Abstract
Objectives
The aim of this study was to assess the performance of a patient-side blood test in determining neuter status in female cats.
Methods
Residual blood samples from female cats of unknown neuter status that were admitted to four cat adoption centres in the UK were tested for luteinising hormone (LH) using the Witness LH test (Zoetis). A positive LH test result indicated that the cat was neutered. Cats were assessed for evidence of a surgical scar suggestive of prior neutering; if none was found, an exploratory laparotomy was performed to confirm neuter status. The LH test performance was assessed (sensitivity, specificity, negative and positive predictive value).
Results
Two hundred and thirty-six cats had both LH test and exploratory laparotomy data. The specificity of the test in detecting neutered cats was 100% (95% confidence interval 96.2–99.9) and the sensitivity was 69% (95% confidence interval 59.3–76.8). The prevalence of neutered cats in this sample was 49%. The positive and negative predictive values were 1 and 0.77, respectively.
Conclusions and relevance
The Witness LH test correctly detected all unneutered cats and thus there were no false-positive results that incorrectly indicated a cat was neutered. This study therefore suggests that positive LH test results avoid the need to perform surgery to confirm neuter status. This has significant welfare benefits for cats as it provides a lower risk, faster and less traumatic alternative to surgery and, in the shelter setting, it will have a positive impact on the cost, speed of assessment and time to rehoming of cats.
Introduction
Between a quarter and a third of cats presenting to animal welfare organisations such as Cats Protection are recorded as strays at admission and many of the female cats are of unknown neuter status.1,2 Currently, the gold standard method of determining neuter status is performing exploratory laparotomy under general anaesthesia, which is invasive, costly and potentially risky. In addition, surgical confirmation of neutering for cats of unknown neuter status is likely to be more time consuming owing to potentially increased time under anaesthetic, more traumatic owing to increased length of incision and tissue handling, and may necessitate longer recovery. Exploratory laparotomy as a method to confirm neuter status may also be frustrating for the surgeon. From a shelter perspective, performing an exploratory laparotomy to confirm neuter status compared with using a blood test involves more surgery-associated costs (anaesthesia, suture material, professional fees) and results in longer length of stay. Clipping hair from the flanks and midline to check for evidence of a surgical scar is used by some practitioners, but this can be unreliable, leading to misdiagnosis of neuter status and occasionally resulting in pregnancy and unwanted litters.
It is widely recognised that circulating levels of luteinising hormone (LH) in unneutered cats are low, apart from a short-term surge of LH that occurs after mating that usually leads to ovulation.3–6 Ovulation can also occasionally occur in the absence of copulatory stimulation, caused by release of LH triggered by other stimuli. 7 Basal levels of LH during anoestrus are maintained by negative feedback from the gonads. Various studies have shown that after the gonads are removed, LH levels rise.5,8,9 It is this principle of higher LH levels in neutered cats that has stimulated interest in using detection of LH levels to differentiate between neutered and unneutered cats. Radioimmunoassay and a plate-based enzyme immunoassay have been used to detect LH in serum, but both are expensive, labour intensive, time consuming, require technical expertise and can only be performed in a laboratory.
The Witness LH test (Zoetis) is a semi-quantitative rapid immune migration assay that was developed for use in dogs to identify the LH surge and thus timing of ovulation. It has more recently been used to distinguish ovariectomised from sexually intact cats and dogs. It is an immunochromatographic assay that uses gold-conjugated antibodies to give a visual line in the presence of LH. A positive result occurs when the LH level in the sample is greater than 1 ng/ml, which indicates that the individual has been neutered (Witness LH direction insert). This test requires little technical expertise and provides a result in 20 mins without the need to send samples to a laboratory for analysis. The test instructions stipulate that any positive test result should be repeated in 2 h with a new sample to rule out a false-positive result that could be caused by the brief, episodic surges of serum LH that have been shown to occur in unneutered cats. 5 The test instructions also say to repeat the test 24 h later when a positive result is obtained in a cat suspected to be in oestrus.
The purpose of this study was to assess if the Witness LH test could be used to accurately assess the neuter status of queens. The study proposed that a single blood sample could be used to reliably predict the neuter status of female cats, thereby reducing the use of exploratory laparotomy to confirm whether a cat is neutered or not, as well as reducing the need for repeated blood sampling, as described above.
Materials and methods
Sample size calculations indicated that 323 samples would be required for 95% confidence and 5% precision of the estimate in a population with a 0.3 prevalence of neutered cats. Based on the findings of a pilot study and the sample size estimate, this study was conducted using all suitable cats admitted to four Cats Protection adoption centres over a 30 month period. This enabled enough data to be collected and assisted in the assessment of any effects related to time of year (ie, ensuring some anoestrous queens during the winter months were assessed).
Inclusion criteria were female cats of unknown neuter status estimated to be ⩾4 months of age. Exclusion criteria were females estimated to be <4 months of age, females that were palpably pregnant, lactating or with kittens, and cats with the tip of the pinna removed from the left ear (indicating likely previous neuter surgery).
Cats that met the inclusion criteria were blood sampled for feline immunodeficiency virus and feline leukaemia virus screening as per normal practice for cats admitted to Cats Protection adoption centres and the patient-side Witness LH test was performed on residual fresh serum within 24 h. The test was run in accordance with the manufacturer’s instructions by trained personnel at the adoption centres and then recorded as a positive, negative or ‘unable to interpret’ result. A photograph was taken of the test kit after the sample was run and this was interpreted by an independent observer who was blinded to the outcome recorded by the adoption centre staff. If there was a discrepancy in interpretation of the result, the photo was sent to the person who ran the test and a second (blinded) interpretation was requested.
The veterinary surgeons involved in the study were blinded to the results of the LH test. Cats were checked at flank and midline locations for evidence of a surgical scar at the time of first veterinary clinical examination, under general anaesthesia before exploratory laparotomy, or on both occasions. If the operating veterinary surgeon was unsure of the neuter status on the basis of any marking on the skin, the cat underwent exploratory laparotomy as per normal practice. The surgery was performed at least 24 h after blood sampling in case the sample happened to be taken during an LH spike in an unneutered cat. Ovariohysterectomy was performed where appropriate or the cat was confirmed neutered. In cases where venepuncture was performed for other clinical purposes, a second blood sample was collected on the day of surgery, in case a second test for LH was needed.
Severely haemolysed or lipaemic samples were excluded from the study, but some slightly haemolysed samples were included. The impact of poor technique in running the test was minimised by having the same person train all operators, having clear written instructions, by conducting a pilot study and by minimising the number of different people running the test. Any LH tests where the control failed were excluded from the study.
Results of the Witness LH test were compared with the results of exploratory laparotomy as gold standard. The outcomes used to assess the performance of the LH assay in this population were sensitivity, specificity, and positive and negative predictive values. Sensitivity, or the LH test’s ability to detect neutered cats, was calculated as the number of cats that the test correctly categorised as neutered (true-positive test result) divided by the total number of cats found to be neutered at exploratory laparotomy (positive gold standard result). Specificity, or the test’s ability to detect unneutered cats, was calculated as the number of cats that the test correctly categorised as unneutered (true-negative test result) divided by the total number of cats found to be unneutered at exploratory laparotomy (negative gold standard result). The positive predictive value (PPV) was calculated as the number of cats correctly identified by the LH test as neutered divided by the total number of positive LH test results. The negative predictive value (NPV) was calculated as the number of cats correctly identified by the LH test as unneutered divided by the total number of negative LH test results.
The study was approved by the University of Bristol’s ethics committee.
Results
Data were collected between 3 November 2014 and 3 May 2017. Four hundred and forty-two cats were enrolled in the study. One hundred and thirty-four cats did not have an LH test result and were excluded from the final analysis (Table 1). Seventy-two cats did not have an exploratory laparotomy and were excluded from the final analysis (Table 2).
Reasons for exclusion from analysis due to no luteinising hormone (LH) test result

FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus
Reasons for exclusion from analysis due to no exploratory laparotomy

LH = luteinising hormone; FeLV = feline leukaemia virus; FIV = feline immunodeficiency virus
Two hundred and thirty-six cats had both an LH test and exploratory laparotomy and were included in the data analysis. Twelve per cent of cats were sedated at time of blood sampling. Overall, 10 different staff members ran and interpreted the tests at the four adoption centres and approximately 19 different vets performed the exploratory laparotomies (some data were missing on name of surgeon performing the exploratory laparotomy). Samples were tested in all months of the year, ranging from 5–36 tests per month overall. There were no post-homing reports of cats coming into season or becoming pregnant 8 months after data collection ended (follow-up ranged from 8–36 months post-neutering, depending on the date the cat entered the study). The test performance results are summarised in Figure 1.
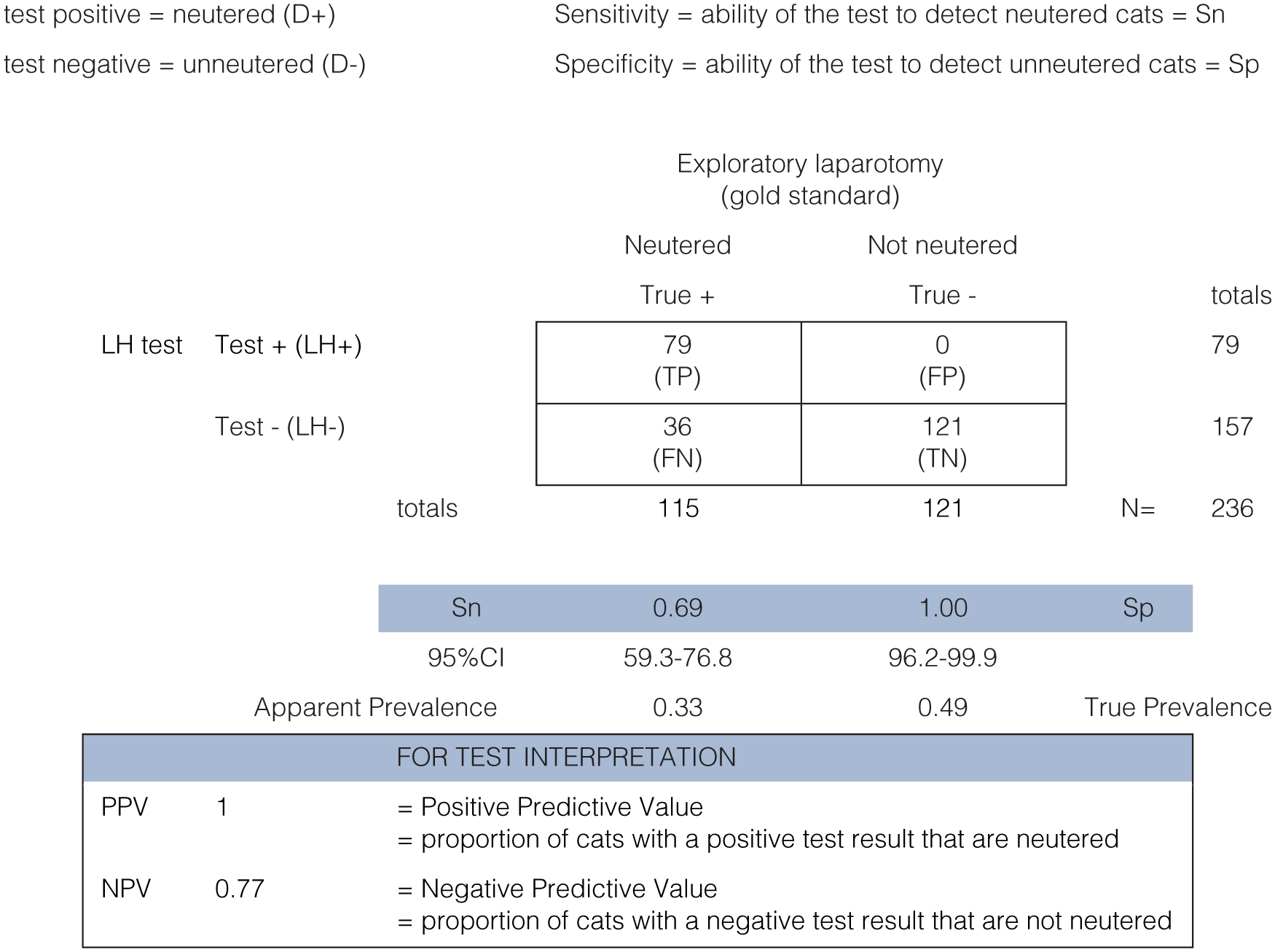
Summary of Witness LH test performance. CI = confidence interval; LH = luteinising hormone; TP = true positive; FP = false positive; TN = true negative; FN = false negative
Discussion
The specificity of the Witness LH test was 100% (95% confidence interval [CI] 96.2–99.9), indicating that the test had an excellent ability to detect unneutered cats. Sensitivity was 69% (95% CI 59.3–76.8), which indicated that a reasonable number of the neutered cats tested positive, but some false-negative results occurred (ie, the LH test indicated the cat was unneutered, but it was found to be neutered at exploratory laparotomy). The primary consideration when using the test to assess neuter status is that every positive test represents a truly neutered cat so that an unneutered cat is not rehomed in the mistaken belief that it is neutered. These data provide evidence that within our sample the Witness LH test fulfilled this requirement (PPV of 1). The NPV of 0.77 for this study (where the prevalence of neutered cats was 49%) indicated that for negative LH test results, an exploratory laparotomy should be performed to confirm the true neuter status of the cat.
A small study by Scebra and Griffin of 50 cats of known neuter status (48% neutered) found that the Witness LH test categorised all neutered cats correctly, but classified two of the unneutered cats as being neutered, resulting in a sensitivity of 100%, a specificity of 92.3%, a PPV of 92% and a NPV of 100%. 10 In contrast to the data presented here, they encountered false-positive results but not false-negative results. This difference between results could be due to the small sample size in the study by Scebra and Griffin, 10 but, ultimately, the reason for this discrepancy is unknown. Rohlertz et al compared the Witness LH test to the gonadotropin releasing hormone-stimulation test in 31 cats of known neuter status (48% neutered) and reported one falsenegative result and no false-positive results with the LH test. 11 These results are similar to the data presented here, but, as with the study by Scebra and Griffin, 10 the sample size was small. Most recently, Alm and Holst compared the Witness LH test to an anti-Müllerian hormone assay in dogs of known neuter and oestrous status and found that in 100 non-oestrus bitches, the Witness LH test produced three false-negative test results and no false-positive test results. 12 The present study was designed to assess the performance of the Witness LH test under the conditions it is most likely to be used, namely, when neuter status of the cat is not known at time of testing and where multiple users are running and interpreting the test.
In this application of the Witness LH test, the consequences of a false-positive test result are greater than that of a false-negative result; if a cat was homed or released as neutered on the basis of a (false) positive LH test result, but was not actually neutered, this has more significant consequences than obtaining a (false) negative result, performing exploratory laparotomy and then establishing that the cat is already neutered. The test is useful because the majority of cats that were already neutered were ruled out of needing an exploratory laparotomy by the positive LH test result. In addition, as there were no false-positive results, all of the unneutered cats that needed to go to exploratory laparotomy were identified by the test.
The lack of false-positive results also indicated that none of the unneutered cats had a pre-ovulatory LH peak, a spontaneous ovulation or an episodic surge of LH at the time of sampling. In most queens behavioural signs of oestrus are very obvious and the other scenarios are probably extremely uncommon. Thus, the results of this study suggest that it is not necessary to do a second LH test with a new sample every time a positive result is obtained. The exception to this would be if a cat is suspected to be showing signs of oestrus, a second test could be performed 24 h later (as per the test instructions); however, if the signs of oestrus were unequivocal, the cat would more likely be taken to surgery for neutering without waiting and repeating the test. In this group of cats, two were recorded by staff as showing signs of oestrus and both of them were negative with the LH test and were found to be unneutered at exploratory laparotomy.
The Witness LH test correctly identified all unneutered cats in this study and the tests were run every month of the year, suggesting that the time of year when the sampling is carried out does not affect the accuracy of the test. This is reassuring in that it indicates the performance of the test is not affected by anoestrus.
There were 36 false-negative test results (36/157 negative test results [23%] ie, when the test indicated the cat was unneutered when it was actually already neutered). Possible explanations for this are testing error, gold standard error, physiological differences between neutered cats, stress or anaesthetic agents.
Testing error can occur owing to factors such as sample quality, poor technique running the test, failure of the control and inaccurate test interpretation (misclassification of the result). The majority of false-negative results occurred when clear serum samples were used compared with slightly haemolysed samples (data not shown), so sample quality did not appear to influence the number of false-negative results in this study.
Inaccurate test result interpretation was likely to be the most significant source of error with this semi-quantitative test because determination of a positive result was based on subjective assessment of the test line being ‘similar or greater intensity to the control line’. Visual interpretation of immune migration assays can be problematic and is a recognised issue. 13 In this case, it was not clear from the test insert instructions whether ‘similar’ includes only test lines that are equal to the control line or whether test lines that are slightly less intense than the control line would be considered ‘similar’ and thus be categorised as a positive test result.
In addition, people have different abilities to detect small differences between colour intensity and this needs to be accounted for when developing patient-side tests that will, by their nature, be run and interpreted by many different people. Post-study assessment of photos of the false-negative results showed that in about half of them the test line was less intense than the control, but it was close enough that it could be interpreted as similar and thus be categorised as a positive result. If this was the case, in this set of data, test performance results for this study would become sensitivity 84%, specificity 100%, PPV of 1 and NPV of 0.87.
If the interpretation parameters for the test could be improved, the frequency of false-negative results could be reduced. One promising solution to this would be the use of a recently developed smartphone application that quantitatively measures the LH level based on a photo of the test. 14
While it is generally accepted that LH levels are higher in neutered cats than in unneutered cats,5,6,8,9 it is possible that some neutered cats naturally have low LH, or LH levels in neutered cats are variable and occasionally fall below the detection limit of assay (1 ng/ml), thus leading to a false-negative result. It has been shown that time since neuter may also affect circulating levels of LH,5,9 but in this study it was not known when the cat was neutered and so it was not possible to attribute the explanation for false-negative results to this. The lack of knowledge of when the cat was neutered does reflect the conditions under which the test would normally be run (ie, to find out whether a cat is neutered or not). A study that measured LH levels over time post-neutering using the Witness LH test would provide more information on how LH levels vary after neutering and whether this impacts on test performance.
Stress has been shown to reduce LH secretion in ewes 15 and in cats it has been shown to reduce pituitary sensitivity to LH-releasing hormone, 16 so this could have had an impact on the number of false-negative results. Degree of stress was not formally assessed in this study, but further studies could examine whether cats that are experiencing a high level of stress are more likely to have false-negative LH test results. Anaesthetic agents have also been shown to inhibit LH release in multiple species, including cats; 5 however, in this study sedated cats were not over-represented in cats that had a false-negative LH test result (data not shown).
Conclusions
In this study, the Witness LH test had excellent specificity and reasonable sensitivity for use in cats of unknown neuter status. There are significant welfare benefits to using this test in cats of unknown neuter status because all cats that test positive can confidently be identified as neutered, without the need for further assessment by a surgical procedure. While this study showed that not every neutered cat will be detected by the LH test, 77% of them were, which means an approximate potential 77% reduction in these unnecessary surgeries.
The Witness LH test is currently the only available non-surgical test for assessing the reproductive status of cats that is easy to run, where a reliable result can be obtained using one blood sample and where an answer is obtained only 20 mins after sampling. This is a major advantage in practical field applications. All other currently available blood tests to determine neuter status require multiple blood samples, injections of hormones and/or need to be sent to a laboratory for analysis, resulting in higher cost and a significantly longer time from assessment to results.
Footnotes
Acknowledgements
The authors wish to thank the adoption centre staff and vets for their diligence in collecting the data for the study.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors wish to acknowledge that the LH test kits used in the study were supplied by Zoetis. JM was funded by Cats Protection.
