Abstract
The ten key characteristics (KCs) of carcinogens are based on characteristics of known human carcinogens and encompass many types of endpoints. We propose that an objective review of the large amount of cancer mechanistic evidence for the chemical bisphenol A (BPA) can be achieved through use of these KCs. A search on metabolic and mechanistic data relevant to the carcinogenicity of BPA was conducted and web-based software tools were used to screen and organize the results. We applied the KCs to systematically identify, organize, and summarize mechanistic information for BPA, and to bring relevant carcinogenic mechanisms into focus. For some KCs with very large data sets, we utilized reviews focused on specific endpoints. Over 3000 studies for BPA from various data streams (exposed humans, animals, in vitro and cell-free systems) were identified. Mechanistic data relevant to each of the ten KCs were identified, with receptor-mediated effects, epigenetic alterations, oxidative stress, and cell proliferation being especially data rich. Reactive and bioactive metabolites are also associated with a number of KCs. This review demonstrates how the KCs can be applied to evaluate mechanistic data, especially for data-rich chemicals. While individual entities may have different approaches for the incorporation of mechanistic data in cancer hazard identification, the KCs provide a practical framework for conducting an objective examination of the available mechanistic data without a priori assumptions on mode of action. This analysis of the mechanistic data available for BPA suggests multiple and inter-connected mechanisms through which this chemical can act.
Keywords
Introduction
Bisphenol A (BPA) is a high production volume chemical with a wide range of consumer and industrial applications. Major uses of BPA are in the production of polycarbonate plastics and epoxy resins. Humans are exposed to BPA predominantly through contaminated food (e.g., via food packaging) and drinking water, although dermal exposure from other uses is also likely. BPA is a recognized and extensively studied xenoestrogen. However, there is a lack of comprehensive reviews of BPA encompassing data from all evidence streams related to carcinogenicity.
In November 2020, BPA was placed in a “high” priority group for evaluation of potential carcinogenicity by the Carcinogen Identification Committee (CIC), a group of scientists designated as the “State’s Qualified Experts” for evaluation of chemical carcinogenicity under California’s Proposition 65. In January 2022, OEHHA solicited from the public information relevant to the assessment of the evidence on its carcinogenicity.
In developing the hazard identification document for BPA for evaluation by the CIC, we conducted a comprehensive search of the scientific literature for BPA studies relevant to potential carcinogenicity, and compiled and summarized evidence from human and animal studies, metabolism studies, and mechanistic studies (OEHHA, 2022). 1 The CIC met on December 14, 2022 and declined to add BPA to the Proposition 65 List of Carcinogens by 6 no votes and 5 yes votes.
The CIC considered the epidemiologic evidence inadequate to evaluate the carcinogenicity of BPA due to limitations such as inadequate exposure assessment. None of these studies were able to reliably characterize long-term BPA exposure due to the use of short-term biomarkers that reflect recent exposure and the lack of longitudinal measurements to characterize long-term exposure or temporal variation. All studies were conducted in the general population with lower exposure than workers highly exposed to BPA. The CIC also reviewed the carcinogenicity studies of BPA in multiple strains of rats and mice, and in gerbils. Tumors were observed in the liver, lymphohematopoietic system, reproductive system, pituitary, thyroid, and mammary glands in studies of BPA-treated rats. In studies of BPA-treated mice, tumors were observed in the liver, lymphohematopoietic system, and pituitary gland. However, some Committee members considered some of these tumor findings to be marginal, noting observations such as apparent non-linear dose-response, high background incidence in controls, or significant for trend with lack of increase in the low dose.
For the mechanistic data, we applied the key characteristics (KCs) of carcinogens to identify, organize, and evaluate the mechanistic data for BPA and to investigate whether the mechanistic evidence supports the possibility that BPA is, or acts, like a carcinogen.
Other frameworks exist that seek to evaluate human health or ecological risks of chemical exposure. One example is the Adverse Outcome Pathway (AOP), which describes a sequential chain of causally linked events at different levels of biological organization that lead to an adverse health or ecotoxicological effect, with the ultimate goal to assist with regulatory decision making. 2 Although there are many specific AOPs that are related to carcinogenesis (e.g., AOP296, oxidative DNA damage leading to chromosomal aberrations and mutations), there are currently no set AOP frameworks that comprehensively evaluate chemical carcinogenesis.
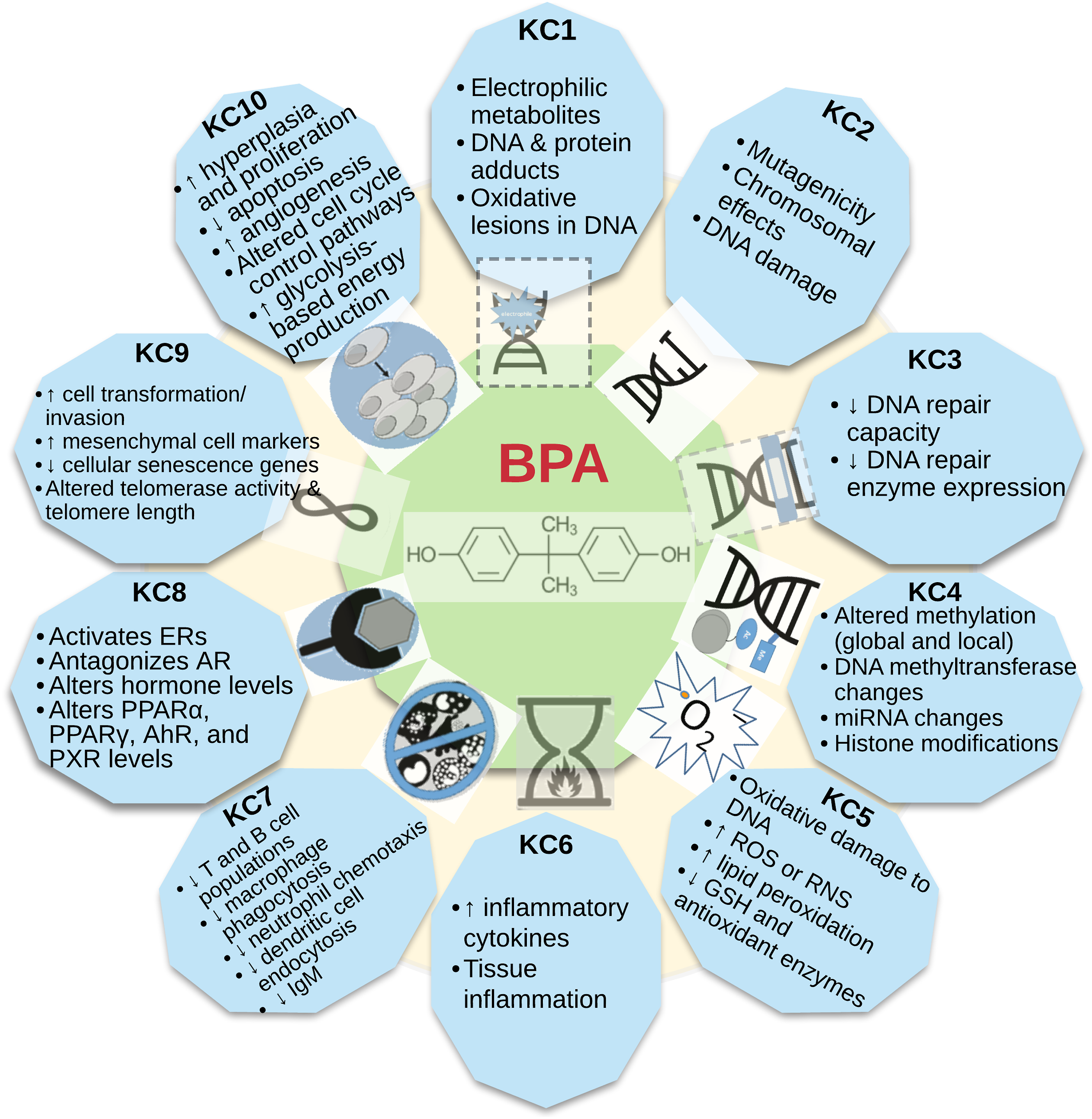
The ten KCs of carcinogens were originally identified via a comprehensive review of the more than 100 agents known to cause cancer in humans.3,4 The KCs approach is constructed “bottom up”, i.e., characteristics of known carcinogenic agents were used to inform which mechanisms are relevant in this multistage process, without requiring a pre-ordained sequence of events, specific tests or methods, or an advanced hypothesis. Specifically, these key characteristics comprise the ability of an agent to: act as an electrophile either directly or after metabolic activation (KC1); be genotoxic (KC2); alter DNA repair or cause genomic instability (KC3); induce epigenetic alterations (KC4); induce oxidative stress (KC5); induce chronic inflammation (KC6); be immunosuppressive (KC7); modulate receptor-mediated effects (KC8); cause immortalization (KC9); and alter cell proliferation, cell death, or nutrient supply (KC10). 3
The KCs distill 40 years of scientific knowledge about the mechanisms through which human carcinogens operate and are used in cancer hazard identification by IARC, NTP, US EPA, and OEHHA. In contrast to modes of action, which are sequences of key events that transform normal cells into malignant tumors, KCs do not require an implicit ordering of events. In this sense, the KCs are analogous to the well-established hallmarks of cancer, which also do not imply any sequence.5,6 On the other hand, the two concepts differ in that the KCs describe the chemicals that cause cancer, while the hallmarks describe the biological features of the cancers. Mode of action identification depends on prior knowledge sufficient to hypothesize how an agent might cause cancer, knowledge that too often is incomplete. The KCs can encompass many types of mechanistic endpoints and are not constrained to previously formulated hypotheses, allowing a broader consideration of multiple mechanistic pathways and hypotheses. Many human carcinogens act via multiple mechanisms, whereas for others only one mechanism is known. There is no requirement as to how many KCs are necessary for a chemical to be identified as a carcinogen. 7 Importantly, the KCs approach also serves as a bridge to innovation in toxicity testing, allowing for mapping data generated from New Approach Methods (e.g., omics) to the KCs and including molecular profiling in the evaluation. 8
In this manuscript, we present findings on the mechanistic data as well as a synthesis of metabolism data, as the oxidative metabolism of BPA gives rise to a complex mixture of reactive and bioactive metabolites that directly support several KCs. Our review highlights how the application of the KCs can facilitate the evaluation of a very large data set and provide insight on possible mechanisms.
Methods
Literature Search Process
Literature searches on BPA, its metabolism, and the KCs were conducted up to December 2021. 1 The goal was to identify peer-reviewed journal articles, print and digital books, and reports that potentially reported mechanistic information on the carcinogenicity of BPA. As part of the development of the hazard identification document for BPA, the literature searches were supplemented with a public data call-in period from January 28 to March 14, 2022.
We used an approach similar to that recommended by the National Toxicology Program (NTP) Handbook for Preparing Report on Carcinogens (RoC) Monographs. 9 This included primary searches in major biomedical databases, such as PubMed (National Library of Medicine), Embase, Scopus, and SciFinder-n. Additionally, other data sources including authoritative reviews and reports, databases, and web resources were searched, and focused searches were conducted as needed. Primary searches were executed using BPA synonyms in combination with search terms for toxicokinetic studies or the KCs. For the KCs, in addition to BPA synonyms, the search terms included cancer terms, RoC “other mechanistic” terms, IARC “key characteristics of carcinogens” terms, and RoC “key characteristics of carcinogens” terms. There were no restrictions in the searches on exposure route or duration of studies, or on publication language.
Literature Screening Tools
Sciome Workbench for Interactive Computer-Facilitated Text-mining (SWIFT) Active Screener was used as a tool to facilitate the initial screening of references from the primary search. 10 In SWIFT Active Screener, two scientists independently screened each title and abstract by following predefined inclusion and exclusion criteria. Selected references were uploaded to Endnote and duplicates were removed.
HAWC (Health Assessment Workspace Collaborative) was used to further screen and tag the literature according to the key subject areas after initial screening in SWIFT Active Screener. We applied two levels of screening in HAWC before tagging. In Level 1 screening, citations that did not contain information on BPA, BPA metabolism, or KCs were eliminated based on their title and abstract. Papers identified for inclusion during Level 1 screening were tagged in HAWC according to the endpoints and test systems associated with the KCs and metabolism. In Level 2 screening, full-text papers for all citations that passed the Level 1 screening were screened by at least one scientist, using similar inclusion/exclusion criteria used in the Level 1 screening. Following Level 2 screening, the tagging of articles according to key topics was updated in HAWC. HAWC search results were updated if additional relevant studies in addition to those cited in the original set of publications (“secondary citations”) were identified.
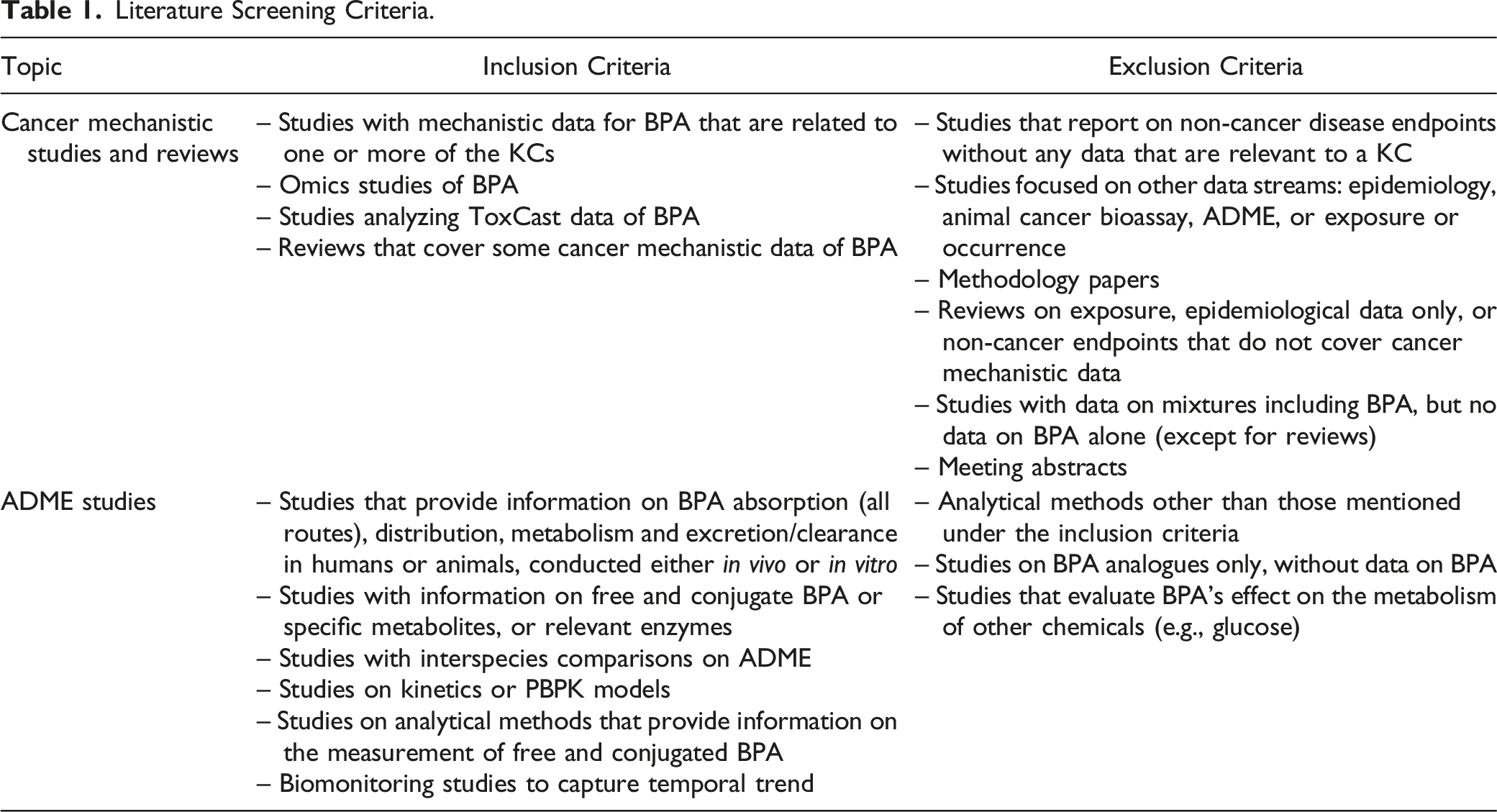
Inclusion and Exclusion Criteria
Literature Screening Criteria.
Use of Comprehensive Reviews
For KCs (or endpoints within KCs) with very large data sets (e.g., for KC8, receptor-mediated effects, over 1600 citations were identified), we used recent, comprehensive reviews identified from the literature screening as a starting point. Reviews were selected based on their utility for inclusion and examined for accuracy by cross-checking reported data and summary statements. 1 Specifically, we focused on reviews that reported original data from studies of BPA with data representing one or multiple endpoints or biomarkers for a KC, and that reported data across test systems or diverse study designs. For endpoints or test systems not included in these reviews, we used data from original studies if they were available. Below we describe some examples of this approach.
For KC2, many of the genotoxicity studies of BPA were published before 2008 and are reviewed and summarized in detail by Chapin et al (2008), 11 part of an NTP report (CERHR 2008). We reviewed the genotoxicity studies (>100) identified by our literature search that were not included in Chapin et al (2008).
Over 500 studies for KC5 (induction of oxidative stress) were identified. While we reviewed the original studies in humans and rodents on endpoints such as oxidative damage to DNA and reactive oxygen species (ROS) or reactive nitrogen species (RNS), we utilized available reviews on other KC5 endpoints, such as lipid peroxidation and changes in antioxidant enzyme activities or levels.12-15
For KC8 (receptor-mediated effects), we identified over 800 studies for estrogen receptor-mediated effects. BPA is a well-recognized xenoestrogen known to mimic and interfere with the actions of estrogen, which has led to a substantial body of evidence that has been well summarized in a number of high-quality reviews.11,16-30 Therefore, we have relied on these reviews to summarize the evidence for estrogen receptor-mediated effects.
Similarly, over 600 studies for KC10 (alteration of cell proliferation, cell death, or nutrient supply) were identified. We relied on reviews, such as Chapin et al 2008; 11 Gassman 2017; 15 and Nomiri et al 2019, 31 for summaries of KC10-related effects of BPA in human calls in vitro. We also reviewed original studies on BPA conducted in animals, in vivo, and studies that covered additional endpoints not summarized in these reviews.
Results
Our literature search identified over 500 references on toxicokinetics and metabolism of BPA and more than 3000 references on the KCs. Below, we summarize key findings from metabolic and mechanistic data sets. For a more detailed summary of all studies and a complete list of citations, see OEHHA. 1
BPA Metabolism and Key Metabolites
BPA is rapidly absorbed in humans by the oral and dermal routes32-34 and is distributed throughout the body, crossing the blood-brain barrier and the placenta.35-37 The half-life of BPA in humans after oral administration is around six hours. Its excretion is rapid and occurs primarily via urine in humans and other primates. Despite rapid elimination, BPA is routinely detected in more than 90% of human urine samples, suggesting frequent repeated exposures, likely from multiple sources. 38
BPA is primarily metabolized via conjugation, leading to the formation of BPA-glucuronide and BPA-sulfate. The extent of conjugative metabolism is determined by life stage, co-exposure to xenobiotics and drugs, certain diseases, and polymorphisms of conjugative enzymes.39-42
A second metabolic pathway is the oxidation of BPA by peroxidases or cytochrome P450 (CYP) enzymes, with key metabolic reactions including hydroxylation, carbon bond cleavage, dimerization, and glutathione (GSH) binding to an arene epoxide intermediate product of CYP-catalyzed oxidative metabolism. Oxidation of BPA produces several reactive and biologically active metabolites such as quinones and various radicals, which can contribute to the formation of ROS and oxidative stress.43,44 Some metabolites have estrogenic and/or cell proliferative properties discussed in more detail below.
An overview of oxidative metabolism of BPA with its key reactive metabolites is presented in Figure 1. An in-depth summary of BPA metabolism, along with a description of metabolites and a complete list of references, is provided in OEHHA 2022.
1
Oxidative metabolism of BPA. The oxidation of BPA is catalyzed by cytochrome P450 (CYP) enzymes and peroxidases (peroxidases not shown). In an initial oxidation step, BPA is hydroxylated to ortho-hydroxy-BPA (ortho-OH-BPA; BPA catechol) and then undergoes further oxidation to BPA-3,4-quinone (BPAQ), via its semiquinone. BPAQ forms DNA and protein adducts. BPAQ can also be conjugated with GSH. In other reactions, the quinone-catechol couple can undergo redox cycling during which reactive oxygen species (ROS) are produced. ROS may also be generated via other reactive metabolites and pathways (not shown). Following metabolism via CYP enzymes and likely involvement of an arene epoxide intermediate, BPA can be conjugated with GSH. Carbon bond cleavage of the parent molecule, via a quinol intermediate, leads to formation of hydroquinone and a carbocation intermediate. Subsequent reactions and rearrangements yield isopropenylphenol (IPP) and hydroxycumyl alcohol (HCA). IPP and its intermediate radical can form the metabolite 4-methyl-2,4-bis(p-hydroxyphenyl)pent-1-ene (MBP). Oxidative metabolism also forms phenol and its GSH conjugate.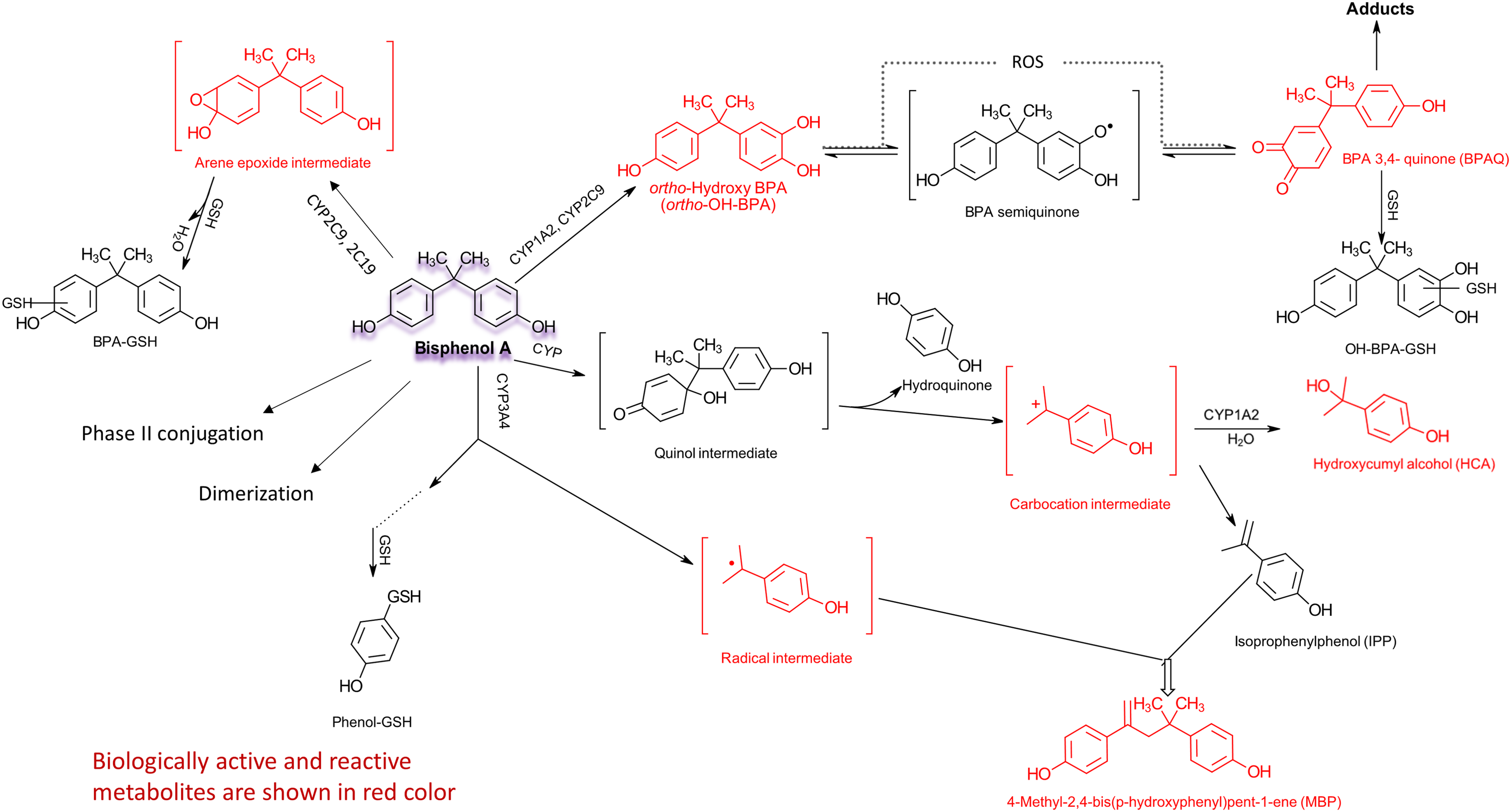
Here we briefly list and describe some of the key metabolites. BPA-3,4-quinone (BPAQ), a well-studied metabolite, forms protein and DNA adducts (e.g., 3-hydroxy-bisphenol A-N7-guanine, 3-hydroxy-bisphenol A-N7-adenine) in vitro and in vivo.45-47 BPAQ can also induce the formation of oxidatively modified DNA bases, measured as 8-OHdG (8-hydroxydeoxyguanosine) in rat hepatocytes. 48 Either type of DNA modification is likely to contribute to genotoxicity. The BPAQ (quinone) - ortho-hydroxy-BPA (catechol) couple can also undergo redox cycling during which ROS are produced, leading to oxidative stress. 15 ROS formation may also occur in other reactions where electrophilic or reactive compounds are formed. Other reactive metabolites include an arene epoxide intermediate, which is formed prior to GSH conjugation 49 ; an isopropenylphenol (IPP) radical, the formation of which involves a carbocation intermediate; and an unidentified electrophilic compound involved in the dimerization of BPA. 50
Biologic activity associated with BPA metabolites includes estrogenicity and induction of cell proliferation. Metabolites with estrogenic activity include ortho-hydroxy-BPA, 4-methyl-2,4-bis(p-hydroxyphenyl)pent-1-ene (MBP) and hydroxycumyl alcohol (HCA).47,51,52 Ortho-OH BPA and MBP can also induce cell proliferation of human breast cancer cells.52,53 Thus, oxidative metabolism of BPA produces several metabolites that are reactive and biologically active. Their relationship to the KCs will be discussed further below.
Mechanistic Data for BPA Organized by KCs of Carcinogens
As mentioned earlier, we identified over 3000 publications on mechanistic data for BPA. Analysis of such a large database would be unwieldy without a framework to organize the studies based on factors related to potential mechanisms. To efficiently analyze this complex dataset, we utilized the KCs approach to organize the studies based on factors related to potential mechanisms. There is evidence for BPA relevant to each KC (Figure 2), with some KCs having stronger evidence than others, based on data availability and consistency of results across endpoints and test systems. Data for the KCs stem from observational studies in humans, experimental studies conducted in animals, in vitro studies with human or animal cells, and studies in cell-free systems. Key findings relating to each KC, along with examples of some data tables, are discussed below. Additional tables and data sets can be found in Appendices F through K of OEHHA 2022.
1
KC1: Is Electrophilic or Can Be Metabolically Activated
Electrophiles are reactive, electron-seeking molecules capable of binding to electron-rich cellular macromolecules, including DNA, RNA, lipids, and proteins, thereby forming covalent adducts. The measurement of covalent adducts on DNA and proteins is the most common method of assessing electrophilic activity. 55
BPA can be metabolized by peroxidases 46 and by CYP enzymes to form electrophilic compounds (see OEHHA 2022 1 and Figure 1), leading to formation of DNA and protein adducts as well as oxidative DNA lesions. 48 DNA adducts formed by BPAQ have been identified as 3-hydroxy-bisphenol A-N7-guanine and 3-hydroxy-bisphenol A-N7-adenine in human in vitro, animal in vivo and in vitro, and cell-free systems.45,46,56-58 BPA can also bind to cysteine residues of proteins and forms protein adducts in the rat in vivo and in a cell-free system. 59
Overall, the evidence for KC1 is consistent across species in vivo and in vitro. In spite of a fairly small data set, the data for KC1 were considered strong during the Committee discussion. In addition, metabolism data strengthen the biological plausibility with the formation of ROS and electrophilic metabolites, which result in oxidative lesions in DNA and adduct formation.
KC2: Is Genotoxic
Genotoxicity refers to the ability of a chemical or other type of agent or biological process to damage DNA or induce changes in the DNA sequence. The link between genotoxicity and carcinogenesis is well established.3,4,55,60 Changes in the DNA sequence include gene or point mutations such as base substitutions, frameshifts, and small deletions or insertions, and chromosomal effects such as chromosomal aberrations, micronuclei, and aneuploidy. Examples of DNA damage include DNA adducts, DNA strand breaks, and oxidative damage to DNA.
Chromosomal and DNA Damage Effects of BPA Exposure in Human Observational Studies.
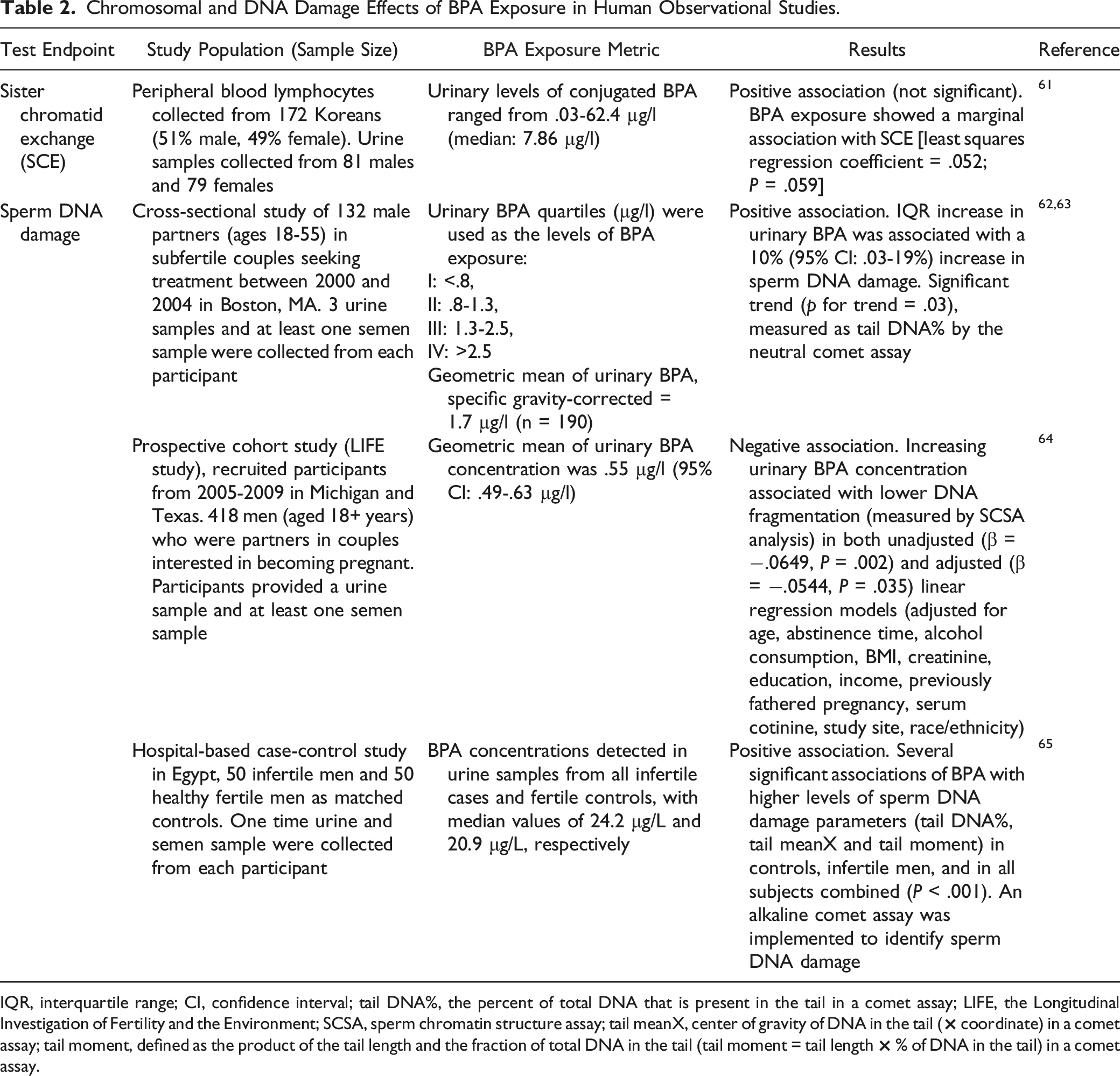
IQR, interquartile range; CI, confidence interval; tail DNA%, the percent of total DNA that is present in the tail in a comet assay; LIFE, the Longitudinal Investigation of Fertility and the Environment; SCSA, sperm chromatin structure assay; tail meanX, center of gravity of DNA in the tail (× coordinate) in a comet assay; tail moment, defined as the product of the tail length and the fraction of total DNA in the tail (tail moment = tail length × % of DNA in the tail) in a comet assay.
Effects of BPA Exposure on Oxidative Damage to DNA (Urinary 8-OHdG) in Human Observational Studies. a

aStudies in the table are sorted by the findings (positive, no significant, or negative association) and then alphabetically by author.
bNew studies not reviewed in Steffensen et al 2020. 12
Genotoxicity Studies of BPA in Human Cells In Vitro.

CBMN, Cytokinesis blocked micronucleus; UDS, Unscheduled DNA synthesis; Fpg enzyme, formamidopyrimidine-DNA glycosylase; Nth, endonuclease III; hOGG1, human 8-oxoguanine DNA glycosylase; γ-H2AX, phosphorylated histone H2AX (Ser139); PHA, phytohemagglutinin; TP53, tumor protein P53; p-p53, phospho-p53 (Thr18); MDM2, MDM2 proto-oncogene; CDKN1A, cyclin-dependent kinase inhibitor 1A; GDD45 A, growth arrest and DNA damage inducible gene alpha; CHEK1, checkpoint kinase 1; p-Chk1, phospho-Chk1 (Ser345); p-Chk2, phospho-Chk2 (Thr68) [phosphorylated checkpoint kinase 2]; ERCC4, ERCC excision repair 4, endonuclease catalytic subunit.
The comet assay is also known as the single cell gel electrophoresis assay (SCGE). In this assay, tail DNA% is a measure of the proportion of total DNA that is present in the tail. The alkaline comet assay lyses cells under alkaline (pH > 13) conditions to detect single and double strand breaks and alkali-labile sites. The neutral comet assay lyses cells under neutral (pH 7 – 9) conditions to detect double strand breaks.
Substantial data on BPA-induced DNA damage are available. More than ten human observational studies reported statistically significant associations between urinary or serum levels of BPA and urinary 8-OHdG, a biomarker of oxidative damage to DNA (see section on KC5 and Table 3 for details). Two human observational studies reported positive associations between urinary BPA levels and sperm DNA fragmentation (Table 2).62,63,65 Increases in DNA adduct formation, DNA strand breaks, oxidative damage to DNA, and γ-H2AX (histone 2AX phosphorylated at serine 139) were observed in multiple experimental systems treated with noncytotoxic concentrations of BPA, i.e., human cells in vitro (Table 4), animals in vivo, animal cells in vitro, and acellular systems. 1 Increases in expression of proteins associated with DNA damage-control were observed in two studies in human cells in vitro122,124 and one study in animals in vivo. 126
Many studies showed that noncytotoxic concentrations of BPA induced chromosomal damage. One study in humans exposed to BPA showed a marginally positive association (P = .059) between urinary BPA levels and sister chromatid exchange frequency in peripheral blood lymphocytes collected from over a hundred Korean men and women (Table 2). 61 A number of in vitro studies in human (Table 4) and animal cells and in vivo studies in rodents and other animals have reported increases in micronuclei, chromosomal aberrations, and various types of chromosomal abnormalities following BPA treatment. 1 Three studies showed that BPA significantly increased chromosomal aberrations in plants.127-129 Two studies showed that BPA induced microtubule abnormalities in acellular systems.130,131
BPA is not mutagenic in bacteria or yeast. 11 The effects of BPA on mutagenicity have not been thoroughly investigated in other systems, although studies have shown that BPA can cause mutations in a normal human cell culture, 88 in a human immortalized cell line in vitro 87 and in male rats in vivo 11
Overall, some Committee members considered there to be strong evidence for KC2, based on mutagenicity, chromosomal effects, and DNA damage induced by BPA. One Committee member noted that while there is certainly evidence that BPA is genotoxic in vitro, the evidence for its genotoxicity in vivo, especially concerning aneuploidy and chromosomal damage, is more limited.
KC3: Alters DNA Repair or Causes Genomic Instability
Maintaining genomic stability and repairing damaged DNA are essential for normal cell function. Exogenous agents may interfere with DNA repair processes, which can lead to mutations and/or genomic instability. Alterations in DNA repair that lead to an increase in mutations can occur, for example, through suppression of high-fidelity DNA repair enzymes or enhancement of error-prone enzymes. Genomic instability can manifest as alterations in copy number variants, increases in chromosomal translocations, or microsatellite instability. Both alterations in DNA repair and genomic instability induced by exogenous agents can contribute to tumorigenesis. 55
Overall, a small number of studies have evaluated the ability of BPA to alter DNA repair or cause genomic instability. A few studies found that BPA decreased capacity to repair various types of DNA damage in human peripheral blood mononuclear cells, 108 a human breast cancer cell line (MELN), 132 mouse peritoneal macrophages, 133 and mouse embryonic fibroblasts. 134 Three studies found decreased expression of DNA repair enzymes (MyH, TP53, mlh1)111,135,136 and one study found small changes in copy number variants. 137
While there is evidence for BPA on impairment of DNA repair or DNA repair enzymes, the Committee considered the overall evidence to be limited, based on the few studies that report findings for KC3.
KC4: Induces Epigenetic Alterations
Epigenetic alterations are “hypothesized to serve as mediators of cancer etiology and progression, in many cases preceding cancer”. 55 Some markers of altered epigenomes typical of cancer cells include site-specific DNA hypermethylation and hydroxymethylation, a global pattern of DNA hypomethylation, alterations in the microRNA profile, and histone modifications.138-140
There is evidence from many studies that BPA exposure can induce epigenetic effects or effects on gene or protein expression that may be caused by epigenetic alterations. Study details can be found in the tables in Appendix G of OEHHA 2022. 1 Human observational studies reported associations between BPA levels and altered methylation of many genes related to cancer pathways, as well as altered global DNA methylation. Interpretation of these results is limited by the one-time measurement of the urinary BPA level. Among the study populations that found associations between BPA levels and altered methylation of specific genes were two publications examining sets of genes from a study of fetal liver tissue samples,141,142 seven cohorts of mother-child pairs, and six cross-sectional studies. The cohorts of mother-child pairs measured BPA in maternal urine during pregnancy and DNA methylation in the placenta, 143 cord blood,144-146 or whole blood of offspring at ages 2, 6, 7, or 8-14 years.147-149 The cross-sectional studies were of children150-153 and adults.154,155 The study details and affected genes can be found in Appendix Table G2 of OEHHA 2022 1 . The majority of these studies reported selected candidate genes or pathways but did not address comprehensive effects on methylation of the entire genome, preventing the possibility of discerning overall patterns in methylation.
Various measures of global methylation were altered in association with BPA levels in fetal liver tissue,141,156 placenta (but not fetal kidney or liver tissue), 157 and cord blood [statistically significant in one study but not in another145,158]. No associations were observed in a study that measured BPA in pregnant mothers and global methylation in 8-14 year-old children. 149 Several studies reported changes of global methylation in sperm (but not peripheral blood) in a cohort of workers in China who were exposed to BPA.159-161
Similarly, BPA exposure to human cells in vitro altered methylation of individual genes and global methylation status. Differentially methylated genes were observed in several cell types following BPA exposure, including breast epithelial cells (both estrogen-dependent and estrogen-independent),122,154,162-164 prostate adenocarcinoma cells, 165 human embryonic stem cells, 166 and placenta cells. 167 No changes were observed in hepatoma cells. 168 Global methylation was altered in several human cell types in vitro, including neuroblastoma cell lines, mesenchymal bone marrow stem cells, embryonic kidney cells, hepatoma cells, embryonic stem cells, breast epithelial cells, prostate cancer cells, colorectal carcinoma cells, and umbilical vein endothelial cells.137,154,165,168-173 Some studies did not observe an effect of BPA on global methylation.162,174,175
Other epigenetic effects observed in human cells in vitro included altered expression of microRNAs, overexpression of DNA methyltransferases (which regulate DNA methylation machinery; overexpression can lead to reduced expression of tumor suppressor genes), 176 and histone modifications. For example, BPA inhibited miR-381-3p expression in MCF-7 cells, which promotes high PTTG1 (Pituitary Tumor-Transforming Gene 1, Regulator of Sister Chromatid Separation, Securin) expression and alters the cell cycle to enhance proliferation. 177 BPA exposure in MCF-7 cells led to increased expression of genes associated with breast and other cancers by increasing histone acetylation and H3K4 trimethylation in promoter regions. This effect was observed in three studies and was mediated by the activation of MLL2, MLL3 (mixed lineage leukemia family of histone methyltransferases) and CBP/p300 (histone acetyltransferases).178-180
All of these observations are also supported by studies in non-human mammals in vivo, mammalian cells in vitro, and fish. Some examples are from experimental data showing that exposure to BPA in utero significantly increased methylation of CpG sites in estrogen receptor genes (Esr1 and/or Esr2) in the brain in mice 181 and rats, 182 prostate in mice, 183 and testis 184 and rats. 185 Global methylation was altered in mice and rats (e.g.,186,187); DNA methyltransferases were altered in rats and mice (e.g.,183,188); miRNAs were altered in mice (e.g., 189 ); and histone modifications were observed in mice, rats, and gerbils (e.g.,190-192). In zebrafish exposed to BPA, global hypomethylation was observed in the testes and ovaries.193,194
Overall, there is consistent evidence from many studies that BPA exposure can induce epigenetic changes in multiple systems, although it was also noted that drawing conclusions is difficult due to the quality of available data and design of those studies. Human observational studies reported associations between BPA levels and altered methylation of genes related to cancer pathways, but these studies had the same limitations in exposure assessment as the human epidemiological studies and may not reflect actual BPA levels. In addition to measurement of BPA levels over time in human observational studies, future studies should be designed to consider the functional consequences of the epigenetic changes observed in the in vitro and animal studies.
KC5: Induces Oxidative Stress
Oxidative stress refers to an imbalance between the production and elimination of reactive oxygen and nitrogen species (ROS, RNS). Oxidative stress may contribute to the carcinogenic process by causing DNA mutations, chromosomal damage, genomic instability, altered cell cycle regulation, and chronic inflammation.195,196 As noted, ROS can be formed during the oxidative metabolism of BPA, either via redox cycling or oxidative reactions. Biomarkers for oxidative stress include 8-OHdG (oxidative damage to DNA), ROS or RNS production, malondialdehyde or 8-isoprostane (two common markers for lipid peroxidation), total antioxidant capacity, glutathione status, and other antioxidant enzyme activities and expression levels.
Over 30 studies, all published since 2009, investigated whether BPA induces oxidative damage to DNA, including 19 observational studies in humans (Table 3), five in vitro studies using human cells and three using animal cells (see Appendix Table H2 of OEHHA 2022 1 ), and five in vivo animal studies. Consistent positive associations between BPA exposure and 8-OHdG in various study populations (e.g., life stage, gender, geographic location) were reported in 13 human observational studies. Increased 8-OHdG levels in BPA-exposed rodents were also reported in three in vivo studies197-199; a dose-dependent and time-dependent relationship was reported by Cho et al. (2009). 198 In vitro evidence also shows similar associations between BPA exposure and 8-OHdG. For example, a significant increase in 8-OHdG (P < .01) was observed in mouse embryonic fibroblast cells treated with 50 μM BPA. 200 Statistically significant increases of 8-OHdG were observed in primary rat hepatocytes exposed to 10 µM of BPAQ, a reactive metabolite of BPA. 48
More than 100 publications were identified with data on ROS or RNS generation associated with BPA exposure in human cells in vitro, rodents in vivo, and rodent cells in vitro (details in Appendix Tables H3-H5 of OEHHA 2022 1 ). Overall, BPA was consistently associated with increased ROS (both intracellular and mitochondrial) or RNS levels in the majority of these studies. These findings are also consistent with an earlier review. 15 Significant increases of ROS were observed in LAPC-4 human prostate cancer cells exposed to 1 nM BPA, an “environmentally relevant low dose” per the study authors, for 15 minutes. 201 Dose-dependent increases of ROS and RNS were observed in some rodent in vivo studies,202-206 studies using human cells in vitro202,204,207,208 and rodent in vitro studies.206,209-212
In addition, increases in lipid peroxidation and reductions in GSH and antioxidant enzyme activities or levels have been reported in many in vivo and in vitro human and animal studies, as reviewed by Steffensen et al (2020), 12 Zhang et al (2022), 13 Amjad et al (2020), 14 and Gassman (2017), 15 and in recent primary research articles on BPA identified by OEHHA (see OEHHA 1 ).
Overall, BPA consistently induces oxidative damage to DNA (measured as 8-OHdG; Table 3) and increases ROS/RNS production (see Appendix Tables H3-H5 of OEHHA 2022 1 ) in numerous studies from human observational studies, studies using human cells in vitro, and rodent studies in vivo and in vitro, with concentration- or dose-dependent relationships in some studies. The Committee considered the evidence of BPA-induced oxidative stress to be strong, and highlighted that BPA induces oxidative stress at high doses, but also importantly, it does so at lower, non-cytotoxic doses. BPA induces ROS that appears to be involved in altering cell signaling pathways, promoting cell proliferation and survival, as well as DNA damage.
KC6: Induces Chronic Inflammation
Chronic inflammation associated with the development of cancer is a prolonged response to persistent infections or irritants that inflict cell death and tissue injury and can trigger cellular events associated with carcinogenesis, such as cellular transformation, promotion and survival of transformed cells, proliferation, invasion, angiogenesis, and metastasis.55,196,213 Many pro-inflammatory molecules have been identified, including cytokines such as tumor necrosis factor (TNF; formerly TNF-α), lymphotoxin-alpha (LT-α; formerly TGF-β) and several interleukins (ILs) (IL-1α, IL-1β, IL-6, IL-8, IL-18), chemokines, and others.196,214
We identified over 200 studies that investigated inflammation due to BPA exposure in humans and animals. Of these, we selected the following for review: human studies that measured inflammatory biomarkers in BPA-exposed populations, and animal studies with longer-term (≥28 days) exposure that measured inflammation either directly via histopathology, or as changes in inflammation biomarkers, or both.
Several human cross-sectional studies found a positive association between BPA levels and inflammatory biomarkers such as C-reactive protein, IL-6, and TNF. One cohort study reported that BPA was positively associated with increased levels of IL-6 in pregnant women although another cohort study in pregnant women did not report any significant associations. 67
BPA-Induced Tissue Inflammation (With Changes in Biomarkers of Inflammation, If Assessed) in Animal Studies In Vivo.
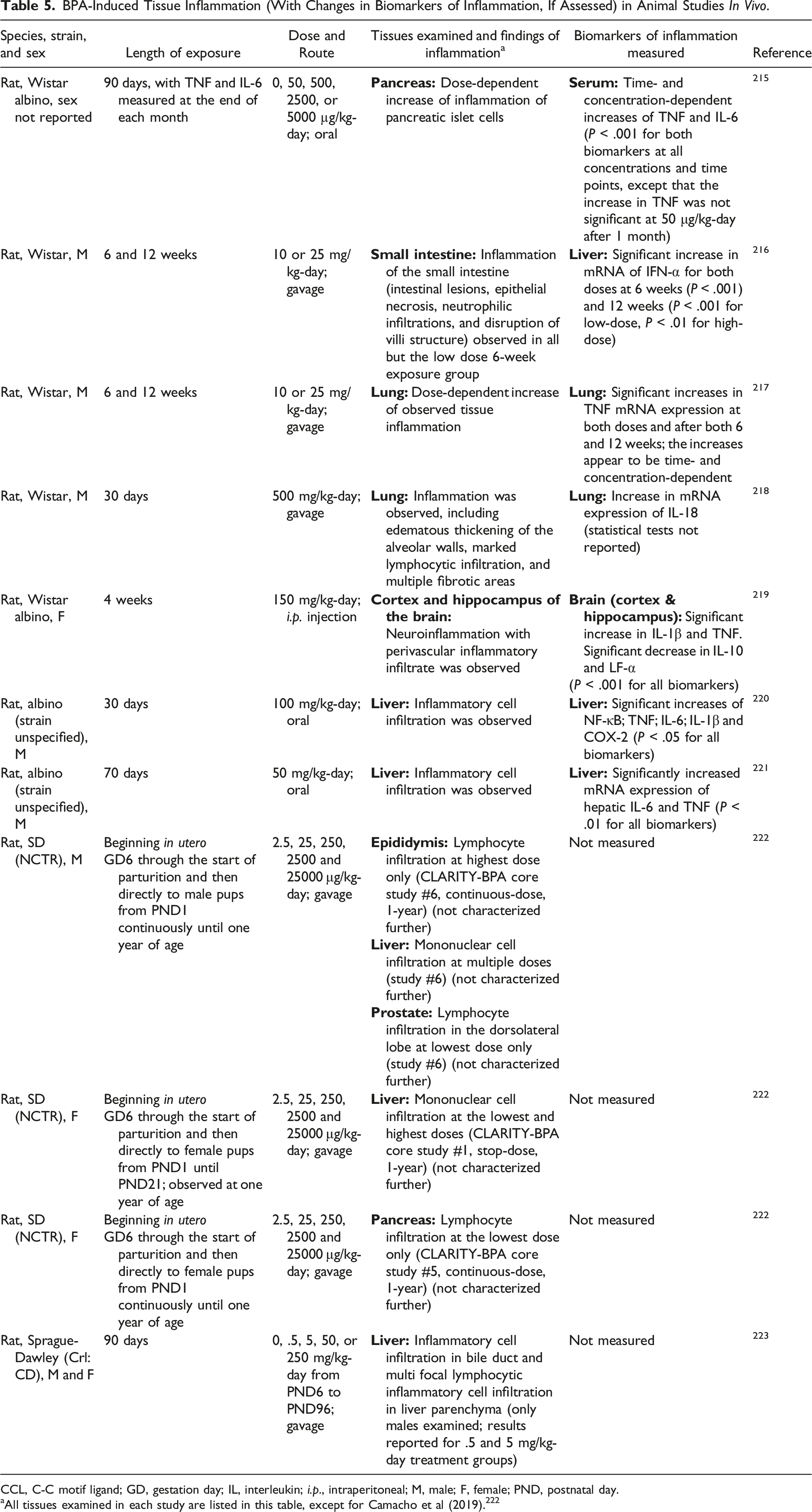
CCL, C-C motif ligand; GD, gestation day; IL, interleukin; i.p., intraperitoneal; M, male; F, female; PND, postnatal day.
aAll tissues examined in each study are listed in this table, except for Camacho et al (2019). 222
Overall, the Committee considered the animal evidence linking BPA exposure to chronic inflammation as robust, and the human evidence for KC6 as weak-to-modest.
KC7: Is Immunosuppressive
Immunosuppression can result in a reduction in the capacity of the immune system to respond effectively to tumor cells. Immunosuppression may allow neoplastic cells to escape immune surveillance and permit the survival and replication of these cells to form tumors. 55 Both the innate and adaptive parts of the immune system participate in immune surveillance, i.e., recognition and removal of malignant cells. The innate immune system is the first line of defense, and key components of the innate (or natural) immune system include natural immunoglobulin M (IgM) antibody-producing B1 or CD5+ cells, macrophages, mast cells, dendritic cells, and natural killer cells. 224 The adaptive immune system consists of a heterogeneous population of infiltrating lymphocytes such as T cells and other immune cells to modulate the anti-tumor response. 225
Our review found that several studies observed significantly decreased T and B cell populations in spleen or lymph nodes in rodents exposed to BPA. Additionally, a few in vitro studies have observed decreased proliferation in human cells following exposure to BPA (e.g., Tran et al (2020), 226 Jang et al (2020), 227 Yan et al (2008) 228 ). Exposure to BPA has been shown to decrease macrophage phagocytotic capacity in animal studies, as well as in studies of human and animal cells in vitro229–232 A few studies in primary human cells and rodents have demonstrated that BPA exposure reduces the chemotactic capacity of neutrophils.233–235 For dendritic cells, one study observed decreased endocytotic capacity in human cells after exposure to BPA, 236 while another study in mice observed decreased numbers of cells in spleen mesenteric lymph nodes. 237 One study found that BPA exposure caused a decrease in levels of splenic natural killer cells in rats. 238 Lastly, one study in mice and one study in juvenile rock bream reported decreased IgM levels after BPA exposure.239,240
While there is evidence for decreases in several immune responses, the Committee expressed that the overall evidence of immunosuppression is insufficient based on the small number of studies available for each endpoint.
KC8: Modulates Receptor-mediated Effects
Chemicals may modulate receptor-mediated effects in a variety of ways, including binding to and either activating or inactivating a receptor, altering receptor levels or function, altering levels of endogenous ligands that are available to bind to the receptor, or otherwise altering receptor-mediated gene expression or intracellular signaling. Many cellular receptors regulate critical cellular pathways, such as those involved in differentiation and proliferation, the disruption of which can contribute to carcinogenic processes. 55
Over 1600 publications were identified investigating modulation of receptor mediated effects caused by BPA. Detailed summaries of data, except those for estrogen receptors (ERs), can be found in the tables in Appendix J of OEHHA 2022. 1
Estrogen receptors (ERs)
BPA is a well-recognized xenoestrogen that mimics and interferes with the actions of estrogen. There is a large body of evidence from observational studies in humans, human cells in vitro, and animal studies in vivo and in vitro indicating that BPA modulates ER-mediated effects through several different ER subtypes and their downstream signaling pathways.16,17,29 While BPA is known to bind to cytosolic ERs and activate ER nuclear translocation and binding to estrogen response elements in DNA, additional ER-related activities of BPA have been identified.18,27,30
These non-canonical ER activities of BPA may explain many observations that are unexpected solely based on the classical ER pathway, such as the rapid onset of extra-nuclear responses, the low-dose effects, and the non-monotonic dose-responses. These latter two effects are also consistent with the female rat mammary tumor response seen in one study arm of the Consortium Linking Academic and Regulatory Insights on Bisphenol A Toxicity (CLARITY-BPA) core study, a 2-year cancer bioassay where rats were exposed to BPA in utero and postnatally by gavage for 21 days. 241 For example, BPA’s effects on membrane-associated estrogen receptors (mERs), G-protein coupled estrogen receptor (GPER/GPR30), and estrogen-related receptor gamma (ERRγ, an orphan nuclear receptor) may each alter different downstream genomic and non-genomic signaling pathways in a cell-type specific fashion. Additionally, BPA can induce epigenetic changes to regulate the expression of ERα, and cancer-related ER target genes.
Other nuclear receptors and hormones
Effects of BPA exposure on other nuclear receptors (androgen receptor, progesterone receptor, thyroid receptor, aryl hydrocarbon receptor, peroxisome proliferator-activated receptor, and pregnane X receptor) and hormones (estrogen, testosterone, progesterone, and prolactin) from various study designs were also reviewed and included within Appendix J of OEHHA 2022. 1
While BPA affected several endpoints, some consistent associations are listed below: • Increased progesterone receptor expression in human and animal studies in vitro (e.g., Samuelsen et al (2001)
242
; Schaefer et al (2010)
243
; Altamirano et al (2020)
244
; Kim et al (2012)
245
). • Antiandrogenic activity and interference with androgen receptor nuclear translocation in human studies in vitro (eg, Lee et al (2019)
246
; Teng et al (2013)
247
). • Decreased serum testosterone levels in male mice in vivo (e.g., Meng et al (2018)
248
; Park et al (2018)
249
). • Antagonistic effects on thyroid hormone receptor β in human studies in vitro (e.g., Moriyama et al (2002)
250
; Sun et al (2008)
251
). • Increased serum prolactin levels in rats in vivo (e.g., Hao et al (2011)
252
; Osman et al (2021)
253
). • Increased aryl hydrocarbon receptor expression in brain, kidney, spleen, and reproductive tissues in rodents exposed in vivo (Meng et al (2018)
248
; Gao et al (2020)
254
; Nishizawa et al (2005)
255
; Hsu et al (2019)
256
). • Increased peroxisome proliferator-activated receptor γ expression in human adipocytes and mouse preadipocytes in vitro and in liver tissue of rodents exposed in vivo (e.g., Boucher et al (2014)
257
; Ahmed and Atlas (2016)
258
; Long et al (2021)
259
). • Increased induction of pregnane X receptor activity in human studies in vitro (e.g., Vrzal et al (2015)
260
; Yu et al (2021)
261
; Kuzbari et al (2013)
262
).
Overall, the Committee noted that the evidence for BPA on receptor-mediated effects is strong for estrogen and estrogen-related receptors. For other receptors, the data were more limited.
KC9: Causes Immortalization
Carcinogens have been shown to increase the frequency of cell transformation from normal to malignant phenotypes and increase such cells’ capacity for tissue invasion. 55 This process can occur through the epithelial-to-mesenchymal transition (EMT), in which altered expression of the ratio of epithelial (e.g., E-cadherin) to mesenchymal (e.g., vimentin) genes allows cells to detach and invade surrounding tissue, a key step towards tumor metastasis. 263 Additionally, carcinogens can alter key factors that lead to cell immortalization, such as decreasing cellular senescence markers (e.g., p21, p53, etc.), and maintaining or extending telomere length through activation of telomerase. 55
There are several studies available on BPA that provide information relevant to its potential to cause immortalization. Two studies reported that BPA is able to induce cell transformation in Syrian Hamster Embryonic (SHE) cells,58,264 while two other studies in SHE cells and one in the A31-1-13 clone of BALB/c3T3 mouse embryo cells did not find significant alterations in cell transformation.265–267 BPA was able to increase cell invasion in three human primary cell lines.268-270 Additionally, increases in mesenchymal cell markers were observed in three human cancer cell lines271-273 and one human epithelial cell line after exposure to BPA, 274 and one study observed a decrease in expression of a cellular senescence gene in a human cancer cell line. 275 Results from studies of the effects of BPA on telomere length, telomerase expression or telomerase activity were mixed. In one cross-sectional study, higher urinary BPA levels were associated with shorter relative telomere length in adult women. 276 Five studies characterized alterations to telomerase expression, activity, or telomere length after BPA exposure in human cells in vitro. Two studies performed in primary human cells found decreases in telomerase activity, hTERT mRNA expression, or telomere length after exposure to BPA.107,226 In three studies performed in human cancer cell lines, increases in telomerase expression or activity were observed in cells exposed to higher BPA concentrations or for longer exposure durations, while no alterations were observed when cells were exposed to lower BPA concentrations or for shorter durations.277-279
Given the limited number of studies and inconsistencies in some endpoints, such as cell transformation and telomere length, the Committee noted that the evidence for BPA on cell immortalization was limited.
KC10: Alters Cell Proliferation, Cell Death or Nutrient Supply
Cancer is a disease involving uncontrolled cell growth. The types of effects indicative of KC10 include increased cell proliferation, decreased apoptosis, changes in growth factors, energetics, and signaling pathways related to cellular replication or cell cycle control, and increased angiogenesis, as described by Smith et al. (2016). 3
In vitro studies in human cells have reported various proliferative effects of BPA, including increased cell proliferation, changes in signaling pathways, alterations of apoptosis, angiogenesis, and glycolysis-based energy production. Studies in various cell types (normal, immortalized, and cancer) have shown that BPA at non-cytotoxic concentrations induces cell proliferation in normal human prostate fibroblasts, osteoblasts, and endothelial cells, as well as human breast, colon, liver, thyroid, lung, ovarian, endometrial, and cervical cancer cells.1,11,15 In several different human cancer cell lines, BPA has been shown to alter the levels of key proteins in cellular signaling pathways, including cyclins, cyclin-dependent kinases (CDKs), p53, and p21, which are related to cellular replication or cell cycle control.31,280-282 In addition, BPA has been shown to upregulate anti-apoptotic proteins and downregulate pro-apoptotic proteins in several human cancer cell lines. 31 Exposure to BPA was found to significantly increase the growth of human umbilical vascular endothelial cells and the number of segments and nodes of vessel-like structures in one study, 283 and to upregulate genes important for vascularization and angiogenesis in another study. 284 BPA exposure also increased expression of vascular endothelial growth factor in estrogen-sensitive breast cancer cells. 285 Exposure to low-dose BPA was found to promote glycolysis-based metabolism in human ovarian cancer cells, 286 exacerbate the metabolic switch observed in human bladder cancer-associated fibroblasts, 287 and increase glucose uptake and cell proliferation in breast cancer cells. 288
BPA has been observed to increase cell proliferation and hyperplasia in rats and mice, as shown in various studies. 1 For example, female rats exposed to BPA showed a statistically significant increase in cystic endometrial hyperplasia in the uterus and ductal hyperplasia in mammary gland,241,289-291 while male rats exposed to BPA showed a statistically significant increase in hyperplasia of bone marrow myeloid cells and prostate gland.241,292 In another study, female rats treated with BPA by gavage for 90 days showed significantly higher scores for proliferating cell nuclear antigen (PCNA)-positive nuclei in the myometrial layers of the uterus. 293 In mouse mammary tumor virus (MMTV)-erbB2/neu transgenic female mice, BPA exposure significantly increased the cell proliferation index in mammary gland epithelial cells, 294 while BPA exposure to female Balb/c mice resulted in mammary gland hyperplasia and endometrial adenomatous hyperplasia.295,296 Additional studies reporting increased cell proliferation in rodents are described in OEHHA 2022. 1
The available evidence for KC10 includes multiple studies showing consistent findings of cell proliferation in vitro and hyperplasia of target organ sites in vivo induced by BPA. Regarding the overall strength of evidence for KC10, the Committee considered the data to be moderate to strong.
Discussion
Here we have shown that applying the KCs approach enabled us to identify specific endpoints and mechanisms that may be pertinent to BPA’s potential carcinogenicity. Using this approach, the mechanistic data impart a clarity that is not readily apparent from the available human and animal studies.
BPA had evidence for all ten KCs, with some KCs (KC8, KC4, KC5, and KC10) having extremely large data sets, comprising hundreds of studies. Evidence for several of the KCs (such as KCs 1, 2, 5, 6, 8, and 10) was considered strong by the Committee, based on consistent findings from multiple studies and across species and test systems. For KCs 3, 7, and 9, the evidence was less clear and often the findings were inconsistent, or there were few studies available for a given endpoint. For KC4, there is consistent evidence of epigenetic changes, but it is difficult to draw conclusions regarding functional consequences. The mere number of KCs exhibited by a chemical is not indicative of its strength or likelihood of carcinogenicity. For some human carcinogens there are data for a single KC, while for other carcinogens there are data showing that they act via multiple KCs that are related to multiple mechanisms, causing various biological changes in the multistage process of carcinogenesis.3,297 In other words, the ability to act via multiple mechanisms may simply mean more pathways through which a chemical can contribute to cellular changes leading to carcinogenesis.
As described in more detail earlier, compelling evidence supports the potential involvement of receptor-mediated effects, oxidative stress, and increased cell proliferation, with consistent results from multiple testing systems. For example, with respect to KC8, studies have shown that BPA modulates ER-mediated effects through several different ER subtypes and their downstream signaling pathways; there were also several non-canonical effects. BPA was also consistently associated with oxidative damage to DNA, and ROS or RNS production (KC5) in numerous human observational studies, studies using human cells in vitro, and rodent studies in vivo and in vitro, with concentration- or dose-dependent relationships in some studies. For KC10, BPA-induced cell proliferation has been observed in many studies using human cells in vitro as well as studies in rats and mice.
Although there were fewer studies for other KCs, the data also strongly supported specific endpoints within these KCs. For example, many studies in both humans and animals demonstrated that BPA induces chronic inflammation (KC6), indicated by significantly increased levels of relevant biomarkers, including dose-dependent increases in animal studies at non-toxic doses and with longer exposure. Many animal studies had concomitant histopathology consistent with tissue inflammation. Another example is KC1, where consistent data on the formation of electrophilic and reactive compounds and the formation of DNA adducts in vivo and in vitro have been observed. In addition, strong support for DNA damage, chromosomal effects, and mutagenicity within KC2 was evident through many studies (including studies in humans).
The evidence also indicates that several KCs may be inter-connected and thus this analysis provides a possible map for the carcinogenic mechanisms of BPA.
Specifically, BPA metabolism data directly support and link KC1, KC2, and KC5. Metabolic oxidation of BPA generates several reactive metabolites, including BPAQ. BPAQ readily forms DNA adducts (BPA-3,4-quinone–guanine-N7 adduct) (KC1) in vivo and in vitro, likely leading to genotoxicity (KC2). Redox cycling of the BPA catechol-quinone couple can generate reactive oxygen species leading to oxidative stress (KC5). A similar mechanism has been reported for estrogens and the synthetic estrogen diethylstilbestrol (DES), which are associated with breast and other cancers.
298
These compounds also form N7-guanine adducts,57,298,299 and the quinone catechol couples of estrogen and DES can also undergo redox cycling to produce reactive oxygen species.
300
Additionally, BPAQ, along with its parent molecule BPA, has been shown to induce oxidative DNA damage in humans and animals (KC2 and KC5). Review of human observational studies demonstrated a consistent positive association between BPA exposure and 8-OHdG in various study populations (e.g., life stage, gender, geographic location) in most studies. Although some of BPA’s reactive metabolites are short-lived, continuous exposure to BPA may continually feed into this metabolic pathway and thus provide a steady stream of reactive metabolites. A summary of the KC-related biological activities from BPA metabolites is shown in Figure 3. Proposed interactions between oxidative metabolites of BPA and some KCs. Oxidative metabolism of BPA generates reactive and bioactive metabolites. The reactive metabolites result in DNA adducts, oxidative DNA damage, and reactive oxygen and nitrogen species. The bioactive metabolites display receptor mediated effects and induce cell proliferation.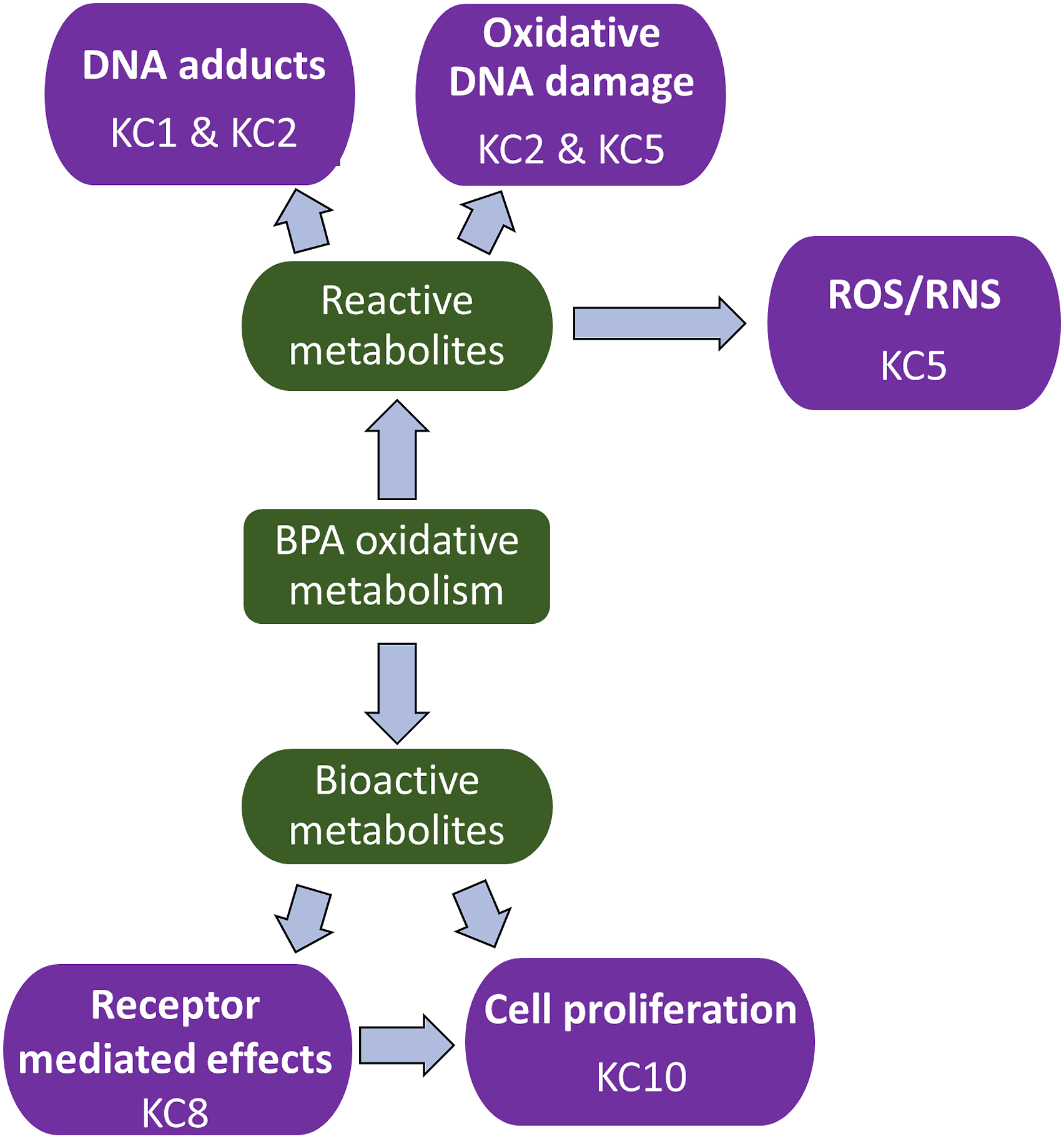
Mechanistic data for BPA also connect KC8, KC9 and KC10. As described in our summary and by numerous reviews, BPA modulates ER-mediated effects (KC8) through several different ER subtypes and their downstream signaling pathways.21,26,28 Modulation of ER is associated with alterations in genes involved in cell immortalization, increased cell invasion (KC9),19,268,301 induced cell proliferation/hyperplasia, inhibition of apoptosis, and increased cell migration (KC10).11,16-30 This is similar to estradiol, which is thought to act through binding to the ERα, resulting in stimulation of cell proliferation and initiation of mutations. 302 BPA’s effects may be amplified or enhanced by estrogenic BPA metabolites, such as MBP and HCA.
Several other KCs of BPA in addition to those already mentioned were identified in our review and may be connected. For example, reduced DNA repair capacity (KC3) may exacerbate BPA’s genotoxicity (KC2) and increase damage induced by oxidative stress (KC5). In BPA treated human peripheral blood mononuclear cells (PBMCs), significant residual DNA damage was observed after a 120-min recovery period.
108
In BPA treated human prostate epithelial cells, expression levels of genes encoding three DNA repair proteins (MyH, OGG1, and APE-1) involved in base excision repair, the key mechanism to repair 8-OHdG, were decreased, with the decrease for MyH being statistically significant.
111
Many genes that are shown to be epigenetically modulated by BPA (KC4) are key players for other KCs (e.g., ESR1 (KC8), IL-6 (KC6), mitogen activated protein kinase 1 (MAPK1, KC10). In another example, oxidative stress (KC5) may exacerbate chronic inflammation (KC6).
303
Taken together, the evidence supports the interaction of several KCs and BPA’s multi-faceted mechanisms of action. Some examples of such interaction of KCs, mediated by BPA or its metabolites, are shown in Figure 4. Examples of how some KCs exhibited by BPA are interconnected. Some of the depicted effects may be mediated by BPA metabolites. This is a visualization of how some of the evidence is interconnected.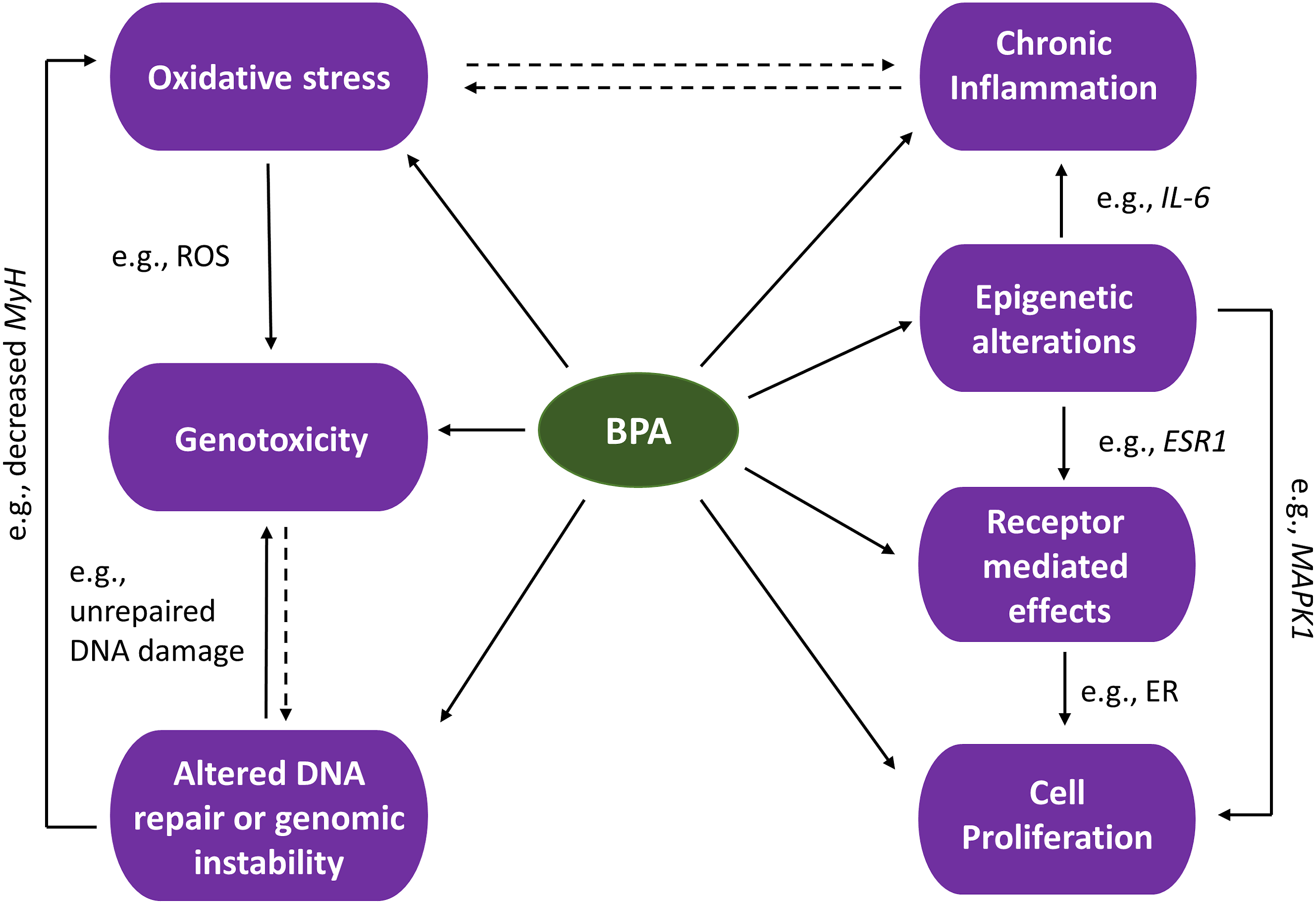
At IARC, strong mechanistic evidence plays a significant part in the overall evaluation of carcinogenic hazards. 4 While hazard identification of carcinogens traditionally relies heavily on evidence from cancer epidemiological studies and animal cancer bioassays, these studies are not always available or adequately done. It is anticipated that mechanistic data will play an increasingly important role in the future because of their expected increasing volume, diversity, and relevance. Furthermore, the KCs approach can serve as a bridge to incorporating both traditional and novel types of toxicity tests. The question remains how much weight should be given to mechanistic data in the absence of sufficient evidence from other data streams, and, in such circumstances, whether a determination of carcinogenicity can be made.
Conclusion
In the case of BPA, the Committee as a whole did not find the carcinogenicity evidence overall reached the threshold of “clearly shown”. They considered the human data inconclusive and were mixed regarding the strengths of the animal data. Several committee members found that the evidence for a number of KCs were either strong, or moderate to strong. The mechanistic data show that BPA shares many characteristics of known human carcinogens, and thus it may be plausible to consider that BPA might contribute to the carcinogenic process via electrophilicity, genotoxicity, oxidative stress, chronic inflammation, receptor mediated effects, and cell proliferation. While it still remains unclear how mechanistic data, either alone or in conjunction with less than sufficient human and/or animal data, will be utilized by individual entities that make cancer hazard determinations, the KCs approach facilitates a broad, holistic, and systematic consideration of mechanistic evidence.
Footnotes
Acknowledgments
The authors thank Dr Nathalie Pham and Dr Ida Flores-Avila for their thoughtful review and helpful input on this work.
Author Contributions
Ricker, K. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Cheng, V. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Hsieh, C. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Tsai, FC. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Osborne, G. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Li, K. contributed to conception and design contributed to acquisition, analysis, and interpretation, and drafted manuscript; Yilmazer-Musa, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Sandy, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Cogliano, V. contributed to conception and critically revised manuscript; Schmitz, R. contributed to design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Sun, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Note
The views expressed are those of the authors and do not necessarily represent those of the Office of Environmental Health Hazard Assessment (OEHHA), the California Environmental Protection Agency, or the State of California.
