Abstract
A 90-day study in Göttingen minipigs was conducted to test the local tolerability and systemic toxicity of ND0612, a novel aqueous solution of carbidopa (CD)/levodopa (LD) intended for the treatment of Parkinson’s disease by continuous subcutaneous administration using a discrete infusion pump. To evaluate tissue site reactions, we used a unique study design involving multiple infusion sites to evaluate the effect of dose per site (270/63, 360/45, and 360/84 mg LD/CD), volume of infusion per site (4.5 and 6 ml per site), formulation concentration (60/14 and 60/7.5 mg/ml LD/CD), daily rate of infusion per site (240 μl/hr for16 hr and 80 μl/hr for 8 hr, 320 μl/hr for 16 hr and 100 μl/hr for 8 hr, or 750 μl/hr for 8 hr), frequency (once every 5, 10, 15, or 20 days), and number of infusions (4, 6, or 9) to the same infusion site. No systemic adverse effects were observed. Histopathological changes at infusion sites started with localized minimal necrosis and acute inflammation that progressed to subacute and chronic inflammatory and reparative changes with evidence of progressive recovery following the final infusion. None of the infusion site effects were judged to be adverse, and clinical exposures to ND0612 are not expected to result in adverse responses.
Keywords
Introduction
Parkinson’s disease (PD) is considered to be one of the most common neurodegenerative diseases, affecting 1% of the people over the age of 65 (Reeve, Simcox, and Turnbull 2014). PD is associated with loss of dopaminergic neurons in the substantia nigra (Han et al. 2015), leading to both motor and nonmotor dysfunction (Choi et al. 2017).
Dopamine receptor agonists are used as the mainstay of treatment for PD. Although considered the gold standard for treatment of PD, long-term use of levodopa (LD) is associated with motor fluctuations and drug-induced dyskinesia. Such complications are related to the pulsatile drug delivery of LD due to its short half-life and irregular gastrointestinal absorption (Poewe and Antonini 2015). Improving the pharmacokinetics (PK) of LD is an important medical need, and several approaches have been utilized to achieve this goal. These include the incorporation of enzyme inhibitors that are responsible for the metabolism of LD and the use of nonoral delivery methods (Poewe and Antonini 2015). However, LD is poorly soluble, a fact that has hindered the development of effective formulations for subcutaneous (SC) delivery.
ND0612 is a novel liquid formulation of LD/carbidopa (CD), intended for continuous SC administration. Preliminary results from small studies in humans revealed that continuous SC administration of ND0612 resulted in more constant plasma concentrations of LD and good tolerability (Poewe and Antonini 2015; LeWitt 2016; Lotia and Jankovic 2016). The purpose of the current study was to assess the local and systemic toxicity of ND0612 following 90 days repeated continuous SC infusion in Göttingen minipigs for up to 90 days. The study was designed to support the envisioned clinical use of ND0612 as well as unplanned overexposure. Since infusion site reactions are common during SC administration of drugs, and can sometimes lead to termination of treatment (Gold et al. 2005; Ramot et al. 2012), and in order to gain better insight on the expected histopathological changes following repeated continuous SC infusion of multiple ND0612 treatment options, we used a sophisticated experimental design to evaluate the effect of dose, rate, volume, frequency, and concentration of test agent infusions (Table 1) based on the intended clinical use (Table 2).
Study Design.
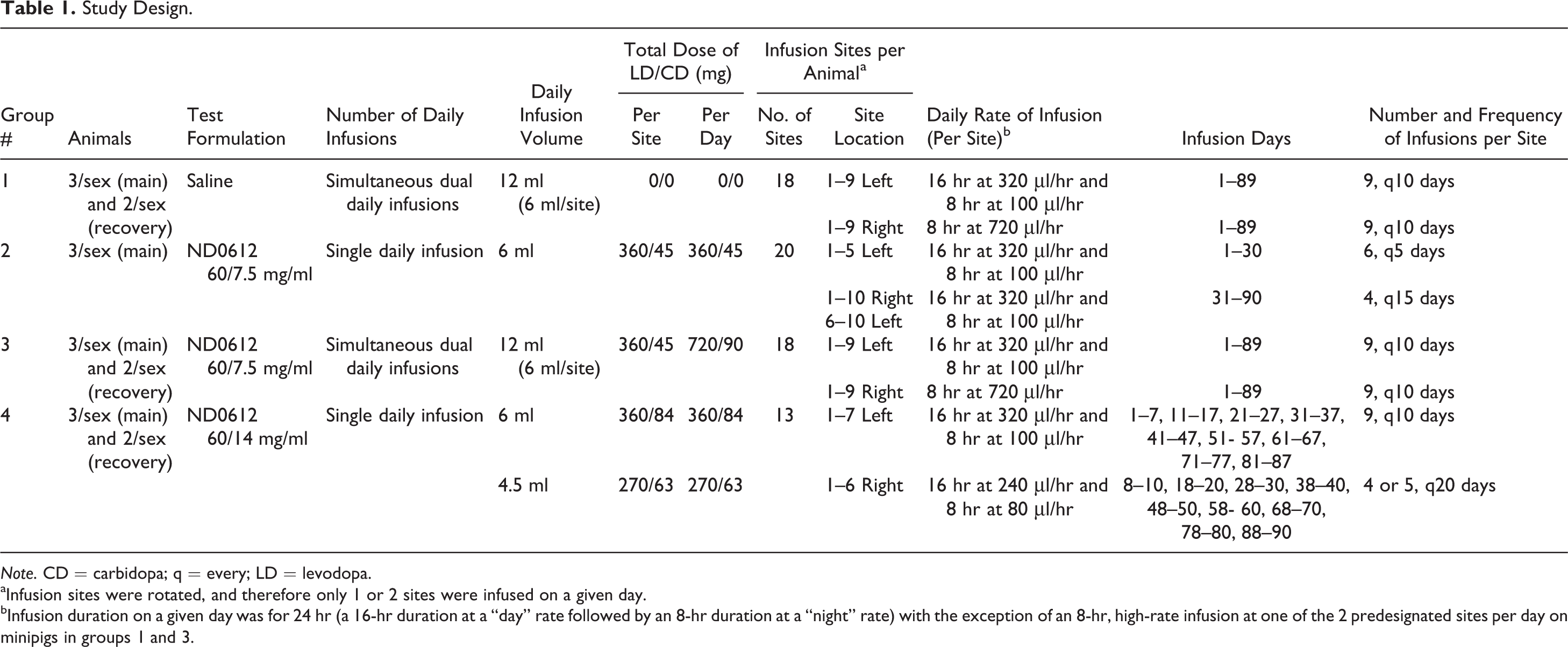
Note. CD = carbidopa; q = every; LD = levodopa.
aInfusion sites were rotated, and therefore only 1 or 2 sites were infused on a given day.
bInfusion duration on a given day was for 24 hr (a 16-hr duration at a “day” rate followed by an 8-hr duration at a “night” rate) with the exception of an 8-hr, high-rate infusion at one of the 2 predesignated sites per day on minipigs in groups 1 and 3.
Intended Clinical Use of ND0612.
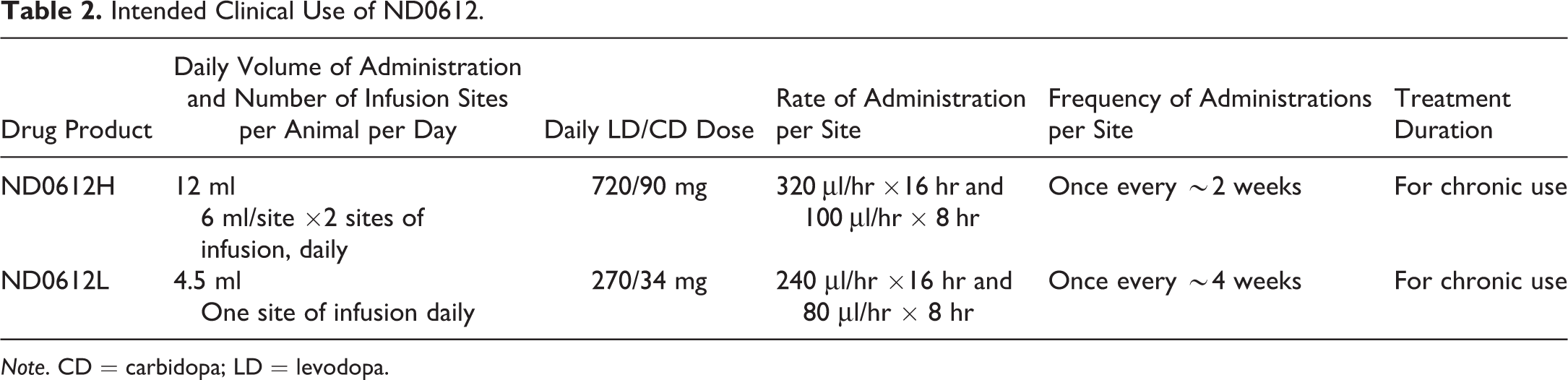
Note. CD = carbidopa; LD = levodopa.
By providing this information, we believe that this report can be of great importance for future preclinical pathological assessments of infusion and injection site reactions in minipigs while complying with the reduction, refinement, replacement (3Rs) guiding principles for more ethical use of animals in testing.
Materials and Methods
Animal Husbandry and Maintenance
A total of 18 male and 18 female Göttingen minipigs (Ellegaard Göttingen Minipigs, Dalmose, Denmark), approximately 9 to 10 months old, weighing 16 to 18 kg, participated in the study. The experiment was conducted at Research Toxicology Centre Spa (Via Tito Speri, 12/14, 00071 Pomezia, Italy). The animals were housed individually in a limited access animal facility. Room controls were set to maintain temperature and relative humidity at 19°C ± 3°C and 55% ± 15%, respectively. There was continuous air exchange, and the rooms were artificially lit for 12 hr each day, except during toxicokinetic (TK) blood sampling. The study animals were acclimatized for at least 4 weeks before treatment initiation.
Drinking water was supplied ad libitum to each enclosure via an automatic “lixit” valve system or water bottles. Each minipig was offered a predefined amount of diet (SMP (E) SQC, Special Diets Services, Witham Essex, England) daily, divided in 2 rations as suggested by the supplier to avoid excessive weight gain. The Göttingen minipig was chosen for this study based on physiologically similar cardiovascular, gastrointestinal, and notably integumentary systems to humans (Manno et al. 2016; Ramot et al. 2015; Vezzali et al. 2011).
The experimental protocol was approved by the ethical committee of the Research Toxicology Centre (Pomezia, Italy), and the study was conducted in accordance with the requirements of the EC Commission Directive 86/609/EEC. This experiment was conducted in facilities approved by the Association for Assessment and Accreditation of Laboratory Animal Care International, and the animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals (Institute for Laboratory Animal Research 1996).
Test Material and Treatment Protocol
The ND0612, the test material, was supplied by NeuroDerm Ltd. (Rehovot, Israel), as either 60/7.5 mg/ml LD/CD or 60/14 mg/ml LD/CD formulations. Sterile saline was used as placebo control.
The main features of the study design are as follows: formulation concentration: 60/7.5 mg/ml and 60/14 mg/ml (LD/CD); volume of infusion: 4.5 and 6 ml/site; rate of infusion: 240 μl/hr for 16 hr and 80 μl/hr for 8 hr, 320 μl/hr for 16 hr and 100 μl/hr for 8 hr, and 750 μl/hr for 8 hr; different intervals between reuse of infusion sites (once every 5, 10, 15, or 20 days); reversibility of effects 1, 2, 4, 5, and 9 weeks after cessation of infusions; and the TK profile of ND0612.
Table 1 summarizes the study design that is based on intended clinical use (Table 2). Each minipig had multiple sites designated in advance (Supplementary Figure 1). Different sites for continuous infusion were alternated daily using 1 site for groups 2 and 4 and 2 sites for groups 1 and 3. Infusion duration on a given day was for 24 hr (a 16-hr duration at a “day” rate followed by an 8-hr duration at a “night” rate) with the exception of an 8-hr, high-rate infusion at one of the 2 predesignated sites per day on minipigs in groups 1 and 3. The study design for recovery allowed for examination of specific sites from 1 up to 64 days following final infusions.
SC infusion was performed using an infusion pump (Crono-ND®, Canè Medical Technologies, Rivoli-Turino, Italy) connected to an infusion set (Accu-chek®, FlexLink, Roche, Indianapolis, IN, USA) with a percutaneous needle of 8 mm length. On each day of treatment, before the application of a new infusion set, the skin pertaining to the treatment site was cleaned, disinfected with alcohol, and accurately wiped with sterile gauze. An adhesive tape was used to protect and hold the infusion set at the injection site, and the infusion pump was placed in a protective pocket held in place by an elastic belt placed around the thorax behind the scapulae of the animal. A tubular elastic bandage was used to further secure the pump and infusion set. Volume of dose administered and the flow rate were recorded for each animal. At the end of a day’s infusion, following pump and infusion set removal, a gauze pad with iodine solution was applied to the site of administration and secured with an adhesive tape.
The treatment regimen of group 2, that is, reinfusion to the same site once every 5 days was planned to be as follows: sites #1 to 5 during days 1 to 30, sites #6 to 10 during days 31 to 60, and sites #11 to 15 during days 61 to 90. During the first month of treatment, 1 male animal exhibited severe erythema with eschar at the sites of infusion. Therefore, from day 31 of the study, animals in group 2 were subjected to a new treatment plan in which infusion sites were revisited once every 15 days (see Table 1).
All animals were dosed continuously for 89 or 90 consecutive days. Necropsy of the main groups was performed on days 90 (groups 1 and 3) to 91 (groups 2 and 4) of the study. Infusion sites were allowed different periods of recovery ranging between 1 and 64 days prior to their excision. Necropsy of the recovery groups 1, 3, and 4 was performed 30 days after the last infusion. All elements of the study design including total doses of LD/CD, infusion volumes, and number of repeat infusions per site are detailed in Table 1.
Clinical Observations and Mortality and Physical Examination
Throughout the study period, all animals were carefully observed twice daily for any signs of ill-health. Once before commencement of treatment and daily during the study (approximately 1 hr from the start of infusion), each animal was observed, and any clinical signs were recorded. Each day prior to refilling the infusion pump, treatment sites of all animals were examined for any local dermal reactions. Erythema and edema were compared to adjacent untreated skin and assigned a numerical value as follows: 0 = no erythema, 1 = very slight erythema, 2 = well-defined erythema, 3 = moderate-to-severe erythema, 4 = severe erythema to eschar formation preventing grading of erythema; 0 = no edema, 1 = very slight edema (barely perceptible), 2 = slight edema (edges of area well defined and raised), 3 = moderate edema (raised approximately 1 mm), 4 = severe edema (raised more than 1 mm and extending beyond the area of exposure; Draize, Woodard, and Calvery 1944). Any other dermal reactions (e.g., alopecia, erosions, cracking, scabs, scaling, desquamation, hemorrhage/bleeding, and hyperpigmentation) were also recorded and described.
During the first month of treatment, 1 male animal from group 2, that is, reinfusion to the same site once every 5 days, exhibited severe erythema with eschar at the sites of infusion. Therefore, from day 31, a new treatment plan was adopted, in which infusion sites were revisited once every 15 days. Since the adoption of this new treatment plan, no more cases of severe erythema and eschar formation were seen in this group, and therefore, the previously reported lesions were not taken into consideration for adversity determination.
Body Weight and Food Consumption
Each animal was weighed upon allocation to treatment group, on the day of commencement of treatment, subsequently at weekly intervals, and prior to necropsy. Weighing was performed prior to feeding. The weight of food consumed by each animal was recorded daily.
Clinical Pathology and TK
Samples of blood (4 ml) were withdrawn from the jugular vein of each animal after overnight food and water deprivation, prior to study initiation, on days 7 and 30 and during week 13 of the study, and for the recovery groups, on the day before necropsy. The blood was analyzed for hematological, coagulation, and biochemical parameters.
Blood samples of 4 ml each were collected from all animals pretest (once), on days 7 and 27 and on week 13 of the study immediately before pump replacement, and 4, 8, 12, 16, 20, and 24 hr after pump replacement, and analyzed for LD/CD content using a validated method. The following TK parameters were obtained or calculated, when appropriate, from the individual plasma values: Area under the curve (AUC)(0–tlast), C max, and T max. Animal exposure was estimated or calculated by the Kinetica™, version 4.4.1, PK/PD Analysis (Thermo Electron Corporation Informatics, Philadelphia, PA) software. Means and/or medians, standard deviations, and coefficients of variation were obtained using a Microsoft Excel worksheet.
Euthanasia and Necropsy
Animals that had completed the scheduled test period were given an intramuscular injection of sedative/tranquillizer and subsequently anaesthetized with intravenous barbiturate and sacrificed by rapid exsanguination. All animals were subjected to a detailed postmortem examination. For each animal, the skin pertaining to all injection sites and a sample of untreated skin (including the SC tissue and, if applicable, the underlying muscle tissue) were collected, and macroscopic evaluation (including size) of treatment site lesions was performed.
Organ Weights
After dissection to remove fat and connective tissue, the absolute and relative (organ-to-body weight ratios) weights of the following organs were recorded: adrenal glands, brain, epididymides, heart, kidneys, liver, ovaries, pituitary gland, prostate gland, spleen, testes, thymus, thyroid gland, and uterus cervix.
Microscopic Observations
Tissues were fixed and preserved in 10% neutral-buffered formalin. Histopathological evaluation was performed on all animals in all (control and treated) groups, and the following tissues were evaluated: adrenal glands, brain, heart, kidneys, liver, inguinal and axillary lymph nodes, all right and left sites of the treated skin, untreated skin, spleen, and any abnormality.
The histopathological sections of the skin samples (i.e., trimming of the tissue) were taken from the middle of the sample, to include the entire thickness, from the epidermis to the subcutis, and, if applicable, underlying muscle tissue. After dehydration and embedding in paraffin wax, sections of the tissues were cut at 5 μm thickness and stained with hematoxylin and eosin.
The histological evaluation of the skin pertaining to treatment sites was made by a subjective description of the observed tissue reaction, based on parameters describing grade of tissue tolerance (i.e., inflammation, necrosis, and fibrosis). The scoring of the lesions was done semi quantitatively, using a 5-point grading scale (0– to 4), taking into consideration the severity of the changes (0 = no change, 1 = minimal change, 2 = mild change, 3 = moderate change, 4 = marked change), based on the criteria explained by Shackelford et al. (2002).
Statistical Analysis
Standard deviations were calculated as considered appropriate. For continuous variables, the significance of the differences among groups was assessed by analysis of variance. Differences between each treated group and the control group were assessed by Dunnett’s test using a pooled error variance. The homogeneity of the data was verified by Bartlett’s test before Dunnett’s test. If the data were found to be inhomogeneous, a modified t-test (Cochran and Cox) was applied.
Results
Clinical Observations
Neither mortality nor adverse systemic signs occurred during the study with all minipigs remaining clinically normal. No treatment-related effects on body weight gain, final body weight, or food consumption were present.
Clinical infusion site observations
In general, the erythema score was low (typically absent or minimal) and in most cases comparable to that seen with saline (data not shown). Moderate-to-severe erythema was recorded in a limited number of sites of male animals from group 2 (mainly 1 animal) that were treated with ND0612 at a high frequency of infusions to a single site (i.e., once every 5 days, 6 infusions per month per site). At the end of recovery phase, erythema resolved in all animals. Edema (very slight-to-slight) was observed following ND0612 infusions but decreased rapidly and was very low prior to reinfusion at the same site. The magnitude of the edema remained relatively constant with repeated doses over the course of the exposure period indicating an absence of cumulative irritation. Any residual edema at the end of the exposure period recovered progressively during the postdosing recovery period. The daily observation of local infusion sites demonstrated similar responses among males and females. Dark skin discoloration and small scabs involving single infusion sites were seen randomly and periodically throughout the study.
The incidence of occasional and transient generalized reddening of the skin in the area of treatment sites (i.e., the back and flanks of the animals) observed during the exposure period was similar among groups. This was associated with generalized desquamation of the skin only in males treated with ND0612 (data not shown).
Clinical Pathology
No effects of toxicologically relevance were observed throughout the study. A slight increase in total and differential white blood cell counts was noted in males from group 2 (mainly in 1 animal, data not shown). This change is consistent with a mild response to the severe local tissue changes observed in one group 2 animal at 3 of the 5 infusion sites, following 6 reinfusions to the same site at high frequency (once every 5 days; see Material and Methods section for more details). This white blood cell effect was not present in treated female minipigs of group 2 and was not present in minipigs of group 3 administered higher doses of ND0612 (data not shown).
TK
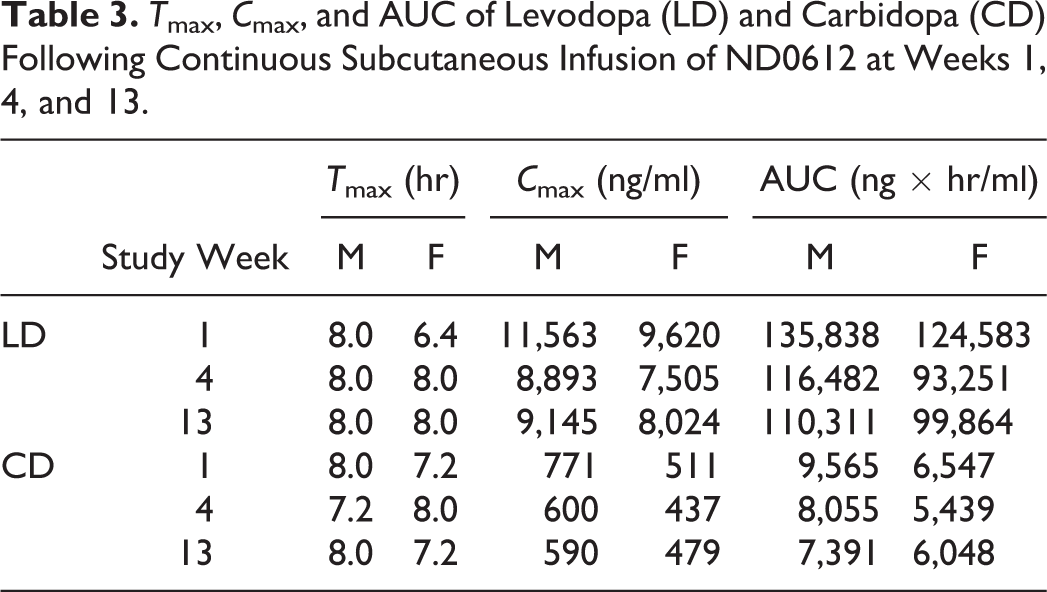
All treated animals were exposed to LD and CD throughout the study with mean AUC levels reaching 135,838 ng/ml × hr and 9,620 ng/ml × hr, respectively. Plasma levels (AUC0–tlast and C max) of LD and CD determined every 4 hr on experimental days 7, 27, and 85 (week 1, 4, and 13, respectively) were dose proportional and reflected the varying doses of infusion over a 24-hr period. Differences in AUC0–tlast and C max were observed between week 1 and 4, but no noteworthy differences were observed between week 4 and 13 (Figure 1 and Table 3).
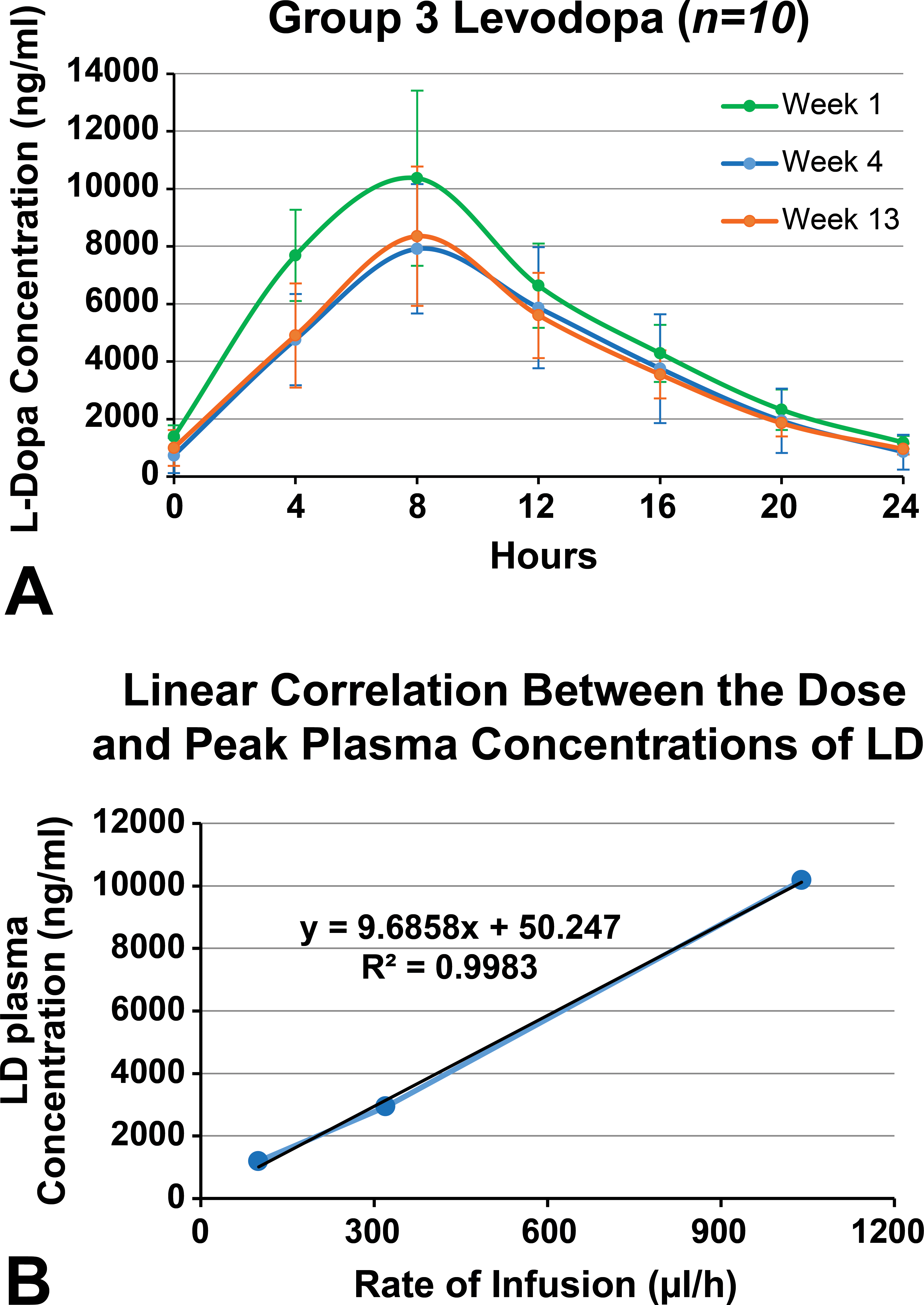
(A) Levodopa plasma concentrations following infusion at weeks 1, 4, and 13. (B) Linear correlation between the dose and peak plasma concentrations of levodopa.
T max, C max, and AUC of Levodopa (LD) and Carbidopa (CD) Following Continuous Subcutaneous Infusion of ND0612 at Weeks 1, 4, and 13.
Macroscopic Observations
No systemic macroscopic treatment-related changes were observed.
Macroscopic infusion site observations
At the end of treatment, the most recently ND0612-infused sites of both male and female animals showed focal or multifocal areas of darkened and/or pale subcutis as well as swelling of the cutis, irrespective of treatment regimen. The last injected sites showed similar effects except that the swelling of the subcutis had resolved in most animals. This is consistent with the in-life clinical observations of the resolution of edema between reinfusions. A pale and creamy aspect to the cut surface, scabs, or pale muscle were infrequently observed across the ND0612-treated groups and were predominantly observed at the most recently injected sites. Abnormal gross findings at infusion sites of animals receiving saline infusions were rare. At the end of the 30-day recovery period, the principal finding was dark foci in the subcutis of the ND0612 infusion sites. However, the dimensions of the foci were smaller at the end of recovery than those observed at the end of the treatment period.
Microscopic Observations
Changes were limited to ND0612 infusion sites. No systemic microscopic treatment-related changes were observed.
Microscopic observations of infusion site
Terminal sacrifice
Minimal to mild multifocal collections of brownish pigment-laden macrophages from previously present hemorrhage were noted. In some of the animals, chronic inflammation, associated with or without necrosis and mineralization, was noted in the deeper muscle (panniculus) layer. The chronic inflammation was characterized by fibroblastic proliferation and collagen deposition, associated with mixed mononuclear cells. The degree of necrosis was reduced from mild to minimal, as time elapsed from the last infusion (1 or 2 weeks postinfusion, respectively).
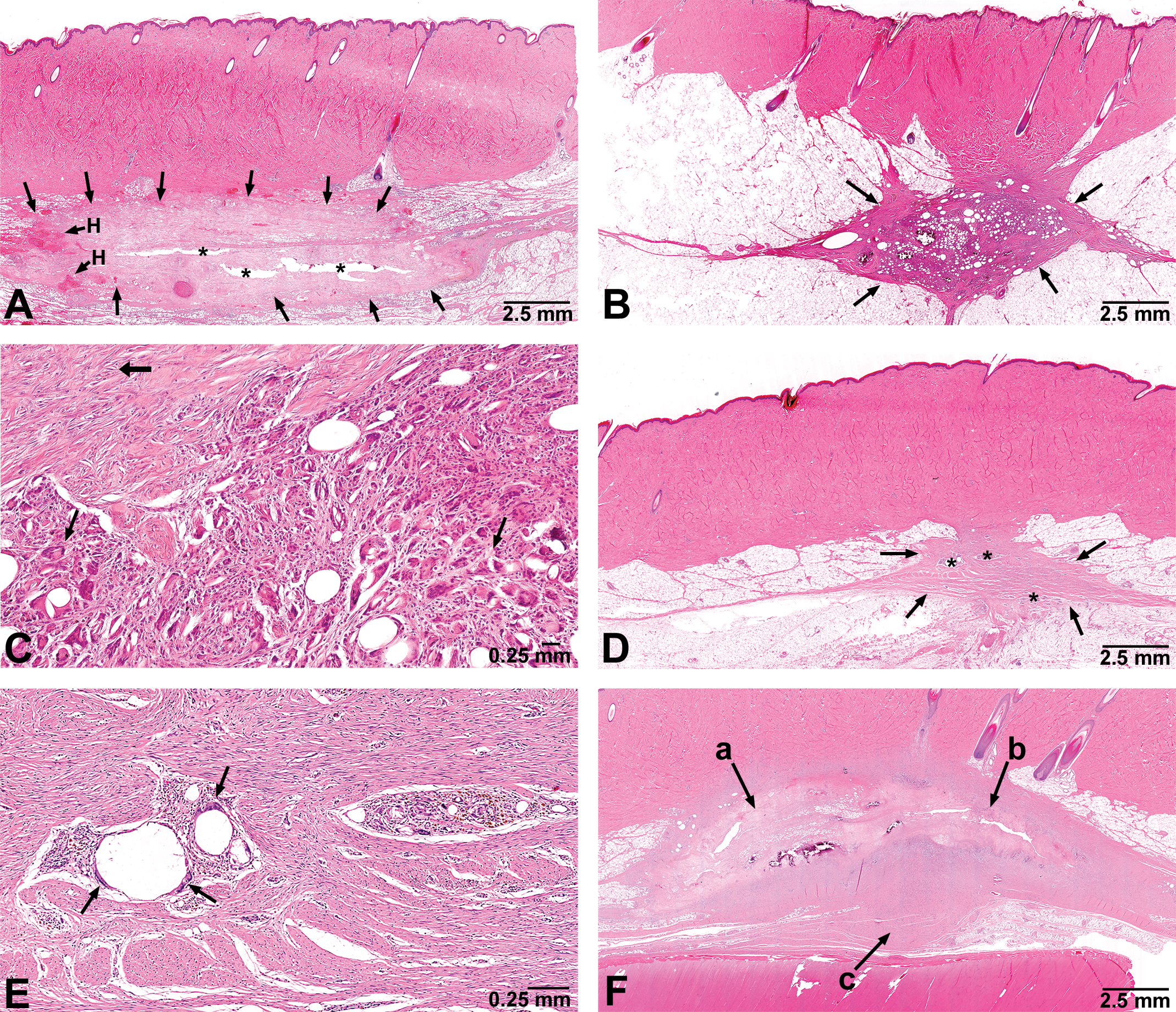
(A) Acute necrosis 3 days following last of 4 infusions to the same site once every 15 days in a male minipig from group 2 (60/7.5 mg/ml, 6 ml/24 hr). Arrows point to a pale pink area of necrosis with hemorrhage (H) and centrally located clear spaces (*) representing the site of test agent and fragile necrotic tissue washed out during tissue fixation and histology slide preparation. (B) Chronic residual reparative inflammation 34 days following last of 9 infusions to the same site once every 10 days in a female minipig from group 4 (60/14 mg/ml, 6 ml/24 hr). The discrete subcutaneous inflammation (arrows) consists of a central area of necrotic fat undergoing phagocytosis surrounded by a band of mature connective tissue. (C) Higher magnification of the previous photo demonstrating deposition of mature collagen (thick arrow), and multinucleated giant cells developed to engage in the phagocytic process (thin arrows). (D) Residual tissue response 61 days following the last of 6 infusions once every 5 days in a male minipig from group 2 (60/7.5 mg/ml, 6 ml/24 hr). An irregularly shaped but quite discrete area of chronic reparative connective tissue (arrows) is surrounded by normal adipose tissue in the subcutis. Small foci of residual phagocytosis (*) are present in the mature connective tissue. (E) Higher magnification of Figure D. Two focal aggregates of residual necrotic fat tissue are being phagocytosed by mononuclear cells and multinucleated giant cells. Multinucleated giant cells (arrows) can be seen at the periphery of clear spaces previously occupied by necrotic fat. Surrounding collagenized connective tissue comprises the majority of this chronic response to infusion. (F) Infusion site reaction demonstrating 3 stages of response in a male minipig from group 3 following 9 infusions once every 10 days to the same site (60/7.5 mg/ml, 6 ml/24 hr): (a) The tissue reaction 5 days following last infusion has a pale pink staining area of necrosis with mineralization. (b) The tissue reaction 15 days following last infusion has a pale pink staining area of necrosis but with a faint basophilic staining peripheral band of granulation tissue. (c) Mature connective tissue with collagen deposition representing a chronic response from an infusion greater than 25 days prior.
Recovery phase
Only sporadically, the chronic inflammation extended into the deeper muscle (panniculus) layer. In 2 animals, minimal focal mineralization was noted. In a single animal, an abscess formation was noted, but this change is considered related to an incidental contamination occurring during injection. The chronic inflammation was characterized by predominant presence of fibrosis and with a minor component of mixed mononuclear cell infiltration.
Discussion
Injection/infusion site reactions are a common adverse event when parenteral drugs are used, with incidence rates of up to 90% with some medications (Gold et al. 2005; Haibel et al. 2005). Such reactions result from local irritation due to pharmacology, toxicity, and/or hypersensitivity mechanisms (Willy et al. 2016). Although most reactions are mild and transient in nature, they can sometimes be severe and lead to tissue necrosis necessitating surgical interventions (Lee et al. 2014; Sato et al. 2014; Willy et al. 2016) and to treatment discontinuation. It is therefore essential to accurately predict and avoid infusion site reactions during preclinical phase testing to avoid unjustified failure of clinical trials and drug withdrawal (Willy et al. 2016). Indeed, severe infusion site reactions can sometimes lead to termination of the drug development program, as in the case of protiramer (Ramot et al. 2012; Varkony et al. 2009).
In this study, we used a complex study design involving multiple infusion sites with different doses and concentrations of LD and CD as well as various rates, volumes, number, and frequency of infusions. This complex study design allowed us to reconstruct the natural history of tissue reaction to infusion and to provide a complete histopathological scenario following injection, while complying with the 3Rs guiding principles for more ethical use of animals in testing.
The histopathological evaluation of the infusion sites demonstrated localized, SC changes, associated with temporal progressive healing. Our study also clearly showed that the extent of histopathological changes at the site of infusion was mainly related to the number and frequency of injections, with longer intervals between infusions to the same site and a smaller number of injections resulting in smaller lesions.
The relative effect of the 2 CD concentrations and the different rates and volumes that were tested in this study had a negligible effect on the extent and the respective rate of repair of local site reactions (data not shown). Thus, longer intervals between infusions resulted in more rapid and complete lesion resolution, having obvious relevance for clinical applications.
In addition, plasma LD and CD concentrations were determined throughout the study to evaluate the effect of local site reactions on drug absorption. Differences in AUC0–tlast and C max were observed between weeks 1 and 4, but no noteworthy differences in AUC0–tlast and C max were observed between weeks 4 and 13. Moreover, there were no differences in T max. These results suggest that frequent repeated infusions to the same site and the respective local site reactions do not impair the absorption of ND0612.
Overall, no systemic adverse effects were observed after 13 weeks of SC infusion with the novel ND0612 formulation of LD 60 mg/ml and CD 7.5 mg/ml or 14 mg/ml and up to a dose level of 720 and 90 mg/animal (approximately 36 and 4.5 mg/kg), respectively. Effects seen in this 90-day study were limited to the sites of infusion and tended to increase in incidence and/or severity with increasing frequency and number of infusions to the same site. However, reinfusion to same site once every 10 days at a dose volume of 6 ml/site, corresponding to a dose of up to 360/83 mg LD/CD at an infusion rate of up to 750 μl/hr, was safe and tolerable. Furthermore, a time-dependent recovery process of local site reactions was observed, initiating after15 days, showing foremost reduction in the incidence and severity of local site reactions 30 days after the last infusion, and suggesting reversibility of local site reactions.
In the scientific assessment of the extent and severity of pathological lesions in a given organ system, complete recovery (full reversibility) within the context of study duration is not essential (Perry et al. 2013). Consideration of the repair capacity of the affected organ system, demonstration of evidence of progressive recovery, and general knowledge of pathobiology allow for the scientific assessment of the clinical significance of recovery. In situations where full reversibility to complete normalcy does not occur, the effect of the residual change, as with the residual fibrotic scar following resolution of infusions in the present study, is not considered adverse when organ function would not be impaired at clinically relevant doses (Perry et al. 2013).
The anticipated clinical infusion regimen uses the same concentration drug product (60 mg/ml LD, 7.5 mg/ml CD) as was administered in groups 2 and 3 in the 90-day minipig study. A comparison of the anticipated clinical regimens with those tested in the 90-day minipig study is shown in Table 4. Thus, local effects observed in the minipig due to the greater exposure are expected to be similar to or somewhat greater than local effects likely to occur in patients receiving ND0612. It is likely, therefore, that patients receiving the clinical regimens may experience a chronic inflammatory response at the infusion sites that is characterized by very minimal erythema and mild edema that resolves between repeated infusions. Scabs, cysts, or abscesses at the infusion sites may infrequently be observed, but these are likely to be minimal in severity and to resolve following cessation of the infusion treatments to the affected site.
Comparison of Planned Clinical Regimens with the Minipig Regimens.

This study also provides support for the safety of the alternate low-dose product intended for mild-to-moderate PD patients. The infusion regimen planned for these mild-to-moderate patients utilizes the same drug product as the high-dose product but at a slower infusion rate (4.5 ml/day). Additionally, the low-dose product is intended to be administered at a single infusion site versus the dual-site regimen planned for the high-dose product, and the time between reuse of infusion sites will therefore be around 30 days. Thus, the low-dose regimen will result in a total daily dose of 270 mg LD and 34 mg CD. Since the injection site volume will be 75% of that for the high-dose product, it is anticipated that injection site reactions may be slightly less. Systemic exposures will be 62.5% lower than the high-dose regimen. Thus, no adverse systemic effects are anticipated.
Given the clear trend for reversibility of the local infusion site and no evidence of behavioral changes or functional impairment associated with these findings, as well as no effect on drug absorption, none of the effects seen in this study were judged to be adverse.
To the best of our knowledge, this is the first systematic and detailed report on the histopathological findings that accompany infusion site reaction in pigs. This report can thus serve as a case study for the preclinical pathological assessment of infusion and injection site reactions in minipigs.
Footnotes
Authors’ Contribution
Authors contributed to conception or design (AN, RM, YT, RAM, GS, OY), data acquisition, analysis, or interpretation (YR, AN, RM, YT, RAM, GS, OY); drafted the manuscript (YR, AN, RM, OY); and critically revised the manuscript (YR, AN, RM, YT, RAM, GS, OY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by NeuroDerm Ltd.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
