Abstract
Background
Conventional ketamine hydrochloride solutions are acidic and hyperosmotic, limiting their tolerability for subcutaneous (SC) delivery. BB106 is a novel ketamine formulation using the multitude of sulfobutylether-beta-cyclodextrin (SBECD) anionic substitutions as ionic counterions to achieve a new ketamine–SBECD salt in solution at near-physiologic pH and isotonicity, thereby enabling SC administration. To support its development for pain and neuropsychiatric indications, we conducted nonclinical toxicology studies in rats and minipigs.
Methods
A 2-week repeated-dose SC injection study in rats and a 4-week continuous SC infusion study in Göttingen minipigs were performed, each with a 2-week recovery phase. Assessments included clinical observations, body weights, food consumption, ophthalmology, electrocardiography, clinical pathology, toxicokinetics (TK), and histopathology.
Results
BB106 was well-tolerated in both species. No mortality or dose-limiting systemic toxicity occurred. Clinical signs were consistent with ketamine pharmacology (eg, transient ataxia) and resolved after dosing. Local site reactions were minimal, histologically mild, and reversible. No treatment-related lesions were observed in any of the systemic tissues examined (including liver, kidney, bladder, and brain). TK analyses confirmed consistent systemic exposure without accumulation. SBECD itself produced no adverse effects at the exposure levels tested.
Conclusion
Repeated SC bolus injections in rats and continuous SC infusion in minipigs demonstrated favorable local and systemic safety profiles for BB106. These findings support its feasibility as an alternative to intravenous ketamine for pain and psychiatric disorders. To our knowledge, this is the first toxicology report of SC administration of an SBECD salt formulation and the results support continued consideration as a potential medicine.
Keywords
Introduction
Ketamine is a small molecule N-methyl-D-aspartate (NMDA) receptor antagonist that has been used for decades as a dissociative anesthetic in both human and veterinary medicine.1,2 In recent years, interest in ketamine has expanded due to its rapid-acting antidepressant and analgesic properties.3-5 In 2019, the S-enantiomer of ketamine received regulatory approval as a nasal spray for treatment-resistant depression, 6 underscoring the therapeutic potential of ketamine beyond anesthesia. Sub-anesthetic dosing is increasingly used off-label for acute and chronic pain, including neuropathic pain,1,7,8 and multiple meta-analyses support its efficacy for postoperative pain relief.4,9-11 Consensus guidelines now recommend ketamine infusions in both acute 12 and chronic pain settings. 13
Despite these benefits, ketamine’s pharmacokinetics complicate delivery. Oral bioavailability is poor (∼8–24%) 4 and systemic clearance is rapid (t½ = 2–4 h), 4 necessitating parenteral administration to achieve consistent therapeutic plasma levels. Continuous intravenous (IV) infusion is effective but often impractical outside hospital environments. 14 Subcutaneous (SC) administration offers a convenient alternative, providing high bioavailability with the potential for both bolus and continuous dosing in ambulatory care.
SC ketamine infusions have been investigated for chronic pain, but wider adoption has been limited by injection-site reactions and the absence of approved formulations or pump-based platforms. Repeated SC injections of ketamine have been associated with irritation, itching, and painful induration.15,16 These local effects likely arise from physicochemical properties known to affect subcutaneous formulation tolerability.17,18 USP ketamine hydrochloride solutions are labeled at pH 3.5-5.5 (REF 19) but are reported at pH 4.09–4.18 for 50 mg/mL formulations and 3.88–3.94 for 100 mg/mL formulations. 1 Prepared ketamine HCl solution at 100 mg/mL is reported to be hyperosmotic (740.1 mOsm/kg at 100 mg/mL). 1 Attempts to correct these by raising pH trigger freebase precipitation, 1 while dilution increases injection volume, both impractical for clinical use.1,17,18
To overcome these limitations, BB106 was developed as a novel ionic ketamine sulfobutylether-beta-cyclodextrin (SBECD) salt using the sponsor’s proprietary SolusaltTM technology. Unlike conventional SBECD inclusion complexes that increase osmolality,20-22 BB106 exploits the multitude of anionic SBECD substituents as counterions, yielding a stable, highly multivalent salt with favorable physicochemical properties. At 70 mg/mL, BB106 is essentially isotonic (∼290–300 mOsm/kg), maintains stability at pH 5.5, and provides buffering capacity against pH drift. 1 This formulation meets the critical requirements for SC tolerability—near-physiologic pH, isotonicity, low injection volume, and solution stability—supporting its advancement as a reformulated ketamine product optimized for SC delivery.
Before clinical evaluation, it was necessary to characterize BB106’s safety profile through nonclinical toxicology studies in 2 species. While ketamine’s pharmacology and acute toxicology are well-characterized for IV and IM routes, data on local tissue effects and subchronic safety via SC dosing are limited. To address these gaps, we conducted 2 GLP-compliant studies in accordance with ICH M3 guidance: (1) a 2-week repeated-dose SC injection study in rats and (2) a 4-week continuous SC infusion study in minipigs. These complementary models simulate the 2 most relevant clinical scenarios—intermittent SC bolus injection and continuous SC infusion—and enabled assessment of both systemic and local toxicity during dosing and after recovery.
Materials and Methods
Regulatory Compliance and Ethical Oversight
These studies were conducted in full compliance with Good Laboratory Practice (GLP) guidelines and the European Directive 2010/63/EU for the protection of laboratory animals. All protocols were approved by the Institutional Animal Welfare Body of the test facility, ERBC, which is AAALAC-accredited. The minipig study design followed OECD and FDA guidelines for subchronic toxicity and TK evaluations in non-rodent species.
Animals and Housing
Sprague-Dawley rats (Hsd:SD strain; Envigo RMS, Italy) were selected for their regulatory acceptance and established use in toxicology. Five groups of 10 males and 10 females (8–9 weeks old at study start) were acclimatized for ∼4 weeks before dosing. Rats were housed in groups of up to 5 per cage in a limited-access facility under controlled conditions (22 ± 2°C and 55 ± 15% relative humidity, 12-hour light/dark cycle). Standard rodent diet and tap water were provided ad libitum, with periodic feed and water analysis.
Twenty-five male and twenty-five female Göttingen minipigs (Ellegaard Göttingen Minipigs A/S, Denmark), 5–11 months of age and weighing 20–27 kg at allocation, were selected as the non-rodent species based on their regulatory acceptance and physiological similarity to human skin. Following a minimum 3-week acclimatization and quarantine period, only animals deemed healthy by veterinary examination and baseline clinical pathology were included. Minipigs were housed individually in indoor pens under controlled environmental conditions (19 ± 3°C, 55 ± 15% relative humidity, 12-h light/dark cycle) with straw bedding and visual contact for enrichment. Animals received SDS SQC diet twice daily with rations adjusted to growth curves, and tap water was provided ad libitum except during designated procedures.
Formulation and Administration of Test Article and Controls
BB106 was provided as a sterile, ready-to-use solution containing racemic ketamine at 70.2 mg per mL, SBECD at 105 mg/mL, and benzethonium chloride at 0.1 mg/mL, with certificate of testing at pH 5.55 and osmolality of 284 mOsm/kg. The vehicle control was provided in water for injection containing SBECD sodium at 107 mg/mL and benzethonium chloride at 0.1 mg/mL, with certificate of testing at pH 5.50 and osmolality of 293 mOsm/kg. Saline control was provided as normal saline solution (0.9%). Sterility was ensured by passing solutions through a 0.22 μm polyvinylidene fluoride (PVDF) filter before administration each day. Filtration was confirmed to not affect test item concentration or pH.
For the minipig study, BB106 or control solutions were delivered by continuous subcutaneous (SC) infusion using a programmable miniature pump (CRONO-SC®, Canè S.p.A., Italy) connected to an infusion set (Neria™ guard, 9 mm Teflon cannula, Unomedical, Denmark). One to two days prior to dosing, hair on the dorsal flanks was clipped to facilitate placement. On Day 1, a sterile 9 mm cannula was inserted into the dorso-lateral SC tissue and secured with adhesive tape; the tubing was covered with a flexible bandage, and the pump was housed in a pouch within a custom-fitted jacket to permit free movement without catheter dislodgment. The pump and SC route were selected to model the intended clinical mode of BB106 administration in humans.
Group Allocation and Dosing Regimens
Experimental Design of the Rat Study.
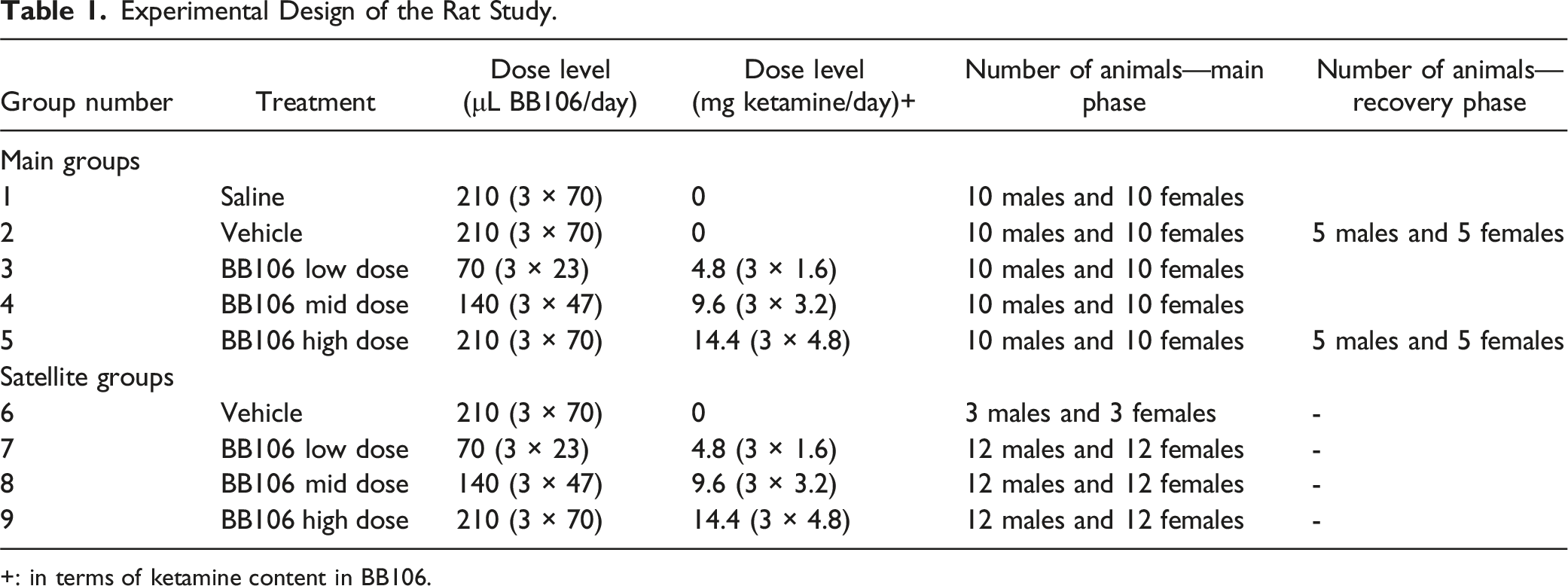
+: in terms of ketamine content in BB106.
Satellite groups of rats were included for toxicokinetic (TK) analysis: Groups 7–9 (12 males and 12 females each) received BB106 at low-, mid-, and high doses, and Group 6 (3 males and 3 females) received vehicle only (Table 1). These animals were dosed on the same schedule as the main groups and used exclusively for serial blood collection. Based on average baseline body weights (females ∼225 g, males ∼350 g), the fixed daily doses corresponded to approximately 21.3–64.0 mg/kg in females and 13.7–41.1 mg/kg in males.
Experimental Design of the Minipig Study.
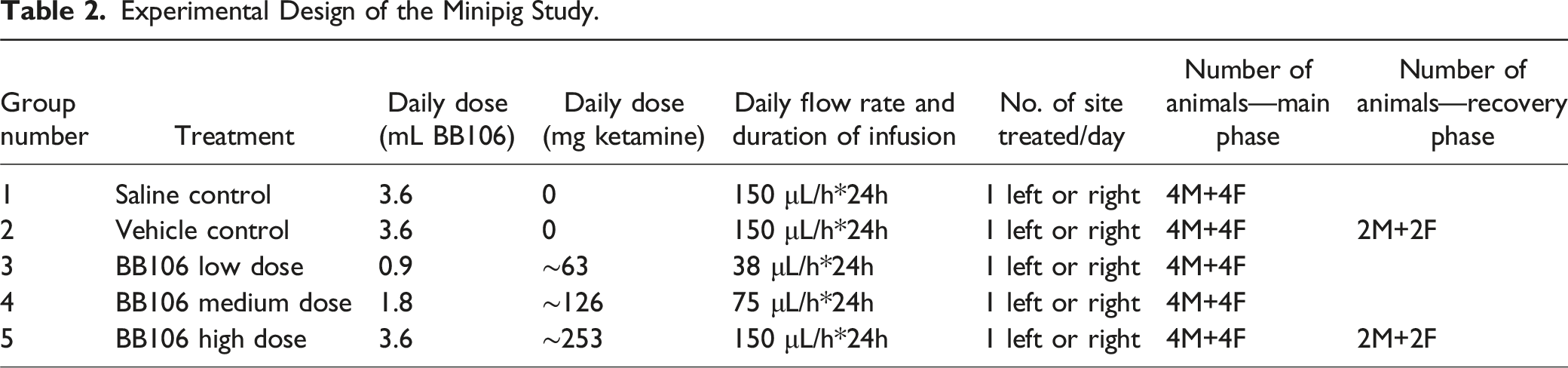
BB106 was administered to minipigs undiluted at a fixed concentration (70.2 mg/mL ketamine) by continuous SC infusion for 28 consecutive days. Flow rates were ∼38 μL/h (0.9 mL/day, 63 mg/day), ∼75 μL/h (1.8 mL/day, 126 mg/day), and ∼150 μL/h (3.6 mL/day, 253 mg/day) for the low-, mid-, and high-dose groups, respectively. Fixed dosing was selected to maintain low infusion volumes across multiple animals; calculated exposures corresponded to ∼2.4–11.2 mg/kg/day in males and ∼2.3–10.3 mg/kg/day in females, based on initial body weights and growth during the study. The low dose approximated the anticipated clinical dose (allometrically scaled), while mid and high doses provided exposure margins. Control groups received saline or vehicle at the highest infusion volume (3.6 mL/day).
Infusion sites were rotated daily among 16 predefined dorso-lateral thoracic/flank locations (8 per side) marked with indelible ink (Supp. Figure 2). Most sites were used only once for a 24-hour infusion; 4 sites were used twice after a ≥1-week interval to assess potential effects of repeat use. Pumps were refilled daily and monitored to ensure proper flow. Animals in Groups 2 and 5 designated for recovery were observed untreated for 14 days following the completion of dosing.
Observations and Measurements
Animals were monitored daily for viability, general clinical condition, and signs of toxicity throughout the in-life phases. Rats and minipigs were observed at least twice daily, with detailed clinical examinations recorded once daily. Particular attention was given to the SC administration sites (bolus in rats, infusion in minipigs) that were inspected for local reactions such as erythema, swelling, induration, or edema; abnormalities were scored and documented. Body weights were recorded before dosing and weekly thereafter, with additional terminal or recovery measurements as appropriate. Food consumption was measured weekly for rats (per cage) and daily for minipigs (per individual). Ophthalmologic examinations were performed pre-dose and during dosing (Week 2 in rats; Week 4 in minipigs), with recovery animals re-examined at study completion. In minipigs, cardiovascular safety was further assessed by 3-lead ECG recordings obtained pre-dose, on Day 1, and during Week 4. Clinical pathology evaluations (hematology, clinical chemistry, coagulation, urinalysis) were conducted at the end of the dosing phases for both species and repeated at the end of the recovery phase for the designated subgroups.
Toxicokinetics
Systemic exposure to ketamine and its primary metabolite (norketamine) was characterized in both species using validated LC-MS/MS. In rats, blood samples were collected from satellite TK groups on Day 1 and Day 14 at pre-dose, at 0.25, 0.5, 2, and 4 hours after the first and second daily injections, and at 0.25, 0.5, 2, 4, 8, and 16 hours after the third daily injection. In minipigs, blood was sampled from all main-study animals on Days 1 and 28 pre-dose, and at 2, 4, 6, 8, and 24 hours after the start of the infusion to characterize early exposure and steady-state profiles. Plasma concentrations were used to calculate TK parameters by non-compartmental analysis.
Terminal Procedures and Histopathology
At scheduled termination (Day 15 for rats and Day 29 for minipigs) or following recovery periods, animals were euthanized under anesthesia (rats: isoflurane with exsanguination; minipigs: barbiturate overdose or exsanguination) and subjected to full necropsy. Gross pathology was recorded, and major organs were dissected and weighed. Organ-to-body and organ-to-brain weight ratios were calculated. In both species, tissues were preserved in 10% neutral-buffered formalin (with eyes/testes in Davidson’s fixative) and processed for histology. Histopathological examination focused on all major organs, with comprehensive evaluation of administration sites. In rats, all SC injection sites were examined; in minipigs, all 16 predefined infusion sites were collected (used and unused) to assess local tolerance. Microscopy was performed on control and high-dose groups for systemic organs, with lower-dose groups examined as needed to clarify target organ findings. Any gross lesions were correlated with histology. Peer review of pathology findings was conducted by a second pathologist for rats and an external reviewer for minipigs.
Statistical Methods
Data from both studies were analyzed using validated statistical software. Continuous variables (body weight, food consumption, clinical pathology, organ weights) were summarized as means ± SD and compared across groups by one-way ANOVA. If significant differences were observed (P < 0.05), Dunnett’s test was applied for pairwise comparisons with controls. Bartlett’s test was used to assess variance homogeneity; if violated, the Cochran–Cox modified t-test was used instead. Incidence data such as histopathological findings were compared using non-parametric methods (eg, Kolmogorov–Smirnov test). Statistical significance was set at α = 0.05, and results were interpreted in the context of biological relevance.
Results
2-Week Repeated-Dose SC Injection Study in Rats
Mortality and Clinical Observations
All rats completed the 2-week dosing and the 14-day recovery phases with no treatment-related mortality. Clinical findings were limited to expected ketamine effects and local injection-site reactions. Ataxia was observed in all mid- and high-dose animals (9.6 and 14.4 mg/day), generally slight to moderate in severity, with 1 high-dose male exhibiting marked ataxia. Injection-site swelling occurred transiently in mid- and high-dose groups, typically mild and occasionally moderate, more frequently in males. Isolated palpable masses were noted in 1 vehicle-control female (mammary region) and 1 high-dose female (ventral abdomen). No abnormal clinical signs were seen during recovery.
Body Weight and Food Consumption
BB106 administration produced only transient effects on body weight and food intake. On Day 4, low-dose males (4.8 mg/day) showed a statistically significant, dose-related reduction in mean body weight versus controls, but values normalized by the end of dosing. Overall weight gain in all treated groups, including during recovery, was comparable to controls aside from minor fluctuations linked to scheduled fasting on Day 15 (Supp Figure 3). Food intake showed a similar pattern: on Day 4, both sexes exhibited dose-dependent reductions (significant in mid- and high-dose groups), which resolved by Day 8. Thereafter, consumption remained comparable across groups, with no differences observed during recovery.
Local Tolerability (Injection Site Reactions)
Local tolerability was acceptable, with dose-related but reversible findings. During in-life observations, injection sites showed only mild, transient swelling without persistent irritation. At necropsy after 2 weeks of dosing, repeated BB106 injections produced subcutaneous discoloration (red to dark areas) occasionally accompanied by slight thickening, swelling, or scab formation. These changes occurred more frequently in treated groups (≥4.8 mg/day) than in vehicle or saline controls. In contrast, single-use sites (on Day 1 only, examined after 14 days of healing) showed no treatment-related, aside from 1 small dark focus in a low-dose female. Following the 14-day recovery phase, no macroscopic injection-site changes were observed in any group.
Ophthalmology and Other Clinical Examinations
Ophthalmic evaluations revealed no treatment-related effects. Week 2 ophthalmoscopy showed no ocular abnormalities in BB106-treated rats relative to baseline. Aside from the expected transient ataxia and mild injection-site swelling, no additional clinical signs or behavioral changes were observed.
Clinical Pathology
Hematology and Coagulation
Minimal changes were observed. Reticulocyte counts increased slightly in BB106-treated rats at ≥9.6 mg/day (∼17–31% above controls in males, smaller increases in females), without associated alterations in red cell indices or signs of anemia. Values normalized after the 2-week recovery period. No treatment-related changes occurred in white cell or platelet parameters, and coagulation values remained within normal ranges.
Clinical Chemistry and Urinalysis
No adverse effects were detected. A modest non-dose-proportional increase in serum cholesterol was noted at the high dose in both sexes (+42% in males, +17–19% in females), but values remained within physiologically acceptable limits and returned to baseline during recovery. Minor shifts in other parameters (eg, protein, urea, electrolytes, and calcium) were small, within normal ranges, and considered incidental. Urinalysis showed no treatment-related changes in renal function or composition; an isolated increase in urine volume in mid-dose males was deemed incidental. No abnormalities persisted into the recovery phase.
Toxicokinetics
Ketamine Exposure
Repeated SC dosing of BB106 produced substantial systemic ketamine exposure in all treated rats, while plasma levels remained undetectable in controls (Supp Figure 4). Ketamine was rapidly absorbed, with peak concentrations (Tmax) typically at ∼0.25 h post-dose. Exposure increased with dose, although increases in Cmax and AUC_0–t_last were less than dose-proportional (eg, Day 14 mean Cmax ∼0.5–1.5 μg/mL across low to high doses). Plasma clearance was rapid (t½ ∼0.5–1 h), and no meaningful accumulation occurred with thrice-daily dosing (Day 14/Day 1 accumulation ratios ∼1.0–1.2). Thus, ketamine was present in circulation for ∼12 h of each 24-hour period.
Norketamine Exposure
The active metabolite norketamine was detectable in all treated animals but absent in controls (Supp Figure 5). Tmax occurred around 0.5 h, reflecting rapid metabolism of ketamine. Exposure increased with BB106 dose in both sexes, though not strictly dose-proportional. On Day 1, mean Cmax in males rose from ∼0.43 μg/mL at 4.8 mg/day to ∼0.71 μg/mL at 14.4 mg/day; in females, from ∼0.77 μg/mL to ∼2.7 μg/mL. As with ketamine, no accumulation was observed (Day 14/Day 1 AUC ratios near or below unity). Notably, females showed 2–3-fold higher norketamine AUC at the high dose compared with males, attributable to both higher mg/kg exposure (64 vs 41 mg/kg/day) and possible sex-related differences in metabolism. In contrast, parent ketamine exposure was similar between sexes after normalization for body weight.
Together, these results demonstrate that BB106 provides dose-dependent systemic exposure, rapid conversion to norketamine, and no evidence of accumulation, with sex-specific differences most evident in norketamine pharmacokinetics.
Gross Necropsy and Organ Weights
At the end of the 2-week dosing phase, no treatment-related gross lesions were observed in internal organs; incidental findings (eg, enlarged pituitary or adrenal) occurred with similar frequency in controls. Terminal body weights were slightly lower in treated males but unaffected in females. Some organ weight differences were noted, including higher mean brain, adrenal, and pituitary weights, lower prostate weight in BB106-treated males, and reduced thymus weight in mid-dose females. These changes were moderate in magnitude (eg, adrenal +20%, thymus −18% vs controls) and unaccompanied by histopathological abnormalities, indicating they were incidental and not treatment-related. All other absolute and relative organ weights were within expected ranges.
Following the 14-day recovery phase, body and organ weights were generally comparable between treated and control animals. Minor residual differences were observed in high-dose groups, with slightly heavier adrenals in males (+19–23%) and lighter thymus weights (up to −18% vs controls), but without microscopic correlates. These small changes were considered biologically insignificant, reflecting normal variability or reversible responses.
Histopathology
Injection Sites
BB106 produced localized, minimal to moderate inflammatory changes characterized by mononuclear cell infiltrates, occasional multinucleated giant cells amid fibroplasia (new blood vessels, fibroblast proliferation, and collagen deposition). These lesions often included focal fat necrosis with loss of adipocytes, hemorrhage with vascular congestion or fibrin deposition, and occasional small cavity formations. Lesions were slightly more frequent and severe in females.
In contrast, SBECD vehicle sites showed only minimal subacute inflammation and necrosis, comparable to saline controls, and resolved fully after 14 days, confirming good vehicle tolerability.
The severity of BB106-related changes was inversely correlated with healing time: lesions were most pronounced at sites sampled 1–5 days post-injection and milder at sites with longer recovery intervals. Repeated injections at the same site did not exacerbate injury relative to single injections of similar recovery time (Supp Figure 6A–F, Supp Figure 7). By the end of the 2-week recovery phase, most sites were fully healed, with only rare minimal residual chronic inflammation in high-dose animals (Supp Figure 6G–L, Supp Figure 8).
Systemic Tissues
Microscopic examination of all major organs revealed no treatment-related lesions. Findings in heart, liver, kidneys, spleen, lungs, GI tract, brain, and other tissues were within the expected spontaneous background range for Sprague-Dawley rats. No dose-dependent or treatment-specific histopathology was observed, and systemic tissues from recovery animals were indistinguishable from controls.
4-Week Continuous SC Infusion Study in Göttingen Minipigs (2-Week Recovery)
Mortality and Clinical Observations
All minipigs completed the 28-day continuous SC infusion and 2-week recovery without mortality or early euthanasia. Clinical monitoring revealed no treatment-related abnormalities. No sedation-like effects (eg, ataxia or depression) were observed. A single episode of emesis in 1 high-dose male was considered incidental. Mild dorsal erythema and occasional neck swelling occurred sporadically across groups, including controls, and were attributed to jacket-related skin irritation rather than BB106. During recovery, all animals remained healthy with no new or persistent clinical signs.
Body Weight and Food Consumption
Continuous SC infusion of BB106 had no effect on body weight or food intake in minipigs. Mean body weights in treated groups paralleled controls throughout the 4-week dosing period, with normal weight gain observed in all individuals. Food consumption was consistent across groups, with only minor day-to-day variation within expected ranges. During the 2-week recovery, feeding behavior remained normal, and previously treated animals showed a slight rebound in weight gain, comparable to controls.
Local Tolerability (Infusion Site Findings)
In-Life Observations
Continuous SC infusion of BB106 and SBECD was well-tolerated, with only minimal, transient local reactions. Slight edema (score 1 on a 0–4 scale) or very mild erythema was observed sporadically in a few animals, including controls, without dose dependence. These findings were short-lived, consistent with minor mechanical irritation from the infusion set or bandaging, and no severe local reactions (eg, ulceration, bleeding, and marked swelling) occurred. During recovery, all sites appeared normal on external inspection.
Gross Pathology
At necropsy, infusion sites from all groups occasionally showed mild subcutaneous discoloration at cannula entry points, more frequent in BB106-treated animals than in controls. Lesions corresponded to small hematomas or localized inflammation and never progressed to necrosis or ulceration. Healing correlated with time since last use: sites with >1 week of recovery appeared largely normal, while recently used sites (1–4 days) showed more visible discoloration. After the 14-day recovery phase, most sites in BB106-treated animals appeared normal, with only faint residual discoloration in a few high-dose animals. No abnormalities remained in vehicle-control recovery groups.
Ophthalmology, ECG, and Other Examinations
Comprehensive ophthalmic and cardiovascular monitoring revealed no adverse effects of BB106. Ophthalmologic examinations conducted pre-study and during Week 4 detected no treatment-related abnormalities, and ocular health remained normal in all animals. Cardiovascular function assessed by ECG on Day 1 and Week 4 showed normal heart rate and rhythm, with no drug-related changes in PR, QRS, or QT intervals. Occasional findings such as transient tachycardia, bradycardia, electrical alternans, or high-amplitude T-waves occurred sporadically across groups without dose dependence and were attributed to normal variability or handling stress.
Routine clinical assessments likewise showed no treatment-related effects. Body temperature, respiratory rate, and other vital signs remained normal throughout dosing and recovery. Minor dermatologic findings, such as mild erythema or superficial jacket-related skin wounds, occurred in both treated and control animals and were unrelated to BB106. No abnormalities in gait, behavior, or neurological function were observed. Overall, 28 days of continuous SC infusion of BB106 was well-tolerated, with no systemic clinical effects detected in minipigs.
Clinical Pathology
BB106 produced no treatment-related changes in hematology, clinical chemistry, or urinalysis.
Hematology
Hematologic values, including red and white cell counts, hemoglobin, platelets, and coagulation times, remained within normal ranges and showed no consistent dose-related trends. Sporadic differences from controls (eg, minor shifts in hemoglobin in some females) were small, lacked dose relationship, were not supported by clinical signs or other lab findings, and were therefore considered incidental. No evidence of bone marrow toxicity or hemolysis was observed, and all values normalized after recovery.
Clinical Chemistry
Serum chemistry profiles were unremarkable, with liver and kidney function markers, electrolytes, glucose, and lipids comparable across groups. No dose-related enzyme elevations or abnormalities in protein levels occurred, and all values remained within reference ranges. Post-recovery, treated animals were indistinguishable from controls.
Urinalysis
Urinalysis likewise revealed no treatment-related findings. Specific gravity, pH, and qualitative parameters (presence/absence of glucose, protein, blood, or ketones) were comparable across groups, and quantitative assessments (urine volume, electrolyte excretion) showed no changes suggestive of renal effects. Overall, clinical pathology evaluations confirmed that continuous SC infusion of BB106 at doses up to 3.6 mL/day (≈253 mg ketamine base/day) did not produce systemic toxicity in minipigs.
Toxicokinetics
Ketamine Plasma Levels
Continuous SC infusion of BB106 produced clear, dose-dependent systemic exposure. As expected, ketamine was undetectable in controls aside from 1 trace-contaminated sample. In treated animals, ketamine was measurable at all sampling times on Days 1 and 28, with plasma concentrations maintained throughout the infusion interval. On Day 1, mean Cmax values in males were ∼36, 93, and 192 ng/mL at low, mid, and high doses, respectively; females showed similar exposure (41, 115, and 138 ng/mL). Corresponding AUC_0–24h values increased with dose, though the rise from low to mid dose was slightly over-proportional. By Day 28, near steady-state was achieved with no meaningful accumulation (Day 28/Day 1 AUC ratios ∼0.8–1.1). High-dose males, for example, had a mean Cmax of ∼166 ng/mL and AUC_0–24h of ∼2738 ng*h/mL, nearly identical to Day 1. Overall, ketamine exposure was dose-dependent, consistent across sexes, and maintained at steady levels under continuous infusion.
Norketamine Plasma Levels
The primary metabolite norketamine was also detected in all BB106 groups, with dose-dependent exposure and marked sex differences (males > females). On Day 1, mean Cmax in males ranged from ∼83 to 286 ng/mL (low to high dose), compared with ∼17 to 63 ng/mL in females. This disparity persisted on Day 28, where high-dose males reached ∼246 ng/mL vs ∼60 ng/mL in females. AUC values showed a similar pattern: Day 28 AUC_0–24h for high-dose males was ∼3874 ng*h/mL, about 6–7 times higher than females (∼985 ng*h/mL). Despite these differences in magnitude, kinetics were otherwise similar between sexes, with both achieving near steady-state by Day 28 and showing little to no accumulation (Day 28/Day 1 AUC ratios ∼1.0–1.5). Norketamine exposure was broadly dose-proportional in females and slightly less than proportional at the highest dose in males.
In summary, BB106 infusion provided reliable, dose-dependent systemic exposure to ketamine and its metabolite. Ketamine pharmacokinetics were comparable between sexes, while norketamine exposure was markedly higher in males, suggesting a sex-related difference in metabolism or clearance. Both analytes reached steady-state without accumulation over the 28-day infusion period.
Gross Necropsy and Organ Weights
At the end of the 28 days of infusion, gross necropsy revealed no treatment-related abnormalities in systemic organs. All major viscera appeared normal, and occasional findings such as small foci of pallor in the liver or mild adrenal enlargement occurred with similar frequency in controls and were considered incidental. Terminal body weights were comparable across groups, confirming normal growth.
Organ weight evaluation identified a few statistically significant differences, but none were dose-dependent or associated with microscopic pathology. In males, adrenal and liver weights were slightly lower than controls at mid and high doses (by ∼23–26%), and thymus weights were reduced at low and mid doses. In females, adrenal weights were slightly higher at low and high doses (+22 to +37%), while thymus weights appeared increased due to 1 control with unusually low values. No consistent trends were observed for other organs, and kidneys, heart, spleen, and reproductive organs showed no differences from controls. Histopathology of the adrenals, liver, thymus, and other tissues revealed no lesions, confirming that organ weight variations reflected normal biological variability rather than BB106 treatment.
Recovery Necropsy (Week 6)
Following the 2-week recovery phase, no gross pathological changes were observed in systemic organs of high-dose animals, which were indistinguishable from recovery controls. High-dose pigs exhibited slightly higher terminal body weights (+10–14% vs controls), consistent with normal growth and overall good health.
Organ weight analysis identified several statistically significant differences between high-dose and control recovery animals; however, these changes lacked histopathological or clinical pathology correlates and showed opposite trends between sexes, arguing against treatment-related effects. In males, modest decreases (∼13–30%) were noted in the weights of the heart, kidneys, thymus, thyroid, spleen, and androgen-dependent organs (epididymides, prostate, testes). In females, adrenal and spleen weights were increased, while ovaries were slightly lighter (∼7–9%). Despite these differences, microscopic examination of all affected organs was unremarkable: thymic involution was age-appropriate, adrenal morphology was normal, and spleens appeared healthy. Clinical pathology results were likewise unremarkable.
Overall, the organ weight variations were considered to be incidental, reflecting biological variability and small group size rather than BB106 exposure. There was no evidence of persistent organ toxicity after 4 weeks of BB106 infusion followed by recovery.
Histopathology
Infusion Sites
Microscopic evaluation showed localized inflammatory and reparative changes consistent with wound-healing responses to SC infusion. Saline controls exhibited minimal alterations, while vehicle controls displayed minor inflammation, indicating slight irritancy of the excipient. BB106-treated sites demonstrated minimal to moderate subcutaneous lesions, including mononuclear and multinucleated giant cell infiltrates, new capillary formation, fibroblast proliferation, collagen deposition, adipocyte necrosis, fibrin deposition, hemorrhage, and small cavities from resolving necrosis—typical of a foreign-body or irritant reaction. Lesions were slightly more pronounced in females but similar in character across sexes (Figures 1 and 2). Representative histopathological findings at subcutaneous infusion sites in minipigs. (A) Female animal, Group 1, saline control, Site 2R—single infusion on Day 25; examined after 3–4 days of recovery. Tissue was histologically unremarkable (H, E). (B) Female animal, Group 1, saline control, Site 1L—repeated infusions on Days 2 and 15; examined after 13–14 days of recovery. No histopathological abnormalities observed (H, E). (C) Female animal, Group 1, saline control, Site 8L—Single infusion on Day 14; examined after 14–15 days of recovery. Tissue is unremarkable (H, E). (D) Female animal, Group 2, SBECD (vehicle), Site 2R—single infusion on Day 25; examined after 3–4 days of recovery. Minimal subchronic inflammation (mononuclear cell infiltrate), minimal subcutis necrosis, and minimal hemorrhage were observed. Arrow indicates lesion site at low magnification; inset: higher magnification (H, E). (E) Female animal, Group 2, SBECD (vehicle), Site 1L—repeated infusions on Days 2 and 15; examined after 13–14 days of recovery. Minimal subchronic inflammation with mononuclear and multinucleated cell infiltrate, extending into the dermis and likely related to needle tract. Minimal subcutis necrosis also present. Arrow indicates lesion site; inset: higher magnification (H, E). (F) Female animal, Group 2, SBECD (vehicle), Site 8L—single infusion on Day 14; examined after 14–15 days of recovery. Tissue is histologically unremarkable (H, E). (G) Female animal, SBECD (vehicle), recovery group, Site 2R—single infusion on Day 25; examined after 17-day recovery. Tissue was histologically unremarkable (H, E). (H) Female animal, SBECD (vehicle), recovery group, Site 1L—repeated infusions on Days 2 and 15; examined after 27-day recovery. Minimal subchronic inflammation (mononuclear and multinucleated cells) and minimal subcutis pigmentation (yellow/brown). Arrow indicates lesion site; inset: higher magnification (H, E). (I) Female animal, SBECD (vehicle), recovery group, Site 8L—single infusion on Day 14; examined after 28-day recovery. Tissue is unremarkable (H, E). (J) Female animal, BB106 (0.90 mL/day; 38 μL/h), Site 2R—single infusion on Day 25; examined after 3–4 days of recovery. Minimal subcutis necrosis and minimal hemorrhage observed. Arrow indicates lesion site; inset: higher magnification (H, E). (K) Female animal, BB106 (0.90 mL/day; 38 μL/h), Site 1L—repeated infusions on Days 2 and 15; examined after 13–14 days of recovery. Minimal subchronic inflammation with mononuclear cell infiltrate. Arrow marks the lesion; inset: higher magnification (H, E). (L) Female animal, BB106 (0.90 mL/day; 38 μL/h), Site 8L—single infusion on Day 14; examined after 14–15 days of recovery. Tissue was histologically unremarkable (H, E). (M) Female animal, BB106 1.8 mL/day (75 μL/h), Site 1L—repeated infusions on Days 2 and 15; examined after 13–14 days of recovery. Mild subchronic inflammation with mononuclear and multinucleated cell infiltrate. Arrow indicates lesion site; inset: higher magnification (H, E). (N) Female animal, BB106 1.8 mL/day (75 μL/h), Site 8L—single infusion on Day 14; examined after 14–15 days of recovery. Mild subchronic inflammation with mononuclear and multinucleated cell infiltrate. Arrow indicates lesion site; inset: higher magnification (H, E). (O) Female animal, BB106 3.6 mL/day (150 μL/h), Site 2R—single infusion on Day 25; examined after 3–4 days of recovery. Minimal subchronic inflammation (mononuclear cells), moderate subcutis necrosis, minimal hemorrhage, and cavity formation. Arrow indicates lesion site; inset: higher magnification (H, E). (P) Female animal, BB106 3.6 mL/day (150 μL/h), Site 1L—repeated infusions on Days 2 and 15; examined after 13–14 days of recovery. Mild subchronic inflammation with mononuclear and multinucleated cell infiltrate. Arrow indicates lesion site; inset: higher magnification (H, E). (Q) Female animal, BB106 3.6 mL/day (150 μL/h), Site 8L—single infusion on Day 14; examined after 14–15 days of recovery. Mild subchronic inflammation, minimal necrosis, and minimal hemorrhage. Arrow indicates lesion site; inset: higher magnification (H, E). (R) Female animal, BB106 3.6 mL/day (150 μL/h), recovery group, Site 2R—Single infusion on Day 25; examined after 17-day recovery. Mild chronic inflammation (mononuclear and multinucleated cells), minimal hemorrhage, with associated fibroblast proliferation and collagen deposition. Arrow marks lesion area; inset: higher magnification (H, E). (S) Female animal, BB106 3.6 mL/day (150 μL/h), recovery group, Site 1L—repeated infusions on Days 2 and 15; examined after 27-day recovery. Mild chronic inflammation characterized by mononuclear and multinucleated cell infiltrate with fibroblast proliferation and collagen deposition. Arrow indicates lesion site; inset: higher magnification (H, E). (T) Female animal, BB106 3.6 mL/day (150 μL/h), recovery group, Site 8L—single infusion on Day 14; examined after 28-day recovery. Tissue was histologically unremarkable (H, E). Incidence and severity of histopathological findings at subcutaneous infusion sites in male and female minipigs at terminal necropsy. Bar graphs summarizing the incidence and severity of local tissue reactions following 28 days of continuous subcutaneous infusion of saline, SBECD vehicle, or BB106 at increasing doses. Documented lesions include subchronic inflammation, subcutis necrosis, hemorrhage, and cavity formation. Data are stratified by sex (males and females) and treatment group, and severity was graded according to standardized toxicologic pathology criteria.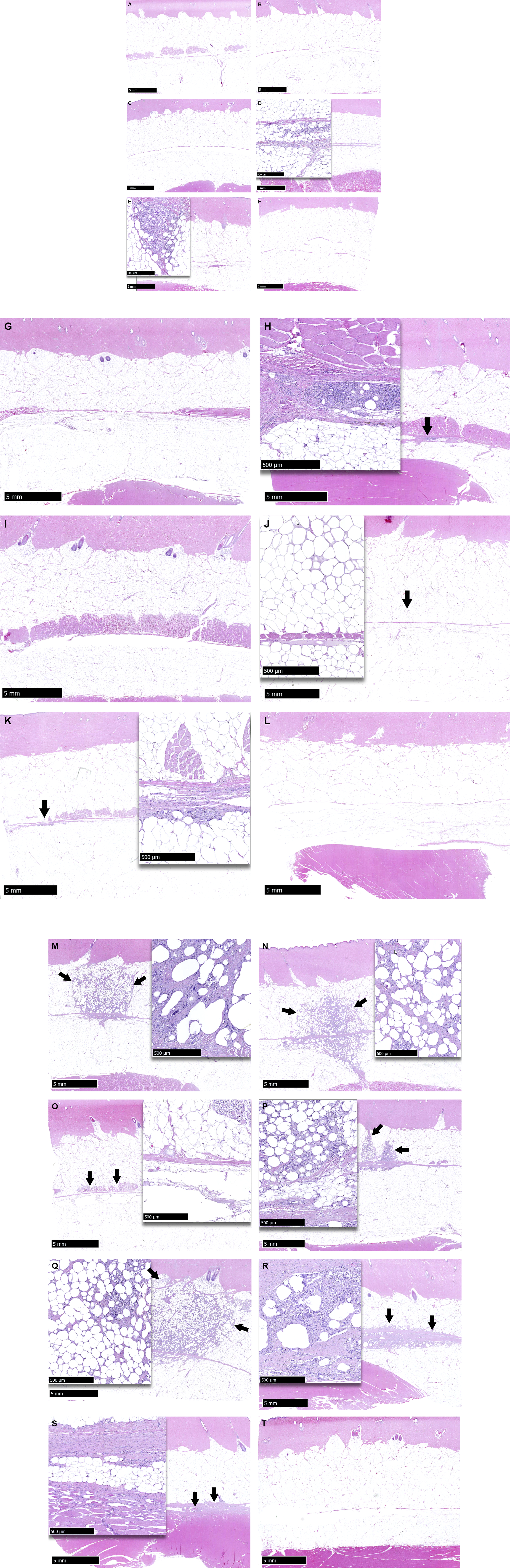
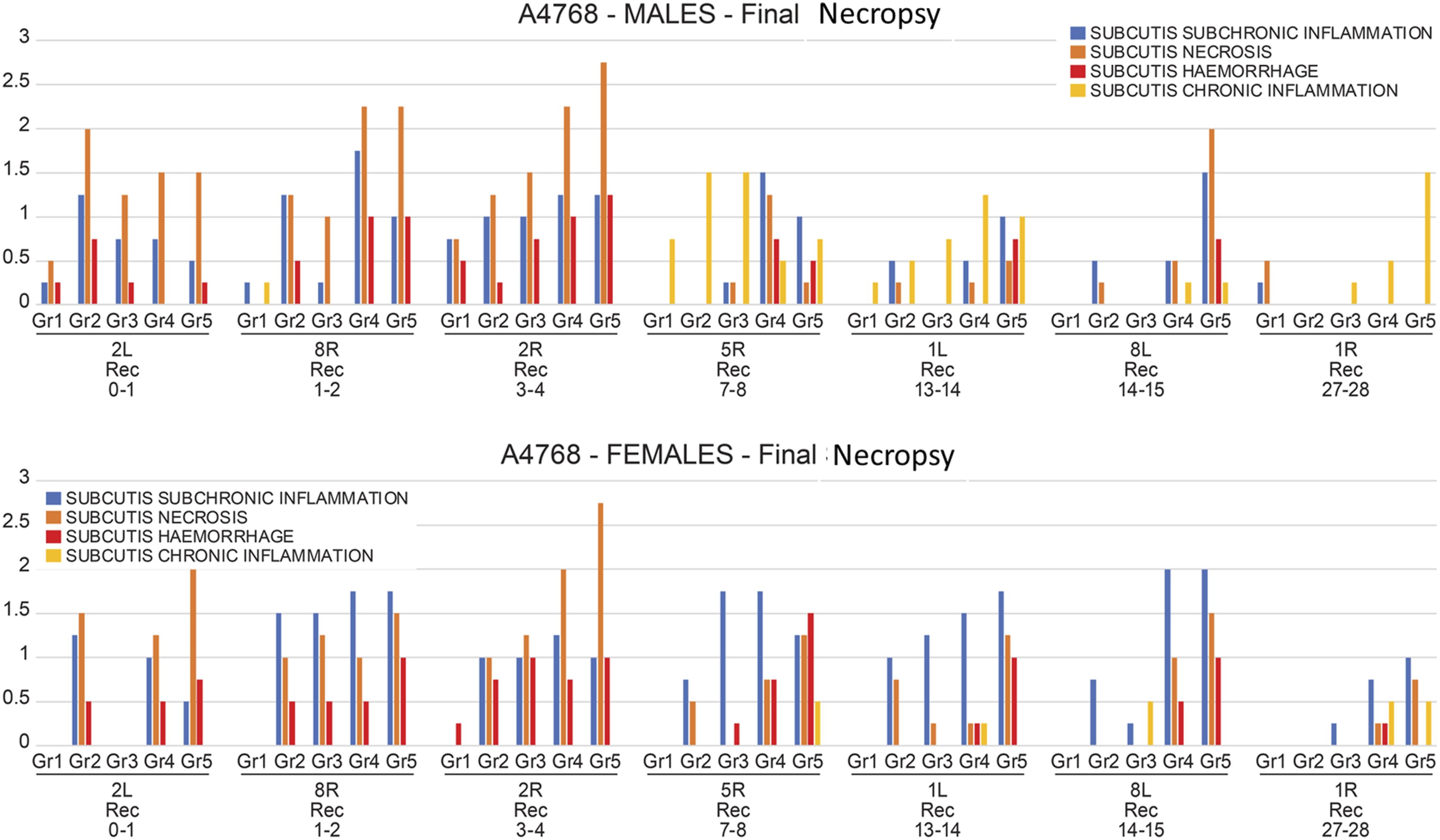
The extent of change correlated inversely with healing time. Sites examined within 1–4 days of last infusion showed the most acute inflammation and necrosis, whereas sites with 1–2 weeks of recovery exhibited primarily chronic inflammation and fibrosis. Sites with 3–4 weeks of recovery were often histologically normal or showed only minimal fibrosis, especially at lower doses. Importantly, multiple infusions into the same site did not exacerbate injury compared with single infusions when recovery intervals were adequate. Dose effects were evident, with higher incidence and severity of inflammation at the high dose compared to vehicle or saline.
By the end of the 2-week recovery phase, most sites across all dose groups showed either complete resolution or only minimal residual inflammation (eg, scattered macrophages or fibrous tissue). Rare residual findings in high-dose animals were mild and limited to sites with the shortest recovery period. No necrosis or severe inflammation persisted. These observations confirm that BB106-related local tissue changes were largely reversible within 2 weeks (Figure 3). Incidence and severity of histopathological changes at subcutaneous infusion sites in male and female minipigs at recovery necropsy. Bar graph summarizing local tissue findings in male and female Göttingen minipigs after a 14-day recovery period following 28 days of continuous subcutaneous infusion with SBECD vehicle or BB106. Histopathological changes include chronic inflammation, necrosis, hemorrhage, and fibrosis-related alterations such as fibroblast proliferation and collagen deposition. Severity grading was based on standard toxicologic pathology criteria. The data reflect reversibility of local effects post-infusion and the residual tissue response at recovery necropsy.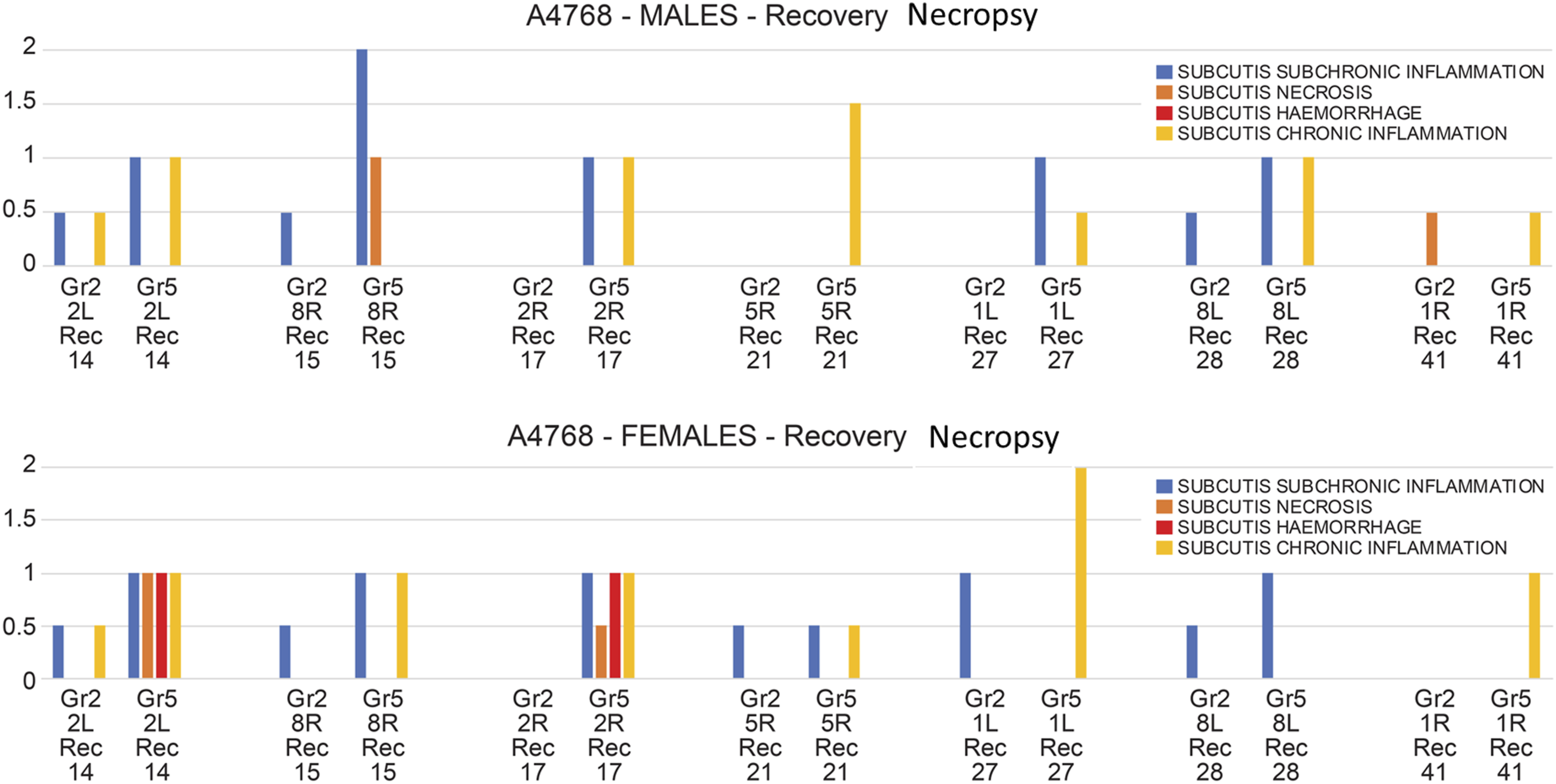
Systemic Tissues
No treatment-related histopathological findings were observed in systemic organs. After 28 days of infusion, all major organs (heart, liver, kidneys, lungs, spleen, GI tract, brain, endocrine, and reproductive organs) appeared normal and indistinguishable from controls. Incidental findings were consistent with background pathology for Göttingen minipigs. No evidence of ketamine-related organ toxicity was present at any dose. After the 14-day recovery period, systemic tissues in high-dose animals remained unremarkable.
In summary, BB106 produced localized, dose-related but reversible subcutaneous reactions at infusion sites, with no systemic organ pathology at infusion volumes up to 3.6 mL/day.
Discussion
These nonclinical studies demonstrate that BB106, a novel ketamine–SBECD salt formulation for subcutaneous delivery, has a favorable toxicological profile. Repeated SC injections in rats and continuous SC infusion in minipigs were well-tolerated, with no evidence of dose-limiting systemic toxicity. Adverse findings were limited to expected ketamine-related pharmacologic effects—such as transient CNS depression (lethargy or ataxia at peak exposure) in rats—and did not involve organ-specific injury. Minipigs receiving BB106 by continuous infusion, which maintained steady plasma levels, showed no pharmacologic signs. Importantly, no histopathological lesions were identified in major organs, and local injection-site changes were generally mild, reversible, and resolved during recovery. Collectively, these findings indicate that BB106 does not induce toxicities beyond those already known for ketamine and support its advancement into clinical development. Fixed daily dosing, necessitated by the small injection/infusion volumes of the formulation, translated to exposure ranges of ∼13.7–64 mg/kg/day in rats and 2.4–11.3 mg/kg/day in minipigs.
A central finding of these studies was the improved local tolerability of BB106 relative to conventional ketamine HCl solutions, which are strongly acidic (pH ∼ 3.88–4.18) and hyperosmotic at clinical concentrations (740.1 mOsm/kg at 100 mg/mL). 1 In contrast, BB106—leveraging SBECD to maintain near-physiologic pH and isotonicity—produced only minimal, transient injection-site reactions. This aligns with formulation principles recommending neutral pH and ∼300 mOsm/kg for injectable solutions to minimize pain and injection-site injury.17,18 Histopathology confirmed only mild, reversible inflammatory changes without necrosis or abscess formation, underscoring the translational potential of this approach.
Systemically, no novel toxicities emerged. In particular, we observed no evidence of urinary tract toxicity, a known risk with chronic exposure in humans and long-term rodent studies.23,24 Minipigs infused for 4 weeks showed normal urinalysis and no bladder or renal lesions. Likewise, no hepatobiliary changes were detected, despite ketamine’s occasional association with liver injury in clinical and abuse contexts. 25 The absence of renal and liver findings at therapeutic exposures suggests that BB106 is not overtly toxic to these systems, though monitoring in clinical trials remains warranted. Neurotoxicity was also not evident: detailed brain histology revealed no neuronal necrosis, vacuolization, or degenerative changes, despite historical concerns about NMDA antagonist-related “Olney’s lesions” in rats under high-dose acute conditions. 26 Only transient, reversible neurological signs were observed, supporting the view that subanesthetic SC dosing via BB106 does not confer significant neurotoxic hazard.
The novel incorporation of sulfobutylether-beta-cyclodextrin as a multivalent counterion to reduce osmolality of solution by approximately 45% 1 was pivotal to BB106’s performance. This design enabled a stable, high-concentration ketamine solution that remained isotonic and near-neutral in pH, thereby reducing local irritation while ensuring reliable systemic exposure. In rats, small-volume injections of BB106 yielded consistent ketamine plasma levels, aided by precision dosing techniques. In minipigs, continuous infusion produced stable systemic concentrations without erratic absorption or depot formation, reinforcing the feasibility of portable pump-based delivery. These results underscore BB106’s translational potential: the SC route is known to provide high bioavailability, and the SBECD excipient itself has a well-established safety profile, with adverse effects only reported at doses far exceeding those used here. 27 Consistent with this, no cyclodextrin-related tissue changes were observed, confirming its suitability for subchronic SC administration.
From a clinical standpoint, even histologically mild lesions could manifest as discomfort in patients (eg, burning, itching, or tenderness) potentially affecting adherence. Thus, local tolerability in animals should be interpreted with attention to human sensory correlates. Site rotation, cannula optimization, and formulation refinement may mitigate these risks. Encouragingly, our results indicate that BB106 substantially reduces the likelihood of such reactions, supporting its use in both intermittent SC bolus injections and prolonged infusions. Importantly, continuous infusion provides steady-state plasma levels without the peaks and troughs of bolus dosing, avoids the need for IV access, and may lower risks of misuse or diversion—advantages particularly relevant for chronic pain or psychiatric applications. Clinical studies have already shown efficacy of SC ketamine bolus dosing in treatment-resistant depression,28,29 but BB106 is the first formulation specifically designed for safe continuous SC infusion, addressing an unmet need for sustained, well-tolerated delivery.
The selection of dosing regimens in this program was tailored to the physiological and technical constraints of each species. While the minipig model utilized continuous subcutaneous infusion—closely mirroring the intended clinical delivery method via ambulatory pumps—technical limitations in the rat model necessitated a different approach. Continuous infusion in rats often requires tethering, which can introduce stress-related artifacts. Therefore, a fractionated dosing regimen (3 injections daily) was employed in rats to achieve high aggregate daily exposure and simulate sustained plasma levels, ensuring a rigorous safety evaluation in a second species.
A distinct sex difference in systemic exposure was observed in the rat study, where males exhibited lower plasma concentrations of ketamine but higher concentrations of its major metabolite, norketamine, compared to females. This finding is attributable to the known sexual dimorphism in rodent hepatic metabolism. 30 Male rats possess significantly higher activity of specific cytochrome P450 enzymes (particularly CYP2C11 and CYP3A2) responsible for the N-demethylation of ketamine. 31 Consequently, males metabolize the parent drug more rapidly, resulting in a higher formation rate of norketamine and lower systemic bioavailability of the parent compound relative to females. This metabolic disparity is a characteristic feature of the rat model and does not necessarily predict a similar gender effect in humans, where such pronounced hepatic dimorphism is not typically observed.
These encouraging data should be interpreted in light of several limitations. Exposure durations were subchronic (2 weeks in rats, 4 weeks in minipigs, plus recovery), so longer-term toxicities cannot be excluded. However, the studies followed ICH M3(R2) guidelines and are appropriate to support short-term human use (<2 weeks), which is the intended therapeutic window. The planned first-in-human trial will last only 5 days, well within the exposure range tested. Species differences remain a consideration: while rats and minipigs are standard models, their metabolism and SC tissue responses may not fully mirror humans. Nonetheless, group sizes met or exceeded regulatory requirements, and consistent findings across preliminary and pivotal studies reinforce robustness of the conclusions.
Conclusion
Together, these data show that BB106 combines pharmacologic efficacy, local tolerability, and formulation stability into a patient-friendly SC delivery option for ketamine. Both rodent and non-rodent studies confirmed the absence of dose-limiting systemic toxicity. Comprehensive evaluation of safety parameters—including clinical observations, body weight, food consumption, ophthalmoscopy, electrocardiography (in minipigs), clinical pathology (hematology, coagulation, clinical chemistry, and urinalysis), and organ weights—revealed no adverse treatment-related effects. Local tolerability was characterized by only mild, reversible inflammatory changes at the infusion sites. The reproducibility of findings across models and study designs supports the suitability of BB106 for short-term SC administration in humans, including via wearable infusion pumps. By mitigating the local tolerability challenges of conventional ketamine formulations and avoiding new systemic toxicities, BB106 offers a strong foundation for clinical development in pain, depression, and related neuropsychiatric conditions.
Supplemental Material
Supplemental Material - Preclinical Safety Evaluation of BB106, a Novel Ketamine–Sulfobutylether-Beta-Cyclodextrin Salt, Following Subcutaneous Administration in Rats and Göttingen Minipigs
Supplemental Material for Preclinical Safety Evaluation of BB106, a Novel Ketamine–Sulfobutylether-Beta-Cyclodextrin Salt, Following Subcutaneous Administration in Rats and Göttingen Minipigs by Yuval Ramot, Isabella Andreini, Giuseppe Sacco, Jeffrey Becker, Kevin Bruhn, Rosa Anna Manno, and Abraham Nyska in International Journal of Toxicology
Footnotes
Acknowledgments
The authors wish to acknowledge the use of ChatGPT for assistance with minor proofreading of the manuscript.
Author Contributions
Ramot, Y. contributed to interpretation and drafted manuscript; Andreini, I. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Sacco, G. contributed to acquisition, analysis, and interpretation; Becker, J. contributed to acquisition, analysis, and interpretation; Bruhn, K. contributed to conception and design and contributed to interpretation; Manno, R.A. contributed to conception and design and contributed to analysis and interpretation; Nyksa, A. contributed to conception and design, contributed to analysis and interpretation, and drafted manuscript. All authors critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jeffrey Becker is a current employee of Bexson Biomedical, Santa Barbara, CA, USA. Kevin Bruhn was an employee of Bexson Biomedical, Santa Barbara, CA, USA. No other co-authors have any conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by Bexson Biomedical, Santa Barbara, CA, USA.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
