Abstract
The IQ Consortium NHP Reuse Working Group (WG) comprises members from 15 pharmaceutical and biotechnology companies. In 2020, the WG developed and distributed a detailed questionnaire on protein non-naïve NHP reuse to the WG member companies. The WG received responses from key stakeholders including principal investigators, facility managers, animal welfare officers and research scientists. This paper’s content reflects the consolidated opinion of the WG members and the questionnaire responses on the subject of NHP reuse within nonclinical programs at all stages of research and development. Many of the pharmaceutical companies represented in the working group or participating in the questionnaire have already achieved some level of NHP reuse in their nonclinical programs, but the survey results suggested that there is significant potential to increase NHP reuse further and a need to understand the considerations involved in reuse more clearly. The WG has also focused carefully on the inherent concerns and risks of implementing protein non-naive NHP reuse and has evaluated the best methods of risk assessment and decision-making. This paper presents a discussion on the challenges and opportunities surrounding protein non-naïve NHP reuse and aims to stimulate further industry dialogue on the subject and provide guidance for pharmaceutical companies to establish roadmaps and decision trees enabling increased protein non-naïve NHP reuse. In addition, this paper represents a solid basis for collaborative engagement between pharmaceutical and biotechnology companies with contract research organizations (CROs) to discuss how the availability of protein non-naïve NHP within CROs can be better leveraged for their use within nonclinical studies.
Introduction
Nonhuman primate (NHP) models are important for pharmaceutical research where their genetic, immunologic, and physiologic similarities to humans have made them excellent models for the robust nonclinical to clinical translation of the safety, pharmacology, and pharmacokinetics of candidate therapeutics. Despite the significant advances in in vitro/in silico alternatives to animal models, and efforts within the pharmaceutical industry to maximize the amount of data generated from the fewest number of in vivo NHP studies, 1 the number of NHPs used in biomedical research has increased in recent years. The most recently available data shows that over 74 000 NHPs were used in the United States in 2017, an increase of 22% relative to 2015.2,3 Recent regulatory guidance acknowledges the influence of the COVID-19 pandemic on the demand for NHPs for the testing of treatments and vaccine candidates and the resulting impact to pharmaceutical development programs. 4 NHPs are often the only pharmacologically relevant species available for safety studies used for the development of therapeutic proteins (e.g., monoclonal antibodies, antibody drug conjugates, fusion proteins, proteins, peptides) as required by international regulatory guidelines (i.e., ICH S6(R1), ICH M3(R2), and ICH S9).5-8 The inherent nature of these modalities (i.e., generally longer lasting pharmacological activity compared to small molecules and greater potential for immunogenicity) has led to the adoption of cautious approaches within the industry relevant to the reuse of protein non-naïve NHPs. The frequency of protein non-naïve NHP reuse across the pharmaceutical industry is not well understood. Hesitancy of pharmaceutical companies to reuse protein non-naïve NHPs may result in missed opportunities to fully utilize healthy NHPs in subsequent fit for purpose studies.
To understand the current thoughts and practices on reuse of protein non-naïve NHPs within the pharmaceutical industry, the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ Consortium) NHP Reuse Working Group (co-sponsored by the Translational ADME and 3Rs Translational and Predictive Sciences Leadership Groups) created a questionnaire to survey member companies, the results of which are presented within this paper. Further discussions within this working group revealed reuse enabling strategies and forward-looking approaches already in use in many pharmaceutical companies. These recommendations, if widely adopted throughout industry, could improve operational sustainability and achieve greater alignment with the principles of refinement, reduction, and replacement (3Rs) of ethical animal use in the discovery and development of new medicines, vaccines, medical devices, and health care products for humans and animals.
The preparation of this manuscript was developed with the support of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ, www.iqconsortium.org). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
Industry Survey on NHP Reuse
The survey questionnaire (see Appendix 1) on protein non-naïve NHP reuse was distributed to the IQ Consortium member pharmaceutical and biotechnology companies that participated in the NHP Reuse Working Group. Personnel completing the survey included principal investigators, facility managers, animal welfare compliance officers, scientists, and management. The survey questions addressed general information about protein non-naïve NHP reuse practices, the study types for which they are used, and any criteria used to determine animal reuse in subsequent studies.
The survey achieved a 100% response rate from all organizations contacted. While most (94%) survey respondents indicated that some form of protein non-naïve NHP reuse was permitted in their organizations, most reported that there were limitations around the practice for scientific reasons. Over half (60%) of the respondents disclosed that protein non-naïve NHP use was restricted to studies involving small molecules. Limiting the use of protein non-naïve NHPs to small molecule studies correlates with perceived potential risk associated with their use in subsequent large molecule studies. More than 84% of respondents identified concern about protein non-naïve NHPs being more susceptible to hypersensitivity responses upon subsequent exposure to therapeutic proteins and over 84% expressed concern that residual therapeutic effects, such as immunomodulation, would confound interpretation of subsequent studies. Variability in previous test article history (57%) and the availability and/or reliability of previous test article exposure history (67%), particularly with NHPs previously used by another pharmaceutical company (Sponsor) at a contract research organization (CRO), were other commonly cited concerns. One half (50%) of respondents indicated that these potentially confounding factors increase the perceived risk of experimental results being refused by regulators, and 57% of the respondents reported that reuse of protein non-naïve NHPs was limited to non-pivotal/non-GLP experiments, such as dose range-finding or investigational toxicology studies. Interestingly, only 8% of respondents indicated they had great concern about receiving explicit regulatory feedback that questioned the appropriateness of protein non-naïve NHP reuse.
Other operational and welfare-related factors that affect reuse decisions were evident in the survey results. Over 90% of respondents identified that extended post-study washout periods were necessary to ensure that long-lasting pharmacological effects or potentially interfering anti-drug antibodies (ADAs) would not affect a subsequent study involving proteins. Extended washout periods result in additional resource demands (e.g., cage space, veterinary care) that were a concern for 46% of the respondents. Utilization of NHPs in nonterminal experiments, regardless of test article modality, carries the additional challenge of managing stable cage mates, a concern for 58% of survey respondents. Effective and informed access to available NHPs, particularly within colonies managed by CROs, was considered a barrier to reuse efforts for 46% of the respondents, some of whom added that they had no direct control over how protein non-naïve NHPs would be utilized.
Practices that Enable Reuse
The most cited concern related to the use of protein non-naïve NHPs in subsequent studies of protein therapeutics was the perception of increased susceptibility to hypersensitivity responses (allergic or anaphylactic response or auto-immunity) mediated by induction of ADAs with potential formation of immune complexes. Moreover, long-term pharmacodynamic effects, which may persist after test article clearance, may not be detectable by typical pre-study assessments such as routine clinical pathology monitoring. According to the survey results, the most common practices employed to enable reuse were washout periods, routine for any type of nonterminal PK/PD work, and ADA screening assays to exclude animals with preexisting ADAs against prospective test articles on subsequent studies. A longer washout period may be necessary for therapeutic proteins with extended half-lives.9-11 The utility of ADA analysis as a prescreening tool has been described in the literature and in presentations.12,13 The screening of prospective protein non-naïve animals prior to initiation of a study is a feasible and practical approach that carries relatively little risk, but requires an assay that reliably detects ADAs cross-reactive with the therapeutic protein to be administered in the subsequent study to ensure effective prescreening. Many therapeutic proteins contain human immunoglobulin domains, which has allowed some organizations to successfully implement generic or universal ADA screening assays. 14 Other organizations reported that a modified test article-specific assay, which utilizes a labeled form of the specific therapeutic protein or antigen, has been used for prescreening. The timing of pre-study ADA testing sample collection is critical to ensure the adequacy of a washout period. Available PK, ADA, and PD data from previously conducted studies involving therapeutic proteins should be utilized to inform the timing of ADA sample collection for banking as well as individual animal selection for future studies. This is most feasible for companies performing studies internally, or with dedicated NHP colonies at CROs, when the prior test article exposure history of individual NHPs is known.
Other lesser used animal selection criteria cited in the survey responses included baseline evaluation of pharmacological endpoints, cytokine monitoring, enhanced clinical signs monitoring, use of in silico tools to predict likelihood of immunogenicity, and in vitro B-cell/T-cell recall assays, used to predict immunologic recall responses in the absence of preexisting ADAs.
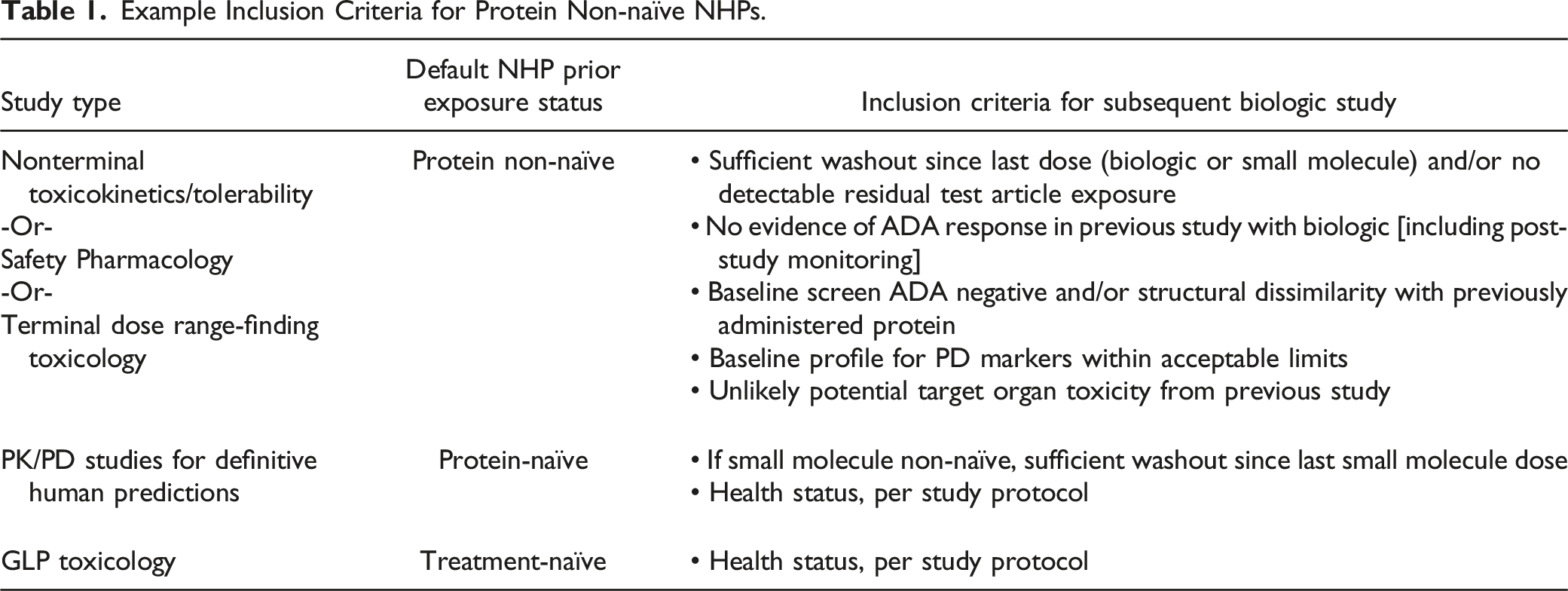
Following the online survey, further discussions in the NHP Reuse Working Group revealed that, rather than individual project teams, outsourcing groups often initiated conversations of NHP reuse opportunities, given their close working relationships with CROs and intimate knowledge of study design. Only one third of companies surveyed have developed formal inclusion/exclusion criteria or other guidelines to drive consistent, enterprise-wide decision-making around protein non-naïve NHP reuse in fit-for-purpose studies. Examples are presented below in Figure 1 and Table 1. Example decision tree for inclusion of protein non-naïve NHPs in subsequent studies with therapeutic proteins. Example Inclusion Criteria for Protein Non-naïve NHPs.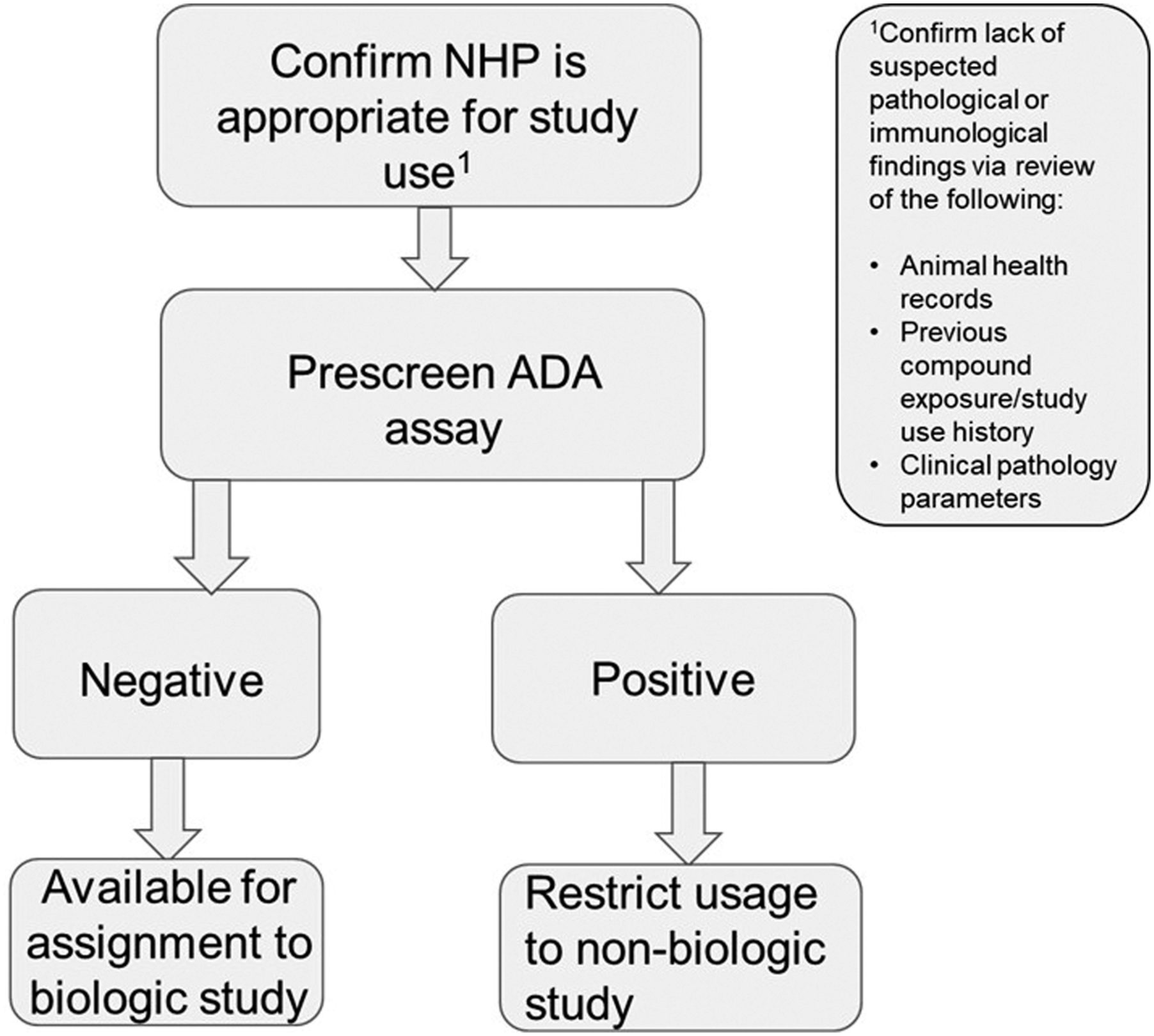
It was acknowledged during WG discussions that the recent FDA guidance that discourages the use of NHPs for general toxicology assessment of small molecules, while highly welcome from a 3Rs perspective, adds complexity to the strategy around appropriate protein non-naïve reuse. 4
Ethical Considerations and the Benefits of NHP Reuse
Harm-benefit analysis, as defined by the American Association for Laboratory Animal Science-Federation of European Laboratory Animal Science Associations (AALAS-FELASA) working group on harm-benefit analysis, is “a systemic process for assessing and comparing the harms and anticipated benefits in a particular animal study.” 15 This includes assurance of the comprehensive consideration of potential harms and benefits during the ethical review process. Preceding discussions have focused on reducing the scientific “harm” posed by unknown or confounding factors on results generated from reused animals. 16 Effective reuse practices can contribute to a reduction in the overall number of NHPs required for biomedical research and development, a commitment to the 3Rs that is shared across industry. Operationally, the benefits include reduced spend on NHP purchases and, in theory, improved scheduling efficiency through utilization of NHPs that have already passed the required quarantine period, been acclimated to the testing facility, and trained for study use. The relationship between transport, facility acclimation, and research animal stress has been well documented.17,18 Regarding harm-benefit analysis, there is an increased weight to the benefit side of the harm-benefit equation when an animal can be appropriately utilized more frequently, compared with an animal being held and not used.
From an ethical perspective, reuse requires consideration of cumulative harm and heightened awareness of the potential for development of an adverse health and welfare complication. As such, proactive health and welfare monitoring and treatment plans should be developed to prioritize animal care, and consideration should be given to the housing and enrichment provided (e.g., EU style housing or USDA housing). The benefits of maintaining socially stable groups of animals should not be overlooked. Some organizations report successful use of protein non-naïve monkeys as non-dosed companion animals on study to maintain socially compatible groups. Scientific management, the Institutional Animal Care and Use Committee (IACUC), individual project teams, and personnel directly responsible for study conduct and animal care should all be aware and supportive of the increased risk and mitigation strategies that provide benefit for the overall animal research operation.
Future Strategies
Increasing the reuse of NHPs within colonies across industry is challenging and requires the commitment and cooperation between pharmaceutical company Sponsors and CROs. Improved awareness of animal availability at CROs to prospective Sponsors will also be critical to advancing protein non-naïve NHP reuse efforts. Adequate health monitoring records and previous test article exposure history are needed to support informed decisions about the appropriateness of reuse of individual animals. To maximize the probability of success of a reuse program, Sponsors will ideally be willing to share general descriptions of test article characteristics such as modality and recommended washout duration, and perhaps high-level study outcomes (e.g., were post-dosing ADAs detected or inferred from PK data). Many elements of descriptive test article information are already collected during pre-study protocol review by ethical animal use committees and could be presented in a blinded manner to other prospective Sponsors before subsequent studies. However, robust data management practices are required to ensure the reliability and security of the data and the Sponsors’ respective proprietary information. A successful CRO protein non-naïve NHP reuse program will provide improved study timelines, reduced study costs, and promote utilization of facility-acclimated NHPs whose suitability for study use will have been demonstrated in previous studies. It should be noted that similar practices could be used within a Sponsor’s own NHP colony, with the exception that redaction of test article information would be unnecessary.
Practices that successfully enable protein non-naïve reuse should be broadly shared across industry. Even without formal internal requirement for tracking reuse within their organizations, many survey respondents indicated that reuse data are regularly reviewed, which suggests that there is interest in these metrics. Furthermore, 92% indicated that they are willing to share their reuse practices for protein non-naïve NHPs, signaling that this is broadly regarded as an important, value-added activity.
Conclusion
The survey results show that, while individual pharmaceutical and biotechnology companies have cautiously approached reuse of protein non-naïve monkeys to protect the scientific integrity of preclinical studies, there remains strong interest in identifying ways to appropriately increase their reuse. Improved accessibility of essential animal history data and sharing of best practices gained through cumulative industry experience present opportunities to appropriately increase the reuse of protein non-naïve NHPs, without compromising animal welfare nor scientific integrity. Widespread adoption of animal screening strategies and increased collaboration with CROs to support pre-study screening sample collection and maintenance of blinded historic treatment data that supports additional reuse are recommended next steps. Industry-wide alignment is needed to facilitate increased, responsible NHP reuse while maintaining rapid development of safe novel therapies for patients.
Footnotes
Author Contributions
Mattis, C., Bratcher, N., Burns, M., Carosino, C., de Zafra, C., Marcus Fancher, R., Georgi, K., Graff, C., Hukkanen, R.R., Johnson, C., Lao, Y., Lange, A., Lepherd, M., Maguire, S., Malisauskas, M., Manuel, M., Miranda, S., Reed, L., Santos, R., Sayers, B., Shaw, D., and Shuster, D. contributed to conception and design and contributed to acquisition, analysis, and interpretation; Lee, D. contributed to acquisition, analysis, and interpretation. All authors drafted manuscript, critically revised manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1: Protein Non-Naïve Nonhuman Primate Reuse Survey
1. Does your organization allow reuse of protein non-naïve NHPs for any preclinical studies? Yes/No
2. If Yes, does your organization apply any of the following restrictions for their reuse: Select all that apply
• Small molecules studies only
• Only for non-pivotal/non-GLP studies (e.g., DRF/investigational tox)
• Screening/pharmacokinetic studies
• Dependent on therapeutic area
• Route of administration/site of action (e.g., local as intraocular vs oral vs parenteral)
• Formulation optimization
• Exploratory biomarker qualification
• Assignment to vehicle control group for subsequent studies
• Other write-in
3. Describe your self-identified restrictions response above
4. Are the following alternative uses permissible for protein non-naïve NHPs? Select all that apply
• Exploratory studies (e.g., preclinical model development)
• Buddy animals (i.e., non-dosed cage mates) to enable group housing
• Tissue harvest for in vitro assay method development
• In-life procedure training
• Necropsy training
• Other write-in
5. Does your organization employ any of the following mitigation strategies to enable reuse of protein non-naïve NHPs? Select all that apply
• Baseline and/or periodic screening for anti-drug antibodies
• Pharmacological sensitivity screening
• Time-bound washout period
• Baseline and/or post-study clinical pathology monitoring
• Enhances clinical signs monitoring
• Consideration of molecular structures from study to study (i.e., similarities that would make an immunological recall response a likely outcome)
• In silico predictivity tools
• In vitro B-cell/T-cell recall assay prior to new experiment
• Limit on total number of additional experiments performed using the same monkeys
• Other write-in
6. To what extent do factors influence decisions on whether the reuse of protein non-naïve NHPs in your organization (e.g., non-naïve status, extended washout period, regulatory feedback, previous exposure history animal health, housing resources, etc.)
7. Roles/groups involved with decisions around the reuse of protein non-naïve NHPs
• Senior management
• Project representatives
• Operations staff
• Quality/Regulatory
• Veterinary Staff
• IACUC
• Others
8. Are practices being tracked within your organization? (Yes/No) and if so who does this and what are their roles?
9. Are data regularly reviewed within your organization?
10. Is there an expectation in your organization to investigate reasons for not reusing monkeys?
11. Is there an established process (e.g., decision tree)?
12. Would you be willing to share your internal practices on NHP reuse with IQ member companies?
