Abstract
Electrocardiographic evaluation is performed in rhesus monkeys to establish the cardiovascular safety of candidate molecules before progressing to clinical trials. These animals are usually immobilized chemically by ketamine (KTM) and tiletamine-zolazepam (TZ) to obtain a steady-state heart rate and to ensure adequate human safety. The present study aimed to evaluate the effect of these anesthetic regimens on different electrocardiographic parameters. Statistically significant lower HR and higher P-wave duration, RR, QRS, and QT intervals were observed in the KTM-anesthetized group in comparison to TZ-anesthetized animals. No significant changes were noticed in the PR interval and p-wave amplitude. Sex-based significance amongst these parameters was observed in male and female animals of TZ- and KTM-anesthetized groups. Regression analysis of four QTc formulas in TZ-anesthetized rhesus monkeys revealed that QTcNAK (Nakayama) better corrected the QT interval than QTcHAS (Hassimoto), QTcBZT (Bazett), and QTcFRD (Fridericia) formulas. QTcNAK exhibited the least correlation with the RR interval (slope closest to zero and r = .01) and displayed no statistical significance between male and female animals. These data will prove useful in the selection of anesthetic regimens for chemical restraint of rhesus monkeys in nonclinical safety evaluation studies.
Keywords
Introduction
Nonhuman primates (NHPs) are humans’ closest known extant relatives.1-3 Along with preclinical rodent studies, information obtained from NHPs and/or other nonrodent studies is often required in establishing efficacy, safety, and/or toxicity of candidate pharmaceuticals or biological formulations before progressing onto clinical trials. For certain biological research molecules, NHPs are the only relevant animal models that can be used in the drug development process.4-12 An Old World nonhuman primate, the rhesus monkey (Macaca mulatta) is one of more than 20 species of NHPs identified under the genus Macaca.7,13 Rhesus monkeys were one of the first studied primate species for understanding human physiology in the 1800s. They have since been extensively employed in translational research.6,14
Cardiovascular safety testing is an important component of the battery of risk evaluation strategies employed in any drug development process. 15 ICH S7A and S7B guidelines direct on procedures employed for assessment of drug effects on hemodynamic parameters along with central nervous system (CNS) and respiratory assessment and assessment of proarrhythmic risk of drugs (QTc prolongation and drug-induced arrhythmias).15-17 Drug-induced QT prolongation is a major cardiovascular concern in the safety assessment of novel drugs and regulatory authorities worldwide warrant a thorough pro-arrhythmic risk evaluation of test molecules.18,19 Rarely, drugs trigger different arrhythmias such as bradyarrhythmia, atrial fibrillation/flutter, atrial tachycardia, monomorphic ventricular tachycardia, Brugada syndrome, and Torsades de Pointes (TdP).20,21 Drug-induced delay in cardiac repolarization can be associated with the development of TdP and other ventricular arrhythmias. Such a delay can be measured as a prolongation of the QT interval on an electrocardiogram (ECG).22,23
ECG examination is an effective tool in assessing the proarrhythmic effects of cardiac and non-cardiac drugs. ECGs play a vital role in regulatory requirements of safety assessment, wherein new drugs undergo a detailed evaluation for their potential to affect ECGs in nonclinical (toxicology and safety pharmacology)17,23 and clinical (thorough QT) studies. 24 NHPs and humans possess anatomical and genetic similarities. 10 Rhesus monkeys share more than 95% of DNA sequence homology with humans. 10 Placement of the monkey heart in the thoracic cavity, coronary distribution, internal structure, and cardiac repolarization processes resemble that of the human heart. These similarities make rhesus monkeys an important and relevant animal model for understanding the effects of test molecules on the human heart.6,10,25-28
To obtain a steady-state heart rate in rhesus monkeys and to minimize harm to animals or personnel while handling conscious animals, electrocardiographic examination can be performed in rhesus monkeys under anesthesia.28,29 Ketamine (KTM) and tiletamine-zolazepam (TZ) are two of the most commonly used anesthetics in NHPs. These agents induce short-term anesthesia required for performing various experimental procedures such as physical examination, ophthalmic examination, and hemodynamic parameters monitoring.29,30 KTM, a dissociative anesthetic, is used in a wide range of NHP species.30,31 KTM induces immobility and analgesia in animals sufficient for short non-invasive procedures. Owing to its wide safety margin, KTM can be used alone or in combination with other anesthetic/analgesic drugs.29,31-33 TZ is a combination regimen of the dissociative anesthetic, tiletamine, and a benzodiazepine-agonist, zolazepam. TZ induces slight tranquilizing action and strong analgesia. TZ induces anesthesia at lower doses compared to KTM and has a comparatively longer duration of action.29,30
Both of these agents have been broadly used in immobilizing rhesus monkeys for performing noninvasive experimental procedures. Although information is available on heart rate and blood pressure, data on the effect of these anesthetic agents on other electrocardiographic parameters of rhesus monkeys is scarce. Through this retrospective study, we have aimed to analyze data on various electrocardiographic parameters such as heart rate; p-wave amplitude and duration; RR, QT, PR, and QRS intervals; and corrected QT intervals obtained from KTM- and TZ-anesthetized rhesus monkeys used in different toxicity studies.
Materials and Methods
Animals
Body Weight Specifications.
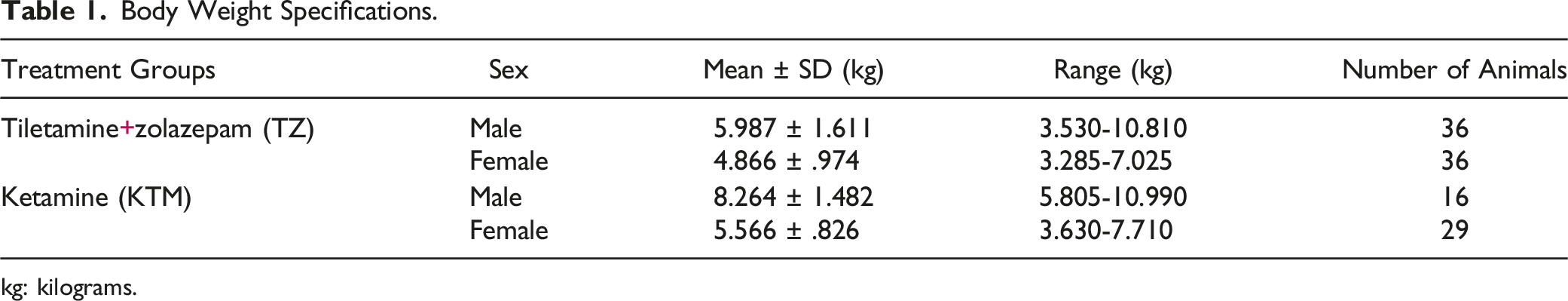
kg: kilograms.
Ethics and Accreditations
These studies were approved by the Institutional Animal Ethics Committee and the Committee for Control and Supervision of Experiments on Animals, Government of India. All experimental procedures involving rhesus monkeys were conducted according to the CCSEA guidelines. 34 The test facility is accredited with a GLP certificate from the National GLP Compliance Monitoring Authority (NGCMA), Government of India, for the conduct of toxicity studies and is additionally accredited by AAALAC International for animal ethics.
Anesthetics
Before recording of ECGs, animals were identified and restrained by the squeeze-back mechanism in home cages by trained personnel and injected by a veterinarian with either ketamine hydrochloride (single dose: 10 mg/kg, i.m.; Ketamax, Troikaa Pharmaceuticals Limited, Ahmedabad, India) or with a combination of tiletamine hydrochloride and zolazepam hydrochloride (single dose: 5 mg/kg i.m.; Zoletil, Virbac Animal Health India Private Limited, Mumbai, India).
ECG Recording
Snapshot ECGs were recorded in the dorsal recumbency position from anesthetized monkeys. Electrode gel was applied to the electrodes to improve conduction. An electrocardiograph machine, CARDIOVIT AT-1(VET) (Schiller AG, Switzerland, software version 8.02) was used to record ECGs. Traces were recorded at a speed of 25 mm/s and ECG sensitivity at 5 mm/mV. The instrument automatically assessed ECG traces. Twelve lead ECGs were recorded; HR and ECG intervals were determined from lead II. The leads were attached at commonly used positions: one electrode each on four limbs and six electrodes on the chest (starting at midline and following the direction of the heart, sequentially) and away from the abdomen to avoid artefacts in the ECG traces. All the procedures were performed in accordance with the standard operating procedures (SOPs) in place at the institution.
ECG Data
Different QT Correction Formulae Used in This Study.

Statistical Analysis
Statistical analysis was performed on GraphPad Prism software (version 9.0.1, 225; San Diego, USA). For KTM- and TZ-anesthetized rhesus monkeys, parameters such as heart rate, RR interval, PR interval, QRS interval, QT interval, and P-wave amplitude and duration were analyzed for normality and lognormality, followed by two-tailed Student’s t test with Welch’s correction or Mann–Whitney U test. Statistical analysis was performed to evaluate the significance between individual parameters of TZ- and KTM-anesthetized groups and to determine the significance between the sexes. P values less than .05 indicated statistical significance.
For TZ-anesthetized rhesus monkeys, linear regression analysis was performed on all four QTc formulae. The correlation between the RR interval and QTc in each formula was assessed using Pearson’s correlation coefficient analysis. The QTc and RR intervals were graphed on a scatter plot, with QTc (milliseconds) on Y axis and RR interval (milliseconds) on X-axis. Different QTc-RR interval scatter plots were generated, one for each QTc formula. The slope of the QTc-RR regression line for each QTc formula was determined and used to compare QTc formulae.
Animals were further divided into four sets: heart rate: >160 bpm and ≤160 bpm; and sex: males and females. QTc-RR scatter plots, means, standard deviations, r-value, and P value were evaluated for each group. Fit plots of QTc-RR variables were created with 95% confidence limits and 95% prediction limits to understand the fitness of a particular correction model in rate correction of QT intervals using Statistical Analysis Software (version 9.4; Cary, NC, USA).
Results
Detailed physical examinations of the animals did not reveal any abnormalities in the present study.
Electrocardiographic Parameters in Anesthetized Indian Rhesus Monkeys.
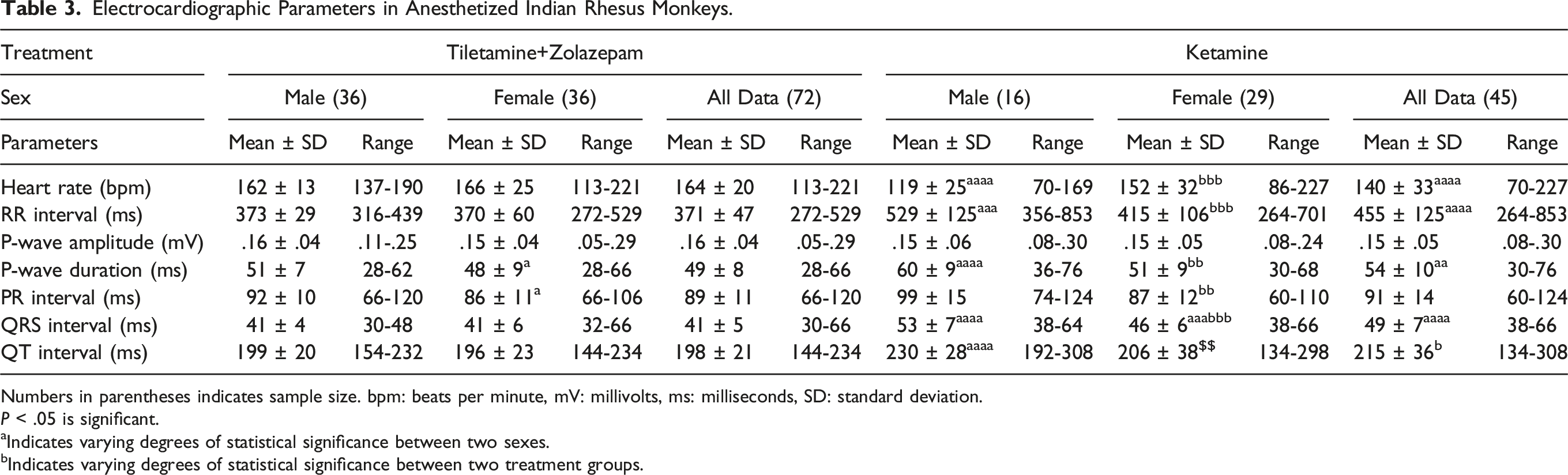
Numbers in parentheses indicates sample size. bpm: beats per minute, mV: millivolts, ms: milliseconds, SD: standard deviation.
P < .05 is significant.
aIndicates varying degrees of statistical significance between two sexes.
bIndicates varying degrees of statistical significance between two treatment groups.
Sex-based significance amongst these parameters was observed in male and female animals of TZ- and KTM-anesthetized animals in addition to the significant difference between the same sexes of both TZ- and KTM-anesthetized groups. While only P-wave duration and PR interval were found to be significant between both sexes of the TZ-anaesthetized group, parameters such as P-wave duration and HR, RR, PR, QRS, and QT intervals displayed sex-dependent significance in the KTM-anaesthetized group of animals. Comparison between male animals of both treatment groups showed statistical significance in HR, RR, QRS, and QT intervals and P-wave duration. A similar comparison between female animals revealed statistical significance in HR and QRS intervals only.
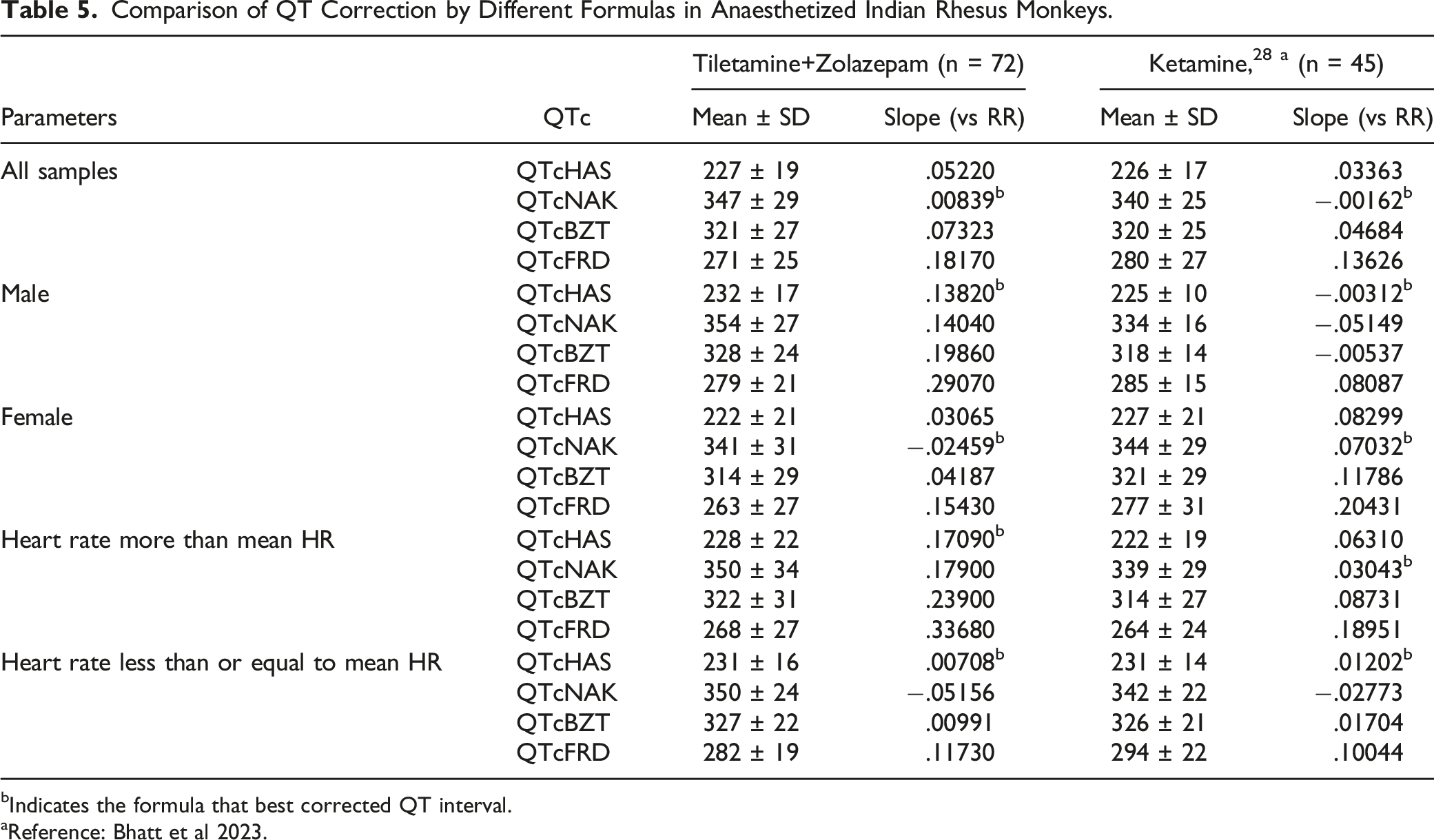
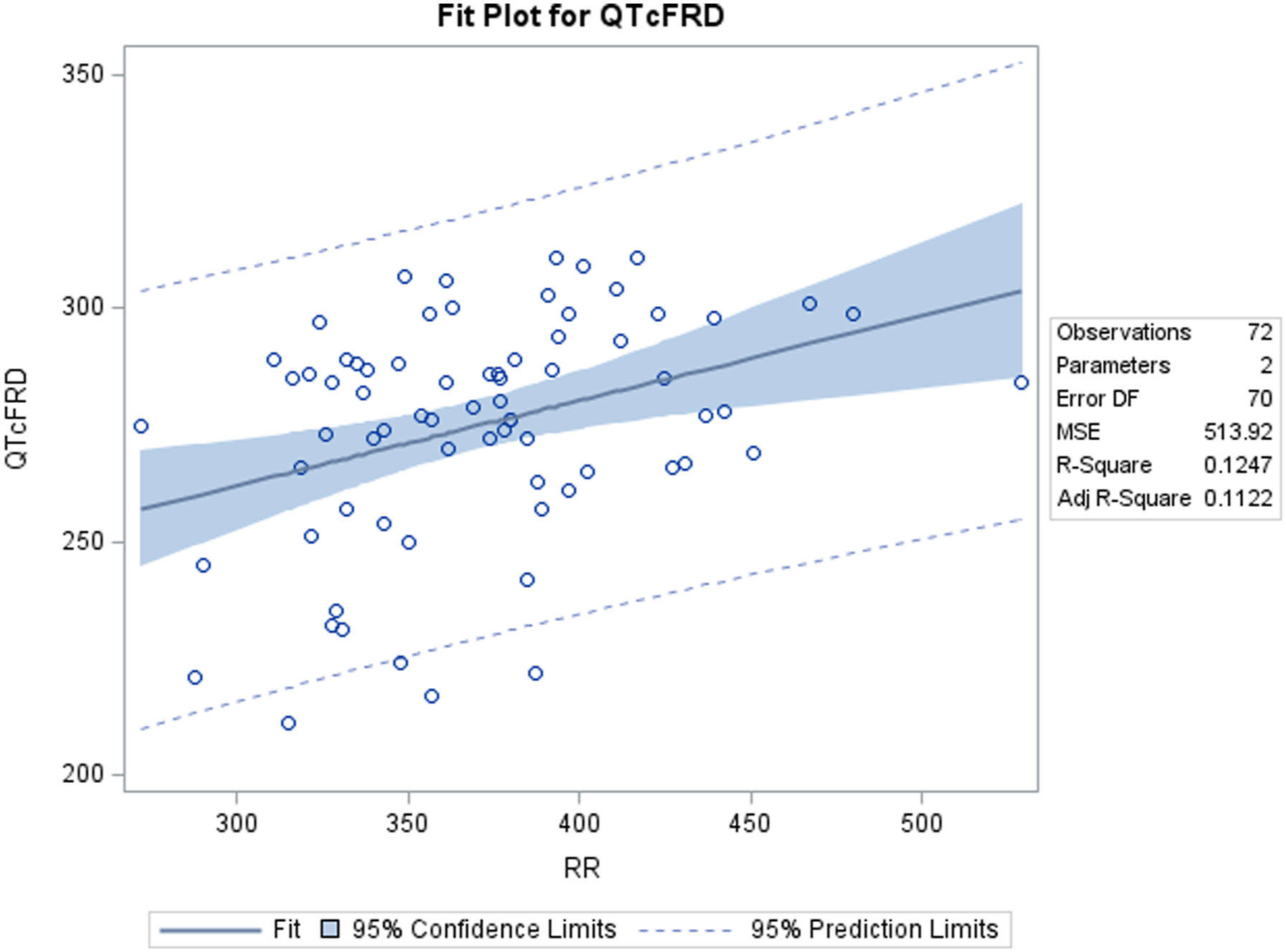
Linear regression analysis was performed on QT values (corrected and uncorrected) vs RR interval values obtained from TZ-anaesthetized monkeys. Regression line slopes and r-values were obtained for the numerical variables of QT/QTc vs RR. In linear regression analysis of two variables, the regression line slope and r-value indicate the strength and extent of correlation between the two variables. The value of ‘r’ is always between −1 (negative) and +1 (positive), where +1 indicates a perfect positive correlation and −1 indicates a perfect negative correlation. Values closest to zero indicate the least amount of correlation between the two tested variables.
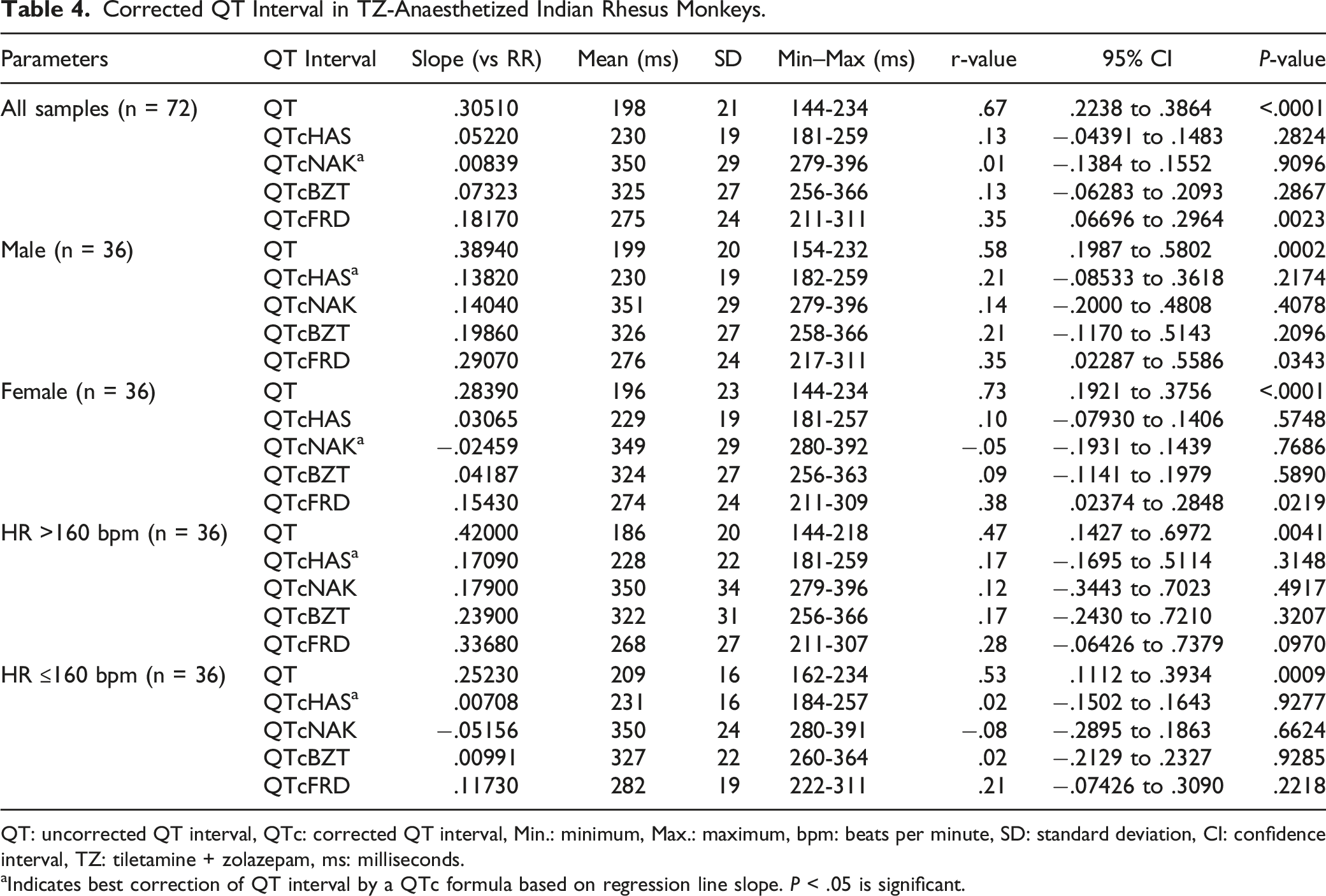
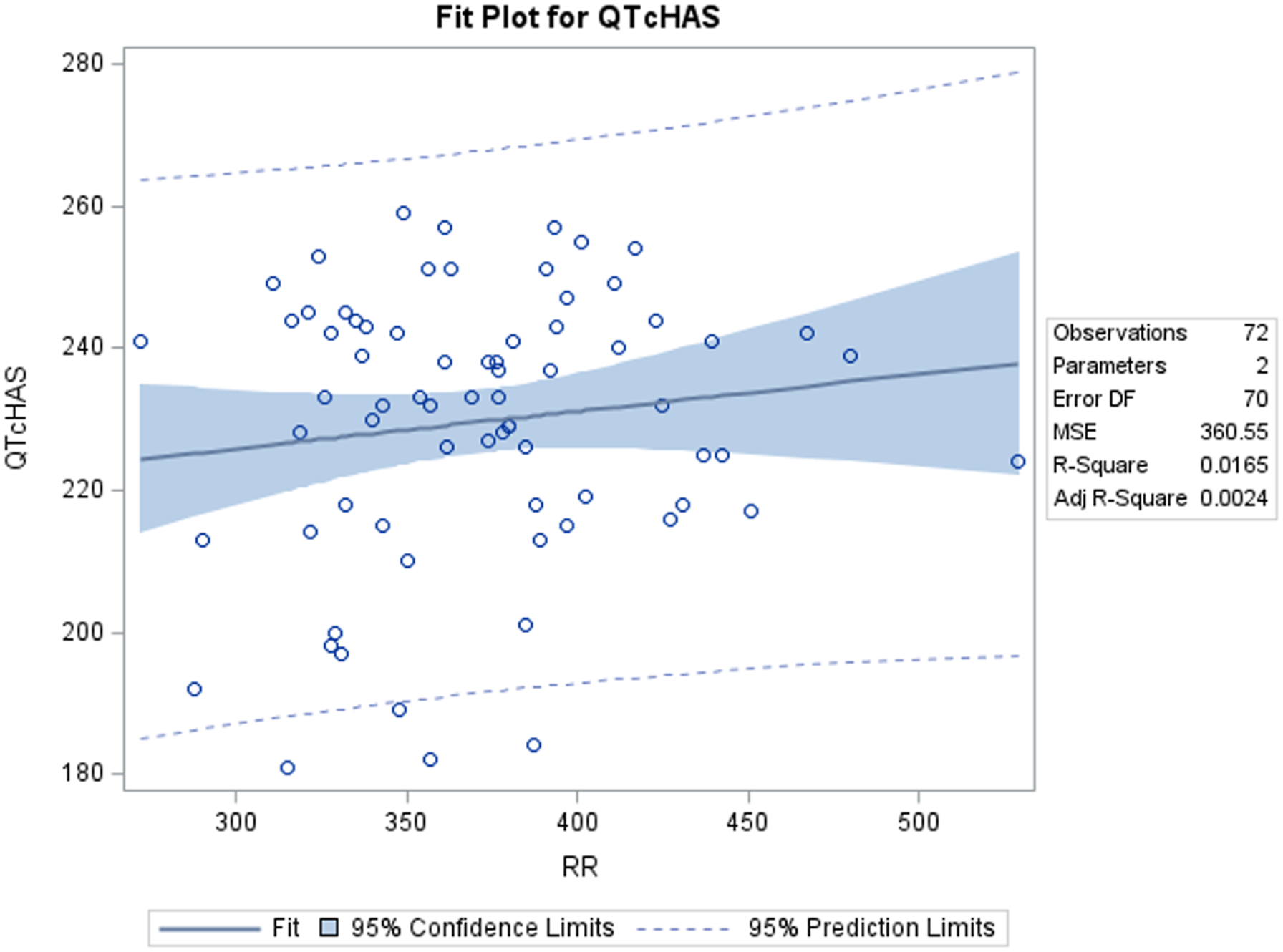
A positive correlation was observed between uncorrected QT and RR values. Out of the four QTc formulas tested, QTcNAK provided the best possible correction of the QT interval for change in the RR interval. The regression line slope obtained from QTcNAK values was closest to zero (.00839), followed by QTcHAS, QTcBZT, and QTcFRD (Figure 1). The incongruence between QTcNAK and RR intervals was further supported by the r-value (.01) and P-value (.9096), indicating both the lack of correlation and statistical non-significance between the two parameters. QTcNAK was the better-correcting formula for female animals, while QTcHAS was found to better correct QT interval in males (Table 4, Figure 2, Figure 3). QTcHAS better corrected QT in groups of animals with heart rates lower or equal to the mean heart rate and in those with heart rates higher than the mean HR (160 bpm) (Table 4, Figures 4, Figure 5). This trend of QT correction observed in TZ-anaesthetized monkeys was similar to the data observed in our previous study on KTM-anaesthetized Indian rhesus monkeys (Table 5).
28
Linear regression plots of uncorrected QT interval and corrected QT interval (QTc) vs RR interval. Corrected QT Interval in TZ-Anaesthetized Indian Rhesus Monkeys. QT: uncorrected QT interval, QTc: corrected QT interval, Min.: minimum, Max.: maximum, bpm: beats per minute, SD: standard deviation, CI: confidence interval, TZ: tiletamine + zolazepam, ms: milliseconds. aIndicates best correction of QT interval by a QTc formula based on regression line slope. P < .05 is significant. QTcHAS vs RR plot for male rhesus monkeys. QTcNAK vs RR plot for female rhesus monkeys. QTcHAS vs RR plot for rhesus monkeys with HR more than 160 bpm. QTcHAS vs RR plot for rhesus monkeys with HR less than or equal to 160 bpm. Comparison of QT Correction by Different Formulas in Anaesthetized Indian Rhesus Monkeys. bIndicates the formula that best corrected QT interval. aReference: Bhatt et al 2023.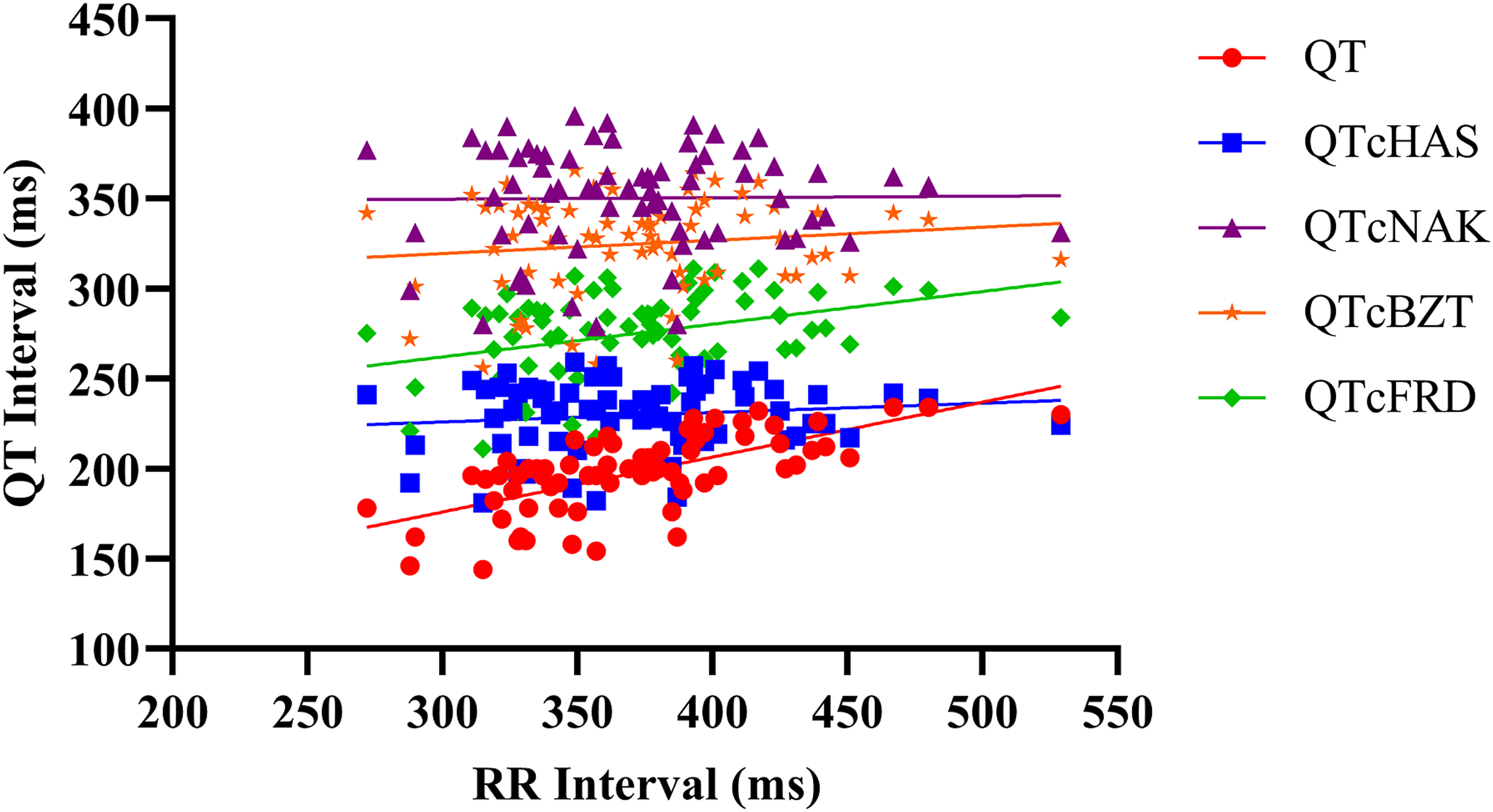
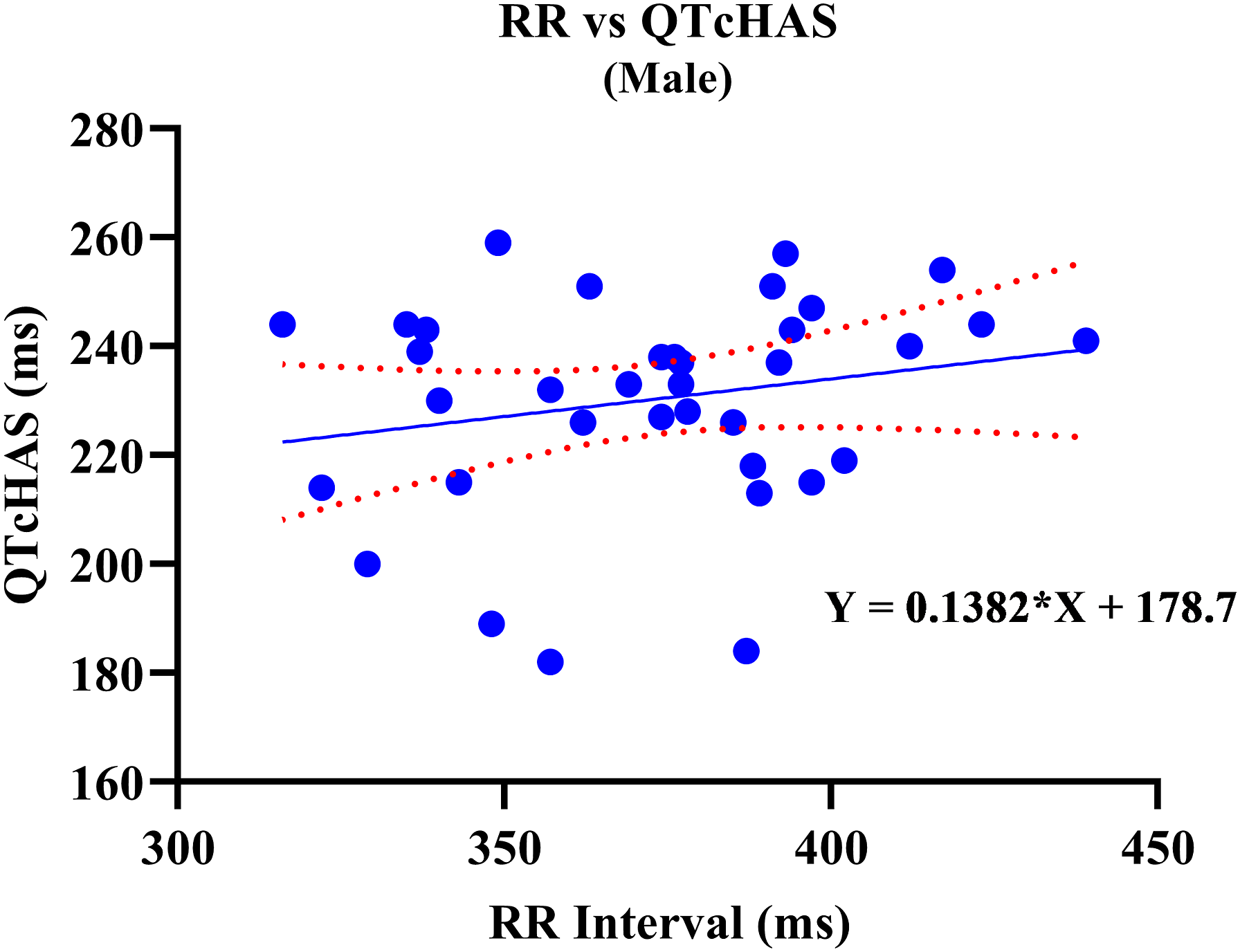
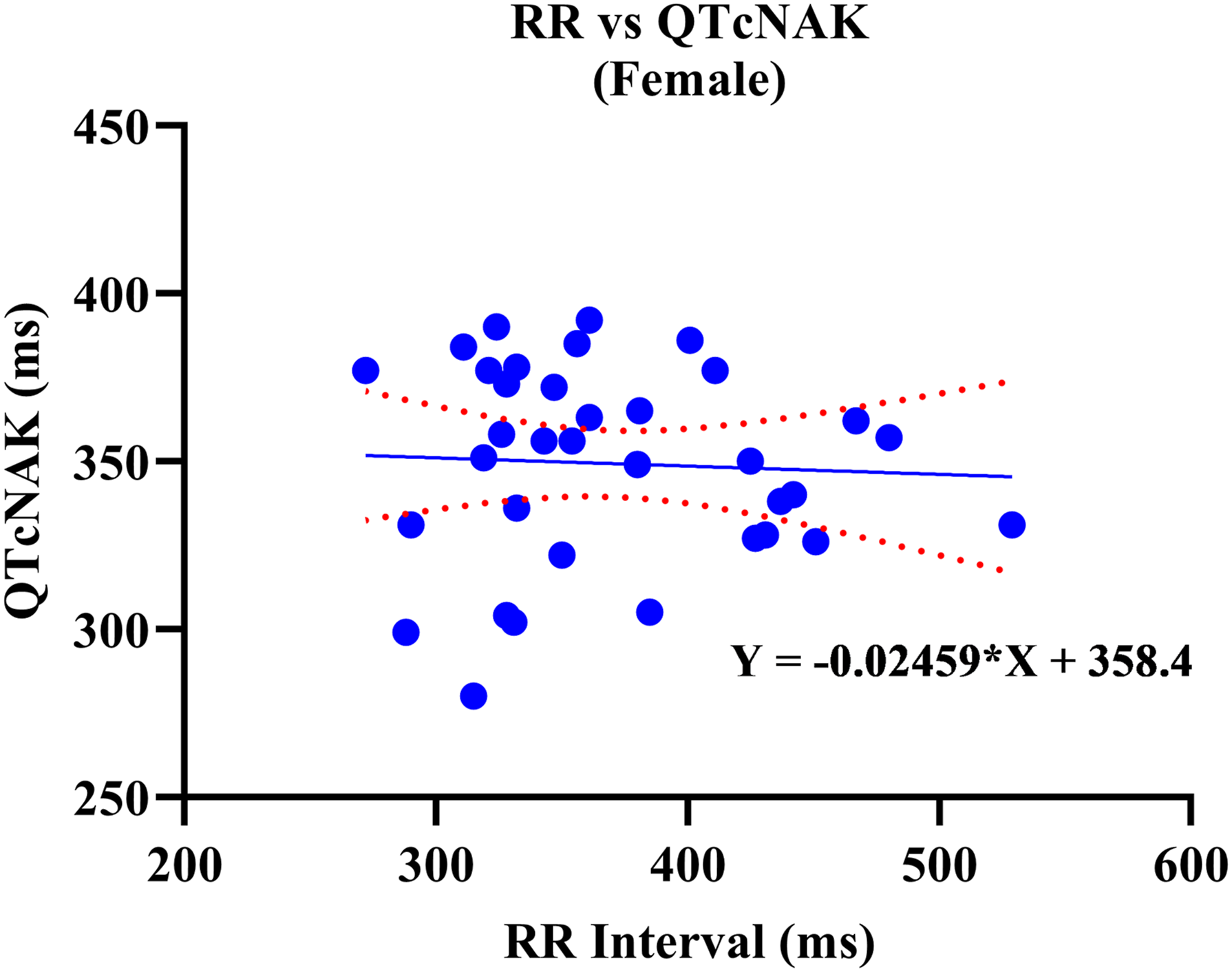
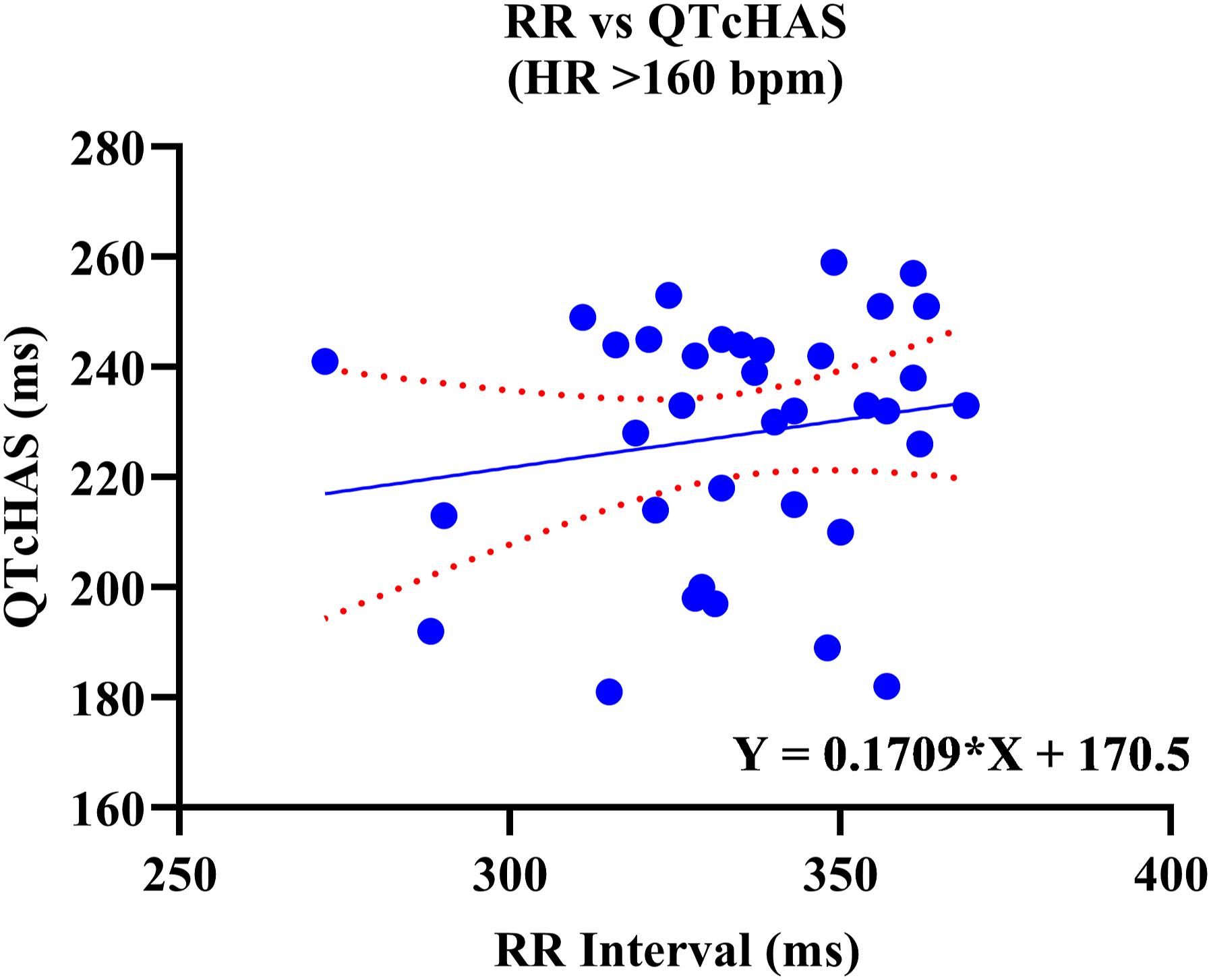
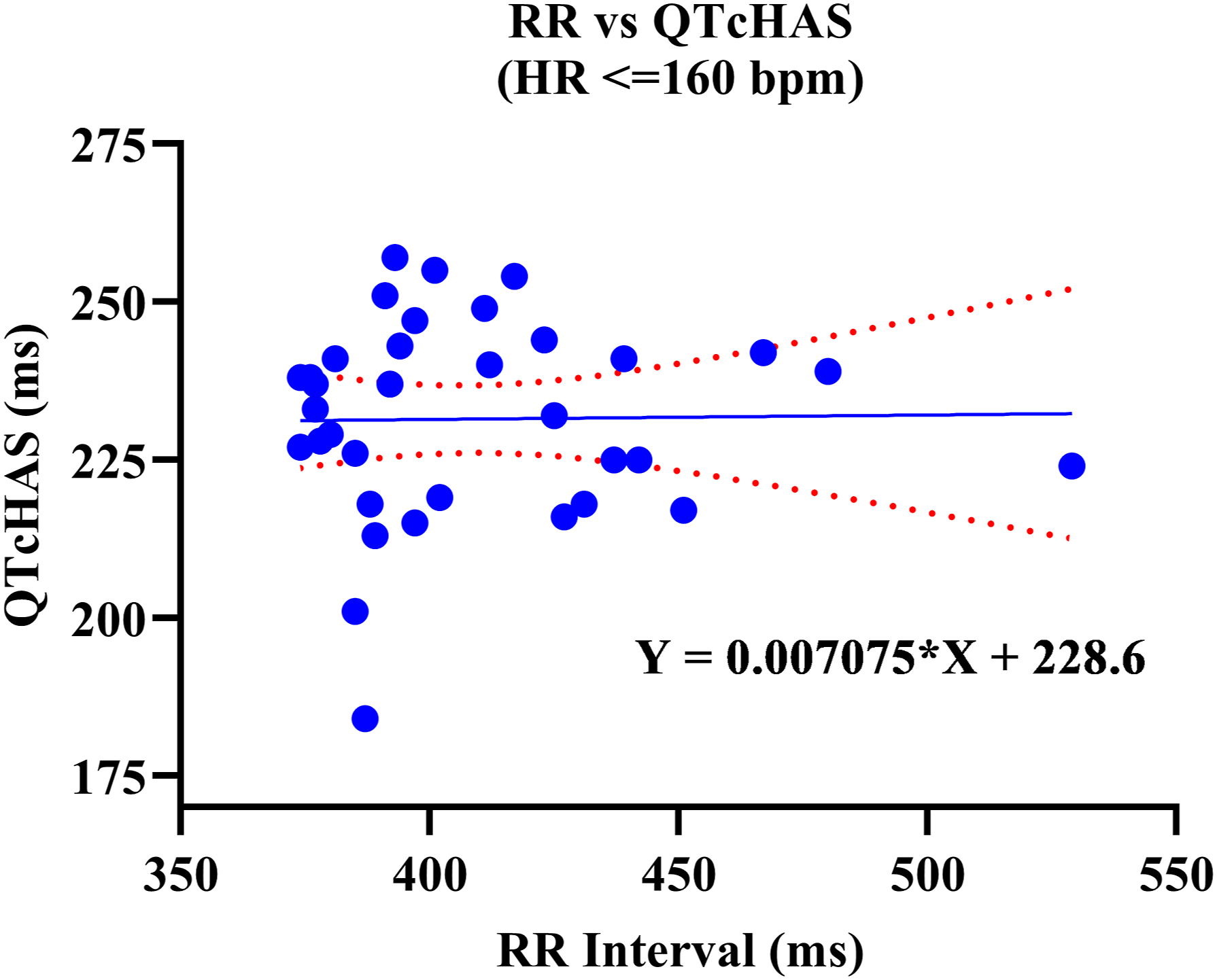
Sex-based significance was not observed in any of the QTc formulas tested in this study in TZ-anaesthetized monkeys.
Discussion
Electrocardiographic examination in NHPs is a critical endpoint of various nonclinical safety evaluation studies.6,12,39,40 Although KTM and TZ are commonly used in toxicology studies for restraint and immobilization of rhesus monkeys, reference data on the effects of these anesthetic agents on electrocardiographic parameters are inadequately available. Through this study, we have presented detailed electrocardiographic data from a large number of rhesus monkeys with specified anesthetic regimens.
Electrocardiographic Parameters
Electrocardiography records the electrical activity of the heart. ECG is considered to be a primary non-invasive tool in the diagnosis of cardiac diseases. Understanding and interpreting different electrocardiographic parameters is crucial in identifying abnormal ECG patterns, ultimately assisting in accurate disease diagnosis.
The mean heart rate in KTM-anesthetized animals in the present analysis was found to be 140 ± 33 bpm, while that for TZ-anesthetized animals was 164 ± 20 bpm. Mean HR values of KTM- and TZ-anesthetized groups were found to be significant upon comparison, wherein the mean heart rate of the KTM group was significantly lower than the HR of the TZ group of animals. Previously reported studies in Macaca mulatta sedated by KTM have reported mean HR values of 134 ± 15 bpm, while the mean HR from KTM-sedated Macaca fusucata 26 was 158 ± 20 and KTM-sedated Macaca fascicularis 41 were 168 ± 22 bpm. It has been observed that anesthetized animals, regardless of the choice of anesthetics, have displayed higher mean HR values. Data from a group of 25 conscious, telemetered rhesus monkeys were found to have a mean HR of 110 ± 12 bpm. In this evaluation, parameters such as RR interval, QRS interval, QT interval, and P-wave duration were found to be statistically significant among KTM- and TZ-anesthetized groups of animals. These parameters were also found to have values ranging nearly similar to the previously published standard reference range in unanesthetized rhesus monkeys. 6
The primary objective of anesthesia is to achieve a consistent heart rate and to reduce artefacts and noise arising from anxiety and body movement. It has been reported that KTM does not majorly affect ECG parameters except HR.26,41,42 In the present study, neither KTM nor TZ treatment resulted in any major changes in common electrocardiographic parameters of rhesus monkeys other than HR.
QT Interval Correction and QT-Heart Rate Independence
The QT interval is inversely related to heart rate. Any change in the heart rate causes a similar opposite change in the normal QT interval, prompting investigators to correct the resultant QT interval for changes in HR. Such a method, termed QT correction, employs the use of various QT correction formulas. The corrected QT interval finds extensive application in pharmaceutical and clinical research.28,43-45 In the current study, we have examined the QT correction in TZ-anesthetized rhesus monkeys. In our previously conducted study on KTM-anesthetized rhesus monkeys, we have extensively evaluated these QTc formulas for their use in NHP toxicology studies. 28 Of the four QTc formulas used in the present study, QTcBZT and QTcFRD are the most clinically relevant. They are widely used across clinical and preclinical studies of cardiac safety assessment.28,44,45 QTcHAS and QTcNAK are the two primate-specific formulas. QTcHAS was designed from studies conducted on rhesus monkeys, while QTcNAK was developed through the data obtained from cynomolgus monkeys.35,36 These formulas were assessed for their ability to correct the QT interval for changes in RR interval based on regression line slopes, r-value, and P-value. The regression line slope indicates the strength of the correlation between two parameters and the r-value describes the extent of such a relationship. For both of these parameters, resultant values closest to zero indicate the least correlation between the two variables analyzed. The P-value indicates significance among analyzed parameters. For the present analysis, P-value <.05 indicated statistical significance.
In the present study, QTcNAK was found to better correct QT interval than other correction formulas. QTcNAK presented a regression line slope (.00839) and r-value (r = .01) closest to zero, indicating the least correlation between the QTc interval and the RR interval. QTcHAS and QTcBZT presented similar regression line slope (.05220, .07323) and r-values (r = .13, .13), indicating that QTc values obtained through both of these formulae were fairly independent of changes in RR interval (Table 4, Figure 1). This observation could be credited to the fact that both QTcNAK and QTcHAS have been derived from the QTcBZT equation (Table 2). QTcBZT employs a square root function (.5 as the exponent ‘n’) over the RR interval, and QTcNAK uses .576 as the exponent. While QTcHAS retains the square root function, the author has added species-specific reference RR-cycle length (500 ms) to the equation. QTcFRD failed to adequately correct the QT interval in this study. Findings from the present evaluation are in correlation to our previous study conducted on KTM-anesthetized rhesus monkeys where QTcNAK emerged to be the best-correcting formula when compared to QTcHAS, QTcBZT, and QTcFRD formulas.
The primary drawback associated with rate-specific QT correction formulas is overcorrection and undercorrection of the QT interval. 46 As the current data shows, uncorrected QT interval increases with an increase in RR interval, indicating a positive correlation and a positive regression line slope of QT vs RR (Figure 1). The goal of any QT correction formula is to produce corrected QT intervals that are not correlated to changes in RR interval and thus provides a zero-regression line slope. QTc formulas that generate a negative correlation and a negative regression line slope are said to overcorrect the QT interval, and those that generate a positive correlation and a positive regression line slope are said to undercorrect the QT interval. 47 Clinically, QTcBZT is known to overcorrect QT at higher HR and undercorrect QT at lower HR. Similarly, QTcFRD also overcorrects QT at higher HR in clinical conditions. 48 Data from the current study indicate that although QTcNAK provided the regression line slope closest to zero, the formula produced QTc values with a positive correlation with RR intervals and a positive regression line slope (.00839), indicating undercorrection of the QT interval. Other three formulas, QTcHAS, QTcBZT, and QTcFRD, also derived positive correlation and positive regression line slope values.
Interestingly, increased or decreased QTc values indicate over- and undercorrection. 46 QTcHAS displays the least amount of overcorrection when compared to the other three QTc formulas. The mean QTc values for these formulas are 230 ms (QTcHAS), 275 ms (QTcFRD), 325 ms (QTcFRD), and 350 ms (QTcNAK) (Table 4). Both QTcBZT and QTcFRD are human-derived QT formulas designed to normalize QT interval to a resting human HR of 60 bpm. Of the four tested formulas, only QTcHAS has been appropriately modified to accommodate NHP-specific reference RR cycle length (500 ms), thereby making it the only physiologically relevant formula, as evident from the mean QTc values. However, in terms of regression line slope and r-value, it is the second-best formula after QTcNAK and a better-correcting formula than QTcBZT and QTcFRD.
The “Best Practice Considerations for in vivo QT Studies” developed by ICH recommends the usage of individual rate-corrected QT formulas as best practice in situations where a sufficient number of QT-RR pairs and a broad range of RR values are available. Additionally, the “E14/S7B Questions and Answers: Clinical and Nonclinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential” document also recommends that conventional QT corrections methods such as formulas by Bazett, Fridericia, and Van de Water can be used but with validation. 49 Individual rate-corrections methods, such as by Miyazaki and Tagawa and Holzgrefe and others, rely on raw QT, raw RR, reference cycle RR and β (the slope of log QT-RR) in calculating QTc for individual animals. The value of β is derived from untreated or vehicle-treated animals, and the ECG data are procured over longer timespans such as 12–20 hours to cover different physiological HR. Repeated dose toxicology studies entail multiple endpoints such as physical examination, ophthalmic examination, and neurobehavioral examination that necessitate frequent handling and result in handling-induced stress in test animals. Snapshot ECGs are fast and cost-effective, can detect irregular HR and major arrhythmias, and can easily be accommodated along with other examinations. However, study designs in GLP-compliant repeat dose toxicology studies do not allow for obtaining large-scale background observations to calculate β-values that are essential in calculating QTc by individual rate-correction methods.28,50,51 Previously conducted studies have recommended usage of individual rate-correction methods in QTc derivation from rhesus monkeys.50–52 Nevertheless, for safety pharmacology studies, where study designs allow the collection of background data from untreated and/or vehicle-treated animals, the authors do recommend following the guidelines stated under the ICH S7B Q&A “Best Practice Considerations for in vivo Studies”.
An ideal QT correction formula must produce QTc values that are independent of RR interval or HR. The independence or absence of correlation in a linear relationship between two parameters can be analyzed by regression line slope and correlation coefficient (r). In the present study conditions, QTcNAK has produced QTc values that are fairly independent of changes in the RR interval, evident from an almost straight regression line parallel to the X axis with a slight negative correlation (Figure 6). Other formulas have produced varying degrees of RR dependence with a fairly positive degree of correlation (Figures 7–9). Fit plots of QTc formulas with 95% confidence limits and 95% prediction limits of QTcNAK. Fit plots of QTc formulas with 95% confidence limits and 95% prediction limits of QTcHAS. Fit plots of QTc formulas with 95% confidence limits and 95% prediction limits of QTcBZT. Fit plots of QTc formulas with 95% confidence limits and 95% prediction limits of QTcFRD.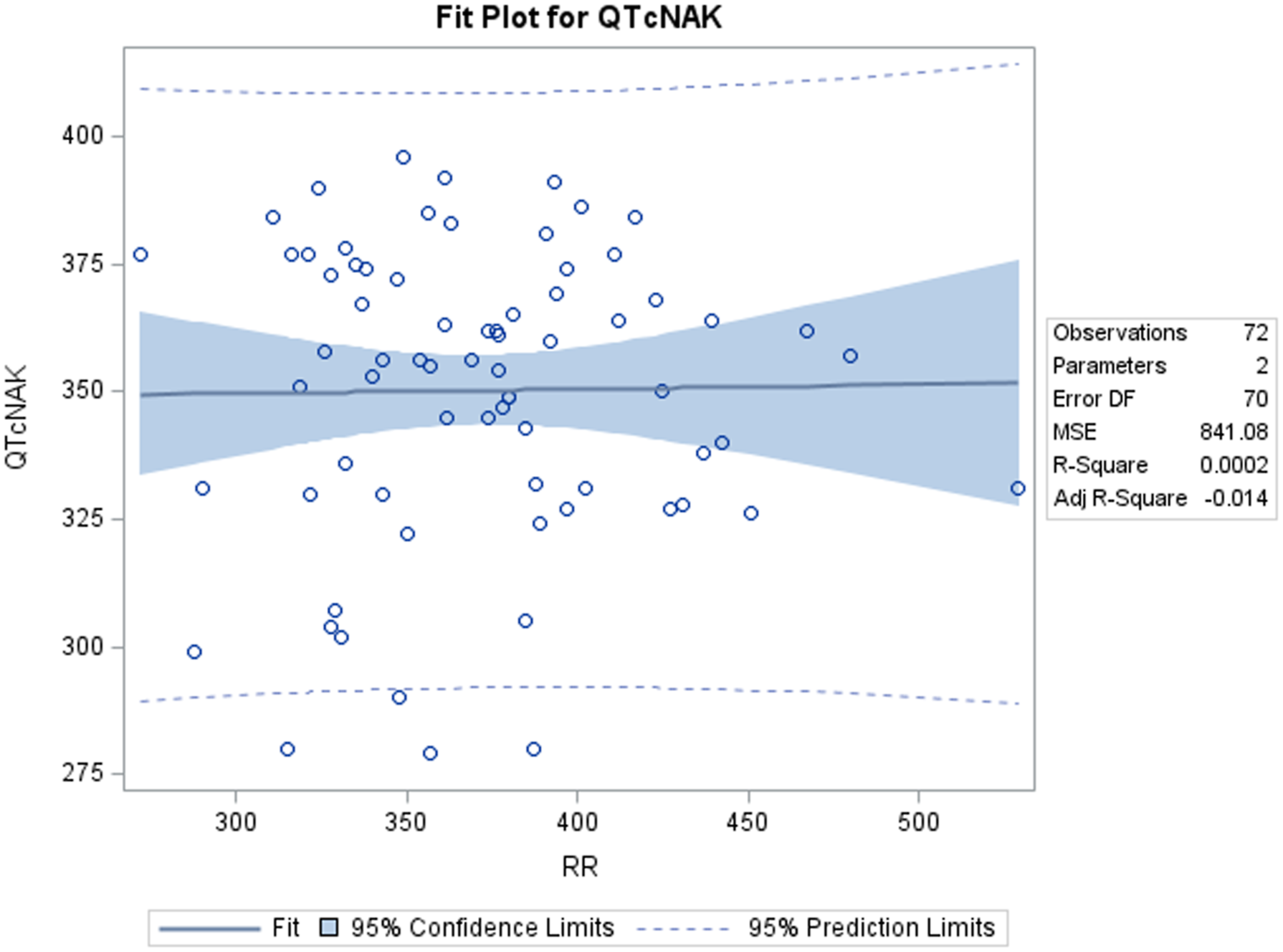
General anesthetics are known to affect thermoregulation.53,54 Hypothermia induced by general anesthetics is a combined result of depression of body temperature regulation centers and heat loss through respiration. 53 Changes in core body temperature (BT) has been associated with changes in the length of cardiac cycles, wherein QT interval prolongation is observed with a decrease in BT.55,56 It can be postulated that if QTc interval is used to assess drug-induced QT interval prolongation during hyper- or hypo-thermic situations, such as anesthesia, the sole QTc formula without appropriate correction for BT changes would be inadequate or even misleading. Both KTM and TZ are known to cause a decrease in core BT.53,54 A limitation of the present analysis is the absence of correlation of QT and QTc evaluation with core BT of rhesus macaques. Hence, further evaluations of QTc formulas for these anesthetics along with BT measurement are suggested.
Conclusion
We retrospectively evaluated the effect of ketamine and tiletamine-zolazepam anesthesia on various electrocardiographic data of Indian-origin rhesus monkeys. Our assessment of QT correction in tiletamine-zolazepam anesthetized animals revealed that the QTcNAK, an NHP-specific QT correction formula, best corrected the QT interval for changes in heart rate in the present study conditions. The authors suggest studies further examining the applicability of species-specific formulas in QT correction. Before this study, information available on reference electrocardiographic parameters of ketamine or tiletamine-zolazepam anesthetized rhesus monkeys was scarce. Through this study, we have made available data that will serve as a historical reference range for ketamine and tiletamine-zolazepam anesthetized rhesus monkeys for use by researchers worldwide.
Footnotes
Acknowledgments
The authors are grateful to the management and scientific team at Zydus Research Centre, Ahmedabad, for their assistance in preparing this manuscript.
Author Contributions
The submitting author affirms that all individuals listed as authors agree that they have met the criteria of authorship and agree to the conclusions of the study. In order to meet the requirements of authorship, each author must have contributed to at least one aspect of each of the four criteria, as listed below. Please note that for Criteria 1 and 2, authors only to meet one of the two items listed. These criteria are not to be used as a means to disqualify colleagues from authorship who otherwise meet authorship criteria by denying them the opportunity to meet criteria 2 or 3. Therefore, all individuals who meet the first criterion should have the opportunity to participate in the drafting, review, and final approval of the manuscript. Any individuals not meeting the criteria may be mentioned in the Acknowledgements section of the manuscript. Per the criteria defined by the International Committee for Medical Journal Editors (ICJME), please note the contribution made by each author listed in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Animal Welfare and Ethics Statement
All experimental procedures involving rhesus monkeys were conducted according to the guidelines provided by the Committee for Control and Supervision of Experiments on Animals, Government of India, and approved by the Institutional Animal Ethics Committee (Facility Registration Number: 77/PO/RcBi/SL/99/CPCSEA).
Contributorship
Bhatt, L.K. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript and critically revised manuscript; Shah, C.R. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Patel, S.D. contributed to design, contributed to acquisition and interpretation, and critically revised manuscript; Patel, S.R. contributed to analysis and critically revised manuscript; Patel, V.A. contributed to interpretation and critically revised manuscript; Patel, R.J. contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Joshi, N.M. contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Shah, N.A. contributed to design, contributed to acquisition, drafted manuscript, and critically revised manuscript. Patel, J.H. contributed to conception and critically revised manuscript; Dwivedi, P. contributed to conception and design, contributed to acquisition, and critically revised manuscript; Sundar, R. contributed to conception and design and critically revised manuscript; Jain, M.R. contributed to conception and design and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
