Abstract
Objectives
This pilot study sought to assess the metabolism of Plavix (Bristol-Myers Squibb/Sanofi) and generic clopidogrel in cats, using a novel assay for the measurement of clopidogrel, clopidogrel carboxylic acid (CCA) and clopidogrel active metabolite (CAM-D).
Methods
This was a prospective, randomized, double-blind study. Four healthy, skeletally mature cats were enrolled into the study. There were two treatment phases during which cats received either Plavix or generic clopidogrel at a dosage of 18.75 mg PO q24h for 7 days with a 2 week washout between phases. During each phase, plasma concentrations of parent drug and active and inactive metabolites were measured along with impedance platelet aggregometry in response to adenosine diphosphate (ADP).
Results
The ratio of CAM-D between generic clopidogrel and Plavix was 0.83 (equivalence reference 1.00, 90% confidence interval 0.80–1.25). Inhibition of ADP-induced platelet aggregation was variable, with two cats classified as non-responders in both treatment phases. The concentrations of CAM-D were not predictive of aggregometry-based responsiveness to either formulation of clopidogrel.
Conclusions and relevance
This is the first study comparing Plavix and generic clopidogrel in cats. Administration of the generic formulation resulted in comparable plasma concentrations of clopidogrel active metabolite when compared with Plavix.
Introduction
Arterial thromboembolism (ATE) is a well-documented and severe sequela of feline cardiomyopathy. Arterial thromboembolism, along with congestive heart failure, are the most common causes of cardiac death in cats with hypertrophic cardiomyopathy (HCM).1–5 Additional risk factors for cardiac death from ATE include previous thromboembolic events, reduced fractional shortening of the left atrium and increased left atrial size.1,2 Enlargement of the left atrium results in stretching of the endothelium, exposing the collagen and initiating platelet activation and adhesion,6–10 leading to decreased blood flow and blood stasis further promoting ATE formation. 11 Additionally, approximately 40% of cats with HCM showed platelet hyperaggregability in vitro. 12 Up to 17% of cats with underlying cardiac disease will develop cardiogenic ATE,1,13 with recurrence rates of 17–75%.14,15 Mortality in these cats has been reported to be 61–100%.11,14–18
Antithrombotic medications are considered standard treatment in cats for prevention of cardiogenic ATE. Thromboprophylaxis protocols have included aspirin, clopidogrel, warfarin, unfractionated and low-molecular weight heparin, and rivaroxaban.14,15,18,19 The only prospective clinical trial evaluating thromboprophylaxis in cats demonstrated that clopidogrel was associated with a lower rate of ATE recurrence and a longer median time to recurrence of ATE than aspirin. 8 There is substantial inter-individual variability in the platelet inhibitory effect of clopidogrel in people and this is associated with an increased risk of recurrent thrombotic events and major cardiac events. 20 Potential causes for this variable clopidogrel response include differential absorption and/or metabolism of the drug.
Clopidogrel is a second-generation thienopyridine that irreversibly inhibits the binding of adenosine diphosphate (ADP) to the P2Y12 receptor on the platelet surface. This blockade results in inhibition of primary and secondary platelet aggregation, as well as inhibiting the ADP-induced conformational change of the glycoprotein IIb/IIIa receptor complex.18,21,22 Clopidogrel is an inactive prodrug that requires activation by hepatic cytochrome P450 (CYP) enzymes. In people, the parent molecule is metabolized to inactive clopidogrel carboxylic acid (CCA), of which approximately 15% is hydrolyzed to the clopidogrel active metabolite (CAM).
Clopidogrel is available in trade (Plavix; Bristol-Myers Squibb/Sanofi) and generic (clopidogrel bisulfate; Dr Reddy’s Laboratories) formulations. Bioequivalence is verified in humans, while pharmacodynamic variability exists with both formulations.23,24 Polymorphisms in the gene encoding for the enzymes CYP2C19 and CYP3A4, and the P2Y12 receptor, have been identified in people and are associated with decreased bioconversion to CAM and reduced platelet inhibition.25–27 Early studies on clopidogrel in cats did not show a similar variability in platelet inhibitory effects; however, a recent study reported that 15% of cats on clopidogrel were classified as non-responders, although the cause for this variability was not reported. 25
The pharmacokinetic and pharmacodynamic differences between Plavix and generic clopidogrel have not been investigated in cats. The quantification of CAM has not been widely investigated as it is unstable in plasma and difficult to measure. A method has recently been validated in cats. It stabilizes CAM by adding 2-bromo-3′methoxyacetophenone (BMAP) to blood tubes. 3 Once stabilized, the derivatized CAM (CAM-D) can be quantitated using high-performance liquid chromatography with tandem mass spectrometry (HPLC-MS/MS). 3
The primary aim of this study was to quantify the clopidogrel parent molecule along with the CCA and CAM-D metabolites in cats and determine if there were any significant differences between Plavix and generic clopidogrel. A secondary aim was to assess the correlation between CAM-D concentration platelet inhibition as measured by platelet aggregation.
Materials and methods
Cats
Skeletally mature cats were recruited with informed consent from the faculty and staff of the veterinary teaching hospital. Health status was confirmed through collection of a detailed history, physical examination, complete blood count, serum chemistry panel, prothrombin time, partial thromboplastin time, retroviral testing and urinalysis. These cats had no history of a disease process that could affect coagulation, and no history of bleeding diatheses or surgical complications. These cats were not receiving any medications other than monthly preventives at the time of enrollment. This study protocol was approved by the Institutional Animal Care and Use Committee (Purdue Animal Care and Use Committee Coeus Protocol Number 1601001366).
Experimental protocol
This randomized (QuickCalcs; www.graphpad.com), double-blind study was composed of two treatment phases in which the cats were administered 18.75 mg Plavix or generic clopidogrel q24h for 7 days. Commercially available Plavix or generic clopidogrel tablets (75 mg) were quartered using a tablet splitter, and each quarter tablet was then weighed to ensure similar dosing. The quarter tablet was then placed into a cellulose capsule to ensure blinding. The quartering and packaging of the clopidogrel formulations was performed by the same pharmacist. There was a washout period of at least 2 weeks between treatment phases.
Two hours after the first administration of clopidogrel in each treatment phase, 1 ml blood was collected for measurement of clopidogrel parent drug and the CCA and CAM-D metabolites. Blood samples were collected atraumatically from the jugular vein using a 23 G 3/4” butterfly needle with a 19 mm tube attached to a syringe (Covidien Monoject). Following collection, the blood was immediately put into a cryogenic tube (Thermo Fisher Scientific) containing 10 µg of a 500 mM BMAP (Sigma-Aldrich) solution and 20 µl of a 500 mM EDTA (Fisher BioReagents) solution. This collection method and timing was validated for quantitation of the clopidogrel active and inactive metabolites in feline plasma. 3 Once collected, blood was centrifuged (1500 g at 4°C for 15 mins) and the plasma harvested and stored at −80°C until shipment on wet ice to the laboratory (Washington State University College of Veterinary Medicine, Pullman, WA, USA) for analysis.
Immediately prior to the first dose (day 1) and 24 h after the final dose (day 8), blood was collected to measure platelet count and ADP-induced whole blood platelet aggregation. Venipuncture was performed using the same technique as described above, except that 4 ml blood was collected directly into a plastic EDTA tube (2 ml; BD Vacutainer) and lithium heparin tube (2 ml; BD Vacutainer) for platelet count and ADP-induced platelet aggregation, respectively. Platelet counts were determined using a hematologic analyzer (Abbott Cell Dyn 3700; Abbott Laboratories) and confirmed by microscopic examination of a blood smear as part of a routine hematologic analysis.
Measurement of clopidogrel parent drug, active and inactive metabolites
Plasma samples for quantification of clopidogrel parent drug, CAM-D and CCA were analyzed at Washington State University through the Program in Individualized Medicine. Samples were processed with HPLC-MS/MS using the previously described methodology. 3 Plasma samples from each cat were analyzed three times on three different dates. Plasma concentrations of clopidogrel, CAM-D and CCA from the three different runs were averaged, and the SD and coefficient of variation were calculated.
Determination of platelet aggregation
As described above, blood was collected directly into lithium heparin tubes. The blood was then allowed to sit at room temperature for 30 mins before being analyzed using an impedance aggregometer (Multiplate; Dynabyte). Briefly, 300 µl preheated isotonic sodium chloride and 300 µl whole blood were added to the aggregometer cuvette and incubated at 37°C for 3 mins while being mixed with a Teflon-coated magnetic stir bar at 800 rpm. Platelet aggregation was induced by the addition of ADP (ADP reagent; Verum Diagnostica) at a concentration of 6.5 µM. Platelet aggregation was measured by documenting the change in electrical resistance over 15 mins, and expressed in aggregation units (AU). These are provided as area under the curve (AUC) values, velocity (AU/min) and maximum aggregation (AU).
Statistical analysis
Normality of distribution for continuous variables was assessed by the Shapiro–Wilk test.
Metabolic ratio was calculated using the below equation with all units converted to mol/l to account for differences in metabolic weight.
As distributions for CAM, metabolic ratio and platelet aggregation velocity did not violate the assumption of normality, data between cats were compared using Student’s t-test and change (day 1 to day 8) within cats was compared using the paired t-test. Assessment of predicting response to clopidogrel was made by logistic regression. Statistical significance was set at a P value of <0.05. Owing to sample size, summary statistics are reported as mean ± SD. Statistical analyses were conducted using commercially available software (StataCorp).
Results
Five cats were enrolled in the study. One of the five cats was unable to complete both treatment phases so was excluded. Demographic and clopidogrel dose information for each cat is presented in Table 1. Mean baseline platelet count was 299 ± 6 × 103/µl and 347 ± 25 × 103/ µl (reference interval 200–500 × 103/µl) for Plavix and generic clopidogrel, respectively. Mean platelet count at day 8 was 447 ± 111 × 103/µl and 391 ± 10 × 103/µl for Plavix and generic clopidogrel, respectively. The platelet counts at baseline were not significantly different when compared with day 8 for either Plavix (P = 0.336) or generic clopidogrel (P = 0.072).
Demographics and clopidogrel dose by body weight for the four, healthy, skeletally mature cats enrolled in the study
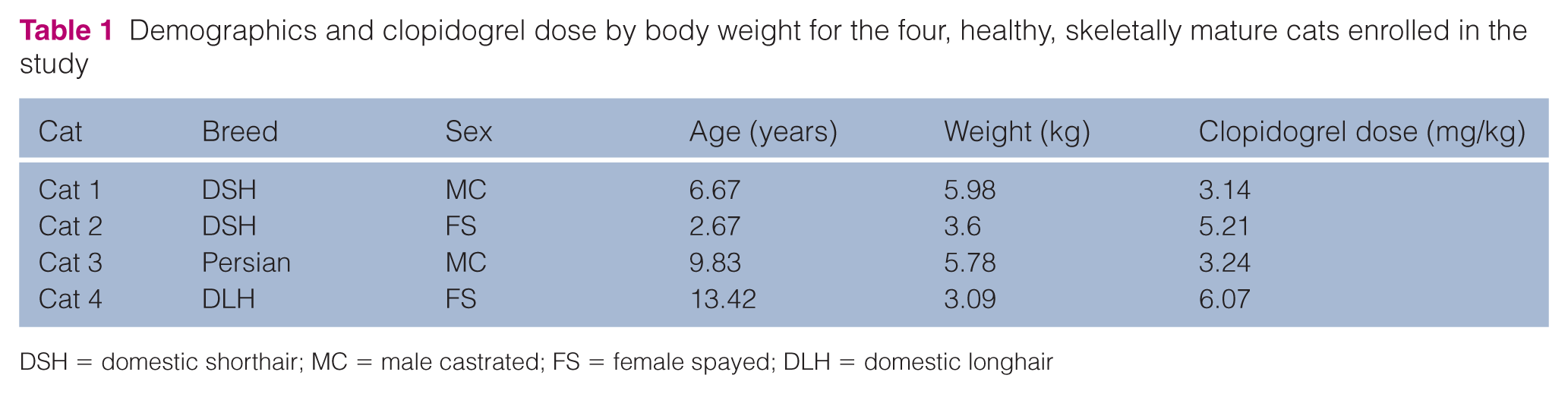
DSH = domestic shorthair; MC = male castrated; FS = female spayed; DLH = domestic longhair
Measurement of clopidogrel parent drug, CAM and CCA
Clopidogrel, CAM-D and CCA were detected in all samples. The concentration of the parent drug was significantly higher when the trade name was administered when compared with the generic drug (P = 0.041), but there was no significant difference between the concentrations of CAM-D (P = 0.230) or CCA (P = 0.367) from the two formulations of clopidogrel (Table 2). The generic:Plavix CAM-D concentration ratio was 0.83 (equivalence reference = 1.0; 90% confidence interval 0.80–1.25). 28
Plasma concentrations of clopidogrel parent drug, clopidogrel active metabolite (CAM-D) and the inactive clopidogrel carboxylic acid (CCA) in cats being treated with trade and generic formulations of clopidogrel

The coefficient of variation (CV%) is calculated based on the results of the assay run three times for each sample. A CV% <20% is considered acceptable
The metabolic ratio for the trade and generic formulations were not significantly different (P = 0.243; Table 3). Cats 2 and 4 demonstrated a higher metabolic ratio with the trade formulation than with the generic formulation, whereas the metabolic ratios for Cats 1 and 3 were more similar regardless of the formulation administered. Owing to the sample size, statistical comparison of these values was not possible.
Metabolic ratios depicting the proportion of clopidogrel active metabolite produced for each treatment phase (trade and generic) in each cat

The mean metabolic ratio for the trade formulation was not significantly different when compared with the mean metabolic ratio for the generic formulation (P = 0.243)
Measurement of platelet aggregation
The day 1 and day 8 platelet aggregometry data, including AUC, maximal aggregation and velocity of aggregation is depicted in Table 4. The velocity of aggregation in response to ADP at day 8 decreased significantly when compared with baseline when cats 1 and 3 are evaluated as a sub-group, regardless of the formulation of clopidogrel administered (P = 0.006; Figure 1). The velocity of aggregation in response to ADP at day 8 compared with baseline when cats 2 and 4 are evaluated as a subgroup did not significantly increase (P = 0.185; Figure 1). From this information, cats 1 and 3 were classified as responsive to clopidogrel, whereas cats 2 and 4 were classified as non-responders. The concentration of CAM-D and the metabolic indices were not predictive of being classified as responsive to clopidogrel (P = 0.260 and P = 0.765, respectively).
Results of whole blood impedance aggregometry using an adenosine diphosphate (ADP) agonist, including the velocity of aggregation, the maximal aggregation detected (Max agg) and the area under the curve (AUC) in cats being treated with trade and generic formulations of clopidogrel

In a previous study of healthy cats at this institution with the same impedance aggregometer and ADP agonist, the mean AUC was 255.8 ± 133.4 and the mean velocity was 15.3 ± 13.1 aggregation units (AU)/min 29

The velocity of aggregation in response to adenosine triphosphate (ADP) prior to and 24 h after completing each treatment phase. The treatment phases consisted of clopidogrel, either trade (solid line) or generic (dashed line), administered at a dosage of 18.75 mg PO q24h for 7 days. Cats 1 and 3 showed a significantly decreased velocity of aggregation in response to ADP at day 8 when compared with baseline, regardless of the formulation administered (P = 0.006). Cats 2 and 4 showed an increased velocity of aggregation in response to ADP at day 8 when compared with baseline. This increase was greater when the trade formulation was administered compared with the generic, but neither difference reached statistical significance.
Discussion
Clopidogrel has become standard treatment for cats with HCM and left atrial enlargement. This drug has greatly reduced the occurrence and recurrence of cardiogenic arterial thromboembolic events in HCM cats and was shown to be superior to low-dose aspirin in a clinical trial. 8 Previous research has demonstrated that platelet aggregation in response to ADP is significantly reduced in cats receiving 18.75 mg Plavix PO q24h and that this response can been seen after 7 days of Plavix administration.30–32 In 2009, the Food and Drug Administration (FDA) approved the generic formulation of clopidogrel. The generic formulation contains the same active salt as the trade-name product: clopidogrel bisulfate. The availability of a generic formulation offered a more cost-effective option for clients who may have previously been unable to afford trade-name clopidogrel. This allowed the standard treatment to be more accessible to clients. The FDA requires evaluation of bioequivalence when approving a generic medication. 28 Plavix and generic clopidogrel were found to be safe, well-tolerated and bioequivalent in people. 23 This is the first study to evaluate and compare both formulations of clopidogrel in cats.
Establishing bioequivalence between a generic and trade drug requires that outcome measurements, primarily pharmacokinetic characteristics, be compared between the two formulations. The ratio of each pharmacokinetic variable of the generic drug to the trade drug is computed with an ideal ratio of 1.00, indicating perfect bioequivalence. The FDA requires that the 90% confidence interval of the pharmacokinetic ratio should lie between 0.8 and 1.25. 28 In the present study, the sample size is too small, and the pharmacokinetic investigation was not comprehensive enough to complete a rigorous test of bioequivalence. Despite the small sample, the ratio of CAM-D of the generic clopidogrel to Plavix was 0.83, which would fall within the acceptable range for bioequivalence.
Despite bioequivalence in people, there is well-documented variability in the platelet inhibitory effects of clopidogrel. The metabolic processing of clopidogrel involves multiple enzymes and important G-protein-coupled receptors, and interindividual genetic variants within these metabolic steps have been shown to affect the antiplatelet function of clopidogrel. 4 Polymorphisms within the cytochrome P450 pathway have been recognized in different individuals, including alleles of CYP2C19 and CYP3A4.5,27 In addition, the ADP receptor (P2Y12) is important for inhibitory binding of CAM-D, and so polymorphisms in this receptor have led to reduced function of the drug. 33 Genetic variants of CYP2C19 are present in 4–25% of people.34–36 This variant of CYP2C19 decreases the conversion of clopidogrel to CAM, and therefore decreases platelet inhibition.34,35 The cytochrome P450 pathway has not been investigated for genetic variation in cats to date. Variability in responsiveness to clopidogrel was identified in Maine Coon cats homozygous for the A31P mutation in MYPBC3, with 2/13 cats classified as non-responsive to Plavix. 25 Beyond the validation, the present study is the first to measure the concentrations of CAM-D, as well as parent drug and CCA, in cats receiving clopidogrel. The present study also represents the first to compare the production of clopidogrel metabolites with Plavix and generic clopidogrel. Metabolic indices were calculated as a method to evaluate the proportion of clopidogrel that is being converted to CAM. Evaluation of the metabolic ratio, rather than absolute CAM concentrations, eliminates the compounding factor of bioavailability. When the cats were compared as groups, the metabolic ratio of Plavix was not different when compared with the generic product. However, when evaluated individually, two cats demonstrated similar metabolic ratios, indicating consistent CAM production, regardless of clopidogrel formulation administered, while the other two cats demonstrated a higher metabolic ratio when Plavix was administered as compared to generic clopidogrel. Interestingly, this difference in metabolic ratio was not predictive of inhibition of platelet function based on impedance aggregometry.
The prevalence of clopidogrel non-responsiveness in people is estimated at 21–26%, overall.27,34,36 This has contributed to approximately 9% of patients receiving clopidogrel having a major adverse cardiovascular event. 34 In addition to people, high individual variability in response to clopidogrel has been noted in horses. 37 Beyond this, there is limited information about clopidogrel resistance in animals. In a recent study, 15% of cats were classified as non-responsive to clopidogrel based on optical aggregometry. 25 In the present study, 2/4 cats did not demonstrate a reduced aggregation response to ADP based on impedance aggregometry and were classified as non-responsive to clopidogrel. While this might represent true variability of responsiveness to the drug, similar to what is seen in people, the method and reliability of measuring platelet function must be scrutinized.
In dogs, the effect of trade-name clopidogrel on ADP-induced platelet aggregation was assessed by three different methods and all were able to detect treatment effects.38,39 In cats, similar comparisons between methods of detecting platelet aggregations have revealed variable responses, making individual results difficult to interpret.40,41 Several studies have identified inconsistency in the measurement of platelet function when different analyzers of platelet aggregation are compared, with the Multiplate analyzer producing a less reliable AUC than PFA-100 closure time or Plateletworks percent aggregation.30,40,42 In the present study, significant baseline variability existed, especially with the AUC and maximal aggregation response. This variation most likely represents problems with sampling (eg, patient stress) or handling (venipuncture technique), which led to early activation and clumping of platelets. 43 In this study, 33% of the platelet counts collected demonstrated significant clumping. For impedance aggregometry, the velocity of aggregation has been shown to be the most sensitive variable to detect the effect of clopidogrel on platelets, 30 so for the present study the change in velocity of platelet aggregation was used to classify the cats as responsive/non-responsive to clopidogrel.
The consistency between formulations within individual cats, given the randomized and blinded nature of this study, might imply true interindividual variation in responsiveness to clopidogrel like that seen in people and horses.35,44,45 The Cmax of clopidogrel in people is expected to be around 20 ng/ml, and similar to previous studies, the plasma concentrations achieved in these cats was higher.3,45 This may be due to the higher dose administered per body weight, but also may indicate a difference in metabolic processing. Despite this, the plasma concentration of CAM-D in these cats was not predictive of functional trends. It is possible that factors beyond production of the active metabolite contribute to a variation in responsiveness. These factors may include bio-availability or variations in ligand-receptor interactions. A larger study in cats with a more comprehensive approach to platelet function assessment, to include multiple tests of platelet function, should be performed in order to evaluate the true variability in platelet responsiveness to clopidogrel.
Beyond the limitations in platelet function testing already discussed, this study was a pilot, and therefore the sample size was not large enough to support any firm conclusions about the bioequivalence of the two formulations of clopidogrel in cats. The low number of cats included in this study precluded meaningful statistical analysis. A larger, prospective study evaluating the clinical effect of both clopidogrel formulations, with the assessment of pharmacokinetic outcome variables, will be required to prove bioequivalence. Additionally, the formulations of clopidogrel should be scrutinized for the distribution of drug within the tablet, especially as quartering tablets of clopidogrel is common practice when administering to cats. There is no currently available information about the distribution of clopidogrel within its tablet, and therefore it cannot be guaranteed that the same dose is administered to each patient, even if tablets are weighed.
Conclusions
The results of the present study suggest that administration of Plavix and generic clopidogrel to cats leads to measurable and similar plasma concentrations of the clopidogrel active metabolite.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

