Abstract
Research has shown that exposure to bisphenol A (BPA), a widely used plasticizer, can lead to meiotic errors, resulting in poor reproductive cell quality and infertility. Health-related concerns have prompted the search for BPA alternatives; however, evidence suggests that currently used BPA analogs, such as bisphenol S (BPS), may pose similar risks to human health. While the effects of BPA on female fertility are well documented, the impact of BPA exposure on sperm quality is poorly understood. To better understand the effects of bisphenol analogs on spermatogenesis, we synthesized a less investigated BPA analog, tetramethyl bisphenol F (TMBPF), and compared its reprotoxic potential to that of widely used BPA and BPS using
Introduction
Infertility affects an estimated 17.5% of reproductive-aged couples worldwide (Cox et al., 2022). Growing evidence indicates that bisphenol A (BPA), a known endocrine disruptor, not only interferes with hormone signaling but also negatively impacts the quality of reproductive cells (Machtinger and Orvieto, 2014; Matuszczak et al., 2019). Since the commercial potential of BPA was recognized in the 1950s, it has been extensively utilized in the production of epoxy resins and polycarbonates (Bilyeu et al., 2005). These polymers have found applications in a wide array of products ranging from food and drink packaging to clothing, electronics, and medical and safety devices (Guimarães et al., 2023). Although BPA has found great utility as a plasticizer, it may leach from damaged and heated plastics (Lopez-Cervantes, 2003), resulting in increased BPA exposure in the general population. Accumulating evidence from both animal and human studies indicates that BPA impairs meiotic fidelity, resulting in poor quality female reproductive cells (Machtinger and Orvieto, 2014; Matuszczak et al., 2019). Although BPA-related health concerns have prompted the search for safer alternatives, research has shown that the most frequently used commercial BPA alternative, bisphenol S (BPS), affects oocyte quality and overall female reproductive health in a similar fashion as BPA (OEHHA, 2023).
The primary route of exposure for both BPA and BPS is through the ingestion of food or beverages contaminated by leachate from packaging materials, while other routes include skin contact and inhalation of indoor dust (Ma et al., 2019). The presence of BPA and BPS has been confirmed in human blood and urine. Median urinary levels of BPA and BPS in the adult U.S. population are approximately 1.2 ng/mL (interquartile range: 0.6–2.5 ng/mL) and 0.4 ng/mL (0.1–0.9 ng/mL), respectively (Lehmler et al., 2018). BPA concentrations in human blood range from 0.5 ng/mL to 10.0 ng/mL, while BPS serum concentrations vary from 0.2 ng/mL (50th percentile) to 2.9 ng/mL (95th percentile) (OEHHA, 2023; Vandenberg et al., 2010). During pregnancy, BPA levels can be also detected in human reproductive and fetal tissues, with concentrations ranging from 0.2 ng/mL to 9.2 ng/mL in fetal plasma and from 1.0 ng/g to 104.9 ng/g in placental tissue (Schönfelder, 2002).
While the effects of BPA on female fertility are well documented, research on the impact of BPA exposure on sperm quality has yielded mixed results, with some studies indicating a significant association with poor semen quality, while others show no such correlation (Castellini et al., 2020; Matuszczak et al., 2019). Specifically, research conducted on animal models suggests that BPA might negatively impact male fertility by altering sperm morphology, decreasing sperm count and its motility (Cariati et al., 2019; Davis et al., 2023; Liu et al., 2021; Rahman et al., 2015). Evidence for the potential effects of BPA on human reproductive health largely comes from observational epidemiological studies (Castellini et al., 2020). Whereas some studies have reported a link between increased urinary BPA excretion and changes in sperm morphology, count, and/or motility (Chen et al., 2022; Li et al., 2011; Omran et al., 2018), other studies have shown no significant associations with any standard sperm quality parameters (Adoamnei et al., 2018; Benson et al., 2021). In addition, recent studies investigating the impact of BPS on male fertility have resulted in inconsistent findings (Benson et al., 2021; Chen et al., 2022; Davis et al., 2023). Given our limited understanding of the effects of bisphenol analogs on male fertility, more research is needed to fully elucidate their impact on spermatogenesis.
In this study, we used Structures of BPA, BPS, and TMBPF. Abbreviations: BPA, bisphenol A; BPS, bisphenol S; TMBPF, tetramethyl bisphenol F.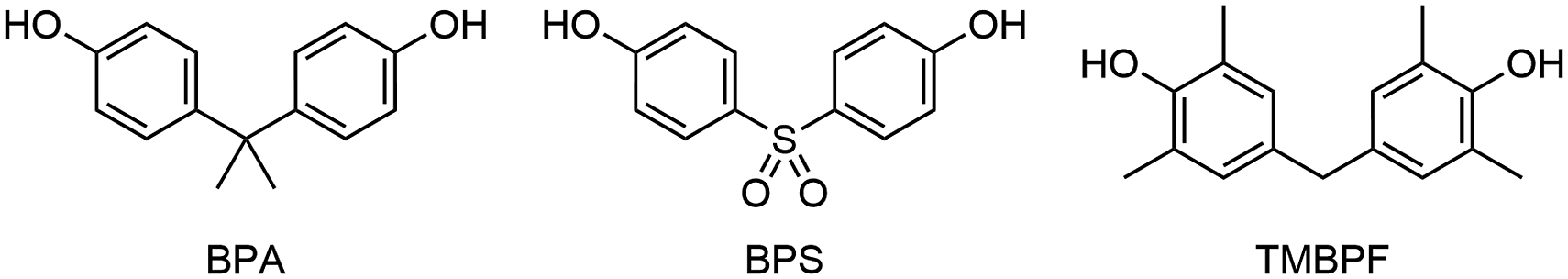
Materials and methods
Synthesis and characterization of TMBPF
General methods
BPA, BPS, and all chemicals used in the synthesis of TMBPF were purchased from Sigma-Aldrich and used without further purification. Thin-layer chromatography (TLC) was performed on polyester-backed plates pre-coated with silica gel containing a fluorescent indicator (200 μm layer thickness and 5–17 μm particle size). Developed TLC plates were visualized using a shortwave UV lamp (254 nm). Nuclear magnetic resonance (NMR) data was recorded using a Varian Mercury 400 MHz spectrometer. Chemical shifts are reported in parts per million (ppm) and were referenced to the DMSO-d6 solvent peak. High-resolution mass spectrometry data was recorded using a Thermo Orbitrap Exploris™ 120 mass spectrometer.
Synthetic methodology
The synthetic protocol used to prepare TMBPF was adapted from a previously published method (Singh et al., 2014). 2,6-Dimethylphenol (1.00 g, 8.19 mmol) was dissolved in petroleum ether (8.0 mL) in a round-bottomed flask containing a magnetic stir bar. An aqueous solution of formaldehyde (37% w/v, 1.40 mL, 13.7 mmol) and concentrated HCl (1.0 mL) were subsequently added to the reaction mixture, which was stirred at room temperature for 2.5 h. The reaction was quenched with 90 mL of distilled water and stirred for an additional 15 min. During this time, a white solid product precipitated from the reaction mixture. After cooling the reaction mixture overnight at −20°C to induce further precipitation, the white solid product was collected via suction filtration and analyzed using TLC (Rf = 0.35; 1:4 ethyl acetate/hexane eluent). The desired TMBPF product was afforded in 36% yield (0.383 g, 1.49 mmol) with no further purification required.
Characterization of TMBPF
1H NMR (DMSO-d6, 400 MHz): δ (ppm) 7.91 (s, 2H, OH), 6.69 (s, 4H, Ph), 3.55 (s, 2H, CH2), 2.09 (s, 12H, CH3); 13C{1H} NMR (DMSO-d6, 100 MHz): δ (ppm) 151.16, 132.49, 128.34, 123.98, 39.83, 16.71; HRMS (ESI)
C. elegans maintenance
Bisphenol treatment
The bisphenols used in the study were dissolved in 100% ethanol. The animals were treated in a liquid medium containing M9 buffer, concentrated OP-50 (Parodi et al., 2015), and bisphenol analogs. The final concentration of bisphenols in the medium was 0.5 mM, while the final concentration of the vehicle, 100% ethanol, was 0.5%. The external concentration of 0.5 mM was selected because it was previously demonstrated that this concentration, following uptake in
Total brood size assay
Following exposure, three males were crossed with an L4-stage female
Germline staining assay
Following treatment, males were fixed in 5 μL of 90% aqueous ethanol on a glass slide. Once the ethanol evaporated, 5 μL of staining solution (1 mg/mL DAPI in 90% ethanol) was applied to the slide. The slide was then incubated in the dark for 5 min before a cover slip was placed and sealed with nail polish. Z-stacks of each germline were collected at 40X using a fluorescence microscope (All-in-One Fluorescence Microscope BZ-X800, Keyence, Japan), and the number of germline nuclei was counted using ImageJ software.
Sperm size assay
This assay was performed according to the published protocol (Hammerquist et al., 2021). Briefly, treated males were transferred into 15 μL of SM buffer containing dextrose on a microscope slide. The worms were dissected using 27-gauge needles and the released spermatids were covered with a cover slip, which was sealed with nail polish. The spermatid images were captured using a 100x objective and a DIC filter (All-in-One Fluorescence Microscope BZ-X800, Keyence, Japan). The diameter and cross-sectional area of the spermatids were measured using ImageJ software.
Spermatid activation assay
This assay was performed according to the published protocol (Hammerquist et al., 2021). Briefly, treated males were dissected in 15 μL of SM buffer containing bovine serum albumin (BSA) on a microscope slide. The released spermatids were exposed to 400 ng/µL pronase and incubated in a humid chamber for 10 min. The number of spermatids observed with a pseudopod was counted under a DIC microscope (All-in-One Fluorescence Microscope BZ-X800, Keyence, Japan). The rate of spermatid activation was calculated as the ratio of the number of sperm with a pseudopod to the total number of spermatids observed.
Data analysis
One-way ANOVA with Tukey’s post hoc test was used for parametric data, while a non-parametric Kruskal–Wallis test followed by Dunn’s multiple comparisons was performed to analyze data if ANOVA assumptions could not be met. A
Results
Synthesis and characterization of TMBPF
TMBPF was prepared by adapting a published electrophilic aromatic substitution protocol involving 2,6-dimethylphenol, formaldehyde, and an acid catalyst (Figure 2) (Singh et al., 2014). The TMBPF product was characterized using 1H and 13C{1H} NMR spectroscopy and high-resolution mass spectrometry to ensure its high purity and identity prior to use in toxicity assays. Electrophilic aromatic substitution reaction used to prepare TMBPF. Abbreviation: TMBPF, tetramethyl bisphenol F.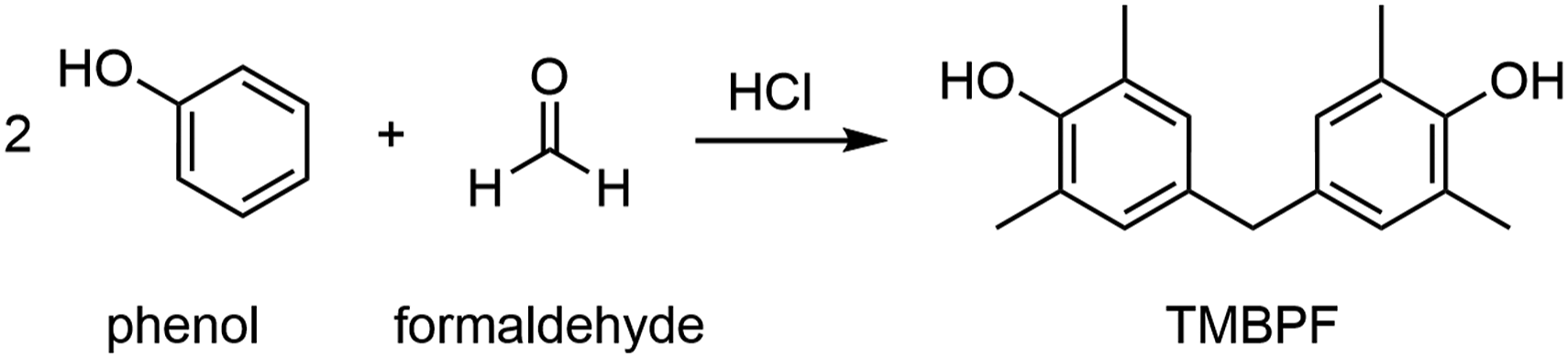
Effect of BPA, BPS, and TMBPF exposure on brood size
To evaluate the effect of bisphenol analogs on male fertility, we first quantified the number of fertilized eggs laid by Effect of bisphenols on fertility and embryonic lethality. (a) Number of fertilized eggs laid in each treatment group. (b) Brood size of 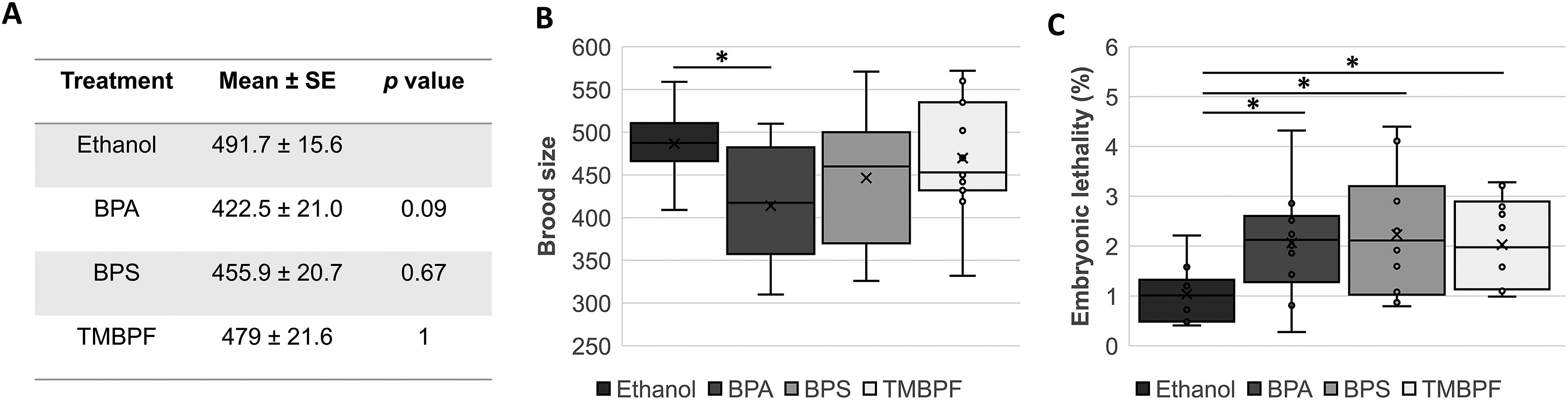
To investigate the effect of bisphenol analogs on brood size, we observed the hatched larvae. As shown in Figure 3(B), all treatment groups exhibited a trend toward a reduced brood size. However, only BPA exposure resulted in a statistically significant decrease in the number of viable progeny (ethanol: 486.5 ± 15.2; BPA: 413.9 ± 20.8; BPS: 446.5 ± 21.3; and TMBPF 469.7 ± 20.9 viable offspring).
Given that embryonic lethality is indicative of meiotic defects and an increase in the incidence of chromosomal aberrations in embryos, we quantified the percentage of unhatched fertilized eggs. All treatment groups experienced a significant increase in embryonic lethality compared with the control (Figure 3(C)). The percentages of unhatched eggs were as follows: 1.1 ± 0.2% in ethanol-treated males, 1.5 ± 0.3% in BPA, 2.1 ± 0.4% in BPS, and 1.9 ± 0.3% in TMBPF treatment.
Assessment of germline nuclei number in males exposed to bisphenol treatments
To further investigate the decrease in brood size, we first assessed the germline size by comparing the total germline nuclei count of males exposed to different bisphenol treatments with that of ethanol-treated males. As shown in Figure 4, the overall germline sizes of the males exposed to BPA, BPS, and TMBPF were not significantly different from the control (ethanol: 277.2 ± 15.0 nuclei; BPA: 268.9 ± 8.5 nuclei; BPS: 285.1 ± 18.2 nuclei; and TMBPF: 255.6 ± 17.5 nuclei). Effect of bisphenol treatments on germline. (a) Total number of germline nuclei in 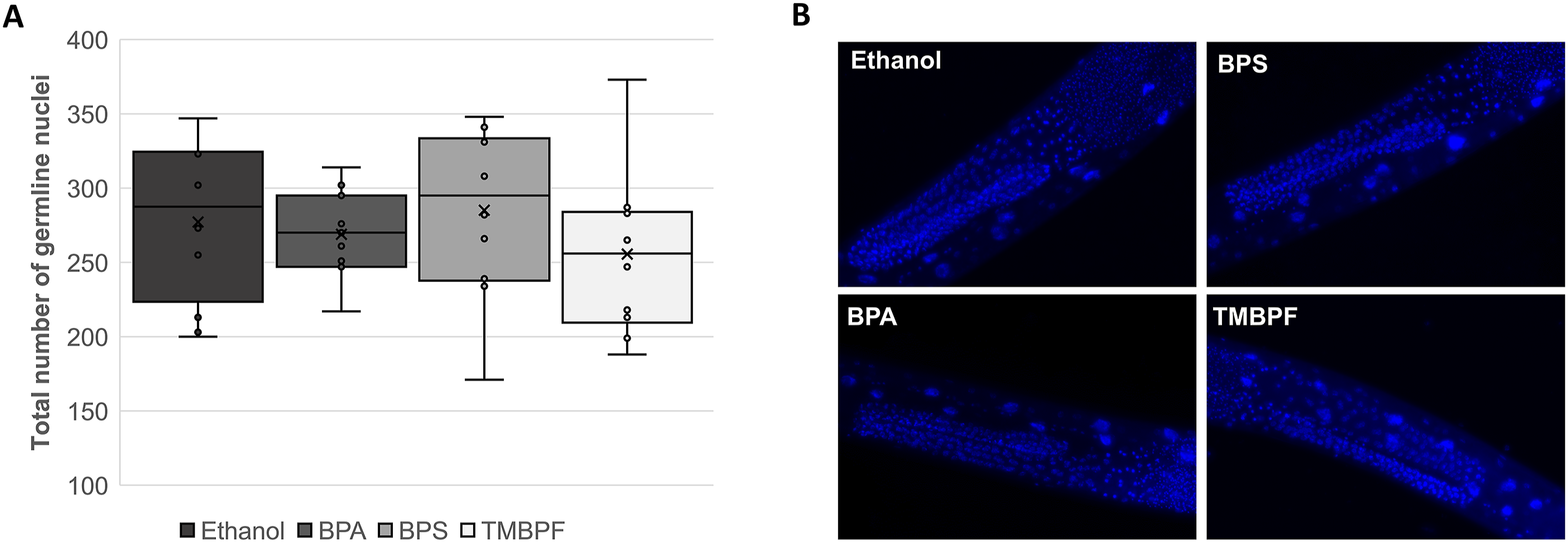
Effect of bisphenol treatments on spermatid size and morphology
To evaluate whether a reduced reproductive capacity was due to changes in sperm size and morphology, we measured spermatid diameter and cross-sectional area. As shown in Figure 5, control group males had an average spermatid diameter of 6.2 ± 0.03 µm and spermatid cross-sectional area of 28.06 ± 0.24 µm2. While the spherical shape of spermatids was not affected by the treatments, all three bisphenol exposures significantly reduced the diameter of the spermatids (BPA: 5.9 ± 0.03 µm; BPS: 5.9 ± 0.03 µm; TMBPF: 5.9 ± 0.03 µm; Effect of bisphenol exposure on spermatid size. (a) Spermatid diameter in 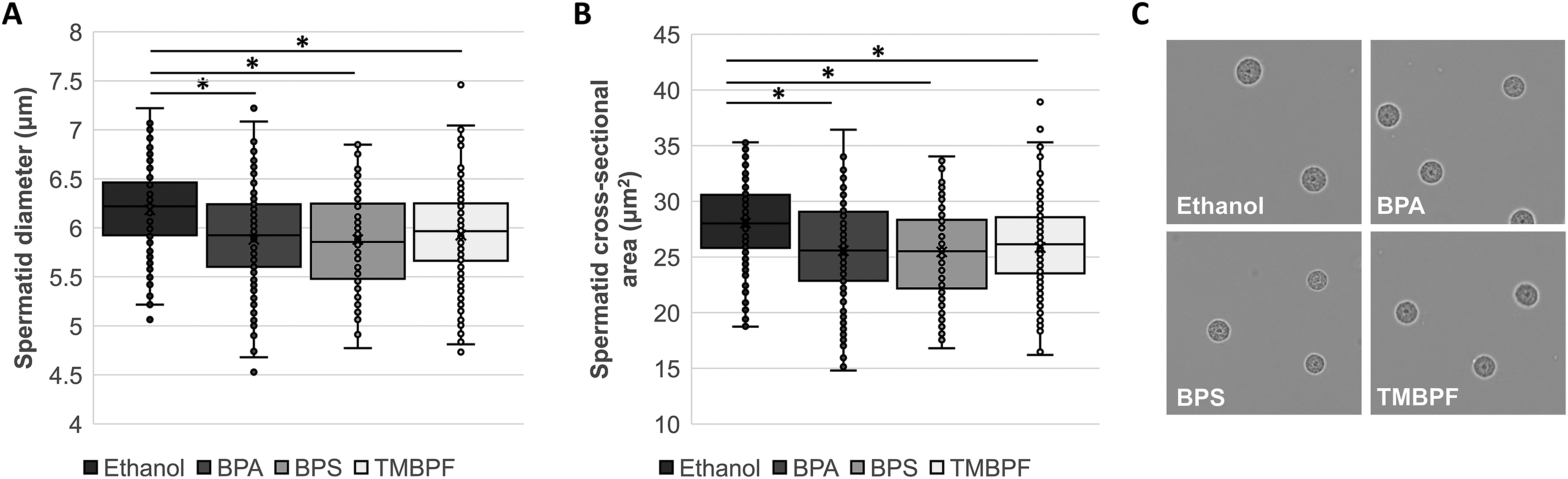
Assessment of spermatid activation in males exposed to bisphenol treatments
To investigate sperm motility, we determined the rate of spermatid activation following exposure to the bisphenol treatments. As shown in Figure 6, we observed 96.96 ± 0.33% spermatid activation in ethanol-exposed worms, 89.10 ± 4.25% in BPA-exposed worms, 94.36 ± 1.76% in BPS-exposed worms, and 97.03 ± 0.48% following TMBPF treatment. A statistically significant reduction in the rate of spermatid activation was observed only in animals exposed to BPA ( Effect of bisphenol exposure on spermatid activation. (a) Rate of spermatid activation in 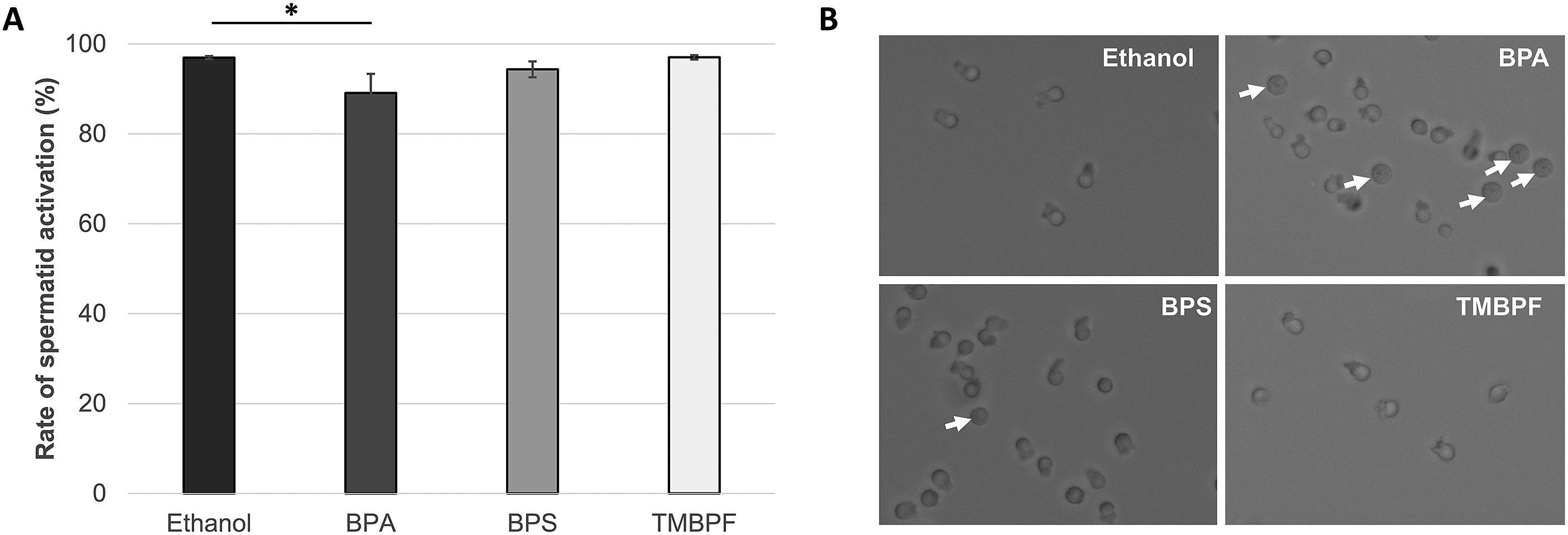
Discussion
Despite the growing body of evidence linking BPA with female infertility (Machtinger and Orvieto, 2014; Matuszczak et al., 2019), research on the impact of BPA exposure on male fertility shows inconsistent results requiring further investigation. Previous studies on animal models have shown that BPA exposure adversely affects sperm count, viability, and motility in animals such as mice, rats, and bulls (Cariati et al., 2019; Davis et al., 2023; Liu et al., 2021; Rahman et al., 2015). However, human studies have yielded mixed results, with some observing a negative impact on certain sperm quality parameters, while others finding no association (Adoamnei et al., 2018; Benson et al., 2021; Chen et al., 2022; Li et al., 2011; Omran et al., 2018).
In recent years,
The number of offspring is often used as a sensitive endpoint marker to assess the reprotoxic potential of chemicals and predict defects in the reproductive capacity of
Although exposure to reproductive toxins can sometimes impact gonad size, in the present study, none of the evaluated bisphenol analogs significantly affected the total germ cell number. The reduction in gonad size previously reported to contribute to decreased reproductive capacity in BPA- and BPS-exposed
Given the observed reduced fecundity and increased embryonic lethality, we next assessed spermatid size, morphology, and activation. Spermatid size has been shown to affect sperm fertilization competence in
Taken together, our findings indicated that exposure to BPA, BPS, and TMBPF at biologically relevant concentrations may have varying impacts on the reproductive health of male
Footnotes
Acknowledgments
The authors thank Dr Nicholas Peraino at Wayne State University for mass spectrometry service, which was supported by the National Institutes of Health under award number R01GM098285. We thank Dr Jim Woodworth at Lawrence Technological University for NMR spectroscopy support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (R01GM098285) and NIH Office of Research Infrastructure Programs (P40 OD010440). Research reported in this publication was also supported by the National Institute of Environmental Health Sciences of the National Institutes of Health under Award Number R15ES034520. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
