Abstract
Objectives
The aim of this study was to determine the prevalence of post-contrast acute kidney injury or comparable side effects on kidney function in cats receiving the non-ionic, iodinated agent ioversol and/or paramagnetic agent gadoteric acid.
Methods
Fifty-two animals were divided into four groups on the basis of contrast medium administration for imaging: ioversol (n = 27), gadoteric acid (n = 12), dual contrast media (n = 4) or control, which received an infusion of isotone intravenous fluids only during anaesthesia (n = 9). Blood and urine samples were obtained three times after contrast administration and compared with values obtained prior to administration of the contrast medium. Creatinine (<1.60 mg/dl), symmetric dimethylarginine (SDMA; ⩽14 μg/dl), urine protein:creatinine ratio (UPC; <0.2) and critical differences for creatinine (<0.3 mg/dl) and SDMA (<5.98 μg/dl) were measured.
Results
No significant short-term effects on mean creatinine, SDMA and UPC measurements were seen. Borderline proteinuria (UPC, 0.2–0.4) was detected in 11.4% of cases after contrast media administration. A UPC of more than 0.2 in five cases indicated that contrast media may affect kidney function, leading to (transient) proteinuria.
Conclusions and relevance
This study found no side effect on renal function following the administration of ioversol or gadoteric acid, provided patients were adequately hydrated. However, the clinical relevance of proteinuria in some cats needs to be evaluated in future studies.
Introduction
The use of advanced diagnostic imaging such as CT and MRI, including intravenous (IV) administration of contrast media, is increasing in pets. In human medicine, the use of IV contrast media has been associated with post-contrast acute kidney injury (PC-AKI) in several clinical studies. 1 PC-AKI is defined in humans as an increase of ⩾0.3 mg/dl serum creatinine or an increase of 1.5−1.9 times baseline serum creatinine 48 h after intravascular contrast administration.1,2 A decline of urine output <0.5 ml/kg/h for 6−12 h is diagnostic for PC-AKI as well; other reasons for renal impairment have to be excluded.1,2 PC-AKI is also referred to as contrast media-induced kidney injury (CIN). 3 Contrast-induced acute kidney injury (CIAKI) describes cases in which a relationship between contrast media administration and decline of renal function has been proven.3,4 Differing study results from previous investigations opened up a controversial discussion about the prevalence, risk factors and prevention of PC-AKI in humans. 5 The findings ranged from a PC-AKI prevalence of more than 10% in an outpatient population to some studies concluding that PC-AKI was overestimated in humans.3,6,7 Some recognised risk factors for contrast media use include altered renal function, diabetes mellitus, older age, non-steroidal anti-inflammatory drug (NSAID) use and severe heart failure.8–10 Thus, an accepted risk factor such as chronic kidney disease (CKD) is common in cats and the toxic effects caused by certain drugs on feline kidneys have been proven; it is possible that renal function may be affected after contrast media administration.11,12 Additionally, the prevalence of CKD in cats increases with older age, resulting in a patient population that matches the risk factors for PC-AKI in humans.8,12
In a retrospective case review, a PC-AKI prevalence of 7.6% was found in a canine population of 86 individuals. 13 One of the dogs also had immune-mediated haemolytic anaemia and was euthanased due to oliguria associated with deteriorating renal function. Necropsy revealed severe tubular degeneration and necrosis, which did not allow the cause of acute kidney injury (AKI) to be defined. 13 However, a toxicological study from 1989 examined different dosages of ioversol administered to rats, mice and dogs in different settings. The acute IV toxicity studies in dogs did not cause clinical alterations in serum biochemistry tests sampled before and on days 1 and 14 after administration of ioversol at different sublethal dosages. Terminal necropsy also revealed no changes. 14
A study in which cats and dogs received contrast media for CT and MRI reported that 24.4% of cats showed mild (12.2%) to moderate (12.2%) hemodynamic alterations after administration of the gadolinium-based agent, gadobutrol, and 31.0% of cats presented with mild (13.8%) to moderate (17.2%) hemodynamic alterations after receiving the ionic iodised agent, iohexol. A change in mean arterial blood pressure (MAP) was detected in both directions after use of the two contrast agents; a change exceeding 20% difference from baseline level correlated with an increase in blood pressure. 15 Pollard et al looked at haemodynamic alterations (heart rate, peak systolic pressure) during anaesthesia of dogs receiving gadolinium-based contrast media, ionic-iodinated and non-ionic-iodinated contrast for diagnostic imaging. A fourth group of dogs that received no contrast media served as the control group.16 They found slightly more cases of hyper- and hypotension in dogs after IV contrast administration in comparison with humans, which possibly confirms that general anaesthesia may play a role in dogs. All measured alterations were categorised as moderate in severity based on the human literature. Ionic iodinated agents were found to have a higher incidence of immediate haemodynamic changes than non-ionic and gadolinium-based agents in dogs. 16 As hypertension has been recognised as a risk factor in the development of CKD in cats, increased arterial pressure after contrast medium administration may contribute to the worsening of kidney function or the development of kidney disease.12,17 Among other alterations, a decreased heart rate after iohexol injection is another reported effect, while an increased heart rate is visible after gadobutrol injection in cats. 15 One retrospective study investigated serum biochemistry with available pre- and post-contrast values of serum creatinine and blood urea nitrogen (BUN) in 27 cats and did not find significant changes. 16
Although the pathophysiology of PC-AKI is not completely understood, alterations in blood flow represent one part of a multifactorial process that induces hypoxia and apoptosis in the renal cells; these alterations are more pronounced after administration of ionic contrast media when compared with non-ionic contrast media. 18 The high concentrations of iodine owing to the filtration of radiocontrast agent cause hypertonic conditions in the renal tubules. 18 A key factor in these alterations is the tubular effect, which is comparable to acute tubular necrosis observed by several in vitro studies on mostly Madin Darby Canine Kidney (MCDK) cell lines; these studies indicate the destruction of the typically polarised cell-layer structure, which leads to apoptosis.18–20
The aims of this study were to identify changes in renal biomarkers, especially PC-AKI-related changes in serum creatinine, and to identify alterations in the urine of cats that received the non-ionic iodinated agent ioversol, and/or the paramagnetic gadolinium-based agent gadoteric acid.
Materials and methods
Study population
Cats that were presented to the Diagnostic Imaging, Department for Small Animals and Horses of the University of Veterinary Medicine, Vienna, for further diagnostic work-ups and/or radiation therapy planning were included in the study after obtaining the owner’s written consent following the standards of the Ethics and Animal Welfare Committee (ETK).
Ethical approval
The project was a prospective clinical cohort study. This work involved the use of non-experimental client-owned animals. The study design was approved by both the ETK of the University of Veterinary Medicine, Vienna, and the Austrian Federal Ministry for Science, Research and Economy. The study was categorised as a low-invasive animal experiment (animal trial number BMBWF-68.205/45-V/3b/2019).
Study groups
The cats were divided into four groups based on the type of contrast media received. One group received only medication for maintenance of general anaesthesia without the use of a contrast medium and served as the control group (ctr). Another group received the non-ionic iodated agent, ioversol (Optiray; Guerbet) (group ioversol [iov]). The third group received gadoteric acid (Dotarem; Guerbet) for the MRI (group gadoteric acid [gad]). A fourth group underwent both modalities and received both contrast media (group ioversol and gadoteric acid [iov + gad]). The radiologist in charge decided, in agreement with the anaesthesiologist monitoring the cat’s clinical state, whether the administration of a contrast medium was necessary. This decision was made completely independently of the authors (Figure 1).

Decision tree for contrast media administration in the patient presented for diagnostic imaging. CM = contrast media; gad = group gadoteric acid; iov = group ioversol; iov+gad = group ioversol+gadoteric acid; ctr = control group
Exclusion criteria
Each routine clinical examination of a cat started with a blood pressure measurement following the Consensus Statement of the American College of Veterinary Internal Medicine on blood pressure measurement, 21 with a cuff width of 30–40% of the circumference of the cat’s forelimb 22 where the cuff was placed. Five measurements were obtained and the average was calculated. Indirect blood pressure measurements with Doppler ultrasonography were obtained to detect hypertension, which was defined as systolic blood pressure of more than 160 mmHg 23 and served as an exclusion criterion. The cat’s stress level was kept as low as possible. If feasible, the cat had time to acclimate to the environment for approximately 10 mins while the room was kept quiet.
The cats underwent a shortened abdominal ultrasound (Philips iU22, 8–5 MHz, micro convex transducer), performed by one of the authors (MLP). The kidneys, ureters, bladder and proximal urethral structure were examined by ultrasound for anatomical variants or major pathomorphological traumatic, neoplastic, congenital, degenerative and vascular findings. If a major abnormality compromising the kidney function was found, the cat was excluded. If the clinical status of the cat was not stable or the cat was already in intensive care, it was excluded. Figure 2 shows the study design.

Study design.
Anaesthesia
The anaesthesia protocol for CT or MRI was standardised as follows. For premedication, butorphanol (Alvegesic; Virbac, 0.2 mg/kg body weight [BW]), midazolam (Dormicum; Cheplapharm, 0.2 mg/kg BW) and ketamine (Ketamidor; Richter Pharma, 1 mg/kg BW) were applied via an IV or intramuscular (IM) injection. For induction, propofol (Propofol; Fresenius, 1–3 mg/kg) or alfaxalon (Alfaxan Multidose; Jurox [UK], 1–2 mg/kg) was administered IV. After orotracheal intubation, anaesthesia was maintained with isoflurane (CT) or sevoflurane (MRI) in oxygen (fraction of inspired oxygen = 1.0). In 12/53 cats, slightly modified protocols were used. For the CT contrast study, ioversol (Optiray; Guerbet) was injected at a dose of 2 ml/kg BW via an injection pump at a flow rate of 0.7 ml/s. For the MRI contrast study, gadoteric acid (Dotarem; Guerbet) was administered manually at a dose of 0.2 ml/kg. During anaesthesia, each cat was infused with isotone IV fluids (Sterofundin; Braun) at a dosage of 3–5 ml/kg/h. During the CT examination, the cats were warmed with forced hot air (Bair Hugger); during the MRI examination, the cats were kept warm with blankets.
Blood tests
Blood samples were drawn after placement of a 22 G catheter (Vasofix; Braunüle) or by needle puncture mainly from the cephalic vein. Blood samples were collected into lithium heparinised tubes and into K2-EDTA tubes.
A complete blood count was performed from EDTA anticoagulated whole blood by a laser-based hematology unit (ADVIA 2120i; Siemens) using veterinary software with feline settings. Quality control included daily measurements of quality control material at three different levels (testpoint low, testpoint normal, testpoint high; Siemens). A blood smear was prepared for each sample and microscopically evaluated by a trained technician or board-certified clinical pathologist (IS) when numerical results exceeded the lower and upper cut-offs for more than 25% or analyses (dot plots and histograms) indicated possible erroneous results. Biochemistry analyses were performed from Li-heparin plasma by a fully selective autoanalyser for clinical chemistry (Cobas 501c; Roche Diagnostics). Tests performed at the hospital laboratory and the respective analytical methods are summarised in Table 1. Plasma osmolality was determined by a freezing point osmometer Modell 3320 (Advanced Instruments). Symmetric dimethylarginine (SDMA) was analysed by an external laboratory (IDEXX) by a high throughput immunoassay. Results were reported in micrograms per decilitre. Test results were evaluated in comparison with the respective reference intervals and the magnitude of changes between time points were compared with the critical differences (CDs; ie, subtraction of day X creatinine, SDMA or urea level [minuend] from the respective day 0 values [subtrahend]) reported in the literature.24–26 In this study we focused on the CDs in creatinine results between day 0 and day 1 (⩾0.30 mg/dl) for each individual.
Analytical methods based on International Federation of Clinical Chemistry and Laboratory medicine recommendations (www.ifcc.org/)
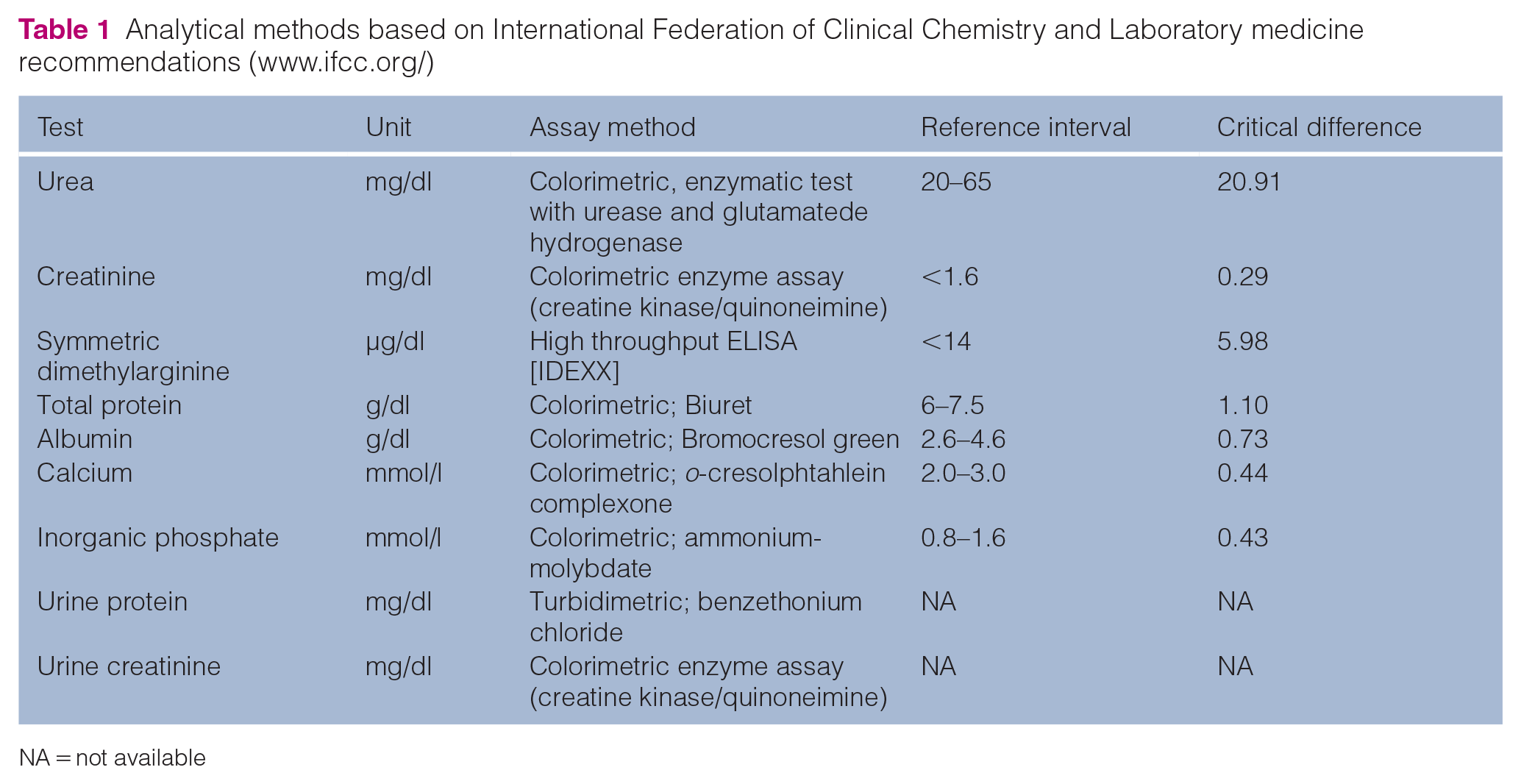
NA = not available
Urinalysis
Urine was taken via ultrasound-guided cystocentesis into uncoated tubes. The urine was processed within 30 mins of collection and the following biomarkers were determined: urine protein:creatinine ratio (UPC), urine osmolarity and urine specific gravity (USG). Urine chemistry tests were performed on the autoanalyser for clinical chemistry (Cobas 501c; Roche Diagnostics) with urine settings and the methods are summarised in Table 1. Osmolarity was measured with the freezing point osmometer described above. USG was measured with a refractometer (Reichert Vet 360; VWR) using the calibration scale for cats.
Time points
Sampling of blood and urine was performed on day 0 (day of contrast media administration) before or after injection of drugs for premedication (base level) and at three selected time points (days 1, 7, 21) after contrast media administration (Figure 2).
Statistics and data evaluation
Statistical analyses were conducted with the commercial software program SPSS (IBM Österreich Internationale Büromaschinen) by means of the Bioinformatics and Biostatistics Platform of Vetmeduni Vienna. The normal distribution of the data was tested with the Kolmogorov–Smirnov test. For the data analysis, descriptive statistical and mixed models with time points and group membership as principal factors were used. Data were reported as mean with both SD and range (minimum and maximum). The significance was set with a P value of 0.05.
Additionally, the data were observed for changes in laboratory results over time (days 0, 1, 7, 21) using absolute numbers and CD values for each individual patient.
Results
The study took place at the Diagnostic Imaging, Department of Small Animals and Horses of the University of Veterinary Medicine, Vienna, from May 2019 to June 2020, with a 1-month pause (April 2020) owing to the COVID-19 pandemic. Included in this study were 52 cats with a sex distribution of 23 male (one intact, 22 neutered) and 29 female (three intact, 26 neutered) individuals. The age distribution ranged from 3 months to 20 years, with a mean age of 9.2 years and a median age of 9.7 years. Among the cats were 44 domestic shorthair cats, three Maine Coons, two British Shorthairs, one Persian, one Ragdoll mix and one Chartreux-Maine Coon mix. The mean weight of the cats was 4.28 kg and the mean body condition score was 5/9.
The most common final diagnoses for the individuals presented to the Diagnostic Imaging Department were as follows: neoplasia (19 cases, 16 confirmed by histopathology) and otitis (15 cases) with comorbidities such as polyps (3), aural neoplasia (3), polyps and aural neoplasia (2), rhinitis (2) or nasopharyngeal stenosis (1). Further final diagnoses included myelopathy (4), rhinitis (3), arthritis (2), idiopathic epilepsy (2), urogenital malformation (ectopic ureter, enlarged bladder neck/urethra) (2), feline infectious peritonitis (1), encephalitis (1), behavioural automutilation (1) and no final diagnosis (2).
The cats were distributed into groups as follows: group ioversol (iov), 27 cats (14 females, 13 males); group gadoteric acid (gad), 12 cats (eight females, four males); group ioversol and gadoteric acid (iov + gad), four cats (three females, one male); and the control group (ctr), nine cats (five males, four females). For 9/52 cats, no follow-up data were available.
No cat was excluded based on ultrasonographic findings or blood pressure measurements.
Before the contrast medium was applied, 17 of the cats had increased plasma creatinine concentration (>1.60 mg/dl), between 1.60 and 3.20 mg/dl, and 35 had values within the reference interval (WRI) on day 0 including the nine cats with no follow-up data available. Three cats (numbers 29, 35 and 52) had known mild to moderate renal dysfunction prior to the study. Two cats (29 and 35) were stage 2 based on the International Renal Interest Society (IRIS) staging for CKD by their referring veterinarian; on day 0, they had creatinine results of 2.70 mg/dl and 1.90 mg/dl, respectively. They had no recent clinical signs reported by the owner and received ioversol (group iov). The third cat (52) was IRIS stage 3 with creatinine results of 2.90 mg/dl, USG of 1017 and no proteinuria (UPC = 0.18) on day 0. The owner reported only weight loss during the past 2 months. This cat did not receive any contrast media (group ctr).
Four cats had a CT/MRI procedure within 1 year before the actual examination and one cat had CT performed within 3 years.
Time points
Figure 2 shows the mean, median, maximum and minimum days after contrast media administration for the optimal sampling time points on days 0, 1, 7 and 21. Day 0 took place on the day of contrast administration before contrast media was administered to each cat. Day 1 was the first consultation after contrast media administration but varied from 24 to 120 h (Figure 2).
Blood analysis
In Table 2, mean values for each group (ctr, iov, gad, iov+gad) at each appointment (days 0, 1, 7, 21) have been calculated for creatinine, urea, SDMA, haematocrit, UPC and USG. An outlier was deleted for the statistical analyses (cat 37, SDMA results, day 7; see Table 3). No statistically significant differences (P value <0.03) were observed with the mixed model concerning both group and date as key factors. Mean CD values for each group between day 0 and days 1, 7 and 21 were calculated for creatinine, urea and SDMA as well. The CDs revealed no significant correlation with time and group as principal factors (Table 2).
Elected mean blood and urinalysis results and calculations of critical differences (CDs) for each group (iov, gad, iov+gad, ctr) and each sampling day (days 0, 1, 7 and 21)

Out of reference interval†No statistically significant differences or correlations (P <0.03) were observed for the mixed modeln = number of cats; ctr = control; gad = gadoteric acid; iov = ioversol; iov+gad = ioversol and gadoteric acid; UPC = urine protein:creatinine ratio; SDMA = symmetric dimethylarginine; NA = no data available; USG = urine specific gravity; CM = contrast media
Blood biomarkers for each individual regarding creatinine, symmetric dimethylarginine (SDMA) and the calculated critical differences (CDs) of creatinine and SDMA showing at least one increased value at any sampling day (days 0, 1, 7, 21)
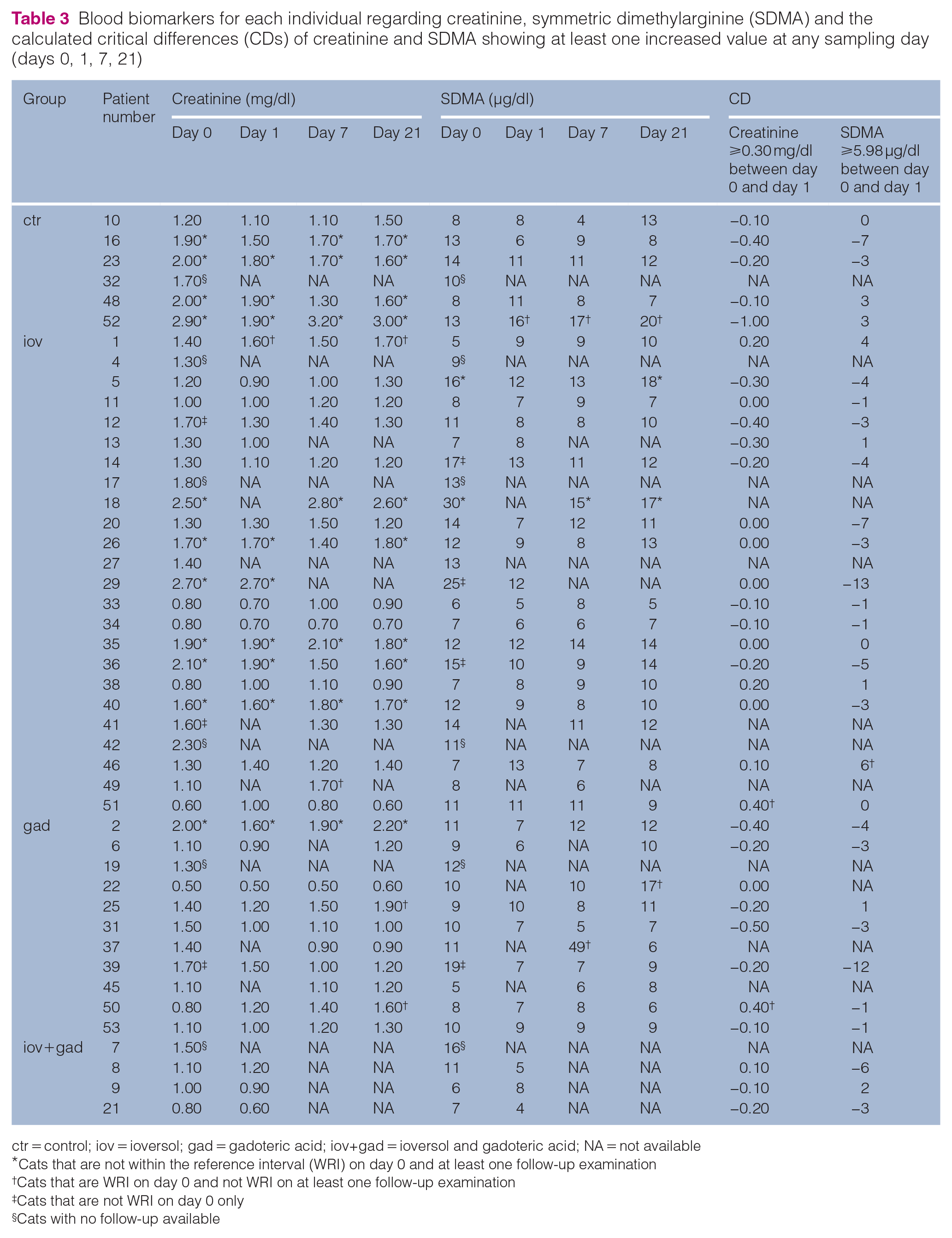
ctr = control; iov = ioversol; gad = gadoteric acid; iov+gad = ioversol and gadoteric acid; NA = not available *Cats that are not within the reference interval (WRI) on day 0 and at least one follow-up examination †Cats that are WRI on day 0 and not WRI on at least one follow-up examination ‡Cats that are not WRI on day 0 only§Cats with no follow-up available
The mean absolute creatinine value was 1.74 mg/dl for the ctr group on day 0 (Table 2, marked with an asterisk), which exceeded the reference interval, whereas the mean laboratory results from the other three groups were WRI (Table 2). In the follow-up tests, the mean creatinine results of the ctr group were WRI for days 1 and 7, but they exceeded the reference interval by about 3.8% on day 21 (Table 2, marked with an asterisk), with 1.65 mg/dl. The mean results for creatinine in the groups receiving the contrast media (iov, gad, iov+gad) were WRI for all appointments and declined by about 4.1% to 18.0% from day 0 to days 1 and 7.
The mean CD values for creatinine were WRI for all evaluated time periods (days 0−1, days 1−7, days 7−21) and revealed no statistically significant correlation among the different groups (Table 2).
Table 3 shows the individual absolute laboratory results for all cats that were not WRI at any sampling date for one of the following laboratory values: creatinine, SDMA and CD calculations of creatinine and SDMA. The UPC and USG results of these cats are shown in Table 4. Of the whole population, 44/53 cats received follow-up examinations, which enabled comparison over the study timeline. Therefore, the subpopulation of 44 patients in this sample corresponds to 100% in the following.
Urinalysis for each individual regarding urine protein:creatinine ratio (UPC) and urine specific gravity (USG) for the sampling days on days 0, 1, 7 and 21

gad = gadoteric acid; iov = ioversol; iov+gad = ioversol and gadoteric acid; ctr = control; NA = no data available *Cats that are not within the reference interval (WRI) on day 0 and at least one follow-up examination †Cats that are WRI on day 0 and not WRI on at least one follow-up examination ‡Cats that are not WRI on day 0 only§Cats with no follow-up available
For both cats 50 and 51, the increase in creatinine was 0.4 mg/dl. Thus, the CD for creatinine was outranked, fulfilling the criteria for suspected PC-AKI despite the fact that both cats were WRI with 0.8 mg/dl and 0.6 mg/dl, respectively. The creatinine concentration of cat 50 doubled to 1.6 mg/dl on day 21, whereas the values of cat 51 remained WRI. Cat 50 suffered from progressive paresis and micturition problems, so a post-renal cause for the azotaemia could not be ruled out. MRI indicated generalised myositis, which was confirmed by a marked increase in creatinine kinase activity. Cat 51 was 3 months old, so the reduced muscle mass contributed to the low baseline creatinine concentration. The cat had urinary incontinence and was not only suspected to have neuromuscular disease but CT indicated malformation of the ureteral opening into the bladder. Cystitis and urethritis completed the clinical picture, so post-renal causes for the increase in creatinine could not be reliably ruled out.
On day 0, 14/44 cats were azotaemic and 11/14 (six iov, one gad, four ctr) maintained increased creatinine concentrations (⩾1.6 mg/dl) on days 1, 7 and/or 21 and 3/14 cats (two iov, one gad) were WRI after contrast administration. Twenty-eight out of forty-four cats were WRI on day 0 and 4/28 cases (two iov, two gad) showed azotaemia after contrast administration with creatinine results ranging between 1.60 mg/dl and 1.90 mg/dl.
The mean absolute group results for SDMA did not exceed the reference interval at any sampling date in any group; mean results of SDMA ranged from 5.67 μg/dl to 11.59 µg/dl. The mean group results for the calculated CDs of SDMA were WRI and negative on each date and in each group (Table 2).
For the time period day 0 to day 1, a single cat (patient number 46) exceeded the reference interval with a CD value of 6 µg/dl (Table 3). The SDMA and creatinine results were WRI for each sampling date and returned from 13 μg/dl on day 1 back to baseline at 7 μg/dl on day 7.
A total of 6/44 cats with follow-up examinations had an increased SDMA (>14 µg/dl) concentration between 16 µg/dl and 30 µg/dl before the contrast media was applied. Two of six cats maintained increased SDMA values for at least one further appointment (two iov) and 4/6 (three iov, one gad) became WRI. For 38/44 cats with follow-up examinations, the SDMA results were WRI, with 3/38 cats showing an increase in SDMA (two gad, one ctr) on at least one additional date.
All mean group values for urea and haematocrit were WRI on each sampling day (Table 2).
Urinalysis
The results of the mean UPC calculation were low grade increased (⩾0.20) for groups ctr (UPC = 0.26), gad (UPC = 0.27) and iov+gad (UPC = 0.21) before the contrast medium was applied on day 0 (Table 2). The group iov+gad increased to a UPC of 0.29 on day 1. During the course of the follow-up examinations, the mean group UPC for ctr decreased to the reference interval and for gad decreased to the reference interval on days 1 and 21 and to a value of 0.20 on day 7. For group iov, the results of the mean UPC calculation were WRI on days 0 and 1, increasing to a UPC of 0.21 on both days 7 and 21.
For 16/44 cases (one ctr, eight iov, four gad, three iov+gad) with available follow-up measurements the UPC results were increased on day 0, ranging between 0.20 and 1.33 (Table 4). For at least one additional appointment, 12/16 cats (one ctr, six iov, three gad, two iov+gad) had persistent increased UPC results. Three of the cats (UPC = 0.73, 0.42 and 0.50) with ongoing proteinuria exceeded a ratio of 0.4. After values WRI on day 0, 5/28 cats showed increased UPC (three iov, two gad) after contrast media was administered with two cases (two iov) exceeding a ratio of 0.4.
Consequently, 11 cats (25.0% of cats with available follow-up examinations) with proteinuria prior to contrast media administration showed persistent proteinuria afterwards. Another five cats (11.4% of cats with available follow-up examinations) developed proteinuria after the contrast media were administered.
For the urinalyses, the mean USG was WRI for every group at each appointment and statistically there were no significant differences among groups (Table 2). In comparison, on day 0, the values for group ctr (1021.50) were much lower than those of the groups iov (1036.27), gad (1031.92) and iov+gad (1027.75).
For the individual USG results, 12/44 cats (four ctr, three iov, four gad, one iov+gad) were not WRI on day 0. For at least two additional sampling dates, 9/12 cats maintained decreased USG results. Cat 50 had a decreased USG on days 0 and 7 of 1017 and 1018, respectively, while cat 51 had a decreased USG (1014) on day 0 but was WRI (1038, 1029, respectively) on days 1 and 21. After contrast medium was applied, 9/32 cats (six iov, one gad, two iov+gad) that had been WRI on day 0 developed a decreased USG on one additional sampling date (mainly day 1).
Discussion
In this study, no cases of PC-AKI were observed after administration of gadoteric acid and ioversol. However, two cats (4.6%, one iov, one gad) had increased CDs of creatinine (0.4 mg/dl) within 48 h post-contrast exceeding the reference change interval (⩾0.3 mg/dl); in both cases, a post-renal cause such as a neurogenic bladder dysfunction was likely based on history, diagnostic imaging results and treatment outcome. In these cats further diagnostic work-up revealed macroscopic post-renal reasons for the rise in plasma creatinine concentrations so that PC-AKI was no longer at the top of the list of differentials, despite that it could not be ruled out completely. In patients with previously known mild azotaemia, no further deterioration of kidney biomarkers was seen. Cats that had low-grade increased UPC before contrast media administration continued to show increased UPC results. Still, five patients (11.4%) without proteinuria before contrast media administration developed detectable proteinuria between day 0 and day 21.
The use of IV iodinated contrast media in human medicine has been intensively investigated.27,28 Recent studies in humans have revealed no to minimal risk in patients without renal compromise, which agrees with the results of our study.27–29 To the best of our knowledge, proteinuria after IV contrast media administration has not been previously described in cats.
Multiple studies have found a variable prevalence of PC-AKI in humans. 7 The intra-arterial administration of iodinated contrast media accompanying cardiac procedures in interventional human radiology, including its injection into the coronary artery during percutaneous coronary intervention, is related to a risk value of up to 3.3% for PC-AKI in humans. 30 The long-term outcome in patients with PC-AKI is therefore worsened, causing higher mortality and hospitalisation costs. 31 The overall incidence of PC-AKI in our examined cat population was small, probably because the amount of IV-administrated contrast media is smaller compared with that used in cardiovascular procedures.
Some reviews, including meta-analyses of large data sets over the past decade, emphasised that many of the studies that focused on PC-AKI lacked a proper design, did not use control groups and did not exclude biased patient populations.5,32
In human medicine, the preventive effect of pre- and post-procedure hydration of patients receiving iodinated, non-ionic low-osmolar contrast agents, especially in cases of cardiac procedures, has been shown.33,34 In contrast to patient management in human medicine, patients in veterinary medicine receive general anaesthesia including a proper fluid regime regularly in the course of diagnostic imaging. Thus, the cats in our study, even without known compromise in renal function, were well hydrated when the contrast media was administered. Furthermore, the blood analysis and hydration status of the patient were checked prior to anaesthesia and contrast media administration (Figure 1). Dehydration serves as a relative contraindication of contrast administration; therefore, the diagnostic imaging is postponed if possible, depending on the patient’s general health status, until the dehydration has been corrected. Both (re-)hydration and accurate assessment of patients’ suitability for anaesthesia potentially biased the results of our study due to their protective effects on renal function, but nevetheless emphasise the necessity of these steps before contrast media is administered. A recent study in healthy Beagle dogs examined the value of pre-anaesthetic blood analysis and described changes in the perioperative anaesthetic management in 79% of cases after reviewing the results. 35 The reason that the mean creatinine value of the control group was higher on day 0 than those of the other groups was probably the effect of pre-anaesthetic laboratory results revealing pre-renal or renal azotaemia leading the clinician, radiologist and anaesthesiologist in charge to reflect on the use of contrast media more critically. Subsequently, in patients with known CKD or those with until then undiagnosed azotaemia, the use of contrast media was more restrictive. This can be related to higher mean creatinine concentrations in the control group. The lower USG in this group can also be explained with some cases of CKD or suspected renal insufficiency.
The adequate fluid regime in patients showing azotaemia before contrast administration in our study has most likely compensated renal malfunction (since renal biomarkers were WRI). The positive effects of providing IV fluids along with contrast media have been proven in human medicine as well. 24 One prospective study in humans identified patients at high risk for development of PC-AKI and treated them 4 h before and after administering contrast media with IV fluids (isotonic saline) at a rate of 250 ml/h, with a special treatment for patients with heart failure or severe renal insufficiency.33 Having found a low incidence of PC-AKI in the patient group with stages 3 and 4 CKD, they concluded that a proper fluid regime has a positive effect. 33 A further study of patients with congestive heart failure and CKD undergoing coronary procedures compared the effects of administering isosorbide dinitrate to one group with adequate infusion therapy (adequate isotonic saline) with a control group that received only routine hydration (routine isotonic saline). There were fewer cases of PC-AKI and cumulative adverse reactions during the 90-day follow-up in the group that received the adequate infusion therapy. 25 Another human study yielded comparable results and additionally proposed no risk for nephrotoxicity in patients with renal insufficiency when proper rehydration is provided. 33 Knowing that there is a high prevalence of CKD in cat populations, IV fluid administration to maintain adequate hydration becomes an important procedural step in cats that are receiving a contrast medium during imaging procedures under general anaesthesia. 26 Two of the three cases of known mild CKD in our study belonged to the group iov because of the unavoidable need for contrast studies after earlier hydration therapy. The third patient did not receive contrast medium and thus was assigned to the control group. All three cases maintained azotaemia but did not deteriorate statistically significantly during the study period; however, further research is needed because the number of cases in our study was low.
The sampling date was on day 1 in order to have immediate control of creatinine. The literature on human medicine proposes a sampling time of 48–72 h after contrast administration.1,2 Strictly, daily sampling after IV contrast medium administration would have been ideal. However, owing to a lack of data regarding the optimal sampling time in cats, and for the purpose of patient owner cooperation as well as patient management and safety, we decided to take three samples over 3 weeks on days 1, 7 and 21. Interestingly, immediate proteinuria was seen between days 1 and 7, so further studies with sampling after 48 and 72 h are needed to explain the origin and the course of transient proteinuria.
During the prospective study, the sampling dates varied slightly between patients depending on owner compliance, although we tried to maintain the same time periods between sampling for all cats to the extent possible. In some cases, the owner was simply unable to bring their cats back at the optimal sampling time point; in other cases, the study dates were customised to the patients’ treatment or control visit to the hospital. Nevertheless, most of the owners brought their cats at the appointed times. Thus, another important aspect in evaluating the biomarkers is the sampling time point, as it relates to possible cyclic rhythms of the selected biomarker. In humans, the level of creatinine is nearly constant during the day because the high tubular creatinine secretion in the circadian rhythm is normalised by the opposing glomerular filtration rate rhythm, whereas in dogs and cats, a circadian rhythm with peak levels in the afternoon has been described.36–38 If it was feasible, study appointments 1, 2 and 3 were undertaken at the same time of day as the day 0 control appointment. Most of the appointments took place in the morning or at noon, which is why a data shift due to circadian rhythm is not probable.
Several studies in human medicine revealed no contraindication of gadoteric acid for use in patients with CKD. In one study, pharmacokinetics and tolerance were tested in a healthy control group and in eight individuals with non-dialysed CKD. Patients who received gadoteric acid experienced a delayed but nearly complete urinary elimination correlating with the plasma creatinine clearance. 39 A further study evaluated serum creatinine and GFR in 20 patients receiving gadoteric acid compared with 10 people in the control group with measurements at two time points after contrast media administration. 40 In cats, evaluating the GFR using an estimate (eGFR), comparable to the Cockcroft–Gault formula in humans, is not recommended because no reliable calculations have been derived. 41 Therefore, the eGFR was not calculated for this study. The best modality to access the kidney function would have been an estimate of the GFR or excretion fraction of some marker. Ethical restrictions related to extra radiation exposure without a clinical indication in the patient’s diagnostic work-up would have been disproportionate, and it would have been difficult to convince the pet owner to allow further examination.
Furthermore, the anaesthesia itself as a reason for AKI is well described in humans, but in veterinary medicine little information exists about this relation. A recent study of dogs showed 12 cases with azotaemia after receiving general anaesthesia. 42 In our study population no statistically significant effect on creatinine concentration was detectable.
Baral et al studied several feline biochemistry values with highly individual laboratory values including creatinine, urea and total protein. The evaluation of the CDs enabled the calculation of individual changes in creatinine results to a basic level for each animal. 43 A later study defined the CD for SDMA in dogs, which we used for our feline population in the absence of cat-specific data.44,45 A very recent study documents small changes in SDMA concentrations that require an analyser-specific reference interval. 46 We used the individual references inter alia for the creatinine and SDMA, but no changes in SDMA between day 0 and days 1, 7 and 21 were found.
There were five cats that received ioversol and gadoteric acid and showed proteinuria (UPC >0.4) or borderline proteinuria (UPC = 0.2–0.4) after the contrast medium was applied, but they were WRI before administration. The levels of proteinuria were not high grade and thus may have been the result of tubular damage or a renal parenchymal inflammation if post-renal causes such as urinary tract disease and pre-renal diseases that increased the filtered proteins, such as Bence Jones proteins in neoplasia, could be excluded. 47 One of the five cats with proteinuria had a neoplasia, one had a urogenital malformation (the ectopic ureters were not seen with ultrasound) and the third had feline infectious peritonitis, which may have led to aberrant urinalyses. Even a glomerular origin of the proteinuria could not be excluded; for example, a transient, undetected hypertension after contrast media administration could induce the pathophysiological process. 47 Another possible reason for proteinuria may have been a physiological stress-induced transient proteinuria (which has been described in dogs) that was likely due to the change of conditions in the hospital and after transportation of the cats in our study to each appointment. 48 Defects in the tubular reabsorption of mainly low molecular weight proteins serve as another reason for renal proteinuria. A suspected damage of the renal tubular cells caused by the contrast agent would lead to a loss of proteins in this segment of the nephron. Thus, an already existing protein-losing nephropathy could worsen or even damage further parts of the nephron.49–51 Furthermore, measuring the UPC may underestimate proteinuria because the test has a high specificity of 99.02%, but only a low sensitivity of 2.0%, as mentioned by Lyon et al. 52 It is important to classify the origin of the proteinuria because human studies showed the negative impact of proteinuria on the glomerular function in addition to the negative prognostic value of an ongoing proteinuria known in cats.53,54 For determining the origin of a renal proteinuria a urinary protein electrophoresis, for example, a sodium dodecyl sulfate-agarose gel electrophoresis (SDS-PAGE), can aid in clarification. Glomerular damage results in a loss of high molecular weight proteins, while low molecular weight proteins dominate in case of tubulointerstitial defects.55,56
The value of biomarkers detecting AKI is limited. A study comparing the value of measuring GFR and serum creatinine levels in azotaemic and non-azotaemic cats demonstrated that GFR is better for monitoring non-azotaemic cats, while serum creatinine is recommended for azotaemic cats. 57 In this study, GFR could have been the best test to measure level of kidney function and determine the stage of kidney disease in non-azotaemic cats. However, hazards to add GFR measurements to our protocol were prolongation of the study design, increased costs for patient management, higher ethical issue intensity and suspected bias of GFR from the administered iodinated contrast agents (iohexol). Benefits of serial serum creatinine measurements were specific and individual baseline levels that we could individually relate further measurements to. For further and more detailed examination of kidney function, the biomarker neutrophil gelatinase-associated lipocalin (NGAL) in urine has been well described in humans. 58 Interestingly, a study of 71 children with cardiac catheterisation and concomitant ioversol administration resulted in an earlier elevation of urinary and plasma NGAL (2 h) than creatinine. 59 In dogs, urinary NGAL seemed to indicate renal impairment earlier than NGAL in blood and creatinine and represented a sensitive biomarker. 60 In cats, more complex forms of urinary NGAL have been described and presented promising results in detecting of AKI, CKD, urinary tract infections and CKD progression. 61
Furthermore, Fowlie et al reported that in healthy dogs receiving contrast media for CT angiography, heterogeneous changes in urinalysis were visible, but osteopontin and clusterin increased significantly over time while the kidney injury molecule, KIM-1, decreased. 62 These changes may be related to the transient proteinuria that we observed in several cases. A further evaluation of these biomarkers may be helpful.
The population of cats in this study may have been biased owing to several effects. Cats with severe diseases hospitalised in the intensive care unit were not included in this study owing to ethical reasons; however, cats that were suspected by the anaesthesiologist to be inadequately hydrated did not receive either anaesthesia or contrast media without previous rehydration. When the examination was not urgent, the appointment was postponed, which may have led to a population of well-hydrated cats that did not meet the conditions to be seen in a private hospital, even if they may have been good candidates for this study. Cats with a special risk for anaesthesia or administration of contrast media were excluded from the study in advance, which probably biased the cat population as a group of lower risk patients. Cats with moderate to severe elevation of renal biomarkers and unstable patients were excluded from contrast media administration or even anaesthesia itself. However, because our setting mirrored real clinical settings, there may not have been a true bias in the study population.
Conclusions
The administration of the non-ionic iodinated contrast agent, ioversol, or the paramagnetic agent, gadoteric acid, did not result in a significant elevation of the tested kidney biomarkers in blood and urine examinations in our sample. However, additional studies are needed to investigate the relevance of the mild proteinuria in 11.4% (five cats) of patients. This conclusion indicates that cats with prior considerably elevated renal biomarkers or marked dehydration should be excluded from anaesthesia or contrast media administration until they have received effective rehydration therapy. The administration of IV fluids is an important condition to keep the patient well hydrated and, consequently, keep renal perfusion stable. Further studies are needed to sustain our findings and examine the effects of both contrast media with one type of anaesthesia.
Footnotes
Acknowledgements
The authors acknowledge the work of the laboratory staff and technicians for providing support. Further, we would like to thank Dr Tichy, Bioinformatics and Biostatistics Platform, Vetmeduni Vienna, for statistical support and the graphic artist Dr Eva Polsterer for her contributions.
Author note
Part of this study was presented in the form of an abstract at the ACVR Annual Meeting, 19–23 October 2020, New Orleans, LA, USA.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee, while not specifically required for publication in JFMS, was nonetheless obtained, as stated in the manuscript.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
