Abstract
Objectives
The purpose of this study was to evaluate symmetric dimethylarginine (SDMA) in hyperthyroid cats before and after treatment with radioactive iodine and to determine how pretreatment SDMA relates to the development of post-treatment azotemia.
Methods
Eighty-four non-azotemic hyperthyroid cats had serum SDMA and creatinine evaluated before and 1, 3 and 6 months after treatment with radioiodine therapy.
Results
Baseline SDMA was increased in 7% (n = 6/84) of cats, whereas SDMA was increased in 19% (n = 15/81), 20% (n = 16/80) and 32% (n = 26/81) at 1 month, 3 months and 6 months after treatment, respectively. Creatinine was not elevated in any of the cats at baseline because of the study design, and was elevated in 6% (n = 5/81), 15% (n = 12/80) and 15% (n = 12/81) of cats at 1, 3 and 6 months after treatment, respectively. SDMA (median 11 μg/dl, range 1–22 μg/dl) was significantly higher at 3 (12 μg/dl, range 6–45 μg/dl; P = 0.005) and 6 months (11 μg/dl, 6–25 μg/dl; P <0.001) compared with baseline (11 μg /dl, range 1–21 μg/dl). The median baseline SDMA was significantly higher in the azotemic group (13 μg/dl, range 11–22 μg/dl) compared with the non-azotemic group (10 μg/dl, range 1–21 μg/dl, P = 0.002). The sensitivity of SDMA for detecting azotemia after treatment was 15.4%, with a specificity of 94.4%. Baseline serum SDMA concentration had a moderately positive association with baseline creatinine concentration (P <0.001, r = 0.437). At 6 months, there was a strong positive correlation between SDMA and creatinine concentrations (P <0.001, r = 0.721). There was no significant correlation with SDMA and thyroxine at baseline (P = 0.772, r = −0.034) or 6 months (P = 0.492, r = −0.078).
Conclusions and relevance
SDMA increases in cats treated for hyperthyroidism with radioactive iodine and likely reflects associated changes in glomerular filtration rate. An increased SDMA concentration above the reference interval prior to treatment has a high specificity but poor sensitivity for the prediction of post-treatment azotemia.
Introduction
Cats develop numerous age-associated conditions, including hyperthyroidism and chronic kidney disease (CKD). Hyperthyroidism is one of the most common endocrine diseases of older cats, with a prevalence estimated at 10% in cats aged 10 years and older.1–6 Likewise, CKD develops in approximately 30% of cats by 15 years of age, 7 with pre-existing kidney disease being present in 14–40% of hyperthyroid cats.8–11 The diagnosis of CKD in cats with concurrent hyperthyroidism is problematic as hyperthyroidism can mask CKD as a result of increased glomerular filtration rate (GFR) and decreased muscle mass resulting in lower serum creatinine concentrations.12–15 This becomes particularly important when determining the ideal treatment and management strategies in hyperthyroid cats.
Radioiodine therapy is considered the treatment of choice as it is curative, non-invasive and has minimal risks. However, iatrogenic hypothyroidism can occur as a result of treatment and further decreases GFR and worsens azotemia. 16 Survival times are shorter in cats with iatrogenic hypothyroidism that develop azotemia, highlighting the serious consequences of decreased GFR in cats with pre-existing CKD. 16 The ability to detect pre-existing CKD in untreated hyperthyroid cats would be advantageous to guide patient monitoring and therapy.
A subnormal pretreatment GFR in cats with hyperthyroidism may predict the risk of developing post-treatment azotemia. 11 Unfortunately, measurement of GFR can be expensive and technically demanding to measure in clinical patients, and is not readily available in most practices. In addition, many cats with a normal GFR prior to treatment develop azotemia after resolution of hyperthyroidism.Pretreatment creatinine and blood urea nitrogen (BUN) are unreliable markers for the development of azotemia after treatment.17,18 While urine specific gravity (USG) has been shown not to predict post-treatment azotemia in most studies, a recent study found that a USG <1.035 was the most sensitive test (92%) for identifying post-treatment azotemia as compared with symmetric dimethylarginine (SDMA) and creatinine, but also the least specific test (71%). Markers of tubular dysfunction, such as reti-nol binding protein, urine protein:creatinine ratio, and N-acetyl-beta-D-glucosaminidase, have failed to predict the development of post-treatment azotemia.17–20 A methimazole trial can be performed to determine the effects of restoration of euthyroidism on renal function. 21 This can be impractical for clients who cannot medicate their cats and potentially harmful to cats that develop adverse reactions, such as facial excoriations, blood dyscrasias and hepatotoxicity, to the drug. 22 Additionally, it is unknown if response to methimazole therapy accurately predicts the presence and severity of CKD in cats that undergo radioiodine treatment.
SDMA is a methylated amino acid derivative that is released by all cells within the body after proteolysis and is excreted exclusively by the kidney. SDMA is more sensitive than creatinine for detecting CKD with decreases in GFR of ⩾40% and ⩾75% leading to increases in serum SDMA and creatinine, respectively. 23 SDMA is not affected by changes in lean body mass giving it another advantage over creatinine, particularly in hyperthyroid cats, which tend to have decreased muscle mass.24,25 Recently, SDMA was evaluated as a predictor of azotemia after treatment with radioiodine therapy in hyperthyroid cats. In the aforementioned study, an elevated SDMA had a sensitivity and specificity of 33% and 97%, respectively, for the detection of post-treatment azotemia. 26 In contrast, another study found that SDMA was not helpful in predicting abnormal GFR in hyperthyroid cats after treatment, but the number of cats with impaired renal function was low. 27 While it appears that SDMA may have some use in predicting kidney function in hyperthyroid cats, the poor sensitivity is a substantial limitation.
The purpose of this study was to evaluate SDMA in hyperthyroid cats before and after treatment with radioactive iodine and to determine how pretreatment SDMA relates to the development of post-treatment azotemia. We hypothesized that cats with an elevated pretreatment SDMA were more likely to become azotemic once euthyroidism was achieved. A second aim of this study was to evaluate SDMA and its relation to thyroid hormone status – in particular, to determine if SDMA could be used as an indication of kidney dysfunction in cats with iatrogenic hypothyroidism. We hypothesized that SDMA would be higher in cats with iatrogenic hypothyroidism than in euthyroid cats post-treatment with radioactive iodine.
Materials and methods
Case enrollment
In this prospective study, cats diagnosed with hyperthyroidism presenting to the Virginia Maryland College of Veterinary Medicine (VMCVM) or Veterinary Referral Associates (VRA) for radioactive iodine therapy were enrolled from December 2015 to May 2017. Cats were excluded if serum creatinine was above the reference interval (RI) prior to radioiodine administration. Cats that were azotemic while receiving medical therapy (methimazole or low iodine diet) were not excluded provided azotemia was absent at the time of presentation for radioactive iodine treatment. Cats that received methimazole or low-iodine diets within 1 week of presentation were excluded. Routine complete blood count, biochemical profile, urinalysis, serum SDMA and thyroid function tests (total thyroxine [T4]; and thyrotropin [TSH]) were performed within 6 weeks of treatment (baseline). Subsequent testing consisting of serum concentrations of T4, TSH, BUN, creatinine and SDMA, and urinalysis was performed 1, 3 and 6 months after radioiodine administration.
Blood and urine samples were analyzed at VMCVM Virginia Tech Animal Laboratory Services (ViTALS) or an IDEXX reference laboratory. Cats were subsequently categorized as azotemic or non-azotemic, as well as euthyroid or hypothyroid, based on data from the last available evaluation (at least 3 months post-treatment). Azotemia was defined as either a serum creatinine concentration above the RI (0.9–2.3 mg/dl) on two occasions or an increased serum creatinine in conjunction with a USG <1.035. 26 Thyroid function was classified based on serum T4 and TSH concentrations. Cats were classified as euthyroid based on serum T4 and TSH concentrations within their respective RIs. Cats were categorized as overtly hypothyroid if serum T4 concentration was below the RI with a serum TSH above the RI and subclinically hypothyroid if serum T4 was low–normal with a serum TSH above the RI. Cats that remained persistently hyperthyroid at the last available evaluation (at least 3 months after treatment with radioactive iodine) were excluded. Cats were categorized as having either a normal or an elevated serum SDMA concentration (RI 0–14 μg/dl).The decision to institute levothyroxine therapy in iatrogenic hypothyroid cats was based on the development of azotemia, clinical signs, time from treatment and clinician discretion. Cats with iatrogenic hypothyroidism received levothyroxine supplementation (initial dose of 0.1 mg/day orally) if azotemic at 3 months after radioactive iodine treatment, or displayed clinical signs of hypothyroidism (lethargy, seborrhea, poor haircoat, obesity) after 3 months.
Data from 13 of the cats in this study were included in another. 28 Dose determination for radioactive iodine was based on a variable dosing protocol in cats treated at VMCVM or based on serum T4 concentrations ± thyroid scintigraphy in cats treated at VRA.
Laboratory analysis
Blood and urine samples were submitted immediately upon collection to the ViTALS of the VMCVM for a biochemistry profile and a urinalysis, or were stored at 4°C before shipping to the IDEXX reference laboratory. Blood for SDMA and creatinine was centrifuged within 1 h of collection. Serum was harvested then stored at either −70°C for up to 2.5 years or was stored at 4°C before shipping to a reference laboratory for analysis.
SDMA concentration was analyzed using a previously validated immunoassay (IDEXX Laboratories). 29 The day-to-day precision analysis of low SDMA concentration samples and high concentration samples showed a coefficient of variation of 7.7% and 2.3%, respectively. 30
Serum urea nitrogen and creatinine concentrations were measured with a Beckman Coulter AU 480 analyzer using an enzymatic method and by a modified Jaffe method, respectively. The upper limit of the RI for creatinine was 2.3 mg/dl, based on a 95% confidence interval of values for healthy cats. The IDEXX creatinine reference intervals were determined following CLIA guidelines using >120 normal cats (EP28-A3c; Defining, Establishing, and Verifying Reference Intervals in the Clinical laboratory; Approved Guideline).
Serum T4 and TSH concentrations were measured using the canine total T4 and TSH chemiluminescent enzyme immunoassays (Immulite 1000; Siemens Healthcare Diagnostics). The RI for serum T4 concentrations at the ViTALS was 16.0–37.7 nmol/l and 10.2–59.9 nmol/l for the IDEXX reference laboratory. The reference interval for TSH was <0.03–0.3 ng/ml.
Statistical analysis
Normal probability plots showed that continuous data were skewed. Accordingly, continuous variables were summarized as medians (range), while categorical variables were summarized as contingency tables. Effect of time on outcomes (SDMA, creatinine, USG) was assessed using Friedman’s χ2 test with cat identification as a blocking factor. P values for all two-way comparisons were adjusted for multiple comparisons using Bonferroni’s procedure. Correlations between creatinine and SDMA were assessed using scatterplots and Spearman’s non-parametric correlation coefficients. Sensitivity and specificity of baseline SMDA as a predictor of azotemia after treatment were generated using receiver–operating characteristics (ROC) curve analysis. Associations between baseline measurements and the development of azotemia was assessed using the Wilcoxon rank sum test. Associations between hypothyroid/euthyroid and azotemia status were assessed using Fisher’s exact test. Concentrations of serum SDMA and creatinine were compared between hypothyroid and euthyroid cats (post-radioiodine treatment) using the Wilcoxon rank sum test. Statistical significance was defined as P <0.05. ROC curve analysis was performed using JMP pro version 14. All other analyses were performed using SAS version 9.4.
Results
Eighty-four cats were enrolled in the study. Median age was 13 years (range 7–19 years). Breeds included were domestic shorthair (n = 66 [78.6%]), domestic longhair (n = 7 [8.3%]), Ragdoll (n = 3 [3.6%]), domestic mediumhair (n = 2 [2.4%]), Maine Coon (n = 2 [2.6%]) and one of each of the following breeds: Egyptian Mau, Himalayan, Persian and Russian Blue. Forty-two (50%) cats were female and 42 (50%) were male; all cats were neutered. Median weight was 4.2 kg (range 2.5–7.2 kg). All cats had follow-up testing for at least two time points during the study. The follow-up time for the study ranged from 3 to 9 months, with a median of 6 months. The median dose of radioactive iodine was 2.6 mCi (range 1.3–4.5 mCi).
Baseline SDMA was above the RI in 6/84 (7%) cats, whereas SDMA was elevated in 15/81 (19%), 16/80 (20%) and 26/81 (32%) at 1, 3 and 6 months after radioiodine administration, respectively. Serum SDMA concentrations were significantly higher at 3 and 6 months compared with baseline (Table 1, Figure 1). Creatinine was elevated in 5/81 (6%), 12/80 (15%) and 12/81 (15%) cats at 1, 3 and 6 months, respectively. USG was <1.035 in 39/76 (51%), 44/74 (59%), 45/76 (59%) and 43/70 (61%) at baseline, 1, 3 and 6 months, respectively. One cat had a USG of 1.007 at 3 months but was 1.013 at 6 months. Serum creatinine and BUN concentrations were significantly higher 1, 3 and 6 months after treatment (Table 1). USG was not significantly different at any time point (Table 1). Serum concentrations of T4 (Figure 2) and TSH (Table 1) significantly decreased and increased compared with baseline, respectively.
Baseline biochemical variables compared with 1, 3 and 6 months after treatment

Data are median (range). Significant differences in data distribution between time points is indicated by the same superscript letter
Lower limit of detection is <0.03 ng/ml and is listed as 0.02 for analyses
SDMA = symmetric dimethylarginine; BUN = blood urea nitrogen; USG = urine specific gravity; TSH = thyrotropin

Box plot of serum symmetric dimethylarginine (SDMA) concentrations prior to treatment (baseline) and 1, 3 and 6 months after treatment with radioactive iodine. Boxes represent the interquartile range (IQR) from the first and third quartile. The horizontal bar in each box represents the median value. The T represents 1.5 times the IQR above the third and below the first quartile ranges. Asterisks represent outlying data points. Shaded box represents data inside the reference interval (0–14 µg/dl)

Box plot of serum thyroxine (T4) concentrations prior to treatment (baseline) and 1, 3 and 6 months after treatment with radioactive iodine. Boxes represent the interquartile range (IQR) from the first and third quartile. The horizontal bar in each box represents the median value. The T represents 1.5 times the IQR above the third and below the first quartile ranges. Asterisks represent outlying data points
SDMA as a predictor of post-treatment azotemia
Follow-up was available for 81 cats at 6 months. In three cats, data at 3 months were used to determine the prevalence of azotemia. Of the 84 cats, 13 (15%) developed azotemia after treatment.Baseline SDMA was elevated in two cats that developed azotemia, whereas the other four cats with an elevated baseline SDMA remained non-azotemic. Post-treatment SDMA was within the RI in one cat that had an elevated baseline SDMA. Median post-treatment creatinine and USG in the six cats with an elevated SDMA were 2 mg/dl (range 1.9–3.0 mg/dl) and 1.015 mg/dl (range 1.013–1.025 mg/dl), respectively. The sensitivity of an elevated pretreatment SDMA (>14 µg/dl) for detecting azotemia after treatment was 15.4%, with a specificity of 94.4%. In comparison, a baseline USG of <1.035 had a sensitivity of 90.9% and specificity of 55.4%. A pretreatment creatinine concentration ⩾1.6 mg/dl had a sensitivity and specificity of 15.3% and 98.6%, respectively, and an increased pretreatment BUN (>32 mg/dl) had a sensitivity and specificity of 33.3% and 97.18%, respectively. ROC curve (area under the curve = 0.84) analysis indicated that a baseline serum SDMA concentration of 11 (100% sensitivity, 57% specificity) acts as a good screening test for azotemia 6 months after treatment with radioiodine.
The median baseline serum SDMA concentration was significantly higher in the azotemic group (13 µg/dl, range 11–22 µg/dl) compared with the non-azotemic group (10 µg/dl, range 1–21 µg/dl [P = 0.002]; Figure 3, Table 2). Cats in the azotemic group were also significantly older, had lower body weights, higher serum BUN and creatinine concentrations, and lower USGs compared with the non-azotemic group at baseline. Baseline serum SDMA concentration had a moderate positive association with baseline creatinine concentration (P <0.001, r = 0.437). At 6 months, there was a strong positive correlation between SDMA and creatinine concentrations (P <0.001, r = 0.721). There were no significant correlations with SDMA and T4 or SDMA and TSH at baseline (P = 0.772, r = −0.034; P = 0.602, r = 0.06) or 6 months (P = 0.492, r = −0.078; P = 0.469, r = 0.084).
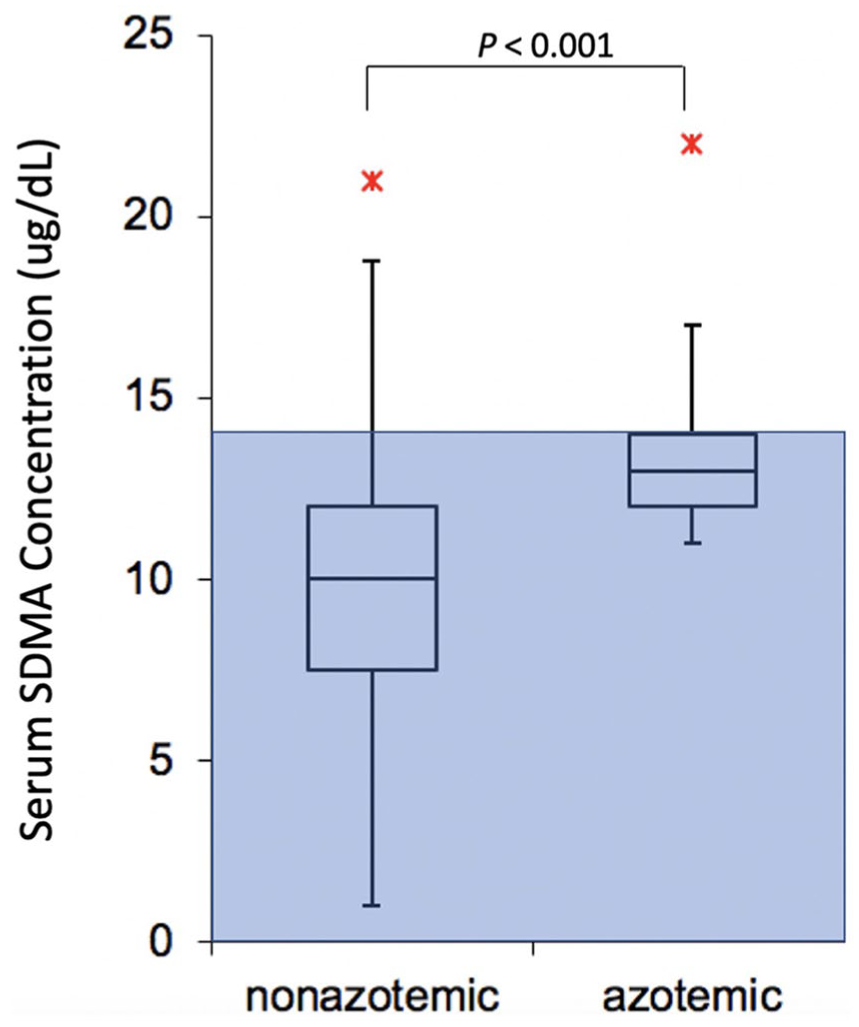
Box plot of baseline serum symmetric dimethylarginine (SDMA) concentrations in azotemic and non-azotemic cats. Boxes represent the interquartile range (IQR) from the first and third quartile. The horizontal bar in each box represents the median value. The T represents 1.5 times the IQR above the third and below the first quartile ranges. Asterisks represent outlying data points. Shaded box represents data inside the reference interval (0–14 µg/dl)
Comparison of baseline variables between cats with and without azotemia 6 months after treatment

Data are median (range)
n = 64 non-azotemic
n = 65 non-azotemic and 11 azotemic cats
n = 64 non-azotemic
SDMA = symmetric dimethylarginine; USG = urine specific gravity; BUN = blood urea nitrogen
SDMA in relation to post-treatment thyroid status
After treatment, 57/84 (68%) cats were euthyroid and 27/84 (32%) cats were hypothyroid. Of the hypothyroid cats, 11/27 (41%) had overt hypothyroidism and 16/27 (59%) had subclinical hypothyroidism. SDMA was above the RI in 16/57 (28%), 4/11 (36%) and 5/16 (31%) of euthyroid, overtly hypothyroid and subclinically hypothyroid cats, respectively (P = 0.82). Post-treatment serum SDMA and creatinine concentrations were not significantly different between hypothyroid or euthyroid cats (Table 3). Azotemia was observed in 8/57 (14%), 2/11 (18%) and 2/16 (12.5%) of euthyroid, overtly hypothyroid and subclinically hypothyroid cats, respectively (P = 0.895). Data collected after treatment with levothyroxine from four cats were excluded from thyroid status analyses.
Comparison of median (range) serum symmetric dimethylarginine (SDMA) and creatinine concentrations between euthyroid and hypothyroid cats 6 months after treatment

Data are median (range)
Hypothyroid cats treated with levothyroxine were excluded
Four hypothyroid cats were treated with levothyroxine starting at 3 months following radioiodine treatment. Two of the four cats were azotemic and had an elevated SDMA when levothyroxine treatment was initiated. Follow-up laboratory tests were available 3 months after initiating treatment with levothyroxine where serum T4 concentration was within the RI in three cats and low in the remaining cat. Serum TSH concentration remained above the RI in all four cats. SDMA decreased in two cats, remained the same in one cat and increased in the other, whereas creatinine remained the same in two cats, increased in one cat and decreased in one cat.
Discussion
Pretreatment SDMA is an insensitive but specific test for the development of azotemia in cats treated for hyperthyroidism with radioiodine therapy. A similar specificity with a slightly higher sensitivity was found in another study of SDMA in hyperthyroid cats. 26 In the present study, SDMA was significantly higher in cats that developed azotemia; however, there was considerable overlap between the groups. Our findings suggest that a hyperthyroid cat with a pretreatment SDMA <10 μg/dl would be unlikely to develop azotemia post-treatment. When evaluating baseline SDMA, BUN and USG, a baseline USG <1.035 was most sensitive (90.9%) and an increased baseline SDMA the most specific. When baseline creatinine was included with a cut-off of ⩾1.6 mg/dl, this test was the most specific for the detection of post-treatment azotemia.
As indirect markers of GFR, both serum SDMA and creatinine concentrations increased after radioiodine treatment of hyperthyroid cats. In addition, SDMA correlated with creatinine but not T4, indicating that changes in SDMA are due to changes in GFR rather than thyroid function. In one study pertaining to hyperthyroid cats after 6 months of treatment with Hill’s y/d, SDMA was significantly correlated with GFR (r = 0.71) and less so with serum creatinine (r = 0.55). 31 In a more recent study of hyperthyroid cats 1 month after treatment with radioactive iodine, GFR was insignificantly correlated with SDMA but significantly correlated with serum creatinine. 27 These studies were limited by sample size, the number of cats achieving euthyroidism and few cats with post-treatment azotemia. Further studies are necessary in a larger group of hyperthyroid cats with a higher prevalence of pre-existing CKD to better ascertain how well SDMA correlates with GFR.
The increase in serum SDMA noted after treatment in hyperthyroid cats may be time dependent. Results reported here are similar to those found by Buresova et al, 27 where SDMA did not significantly increase 1 month after treatment. However, a significantly increased SDMA level was found with longer follow-up in the present study, as previously reported by Peterson et al. 26 Interestingly, mild increases in SDMA in a few cats may also normalize after treatment, which was seen in one cat in the current study.26,27
Baseline SDMA, creatinine, USG, age and body weight were significantly different between azotemic and non-azotemic cats after radioiodine treatment. One non-azotemic cat in this study had a USG <1.008 at 3 months but was 1.013 at the end of the study. The occurrence of hyposthenuria in this cat was unclear. Baseline SDMA was elevated in four cats that did not develop azotemia. It is possible that cats had an increased SDMA but normal creatinine concentrations due to the effects of muscle mass on creatinine concentrations but not SDMA.24,25 Additionally, a higher proportion of cats had an increased SDMA compared with creatinine. About half of treated hyperthyroid cats may fail to regain normal muscle mass despite improvement in body weight. 32 A reduced muscle mass might lead to lower creatinine concentration in a cat that would otherwise be azotemic where SDMA indicates impaired renal function. Additionally, depending on the degree of GFR reduction, serum SDMA increases prior to serum creatinine as it is more sensitive for detection of decreases in GFR in cats with CKD. 23 We did not collect data regarding muscle condition scores or concurrently measured GFR to investigate if SDMA is a better reflection of renal function than serum creatinine in hyperthyroid cats before and after treatment.
The prevalence of increased SDMA and azotemia and concentrations of serum SDMA and creatinine were not significantly different between hypothyroid cats and euthyroid cats. These findings are in contrast to some other studies where hypothyroid cats are more likely to be azotemic than euthyroid cats.16,26,33 The inability to detect a significant difference in our study could be due to low statistical power given the small numbers of azotemic and hypothyroid cats. Additionally, our study had a lower prevalence of pre-existing CKD in cats that developed hypothyroidism, as well as differences in the definition of hypothyroidism vs previous studies. SDMA as tool for monitoring treatment for hypothyroidism as a surrogate for GFR has not been evaluated. There were only four hypothyroid cats treated with levothyroxine during the study and therefore we were unable to evaluate the use of SDMA in monitoring hypothyroid cats treated with levothyroxine. Further studies are needed to investigate this, as well as the survival of hypothyroid cats with an elevated SDMA compared with euthyroid or hypothyroid cats without elevated SDMA.
This study was performed at two locations and SDMA and creatinine were analyzed after samples were stored frozen for up to 2.5 years at one location, and the analytes were measured on refrigerated serum soon after collection from the other location. While there has been no study evaluating the long-term stability of SDMA in serum stored at −70°C, numerous reports have used serum stored for more than 5 years without apparent effect. Separate analysis from each institution revealed similar results. Additionally, SDMA and creatinine concentrations were evaluated at the same laboratory (IDEXX) to limit variability. Data from treated cats that were on levothyroxine at the 6-month evaluation were included in analyses except for post-treatment thyroid status in relation to SDMA and azotemia. However, we ran all analyses with and without 6-month data from these cats, which yielded similar results. Another limitation is that the study was likely underpowered to detect changes in creatinine and SDMA in relation to thyroid status. The definition of azotemia in this study did not include increased serum BUN and five cats did not have documented persistent hypercreatininemia as this was limited by study duration. For these cats to be considered azotemic in this study, a concurrent inappropriately concentrated USG was necessary in conjunction with an increased serum creatinine concentration. This could have had the potential to overestimate azotemia. Additionally, as BUN can be affected by other diseases besides renal function, one being hyperthyroidism due to increased protein metabolism, we did not use this as a criterion. Lack of BUN in the definition of azotemia could have underestimated azotemia in our population.
Conclusions
SDMA increases in cats treated for hyperthyroidism with radioactive iodine and likely reflects associated changes in GFR. Baseline SDMA has a high specificity but poor sensitivity for the prediction of post-treatment azotemia. Given the high prevalence of hyperthyroidism and CKD in older cats, SDMA concentrations should be considered in conjunction with serum creatinine and USG when evaluating kidney function. Further studies are necessary to determine if SDMA is a more reliable indicator of kidney function than serum creatinine in hyperthyroid cats before and after treatment.
Footnotes
Acknowledgements
SDMA assays were performed by IDEXX free of charge.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported in part by the Virginia-Maryland College of Veterinary Medicine Veterinary Memorial Fund.
