensin II and vasopressin concentrations increased in all groups. In conclusion, xenon/remifentanil anaesthesia maintains MAP but reduces heart rate and CO and is associated with a considerable stimulation of vasopressor hormones in Beagle dogs. Therefore, xenon/remifentanil exerts a new quality of adverse haemodynamic effects different from volatile anaesthetics and may not perform better during studies of cardiovascular physiology.
It is generally agreed that cardiovascular physiological research should as far as possible be conducted on conscious individuals or animals in order to avoid anaesthesia-induced haemodynamic and hormonal side-effects (Burchardi & Kaczmarczyk 1994, Hettrick et al. 1996, Preckel et al. 2004). However, if the experimental protocol includes painful and stressful procedures general anaesthesia is essential. It is not yet clear what the optimal anaesthetic technique for this type of study is. A wide range of anaesthetic agents are now available and can be used especially in bigger animals like pigs and dogs as well as in humans. Xenon as an anaesthetic may prove to be superior to other anaesthetic agents (Lane et al. 1980, Lynch et al. 2000). It is of special interest to the physiologist because it is highly cardiostable. Blood pressure and left ventricular performance are not or minimally affected in dogs (Hettrick et al. 1998), pigs (Marx et al. 1997) and humans (Luttropp et al. 1993, Ishiguro 2001). Moreover, a randomized, controlled multicentre trial by Rossaint et al. (2003) has shown xenon anaesthesia to be clinically effective and safe.
However, there are reports that xenon may potentiate parasympathetic activity, depress heart rate (HR) variability and depress baroreflex sensitivity to a greater extent than isoflurane (Ishiguro et al. 2000). The literature on catecholamine release during xenon anaesthesia is equivocal. Some studies report a decrease of plasma catecholamine concentrations during anaesthesia in pigs (Marx et al. 1997), and unaltered or elevated catecholamine concentrations during xenon anaesthesia in human patients (Boomsma et al. 1990). Against this background of conflicting reports regarding the cardiovascular and hormonal effects of xenon, we performed this randomized controlled study in one of the most widely studied animal models of cardiac physiology – the Beagle dog. We compared a combination of xenon and remifentanil with two well-established regimens: isoflurane/remifentanil and isoflurane/N2O. We hypothesized that a combination of xenon and remifentanil results in less cardiovascular and hormonal disturbance than two widely used isoflurane-based regimens.
Materials and methods
The study was approved by the local Governmental Animal Protection Committee (Permission No. G0424/99) and adheres to the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Research, Commission on Life Sciences, National Research Council 1996).
Animal maintenance and dietary regimen
Healthy, pure-bred female Beagle dogs (mean body weight: 13.4 ± 1.3 kg) of, on average, two years of age were kept under highly standardized environmental conditions (air-conditioning at 21°C and 55–60% humidity) and received a dietary regimen which was standardized for sodium (2.5 mmol/kg body weight per day) and water (91 mL/kg body weight per day) intake as described previously (Höhne et al. 2002, Francis et al. 2004). Daily check-ups on physical health included body weight and temperature measurement.
Eight days prior to the experiments, an aliquot (max. 80 mL) of each dog's own blood was collected and stored in a blood bag at 4°C (Biopack®; Biotrans, Dreieich, Germany). The blood served to replace the blood withdrawn for analysis during the experiments. We maintained an interval of at least 14 days between experiments in the same dog in order to avoid possible interactions between the different anaesthetics. Six months after all experiments had been performed, the dogs were subject to a final veterinary examination for health and wellbeing, and were then removed from the laboratory to live with individual families in private housing. The families were all familiar with dog keeping and were aware of the dogs' past history.
Experimental protocols
A total of six dogs were investigated. During a training period of at least six weeks, the dogs were gradually familiarized with the laboratory environment, the staff of investigators and the experimental procedures. The training period has been described in detail previously (Höhne et al. 2002, Francis et al. 2004). The staff of investigators could provide a calming influence on the dogs by voice and petting. None of the dogs displayed noticeable problems regarding their psychological status in the training period, and there was no adverse social behaviour towards the staff of investigators nor towards the other dogs which were kept in close company. Each dog was subject to three different treatments in random order, which resulted in a total of 18 experiments. Only one experiment was performed per day. Preparation of the dogs started at 07:30 h. Body weight and temperature were recorded. The bladder was catheterized with a self-retaining transurethral Foley catheter and a venous cannula was placed into a foreleg vein.
Insertion of intravascular catheters was performed under local anaesthesia (lidocaine 1%). An arterial line (20 gauge, No. 4235-8; Ohmeda, Erlangen, Germany) was positioned in the abdominal aorta via the femoral artery and was used for pressure monitoring and blood sampling. A pulmonary artery catheter (5 French, No. 132F5; Baxter, Unterschließheim, Germany) was then inserted via the right external jugular vein for monitoring of mean pulmonary artery pressure (MPAP), pulmonary capillary wedge pressure (PCWP), determination of cardiac output (CO) (using the transpulmonary thermodilution method) and blood sampling. The arterial line and the pulmonary artery catheter were connected to pressure transducers (Viggo Spectramed, DF-XX, Spectramed GmbH, Düsseldorf, Germany) which were attached to a vertical upright centimetre scale on the table. The transducers were levelled at the right atrium of the dogs (on the centimetre scale this is about 10–11 cm above the table) who were lying on their right side throughout the experiment. The transducer position was recorded and used for all further experiments in the respective dog. Following this, the conscious dogs were allowed to adjust for at least 30 min before the experiment was started at 09:30 h.
After a baseline period of 30 min in the conscious state, anaesthesia was induced with propofol 8–10 mg/kg (Disoprivan® 1% lipid emulsion; AstraZeneca GmbH, Wedel, Germany) and the dogs were intubated with a cuffed Woodbridge tube (34–36 Charrière) and mechanically ventilated on a closed circuit system (PhysioFlexTM, Dräger Medical, Lübeck, Germany) in a volume-controlled mode with an I/E ratio of 1:2. The respiratory rate was set to 12 breaths/min, the positive end-expiratory pressure to 4 mmHg and the tidal volume was adjusted to keep the arterial carbon dioxide tension between 35 and 40 mmHg. General anaesthesia was then maintained by one out of three different anaesthetic regimens (described below).
Isoflurane/N2O
The inspired concentration of isoflurane was adjusted to maintain an end-tidal concentration of 1.2 ± 0.03 vol% in a mixture of 30% oxygen and 70% N2O. This was equivalent to 1.3 times the minimal anaesthetic concentration (MAC) in dogs (5), because 1 MAC is 1.28 vol% for isoflurane and 188 vol% for N2O in dogs. These two MAC values are additive (Cullen 1986).
MAC of an inhaled anaesthetic is defined as that concentration at 1 atmosphere that prevents skeletal muscle movement in response to a supramaximal painful stimulus (e.g. tail clamp or surgical incision) in 50% of patients. A MAC of 1.3 prevents response in 95% of subjects.
Isoflurane/remifentanil
The inspired concentration of isoflurane was adjusted to an end-tidal concentration of 0.8 ± 0.01 vol% in a mixture of oxygen and air (inspiratory oxygen concentration 26 ± 1%). Remifentanil was simultaneously infused at 0.5 µg/kg/min. This dosage of remifentanil has been shown to reduce the MAC of enflurane by about 50% in dogs (Michelsen et al. 1996). We were unable to locate any data on the effect of remifentanil on the MAC of isoflurane in dogs. It is known that remifentanil at a whole blood concentration of 1.37 ng/mL reduces the MAC of isoflurane in humans by 50% (Lang et al. 1996). We therefore presumed that in dogs the MAC of isoflurane should be reduced by approximately 50% when combined with a similar dosage of remifentanil and so targeted an end-tidal isoflurane concentration of 0.8 vol%, which we estimated to be equivalent to a MAC of 1.3.
Xenon/remifentanil
We maintained an end-tidal concentration of xenon of 63 ± 1 vol% with an inspiratory oxygen concentration of 29 ± 1%. Remifentanil was infused at 0.5 µg/kg/min. The MAC of xenon in dogs is 120% (Eger et al. 1965a) and so is impossible to apply under normobaric conditions. We therefore decided to use the highest concentration of xenon possible without risking hypoxia combined with the same dosage of remifentanil (0.5 µg/kg/min) as desribed above.
Depth of anaesthesia was assessed by eyelash reflex testing and by standardized tail-clamping (Eger et al. 1965b) and observation for signs such as tail wagging, eyelash reflex or movement of the limbs. Depth of anaesthesia was determined to be adequate in all cases. Animals were unrestrained and no neuromuscular blockade was used.
Measurements and calculations
HR, mean arterial blood pressure (MAP) and central venous pressure (CVP) were measured continuously, and data were stored on a computer. Measurements were begun before induction of anaesthesia and continued for 60 min during anaesthesia. CO was determined by transpulmonary thermodilution (5 mL injection volume at 5–10°C; Vigilance®, Baxter, Unterschleißheim, Germany). Five consecutive measurements were performed, the highest and the lowest values were rejected and the mean CO was calculated from the three remaining values (Jansen et al. 1990). Systemic (SVR) and pulmonary vascular resistance (PVR) and stroke volume (SV) were calculated by standard formulas: SVR = (MAP–CVP) × 80/CO; PVR = (MPAP−PCWP) × 80/CO; SV = CO/HR. Blood samples were taken to determine plasma hormones, plasma osmolarity (Osmometer 2, Fa. Roebling, Berlin, Germany), plasma electrolytes (flame photometry, AFM 5052, Eppendorf GmbH, Hamburg, Germany), arterial and mixed venous blood gases and base excess (ABL 505 analyser, Radiometer, Copenhagen, Denmark). Systemic oxygen consumption and delivery were calculated by standard formulas:
D˙O2(mL/min) = CO(SaO2[%] × Hb[g/dL] × 1.34[mL/g] + PaO2 [mmHg] × 0.003)
V˙O2(mL/min) = CO(Hb [g/dL] × 1.34 [mL/g] × (SaO2 [%]–SvO2 [%]) + 0.003 (PaO2 [mmHg]–PvO2 [mmHg])).
The blood withdrawn for analysis was immediately replaced with an equal amount of each dog's own blood precollected eight days previously. Blood samples were cooled and centrifuged at 4°C. Plasma was then stored at –20°C until analysis of plasma renin activity (New England Nuclear, North Billerica, MA, USA), angiotensin II (Eurodiagnostika, Arnhem, The Netherlands), atrial natriuretic peptide (Eurodiagnostika), aldosterone (DiaSorin GmbH, Düsseldorf, Germany) or vasopressin (Vasopressin-RIA, Biermann GmbH, Bad Nauheim, Germany) with radioimmunoassays, or with enzyme-linked immunosorbent assay as for plasma endothelin-1 (Biomedica, Austria). Adrenaline and noradrenaline were determined by high-performance liquid chromatography (Chromosystems, Munich, Germany) in serum that had been stored at –80°C.
Statistical analysis
All values are quoted as means ± SEM. A general linear model of analysis of variance (GLM ANOVA) for repeated measures was applied. Based on the ANOVA results, post hoc testing was performed with Student's t-test. The level of significance for first-order error was adjusted according to Holm's procedure (Schaffer 1986). Statistical significance was assumed at P < 0.05.
Results
Haemodynamics
Haemodynamic parameters
Values are presented as means ± SEM; n = 6. Values were measured after 30 min awake and after 60 min of anaesthesia
*P < 0.05 versus awake
† P < 0.05 versus xenon/remifentanil
N2O: nitrous oxide; MAP: mean arterial blood pressure
Plasma hormones
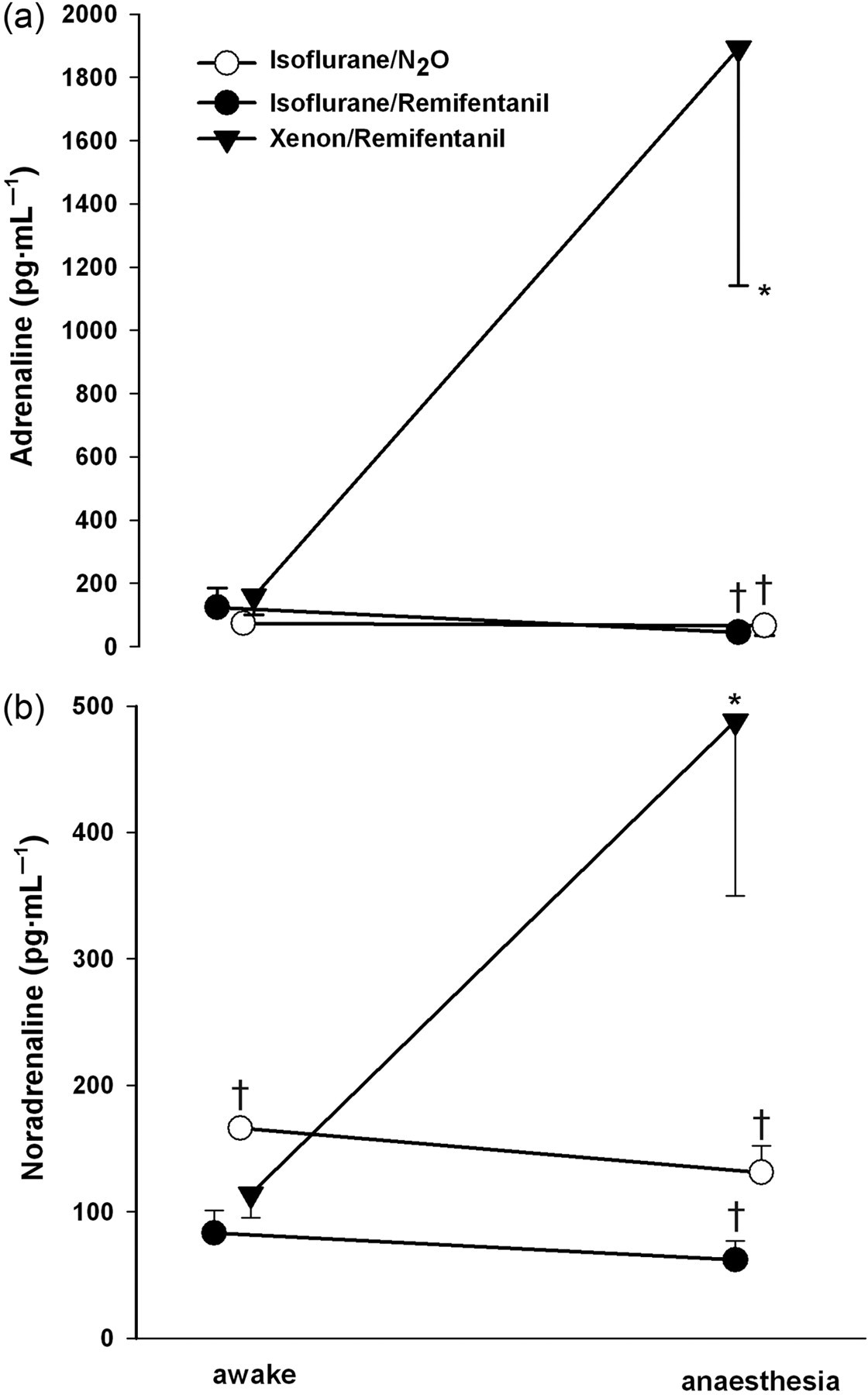
Plasma hormone concentrations were similar in awake dogs (Table 2, Figures 1 and 2). Plasma renin activity increased up to 10-fold during anaesthesia with no significant differences between protocols. Simultaneously, angiotensin II levels (Figure 1) increased during all types of anaesthesia, but this increase was attenuated during isoflurane/N2O anaesthesia. Angiotensin II plasma concentrations were parallelled by plasma concentrations of aldosterone (Table 2). Vasopressin concentrations increased during all anaesthesia regimens, but were highest during xenon/remifentanil anaesthesia (Figure 1). Interestingly, adrenaline and noradrenaline concentrations (Figure 2) rose only during xenon/remifentanil anaesthesia, but did not rise during both the isoflurane protocols. No substantial changes in plasma endothelin concentrations could be observed during the experiments. Only small variations were found for atrial natriuretic peptide during anaesthesia (Table 2).
Plasma hormones Values are presented as means ± SEM; n = 6. Values were measured after 30 min awake and after 60 min of anaesthesia *P < 0.05 versus awake
†
P < 0.05 versus xenon/remifentanil N2O: nitrous oxide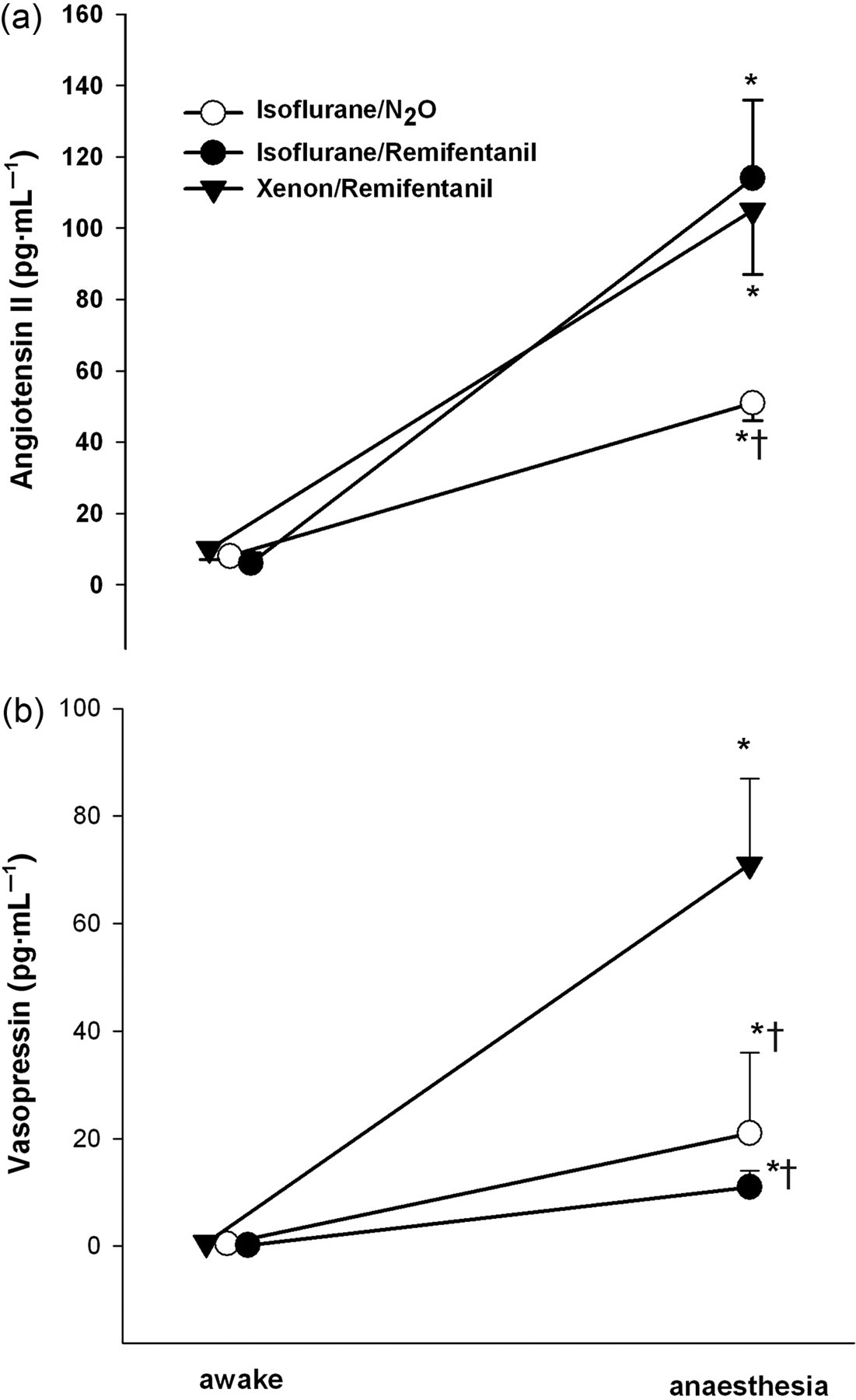
Blood gases and oxygen consumption
Inspired fraction of oxygen, blood gases, pH and base excess
Values are presented as means ± SEM; n = 6. Values were measured after 30 min awake and after 60 min of anaesthesia
*P < 0.05 versus awake
† P < 0.05 versus xenon/remifentanil
N2O: nitrous oxide; FiO2: inspired fraction of oxygen; PaO2: arterial oxygen tension; PvO2: mixed venous oxygen tension; PaCO2: arterial carbon dioxide tension; pHa: arterial pH; BEa: arterial base excess; SaO2: arterial haemoglobin oxygen saturation; SvO2: mixed venous haemoglobin oxygen saturation
Oxygen delivery, consumption and extraction
Values are presented as means ± SEM; n = 6. Values were measured after 30 min awake and after 60 min of anaesthesia
*P < 0.05 versus awake
† P < 0.05 versus xenon/remifentanil
N2O: nitrous oxide; D˙O2: total body oxygen delivery; V˙O2: total body oxygen consumption; C(a−v)O2: arterial to mixed venous oxygen content difference
Plasma electrolytes and osmolarity
Plasma osmolarity and plasma concentrations of sodium and potassium did not differ between protocols and were not substantially changed by any anaesthesia regimen.
Ventilatory parameters
Ventilatory parameters did not differ between protocols. Tidal volume was 195 ± 18 mL during isoflurane/N2O, 221 ± 6 mL during isoflurane/remifentanil, and 216 ± 8 mL during xenon/remifentanil anaesthesia. Respectively, minute volume amounted to 2.4 ± 0.2, 2.6 ± 0.1 and 2.5 ± 0.1/L/min, with a respiratory rate of 13 ± 1, 12 ± 1 and 12 ± 1/min, a mean airway pressure of 13 ± 1, 14 ± 1 and 14 ± 1 mmHg, and an end-tidal carbon dioxide tension of 35 ± 1, 37 ± 1 and 36 ± 1 mmHg, during the respective anaesthesia regimen.
Discussion
The objective of this study was to determine whether xenon/remifentanil has less cardiovascular and hormonal adverse effects than isoflurane/remifentanil or isoflurane/N2O anaesthesia. It was found that:
Xenon/remifentanil appears to reduce CO and HR to a greater extent than either isoflurane/remifentanil or isoflurane/N2O anaesthesia. MAP was unaffected by xenon/remifentanil anaesthesia, but decreased during isoflurane/N2O and isoflurane/remifentanil anaesthesia. Compared with the two isoflurane regimens, xenon/remifentanil anaesthesia produced higher plasma concentrations of adrenaline, noradrenaline and vasopressin resulting in excessively high SVR values in this group. Systemic oxygen consumption was unaffected by isoflurane/remifentanil anaesthesia, but reduced by isoflurane/N2O and xenon/remifentanil anaesthesia.
Haemodynamics
During isoflurane/N2O anaesthesia we found an increase in HR which is consistent with previous studies in dogs (Picker et al. 2001), and has been associated with reduced cardiac vagal activity. When isoflurane was combined with remifentanil we found a decrease in HR, as a result of the dose-dependent bradycardic effect of remifentanil in dogs (Michelsen et al. 1996). However, bradycardia was more pronounced when remifentanil was combined with xenon rather than isoflurane (Table 1). It therefore seems reasonable to conclude that in xenon/remifentanil anesthetized dogs bradycardia cannot be attributed to remifentanil alone. Indeed, a certain degree of bradycardia has also been described during xenon anaesthesia in pigs (Vagts et al. 2004) and humans (Rossaint et al. 2003, Coburn et al. 2005).
CO decreased during all anaesthesia regimens tested in our study. The decrease in CO was most pronounced during xenon/remifentanil anaesthesia (Table 1). As SV was only moderately reduced during xenon/remifentanil anaesthesia, the reduced CO is likely to be a consequence of bradycardia.
Vascular resistance was calculated from MAP and CO. For this reason, one cannot readily conclude that the high SVR observed in the xenon/remifentanil group was a response to the decrease in CO, e.g. due to direct myocardial effects. It could likely be reflective of neuro-hormonally mediated constriction of peripheral vessels (see below) elevating left ventricular afterload and thereby impairing CO.
Hormones
Activation of the renin–angiotensin–aldosterone system can be caused either by intrarenal stimuli of renin release (renal barostat and macula densa mechanisms) or by extrarenal stimuli (renal nerves or elevated plasma adrenaline and noradrenaline concentrations) (Davis & Freeman 1976). During xenon/remifentanil anaesthesia, the renin–angiotensin system was activated in the face of unaltered arterial blood pressure. Consequently, extrarenal factors such as plasma catecholamines would seem to be the important stimuli of the renin–angiotensin system when xenon is used. Indeed, xenon anaesthesia resulted in a four-fold increase in adrenaline and a 12-fold increase in noradrenaline concentrations (Figure 2). Since remifentanil has been shown to leave catecholamine concentrations unaltered and to suppress rather than to stimulate the sympathetic nervous system (Myre et al. 2003), we think that the increase of the catecholamines can be attributed to xenon rather than to remifentanil in the xenon/remifentanil protocol.
Vasopressin increased to vasopressor concentrations under all three regimens. We assume that the relatively higher levels of vasopressin, which were observed during xenon/remifentanil anaesthesia when compared with isoflurane/N2O or isoflurane/remifentanil anaesthesia, were most likely due to the xenon-induced adjustments of the sympathetic nervous system and release of adrenaline and noradrenaline (Figure 2). Other factors that might stimulate vasopressin release are linked either to a barostat and/or volustat mechanism that depends on left ventricular, aortic and carotid sinus receptors (Wang et al. 1988), or to changes in plasma osmolarity. None of these factors were present during the application of xenon in this study.
In summary, our results seem to suggest that the increase in SVR following xenon/remifentanil anaesthesia is a result of a neuro-hormonal compensatory response to the negative chronotropic effects of xenon. It could also be argued that the prominent increase in afterload is the cause of rather than the response to decreased CO. However, in this case one would have expected a more pronounced reduction of SV, which was only minimally affected. This might reflect a catecholamine-related positive inotropic effect to prevent a major reduction of SV in the face of increased afterload.
It was not possible to measure the effect of the various regimens used on myocardial function directly (e.g. by echocardiography). It is, however, known that xenon does not share the inhibitory effects of volatile anaesthetics on myocardial calcium channels and contractility of isolated muscle bundles (Hüneke et al. 2001, Schroth et al. 2002). Further it has not been shown to have any influence on left ventricular performance in dogs (Hettrick et al. 1998) or on fractional area change in transoesophageal echocardiography in humans (Luttropp et al. 1993).
Catecholamines during xenon anaesthesia
The increase in circulating catecholamines as encountered during xenon/remifentanil anaesthesia in our study has not previously been described. Existing studies have reported decreased or slightly increased catecholamine levels during xenon anaesthesia in pigs (Marx et al. 1997) and humans (Boomsma et al. 1990). In an acutely instrumented pig model (Vagts et al. 2004) xenon was associated with stable arterial blood pressure along with a distinct reduction in CO and intestinal perfusion. In that study, adrenaline concentrations of 3.35 nmol/L (∼615 pg/mL) and noradrenaline concentrations of 3.41 nmol/L (∼578 pg/mL) were measured in plasma when no xenon was added to a baseline intravenous anaesthesia, and no increase in catecholamines was observed upon the addition of up to 65 vol% of xenon (Vagts et al. 2004). That is, their catecholamine concentrations without xenon were already in a range found only after an increase during xenon/remifentanil anaesthesia in our study (Figure 2). Apart from species differences, this might reflect a certain degree of stress being put on the pigs during instrumentation as compared with our dogs that have been accustomed to the experimental situation through weeks of training (Francis et al. 2004). Beyond this, xenon was used on top of an established intravenous anaesthesia with ketamine and flunitrazepam. Both ketamine and xenon do inhibit the N-methyl-D-aspartate (NMDA) receptor (de Sousa et al. 2000, Gruss et al. 2004, Hüneke et al. 2004), and by this means, ketamine is involved in the release, metabolism and reuptake of adrenaline and noradrenaline (Kienbaum et al. 2000). Therefore, the high baseline catecholamine concentrations observed in the instrumented pig model may be caused by the ketamine masking or even abolishing a xenon-related increase of catecholamine concentrations. In contrast to volatile anaesthetics, xenon has been shown to be an NMDA receptor ligand (Yamakura & Harris 2000) and to increase catecholamine release in the brain (Yoshida et al. 2001). Stimulation of noradrenergic neurons in the hypothalamus may be one mechanism contributing to the sympathotonic effects of xenon (Preckel et al. 2006). Therefore, the release of catecholamines during xenon anaesthesia in our study might be related to an NMDA receptor-mediated effect in a ketamine-like fashion. Our study was not however designed to elucidate in detail by what means xenon leads to the increase of plasma catecholamines.
Depth of anaesthesia
It might be argued that high plasma catecholamine concentrations during xenon/remifentanil anaesthesia could reflect inadequate depth of anaesthesia. Indeed, our vol% dose of administered xenon was 63 ± 1%, whereas the MAC of xenon is actually 120% in dogs (Eger et al. 1965a). However, had the depth of anaesthesia been inadequate one would have expected to see a certain degree of stress response with its consequent pronounced activation of the renin–angiotensin system and tachycardia. This was not the case, however. To test the depth of anaesthesia, a tail-clamping technique was used. The tail-clamp (Eger et al. 1965a) is the equivalent of a skin incision in humans and is a recognized ‘golden standard’ in the assessment of MAC. MAC is defined as the anaesthetic concentration required to prevent 50% of patients from responding to a painful stimulus (skin incision, tail-clamp) with gross purposeful movements. The tail-clamp was firmly placed for 5 min immediately after induction and then at every 20 min interval during anaesthesia. We did not observe an increase in HR or blood pressure, withdrawal reactions, movement of the tail or limbs, wide pupils or the return of the eyelash reflex during any of the cases. The tail-clamp results were negative for all anaesthetic methods. Electrophysiological brain monitoring with bispectral index (BIS) has been used to monitor the degree of CNS depression in the anaesthetized dog (Greene et al. 2003) but it does not correlate with haemodynamic changes upon nociceptive stimuli (Nakayama et al. 2003, Carrasco-Jimenez et al. 2004) or MAC values and consequently cannot replace the tail-clamp in the assessment of adequate depth of anaesthesia.
Oxygen metabolism
Due to both the reduction in CO and the depth of anaesthesia, oxygen delivery declined during all protocols. During xenon/remifentanil anaesthesia oxygen consumption decreased along with a prominent decrease (61%) in CO. As a consequence, peripheral oxygen extraction, as expressed by c(a-v)O2, increased and was greater than during both isoflurane protocols (Table 4)
In contrast, Picker et al. (2002) have reported that xenon, when added to an established isoflurane anaesthesia, induces an increase in oxygen consumption without significantly altering CO. However, as isoflurane was used as an anaesthetic in combination with xenon their results are not comparable with our study.
Regarding metabolic changes (Table 3), base excess was not significantly different between protocols. Mixed venous oxygen saturation remained unchanged during isoflurane/N2O, where CO was reduced least, but decreased during isoflurane/remifentanil and xenon/remifentanil anaesthesia where CO was impaired most.
In summary, in our experimental dog model, xenon/remifentanil does not reduce anaesthesia-related cardiovascular and hormonal effects but actually produces its own unique set of haemodynamic and hormonal disturbances. Specifically, xenon/remifentanil does not alter MAP but nevertheless reduces CO to a greater extent than isoflurane-based anaesthesia. This is attributable to bradycardia along with extraordinarily high SVR and an exaggerated release of vasopressor hormones.
While xenon is well reputed in numerous investigations on haemodynamics that emphasize its haemodynamic neutrality, we demonstrated that the maintenance of MAP alone in the context of examining cardiovascular physiology is almost meaningless unless hormonal alterations and other haemodynamic parameters are taken into account.
These findings should be considered in the planning, performance and interpretation of investigations on cardiovascular and hormonal physiology in anaesthetized dogs and humans. They should encourage further research with emphasis on cardiovascular hormones during xenon anaesthesia in patients to evaluate in more detail whether xenon exerts potentially adverse haemodynamic or hormonal effects in humans.
Footnotes
Acknowledgements
We thank Rainer Mohnhaupt (Research Engineer), Daniela Bayerl and Birgit Brandt (Research Laboratory Technicians) for their valuable assistance during the experiments, and Martin McGuill for editorial assistance. The PhysioFlex™ ventilator was generously supplied by Dräger Medical AG, Lübeck, Germany, and xenon was provided by Air Liquide Deutschland GmbH, Germany.
