Abstract
Introduction: Patients with diabetes are prone to infections. An elevated C-reactive protein (CRP) level indicates an underlying infection/inflammation; hence, a reliable point-of-care (POC) CRP measurement, independent of glucose level, would be advantageous in rapid detection of an inflammatory process and allowing for appropriate therapy in a timely manner. Methods: A validation study of patients with type 2 diabetes mellitus diagnosed with an inflammatory condition either infectious or non-infectious underwent a capillary CRP measurement on the same blood drop used for the measurement of capillary glucose level. Results: 154 samples from patients with diabetes across a broad range of CRP levels that underwent capillary CRP measurement, demonstrating a linear profile of the dynamic range compared with the wide range CRP assay, with significant correlation of R2 = 0.9 and a slope of 0.98 (95% C.I: 0.91–1). Bland Altman analysis, presents a positive bias profile; the total difference between assays is 6.6 mg/L. The mean bias between methods is 13.34% (C.I 95% 9.8–17.5%). The total agreement between the methods is 89%, the observed Kappa index is: 0.71 with confidence interval of 0.57–0.84 indicating substantial agreement between the methods. C-reactive protein level is independent of glucose concentration. Conclusion: Point-of-care capillary CRP testing is a useful mean of early detection of infection/inflammation in patients with diabetes and enables the treating physician to rapidly assess and treat the patients underlying inflammatory process.
Introduction
It is well established that patients with diabetes are prone to infections.1–4 Infectious as well as non-infectious inflammation in patients with diabetes is associated with several pathophysiologic mechanisms including endothelial dysfunction, formation of advanced glycosylation end products, accelerated atherosclerosis, and altered extracellular matrix composition.5–7
Biomarkers are helpful in the diagnosis and management of infection/inflammation. Moreover, they enable the treating physician to assess the severity of the underlying inflammatory process and its response to treatment. 8 C-reactive protein (CRP) has the advantage of rising rapidly at the initiation and propagation of the inflammatory process and subsiding rapidly upon its termination compared to other inflammatory biomarkers.9–11
Point-of-care (POC) CRP in this context, might be attractive for real time assessment of the developing inflammatory process, and therefore is suitable for use at the diabetic clinic12,13 and was demonstrated to contribute to antibiotic stewardship. 14
Although POC CRP testing is well established in general medicine, it has not been specifically tested in a cohort of patients with diabetes, which is the focus of the present study which is the first study exploring the subject to our knowledge.
Materials and methods
Study design and setting
A validation study of patients diagnosed with type 2 diabetes mellitus according to American Diabetes Association guidelines 15 and admitted between 1 April 2020 and 31 July 2020 to the department of internal medicine in the Sourasky Tel Aviv Medical Center. Wide range C-reactive protein (wrCRP) test is included in the routine chemistry panel in all participants. This was a retrospective analysis of data collected from patients who were routinely monitored during their hospitalization for their glucose level by using a glucometer device.
The study includes patients with diabetes diagnosed with an inflammatory condition either infectious or non-infectious. Infections were determined by using clinical signs and symptoms as well as radiological and microbiological workup and were discussed for each patient with an infectious diseases specialist (AW).
Patients aged younger than 18 years of age, or patients not diagnosed with diabetes mellitus and an inflammatory condition were excluded from the study as well as patients who were not admitted to the department of internal medicine. The study was reviewed and approved by the Institutional Review Board (number 0491–17). The minimum sample sizes have been calculated by WinPepi software under the assumptions of type I error of 0.05 and 80% power. The interclass correlation set on 0.9, under those assumptions the required sample size is 14. 16
Laboratory methods
The same blood drop drawn from the patient’s fingertip is used to measure the capillary glucose level which is performed by the StatStrip glucose meter (Nova biomedical, Waltham, Massachusetts, United States) and the capillary CRP which is performed by the EUROLyser integrated capillary test kit (EUROLyser Diagnostica GmbH, Salzburg Austria). This POC method measures CRP concentration in capillary whole blood by polystyrene-enhanced immunoturbidimetric assay by photometric measurement at 546 nm or 700 nm of antigen-antibody reaction between antibodies to human CRP bound to polystyrene particles and CRP present in the sample. The capillary CRP precision is assessed by the control sample. In total, the capillary CRP precision is good with the total (within laboratory) coefficient of variation (CV) ranging from 9% for low concentration control (13 mg/L) and up to 5% for the high concentration control (47 mg/L). Control samples are performed for 90 days without any outliers (Figure 1). Levey Jennings figure of controls performance (90 days). Controls were performed for 90 days by the Eurolyzer equipment. Each point represents one day. The X-axis represents days passed from the starting point of the controls measurements. The zero line represents the controls' target value: Low control (14 mg/L) and high control (52.9 mg/L). The Y axis represents the deviation from the target value. The dotted line represents 2 standard deviation from the target value. The target value was report by the company. S.D: standard deviation.
The limit of quantitation (LOQ) of the capillary CRP is 2.1 mg/L comparing the ADVIA® wrCRP 0.03 mg/L, and the limit of linearity for wrCRP is 155 mg/L. Due to the discrepancy between the methods, we analyzed the patient results within the concentration of 2.1–155 mg/L by Deming Weighted regression separately from the concentrations lower or higher than the dynamic range (Figure 2). The cohort is divided into three groups according to the limits of the dynamic range (<2.1, 2.1–155, >155) in order to assess the agreement between the methods across the whole concentration range. Deming Weighted Linear regression between wrCRP and capillary CRP based on patient samples. Each measurement represent by dot. The solid regression line coefficient of determination for CRP range 2.1–155 mg/L is r2 = 0.9 with 0.98 slop (95% C.I: 0.91–1) and −5.6 intercept (95% C.I −10 to −0.4). Capillary CRP is measured by EUROLyser equipment.
Wide range C-reactive protein measurements are performed by ADVIA® (Siemens Healthcare Diagnostics Inc., Tarrytown, NY, USA). The ADVIA® Chemistry wrCRP method measures CRP in blood by a latex-enhanced immunoturbidimetric assay. It is based on the principle that the analytic concentration is a function of the intensity of scattered light caused by the latex aggregates. The latex particles coated with anti-CRP antibodies rapidly agglutinate in the presence of CRP-forming aggregates. Agglutination takes place increasing in turbidity, which is measured at 571 nm. This method measures the wrCRP concentration range of 0.03–155 mg/L. When the measured concentrations exceed 155 mg/L, a dilution of 1:4 is performed to extend this range. Point-of-care CRP and wrCRP were measured simultaneously within 4 hours after blood withdrawal.
Statistical methods
The agreement between capillary and wrCRP is evaluated by using a cohort of patients’ samples. Samples run after quality control (QC) testing. Two QC levels are used to evaluate CRP precision and determine the CV. Those QC materials (L1 = 14 mg/L, L2 = 52.9 mg/L) are measured daily prior to samples measurement during 90 days. The maximum allowable CV was set arbitrarily at 10%. Controls repetition is plotted using Levey Jennings method (Figure 1). The mean and the standard deviation of the graph are according to the manufacturing report.
The criteria for agreement between capillary CRP and wrCRP are defined as follows: the 95% confidence interval (CI) of the slope should include 1 and 95% CI of the intercept includes 0. The proportion of explained variation (R2) > 0.9, these parameters present a minimum of permanent or systematic error. The bias between the methods assessed also by Bland–Altman analyses showing the relationship between the methods and their average.
Statistical analyses performed with SPSS (IBM SPSS Statistics for Windows, version 27, IBM Corp., Armonk, NY, USA, 2020) combined with the Prism GraphPad Software (San Diego, California, USA).
Results
Patients characteristics.

CHF- congestive heart failure, CRP- C-reactive protein, IQR- interquartile range, MI- myocardial infarction, UTI- urinary tract infection.
Quality control samples have been done before samples measurements during 90 days. The QC data are within −1 SD under the target, suggesting a negative bias or need for some correction for the target value.
The capillary CRP has a dynamic range between 2.1 and 155 mg/L in this range the agreement between the methods assessed using regression line (Figure 2). The proportion of explained variation R2 = 0.9 with a slope of 0.98 (95% C.I: 0.91–1) and intercept of −5.6 (95% C.I: −10 to −0.4).
Bland Altman analysis, which represents the agreement between the two CRP measurement methods, presents a positive bias profile; the total difference between the assays is 6.6 mg/L. The mean bias between methods is 13.34% with 95% C.I 9.8–17.5% in total the 95% of the differences between the measurements range from −28.5% to 56.2% (Figure 3). Agreement between the wrCRP method and capillary CRP method according to CRP concentration. Y axis represents the different (%) between the capillary CRP and wrCRP methods %Bias = (wrCRP-capillary CRP)/wrCRP*100 according to CRP average concentrations (X axis). Interval includes 95% of the differences represented by dotted line’ and the bias represents by dashed line. CRP: C-reactive protein; wrCRP: wide range C-reactive protein.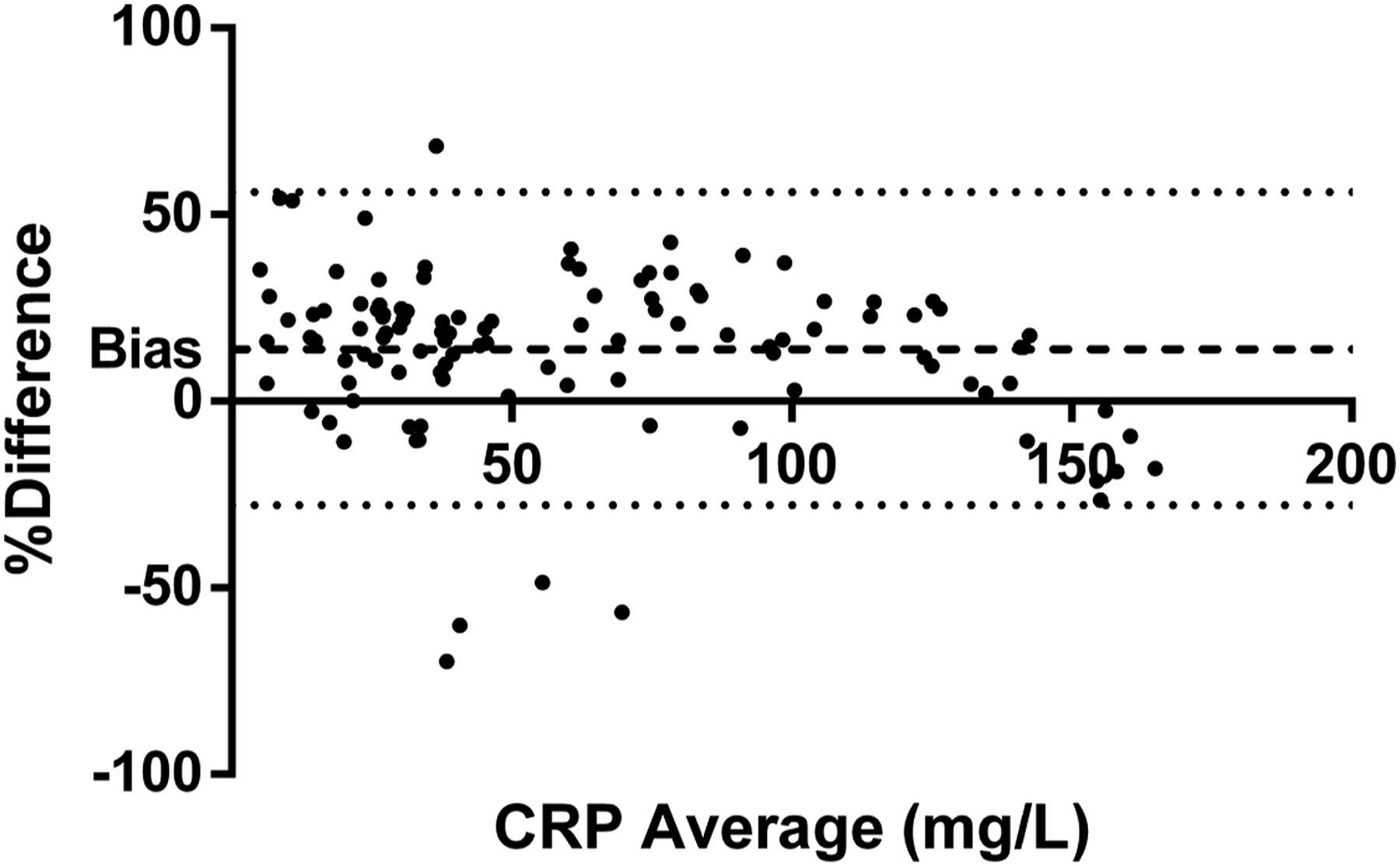
The agreement between capillary CRP and wrCRP methods in concentrations out of the dynamic range.

154 measurements divided into three groups according to the CRP methods. The number of cases in each group and the percentage from the total are shown.
CRP: C-reactive protein; wrCRP: wide range C-reactive protein.
The 8 cases of disagreement between capillary CRP and wrCRP methods.

Discussion
Point-of-care CRP testing has been evaluated previously and was found to be valid in forensic medicine, 17 it enabled to reduce immediate antibiotic prescribing in adults and children18,19,20 and was useful for veterinary care of canines. 21
This study demonstrates the usefulness of POC capillary CRP testing in the presence of acute infection/inflammation in a cohort of patients with diabetes and its correlation with the wrCRP testing in this population of patients.
The main finding of this study is that the capillary POC CRP test, correlates with the wrCRP test among patients with diabetes. CRP values below 2.1 mg/L are not accurately discriminated by the POC CRP test. Therefore, we decided to address all CRP values below 2.5 mg/L as one group since the difference between CRP values below 2.5 mg/L is clinically less significant. Similarly, since the wrCRP test performs dilution of all samples with CRP values higher than 155 mg/L, which is not performed by the POC capillary CRP test, we have considered all patients with CRP values higher than 155 mg/L as one group of patients. The CRP value of each patient in this group is consistent with mega inflammation and the exact value of CRP is clinically less significant, since the appropriate management of this inflammatory process would not change between different CRP values above 155 mg/L. Moreover, we believe that for the everyday management of patients at their bedside, the clinically significant and relevant range of POC capillary CRP is 2.5–155 mg/L, which correlates well with their inflammatory state. Although systemic or constant error between methods are present and controls has shown systemic lower values (Figure 1). In addition, the proportion of explained variation R2 = 0.9 with a slope of 0.98 (95% C.I: 0.91–1) and intercept of −5.6 (95% C.I: −10 to −0.4) strengthens the assumption for low probability of constant or systematic error. In patients with mega inflammation the bias between the methods increases (Table 3) but at these CRP concentrations it is clinically less significant since a substantial underlining inflammatory process is taking place as expressed by these CRP concentrations.
Point-of-care capillary CRP allows the physician to rapidly assess the developing inflammatory process responsible for the patient with diabetes seeking of medical assistance. It allows for quick evaluation and a simple mean of triage of patients with diabetes according to the severity of ongoing inflammation. Moreover, the POC capillary CRP is a mean to perform rapid antibiotic stewardship allowing for appropriate antibiotic therapy given in a timely manner and avoiding unnecessary antibiotic treatment. 14 Furthermore, utilizing POC capillary CRP allows for substantial healthcare cost saving. 22
Patients with diabetes and their physicians are accustomed to measuring blood glucose levels by means of POC capillary glucose test, hence a similar technique using the same drop of blood for measuring POC CRP will be comfortable and easy to adapt to as part of a routine assessment of the medical condition of patients with diabetes.
One limitation of this study is that although its precision is verified for a long time interval, a protocol suggested by the literature (for example, CLSI) was not applied; however such a protocol will be useful for future studies. Another limitation of the study is its retrospective analysis of existing data. In addition, although HbA1C levels and ratio of insulin treatment were not examined in this study; we believe it should be a topic of future study to examine their impact on CRP levels in diabetic patients with an inflammatory condition.
This is a validation study of the technology performed in our laboratory, which demonstrates the feasibility of POC methods to assess inflammatory infectious and non-infectious processes in patients with diabetes. This is the first time that glucose and CRP levels in patients with diabetes measured by POC methods are shown to be independent of each other. The POC CRP test and POC glucose test can be performed on the same drop of blood (as performed in the present study). By using one drop of blood for both tests, an assessment of both inflammatory process and glucose level can be achieved rapidly, efficiently with no need for any additional potentially cumbersome blood draw. This study is relevant for clinicians who treat diabetic patients outside of the hospital or relatively large medical facilities for evaluation of potential inflammatory burst.
We consider the vast variety of inflammatory etiologies whether infectious or non-infectious of the patient in our cohort to be an advantage, demonstrating the generalizability of our results to patients with diabetes.
Conclusions
In conclusion, we suggest including capillary CRP in the arsenal of diabetic clinics to help early signaling of the development of acute infection/inflammation if a clinical suspicion is present.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the local ethics committee (number 02–049) approved the study
Informed consent
Informed consent was not sought for the present study due to its retrospective design and anonymous medical chart review process.
Trial registration
Not applicable.
