Abstract
Cynomolgus macaques, the most commonly utilized nonhuman primate in nonclinical toxicology studies, are acquired from purpose-bred colonies across various geographic locations, including China, Cambodia, and Vietnam. Importation challenges and limited availability have restricted animals suitable for inclusion in nonclinical studies. The coronavirus disease 2019 (COVID-19) outbreak further stressed supply chains, reducing the ability to source animals from a singular location to complete a drug development program. These challenges raised concerns of increased variability in study endpoints due to heterogeneity of animals and that this could subsequently impact historical control data and toxicology study interpretation. To investigate the impact of Chinese, Vietnamese, or Cambodian geographic origin on standard nonclinical toxicology study endpoints, historical control data from studies conducted at a single facility from 2005 to 2020 were compiled and evaluated for the following: clinical observations, body weight, ophthalmoscopic examinations, and clinical and anatomic pathology data. Study populations consisted of 2- to 5-year-old cynomolgus macaques sourced from China (n = 750 males/741 females), Cambodia (n = 282 males/271 females), and Vietnam (n = 122 males/120 females). Interpretation of the various data demonstrated no notable differences in standard toxicology study endpoints or background findings among cynomolgus macaques originating from China, Cambodia, or Vietnam.
Keywords
Introduction
Cynomolgus macaques, the most common nonhuman primate (NHP) species selected for use in nonclinical toxicology studies, are generally derived from purpose-bred sources in a variety of geographic locations such as China, Cambodia, Vietnam, Mauritius, and Indonesia; however, their importation remains a significant challenge due to a shortage in availability. This shortage has been exacerbated recently by restrictions put in place in response to the coronavirus disease 2019 (COVID-19) outbreak, including the cessation/reduction of NHP exports from mainland China in 2020. To meet increased research demands, importers of NHPs have expanded and diversified the supply chain. At American Charles River Laboratories sites, this resulted in decreased importation from China, which has traditionally accounted for the greatest proportion of NHP importation, with increased utilization of cynomolgus macaques from other locations including Cambodia, Mauritius, and others (Figure 1).
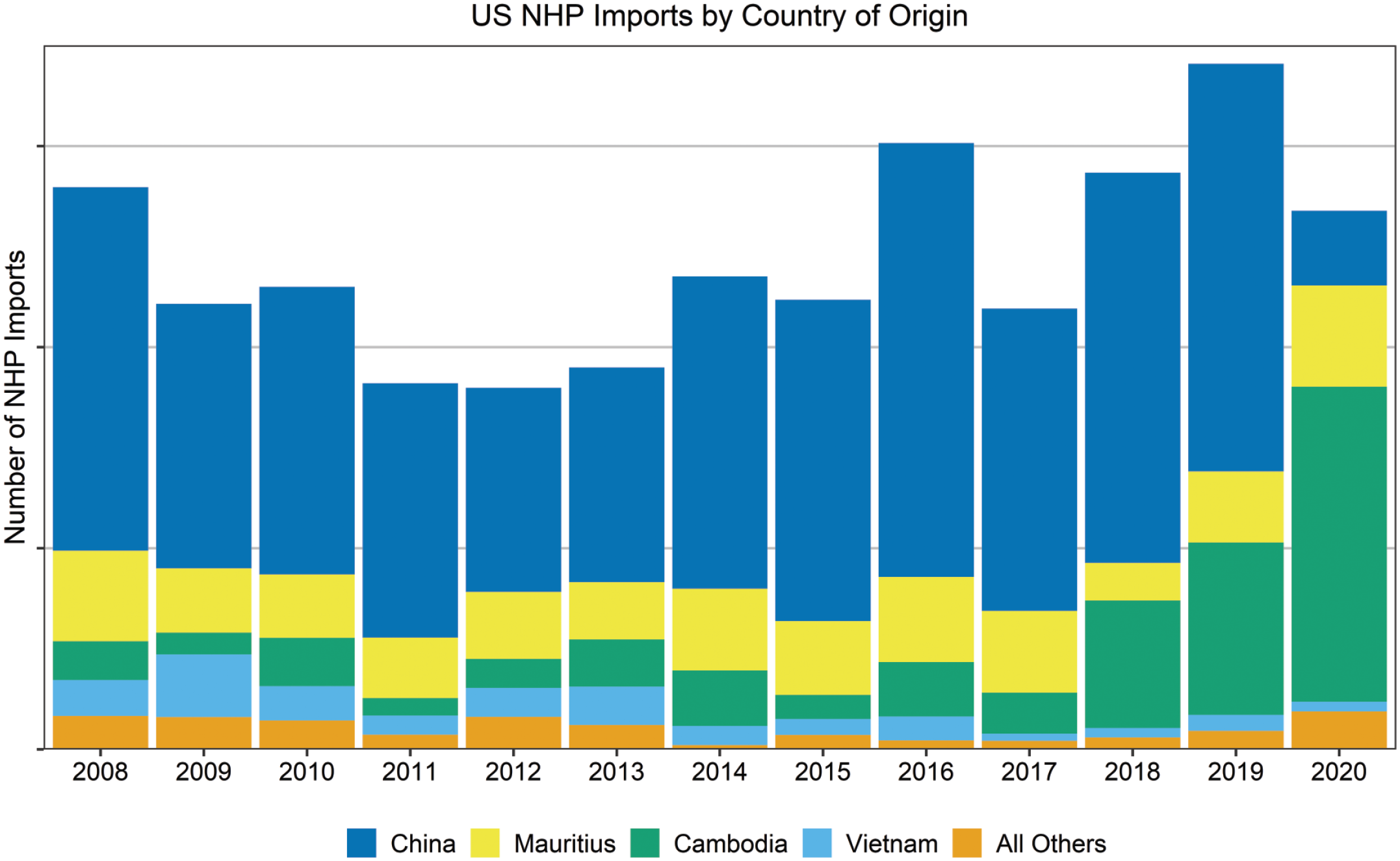
Importation of nonhuman primates by geographic origin for American sites of Charles River Laboratories. NHP indicates nonhuman primate.
Importantly, considerable genetic and environmental variability associated with an animal’s geographic origin has the potential to affect the conduct of nonclinical toxicology studies and the interpretation of study results. Previous studies have examined the genetic differences among subpopulations by analysis of mitochondrial DNA, Y chromosome DNA, and single-nucleotide polymorphisms, generally subdividing Macaca fasicularis into 2 groups. The “continental” group contains the Indochinese and a subset of Indonesian haplotypes while the “insular” group encompasses the subpopulations of the Philippines, Mauritius, and the majority of the Indonesian haplotypes.1,2 Despite increased genetic diversity among continental or Indochinese populations relative to the insular or Indonesian and Filipino populations, animals from these locations have a shared genetic background supporting their interchangeable use in biomedical research. 2 Other factors may also contribute to variability based on geographic origin, such as environmental factors, husbandry considerations, and potential health impacts/hazards that may vary by location.
The current study’s objective was to compile historical control data from healthy cynomolgus macaques originally sourced from China, Cambodia, and Vietnam utilized in good laboratory practice (GLP)-compliant nonclinical toxicology studies at Charles River Laboratories in Mattawan, Michigan, USA (known as MPI Research prior to April 2018) in order to assess the impact of these evaluated geographic origins on standard nonclinical toxicology study endpoints including clinical observations, body weight, ophthalmoscopic examinations, and clinical pathology and anatomic pathology parameters. Cynomolgus macaques from other geographic locations (Mauritius, Philippines, Indonesia, etc.) were necessarily excluded from evaluation as sufficient data were not available at Charles River Laboratories, Mattawan for robust statistical evaluation.
Materials and Methods
Animals
Historical control data were compiled from GLP-compliant nonclinical toxicology studies (n = 374) conducted at Charles River Laboratories, Mattawan (formerly MPI Research). These studies ranged in duration from 1 to 52 weeks, were conducted between November 2004 and March 2020 and represented various routes of administration (Tables 1 through 3) of assorted control articles used in control groups (eg, vehicle controls, saline, etc.). Data were collected prior to the dosing phase or “pretreatment” and at the end of each study or “final” from control group animals (eg, from vehicle, placebo, or sham dosing groups). The animals were purpose-bred for laboratory use and sourced from accredited suppliers from China (n = 1491), Cambodia (n = 553), or Vietnam (n = 242). Each study utilized animals from a single geographic origin.
Number of studies included in data by origin and route of administration of various materials used in control groups.
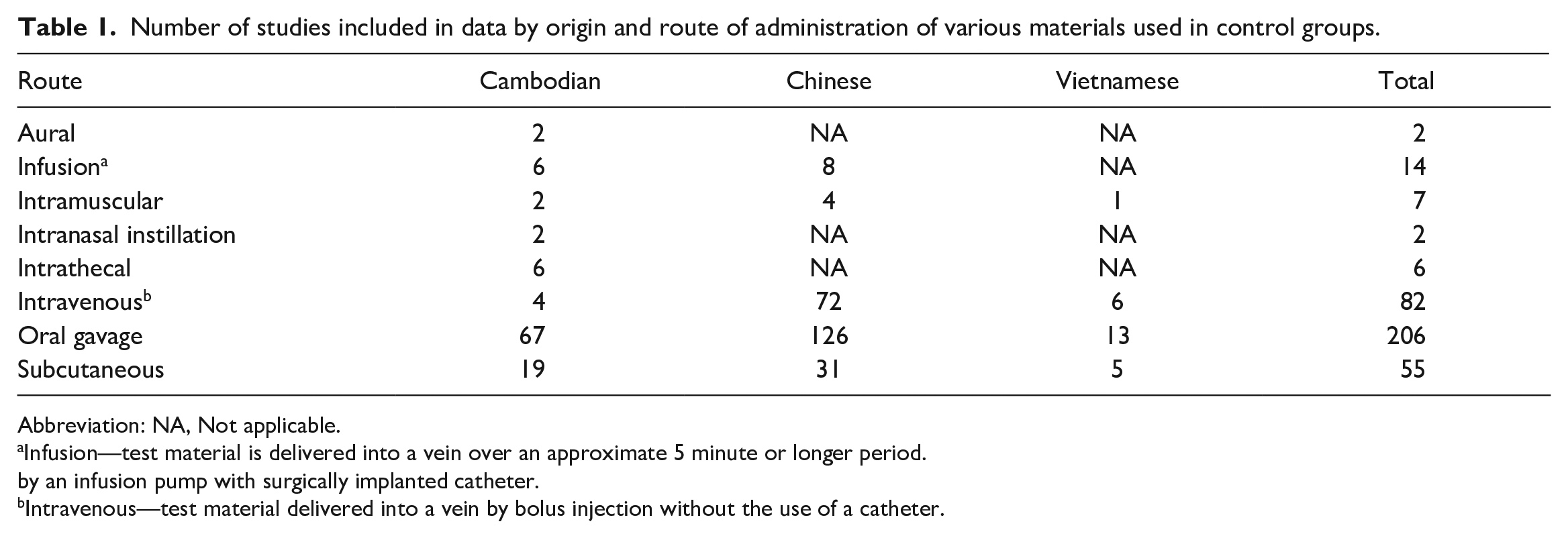
Abbreviation: NA, Not applicable.
Infusion—test material is delivered into a vein over an approximate 5 minute or longer period.
by an infusion pump with surgically implanted catheter.
Intravenous—test material delivered into a vein by bolus injection without the use of a catheter.
Number of cynomolgus macaques included in data by origin and route of administration of various materials used in control groups.
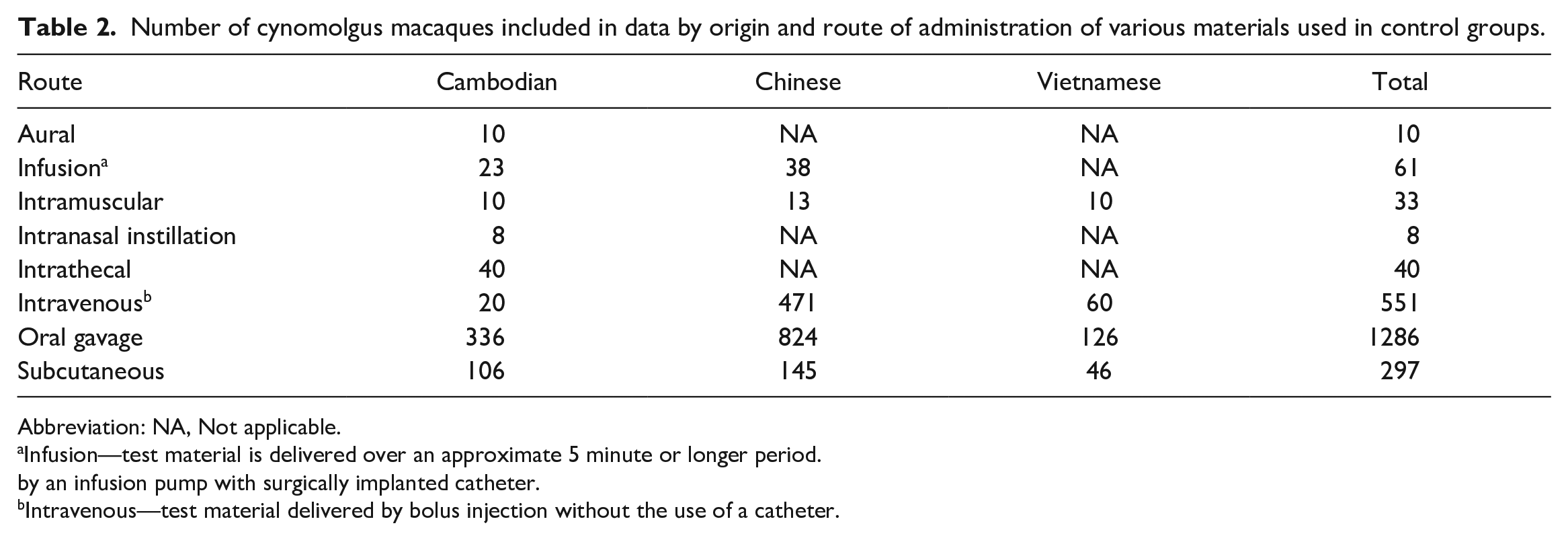
Abbreviation: NA, Not applicable.
Infusion—test material is delivered over an approximate 5 minute or longer period.
by an infusion pump with surgically implanted catheter.
Intravenous—test material delivered by bolus injection without the use of a catheter.
Range of dates of study conduct by origin.

The animals were between 24 and 60 months of age at the initiation of dosing on study. Upon receipt at the Testing Facility, the animals were quarantined (at least 10 days) from the other facility animals by group housing (2-4 per cage; single sex) in appropriately sized caging as specified in the USDA Animal Welfare Act regulations (Code of Federal Regulations, Title 9) 3 and as described in the Guide for the Care and Use of Laboratory Animals. 4 Environmental conditions were controlled throughout the facility housing NHPs to maintain temperature between 17°C and 29°C, humidity between 30% and 70%, and fluorescent lighting provided via an automatic timer for approximately 12 hours per day. Tap water was supplied ad libitum to all animals via an automatic water system. Commercial primate diet (PMI Nutrition International, Inc., Richmond, Indiana) was provided to all animals twice daily except during fasting periods.
During the quarantine period, animals were examined by veterinary staff, weighed weekly, and tested multiple times for tuberculosis. Following the quarantine period, healthy animals were transferred to the pretreatment phase of study where study-specific screening evaluations were performed (veterinary physical examination, clinical pathology, electrocardiography, ophthalmology), and quarantined in separate room until transferred to study.
In-Life Assessments
Detailed clinical observations and body weight data were collected following Test Facility standard operating procedures (SOPs). Ophthalmoscopic examinations were performed via indirect ophthalmoscopy by a Diplomate of the American College of Veterinary Ophthalmology. Observations that were known to have been recorded inconsistently due to change in facility SOPs over the timeframe of this study were excluded from analysis.
Clinical observation data from healthy control cynomolgus macaques recorded by technical staff, veterinarians, and veterinary ophthalmologists during both regularly scheduled and unscheduled observations were evaluated. Detailed ophthalmology, clinical observation, and body weight data are provided in Supplemental Tables 1 through 7.
Clinical Pathology
Historical data from control monkeys for hematology, coagulation, and clinical chemistry were evaluated from pretreatment and final collections. Hematology parameters were evaluated in EDTA-anticoagulated whole blood using Siemens ADVIA® 120 or 2120 instruments and Siemens reagents and included: total white blood cell count or total white blood cells (WBC), absolute neutrophil, lymphocyte, monocyte, eosinophil, basophil, and large unstained cell (LUC) counts, red blood cell (RBC) count, hemoglobin concentration, hematocrit, absolute reticulocyte count, mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), red blood cell distribution width (RDW), and platelet count. Coagulation parameters evaluated in citrated plasma using Stago STA Compact®, STA Compact Max®, or STA-R Evolution® instruments included activated partial thromboplastin time (APTT) using the Siemens Dade APTT reagent, and prothrombin time (PT) and fibrinogen concentration using Stago reagents. Clinical chemistry parameters evaluated in serum using Olympus AU640, Olympus AU2700, or Beckman Coulter AU5800 instruments and Olympus or Beckman Coulter reagents included aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), creatine kinase (CK) activities, total bilirubin, urea nitrogen, creatinine, glucose, total cholesterol, triglycerides, total protein, albumin, globulin, total calcium, phosphorus, sodium, potassium, and chloride concentrations and albumin/globulin ratio. Hematology, coagulation, and clinical chemistry results were recorded in a computerized database (Provantis 9, Instem, Philadelphia, PA, USA). Urinalysis data were not compared across geographic origin due to the low sample size for urinalysis results from monkeys of Vietnamese origin, a change in standard duration of urine collection during the timeframe of the study (eg, 12-16 hour overnight pan collections versus same day [morning] collections), and the potential for contaminant interference on urine reagent test strip analysis.5,6 Hematology, coagulation, and clinical chemistry data and results of statistical analysis are provided in Supplemental Tables 8 through 16.
Anatomic Pathology
Animals were humanely euthanized and necropsies were performed in accordance with approved SOPs and protocols. Necropsies were performed under the supervision of a veterinary pathologist, organs were weighed, and macroscopic observations were recorded. Tissues were collected into 10% neutral buffered formalin and sections were embedded in paraffin wax prior to sectioning to a 4 to 5 μm thickness and stained with hematoxylin and eosin. Organ weights, macroscopic observations, and histopathologic findings were entered into a computerized database (Provantis 9, Instem, Philadelphia, PA, USA). Anatomic pathology data included organ weights, gross lesions, and microscopic lesions. Microscopic observations were generated by Diplomates of the American College of Veterinary Pathologists (DACVP) and many studies included pathology peer review. 7 Histopathologic findings, macroscopic observations, and organ weight data and results of statistical analysis are provided in Supplemental Tables 17 through 29.
Clinical and Anatomic Pathology Assessments
The clinical pathology and anatomic pathology data were reviewed by Diplomates of the American College of Veterinary Pathologists (authors C.C., M.L., and D.P.), and the results depicted herein are limited to representative parameters. Microscopic diagnoses were reviewed, and similar/synonymous diagnoses from the historic control database were combined using current International Harmonization of Nomenclature and Diagnostic Criteria (INHAND). 8 For example, diagnoses of “infiltrate, lymphoid” and “infiltrate, lymphoplasmacytic were listed as “infiltrate, mononuclear””; “degeneration” was listed as “degeneration/necrosis”; “necrosis,” “necrosis (focal),” and “necrosis (hepatocytes)” were listed as “necrosis,” and so on.
Data Analyses
Statistical analysis was performed using R v4.1.0 (R Core Team, 2019, Vienna, Austria) to evaluate the potential impact of geographic origin and study phase on results. All relevant result records were obtained through database queries incorporating the previously mentioned inclusion criteria of a single electronic data capture system in use from November 2004 to March 2020 (Provantis 9, Instem, Philadelphia, PA, USA). To minimize potential impact of individual variation on wholistic interpretations, data were further filtered to include only those individuals with extant results for all result parameters evaluated within this study. Data for each sex and the 2 distinct time intervals (pretreatment and final, where applicable) were subsequently analyzed separately. Due to potential confounds in individual study conduct, available data were inadequate to empirically demonstrate the validity of individual study’s use as a factor variable. 9 As such, study-level impacts could not be determined rigorously enough to serve as an independent variable in further analyses. However, as all studies, regardless of geographic origin were all conducted at a single location, using GLP requirements, with all data being collected by staff trained to facility SOPs, under standardized environmental controls, individual study effects were unlikely to be a significant contributor to variability.
Body weight, organ weight, and all clinical pathology endpoints were evaluated using a semiparametric method to investigate the effect of geographic origin. Data were grouped into the three respective origin classes, and the corresponding F statistic was calculated. 10 Comparisons of intra-class variances to pooled variances and Levene’s test were used to assess homogeneity for each endpoint. 11 This procedure was repeated on simulated datasets generated via pooled non-parametric bootstrap resampling with replacement (1000 iterations) and the resultant F statistic values used to generate an empirical cumulative distribution function (CDF). 12 Rejection of the null hypothesis occurred when the observed data’s F statistic fell outside the central 95% of the generated CDF. In the presence of significant main effects, post hoc pairwise comparisons of the differences in group mean values were performed via pooled non-parametric bootstrap resampling with replacement (1000 iterations) employing a false discovery rate multiplicity adjustment.13,14 To maintain equal probability of a sampling with respect to origin given varying sample sizes, resampling populations were established by replicating the observed data of each origin until reaching the least common multiple in length, pooling the replicated datasets across origins, and assigning individuals values at random from this cumulative pool. Such resampling techniques make no assumptions regarding distribution of data. Consequently, outlier and normality tests were not performed.
Incidence counts for clinical and ophthalmoscopic observations were expressed as a proportion relative to each animal and analyzed for an effect of geographic origin using a generalized linear model fit to a beta-binomial distribution. 15 This method was deemed most appropriate to address clinical observation result events lacking independence across replication (eg, outstanding health effects cumulatively increase probability of the same result being recorded at future intervals). Subsequently, no collective post hoc pairwise comparisons were performed, as such methodology would not properly account for said temporal variation in probabilities. Effect of geographic origin on gross and microscopic lesions was examined through logistic regression of the observed frequency of pathological findings. In the presence of significant main effects, post hoc pairwise comparisons between origin were performed as Wald chi-squared tests with false discovery rate multiplicity adjustment. 16 The results of all statistical analyses are presented alongside the summarized data in the Supplemental Table. As the intended purpose of this paper was to compare across geographic origin with separation of pretreatment and final intervals when both available, no analyses were performed to generate reference intervals for clinical pathology data as this was considered beyond the scope of this investigation.
Historical control data and statistical analyses were reviewed to determine whether variation between geographic subpopulations present represented toxicologically meaningful differences. For the purposes of this evaluation, toxicologically meaningful differences were defined as differences between geographic origin that might confound (due to magnitude, character, incidence, etc.) the interpretation of nonclinical toxicity study data or result in erroneous attribution of inherent or biologic variability to administration of a known chemical entity (ie, test article-related effects). Although statistically significant differences were considered, the determination of the presence or absence of a meaningful difference also took into account the potential study impact, biologic relevance or anticipated physiologic significance, preanalytical and analytical variability, incidence, and magnitude of variation.
All study procedures were conducted in accordance with protocols that were in compliance with applicable animal welfare regulations and were approved by the Institutional Animal Care and Use Committee (IACUC). Charles River Laboratories, Mattawan, was accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC).
Results
In-Life
Clinical and ophthalmoscopic observations
There were no toxicologically meaningful geographic origin-related differences in ophthalmology or clinical observations among historical control cynomolgus macaques. Males and females from Vietnam had an increased frequency of excretion abnormalities (specifically feces soft/watery) when compared to male and female NHPs from Cambodia and China (Figure 2). However, this difference was only present in studies conducted in 2015 to 2017 and the frequency of this observation was comparable between origins, if not lower, in Vietnamese origin monkeys in all other years evaluated (2004-2014 and 2018-2020).
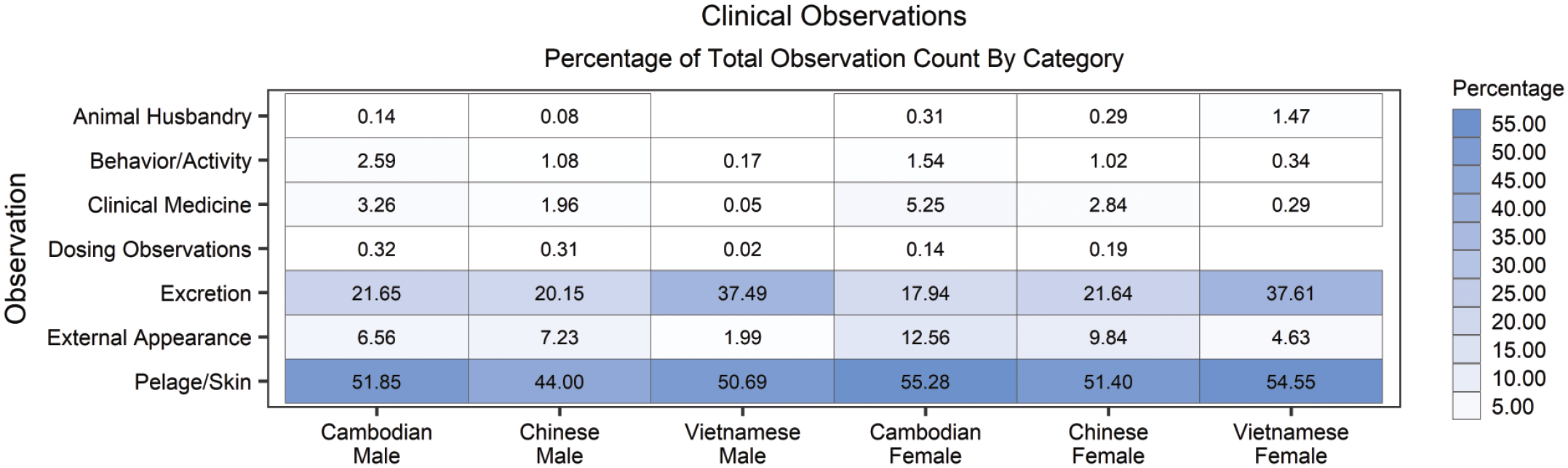
Percentage of observation occurrence by observational category, sex and origin. There were no toxicologically meaningful geographic origin-related differences in clinical observations among historical control cynomolgus macaques. Statistically significant findings can be found in Supplemental Table 3.
Geographic origin-related differences that were not considered toxicologically meaningful included an increased incidence of antemortem findings in Cambodian origin monkeys that primarily included small deformities of the ear or foot and increased incidence of tail bent or missing that were likely the result of husbandry differences at the vendor rather than true origin related differences (Figure 2).
Other factors that affected variations in percentages of total instances of observational categories (behavior/activity, excretion, pelage/skin, clinical medicine, external appearance) among origins in the clinical observations and ophthalmology data (see Supplemental Tables 1 through 5) included a varied distribution of geographic origin among studies of different routes of administration such as emesis on studies with oral gavage as a route of administration and scabbing on studies with subcutaneous/intravenous routes of administration. Likewise, the number of animals from each origin used on study each year were not equivalent.
The most common finding noted was in the category of Pelage/Skin (Figure 2). This category includes the finding of hair sparse/absent, which is the result of grooming behavior of the socially housed NHPs. This finding was equally distributed among males and females of each origin and was not considered toxicologically meaningful.
The most common ophthalmologic findings included cataract and chorioretinitis (Figure 3). Findings of cataract were statistically significant in males (P value = .01) and females (P value = .003) and chorioretinitis was statistically significant in females (P value = .02); however, they were not considered toxicologically meaningful due to low incidence.
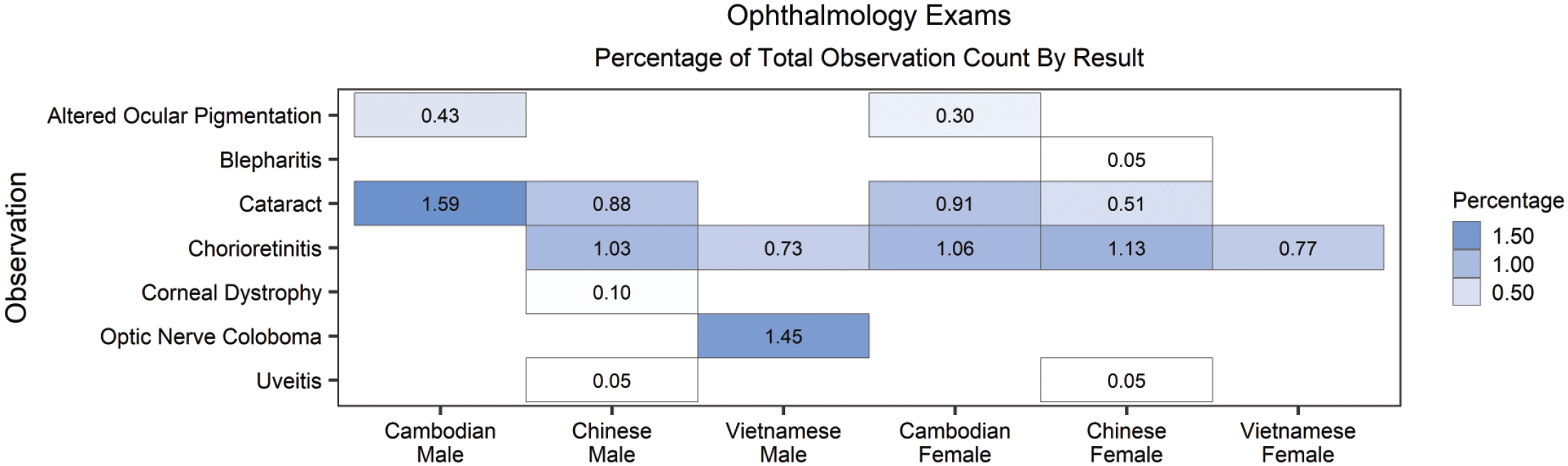
Percentage of ophthalmology observation occurrence by observation result, sex and origin. There were no toxicologically meaningful geographic origin-related differences in clinical observations among historical control cynomolgus macaques. Statistically significant findings (P ≤ .05) included cataract in males and females and chorioretinitis in females (see Supplemental Tables 1 and 2).
Body weights
There were no toxicologically meaningful geographic origin-related differences in body weight among historical control cynomolgus macaques (Figure 4). No significant difference in average body weight was noted at assignment to study (pretreatment) or at the end of study in males or in females (Figure 4, Supplemental Tables 6 and 7) between origins. Variations noted within an origin/sex were likely the result of age disparities at assignment to study. Despite uneven distribution of origin of animals among studies of varying durations, body weights at the final collection between origins were comparable.
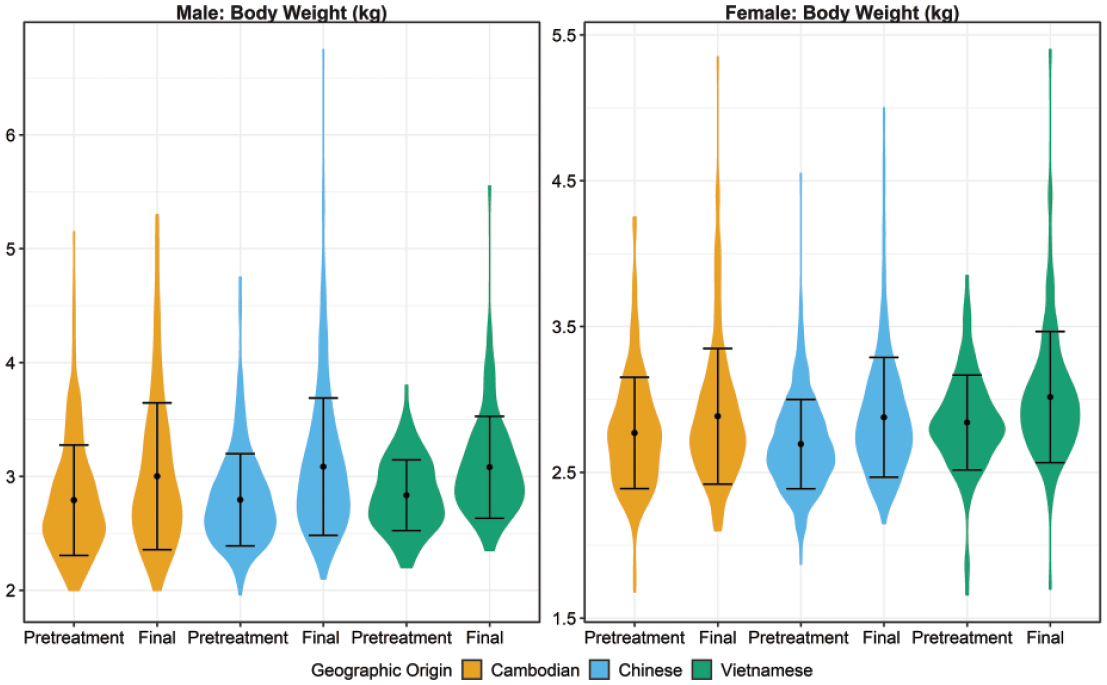
Violin plots for male and female body weights (kg) by origin demonstrating the mean, standard deviation, and distribution of results for comparison of data across geographic origin for each sex at pretreatment and final study intervals. Statistically significant mean pairwise comparisons (P ≤ .01) were observed between origin groups at the pretreatment and final interval for female Cambodian-Chinese, Cambodian-Vietnamese, and Chinese-Vietnamese comparisons.
Clinical Pathology
There were no toxicologically meaningful geographic origin-related differences in routine hematology, coagulation, or clinical chemistry parameters. Parameters included in Figures 5 through 8 were selected to demonstrate that there were no toxicologically meaningful differences in parameters representing major organ systems or processes that may be affected by test article administration, including but not limited to hematopoiesis (eg, absolute neutrophil counts and absolute reticulocyte counts), inflammation (eg, absolute neutrophil counts and fibrinogen concentration), the hepatobiliary system (eg, ALT and ALP activities), and renal function (eg, urea nitrogen, sodium, and chloride concentrations).
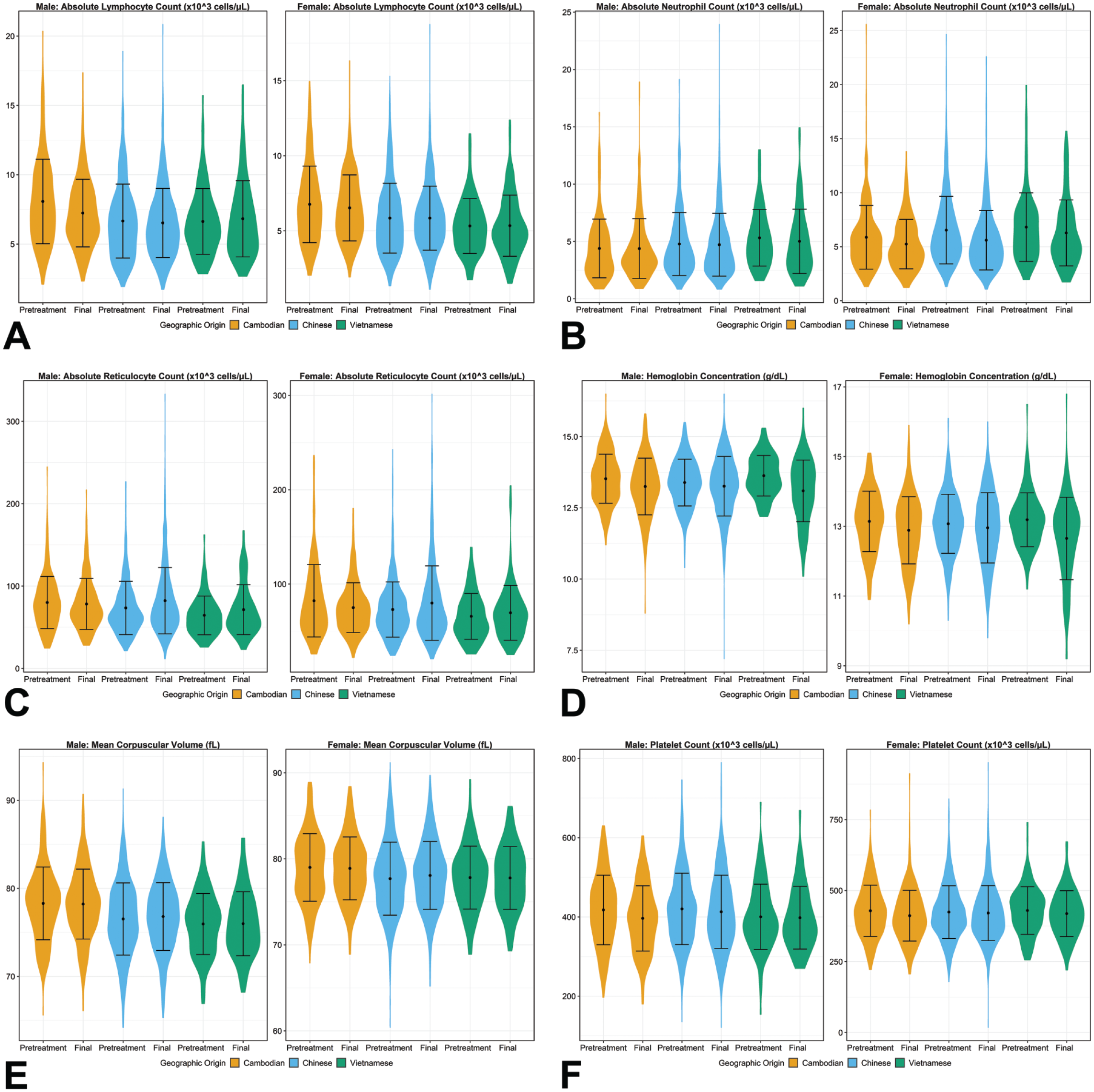
Violin plots for selected hematology parameters ([A] absolute lymphocyte count, [B] absolute neutrophil count, [C] absolute reticulocyte count, [D] hemoglobin, [E] mean corpuscular volume [MCV], and [F] platelet count) demonstrating the mean, standard deviation, and distribution of results for comparison of data across geographic origin for each sex at pretreatment and final study intervals. Statistically significant mean pairwise comparisons (P ≤ .05) were observed between origin groups at the pretreatment interval for absolute lymphocyte count in males for Cambodian-Chinese and Cambodian-Vietnamese comparisons; for absolute neutrophil count in males for all three comparisons; for absolute reticulocyte count in males for all three comparisons; for hemoglobin in males for Cambodian-Chinese and Chinese-Vietnamese comparisons; and for MCV in males for Cambodian-Chinese and Cambodian-Vietnamese comparisons. Statistically significant mean pairwise comparisons (P ≤ .05) were observed at the final interval for absolute lymphocyte count in males for the Cambodian-Chinese comparison and in females for all three comparisons; for absolute neutrophil count in females for all three comparisons; for absolute reticulocyte count in males for Cambodian-Vietnamese and Chinese-Vietnamese comparisons and in females for Cambodian-Chinese and Chinese-Vietnamese comparisons; for hemoglobin in females for the Chinese-Vietnamese comparison; for MCV in males for all three comparisons and in females for Cambodian-Chinese and Cambodian-Vietnamese comparisons; and for platelet count in males for the Cambodian-Chinese comparison.
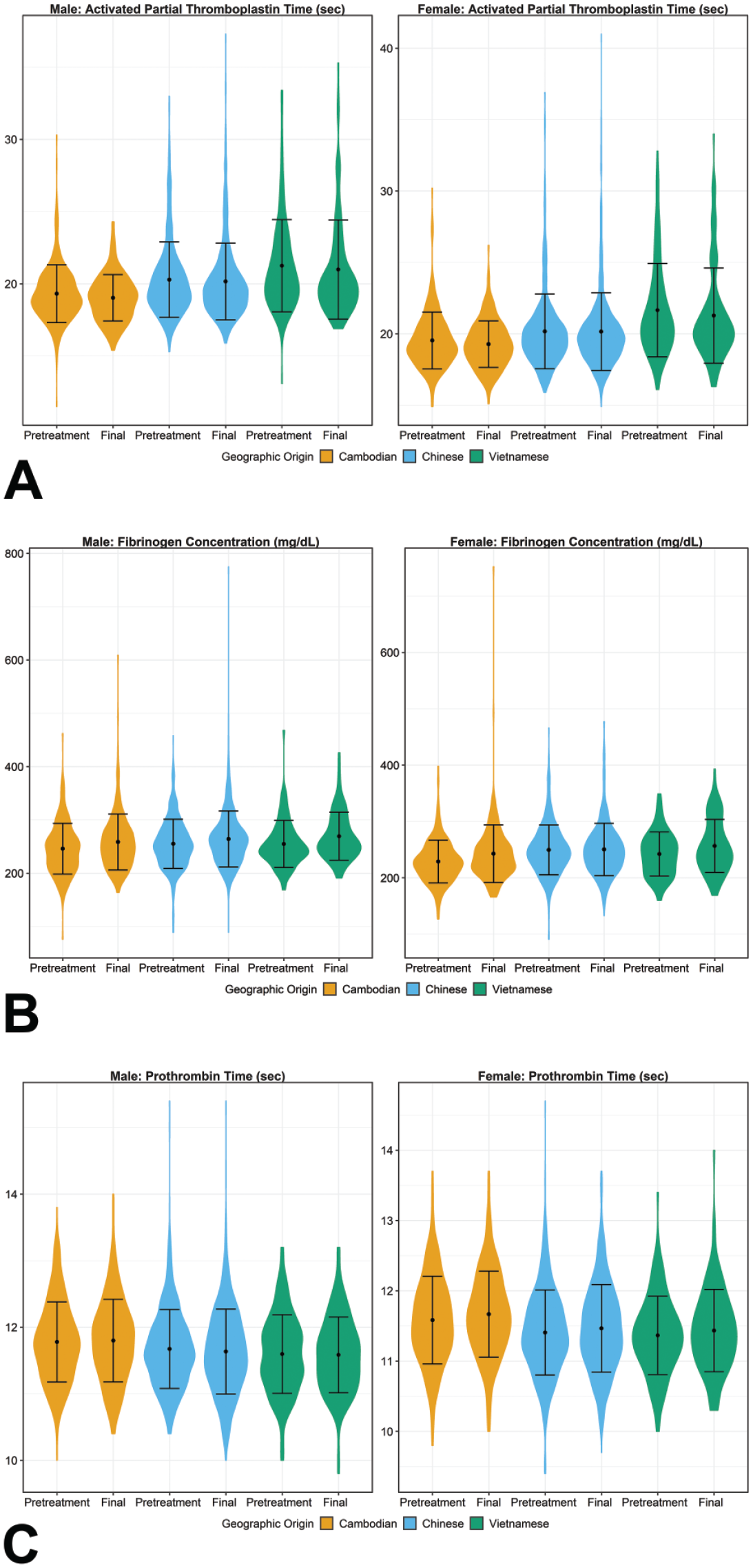
Violin plots for (A) activated partial thromboplastin time (APTT), (B) prothrombin time, and (C) fibrinogen concentration demonstrating the mean, standard deviation, and distribution of results for comparison of data across geographic origin for each sex at pretreatment and final study intervals. No statistically significant mean pairwise comparisons were observed among the three origins at the pretreatment or final interval for coagulation parameters.
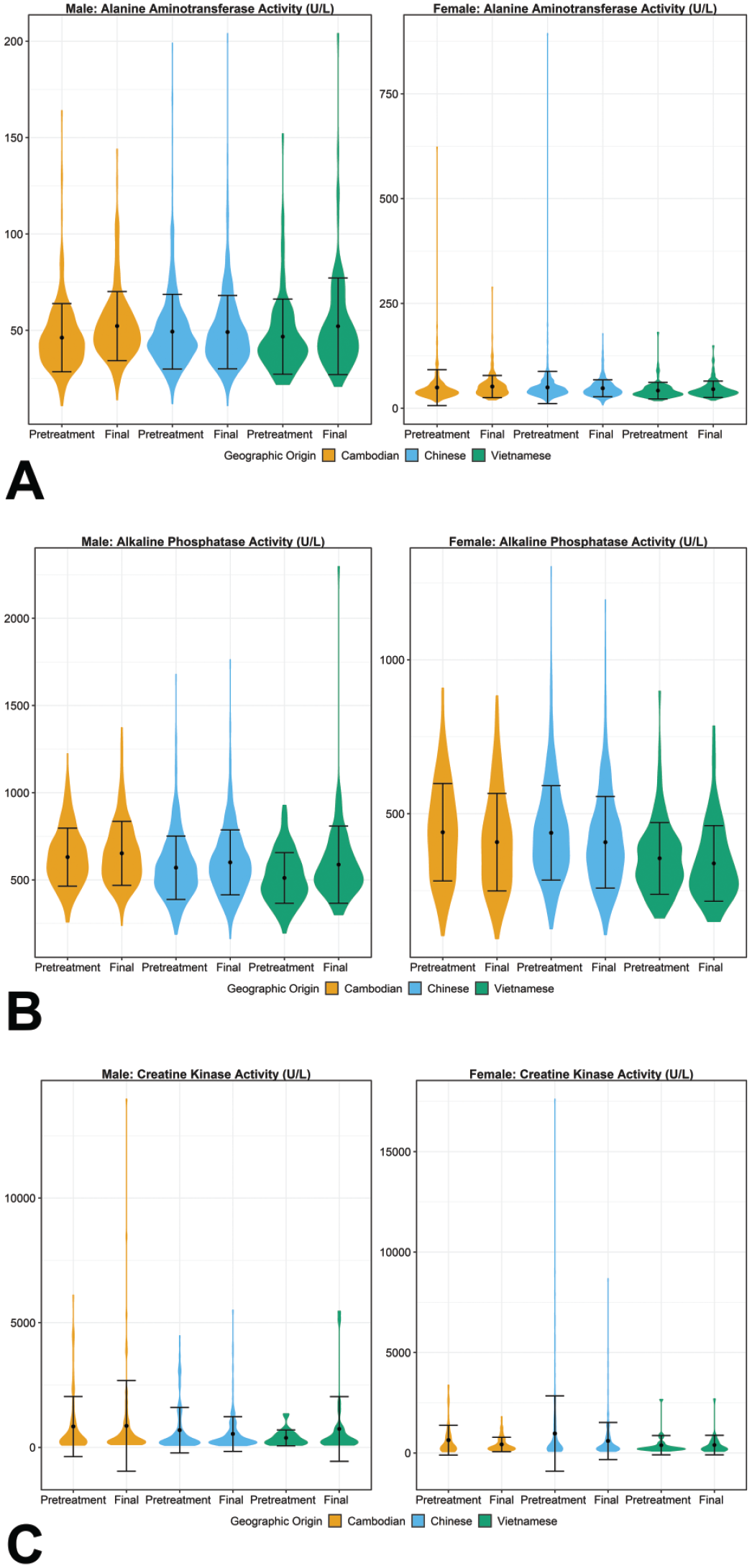
Violin plots for selected clinical chemistry enzyme activities ([A] alanine aminotransferase, [B] alkaline phosphatase, and [C] creatine kinase activities) demonstrating the mean, standard deviation, and distribution of results for comparison of data across geographic origin for each sex at pretreatment and final study intervals. Statistically significant mean pairwise comparisons (P ≤ .05) were observed between origin groups at the pretreatment interval for alkaline phosphatase activity in males for all three comparisons and in females for Cambodian-Vietnamese and Chinese-Vietnamese comparisons. Statistically significant mean pairwise comparisons (P ≤ .05) were observed between origin groups at the final interval for alanine aminotransferase activity in females for Cambodian Chinese and Cambodian-Vietnamese comparisons, and for alkaline phosphatase activity in males for Cambodian-Chinese and Cambodian-Vietnamese comparisons and in females for Cambodian-Vietnamese and Cambodian-Vietnamese comparisons.
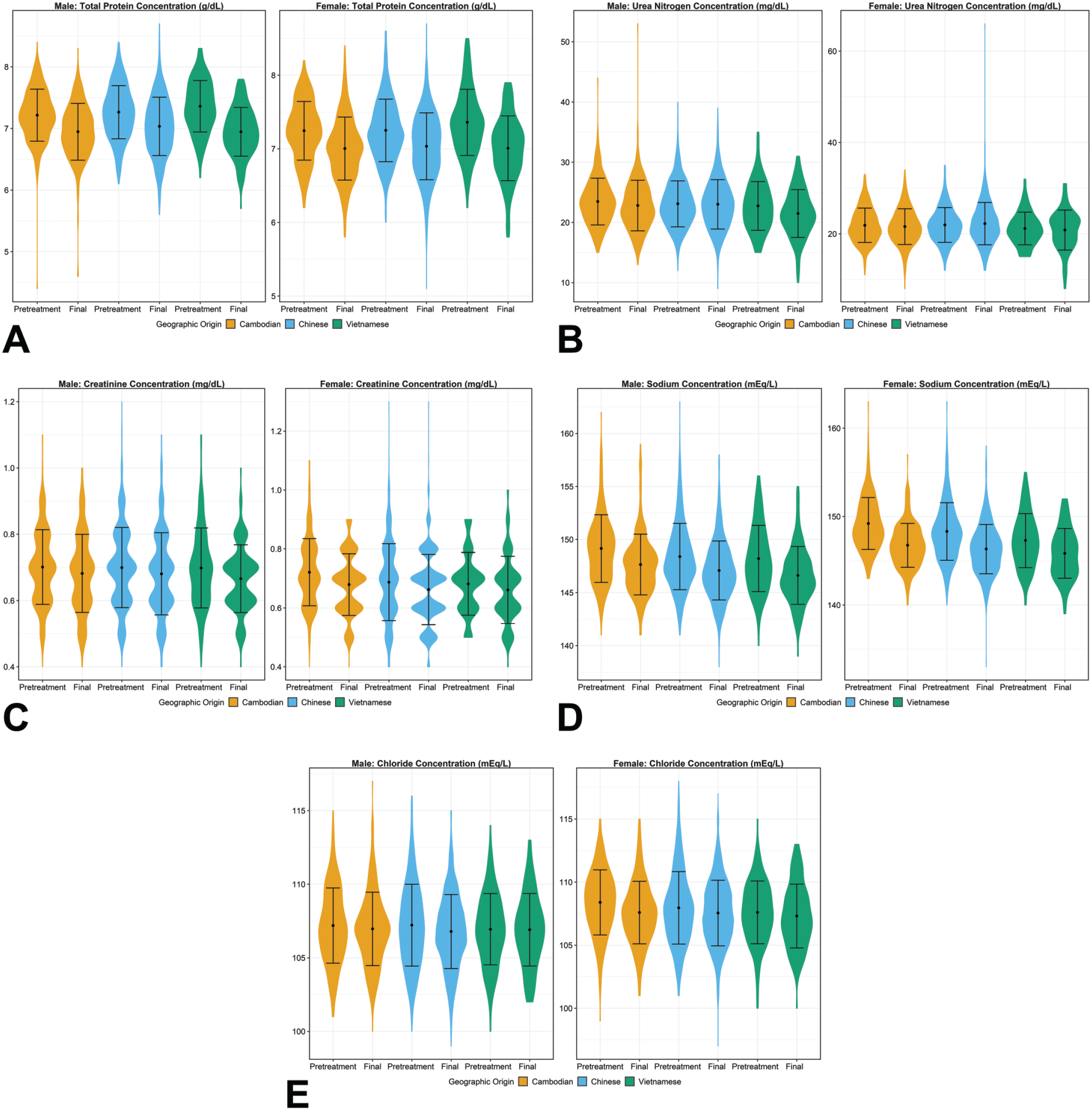
Violin plots for selected clinical chemistry parameters ([A] total protein, [B] urea nitrogen, [C] creatinine, [D] sodium, and [E] chloride concentrations) demonstrating the mean, standard deviation, and distribution of results for comparison of data across geographic origin for each sex at pretreatment and final study intervals. Statistically significant mean pairwise comparisons (P ≤ .05) were observed between origin groups at the pretreatment interval for total protein concentration in both sexes for Cambodian-Vietnamese and Chinese-Vietnamese comparisons; for creatinine concentration in females for Cambodian-Chinese and Cambodian-Vietnamese comparisons; for sodium concentration in males for Cambodian-Chinese and Cambodian-Vietnamese comparisons and in females for all three comparison; and for chloride concentration in females for Cambodian-Chinese and Cambodian-Vietnamese comparisons. Statistically significant mean pairwise comparisons (P ≤ .05) were observed between origin groups at the final interval for total protein concentration in males for Cambodian-Chinese and Chinese-Vietnamese comparisons; for urea nitrogen concentration in males for Cambodian-Vietnamese and Cambodian-Vietnamese comparisons and in females for Cambodian-Chinese and Chinese-Vietnamese comparisons; and for sodium concentration in both sexes for Cambodian-Chinese and Cambodian-Vietnamese comparisons.
Hematology
Males and females from Cambodia had minimally higher mean absolute lymphocyte counts than males and females from both China and Vietnam at the pretreatment and final intervals (Figure 5). Additionally, mean absolute lymphocyte counts were statistically significantly different between Cambodian and Vietnamese males and females at both intervals (all P < .0001), between Cambodian and Chinese males at pretreatment (P < .0001) and females at both intervals (both P < .0001), and between Chinese and Vietnamese females at both intervals (pretreatment P = .01 and final P = .01). However, the differences in absolute values were minimal (≤ 1.44 x 103 cells/µL) and the central 95% ranges overlapped considerably across groups (Figure 5). Violin plots for representative hematology parameters are presented in Figure 5. For the remaining hematology parameters, despite some pairwise comparisons for which mean values were statistically significantly different between geographic origins (see Supplemental Tables 8 through 10), the overall magnitudes of the differences were considered negligible with consideration of physiologic relevance and analytical variability. Additionally, all geographic origins had considerable overlap between central 95% ranges at pretreatment and final intervals (Figure 5). Therefore, none of these differences were considered toxicologically meaningful.
Coagulation
Mean coagulation times (ie, APTT and PT) and fibrinogen concentration were comparable across geographic origins at both pretreatment and final intervals (Figure 6). Despite statistically significant differences between geographic origins for all 3 parameters in both sexes at 1 or both intervals evaluated (see Supplemental Tables 11 through 13), these differences between mean coagulation times remained ≤ 2 seconds different and mean fibrinogen concentrations were ≤ 21 mg/dL different across geographic origins, which were considered within biologic and analytical variation. None of these parameters were considered to have toxicologically meaningful differences due to the negligible magnitude of difference and considerable overlap in central 95% ranges (Figure 6).
Clinical Chemistry
Males and females from both Cambodia and China had minimally higher mean CK activity than in males and females from Vietnam at the pretreatment interval (Cambodian and Chinese origin means up to a 2.5-fold of the Vietnamese origin means) (Figure 7). However, these differences were not statistically significant between geographic origins following either initial analysis or follow-up pairwise comparisons. Additionally, this difference was primarily due to low percentage of Cambodian-origin and Chinese-origin males and females with notably higher CK activity than expected for apparently healthy untreated cynomolgus macaques, and median values for CK activity remained comparable across origins. The hemolytic index was reviewed for these samples when available. Most of these samples had a negative (0) or minimal (1 out of 6) hemolytic index, including samples with CK activity > 12,000 U/L, supporting that sample hemolysis did not contribute significantly to the higher CK activities. The sporadic notably higher CK activity was considered most likely due to a low incidence of procedural (eg, handling or restraint-related) skeletal muscle effects and was not considered to be toxicologically meaningful.
Statistically significant differences were observed for ALT activity between females of Cambodian origin versus both Chinese and Vietnamese origins at the final interval (both P = .01). Additionally, a statistically significant difference was observed for AST activity between females of Cambodian and Chinese origins at the pretreatment interval only (P < .0001). Although statistically significant, these differences in ALT and AST activities were negligible in magnitude (all ≤ 7 U/L differences in group means) and therefore not considered to be toxicologically meaningful.
For ALP activity, statistically significant differences were observed at the pretreatment interval among males of all three origins (all P < .0001) and between females of Vietnamese origin versus both Chinese and Cambodian origins (both P < .0001). Statistically significant differences also were observed for ALP activity at the final interval between males of Cambodian origin versus both Chinese and Vietnamese origins (P < 0.0001 and P = .003, respectively) and between females of Vietnamese origin versus both Chinese and Cambodian origins (both P < .0001). For GGT activity, there were statistically significant differences between males of Cambodian origin versus both Chinese and Vietnamese origins at the pretreatment interval only (both P < .0001). Although the differences between group means were larger for ALP activity (all ≤ 120 U/L) than for GGT activity (all ≤ 10 U/L) or ALT and AST activities discussed above, the higher absolute values for ALP activities than for GGT, ALT, and AST activities contributed to the larger absolute differences between group means. The maximum fold change between group means for ALP activity was 1.24x, which was considered negligible. Due to the overall small differences and the considerable overlap of central 95% ranges across groups for both ALP and GGT activities, the statistically significant differences for these 2 parameters were also not considered to be toxicologically meaningful.
Violin plots for representative clinical chemistry parameters are presented in Figures 7 and 8. For the remaining clinical chemistry parameters, although some pairwise comparisons for which mean values were statistically significantly different between geographic origins (see Supplemental Tables 14 through 16), the overall magnitudes of the differences were considered negligible with consideration of expected physiologic relevance and analytical variability. Additionally, all geographic origins had considerable overlap between central 95% ranges at pretreatment and final intervals. Therefore, none of these differences were considered toxicologically meaningful.
Anatomic Pathology
Slight variations were noted between males and females of Chinese, Cambodian, and Vietnamese origin; however, there were no toxicologically meaningful geographic origin-related differences in organ weights, macroscopic observations, or histopathologic findings.
The findings of highest incidence in males and females of Cambodian, Chinese, and/or Vietnamese origin are noted in Tables 4 and 5. Full incidence tables can be found in Supplemental Tables 17 through 26.
Findings of highest incidence in males of Cambodian, Chinese, and Vietnamese origin.
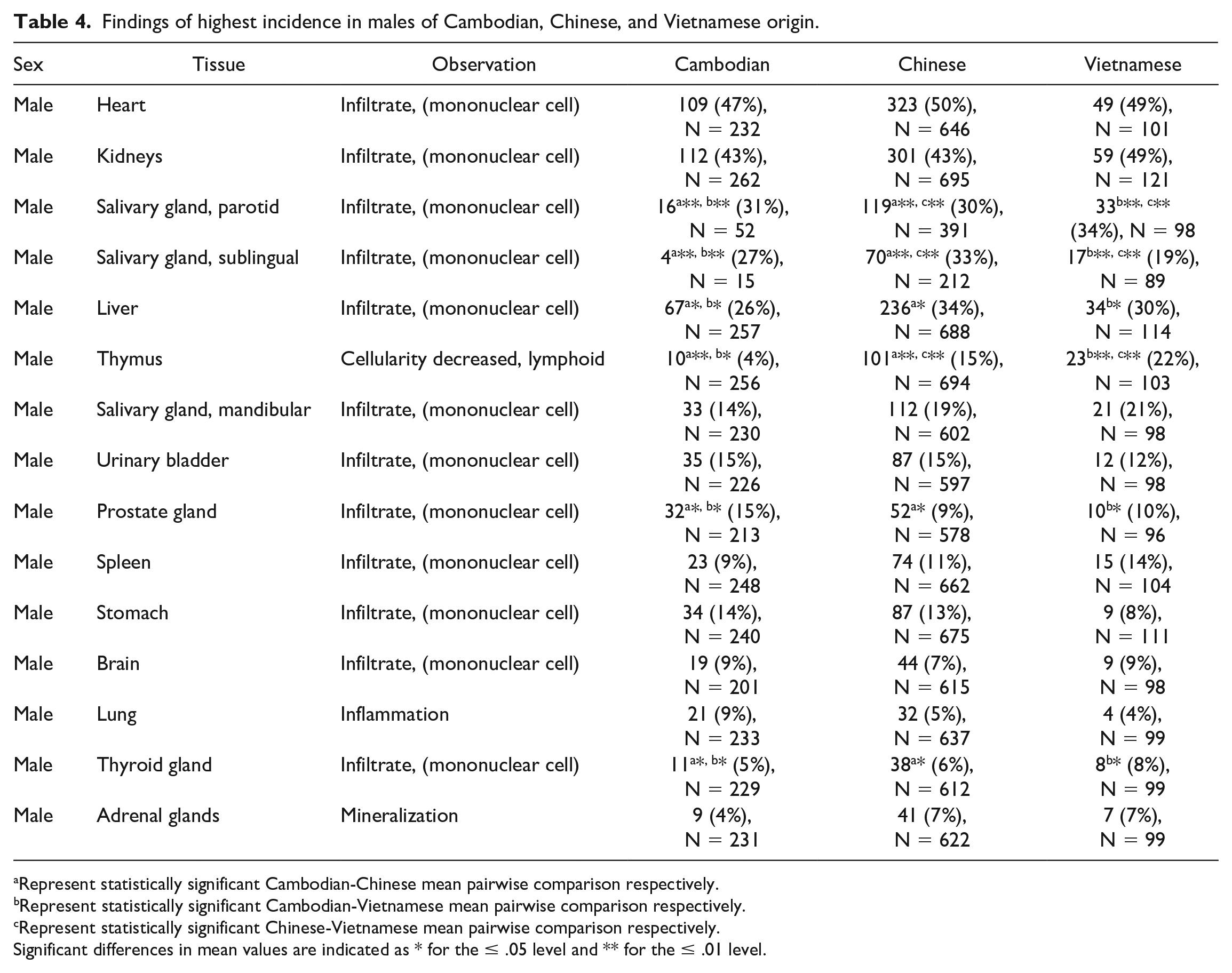
Represent statistically significant Cambodian-Chinese mean pairwise comparison respectively.
Represent statistically significant Cambodian-Vietnamese mean pairwise comparison respectively.
Represent statistically significant Chinese-Vietnamese mean pairwise comparison respectively.
Significant differences in mean values are indicated as * for the ≤ .05 level and ** for the ≤ .01 level.
Findings of highest incidence in females of Cambodian, Chinese, and Vietnamese origin.
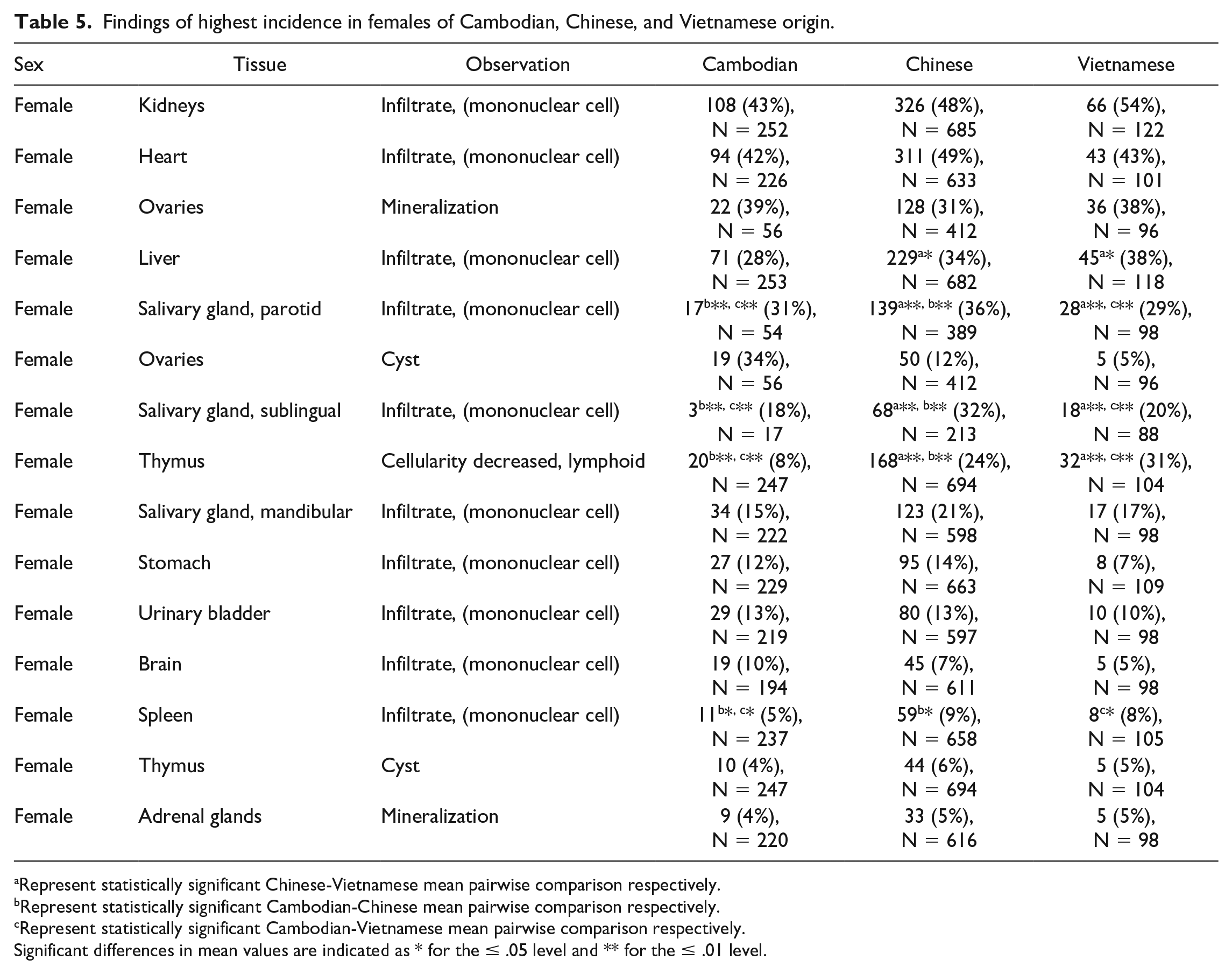
Represent statistically significant Chinese-Vietnamese mean pairwise comparison respectively.
Represent statistically significant Cambodian-Chinese mean pairwise comparison respectively.
Represent statistically significant Cambodian-Vietnamese mean pairwise comparison respectively.
Significant differences in mean values are indicated as * for the ≤ .05 level and ** for the ≤ .01 level.
Incidental findings that were noted to be statistically different in 1 geographic origin over others included decreased thymic lymphoid cellularity in males and females (4%/8% incidence in Cambodian, 15%/24% in Chinese, 22%/31% in Vietnamese males and females respectively; P = .00002), sternal bone marrow lymphoid hyperplasia in males (0% incidence in Cambodian, 2% in Chinese, 6% in Vietnamese; P = .03), cardiac degeneration/necrosis in males (1% incidence in Cambodian, 4% in Chinese, 2% in Vietnamese; P = .03), mammary gland dilatation in females (1.84% incidence in Cambodian, 0.17% in Chinese, 0% in Vietnamese; P = .05). Additionally, statistically significant variation in findings of mononuclear infiltrate were noted in the following organs: parotid salivary gland, sublingual salivary gland, liver, prostate gland, thyroid, kidney, heart, lung, and spleen (see Tables 4 and 5). While these findings were statistically significant, they were not considered to be toxicologically meaningful as they are consistent with previously reported background/incidental findings in cynomolgus macaques and should be interpreted in the context of the study.17-19 For a few of these variations, there was enough of a difference that mixing of sources within a study could lead to numerical imbalances and potentially confound the accurate identification of test article-related changes. This could be particularly impactful for anatomic pathology since there is no pretreatment baseline.
Other common incidental findings that were noted to have increased variation (but were not statistically significant) among males and females from various geographic origins included, increased alveolar macrophages in males (7% incidence in Cambodian, 4% in Chinese, 2% in Vietnamese), pigment in the lung of males and females (0%/0% incidence in Cambodian, 3%/3% in Chinese, 5%/1% in Vietnamese males and females respectively), hepatic fibrosis in females (0% incidence in Cambodian, 1% in Chinese, 3% in Vietnamese), thyroid gland developmental anomalies in males and females (8%/7% incidence in Cambodian, 3%/3% in Chinese, 0%/0% in Vietnamese males and females respectively), adrenal gland mineralization in males (4% incidence in Cambodian, 7% in Chinese, 7% in Vietnamese), ovarian mineralization (9% incidence in Cambodian, 20% in Chinese, 35% in Vietnamese), ovarian cysts (34% incidence in Cambodian, 12% in Chinese, 5% in Vietnamese), pituitary gland cyst in females (7% incidence in Cambodian, 4% in Chinese, 8% in Vietnamese), increased lymphoid cellularity in the mandibular lymph node of males and females (0%/3% incidence in Cambodian, 3%/3% in Chinese, 5%/7% in Vietnamese males and females respectively), lung inflammation in males (9% incidence in Cambodian, 5% in Chinese, 4% in Vietnamese), and decreased lymphoid cellularity in the spleen in females (3% incidence in Cambodian, 3% in Chinese, 6% in Vietnamese). With the exception of decreased lymphoid cellularity in the spleen, these findings are largely consistent with previously reported background/incidental findings in cynomolgus macaques.17-19 Overall, these variations were not considered to be impactful on the interpretation of anatomic pathology data due to low incidence, low magnitude of variation, common biologic presentation, and recognition as common incidental findings.
Macroscopic Observations
Macroscopic observations were noted at low incidence in all geographic groups (see Supplemental Table 27). Epididymal nodules were noted in 5.66% of Vietnamese males, a finding not noted in Cambodian or Chinese males. While statistically significant, this finding was not considered meaningful due to low incidence and lack of any correlative epididymal-related differences in the microscopic data of Vietnamese NHPs compared to Cambodian or Chinese NHPs. Statistically significant variation was noted in red discoloration of the skin in males and females for all geographic locations (see Supplemental Table 27) with increased incidence noted Cambodian and Chinese NHPs and low incidence in Vietnamese NHPs; this finding was not considered meaningful due to lack of correlative data. All other differences in macroscopic observations were attributed to normal biologic variation. There were no trends, patterns, or supportive data to suggest that findings were toxicologically meaningful.
Organ Weights
There were no toxicologically meaningful geographic origin-related differences in organ weight among historical control cynomolgus macaques. Organ weights that were noted to be statistically significant (P ≤ .05) in one geographic origin over others included heart, lung, kidney, and thymus in males and females; brain, salivary gland, and spleen in females; and pancreas and thyroid/parathyroid gland in males. Variations noted within an origin/sex were likely the result of age disparities (studies were not age-matched) or physiologic variation present at assignment to study and/or were not statistically significant. See Supplemental Tables 28 and 29 for organ weight tables.
Discussion
The current study objective was to compile historical control data from healthy cynomolgus macaques originally sourced from China, Cambodia, and Vietnam utilized in nonclinical toxicology studies conducted at a single testing facility (Charles River Laboratories in Mattawan, Michigan, USA) to assess the impact of continental geographic origin on standard nonclinical toxicology study endpoints. Previous studies have demonstrated meaningful differences among various toxicology study endpoints between cynomolgus macaques of Mauritian origin and other insular and/or continental origins. A retrospective study of 90 monkeys (approximately equal number of animals per origin) originating from Mauritius, Vietnam, or the Philippines identified differences in body weight, clinical pathology, and anatomic pathology among those three origins. 20 A prospective study of 60 monkeys (10 monkeys per sex per group) from Mauritius, Cambodia, and a mixed Asian origin (including monkeys originating from Cambodia, Vietnam, and Indonesia) identified differences between monkeys of Mauritian and mixed Asian origins. Except for lower AST activity in the Cambodian only origin group than in the mixed Asian origin group, no other meaningful differences were observed between monkeys of the Cambodian only and mixed Asian origins for clinical, ophthalmology, clinical pathology, or anatomic pathology parameters evaluated. 21 However, neither of these studies evaluated among the distinct continental subpopulations of monkeys originating from China, Cambodia, and Vietnam. In this study, the retrospective comparison of historical clinical, ophthalmology, clinical pathology, and anatomic pathology data enabled the use of a robust sample of control cynomolgus monkeys originating from suppliers in China (n = 750 males/741 females), Cambodia (n = 282 males/271 females), and Vietnam (n = 122 males/120 females) to assess for any toxicologically meaningful differences among these continental subpopulations.
Our results indicate that there were no toxicologically meaningful baseline differences observed among cynomolgus macaques sourced from China, Cambodia, or Vietnam for body weight, clinical observations, ophthalmoscopic observations, clinical pathology parameters or anatomic pathology parameters in this study. Overall, the in-life, clinical pathology, and anatomic pathology data analyzed were quite similar amongst the three geographic origins. The findings of this study suggest that genetic differences and variations in husbandry at the animals’ site of origin had little impact on standard nonclinical toxicology study endpoints for cynomolgus macaques originating in China, Vietnam, and Cambodia.
For end points for which pretreatment data are available, such as body weight, hematology, coagulation, and clinical chemistry, comparison to concurrent controls provides additional support and context for interpretation of potential test article effects. Unfortunately, pretreatment data are not available for macroscopic observations, histopathologic findings, and organ weight data. Findings noted with statistically significant differences among the groups, such as decreased thymic lymphoid cellularity in Cambodian males and females, sternal bone marrow lymphoid hyperplasia in Vietnamese males, decreased cardiac degeneration/necrosis in Cambodian males, increased mammary gland dilatation in Cambodian females, and variable mononuclear infiltrates in many organs were observed; however, these findings were not considered toxicologically meaningful because they are common incidental findings in cynomolgus macaques and should be interpreted within the context of the study at hand.17-19 Notably, this variability could complicate study interpretation if cynomolgus macaques with different geographic origins are intermixed within a single study as they could introduce numeric differences among groups for these parameters.
To our knowledge, this is the largest systematic study of potential variability among cynomolgus macaques sourced from China, Cambodia, and Vietnam. This study had several strengths including large sample size and restriction of assessment to a single study site. Focusing on animals utilized at Charles River Laboratories, Mattawan allowed for review of a large cohort of animals maintained under relatively consistent husbandry practices with standardized practices for in-life assessments and clinical and anatomic pathological assessment. Limitations of this retrospective study include lack of genetic analysis that would confirm animal origin, unknown variation in husbandry and housing practices at site of origin, and the inherent variability in clinical, macroscopic, and microscopic observations across husbandry, clinical, and pathology staff. Additionally, our analysis is restricted to animals imported from China, Cambodia, and Vietnam; future studies are required to analyze the impact of the other geographic locations that are increasingly utilized for nonclinical toxicology studies, such as Mauritius, the Philippines, and Indonesia.
Our findings suggest that Chinese, Vietnamese, and/or Cambodian geographic origin has little impact on baseline variability in the standard nonclinical toxicology study parameters evaluated in this study and demonstrate numerous similarities between cynomolgus macaques sourced from these subpopulations. Regardless of geographic origin, animals undergo multiple evaluations to screen for infectious disease and evaluate overall health prior to import and subsequent placement on a study which helps to minimize interanimal variation, improve study reproducibility, and support animal welfare. While efforts should still always be made to source animals from a single country of origin for each specific study, these findings support the interchangeable use of animals from Chinese, Vietnamese, and Cambodian geographic sources within a drug development program when sourcing of animals from only one geographic origin for the entire program is not possible due to supply, import restrictions, economic, or geopolitical factors.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-1-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-10-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-10-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-11-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-11-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-12-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-12-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-13-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-13-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-14-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-14-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-15-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-15-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-16-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-16-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-17-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-17-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-18-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-18-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-19-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-19-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-2-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-2-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-20-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-20-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-21-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-21-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-22-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-22-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-23-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-23-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-24-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-24-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-25-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-25-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-26-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-26-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-27-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-27-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-28-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-28-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-29-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-29-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-3-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-3-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-30-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-30-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-4-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-4-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-5-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-5-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-6-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-6-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-7-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-7-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-8-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-8-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Supplemental Material
sj-docx-9-tpx-10.1177_01926233221103181 – Supplemental material for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin
Supplemental material, sj-docx-9-tpx-10.1177_01926233221103181 for A Comparison of Historical Control Data From Cynomolgus Macaques (Macaca Fascicularis) of Chinese, Cambodian, and Vietnamese Origin by Molly Liepnieks, Caitlyn Carter, Michael J. Caruso, Zac Lloyd, Michael Muzyka and Daniel Patrick in Toxicologic Pathology
Footnotes
Correction (December 2022):
Michael Caruso and Daniel Patrick’s previous organization has been included below the list of affiliations and their preceding/current affiliations are published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
