Abstract
Thyrotropin-releasing hormone (TRH) and TRH-like peptides carry a therapeutic potential for neurological conditions. Nanoparticles (NP) made of the biodegradable polymer, Poly(Sebacic Anhydride) (PSA), have been developed to carry TRH, intended for intranasal administration to patients. There is limited information on the safety of biodegradable polymers when given intranasally, and therefore, we have performed two preclinical safety and toxicity studies in cynomolgus monkeys and rats using TRH-PSA nanoparticles. The rats and monkeys were dosed intranasally for 42 days or 28 days, respectively, and several animals were followed for additional 14 days. Animals received either placebo, vehicle (PSA), or different concentrations of TRH-PSA. No systemic adverse effects were seen. Changes in T3 or T4 concentrations were observed in some TRH-PSA-treated animals, which did not have clinical or microscopic correlates. No effect was seen on TSH or prolactin concentrations. In the monkey study, microscopic changes in the nasal turbinates were observed, which were attributed to incidental mechanical trauma caused during administration. Taken together, the TRH-loaded PSA NPs have proven to be safe, with no local or systemic adverse effects attributed to the drug loaded nanoparticles. These findings provide additional support to the growing evidence of the safety of peptide-loaded NPs for intranasal delivery and pave the way for future clinical trials in humans.
Introduction
Thyrotropin-releasing hormone (TRH) is a well-characterized neurohormone that controls the activity of the hypothalamus-pituitary-thyroid axis. 1 In addition, TRHergic neurons that are non-hypophysiotropic are common in other locations of the brain, and bear neuromodulatory effects that can affect mood-associated activities. 1 Therefore, TRH and TRH-like peptides carry a therapeutic potential for different neurological conditions, such as bipolar disorders, feeding regulation, Parkinson’s diseases, depression, epilepsy, acute spinal trauma, schizophrenia, anxiety, and Alzheimer’s disease.2-13 However, major hurdles in the clinical use of TRH is its rapid metabolism in the liver, kidney and stomach.14,15 and its very short plasma half-life of 8 minutes, resulting mainly because of its quick degradation by the TRH specific pyroglutamyl aminopeptidase serum enzyme. 16 To overcome these obstacles, large amounts of TRH are administered intravenously, leading commonly to unwanted effects, such as nausea, flushing and cardiovascular symptoms from the early high plasma concentrations. 17
In recent years, there is a growing interest in the intranasal route for drug administration, as a non-invasive way for local, systemic or central nervous system distribution.18,19 Since intranasal administration can expedite the crossing of the blood-brain barrier of several peptides,20,21 it has also been suggested as a potential route for TRH administration.22,23 Nevertheless, when administered in aqueous solution, it is challenging to enable nasal absorption and entrance through the nasal epithelium of the hydrophilic and charged TRH peptide. 24 To overcome these hurdles, it is possible to use TRH loaded/encapsulated polymeric nanoparticles (NPs), which can facilitate permeability, absorbability, and retention of the peptide drug.25-28 The effectiveness of TRH delivery systems to the brain have been demonstrated by this team.21,23,29-31
Therefore, a novel development for TRH administration utilizes NPs that are based on fast degrading biodegradable PSA, which degrades in a solely aqueous hydrolysis to the naturally occurring sebacic acid, in a rapid rate. 32 In addition, PSA is a constituent of the Gliadel® brain implant, which has been used in humans for over two decades and proven to be safe. 33 In vitro studies have previously shown the safety of TRH-PSA NPs in low concentrations in cell toxicity assays, as well as its degradation and cell adsorption characteristics. 27 Nevertheless, it is essential to also test the safety of TRH-PSA in well-designed animal toxicity studies. Therefore, we performed two animal toxicity studies in cynomolgus monkeys and rats for which the study design was based on the principles of the Food and Drug Administration Center for Drug Evaluation and Research (CDER)/International Conference on Harmonisation (ICH) Harmonised Tripartite Guidelines ICH-M3(R2), Nonclinical Safety Studies for the conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals (CDER, January 2010). The studies were performed in Covance Laboratories, which is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). All procedures were in compliance with applicable animal welfare acts and were approved by the local Institutional Animal Care and Use Committee (IACUC).
Materials and Methods
The test article, THR-loaded PSA nanoparticles, was produced under GMP conditions at Nextar Chempharma Solutions, Weizmann Science Park, Ness Ziona, Israel. 27 The safety study was performed in Labcorp Early Development Laboratories Inc., as service to Indiana University.
Thyrotropin-releasing hormone (TRH), clinical grade, was procured from Bachem Americas, Inc. PSA was synthesized from melt condensation of sebacic acid (SA) in the presence of acetic anhydride. 27 The anhydride bonds of the formed PSA were confirmed by FTIR, melting point of 82°C by differential scanning calorimetry and molecular weight of 14,000 as determined by gel permeation chromatography, TRH-loaded poly(sebacic acid), PSA, NPs were prepared using an antisolvent nanoprecipitation method, as follows: 54 mg PSA and 6 mg of TRH were separately dissolved in 2.85 mL dichloromethane and 0.5 mL of ethanol, respectively. Then, the TRH solution was mixed into the polymer solution and added to the antisolvent, heptane, by means of a syringe with 30G needle positioned directly in the medium very slowly under moderate magnetic stirring, such that the injection lasted for 3 min. The dispersing antisolvent medium was heptane with 240 mg of lyophilized mannitol and 1 mg/mL of spanTM 80 (Sigma-Aldrich). The formed NPs attached to the mannitol microparticles were isolated from the antisolvent by centrifugation for 20 min at 6000 rpm and decantation of the solvent. The freshly formed TRH-PSA NPs were then dried under vacuum in a desiccator. The final product was kept in a sealed glass vial at −20°C until use. The final particle size of the nanoparticles was 240 + 20 nm (Supplementary Figure 1). Details on the preparation and characterization of the nanoparticles used in the studies have been described previously. 27
Rat Study
Rats have been selected for this study since they have historically been used in safety evaluation studies and are recommended by appropriate regulatory agencies. Sixty male and female each Crl:CD(SD) rats were received by Labcorp from Charles River Laboratories (Raleigh, North Carolina). At initiation of dosing, animals were 8 to 9 weeks old, and their body weights ranged from 285 to 332 g for males and 207 to 263 g for females. Animals were group-housed (up to three animals/sex/cage) and were offered Certified Rodent Diet #2014C and water ad libitum. They were maintained in a temperature range of 20 to 26°C, a relative humidity range of 30 to 70%, eight or greater air changes/hour, and a 12-hour light/12-hour dark cycle.
Rat Study Design.
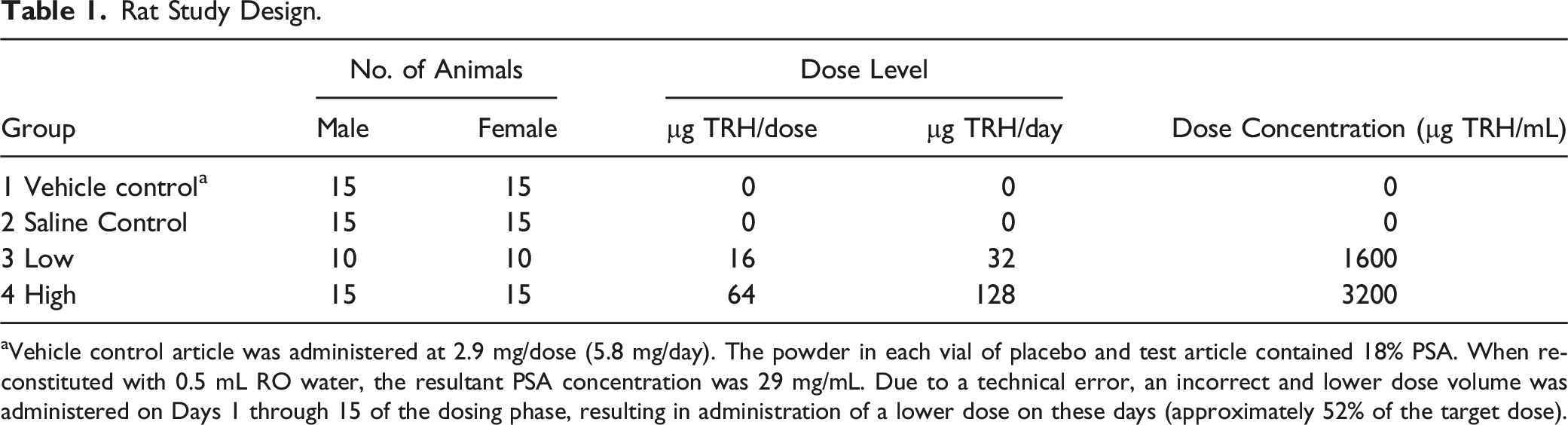
aVehicle control article was administered at 2.9 mg/dose (5.8 mg/day). The powder in each vial of placebo and test article contained 18% PSA. When reconstituted with 0.5 mL RO water, the resultant PSA concentration was 29 mg/mL. Due to a technical error, an incorrect and lower dose volume was administered on Days 1 through 15 of the dosing phase, resulting in administration of a lower dose on these days (approximately 52% of the target dose).
Intranasal dosing was conducted using an adjustable micropipette, and a new pipette tip was used for each animal. Appropriate care was taken to avoid potential tissue damage resulting from contact of intranasal tissues with the pipette tip. The animal was held in an upright position that resulted in the head being tilted back during administration and the dose volume was instilled into a nostril. The animal remained in this position for at least 5 seconds after completion of dosing. The left nostril was administered the dose first, followed by the right nostril.
All animals were assessed for mortality, clinical observations, body weights, food consumption, ophthalmic observations, and clinical and anatomic pathology. Full organ list was checked microscopically in all animals at terminal sacrifice in Groups 1, 2 and 4 and from macroscopic lesions from all Group 3 animals. Blood samples were collected for prolactin and thyroid hormone analyses. On Day 43 of the dosing phase, 10 animals/sex/group were euthanized, and all other animals were euthanized after 15 days (recovery phase).
Cynomolgus Monkey Study
Monkeys have been selected for this study since they have historically been used in safety evaluation studies and are recommended by appropriate regulatory agencies. Twenty-one male and 21 female cynomolgus monkeys (Macaca fascicularis), of Asian origin, were received from the facility now operated by Envigo in Alice, Texas. At initiation of dosing, animals were 29 to 35 months old, and their body weights ranged from 2.3 to 3.5 kg for males and 2.2 to 2.8 kg for females. Animals were housed in stainless steel cages. When possible, animals were socially housed by sex, with up to three animals/cage. Animals were individually housed for study-related procedures.
Animals were offered Certified Primate Diet #5048 (PMI Nutrition International Certified LabDiet®) one to two times daily and had free access to water. They were maintained in a temperature range of 20 to 26°C, a relative humidity range of 30 to 70%, eight or greater air changes/hour, and a 12-hour light/12-hour dark cycle.
Cynomolgus Monkey Study Design.
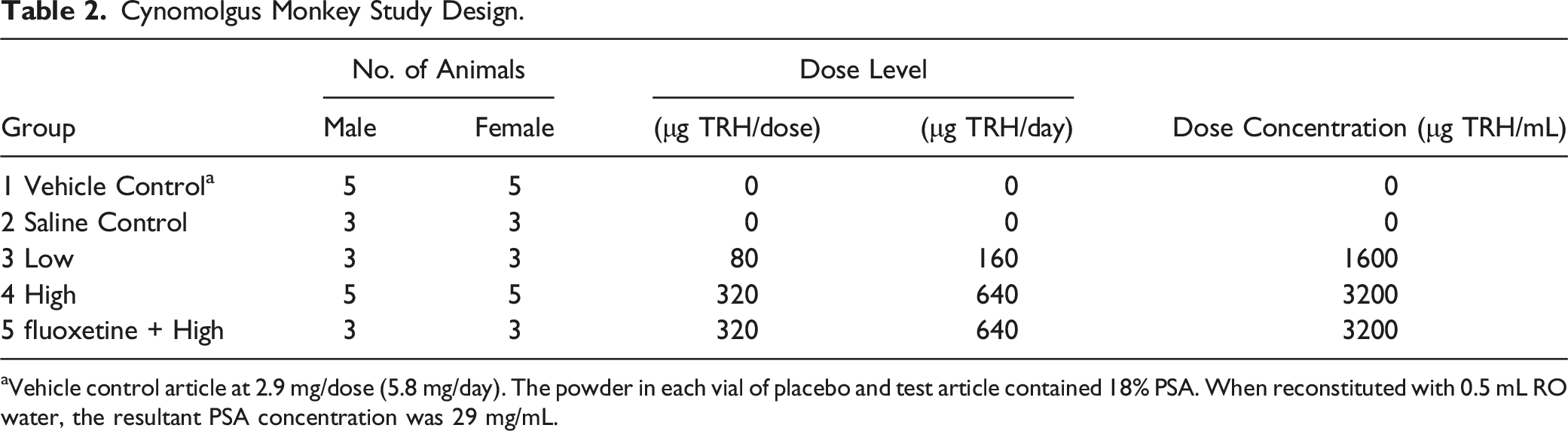
aVehicle control article at 2.9 mg/dose (5.8 mg/day). The powder in each vial of placebo and test article contained 18% PSA. When reconstituted with 0.5 mL RO water, the resultant PSA concentration was 29 mg/mL.
The animal was held in an upright position during dosing. The left nostril was administered the dose first, followed by the right nostril, allowing at least 20 seconds between the doses. A cleaned nhpPOD® (Precision Olfactory Delivery nasal device (Impel NeuroPharma) tip was used for each animal/dose. The tip was inserted just beyond the nostril opening, approximately 5 mm in depth. The tip was first rinsed by drawing and expelling 70% ethanol through the tip multiple times or until clean. The rinsed tip was then dried by repeatedly expelling air through the tip with a syringe until no visible residue remained. Tips were allotted to specific groups and stored following cleaning in a group specific container for reuse as needed, in order to minimize the chance for cross contamination.
All animals were assessed for mortality, clinical observations, body weights, body weight change, qualitative food consumption, ophthalmic observations, electrocardiographic (ECG) measurements, and clinical and anatomic pathology. Full organ list was checked microscopically in all animals from all groups. On Day 29 of the dosing phase, three animals/sex/group were euthanized, and all other animals were euthanized after 15 days (recovery phase).
Results
The test material, TRH loaded nanoparticles, was prepared by anhydrous precipitation of the PSA polyanhydride and TRH in ethanol/dichloromethane solution in excess heptane containing mannitol microparticles. Nanoparticles of about 150 nm were obtained and saved under anhydrous conditions to avoid hydrolysis. TRH loading in the particles was 10% w/w. The preparation method, particle visualization and in vitro release are given in Figure 1. Preparation of THR-PSA nanoparticles: preparation process (A), nanoparticles visualization by SEM (B) and in vitro release of TRH in phosphate buffer pH7.4 at 37°C (C).
Rat Study
All rats survived to their scheduled necropsy. No differences in clinical observations, body weight, food consumption, or ophthalmic observations were attributed to either PSA or TRH-PSA in animals administered 32 or 128 µg TRH/day. No PSA or TRH-PSA-related change in hematology, coagulation, clinical chemistry, or urinalysis parameters or thyroid stimulating hormone (TSH) concentrations occurred in animals administered 32 or 128 µg TRH/day. No changes in organ weights or any macroscopic observation were observed during the dosing or recovery phase, nor any microscopic observations during the dosing phase were found.
Thyroid Hormone Levels in the Rat Study.
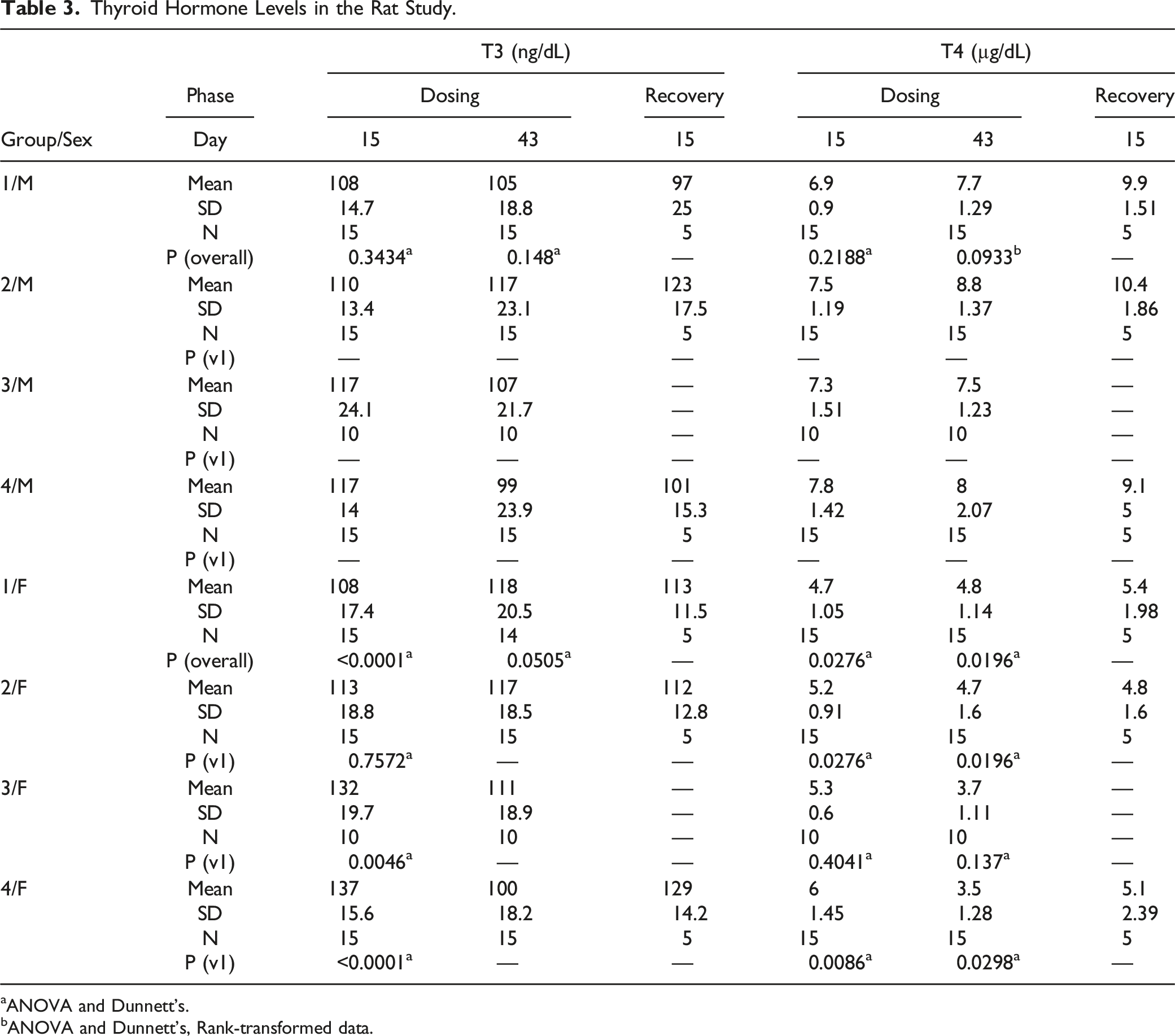
aANOVA and Dunnett’s.
bANOVA and Dunnett’s, Rank-transformed data.
Cynomolgus Monkey Study
No PSA or TRH-PSA related mortality occurred. All animals survived until their scheduled necropsy interval. No differences in clinical observations; body weights; qualitative food consumption; ophthalmic observations; ECG changes; hematology, coagulation, or urinalysis tests; organ weights; or macroscopic findings were attributed to TRH-PSA or fluoxetine + TRH-PSA.
Thyroid Hormone Levels in the Monkey Study.
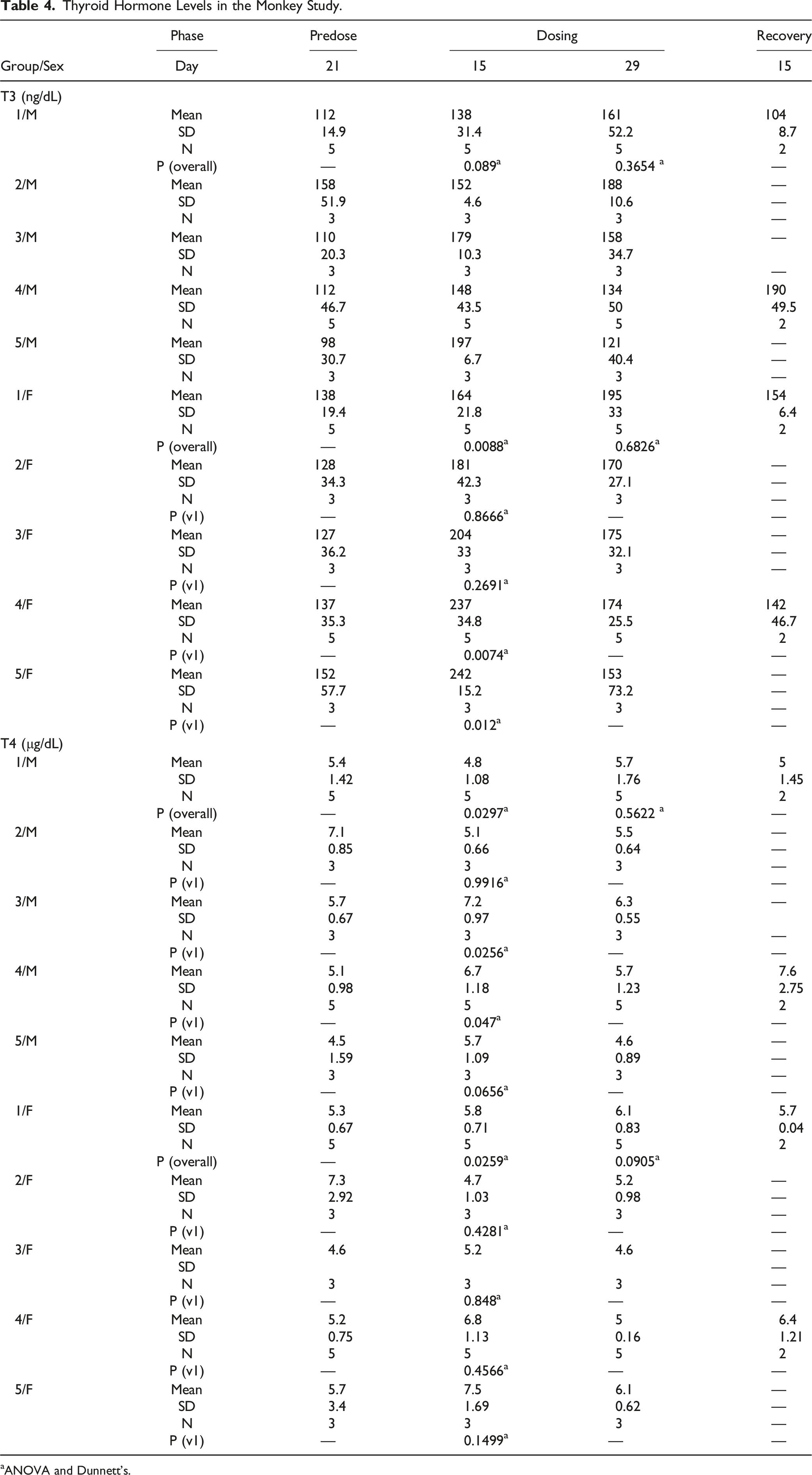
aANOVA and Dunnett’s.
Incidence and Severity of Selected Microscopic Findings in the Nasal Sections of Monkeys-Terminal Sacrifice.
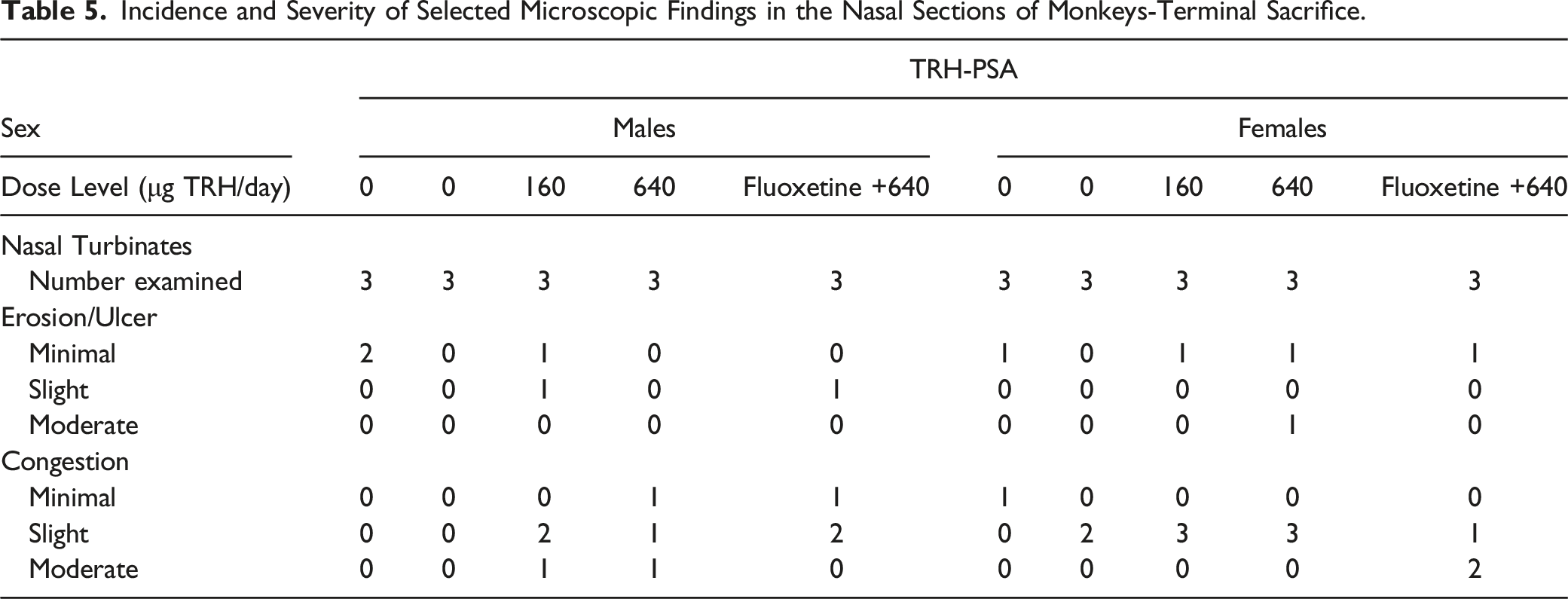
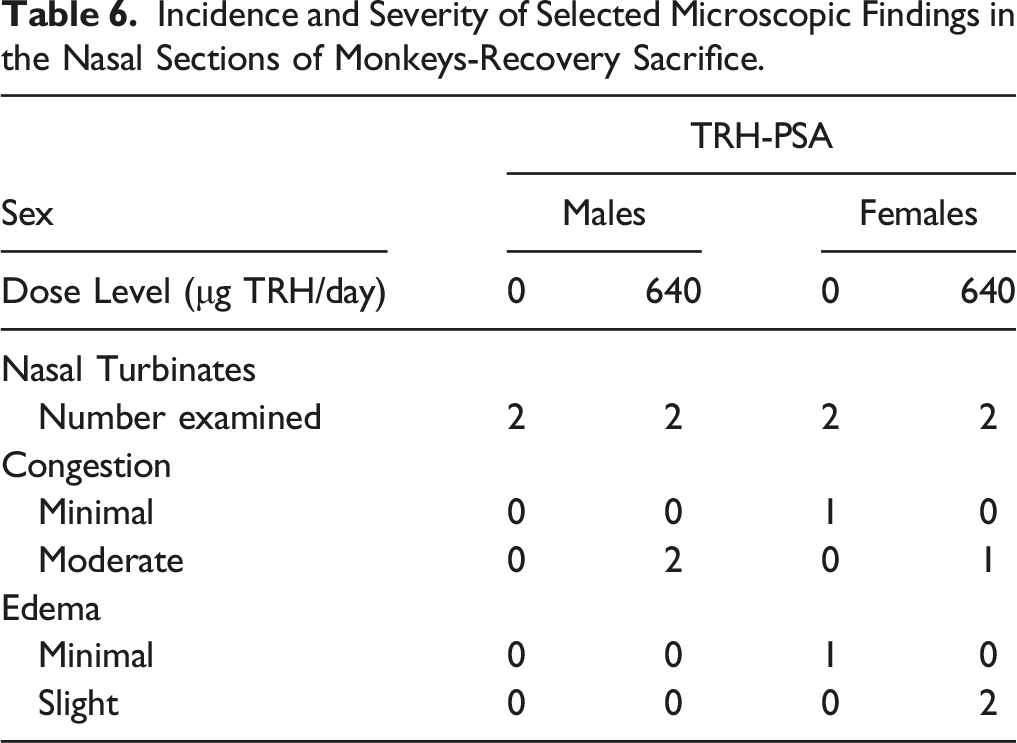
Incidence and Severity of Selected Microscopic Findings in the Nasal Sections of Monkeys-Recovery Sacrifice.
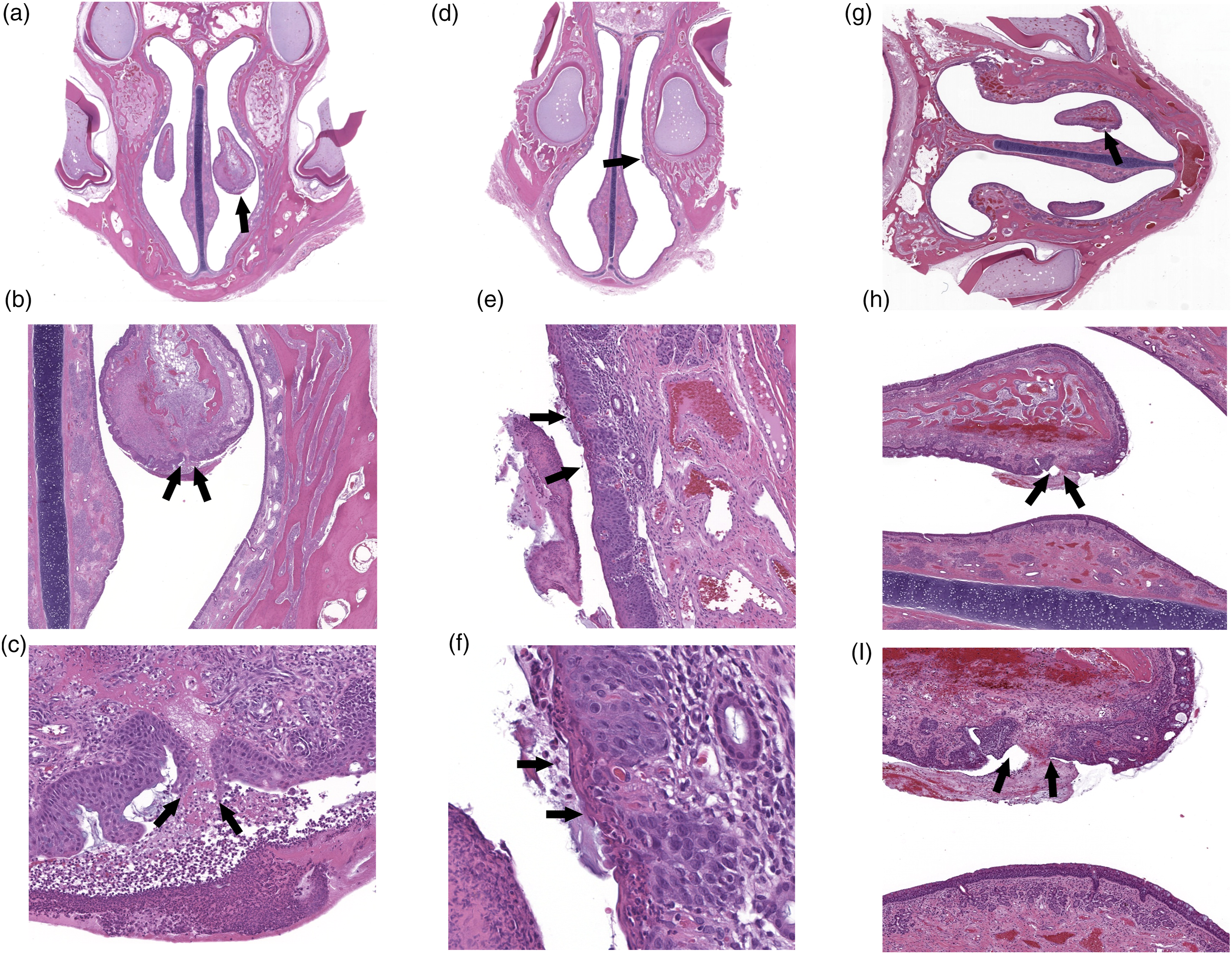
A-C. Section of the nose, level I, from a male vehicle control monkey (Group 1). Note minimal focal erosion/ulcer (arrows), consisting of lost squamous epithelium with neutrophilic exudate in the lumen, with inflammation in the underlying submucosa, located in the maxilloturbinates. The lesion is considered to be caused by incidental mechanical trauma from the nasal device. A-original magnification ×1; B-original magnification ×10; C-original magnification ×20). D-F. Section of the nose level I, from a female vehicle control monkey 2. (Group 1). Note minimal focal erosion/ulcer (arrows), consisting of lost squamous epithelium with neutrophilic exudate in the lumen, with inflammation in the underlying submucosa, located in the lining epithelium of the lateral wall. The lesion is considered to be caused by incidental mechanical trauma from the nasal device. D- original magnification ×1; E-original magnification ×10; F-original magnification ×20). G-I. Section of the nose, level I, from a male low dose monkey (Group 3). Note minimal focal erosion/ulcer (arrows), consisting of lost squamous epithelium with neutrophilic exudate in the lumen, with inflammation in the underlying submucosa, located in the maxilloturbinates. The lesion is considered to be caused by incidental mechanical trauma from the intranasal device. G-original magnification ×1; H-original magnification ×10; I-original magnification ×20).
As the lesions were seen in both the control and treated groups, and characteristically were focal, localized to the mucosa and submucosa in the nasal epithelium and the maxilloturbinates of section level I, and without suggestion of dose-relations, the lesions were not considered as related to the treatment, but rather caused by incidental mechanical trauma from the nasal device.
Discussion
There is growing evidence that NPs can be a safe and effective way of delivering drugs to the brain when administered intranasally, and there are several products being developed as therapeutic and diagnostic tools. 34 Unfortunately, there is limited information in the literature on the in vivo safety studies of nanomaterials when administered intranasally. The available information generally shows good safety data. For example, polycaprolactone-encapsulated NPs were found to be safe when administered to rats, even when given repeatedly. 35 N,N-dimethyl-3β-hydroxycholenamide incorporated in PEG-nanometric micelles and administered intranasally to mice had no safety issues and was shown to be biodegradable. 36 Another example is the use of cell-penetrating peptides, which were modified on the surface of PEG-PLA NPs and given intranasally to rats. Here, as well, the NPs were found to be safe with no prominent adverse events.
However, the use of biodegradable materials can be accompanied by adverse effects, such as evoking inflammatory responses and a foreign body reaction. Therefore, the development of a new delivery device should include well-designed, regulated in vivo safety and toxicity studies.37-45 Therefore, we performed two animal safety studies, one in rats another in cynomolgus monkeys, which historically have been used in safety evaluation studies and are recommended by appropriate regulatory agencies. Furthermore, it was essential to test whether the use of TRH in an intranasal route of administration can lead to any adverse events, since there is very little information on the in vivo safety of TRH when given intranasally.22,23 Our detailed safety evaluation did not reveal any local or systemic adverse effects following the intranasal administration of TRH-PSA, further confirming the safety of this biodegradable polymer and the safety of PSA when administered to the brain. 33
Microscopic findings were noted in the nasal turbinates of the treated monkeys, consisting mostly of congestion, and, less commonly, erosion/ulceration. Such changes were found in both the TRH-PSA-treated animals and in the control animals (both vehicle and saline) and did not manifest clinically in the animals. Local histopathological changes in the nasal application sites have been reported before in other animal models where different nasal application devices were used. These included, for example, inflammation and ulceration, 19 which were attributed to mechanical trauma. Such traumatic effects can also be seen in humans using intranasal sprays, including reports on epistaxis and, rarely, ulceration and septal perforation. 46 In our monkey study, the Precision Olfactory Delivery (POD) nasal device (Impel NeuroPharma) was used for intranasal dosing. This is a propellant activated device that has been used in humans. 47 In the SNIFF trial, which included 289 adult patients with Alzheimer disease, between 5-6% of adverse events were rated as associated with the device. 48 To the best of our knowledge, the results of the additional clinical trials in which this device was used are not yet available. 49
While there is a significant similarity between the human and Cynomolgus monkeys’ anatomy, differences in anatomy between the two species exist. 50 Therefore, devices that have been developed for humans might result in mechanical damage when used in monkeys. The POD device that was used in this study was also reported in the past for intranasal delivery in primates, in both cynomolgus monkeys, 51 as in the current study, and in Rhesus macaques. 52 In both cases, no damage was reported in the monkeys, but in both studies, histopathological evaluation was not performed, and the administration was performed once, and was not repeated administration, as in our study.
Overall, the newly developed TRH-loaded PSA NPs have shown to be safe in both rat and monkey studies, with no local or systemic adverse effects that are attributed to the drug. These findings add to the growing evidence of the safety of peptide-loaded NPs for intranasal delivery and pave the way for future clinical trials in humans.
Supplemental Material
Supplemental Material - Preclinical In-Vivo Safety of a Novel Thyrotropin-Releasing Hormone-Loaded Biodegradable Nanoparticles After Intranasal Administration in Rats and Primates
Supplemental Material for Preclinical In-Vivo Safety of a Novel Thyrotropin-Releasing Hormone-Loaded Biodegradable Nanoparticles After Intranasal Administration in Rats and Primates by Yuval Ramot, Yakir Rottenberg, Abraham J. Domb, Michael J. Kubek, Kevin D. Williams and Abraham Nyska in International Journal of Toxicology
Footnotes
Author Contributions
Ramot, Y. contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Rottenberg, Y. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Domb, A.J. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Kubek, M.J. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Williams, K.D. contributed to analysis and interpretation and critically revised manuscript; Nyska, A. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The TRH nanoparticles were tested for safety as part of FDA submission of this formulation as nasal spray delivery of TRH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was a service work performed by Labcorp Early Development Laboratories Inc., WI, USA, sponsored by Indiana University, Indianapolis, Indiana, USA.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
