Abstract
Respiratory syncytial virus (RSV) is a leading cause of acute lower respiratory tract infections (LRTI) in infants, and toddlers and vaccines are not yet available. A pediatric RSV vaccine (ChAd155-RSV) is being developed to protect infants against RSV disease. The ChAd155-RSV vaccine consists of a recombinant replication-deficient chimpanzee-derived adenovirus (ChAd) group C vector engineered to express the RSV antigens F, N, and M2-1. The local and systemic effects of three bi-weekly intramuscular injections of the ChAd155-RSV vaccine was tested in a repeated-dose toxicity study in rabbits. After three intramuscular doses, the ChAd155-RSV vaccine was considered well-tolerated. Changes due to the vaccine-elicited inflammatory reaction/immune response were observed along with transient decreases in platelet count without physiological consequences, already reported for other adenovirus-based vaccines. In addition, the biodistribution and shedding of ChAd155-RSV were also characterized in two studies in rats. The distribution and persistence of the ChAd155-RSV vaccine candidate was consistent with other similar adenovector-based vaccines, with quantifiable levels of ChAd155-RSV observed at the injection site (muscle) and the draining lymph nodes up to 69 days post administration. The shedding results demonstrated that ChAd155-RSV was generally not detectable in any secretions or excreta samples. In conclusion, the ChAd155-RSV vaccine was well-tolerated locally and systemically.
Introduction
Human respiratory syncytial virus (RSV) is an RNA virus of the Paramyxoviridae family. 1 RSV is a highly contagious pathogen that can produce respiratory tract infections in people of all ages. In temperate climates throughout the world, RSV causes fall-winter epidemics,2,3 whereas viral activity in sub-tropical and tropical regions is more endemic. 1
RSV is one of the major causes of lower respiratory tract infections in very young children. 4 RSV disease spectrum includes rhinitis, otitis, pneumonia, and bronchiolitis, the two latter being the main causes of RSV-associated morbidity and mortality.5,6 During the first year of life, 50-70% of infants are infected with RSV and most children have had an RSV infection by their second birthday.7,8 In 2015, it was estimated that there were 33.1 million episodes of RSV-associated lower respiratory tract infections (LRTI) which resulted in 3.2 million hospital admissions and 27 300 in-hospital deaths globally. Incidence of RSV is higher in children born prematurely or those with cardio-pulmonary diseases.9,10 These populations are considered particularly at risk for severe RSV infections. In the elderly, RSV is increasingly recognized as an important cause of morbidity and mortality, provoking influenza-like illnesses.11-15
There is currently no licensed vaccine to protect against RSV despite multiple attempts and persistent efforts during the last 50 years.16-18 A critical hurdle facing the pediatric strategy was the vaccine-induced enhanced respiratory disease (ERD) which was seen after natural infection in RSV-naïve infants previously vaccinated with a formaldehyde-inactivated whole virus vaccine (FI-RSV) in the late 1960s.19,20 ERD was believed to be due to the formalin inactivation step, suspected of favoring the production of low quality, non-neutralizing antibodies in RSV-naïve infants. 21 In addition, it is hypothesized that ERD may have been linked to the induction of a Th2-skewed CD4+ T-cell immune responses. 22 This legacy and liability has increased the importance of robust and thorough preclinical and clinical RSV vaccine safety evaluation, especially when vaccinating a pediatric population.
Adenovirus-based vaccines are not novel and are being developed or have been used to vaccinate people against numerous pathogens including SARS-CoV2. 23 Adenoviral vectors are potent inducers of CD8+ T cells producing IFN-γ and antibodies against expressed antigens.24,25 GSK is developing a pediatric vaccine using an adenovirus-based vector (ChAd155-RSV) for active immunization of infants for the prevention of RSV-associated LRTI. The ChAd155-RSV vaccine consists of a recombinant replication-deficient simian (chimpanzee-derived) adenovirus group C vector, ChAd155, engineered to express the RSV antigens for the structural proteins, F (fusion), N (nucleocapsid), and M2-1 (transcription anti-termination). The ChAd155 vector belongs to the subgroup C of human adenoviruses as based on the hexon protein sequence. The ChAd155 vector is derived from the wild-type chimpanzee Ad type 155 genome and was isolated from a healthy young chimpanzee using standard procedures. 26 ChAd155 genome sequence analysis shows very high similarity with other subgroup C adenoviruses, suggesting that it can use the same receptor (Coxsackievirus and adenovirus receptor, CAR) to enter the host cell. Several adenovectors developed from adenoviruses belonging to subgroup C (human Ad5, Ad6, chimpanzee-derived ChAd3 27 and the Pan Paniscus-derived PanAd3 28 ) have been tested in clinical trials and have all shown a very good safety profile.24,29,30 The RSV F protein is a major surface glycoprotein, highly conserved among RSV sub-groups and strains, that mediates viral fusion to target cells.31-33 The F protein, particularly in its prefusion conformation, is a target for neutralizing antibodies, including the prophylactic RSV-neutralizing monoclonal antibody palivizumab. 34 The nucleoprotein N is an internal viral protein and is highly conserved among RSV strains and known to display T-cell epitopes. 35 The primary function of the N protein is to encapsulate the virus genome for the purposes of RNA transcription, replication, and packaging and protects the viral genome from ribonucleases. 36 M2-1 is another well-conserved internal viral protein known to be a source of T-cell epitopes. 37 The M2-1 protein is a transcription anti-termination factor that is important for the efficient synthesis of full-length messenger RNAs and for the synthesis of polycistronic readthrough mRNAs. 38 The purpose of including the N and M2-1 antigens in the vaccine construct was to serve as a source of T-cell epitopes for the induction of cell-mediated immunity (CMI).
To support the clinical development of the ChAd155-RSV vaccine, several non-clinical toxicity studies were performed. This article describes the non-clinical findings on three of these studies: a repeated-dose toxicity study performed in rabbits and two studies in rats, a biodistribution study and a shedding study. The objective of the first study was to evaluate the potential local and systemic toxic effects of the ChAd155-RSV vaccine, following three intramuscular administrations at 2-week intervals in rabbits. The objective of the second study was to evaluate by qPCR the biodistribution of ChAd155-RSV in Sprague-Dawley rats following a single intramuscular administration of ChAd155-RSV candidate vaccine and to assess its potential toxicity. The objective of the third study was to evaluate the shedding of the ChAd155-RSV vaccine by qPCR in secretions and excreta samples (feces, urine, throat swabs, saliva, and nasal fluid), following a single intramuscular administration in Sprague-Dawley rats. In addition, a non-GLP infectivity assay was conducted on the positive samples to evaluate the presence of infectious vector particles.
Materials and Methods
Regulatory Guidelines and Data Quality
Each study was conducted in a laboratory that complied with Good Laboratory Practice (GLP) standards, with the exception of the anti-F ELISA. The repeated-dose study was carried out in compliance with the principles of the note for guidance on preclinical pharmacological and toxicological testing of vaccine from the Committee for proprietary medicinal products, 39 the WHO guidelines on non-clinical evaluation of vaccines, 40 the Guidelines on the non-clinical evaluation of vaccine adjuvants and adjuvanted vaccines, 41 the Note for guidance on non-clinical local tolerance testing of medicinal products, 42 and the Guideline on repeated-dose toxicity from the Committee for Human Medicinal Products. 43
The rat biodistribution study was performed in compliance with the Guideline on repeated-dose toxicity from the Committee for Human Medicinal Products 43 and other relevant guidelines.40,41,44
The shedding study in rat was performed in compliance with the ICH M3(R2) Guidance on non-clinical safety studies for the conduct of human clinical trials and marketing authorization for pharmaceuticals, 45 the WHO guidelines on non-clinical evaluation of vaccines, 40 the WHO guidelines on the non-clinical evaluation of vaccine adjuvants and adjuvanted vaccines, 41 the EMEA General principles to address virus and vector shedding, 46 and the FDA Guidance for industry—Design and analysis of shedding studies for virus or bacteria-based gene therapy and oncolytic products. 47
Animals and Husbandry
Specific pathogen free-bred male and female New Zealand White albino rabbits were obtained from Centre Lago (Vonnas, France). Male and female Sprague-Dawley rats were obtained from Janvier (Le Genest Saint Isle, France) for the biodistribution study or from Charles River Canada Inc. (St-Constant, Québec, Canada) for the shedding study. The animals were allowed to acclimate prior to the first day of dosing. They were kept individually in polycarbonate cages, except for the shedding study where they were group-housed when not involved in measurement or activities. For urine collection, animals were placed individually in metabolic cages, under non-fasting conditions. The animal rooms were equipped with filtered air (10 or greater air change/hour) and kept at a mean temperature range of 15-21°C (rabbits) or 18°C to 24°C (rats) and relative humidity in the range 30-70%. The lighting followed a 12-hour light/12-hour dark cycle. Both rats and rabbits were provided ad libitum a certified laboratory diet and had free access to domestic-quality drinking water. The animals were given various cage-enrichment devices and/or dietary enrichment.
Animal Welfare
For the repeated-dose and biodistribution studies, animal husbandry and experiments were ethically reviewed by the CiToxLAB France Ethical Committee and carried out in accordance with the European Directive 2010/63/EU. The shedding study was approved by the Institutional Animal Care and Use Committee of Charles River Laval (Québec, Canada).
Control and Test Items
ChAd155-RSV was provided frozen at −70°C or −80°C. The storage buffer was composed as follows: 10 mM TRIS, 10 mM histidine, 75 mM NaCl, 5% sucrose, 1 mM MgCl2, 0.02% (w/v) polysorbate-80, 0.1 mM EDTA, 0.5% ethanol (w/v), pH 7.4. ChAd155-RSV concentration in each vial was approximately 1 × 1011 viral particles (Vp)/mL. ChAd155-RSV vaccine was thawed, kept at room temperature, and used within one hour after thawing. The control item was a ready-to-use formulation of 0.9% saline, non-pyrogenic, and sterile.
Study Designs
Repeated-dose toxicity study in rabbits
Rabbits were randomly allocated in four groups of 20 animals each, composed of 10 males and 10 females. On the first day of dosing, the rabbits were 3 to 4 month-old and weighed 2825-3425 g (males) or 3030-3655 g (females). The animals were given 0.5 mL of the ChAd155-RSV vaccine by intramuscular injection in a thigh muscle on Days 1, 15, and 29, corresponding to a dose of 5 × 1010 Vp for each injection. The study schedule was defined in order to cover the number of injections to be given in humans plus one extra. Each injection was made at a different site, on Day 1 in the posterior part of the right hind leg, on Day 15 in the posterior part of the left hind leg, and the last injection on Day 29 in the anterior part of the right hind leg. The control group was administered saline in the same conditions as the item-treated group. The first five rabbits/sex/group were euthanized at the end of the treatment period on Day 32 (3 days after the last treatment). The remaining rabbits were maintained on study for an additional four weeks, then euthanized at the end of a 28-day treatment-free period (Day 57).
Toxicity was evaluated based on mortality, visible signs of ill health or behavioral changes, body temperature, body weight, food consumption, ophthalmology, clinical pathology, and post-mortem examination. On each dosing occasion, local reactions at injection sites were recorded pre-dose and 3, 24, 48, and 72 hours after injection; the injection site was scored/graded using a modified Draize scale, based on the extent of erythema and edema. Rectal temperature was recorded in each animal once before the treatment period, and then for each injection, just before and 3, 8, 24, and 48 hours following injection. Body weights and qualitative food consumption were recorded at least once during the pre-dose phase, prior to dosing and twice a week over 3- to 4-day intervals after the first dosing through the remainder of the study. Ophthalmic examinations were conducted by a board-certified ophthalmologist during the pre-dose phase, and during the recovery period. Mydriasis was induced in each eye to allow examination by indirect ophthalmoscopy, and a slit-lamp biomicroscope was used to investigate the anterior segment of the lens.
Laboratory evaluations (hematology, coagulation, clinical chemistry, and antibody levels) were performed once prior to the start of treatment and again on Days 32 and 57 (antibody levels), and Days 2, 8, 25, 30, 36, and 57 (hematology, coagulation, and clinical chemistry). Blood was collected from the ear vein in non-coated serum tubes (for antibody levels), tubes with EDTA (for hematology) or citrate (for coagulation), or in heparinized tubes (for clinical chemistry). Hematology parameters included erythrocyte counts, hemoglobin level, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, absolute reticulocyte count, thrombocyte count, leukocyte count, and absolute neutrophil, lymphocyte, monocyte, eosinophil, basophil, and large unstained cell counts. Coagulation endpoints included prothrombin time, activated partial prothrombin time (APTT), and fibrinogen. Clinical chemistry measurements included alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, creatine kinase, gamma glutamyl transferase, and lactate dehydrogenase activity, as well as glucose, total protein, albumin (and ratio albumin to globulin), urea, creatinine, bilirubin, cholesterol, triglycerides, phospholipids, calcium, sodium, potassium, chloride, inorganic phosphorus, and C-reactive protein (CRP) concentration. Vaccine take was evaluated by measuring the concentrations of anti-F IgG using a specific ELISA.
At study termination on Day 32 (five animals/sex/group) and on Day 57 (all remaining rabbits) the animals were euthanized, necropsy was performed, organ weights were recorded, and gross histopathologic examination was conducted. Tissues were weighed and preserved in phosphate-buffered neutral 10% formaldehyde. All tissues submitted to histopathological evaluation were embedded in paraffin wax, sectioned at 5 µm and stained with hematoxylin and eosin.
Biodistribution study in rats
Study design
Rats in the treatment group were randomly allocated to four sub-groups (one for each time point) of 10 animals, each composed of five males and five females. On the day of injection, the animals were approximately 7-week-old. These animals received one intramuscular injection (100 µL) of the test item ChAd155-RSV (1 × 1010 Vp/animal) in the right quadriceps. This represents 1/5th of a human dose. Treated animals were necropsied 24 hours or 7, 28, or 69 days after administration (Days 2, 8, 29, and 70, respectively). In the control group, four males and four females received an injection of saline solution, and one animal/sex was necropsied at the same timepoint as in the treated group.
The animals were checked daily for mortality and clinical signs of illness. The body weight of each animal was recorded before treatment, on the first day of treatment and then at least once a week. Food consumption was recorded at least once a week and was calculated per animal and per day based on group average.
On the day of sampling, animals were euthanized and a full macroscopic post-mortem examination was performed. Designated organs were weighed, and selected tissue specimens were preserved for ChAd155-RSV tissue distribution analysis by qPCR, as described below.
Evaluation of biodistribution by qPCR
Blood was collected before necropsy from the abdominal aorta (under deep anesthesia) into potassium EDTA tubes. Blood was aliquoted, snap frozen in liquid nitrogen, and stored at −20°C until DNA extraction.
Tissue samples were collected in the following order: brain, lung, liver, spleen, kidneys, heart, gonads, lymph nodes (popliteal, inguinal, and iliac), and muscle at the injection site. Samples were rinsed with phosphate-buffered saline with calcium and magnesium. Adipose tissue was carefully removed from the organs. For large organs, three samples of approximately 27 mm³ were taken. Small organs were sampled as a whole. After weighing, tissue samples were snap frozen in liquid nitrogen and stored at −20°C until DNA extraction.
DNA was extracted from blood using the Blood Quick Pure kit (Macherey Nagel). DNA was extracted from tissue samples using the NucleoSpin tissue kit (Macherey Nagel), according to the manufacturer’s instructions. Briefly, tissue sample was first incubated in a proteinase K solution before DNA extraction. Chaotropic salts and ethanol were added to the lysate to obtain appropriate conditions for DNA binding to the silica membrane. After washing, DNA was eluted under low ionic strength conditions in a slightly alkaline elution buffer. Tissues from control animals were extracted first.
Extracted DNA was used as template for PCR amplification with a validated qPCR test carried out on an ABI 7900 HT Fast Real-Time PCR system (Life Technologies). Primers were designed that are specific for the detection of ChAd155-RSV (forward primer: 5’- CATCTACGTATTAGTCATCGCTATTACCA-3’; reverse primer: 5’- GACTTGGAAATCCCCGTGAGT -3’, TaqMan probe: 6FAM-5’- ACATCAATGGGCGTGGATAGCGGTT -3’-TAMRA). These sequences allow the amplification of the CMV promoter region of the vector DNA.
DNA (0.4 µg, or 0.2 µg for lymph nodes and blood) in elution buffer was amplified in a mixture containing TaqMan Universal master mix, DNase/RNase-free water, both primers, and the TaqMan probe. Amplification was detected in real-time over 40 cycles (including an elongation step at 60°C) by following the evolution of the fluorescent signal generated by the degradation of the probe. Samples were analyzed in duplicate, in parallel with a calibration curve performed with known amounts of ChAd155-RSV. The Ct values were plotted against the logarithm of the Vp numbers of calibration points, and the resulting curve was linearized. For all samples, test item Vp numbers were calculated by interpolation from the calibration curve and adjusted by a dilution factor to express the results in Vp number/μg DNA and further in Vp number/g of tissue. The lower limit of quantification was 100 Vp/well (corresponding to 250 Vp/µg DNA), and the limit of detection was 50 Vp/well (corresponding to 125 Vp/µg DNA).
DNA extracted from biological samples may contain PCR inhibitors, leading to artificial underestimation of the results. Thus, PCR inhibition was verified in all tested samples by spiking a known quantity of genomic DNA from the ANTP gene from Drosophila in the samples and measuring this DNA by qPCR. Inhibition was revealed by discrepancy between expected and measured amounts of this exogenous DNA.
Shedding study in rats
Rats were randomly allocated to two groups: the treated group, composed of five males and five females, and the control group comprising one male and one female. On the day of injection, the animals were approximately 10-weeks old. The vaccinated animals received one intramuscular injection (125 µL) of the test item ChAd155-RSV (1 × 1010 Vp/animal) into the posterior part of the thigh muscle of the right hind limb. In the control group, the animals received an injection of saline solution. After injection, the animals were checked twice daily for mortality and clinical signs of illness. The body weight of each animal was recorded before assignment, one day before treatment and then twice weekly.
Fecal samples, nasal fluid, throat swabs, and saliva were collected before injection and 6 hours, 24 hours, 3 days, 7 days, 14 days, 28 days, 41 days, and 69 days post injection (corresponding to Days 1, 4, 8, 15, 29, 42, and 70). On Day 70, iliac lymph nodes were also collected, and the right iliac lymph node was kept for qPCR analysis. Secretions and excreta samples were collected with special attention to minimize cross contamination (i.e., gloves changed or sprayed with 10% bleach between each sample collection and each animal).
Urine collection was performed pre-dose over a 12-hour period and over time intervals ±15 minutes post dose, 0 to 6 hours post dose (for timepoint 6 hours post dose), 6 to 24 hours post dose (for timepoint 24 hours post dose), and over a 12 hour period on subsequent collection days (4, 8, 15, 29, 42, and 70), except during designated activities. Urine was collected separately in containers kept on wet ice. Fecal samples were collected from all animals by restraining the animal above a sterile pad until a fecal pellet fell on the pad. Manual massage on the abdomen of the animal was performed when deemed necessary. Nasal fluid was collected using a calibrated micropipette filled with 50 µL of sterile NaCl 0.9%/nostril injected once in each nostril of the rats positioned face down and was absorbed immediately by sterile dry cotton-tipped swabs. The saliva sample was collected using a small cotton swab placed into the oral cavity. The cotton swab was wiped all over the oral cavity in order to absorb the maximum volume of saliva and was kept in the mouth for 30 to 60 seconds. Throat swabs were collected using a small cotton swab, previously soaked in sterile NaCl 0.9%, and gently inserted into the throat of the animal making sure the cotton swab touches different areas in the throat. The order of collections was as follows: fecal sample, nasal fluid, saliva, and throat swabs. All samples were separated in two parts. One part was transferred into a sterile DNase/RNase-free cryotube, snap frozen in liquid nitrogen, and placed on dry ice pending storage in a freezer set to −80°C until qPCR analysis. The second part of the sample was transferred in the medium in a Remel MicroTest M4RT tube, snap frozen in liquid nitrogen, and placed on dry ice pending storage in a freezer set to −80°C for infectivity assay analysis.
Infectivity assay
For the infectivity assay, 2.5 × 105 HEK-293 cells were seeded onto poly-L-lysine-coated 24-well tissue culture plates for the immuno-staining procedure, and 1 × 106 HEK-293 cells were seeded onto poly-L-lysine-coated 35 mm dishes for the plaque assay. In addition, 2.5 × 105 A-549 cells were seeded onto uncoated 24-well tissue culture plates for immuno-staining procedure and 1 × 106 cells onto uncoated 6-well plates for plaque assay. Cells were incubated for 60 minutes in an incubator at 37°C with 5% CO2. Following incubation, 100 μL of the inoculum was added per well in the 24-well plates or 500 μL of the inoculum was added in 35-mm dishes or in each well of the 6-well plates at various concentrations (from 4.0 × 108 to 1.0 × 101 vp/mL). When using excreta/secreta, fungizone was added to the cell culture medium. Then, samples were filtered using 0.8-μM and 0.22-μM filters before inoculation. Cells were monitored for a cytopathic effect for 5 to 10 days in 35 mm dishes or 6-well plates for plaque assay and were fixed and stained 2 to 5 days after the incubation for immunochromatographic assay (ICA) and indirect immunofluorescence assay (IFA) procedures.
For IFA or ICA, the cells were fixed for 10-15 minutes with 4% paraformaldehyde and then blocked with blocking buffer (1% bovine serum albumin in phosphate-buffered saline). The primary antibody (anti-adenovirus B025/AD51, diluted 1/1000) was added to each well and incubated for 60 minutes at room temperature with shaking. After the incubation, the cells were washed with PBS three times. The secondary antibody anti-mouse-HRP was used at a dilution of 1:20 for ICA, and the anti-mouse-Alexa Fluor 488 was used at a dilution 1:800 for IFA and both were incubated for 60 minutes at room temperature with shaking. After the incubation, the secondary antibody solution was discarded and cells were washed with PBS three times. For the ICA method, a 1X DAB working solution was then added and incubated for 10-15 minutes. Cellular monolayers were examined using an appropriate microscope for positively infected cells.
qPCR analysis
DNA was extracted from lymph nodes using the DNeasy blood and tissue kit (Qiagen), from urine and fecal samples by using RTP DNA/RNA Mini Kit (Invitek Molecular), and by using Nucleospin DNA forensic kit (Macherey Nagel) for nasal fluid, throat swabs, and saliva, all according to the manufacturers’ instructions. DNA concentration and purity measurements were made for the lymph node samples only, using the NanoDrop Lite spectrophotometer (Thermo Fischer Scientific). This procedure was not applied to feces, urine, nasal fluid, saliva, and throat swabs, since the DNA concentration was considered negligible in these samples or the procedure considered irrelevant due to the presence of RNA carrier (like in urine and feces samples, for example).
Extracted DNA was used as template for PCR amplification with a validated qPCR test carried out on a QuantStudio 5 Real-Time PCR System (Applied Biosystems). Primers were designed that are specific for the detection of human cytomegalovirus promoter sequence, which is part of ChAd155-RSV (forward primer: 5′- CATCTACGTATTAGTCATCGCTATTACCA -3′; reverse primer: 5′- GACTTGGAAATCCCCGTGAGT -3′, TaqMan probe: 6FAM 5′- ACATCAATG-ZEN-GGCGTGGATAGCGGTT-IABKFQ -3′). These sequences allow the amplification of the CMV promoter region of the vector DNA.
DNA (1 µg) was amplified in a mixture containing TaqMan Universal master mix, DNase/RNase-free water, both primers, and the TaqMan probe. Amplification was detected in real-time over 40 cycles (consecutive to an elongation step at 50°C) by following the evolution of the fluorescent signal generated by the degradation of the probe. The QuantStudio Design and Analysis software (Applied Biosystems, version 1.4.2) was used for data acquisition and/or data analysis. Samples were analyzed in duplicate, in parallel with a calibration curve performed with known amounts of ChAd155-RSV, towards which the sample results were evaluated. The Ct values were plotted against the logarithm of the Vp numbers of calibration points and the resulting curve was linearized. For all samples, test item Vp numbers were calculated by interpolation from the calibration curve and adjusted by a dilution factor to express the results in Vp number/μg DNA. The lower limit of quantification and the lower limit of detection were both 50 Vp/well.
Anti-F ELISA
Briefly, the method involved overnight coating of ELISA plates with F protein. On the next day, after washing and blocking, diluted standard/control sera and samples were added. After an incubation step, the plates were washed and any bound antibodies were detected using peroxidase-labeled goat anti-rabbit antibody. Then absorbance was read at 450 nm using an ELISA plate reader after addition of tetramethylbenzidine. The concentration of anti-F IgG present in the unknown samples was calculated by interpolating on a standard curve the optical density of sample dilutions falling within the linear range of the standard curve, adjusting for sample dilution, and was expressed in µg/mL.
Statistical Analyses
For the comparison of body weight, body temperature, hematology, coagulation, or clinical chemistry values between treated and control groups, the hypotheses of normality and homogeneity of variance were first evaluated by a Kolmogorov-Lilliefors and a Fisher test, respectively. Comparisons were made either with a Dunnett’s test or a Mann-Whitney test, depending on the results of the normality and homogeneity tests. The statistical analysis of organ weight data followed a different sequence. Normality and homogeneity of variance were evaluated with a Kolmogorov test and a Bartlett test. Depending on the results obtained with the normality and/or homogeneity tests, either a t-test (parametric) or a Wilcoxon test (non-parametric evaluation) was used.
The data obtained in the biodistribution and shedding studies were descriptive and not analyzed statistically.
Results
Repeated-Dose Toxicity Study in Rabbits
Serology
ELISA results confirmed that animals receiving the ChAd155-RSV vaccine had mounted an immune response, as demonstrated by the presence of anti-RSV F IgG antibodies (Figure 1). All tested animals in the ChAd155-RSV-treated groups had seroconverted on Day 32 or Day 57, after having received three intramuscular administrations of test item. In contrast, no anti-RSV F IgG antibodies were detected in the control groups at any timepoint. Repeated-dose toxicity study. Rabbit serum was collected before immunization and 3 days (Day 32) and 4 weeks (Day 57) after the third immunization. Anti-F antibody titers in rabbit serum were measured by enzyme-linked immunosorbent assay (ELISA). Each symbol represents an animal. Arrows indicate immunizations.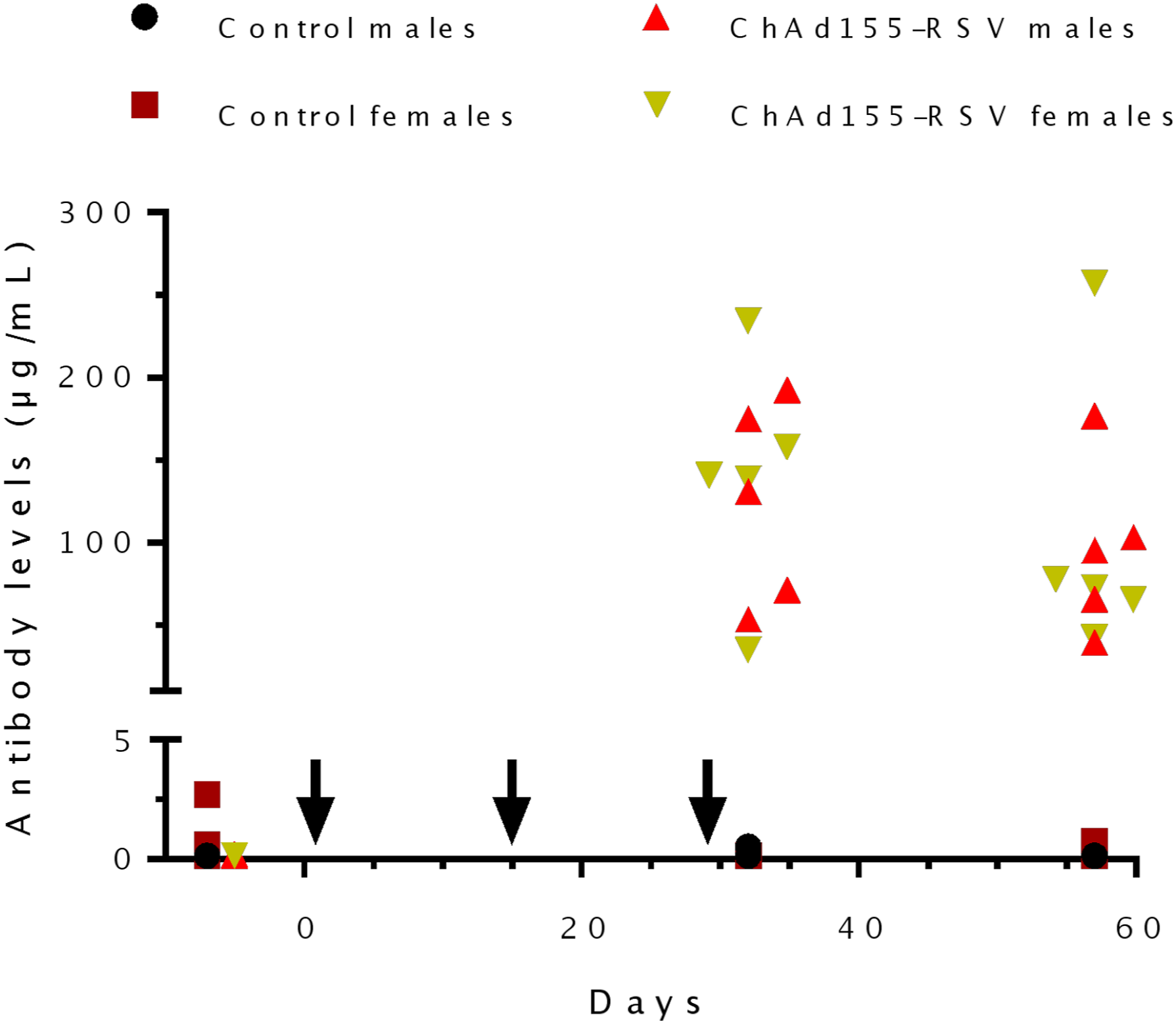
Observations and Measurements
No treatment-related clinical signs were observed, except scabs that were observed at a higher incidence in item-treated animals (3/10 test item-treated males and 4/10 test item-treated females vs 2/10 control females). Hematomas were noted at the same incidence at injection sites in both groups and were considered related to the administration procedure.
No treatment-related effects on ophthalmology, body weight, or food consumption were observed. Increases in body temperatures (≥40°C) were noted sporadically in 1 or 2/5 males and 2/5 females at 8 and/or 24 hours after the first and/or second injection, and in one male and one female 48 hours after the first or third administration of ChAd155-RSV vaccine. Statistically significant higher mean body temperature values were measured 24 hours after the first injection of the test item in males and females, compared with controls (p < 0.01). However, similar mean body temperature values were measured in controls at other time-points or before dosing in treated animals. Therefore, the effect of treatment with the test item on this parameter is uncertain.
Repeated-dose toxicity rabbit study. The animals were immunized on Days 1, 15 and 29. Clinical pathology most affected by treatment.
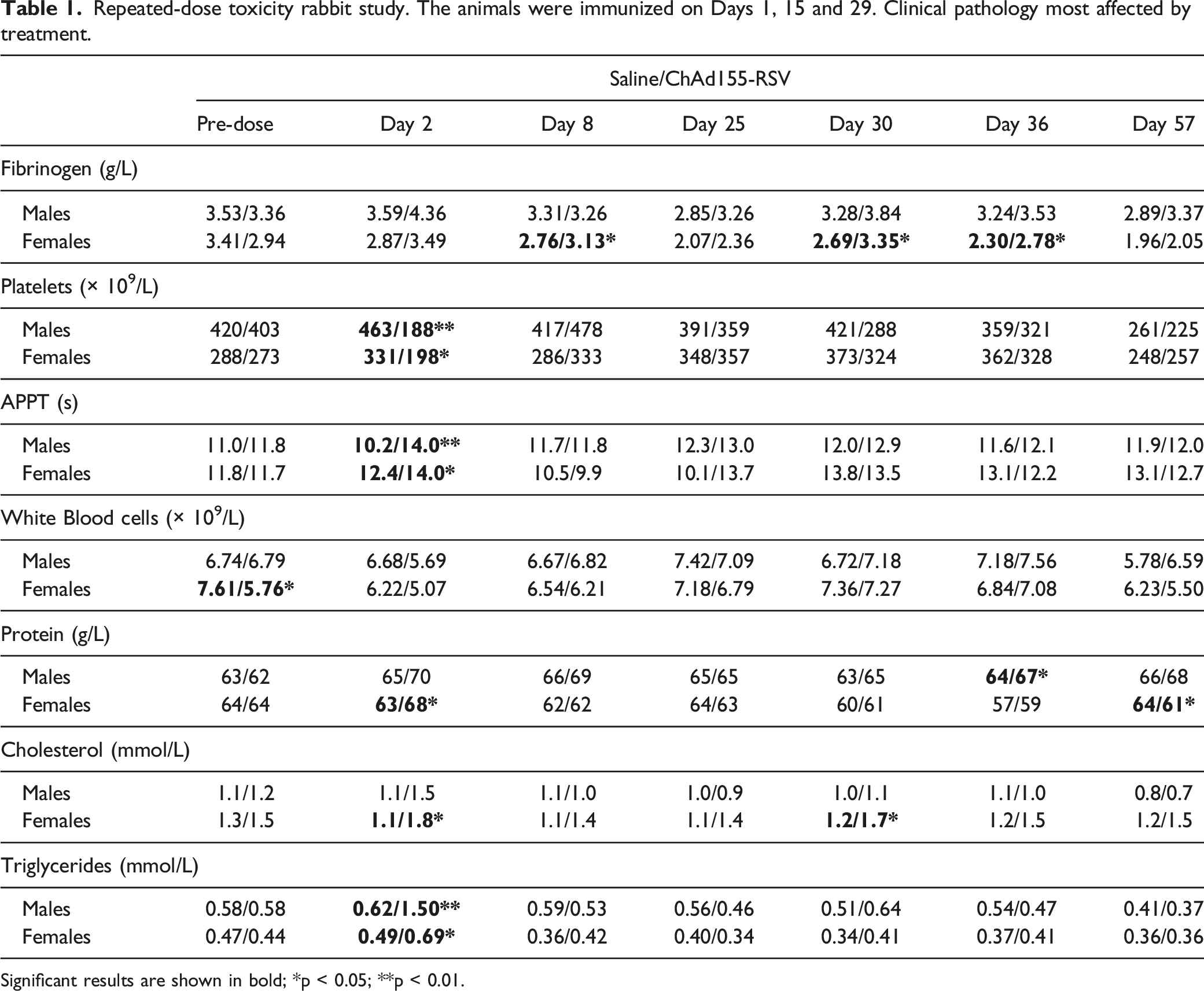
Significant results are shown in bold; *p < 0.05; **p < 0.01.
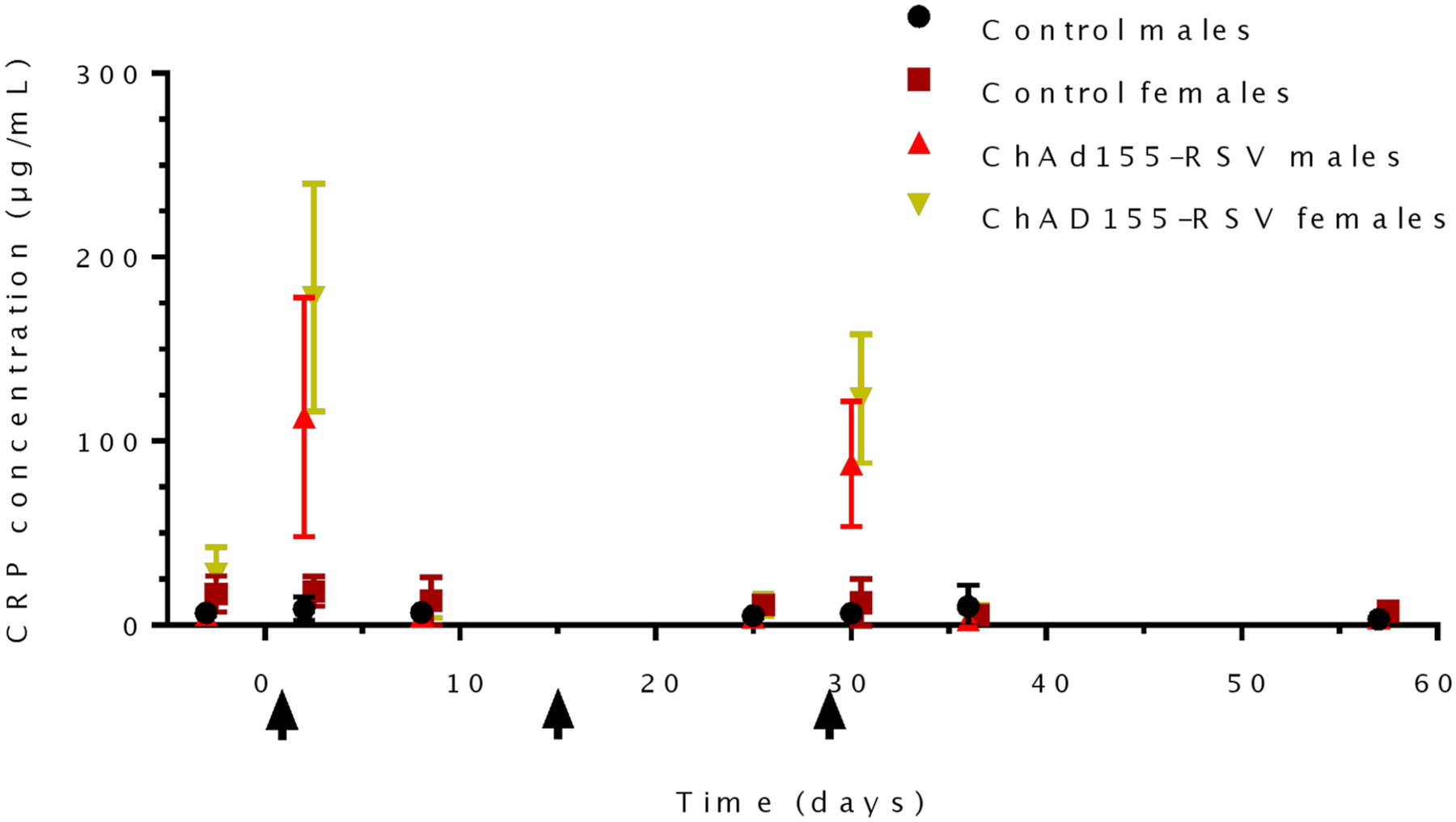
Repeated-dose toxicity study. Rabbit serum was collected before immunization, 1 and 7 days after the first immunization (Days 2 and 8), 4 days before the third immunization (Day 25), 1 and 7 days after the third immunization (Days 30 and 36), and 4 weeks after the third immunization (Day 57). C-reactive protein (CRP) concentration in rabbit serum was measured by enzyme-linked immunosorbent assay (ELISA). Values are means and SD. Arrows indicate immunizations.
A transient fall in platelet counts was observed following vaccine injection on Days 2 and 30. This decrease in platelet counts was present in both male and female rabbits, and mean platelet counts on Day 2 were lower than pre-test values (−53% in males and −27% in females) and lower than concurrent control values (−59% in males; p < 0.01; and −40% in females; p < 0.05). A smaller decrease was noted in males after the third vaccine administration (−29% vs pre-test and −32% vs control; not significant). The effects on platelet counts were reversible, as shown by values on Day 8 and Day 36. On completion of the treatment-free period, on Day 57, platelet counts were similar in both control and vaccine-treated groups and there was no evidence of a persistent effect of vaccine treatment.
Increased triglyceride levels were observed on Day 2 in vaccine-treated animals (+159% in males and +57% in females, vs pre-test; +142% in males and +41% in females vs controls) and to a lesser extent on Day 30 in females only (+21% vs control). This was accompanied by higher cholesterol levels on Day 2 (+25% in males and +20% in females, vs pre-test; +36% in males and +64% in females vs controls) and Day 30 in females only (+13% vs pre-test and +42% vs controls).
Repeated-dose toxicity rabbit study. The animals were immunized on Days 1, 15 and 29. Differences in absolute and relative organs weights on Day 32 (expressed in %).
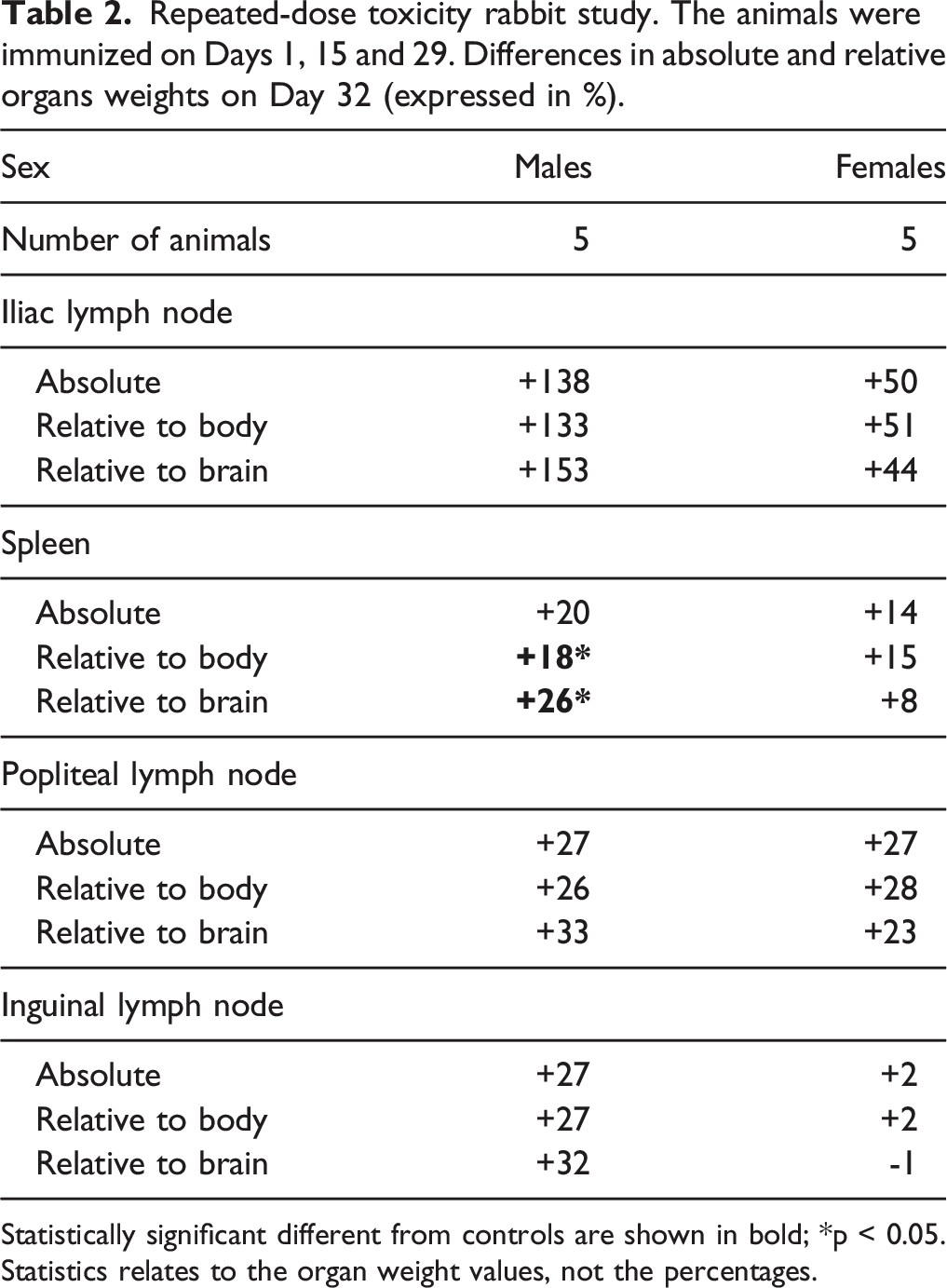
Statistically significant different from controls are shown in bold; *p < 0.05.
Statistics relates to the organ weight values, not the percentages.
The absolute and relative (to body and brain weights) spleen weights were higher, reaching statistical significance for both relative weights in males (p < 0.05; compared with controls). This correlated histologically with the development of germinal centers in the spleen. These observations were considered to be vaccine-related, reflecting the establishment of an immune response. The effects of immunization on the popliteal and inguinal lymph nodes were less consistent, with no direct histopathological correlates. Some other variations were noted in mean organ weights in one or both sexes, but they were sporadic or not consistent and thus were not considered to be vaccine-related.
Microscopic observations at the site of injection, collected 3 days after the third vaccine administration revealed minimal to slight degeneration of the myofibers, mononuclear cell infiltrates (plasma cells, macrophages) and few heterophils, minimal to slight eosinophilic fibrillar material, and minimal to slight hemorrhage. On Day 57, only remnants of these inflammatory reactions were observed.
Biodistribution Study in Rats
Clinical signs
There were no deaths and no clinical signs related to the treatment during the study. Non-adverse and reversible erythema, scabs, and/or dryness were observed sporadically at the injection site in the control and vaccine-treated groups. All slight and reversible changes were considered to be unrelated to the treatment. Body weight and food consumption were not affected by the treatment (data not shown).
Biodistribution
Biodistribution rat study—one immunization on Day 1. ChAd155-RSV candidate vaccine quantities in tissue and blood samples (vp/g tissue or µL blood).
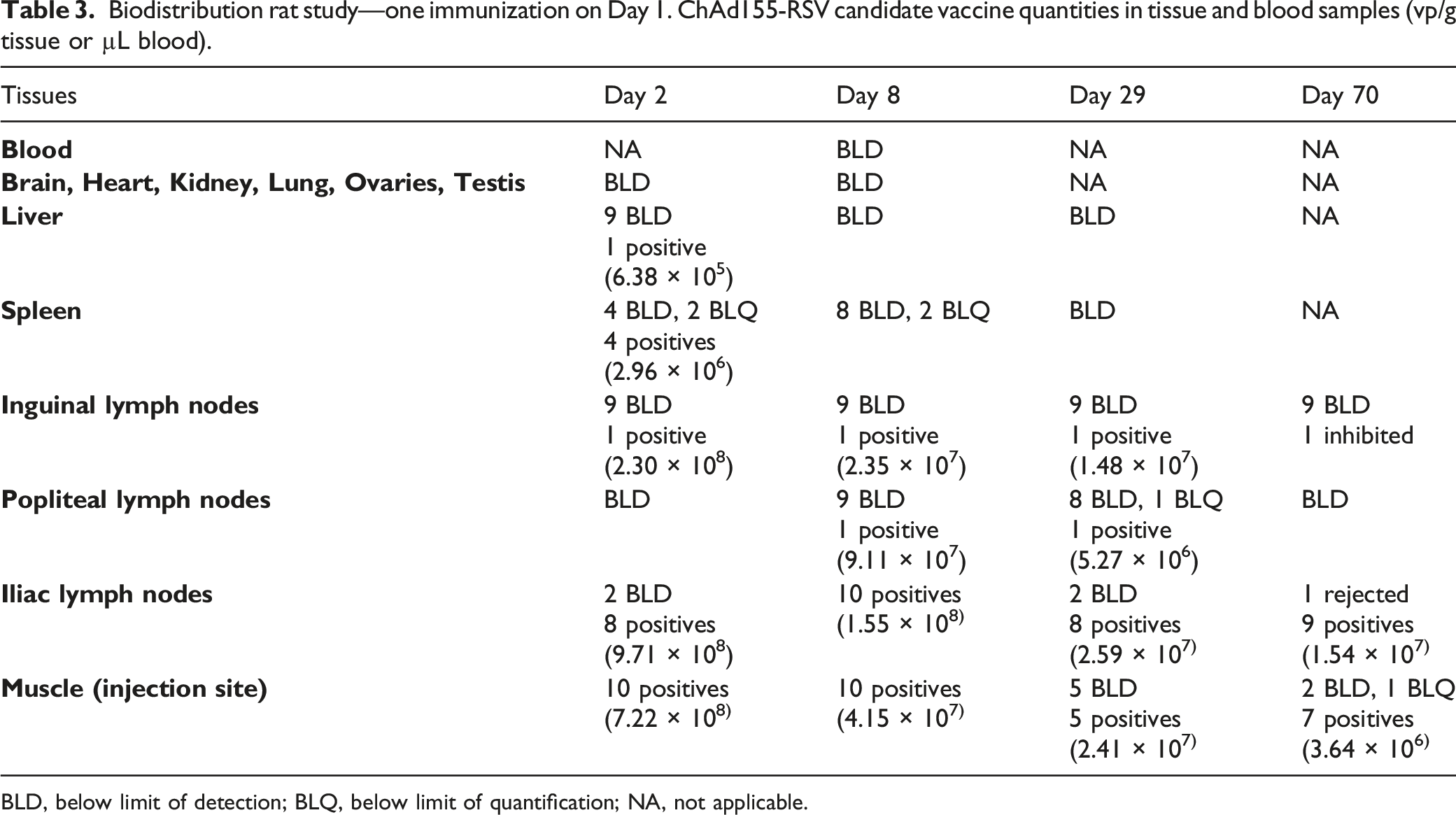
BLD, below limit of detection; BLQ, below limit of quantification; NA, not applicable.
ChAd155-RSV was not detected in any tissues at any time point in the control group. It was neither detected in the brain, heart, kidney, lung, ovary, and testis samples from animals of the ChAd155-RSV-treated group. These results were not false negative, as no PCR inhibition was detected here.
In contrast, in the vaccine-treated group, ChAd155-RSV DNA was found at the injection sites and in the draining lymph nodes from Day 2. In these tissues, ChAd155-RSV DNA concentration progressively decreased, but remained detectable until Day 70 at the injection site and in the iliac lymph nodes. Only 1/10 animals showed ChAd155-RSV DNA-positive inguinal and popliteal lymph nodes up to Day 29. ChAd155-RSV DNA was also detected in the spleen and liver, only on Day 2, but with quantity relatively low compared with the injection sites and lymph nodes.
Shedding Study in Rats
To assess the potential release of infectious ChAd155-RSV particles in excretions and biological fluids, samples were first tested for the presence of ChAd155-RSV DNA by specific qPCR assay. All samples in which the target DNA was detected, even if below the defined LOD, were further tested using an in vitro infectivity assay.
Shedding rat study—one immunization on Day 1. Number of viral particles in secretions and excreta (vp/µg DNA).
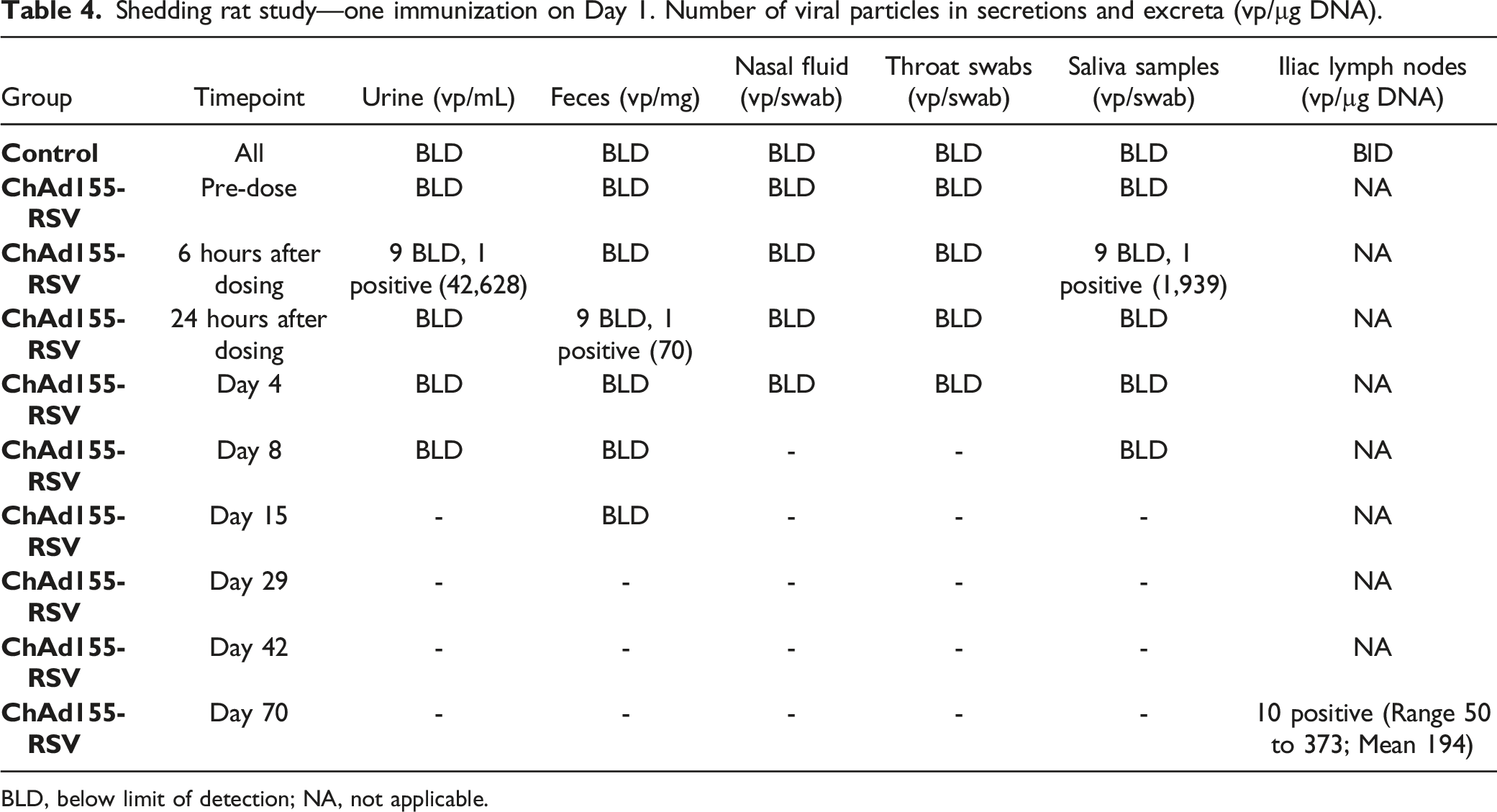
BLD, below limit of detection; NA, not applicable.
Based on the low number of positive samples containing DNA and the lack of a clear signal that infectious particles were being shed, it was concluded that it would be unlikely for significant amounts of infectious ChAd155-RSV to be shed post-vaccination.
Discussion
The toxicology studies conducted with ChAd155-RSV showed no premature deaths, no adverse clinical signs, and no treatment-related effects on body weight or food consumption. During the repeated-dose study, there was a transient increase in body temperature in a few animals, but often values remained within the control range, hence the finding was not considered toxicologically significant. Inflammatory reaction at the site of injection, and immune stimulation in draining lymph nodes and spleen were consistent with the administration of an immunogenic vaccine.
Repeated IM doses of ChAd155-RSV in rabbits were associated with reversible effects on hematology (decreased WBC) and clinical pathology parameters (prolonged APTT and increased fibrinogen, CRP, triglycerides, and cholesterol). Asymptomatic and transient neutropenia or leukopenia have also been reported clinically after administration of an adenoviral vector vaccine, and those observations are consistent with margination and thus related to the vaccine-induced innate immune response. 30 Variations of APTT have been described in rabbits after administration of adenovirus-vector vaccines and are discussed as a reflection of acute inflammation due to the immune response after vaccination. 50 It is unclear if this effect could be attributed to vector-induced anti-phospholipid antibodies leading to an artefact in the assay, an hypothesis discussed in literature to explain the APPT prolongation observed in clinical trials. 30 Fibrinogen and CRP are acute phase protein markers and are often found to be increased following vaccination in toxicity studies. 49 Transient and mild increases in cholesterol and triglycerides mainly after the first administration in the repeated-dose study were considered of minor importance and non-adverse. Since all these changes were transient, returning to baseline 7 days post injection and not related to clinical signs or histopathological findings, they were deemed to be of no toxicological significance, and interpreted as signs of the inflammatory reaction following vaccination. 50
The most remarkable effect was a decrease in platelets (up to 59% in males and 40% in females relative to concurrent controls) one day after the first and the third administration (on Days 2 and 30, respectively). This decrease had fully reversed by Day 8 and Day 36 (7 days after vaccine administration). The observed transient decrease in platelets was interpreted as a direct consequence of the presence of adenoviral particles in blood following their drainage from the injection site. A decrease in platelet count has been reported in the literature after adenovirus vector parenteral administration in different animal models51-54 and in humans. 55 A proposed mechanism is that adenovirus particles bind to platelets and the resulting complexes are subsequently sequestered and degraded in the liver. 56 In our study, the effect on thrombocytes was limited in time and was not associated with systemic clinical signs or hemorrhages during the in-life phase or at necropsy.
The adenoviral vector used to carry the RSV antigen is replication-deficient, entailing that no de novo functional viral particles can be generated in the vaccine-recipient. Nevertheless, the vector may disseminate outside the injection site and there is need to identify the potential dissemination in secretions/excreta. After a single injection of ChAd155-RSV in rats, there was no clear evidence of the presence of the vector in secretions or excreta samples. Only three samples from three different animals collected within 24 hours after injection tested positive for ChAd155-RSV DNA, none being associated with the presence of infectious viral particles. Therefore, based on these results in rats, the dissemination of ChAd155-RSV by humans after vaccination in secretions or excreta is considered unlikely. Further, the risk of recombination of ChAd155-RSV with circulating adenoviruses is considered only hypothetical, due to little homology between the backbones and E1 sequences, and its probability very low since the routes of natural infection would not lead to colocalization of the circulating wild-type viruses in the same cells than those infected by the vector vaccine injected intra-muscularly. 57 In fact, the authors are not aware of any recombination events reported in the literature with replication-defective vectors.
After a single IM administration of the candidate vaccine, ChAd155-RSV DNA was mainly found at the injection site and in the draining lymph nodes, peaking one day after vaccination. Localization in the draining lymph nodes reflects the capture of the test item by the immune system of the host. The amount of viral DNA decreased over time in these organs up to Day 70, indicating progressive clearance. Vaccine DNA was found sporadically in other lymph nodes, in spleen and in the liver, but was not detected in brain, heart, kidneys, lungs, ovaries, and testes. Overall, these results are in line with earlier observations in rabbits made with human adenovirus type 5- and type 35- and chimpanzee adenovirus type 3-vectored vaccines.50,58 One may note the persistence of adenoviral DNA at the injection sites and in the draining lymph nodes more than 2 months after vaccine administration (Day 70). Such persistence is not uncommon and has already been observed previously,50,58 which may indicate a common feature of adenovirus-based vaccination. It should be noted that our biodistribution data are based on an assay targeting the CMV promoter of the vector DNA so that what is detected on Day 70 might only be CMV promoter DNA residues. In addition, it must be emphasized that the quantities detected at the final time point (5.31 × 103 copies/µg DNA in the muscle, on Day 70) were below the threshold level (30 × 103 copies/µg host DNA) above which further studies would be required,59,60 and that the dose relative to body weight administered to rats exceeded by 30-fold the human dose.
In conclusion, under the conditions of these studies, repeated administration of the ChAd155-RSV candidate vaccine was locally and systematically well-tolerated, with the expected inflammatory reaction/immune response triggered by vaccination and a transient drop in platelet count without physiological consequences, as previously observed after administration of other adenovirus-based vaccines. No unexplained vector dissemination or shedding was observed.
These data are supportive of the ChAd155-RSV vaccine being suitable for human clinical trials in healthy volunteers.
Footnotes
Acknowledgments
We thank Pascal Cadot (GSK, Rixensart, Belgium) who provided scientific writing services in the manuscript development. Robert Lin (GSK, Rockville, US) and Pascal Cadot coordinated the manuscript development.
Author Contributions
Alan H. Stokes contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Camille Planty contributed to conception and design and critically revised manuscript; Johanne Pion contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Philippe Ancian contributed to acquisition and analysis and critically revised manuscript; Alexandra Rogue contributed to acquisition and analysis and critically revised manuscript; Carine Bansard contributed to acquisition and analysis and critically revised manuscript; Jérémy Silvano contributed to design, contributed to acquisition and analysis and critically revised manuscript; Dominique Papineau contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Nawel Ben Abdeljelil contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Giulietta Maruggi contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Haifeng Song contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Catherine Spickler contributed to design, contributed to analysis, and critically revised manuscript; Karine Blouin contributed to acquisition and analysis, and critically revised manuscript; Guillaume Dubois contributed to acquisition, and critically revised manuscript; Luis-Alexander Rodriguez contributed to conception, contributed to analysis and interpretation, and critically revised manuscript; Judith Baumeister contributed to conception, contributed to analysis and interpretation, and critically revised manuscript; Ann-Muriel Steff contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Eric Destexhe contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Disclosures
AHS, CP, ED, GM, HS, LAR, JB, and AMS are, or were at the time of the study, employees of the GSK group of companies. AHS, AMS, ED, GM, and LAR report ownership of GSK stocks. JP, AR, CB, JS, DP, CP, KB, PA, GD, and NBA are employees of Charles River Laboratories, a Contract Research Organization contracted by GSK in the context of these studies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored and financially supported by GlaxoSmithKline Biologicals, SA. The costs associated with the development and publishing of the manuscript were also covered by GlaxoSmithKline Biologicals SA.
