Abstract
Occurrence of venous thromboembolism in cancer patients (patients) undergoing chemotherapy is a remarkable concern for the oncologist. In addition, careful attention has to be paid to the possible major bleeding when patients carrying gastrointestinal cancer need antithrombotic therapies. To date some Cancer Associated Thrombosis (CAT) risk scores as Khorana and PROTECHT score have been developed to identify the cancer population at high-risk for venous thromboembolism (VTE). Consensus guidelines recommend to consider also low molecular weight heparin (LMWH) for primary thromboprophylaxis in high-risk patients. This is a report on a retrospective case series of 15 intra-luminal not surgically treated gastrointestinal cancer patients deemed high risk for VTE. The patients had a Khorana or PROTECHT score of 2 points or more (at least
Introduction
Since venous thromboembolism (VTE) is a common complication in cancer patients many efforts have been made to prevent and treat this potentially deadly event.1,2 Some risk-assessment models (RAMs) are specifically developed for cancer-related VTE such as the Khorana and PROTECHT score. 2 The Khorana score is based on the tumor type, white blood cell count, platelet count, hemoglobin concentration or use of erythropoietin stimulating agents, and body mass index (BMI). 2 The PROTECHT score is based on a modified Khorana score by adding the type of chemotherapy as a further risk factor. 2 No doubt, antithrombotic therapy poses a potentially increased risk for hemorrhage in intraluminal gastrointestinal patients (ILGC). The use of LMWH is allowed for both preventive and curative intervention in cancer-related VTE. 3 As to direct oral anticoagulants (DOACs), they might not be an optimal choice for high bleeding risk cancer patients with intact intraluminal gastrointestinal cancer or for those treated with concomitant inhibitors of CYP3A4 and/or P-gp.4,5 In accordance with the still in force instructions of the regional Health Authorities all patients admitted in hospital have to be assessed for venous thromboembolism risk and, if it is the case, given a thromboprophylaxis. The physicians of the Unità Operativa Complessa di Oncologia Medica of San Vincenzo Hospital complied with the guidance of their own institutionally based guidelines to assess the risk for VTE and the duration of primary thromboprophylaxis in cancer patients treated with chemotherapy. Accordingly patients were basically considered at high risk for VTE in accordance with the PROTECHT risk score. Plus, ipomobilty/immobility and inserted central venous catheter in metastatic patients are considered further risk factors. Therefore, a low-weight adjusted dose of LMWH was administered during and 48 days after chemotherapy. We describe our institutional experience to report short time clinical outcomes in patients suffering from ILGC and treated with a LMWH-based thromboprophylaxis regimen. Occurrence of gastrointestinal bleeding and VTE event were the primary and secondary study outcomes.
Case
Medical charts of patients suffering from intact intraluminal gastrointestinal cancer considered at high risk for venous thromboembolic event who uderwent first line chemotherapy and treated with low molecular weight heparin based thromboprophylaxis from November 2014 to February 2021 were retrospectively scrutinized. Our institution does not require ethical approval for reporting individual cases or case series. Written informed consent was obtained from the patients for their anonymized information to be published in this article according to the Regulation (EU) 2016/679. The patients had a Khorana or a PROTECHT score
Baseline patient characteristics.

Antithrombotic treatment.
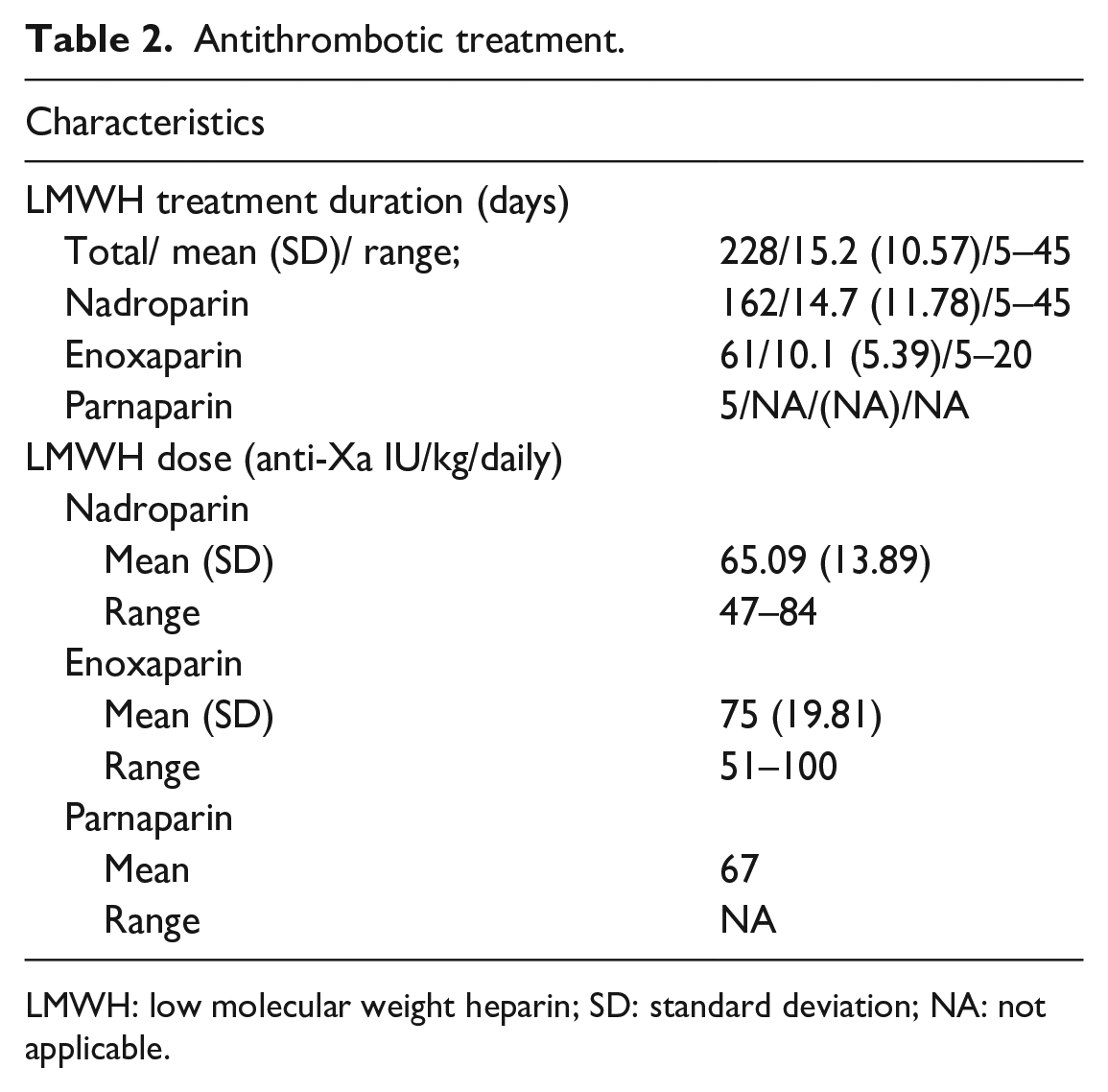
LMWH: low molecular weight heparin; SD: standard deviation; NA: not applicable.
Discussion
Undoubtedly the patients included in this study had characteristics which made them at high risk for VTE either for the histotype itself or chemotherapy regimen (five patients were administered cisplatin) and vulnerable to bleeding at once. As to RAMs, most of the leading experts in the field agree in setting high risk level at 2 or more in accordance with the Khorana score even if some author reported that the majority of VTE events occur in cancer patients with a lower score.6,7 Notably in our case series only chemonaive patients have been investigated. Therefore, they were much more likely to respond to a standard first-line chemotherapy. Fluoropyrimidine and platinum-containing treatment is standard-of-care first-line chemotherapy for advanced gastric, gastroesophageal junction (GEJ) and colon cancer.8–10 Response to treatment usually leads to a reduction in the extension of the neoplasm also in the intestinal lumen and as a results to a reduction in the risk of bleeding either spontaneously or induced by any anticoagulant treatment. DOACs often meet both patient’s and physician’s preference when antithrombotic therapy is required because there is no need of daily subcutaneous injection. Unfortunately and according to relevant clinical trials DOACs are not a safer alternative to LMWH when given to patients with gastrointestinal cancer.11–13 With regard to low molecular weight heparins, they represent a well-established treatment tool in cancer associated thrombosis (CAT). 11 It is intuitive that regardless of what antithrombotic drug is chosen to tackle thromboembolism, the longer it is administered the higher the risk of hemorrhage might be in patients with ILGC and mostly in patients not responsive to anticancer treatments. Still the optimal duration of thromboprophylaxis remains to be well established. 14 Of course, the study has a too short follow-up period, and this clearly is a limitation in order to draw fully acceptable conclusions.
Conclusion
Despite the authors report their experience on a small number of patients some considerations can derive from it. Patients with well-selected intra-luminal gastrointestinal cancer may undergo or started on short-term thromboprophylaxis. An accurate evaluation of the endoscopic cancer feature is mandatory. Of course LMWHs still have a prominent role in prevention and treatment of CAT and should continue to be standard of care for many patients with ILGC.
Footnotes
Acknowledgements
We thank Professor Mario Leonardi for his guidance through statistical analysis process.
Author contributions
Contributors Felice Vito Vitale, Valentina Giaimo and Mario Raffaele are responsible for the conception of the work. All the authors are responsible for the acquisition, analysis and interpretation of data. Alessandro D’Angelo, Fabio D’Amore, Alessia Dottore, and Paolo Colina have drafted the work. All the authors revised the manuscript critically and approved the final version.
Authorship
The seven authors have given substantial contributions to the conception and design of the work; to the acquisition, analysis, and interpretation of data for the work. In addition they drafted the work, revised it critically for important intellectual content and final approval of the version to be published. Moreover the authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
