Abstract
Respiratory syncytial virus (RSV) is a leading cause of acute lower respiratory tract infections, and vaccines are needed to treat young children and older adults. One of GSK’s candidate vaccines for RSV contains recombinant RSVPreF3 protein maintained in the prefusion conformation. The differences in immune function of young children and older adults potentially require different vaccine approaches. For young children, anti-RSV immunity can be afforded during the first months of life by vaccinating the pregnant mother during the third trimester with unadjuvanted RSVPreF3, which results in protection of the infant due to the transplacental passage of anti-RSV maternal antibodies. For older adults with a waning immune response, the approach is to adjuvant the RSVPreF3 vaccine with AS01 to elicit a more robust immune response.
The local and systemic effects of biweekly intramuscular injections of the RSVPreF3 vaccine (unadjuvanted, adjuvanted with AS01, or coadministered with a diphtheria-tetanus-acellular pertussis vaccine) was tested in a repeated dose toxicity study in rabbits. After three intramuscular doses, the only changes observed were those commonly related to a vaccine-elicited inflammatory reaction. Subsequently, the effects of unadjuvanted RSVPreF3 vaccine on female fertility, embryo-fetal, and postnatal development of offspring were evaluated in rats and rabbits. There were no effects on pregnancy, delivery, lactation, or the pre- and postnatal development of offspring.
In conclusion, the RSVPreF3 vaccine was well-tolerated locally and systemically and was not associated with any adverse effects on female reproductive function or on the pre- and postnatal growth and development of offspring.
Keywords
Introduction
Respiratory syncytial virus (RSV) is a major cause of severe disease in two human populations, young children and older adults (≥60 years of age). In the former population, during the first year of life, 50% to 70% of infants are infected with this pathogen, and essentially 100% will have an RSV infection by their second birthday. 1 Severe disease occurs mostly in infants ≤6 months of age for whom it is a leading cause for hospitalizations. 1 -5 In the elderly individuals, RSV is one of the important viral pathogens identified as being responsible for acute respiratory infections. 6 RSV-induced respiratory disease was shown to develop in 3% to 7% of healthy older adults and 4% to 10% of high-risk adults. 6 In 2015, it was estimated that 1.5 million of RSV-induced acute respiratory infections occurred in industrialized countries, of which 14.5% led to hospitalizations. Further, a total of 14,000 in-hospital deaths were attributed to RSV globally. 7 No preventive intervention is currently available that adequately addresses this significant global health problem, neither in children nor in older adults. Developing an RSV vaccine or age-tailored RSV vaccines would help to address this global public health priority.
If a common RSV antigen can be used for both patient populations (children and older adults), it should be recognized that both target populations have specific immunological characteristics that need to be considered to produce effective vaccination strategies. Older adults are confronted with the phenomenon of immunosenescence, meaning that the potency of the immune response fades with age. 8 Consequently, the vaccine-induced immune responses in older adults need to be enhanced to provide better protection, which can be achieved by using an adjuvant. Adjuvants are immunostimulatory molecules or combinations of such molecules associated with an antigen to improve immunogenicity for long-term protection against disease. Adjuvants have been shown to increase antibody titers; increase seroconversion rates in populations with reduced responsiveness due to age, disease, or therapeutic interventions 9,10 ; facilitate the use of smaller doses of antigen(s) in response to urgent large-scale vaccinations and/or when manufacturing facilities are limited 11 -13 ; and reduce the frequency of treatments with the vaccine, which could be an important consideration given the logistical challenges in some parts of the world without ready access to medicine(s) or local health care facilities. 14 In summary, use of adjuvants in vaccine treatment programs has widespread utility and potential advantages that could be paramount to the successful treatment of RSV disease globally, particularly in patient populations that struggle with immature or weakened immunity and those with limited access to medicine. The Adjuvant System, AS01, is under consideration for use with the RSVPreF3 vaccine, since this adjuvant is already successfully used in the frame of a zoster vaccine for older adults 15 and due to its potential to induce robust specific CD4+ responses and rapid and durable humoral and cellular responses when combined with a protein antigen. 16 -19
The development of an RSV vaccine for neonates and infants is challenging due to the immaturity of the immune system in this population and also because RSV is reported to trigger a suboptimal innate immune response. 20,21 To address these challenges, GSK is attempting to use the potential of passive protection of the young infants that is afforded by the mother’s immunity through transplacental passage of maternal antibodies. This approach is now accepted for inactivated influenza, pertussis, and tetanus vaccines. 22 Since most women of childbearing potential would have preexisting immunity to RSV, due to prior infection, an RSV vaccine given during the third trimester of pregnancy is likely to boost pre-existing antibody levels and thus proportionally increase the amount of anti-RSV immunoglobulin G (IgG) transferred from mother to child. Given that serum neutralizing antibodies are considered key players to defeat RSV infection, 23 -26 this approach would be expected to provide protection to the infant through the transfer of maternal antibodies for the first few months of life.
The antigen used in the RSVPreF3 vaccine is an engineered version of the RSV fusion (F) surface glycoprotein, stabilized in the naturally occurring prefusion conformation by introduction of cysteine residues leading to the formation of a disulfide bond and filling of hydrophobic cavities. 27 The prefusion conformation, rather than the postfusion conformation, is the main target of naturally induced anti-RSV antibodies in human serum. 28 -30
To support the clinical development and eventual licensure of the RSVPreF3 vaccine, several nonclinical toxicity studies were performed. This article reports on three of the studies; a repeated dose toxicity study performed in male and female rabbits and two developmental and reproductive toxicity (DART) studies performed in rats and rabbits. The objectives of the repeated dose toxicity study were to evaluate potential local and/or systemic toxic effects of the RSVPreF3 vaccine after three separate biweekly intramuscular (IM) injections and onset, persistence, or reversibility of these effects. The vaccine was administered unadjuvanted, adjuvanted with AS01, or coadministered nonadjuvanted with a diphtheria-tetanus-acellular pertussis (DTPa) vaccine (Boostrix). Coadministration with Boostrix was studied in the repeated dose toxicity study to determine the immunogenicity and potential toxicity of the coadministration. Boostrix is currently recommended in certain countries to be given to pregnant women in the third trimester of pregnancy. Furthermore, as pregnant women are one of the target populations of the RSVPreF3 vaccine, understanding the potential for adverse effects of vaccination on a pregnancy or in women who are planning to become pregnant is paramount from a risk–benefit perspective. Thus, DART studies that evaluated fertility, pregnancy, embryo-fetal development (including assessment of teratogenicity), parturition and lactation, as well as the postnatal growth and development of offspring were performed in rats and rabbits using the nonadjuvanted form of the RSVPreF3 vaccine.
Materials and Methods
Regulatory Guidelines and Data Quality
The rabbit toxicity study was carried out in compliance with the principles of the Food and Drug Administration Center for Drug Evaluation and Research (CDER, January 2010), International Conference on Harmonisation (ICH) Harmonised Tripartite Guideline ICHM3(R2) on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals (June 2009), and the World Health Organization (WHO) Guideline on the Nonclinical Evaluation of Vaccines (2005).
The rat and rabbit reproduction studies were carried out in compliance with the ICH Harmonised Tripartite Guidelines M3(R2) Nonclinical Safety Studies for the Conduct of Human Clinical Trials for Pharmaceuticals (June 2009) and S5(R2) Detection of Toxicity to Reproduction for Medicinal Products and Toxicity to Male Fertility (November 2005), and S6(R1) Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals, as well as the US Department of Health and Human Services Food and Drug Administration (CBER), Guidance for Industry: considerations for developmental toxicity studies for preventive and therapeutic vaccines for infectious disease indications (February 2006), and WHO Technical Report Series, No. 927, Annex 1 (2005) Guidelines on the Nonclinical Evaluation of Vaccines.
Each study was conducted in a laboratory that complied with Good Laboratory Practice (GLP) standards and guidelines for the conduct of toxicity studies as outlined by the US Department of Health and Human Services, Food and Drug Administration, as well as standard operating procedures established for the laboratory. The studies were monitored by Test Facility Quality Assurance (QA) personnel to ensure the facilities, equipment, personnel, methods, practices, records, and controls used were in conformance with GLP standards for conduct of nonclinical studies on the safety of drugs. The QA personnel also reviewed the study protocols and amendments, conducted inspections at intervals considered adequate to assure the integrity of each study, and audited the final reports to assure that they accurately described the methods and standard operating procedures followed and that reported results were an accurate reflection of the raw data.
Animals and Husbandry
Specific pathogen free-bred male and female Hra: New Zealand White albino rabbits were obtained from Covance Research Products, Inc, and female Crl: WI Han (Han Wistar) rats were obtained from Charles River Laboratories Inc. The animals were allowed to acclimate prior to the first day of dosing. They were kept individually in stainless steel cages (rabbits) or bedding-filled plastic nesting boxes (rats), housed in dedicated facilities with filtered air (10 or greater air changes/hour) at a mean temperature range of 16 °C to 22 °C (rabbits) or 20 °C to 26 °C (rats) and relative humidity in the range of 30% to 70%. The lighting followed a 12-hour light/12-hour dark cycle. Both rats and rabbits were provided ad libitum a certified laboratory diet and had free access to domestic-quality drinking water. The animals were given various cage-enrichment devices and/or dietary enrichment.
Animal Welfare
The rabbit repeated dose toxicity study was conducted in the laboratories of Covance (Greenfield, IN, USA), while the rat and rabbit DART studies were conducted at Charles River laboratories (Horsham, PA, USA). Both laboratories were compliant with GLP standards. The welfare of the animals was maintained in accordance with the general principles of the Association for Assessment and Accreditation of Laboratory Animal Care for the repeated dose study. The DART studies were in accordance with all applicable sections of the Final Rules of the Animal Welfare Act regulations (Code of Federal Regulations, Title 9), the Public Health Service Policy on Humane Care and Use of Laboratory Animals from the Office of Laboratory Animal Welfare, and the Guide for the Care and Use of Laboratory Animals from the National Research Council. The procedures in the protocols were in compliance with all applicable animal welfare acts and were approved by the local Institutional Animal Care and Use Committee (Approval No. ACUA 18-109 for the repeated-dose study and 20152506 and 20152507 for the DART studies).
Control and Test Items
AS01 is a liposome-based formulation containing 3-O-desacyl-4′-monophosphoryl lipid A (MPL) and QS-21 (Quillaja saponaria Molina, fraction 21; licensed by GSK from Antigenics LLC, a wholly owned subsidiary of Agenus Inc, a Delaware, US corporation). AS01 used in this work is the AS01B variant containing 50 µg MPL and 50 µg QS-21 per 500 µL. MPL is the detoxified derivative of lipopolysaccharide from Salmonella minnesota and stimulates activation of innate immunity via toll-like 31 receptor 4; QS-21 is a saponin molecule extracted from the bark of the South American tree Quillaja saponaria Molina, fraction 21 and is a water-soluble triterpene glycoside with amphiphilic character known for its hemolytic activity and promotion of antigen-specific antibody responses. 16 The combination of MPL and QS-21 components in AS01 is required to achieve the highest antigen-specific adaptive response. 32 The vaccine Boostrix (GSK) is a reduced antigen-content DTPa vaccine, developed for booster vaccination of children, adolescents, and adults. The RSVPreF3 vaccine, AS01B and Boostrix were supplied by GSK and maintained under refrigerated conditions for the duration of the study. RSVPreF3 was provided freeze-dried, ready for reconstitution with either 0.7 mL of 0.9% saline or AS01, as applicable. AS01 was provided as a monodose vial. Boostrix was used as supplied. All RSVPreF3 vaccine formulations were prepared fresh on the day of use, maintained under ambient conditions, and used within 6 hours after reconstitution. The control item used in the toxicology studies was a ready-to-use formulation of 0.9% saline, nonpyrogenic, and sterile.
Study Designs
Repeated dose toxicity study in rabbits
Rabbits were randomly allocated in fpur groups of 20 animals each, comprising 10 males and 10 females. On the first day of dosing, the rabbits were 14 to 15 weeks old and weighed 1.83 to 2.46 kg (males) or 1.97 to 2.48 kg (females). The animals were dosed by IM injection (2×0.5 mL in a thigh muscle) on days 1, 15, and 29. At each dosing occasion, all animals were administered two injections, which were right/left adjacent in the same leg (ie, homolateral injection). For each vaccination, the control group was administered two injections of saline. The unadjuvanted group was administered one injection of RSVPreF3 and one injection of saline. The adjuvanted RSVPreF3 group was administered one injection of RSVPreF3/AS01 and one injection of saline, and the coadministration group was administered one injection of RSVPreF3 and one injection of Boostrix. For the RSVPreF3 vaccinations, each 0.5 mL dose volume contained 240 µg of the RSVPreF3 antigen. The first five rabbits/sex/group were euthanized at the end of the treatment period on day 32. The remaining rabbits were maintained on study for approximately four weeks then euthanized at the end of the treatment-free period (day 57).
Toxicity was evaluated based on changes in viability, visible signs of ill health and any behavioral changes, increases in body temperature, body weight, food consumption, and ophthalmology. On each dose occasion, rectal temperature was recorded predose, and the injection sites were observed for reactions; the injection site was scored/graded using a modified Draize scale, based on the extent of erythema and edema present. Both measurements were also collected at approximately 6, 24, and 48 hours after completion of immunizations for each sex/group. Body weights and qualitative food consumption were recorded at least twice during the predose phase, prior to dosing and at least twice weekly (body weights) or daily (food consumption) through the remainder of the study to closely monitor for changes in general health of the animals. Ophthalmic examinations were conducted by a board-certified ophthalmologist during the predose phase, within three days after the last dose and within seven days of the end of the treatment-free period. Mydriasis was induced in each eye to allow examination with a direct ophthalmoscope and a slit-lamp biomicroscope.
Laboratory evaluations (hematology, coagulation, clinical chemistry, and serology) were performed once prior to the start of treatment and again on days 32 and 57 (serology), and days 2, 8, 30, 36, and 57 (hematology, coagulation, and clinical chemistry). Blood was collected from the ear vein in noncoated serum tubes (for serology), tubes with EDTA (for hematology) or citrate (for coagulation), or in heparinized tubes (for clinical chemistry). Hematology parameters included erythrocyte counts; hemoglobin level; hematocrit; mean corpuscular volume; mean corpuscular hemoglobin; mean corpuscular hemoglobin concentration; red cell distribution width; absolute reticulocyte count; platelet count; leukocyte count; and absolute neutrophil, lymphocyte, monocyte, eosinophil, basophil; and large unstained cell counts. Coagulation end points included prothrombin time, activated partial prothrombin time, and fibrinogen. Clinical chemistry measurements included alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, creatine kinase, γ-glutamyl transferase and lactate dehydrogenase activity, as well as glucose, total protein, albumin (and ratio albumin to globulin), urea, creatinine, bilirubin, cholesterol, triglycerides, phospholipids, calcium, sodium, potassium, chloride, inorganic phosphate, and C-reactive protein (CRP) concentration.
At study termination on day 32 (five animals/sex/group) and on day 57 (all remaining rabbits), the animals were euthanized and necropsy was performed, organ weights were recorded, and histopathologic examination of protocol required tissues completed. Tissues were weighed and collected for histopathologic examination following the WHO guidelines for adjuvanted vaccines 33 and were preserved in phosphate-buffered neutral 4% formaldehyde. All tissues submitted for histopathological evaluation were embedded in paraffin wax, sectioned at 5 µm, and stained with hematoxylin and eosin prior to microscopic examination.
Developmental and reproductive toxicity study in rats and rabbits
The designs of the rat and rabbit DART studies are illustrated in Figure 1. Selected females (n = 96) were randomly assigned either to a saline control group (n = 48) or to an RSVPreF3 group (n = 48) using a computer-based randomization program balanced by body weight. Once mated, females were further allocated to either a cesarean delivery cohort (n = 24/group) or a natural delivery cohort (n = 24/group). For the RSVPreF3 vaccinations given to rabbits, each 0.5 mL dose volume contained 120 µg of the RSVPreF3 antigen. The concentration of RSVPreF3 given to rats was 48 µg/injection in 0.2 mL/injection. Animals were dosed by IM injection on day 1 of study (DS1, which was 28 days prior to mating), DS15 (14 days prior to mating), during gestation (rat: gestation days [GD] 3, 9, and 15; rabbit: GD3, 11, 16, and 24), and once during the lactation period (lactation day [LD] 7). The frequency of dosing was expected to maintain the immunogenic response throughout all phases of the female reproductive cycle including ovulation, implantation, early embryonic development, organogenesis, parturition, and lactation, all of which facilitated passive transfer of RSVPreF3 antibodies to offspring. Toxicity was evaluated by assessing changes in clinical signs, behavior, body weight, and food consumption; females were examined within one to three hours after each dose for evidence of injection site reactions. Any positive findings were scored/graded using a modified Draize scale, based on the extent of erythema and edema present. Starting on DS28, female rats and rabbits were mated with breeder males (1:1) of the same strain and source. Evidence of sperm in a vaginal lavage and/or presence of an intravaginal copulatory plug confirmed insemination. In rats and rabbits, mating was confirmed by observation of copulation with at least two breeder males. The day of mating was designated as GD0. Within two hours of confirmed mating, female rabbits were given 20 USP units/kg of human chorionic gonadotropin to induce ovulation.
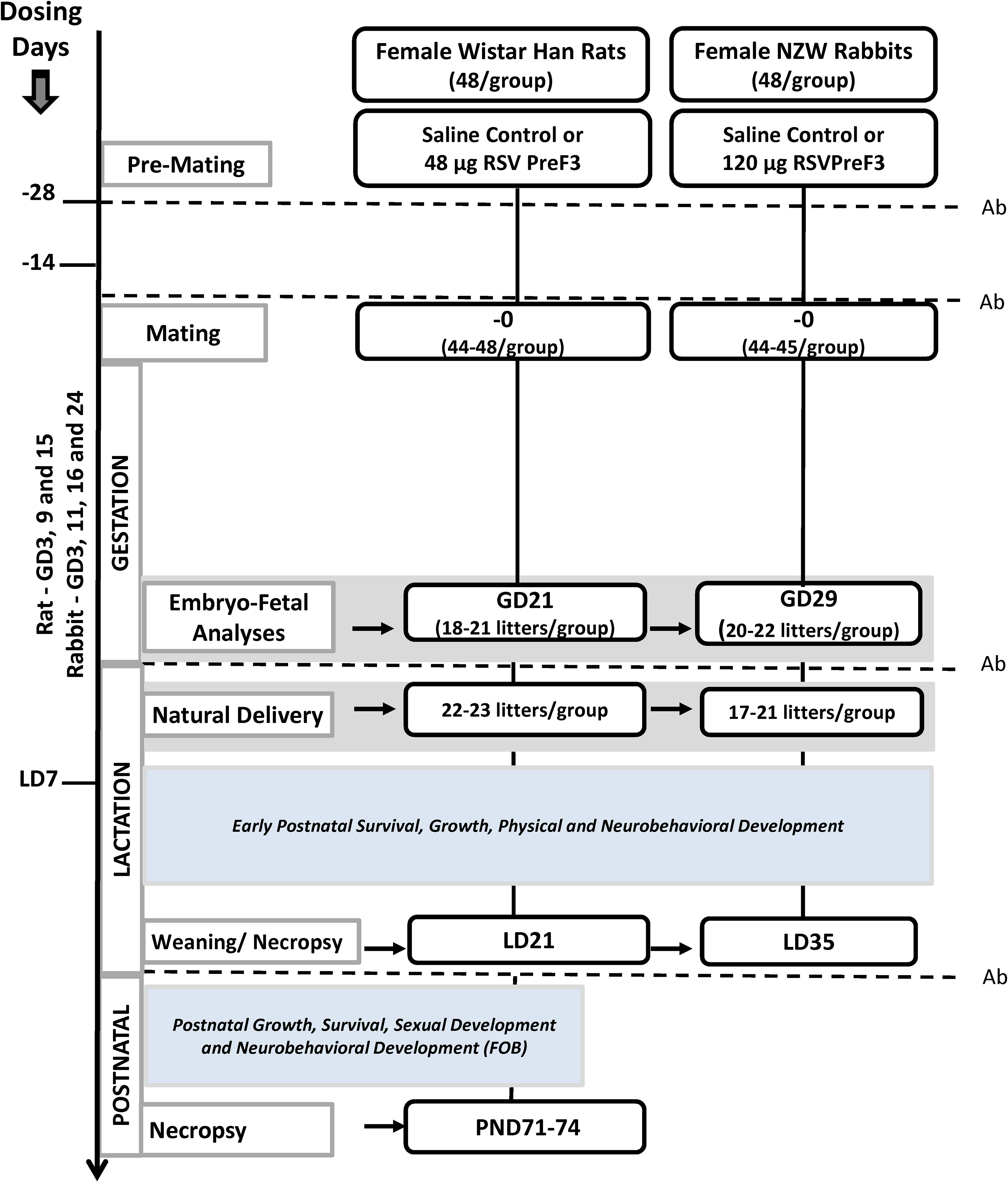
Reproductive and developmental study designs. Animals were dosed at 14 and 28 days prior to mating, during gestation (rats: GD3, 9, and 15; rabbits: GD 3, 11, 16. and 24 and on lactation day (LD) 7. Pregnant females were divided and assigned to a cesarean section cohort or a natural delivery cohort. Ab indicates RSVPreF3 antibody titers measurement; GD, gestation day; PND, postnatal day.
Mated female rats and rabbits assigned to the cesarean section cohort were euthanized and necropsied on GD21 (six days after last dose) or GD29 (five days after last dose), respectively. The number and distribution of corpora lutea, implantation sites, early and late resorptions, and live and dead fetuses were recorded. To eliminate bias, all fetal examinations were conducted blind to identification and treatment. Fetuses were weighed prior to euthanasia via intraperitoneal injection of sodium pentobarbital then were examined fresh (50% of rat fetuses/litter and 100% rabbit fetuses), using a dissecting microscope, for external and visceral abnormalities following microdissection techniques published earlier. 34 -36 The same fetuses were stained with alizarin red S, and skeletal examinations were performed. 35 The heads of the remaining 50% of rat fetuses in each litter were humanely removed and fixed in Bouin solution for subsequent examination following Wilson free-hand razor sectioning technique, 37 whereas all of the rabbit fetuses received fresh eye and brain examinations as described earlier. 38 After completion of the fresh head examination, the stained rabbit fetuses were examined for skeletal abnormalities. 35
Mated rats and rabbits assigned to the natural delivery cohort were monitored hourly between 8:00 and 18:00 hours starting on GD21 (rats) or GD29 (rabbits) for onset/completion of parturition. The progress of active rat deliveries was evaluated at least twice every hour to determine the number of live and dead pups born (nonviable status was confirmed as stillborn via lung floatation test) to facilitate calculation of gestation length, duration of delivery period, and parturition rate (pup delivery time). Pregnant rabbits were monitored frequently each day for onset of delivery and upon completion were left undisturbed for four days to bond with their kits and to minimize the risk of maternal stress-induced adverse outcomes postdelivery. Completed deliveries were considered LD1 for mothers and postnatal day (PND) 1 for the offspring. Litters were examined daily for viability, and external abnormalities and assessments of growth (body weights and tibia measurements) were performed at least weekly during the preweaning period. Early assessments of physical growth and development (hair growth, pinna unfolding, incisor eruption, eye opening, pupil constriction) and neurobehavioral and reflex development (surface righting reflex, acoustic startle reflex, and forelimb grip reflex) were performed at various ages throughout the preweaning period.
Maternal female rats and rabbits and their offspring, except where noted below, were euthanized and necropsied on LD/PND21 or LD/PND35, respectively, following completion of weaning procedures. Organ weights (adults: paired ovaries/uterus; offspring: brain) were recorded. A select number of weaned rat pups (n = 24/sex/group; 1 pup/sex/litter as applicable) were randomly chosen to remain on study for continued postnatal observations of growth and development which included body weight measurements that were performed at least weekly and attainment of physical sexual maturation (vaginal opening starting on PND27 and balanopreputial separation starting on PND39). In addition, sensorimotor and functional neurobehavioral development testing was performed when the male and female rat pups reached adulthood starting on PND70. 39 -42 Testing was conducted with the observer unaware of group assignment and took place while the animal was in its home cage, during handling, and while in an open field. Examinations included assessment of clinical signs; body temperature; autonomic functions; reactivity and sensitivity to audio, visual, tactile, and painful stimuli; level of excitability; movement (gait and coordination); and grip strength. Upon completion of behavioral testing (approximately PND71-PND74), offspring were euthanized and necropsied and brain weights were recorded.
Serology Sample Collection and Analysis
Blood was collected from each pregnant rat/rabbit on DS1 (prior to dosing), on DS24, GD21/29 (cesarean-section cohort), and LD21/35 (natural delivery cohort) following standard methods of the laboratory (conscious rats/rabbits: jugular vein or medial auricular artery or following euthanasia: vena cava). In addition, umbilical cord blood was collected from all viable rat and rabbit fetuses on GD21 or GD29, which was the day of scheduled caesarean delivery, respectively, using microcapillary tubes. The blood collected from each fetus within a given litter was pooled together into one sample tube to ensure sufficient testing volume for analysis (at least 250 µL/sample). Finally, a terminal blood sample (up to 1.0 or 2.0 mL/sample, respectively) was collected from weaned rats and rabbits on PND21 or PND35, respectively. All of the blood samples were processed to serum and analyzed for anti-RSVpreF3 antibodies at Covance Laboratories, Inc.
Anti-RSVPreF3 and anti-diphtheria (DT) antigen antibodies were measured with standard enzyme-linked immunosorbent assays (ELISA). Briefly, the serology method involved overnight coating of ELISA plates with RSVPreF3 or DT antigen as appropriate. On the next day, after washing and blocking, diluted standard control sera or test samples were added. After an incubation step, the plates were washed and any bound antibodies were detected using horseradish peroxidase (HRP)-labeled goat antirabbit IgG for the repeated dose study; the detection system used for DT involved peroxidase-labelled antirabbit IgG. For the samples collected on the DART studies, a biotinylated goat antirat IgG was used, followed by streptavidin-HRP. Then absorbance was read at 450/620 nm using an ELISA plate reader after addition of a peroxidase substrate. A standard curve was generated by applying a 4-parameter logistic regression fit to the reference standard results. Antibody titer values in the control serum or in samples (expressed in EU/mL) were calculated by interpolation from the standard curve. The antibody titers were obtained by averaging the values from dilutions that fell within the 20% to 80% segment of the standard curve. Mean titers with the coefficient of variation greater than 20% were repeated under similar or revised dilutions.
Statistical Analyses
Repeated dose toxicity study in rabbits
Data for each sex were analyzed separately. Analysis of variance (ANOVA) and pairwise comparisons were used to analyze absolute body weight, body weight change, continuous clinical pathology values, terminal body weight, absolute organ weight, organ/body weight percentage, and organ/brain weight percentage in the repeated dose study.
Reproduction and developmental toxicity studies in rats and rabbits
All statistical tests were performed at the 5% level of significance. Numerical data were analyzed according to sex and occasion; the litter was the unit of analysis for data on conceptuses through preweaning period. Data on nonpregnant animals and incidences of clinical signs and pathological findings were excluded from data analysis. Descriptive statistics and inferential statistics were performed as appropriate.
The parametric and nonparametric data including body weights, food consumption, gravid uterus weight, parturition rate, fetal/pup weights, organ weights, postnatal gender distribution per litter, physical development landmark variables, tibia length, auditory startle, and quantitative functional observational battery (FOB) variables were analyzed using the Levene test to assess the homogeneity of group variances and the ANOVA test, when appropriate (ie, Levene test was not significant [P ≥ 0.05]). If the ANOVA was significant (P ≤ 0.05), Dunnett test was used to identify the statistical significance. If the ANOVA was not appropriate (ie, Levene test was significant [P ≤ 0.05]), the Kruskal-Wallis test and Dunn test were conducted, and Dunn test was used to identify the statistical significance. Group comparisons were evaluated at the 5.0%, 2-tailed probability level. If the ANOVA was not significant (P ≥ 0.05), no further analyses were conducted.
Some nonparametric data (fetal abnormality index, fetal sex ratio, duration of mating, gestation length, number of corpora lutea and implantations, litter size indices, survival indices, early physical development and reflex data [litter mean and day], day of sexual maturation, and qualitative FOB variables) were analyzed by conducting the Kruskal-Wallis and Dunn tests, and Dunn test was used to identify the statistical significance. Incidence data were analyzed using Fisher exact test. Data sets with only two groups were analyzed.
Incidence data (incidence of litters with fetal abnormalities, number or percentage of mated and pregnant, percentage of live births and stillbirths, fertility, and postnatal survival indices) were analyzed using a Fisher exact test.
Results
Repeated Dose Toxicity Study in Rabbits
Serology
The ELISA results showed that immunization took place in the three groups injected with RSVPreF3 vaccine, unadjuvanted, adjuvanted with AS01, or coadministered with Boostrix, with anti-RSVPreF3 antibody titers ranging from 6,205 to 995,405 EU/mL (Figure 2). No anti-RSVPreF3 IgG antibodies were detected in samples collected during the predose phase or in samples collected from control group animals during the dosing phase. The ELISA data characterizing the anti-DT immune response demonstrated IgG antibodies in all animals administered RSVPreF3 + Boostrix, which was indicative of an exposure to the DTPa vaccine (data not shown).
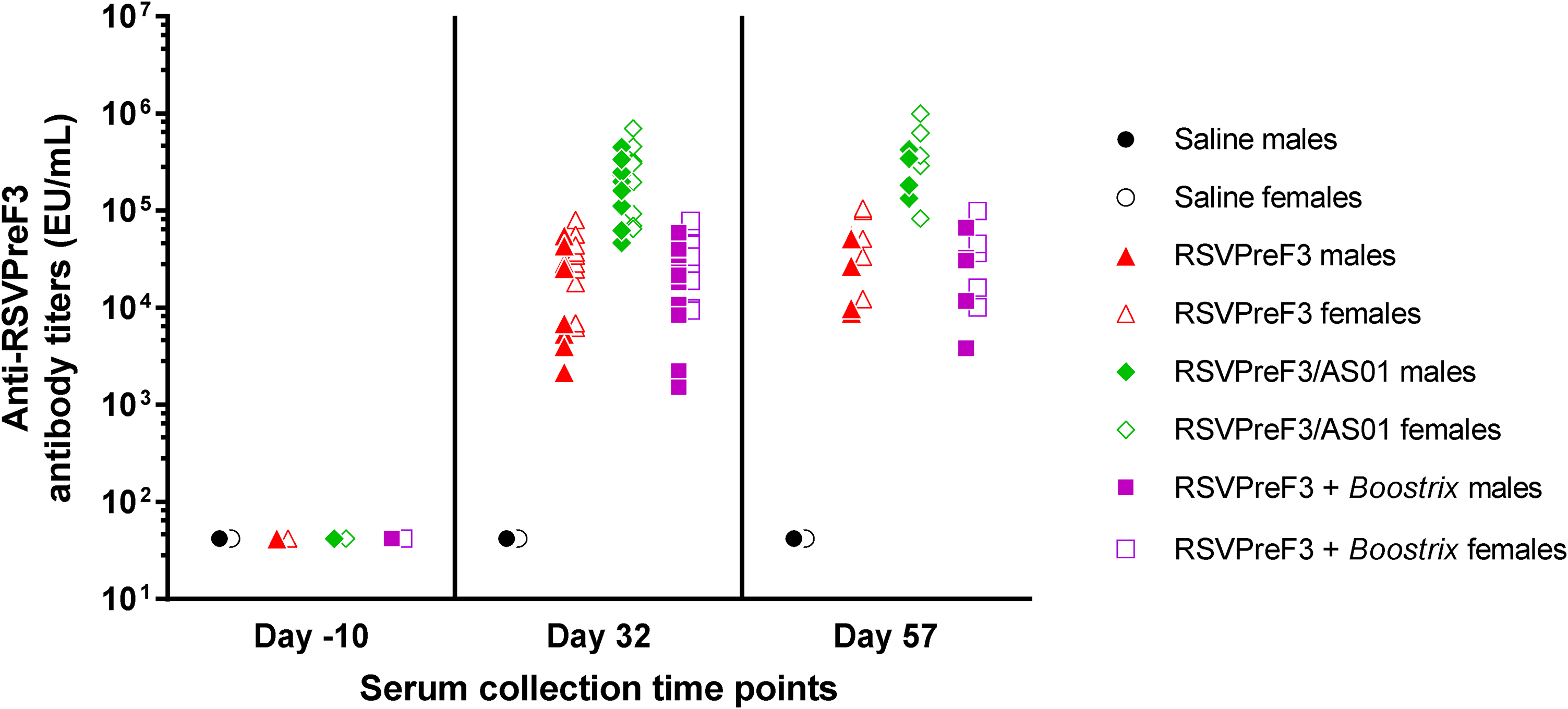
Repeated dose toxicity study. Rabbit serum was collected before immunization (day −10) and three days (day 32) and four weeks (day 57) after the third immunization. Anti-RSVPreF3 antibody titers in rabbit serum were measured by ELISA. Each symbol represents an animal. ELISA indicates enzyme-linked immunosorbent assay.
Observations and measurements
An overview of the study end points evaluated, and outcomes is presented in Table 1. No treatment-related clinical signs were observed, including findings of scabs, skin, or hair coat discoloration. These findings were noted primarily at injection site locations and attributed to blood collection and/or dosing procedure and not considered related to any of the tested vaccines because these findings occurred with similar frequency in animals given saline or noted initially prior to dosing.
Overview of Study End Points and Outcomes.a
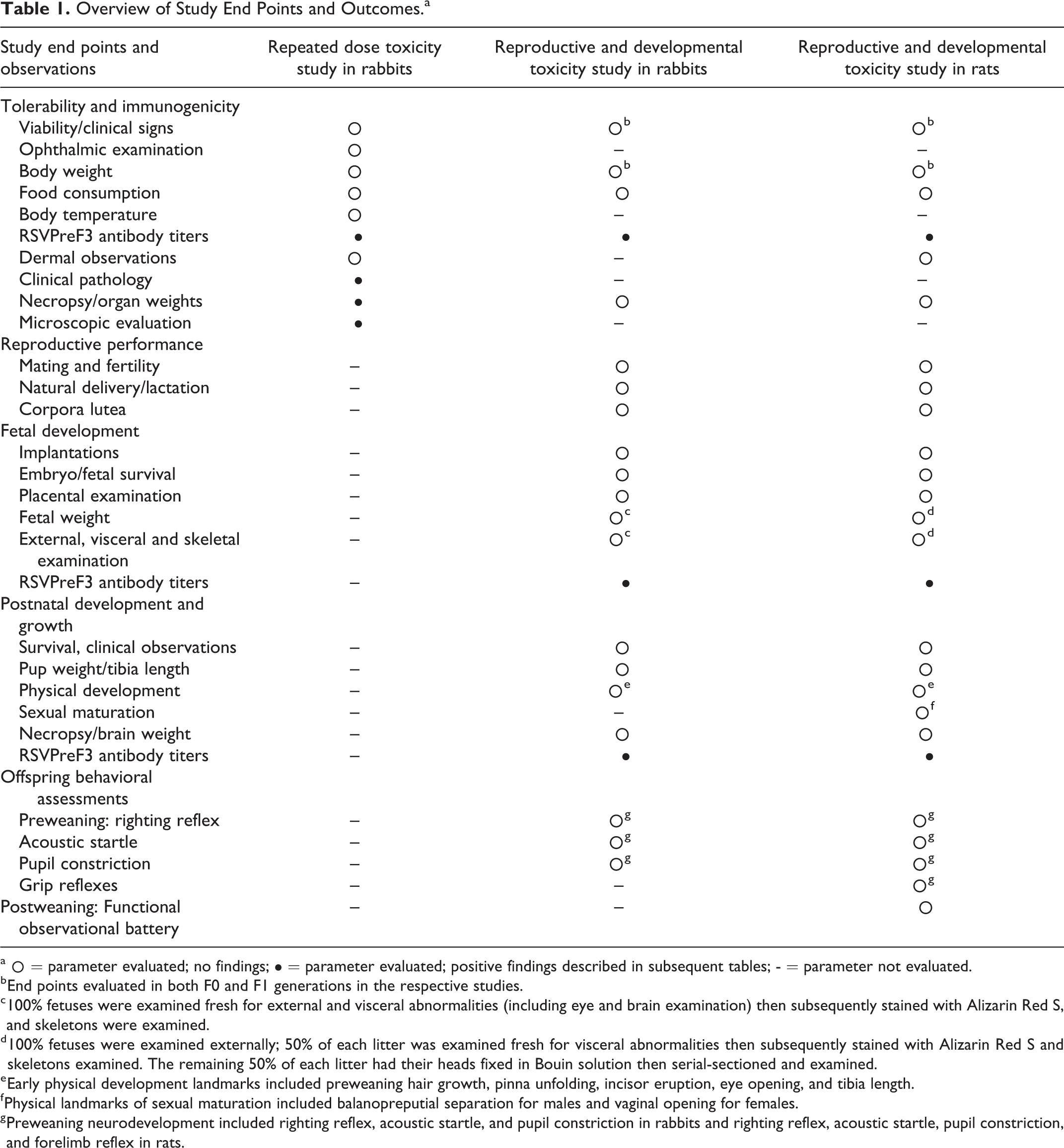
a ˆ = parameter evaluated; no findings; • = parameter evaluated; positive findings described in subsequent tables; - = parameter not evaluated.
b End points evaluated in both F0 and F1 generations in the respective studies.
c 100% fetuses were examined fresh for external and visceral abnormalities (including eye and brain examination) then subsequently stained with Alizarin Red S, and skeletons were examined.
d 100% fetuses were examined externally; 50% of each litter was examined fresh for visceral abnormalities then subsequently stained with Alizarin Red S and skeletons examined. The remaining 50% of each litter had their heads fixed in Bouin solution then serial-sectioned and examined.
e Early physical development landmarks included preweaning hair growth, pinna unfolding, incisor eruption, eye opening, and tibia length.
f Physical landmarks of sexual maturation included balanopreputial separation for males and vaginal opening for females.
g Preweaning neurodevelopment included righting reflex, acoustic startle, and pupil constriction in rabbits and righting reflex, acoustic startle, pupil constriction, and forelimb reflex in rats.
No treatment-related effects on ophthalmology, dermal observations, body weight, food consumption, or body temperature were observed. Animals given RSVPreF3/AS01 or RSVPreF3 + Boostrix had significant increases or decreases in body weight and body weight gain, when compared with the saline control group. These differences occurred periodically with no consistent pattern or trend and did not have impact overall on body weight gains over the 32-day dosing period or during the treatment-free period. Therefore, these differences were attributed to normal, nontest article-related variability in the data. Overall, no consistent pattern or trend was noted in body temperature changes following the administration of unadjuvanted RSVPreF3, adjuvanted RSVPreF3/AS01, or unadjuvanted RSVPreF3 + Boostrix when compared with the saline control group including occasional 40 °C or greater body temperature fluctuations (Figure 3). These changes were observed transiently, occurred primarily prior to dosing, and had no consistent trend related to treatment, gender, day of study, or duration of effect and therefore considered incidental and within the normal variation of data. One individual male given RSVPreF3/AS01 had a temperature of 40 °C, prior to dosing on day 15 and 40.1 °C 24 hours after dosing on day 16. Similar findings were not evident for this rabbit on the days prior to or after day 15/16, and as there were no other noteworthy changes that would suggest the cause to be treatment related, the slightly elevated temperature was considered unrelated to vaccination.
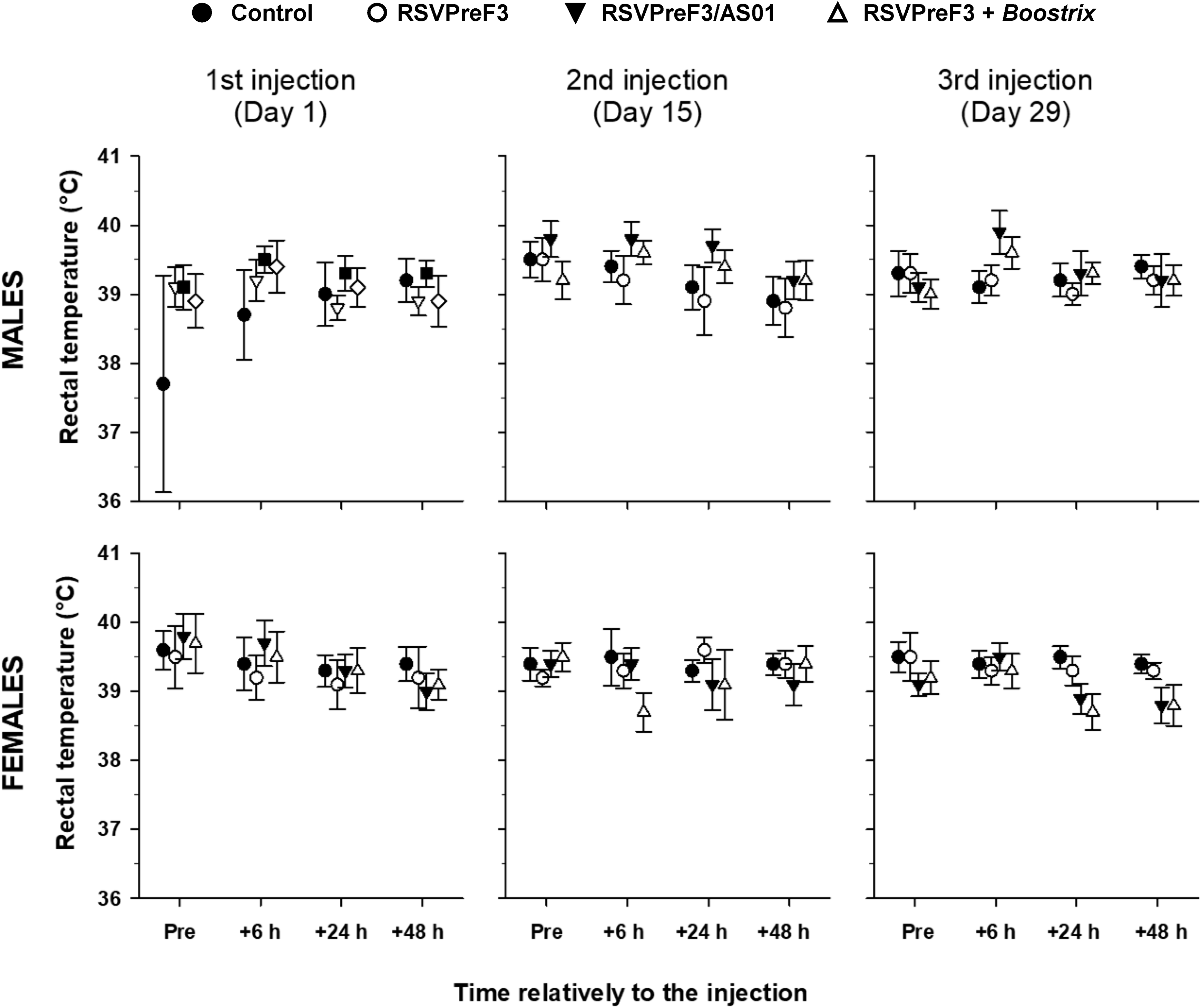
Repeated dose toxicity study. Body temperature in rabbits before (pre) and 6, 24, and 48 hours after the first, second, and third injection of saline control, RSVPreF3, RSVPreF3/AS01, or RSVPreF3+Boostrix formulations.
Test article-related clinical pathology findings consistent with inflammatory/acute phase response were observed on days 2 and 30 and were expected changes commonly observed with vaccine administration 43 ; these are summarized in Table 2. Clinical pathology findings for the unadjuvanted RSVPreF3 group were limited to female rabbits and included minimal increases in fibrinogen (1.2X, on day 2, with X representing the control value) and CRP concentration (3.3X, on day 30) and decreased A/G ratio (0.88X, on days 2 and 30). On day 2 and/or 30, rabbits given RSVPreF3/AS01 or RSVPreF3 + Boostrix showed increases in white blood cell (1.1X-1.4X) and absolute neutrophil (1.4X-2.2X) counts, fibrinogen (1.1X-1.6X, on day 2 and/or 30) and CRP concentrations (5.7X-35X and 2.7X-10X, respectively), and decreases in albumin (0.91X-0.98X) and A/G ratio (0.77X-0.94X) were noted on days 8 and 30. Minimally decreased calcium (0.93X-0.94X) on day 30 of the dosing phase in females may have been secondary to decreased albumin. These clinical pathology findings correlated with mixed cell infiltration and/or myofiber degeneration/necrosis at the injection site, noted microscopically. All findings exhibited reversibility by the end of the 4-week treatment-free period, except A/G ratio that exhibited partial reversibility but remained lower than control. Other differences from control, including values that were statistically significant, were characterized by small magnitude, inconsistency over time and/or between sexes, absence of correlative findings, and or similarity to differences present before initiation of dosing, and therefore were not considered related to any of the RSVPreF3 treatments.
Repeated-Dose Rabbit Study—List of Blood Parameters Most Affected by Treatment.a,b
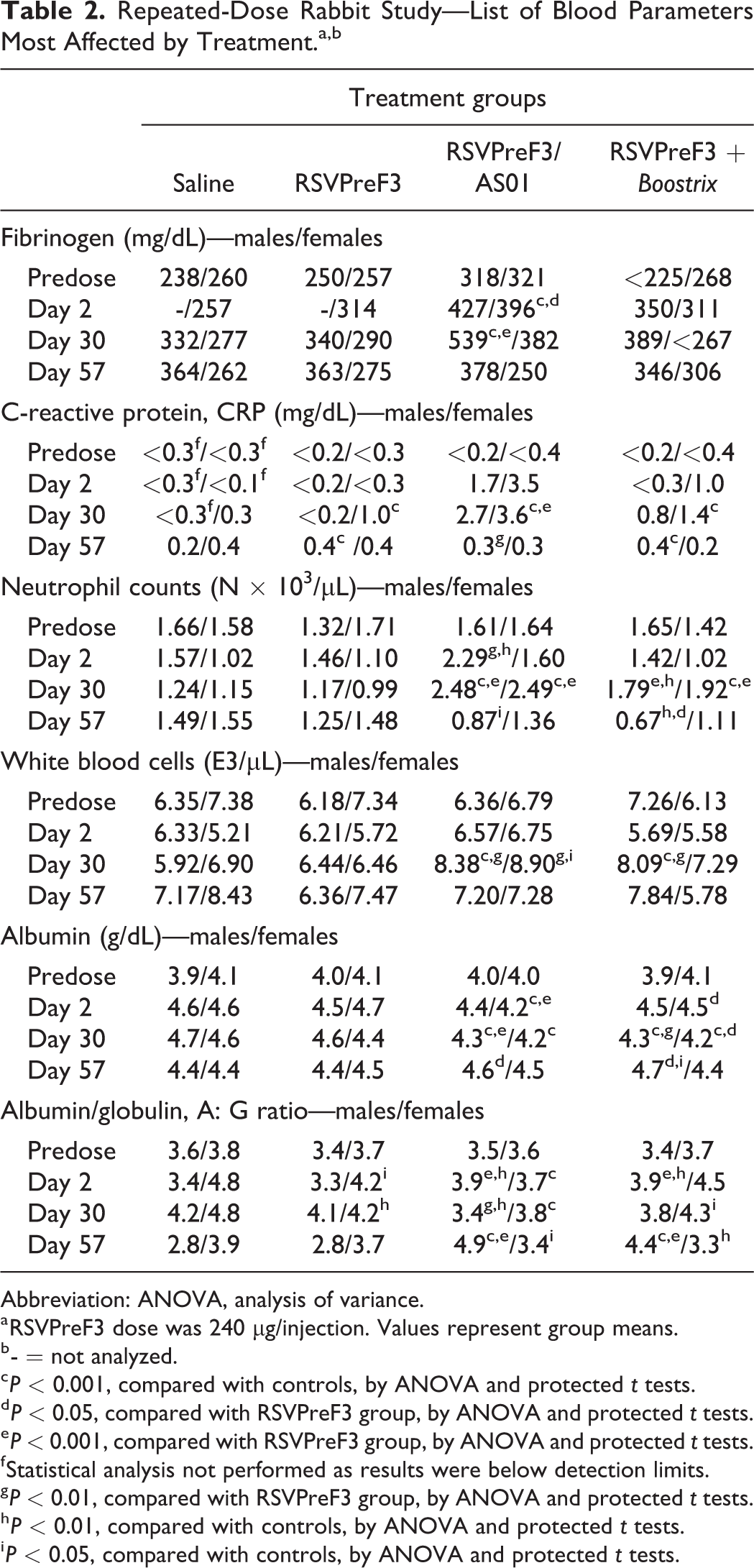
Abbreviation: ANOVA, analysis of variance.
a RSVPreF3 dose was 240 µg/injection. Values represent group means.
b - = not analyzed.
c P < 0.001, compared with controls, by ANOVA and protected t tests.
d P < 0.05, compared with RSVPreF3 group, by ANOVA and protected t tests.
e P < 0.001, compared with RSVPreF3 group, by ANOVA and protected t tests.
f Statistical analysis not performed as results were below detection limits.
g P < 0.01, compared with RSVPreF3 group, by ANOVA and protected t tests.
h P < 0.01, compared with controls, by ANOVA and protected t tests.
i P < 0.05, compared with controls, by ANOVA and protected t tests.
Test article-related microscopic findings are presented in Table 3 and were generally reversible and included minimal to moderate mixed cell infiltration and/or minimal to slight myofiber degeneration/necrosis at the injection sites in animals given any of the three RSVPreF3 vaccine presentations (unadjuvanted, adjuvanted with AS01, or coadministered with Boostrix) and/or the connective tissue around the sciatic nerve (RSVPreF3/AS01 or RSVPreF3 + Boostrix). Increased lymphocytes and neutrophils were also noted in the iliac lymph nodes that drained the vaccination site. Injection site reactions varied between animals but included a mixed cell infiltration of the subcutis and muscle with more severe changes in animals administered RSVPreF3/AS01 or RSVPreF3+Boostrix. In a few animals, mixed cell infiltration of slight or moderate severity was associated with minimal to slight myofiber degeneration/necrosis. The mixed cell infiltrates in the subcutaneous connective tissue surrounding the sciatic nerve and muscle were characterized by macrophages, granulocytic cells, and lymphocytes. Myofiber degeneration/necrosis was characterized by loss, swelling, and fragmentation of the myofibers. Additionally, a large internal iliac lymph node was noted for one rabbit each in the RSVPreF3/AS01 and RSVPreF3 + Boostrix groups, which correlated with the increased organ weights and/or the microscopic finding of increased lymphocytes. An increase in iliac lymph node weights was evident for all three RSVPreF3 vaccine groups with an increased incidence and severity in animals given RSVPreF3/AS01 or RSVPreF3 + Boostrix (up to 0.057 g in rabbits given RSVPreF3 alone, 0.095 g in rabbits given RSVPreF3/AS01, and 0.157 g in rabbits given RSVPreF3 + Boostrix, vs 0.031-0.038 g, control; Table 4). Following the 4-week off treatment period, iliac lymph nodes were macroscopically normal in all animals, consistent with full reversibility. All other differences in iliac lymph node organ weight parameters, including statistically significant values, were attributed to normal variation due to spurious occurrence, changes from controls that lacked biological significance, lack of dose dependency and/or incongruency across weight parameters.
Repeated-Dose Rabbit Study—Treatment-Related Microscopic Findings.a
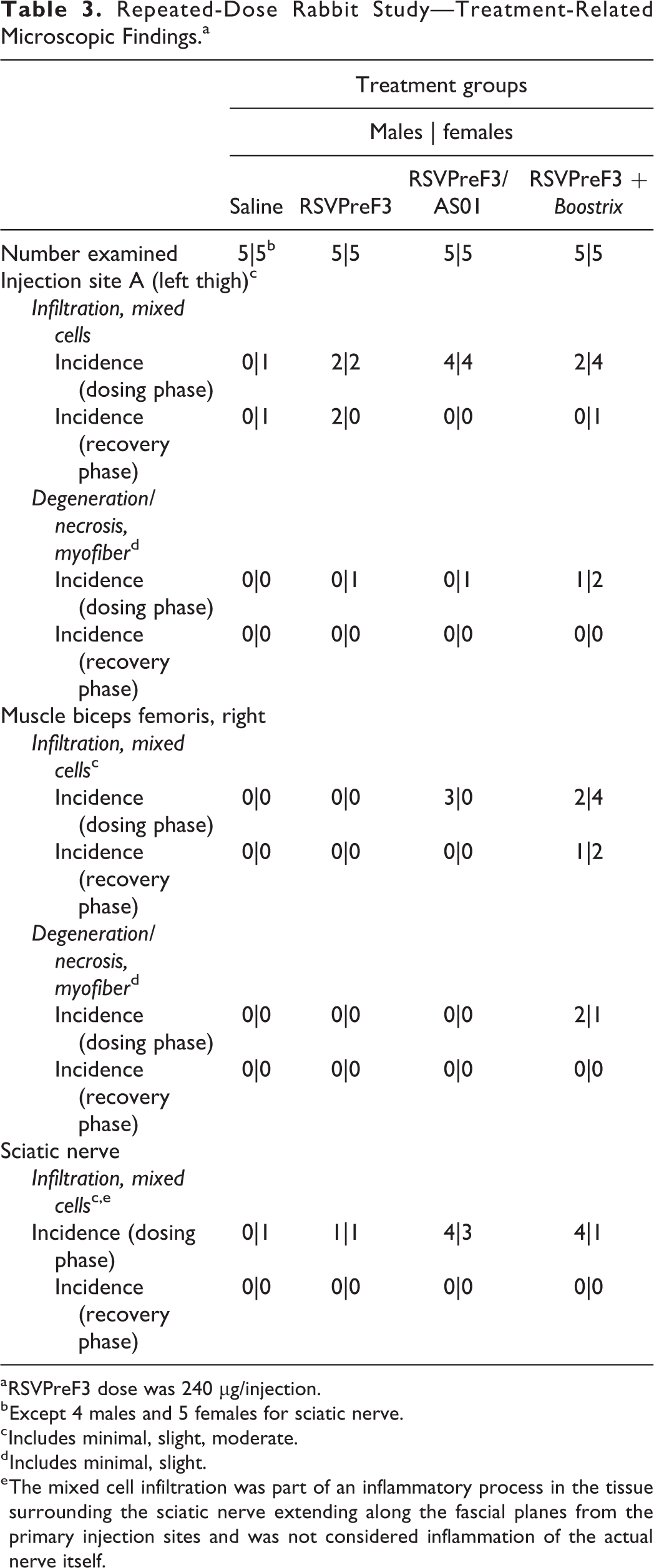
a RSVPreF3 dose was 240 µg/injection.
b Except 4 males and 5 females for sciatic nerve.
c Includes minimal, slight, moderate.
d Includes minimal, slight.
e The mixed cell infiltration was part of an inflammatory process in the tissue surrounding the sciatic nerve extending along the fascial planes from the primary injection sites and was not considered inflammation of the actual nerve itself.
Repeated-Dose Rabbit Study—Iliac Lymph Node Weight Changes at Terminal Necropsy.a
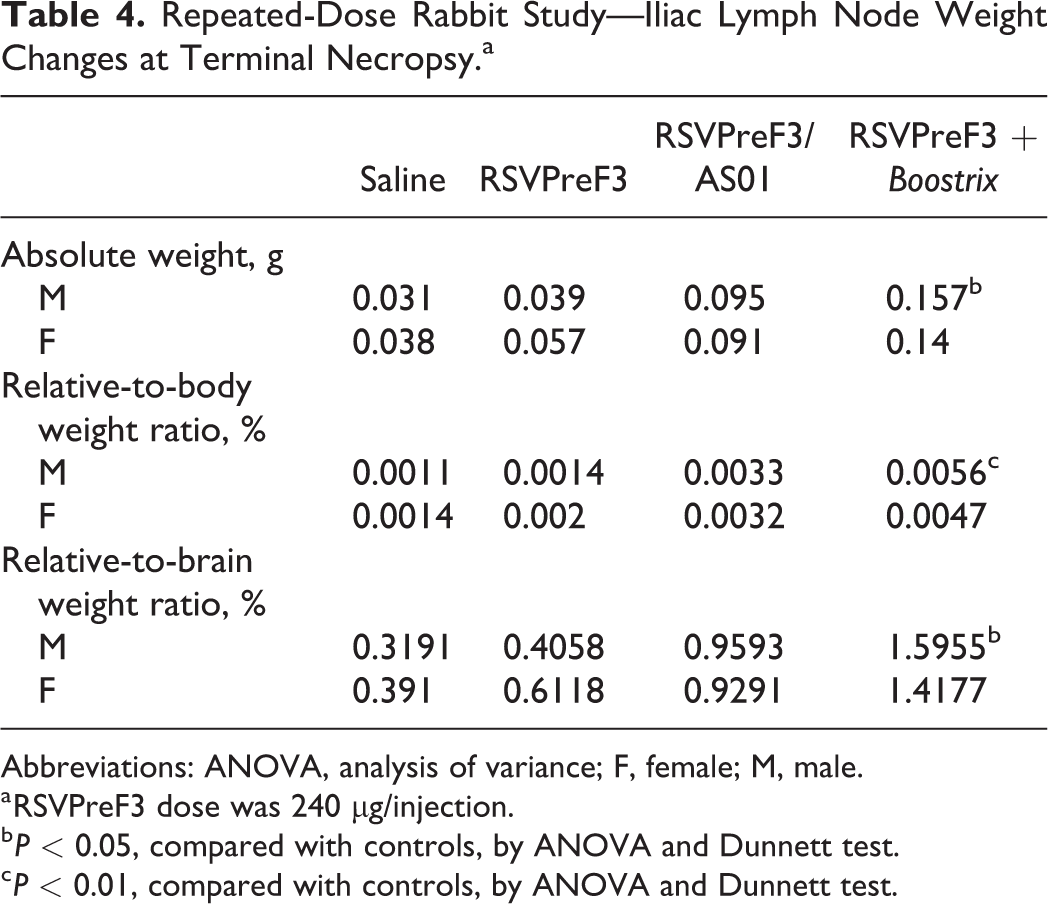
Abbreviations: ANOVA, analysis of variance; F, female; M, male.
a RSVPreF3 dose was 240 µg/injection.
b P < 0.05, compared with controls, by ANOVA and Dunnett test.
c P < 0.01, compared with controls, by ANOVA and Dunnett test.
Developmental and Reproductive Toxicity Studies in Rats and Rabbits
Serology
In the rats (Figure 4A), anti-RSVPreF3 IgG antibody titers were below the limit of detection (0.25 EU/mL) in all predose samples and in all animals (females, fetuses, and pups) in the saline control group at all sample collection time points, suggesting no pre-existing anti-RSVPreF3 immune responses and no incidental immunization in the course of the study. The majority of RSVPreF3-treated rats (31 of 48) had detectable anti-RSVPreF3 IgG titers after the first two injections, prior to mating. The titers ranged from 26.2 to 18,465.0 EU/mL. Of these females, 14 also had detectable anti-RSVPreF3 IgG titers during the gestation period and 16 had measurable titers during lactation. Sixteen females had no detectable anti-RSVPreF3 antibodies in the premating period but five of these animals had antibodies detected during the gestation period; three other females with no premating or gestational antibodies were not pregnant. Females with detectable IgG titers at DS24, GD21, and LD21 showed a mean titer of 2,302.90, 1,827.51, and 2,449.90 EU/mL, respectively. Passive immunity was observed with transfer of anti-RSVPreF3 antibodies from F0 to F1 animals. In the offspring, the majority of the pooled samples (10/18 litters) had detectable levels at GD21, ranging from 61.3 to 2,953.0 EU/mL. At PND21 (14 days after the last maternal immunization), 17 of 22 litters had detectable levels of anti-RSVPreF3 antibodies ranging from 53.5 to 44,086.0 EU/mL. As would be expected, F1 offspring from nonseroconverted F0 females had no detectable IgG against RSVPreF3.
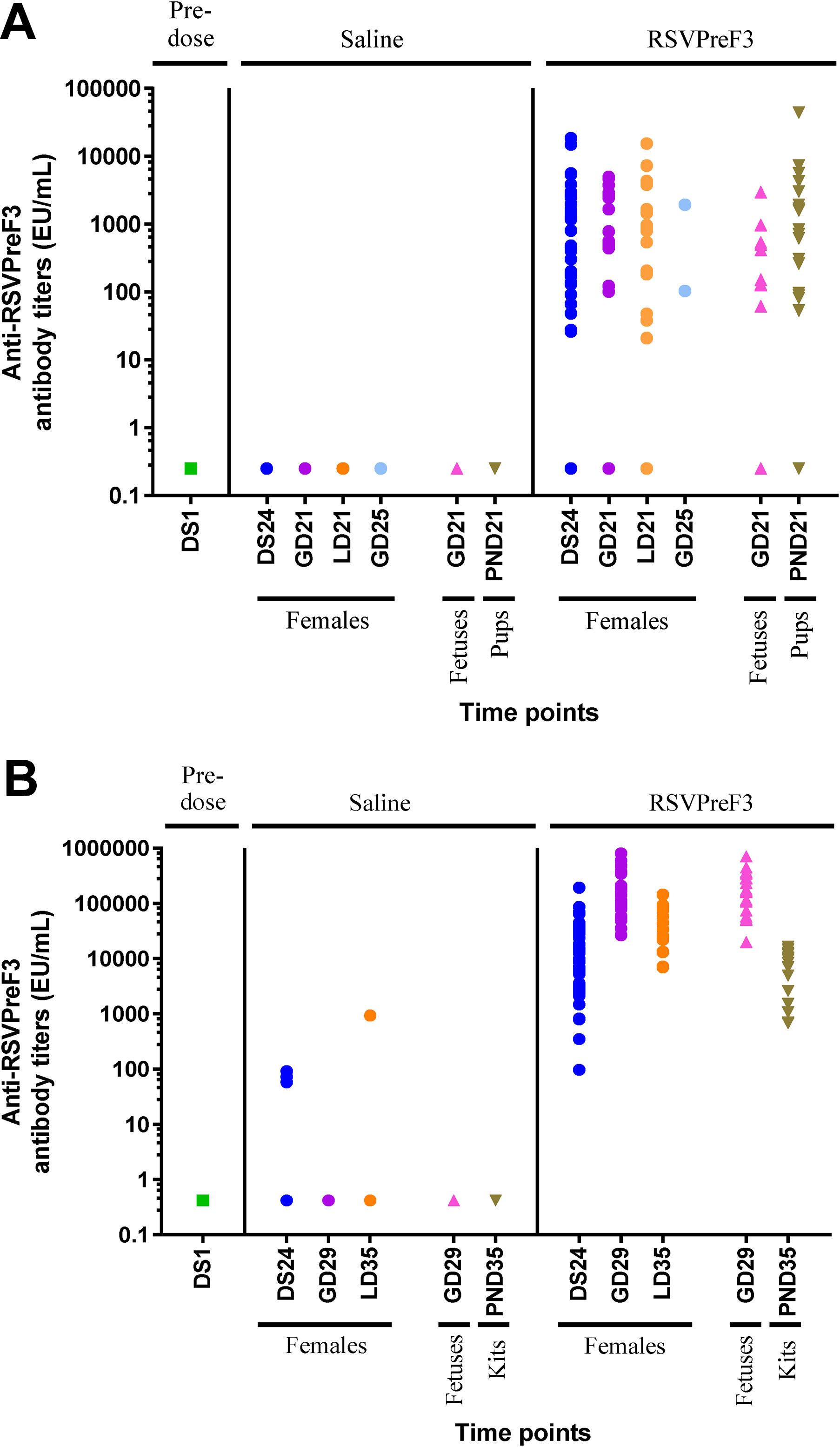
Reproductive and developmental toxicity studies in rats (A) and rabbits (B). Animal serum was collected before immunization (DS1, which was 28 days prior to mating) and at several time points as indicated in the figure. Anti-RSVPreF3 antibody titers in rabbit serum were measured by ELISA. Each symbol represents the antibody titer from a single animal or pool of animals (fetuses and pups/kits). DS indicates day of study; ELISA, enzyme-linked immunosorbent assay; GD, gestation day; LD, lactation day; PND, postnatal day.
In the rabbits (Figure 4B), anti-RSVPreF3 IgG antibody titers were below the limit of detection in all predose samples. All rabbits given 120 µg/injection had anti-RSVPreF3 IgG antibody titers above the lowest limit of detection (0.42 EU/mL), indicating all animals had generated an IgG response upon vaccination (ie, 100% seroconversion). Median titers in rabbits injected with the RSVPreF3 vaccine were 11,237 EU/mL (range: 97.57-194,091 EU/mL) on DS24, 128,037 EU/mL (range: 26,724-802,027 EU/mL) on GD29, and 34,341 EU/mL (range: 7,031-292,303 EU/mL) on LD35. These were nine, five, and 28 days after the previous vaccination, respectively. As reported earlier for the rats, the anti-RSVPreF3 IgG titers in rabbits were detectable throughout critical periods of the reproductive cycle. The mean titer values of the fetal samples and their corresponding mother were comparable at GD29 (geometric mean: mother: 145,873 EU/mL; fetus: 159,228 EU/mL), indicating the passive transfer of antibodies to the fetus. Mean titer values for the offspring were lower compared with those of their mothers on LD35 (mother: 39,405 EU/mL; offspring: 5,481 EU/mL) but was indicative of passive transfer of maternal antibodies to F1 animals.
In-life observations and measurements
An overview of the evaluated end points and outcomes is presented in Table 1. There were no RSVPreF3 vaccine-related effects compared with the concurrent saline control group on clinical observations, dermal irritation at the injection sites, mean body weights, mean body weight gains, food consumption, organ weights, and necropsy observations at the end of gestation or lactation periods.
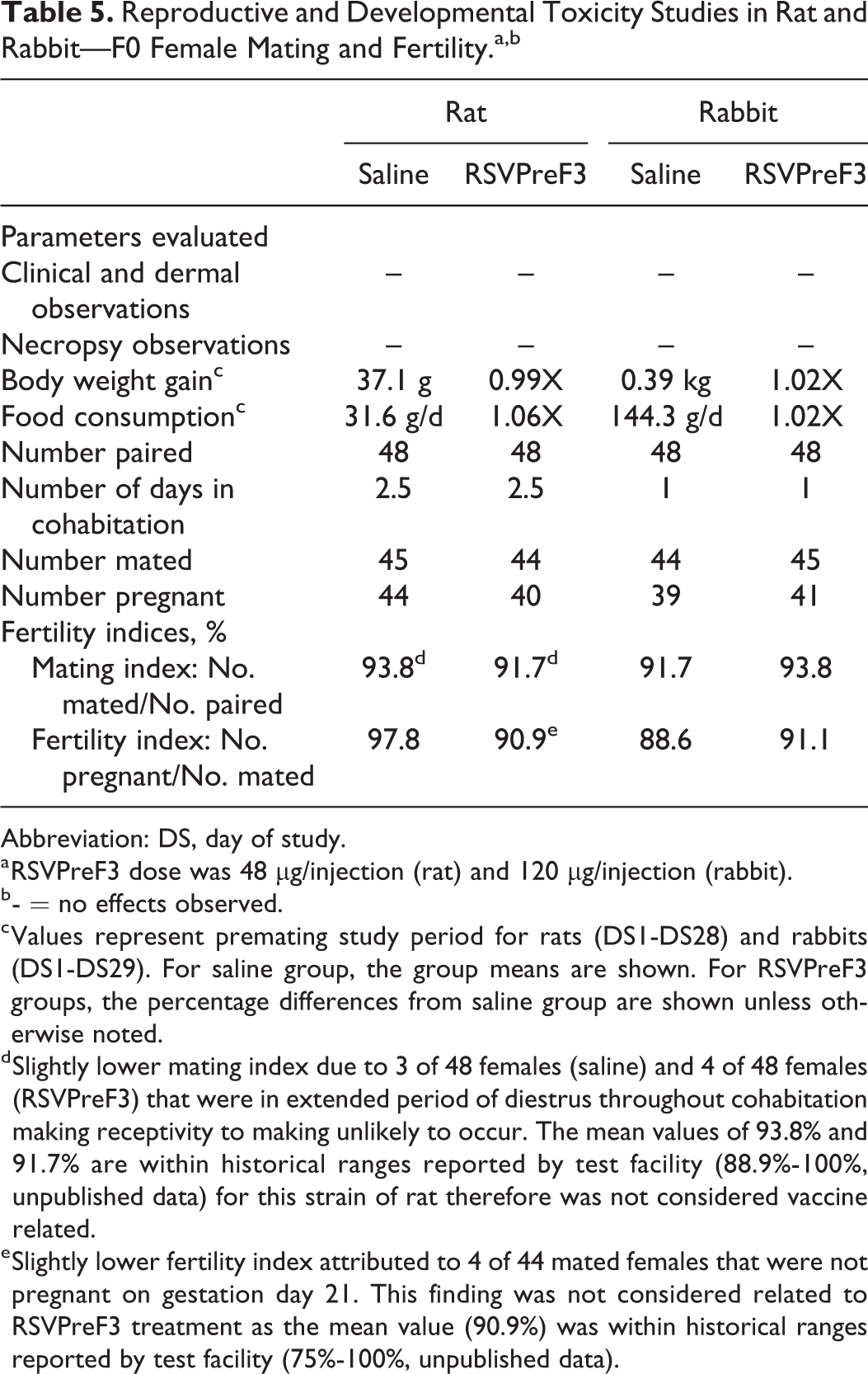
There were no effects of RSVPreF3 immunization on female mating or fertility (Table 5). The number of animals that mated was 93.8% and 91.7% for rats (control and 48 µg RSVPreF3, respectively) and 91.7% and 93.8% for rabbits (control and 120 µg RSVPreF3, respectively). The number of fertile pregnancies in these same groups was 97.8% and 90.9% (rat) and 88.6% and 91.1% (rabbit), respectively. The relatively lower mating index for the female rats in the RSVPreF3 group was not thought to be caused by the vaccine but rather that 4 of 48 females were in a persistent diestrus phase of their estrous cycle throughout the mating period, making receptivity to copulation less likely. A similar number of control females (three of 48 female rats) were noted with identical findings (5-day diestrus phase of estrous cycle and no positive sign of mating after 5 days in cohabitation). Uterine examination at the time of necropsy confirmed that mating had not occurred for any of these females. It should also be noted that a group value of 91.7% mating index is considered normal based on historical ranges reported by the testing facility for the Han Wistar strain of rat (mating index: 88.9%-100%). Four of 44 RSVPreF3 female rats that showed evidence of insemination after just one day in cohabitation (two females had sperm present in a vaginal lavage and two females had presence of intravaginal copulatory plugs) were determined not to be pregnant at necropsy. The lower number of pregnant females in this group was considered unrelated to RSVPreF3 because a fertility index of 90.9% is within normal background data for this strain of rat based on historical ranges reported by the test facility (fertility index: 75%-100%) and because there was no correlation between the antibody titers for RSVPreF3 and pregnancy status to suggest a possible immune-mediated adverse effect on pregnancy.
Reproductive and Developmental Toxicity Studies in Rat and Rabbit—F0 Female Mating and Fertility.a,b
Abbreviation: DS, day of study.
a RSVPreF3 dose was 48 µg/injection (rat) and 120 µg/injection (rabbit).
b - = no effects observed.
c Values represent premating study period for rats (DS1-DS28) and rabbits (DS1-DS29). For saline group, the group means are shown. For RSVPreF3 groups, the percentage differences from saline group are shown unless otherwise noted.
d Slightly lower mating index due to 3 of 48 females (saline) and 4 of 48 females (RSVPreF3) that were in extended period of diestrus throughout cohabitation making receptivity to making unlikely to occur. The mean values of 93.8% and 91.7% are within historical ranges reported by test facility (88.9%-100%, unpublished data) for this strain of rat therefore was not considered vaccine related.
e Slightly lower fertility index attributed to 4 of 44 mated females that were not pregnant on gestation day 21. This finding was not considered related to RSVPreF3 treatment as the mean value (90.9%) was within historical ranges reported by test facility (75%-100%, unpublished data).
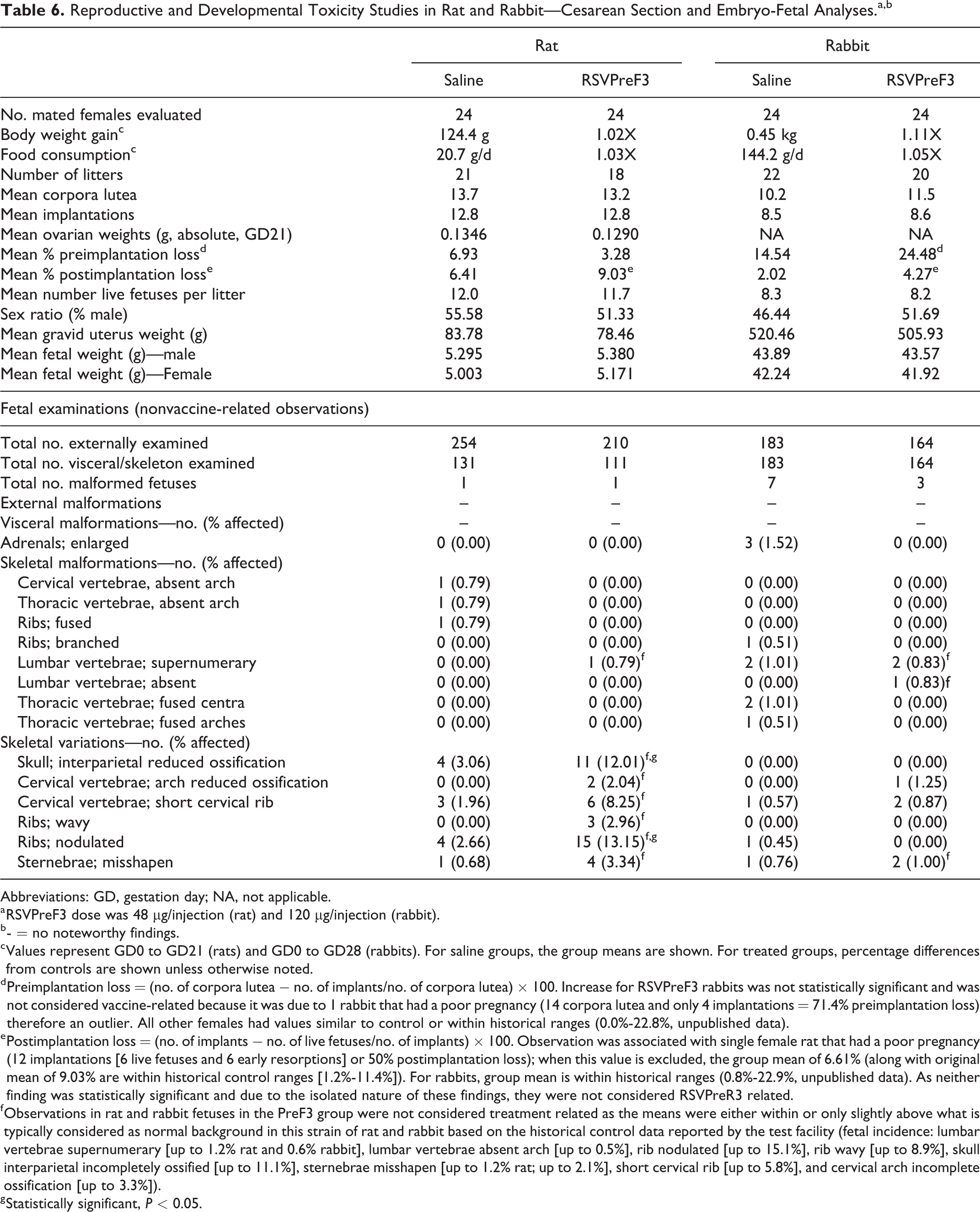
Likewise, RSVPreF3 did not induce any effect on cesarean delivery parameters including corpora lutea, implantations, pre- and postimplantation loss, live fetuses per litter, sex ratio, fetal body weight, gravid uterine weight, or placental morphology in either pregnant rats or rabbits (Table 6). Slightly higher group preimplantation loss value was evident for rabbits (24.48% vs 14.54% for the control, not statistically significant) and was due to one female that had an individual value of 71.4% (total of 14 corpora lutea and only four implantations) compared with other females that had values within concurrent and historical control ranges. If data for this female are excluded from analysis due to the isolated nature of the observation, the group mean is reduced to 22.0% and is within historical control ranges for the laboratory (0.0%-22.8%). Additionally, published literature supports a correlation between high numbers of corpora lutea and preimplantation loss. 44 In both rats and rabbits, slightly higher postimplantation loss was evident (rat: 9.03% vs 6.41% for the control; rabbit: 4.27% vs 2.02% for the control). For rats, this observation was associated with a single female that had a poor pregnancy (litter consisted of 12 implantations [six early embryonic losses and six live fetuses or 50% postimplantation loss]) and when these data are excluded from analyses because of the isolated nature of the observation, the group mean value is reduced to 6.61%; both 9.03% and 6.61% values are within test facility historical control data (1.2%-11.4%; unpublished data). The individual postimplantation loss values for the majority of rabbits were similar to concurrent controls and were within expected historical ranges (0.8%-22.9%; unpublished data). Furthermore, as results were not statistically significant and did not translate to lower numbers of live fetuses per litter in either study, the minor differences from the saline control group were not attributable to RSVPreF3 immunization.
Reproductive and Developmental Toxicity Studies in Rat and Rabbit—Cesarean Section and Embryo-Fetal Analyses.a,b
Abbreviations: GD, gestation day; NA, not applicable.
a RSVPreF3 dose was 48 µg/injection (rat) and 120 µg/injection (rabbit).
b - = no noteworthy findings.
c Values represent GD0 to GD21 (rats) and GD0 to GD28 (rabbits). For saline groups, the group means are shown. For treated groups, percentage differences from controls are shown unless otherwise noted.
d Preimplantation loss = (no. of corpora lutea − no. of implants/no. of corpora lutea) × 100. Increase for RSVPreF3 rabbits was not statistically significant and was not considered vaccine-related because it was due to 1 rabbit that had a poor pregnancy (14 corpora lutea and only 4 implantations = 71.4% preimplantation loss) therefore an outlier. All other females had values similar to control or within historical ranges (0.0%-22.8%, unpublished data).
e Postimplantation loss = (no. of implants − no. of live fetuses/no. of implants) × 100. Observation was associated with single female rat that had a poor pregnancy (12 implantations [6 live fetuses and 6 early resorptions] or 50% postimplantation loss); when this value is excluded, the group mean of 6.61% (along with original mean of 9.03% are within historical control ranges [1.2%-11.4%]). For rabbits, group mean is within historical ranges (0.8%-22.9%, unpublished data). As neither finding was statistically significant and due to the isolated nature of these findings, they were not considered RSVPreR3 related.
f Observations in rat and rabbit fetuses in the PreF3 group were not considered treatment related as the means were either within or only slightly above what is typically considered as normal background in this strain of rat and rabbit based on the historical control data reported by the test facility (fetal incidence: lumbar vertebrae supernumerary [up to 1.2% rat and 0.6% rabbit], lumbar vertebrae absent arch [up to 0.5%], rib nodulated [up to 15.1%], rib wavy [up to 8.9%], skull interparietal incompletely ossified [up to 11.1%], sternebrae misshapen [up to 1.2% rat; up to 2.1%], short cervical rib [up to 5.8%], and cervical arch incomplete ossification [up to 3.3%]).
g Statistically significant, P < 0.05.
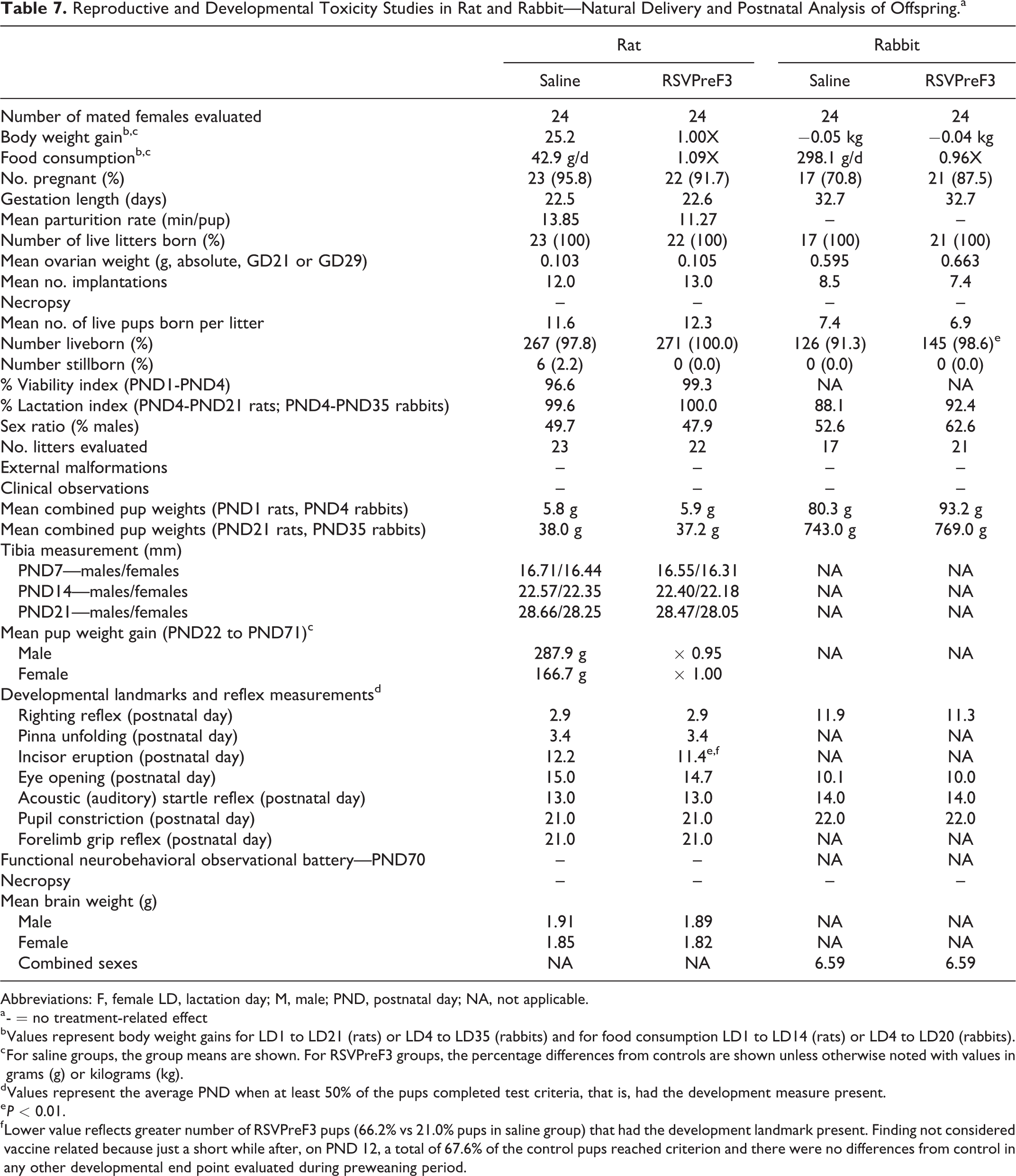
For pregnant rats and rabbits that naturally delivered their litters, no RSVPreF3-related effects were observed on gestation length, natural delivery, litter observations, or lactation (Table 7). Specifically, the mean values for the number of mated females that delivered live litters, the duration of gestation, parturition time, number of implantation sites per delivered litter, and live birth weight were similar between the RSVPreF3 and control groups.
Reproductive and Developmental Toxicity Studies in Rat and Rabbit—Natural Delivery and Postnatal Analysis of Offspring.a
Abbreviations: F, female LD, lactation day; M, male; PND, postnatal day; NA, not applicable.
a - = no treatment-related effect
b Values represent body weight gains for LD1 to LD21 (rats) or LD4 to LD35 (rabbits) and for food consumption LD1 to LD14 (rats) or LD4 to LD20 (rabbits).
c For saline groups, the group means are shown. For RSVPreF3 groups, the percentage differences from controls are shown unless otherwise noted with values in grams (g) or kilograms (kg).
d Values represent the average PND when at least 50% of the pups completed test criteria, that is, had the development measure present.
e P < 0.01.
f Lower value reflects greater number of RSVPreF3 pups (66.2% vs 21.0% pups in saline group) that had the development landmark present. Finding not considered vaccine related because just a short while after, on PND 12, a total of 67.6% of the control pups reached criterion and there were no differences from control in any other developmental end point evaluated during preweaning period.
The RSVPreF3 vaccine was not teratogenic (Table 6). A low incidence of malformations affecting primarily axial skeleton development was observed for RSVPreF3 and control rat and rabbit fetuses alike and included observations of absent cervical and thoracic vertebral arches as well as fused ribs (one control rat fetus), supernumerary lumbar vertebrae (one RSV rat fetus), supernumerary lumbar vertebrae (and enlarged adrenals) in the same fetuses (up to three control rabbit fetuses), fused thoracic vertebrae with and without branched ribs (up to two control rabbit fetuses), absent lumbar vertebra (one RSVPreF3 rabbit fetus), and supernumerary lumbar vertebrae (two RSVPreF3 rabbit fetuses). In addition, a small number of skeletal variations (reduced ossification of interparietal skull bones or cervical vertebrae, nodulated or wavy ribs, and misshapen sternebrae) that are common to the Han Wistar rat occurred at a higher frequency in the RSVPreF3 group than in control. All observed malformations and variations were considered unrelated to RSVPreF3 treatment because the abnormality was limited to a single fetus or occurred at a similar incidence as that reported for the control group or because the litter and/or fetal incidence was within the expected background range based on laboratory historical control data.
Just as there was no RSVPreF3 vaccine-related effect on embryo-fetal development and survival among cesarean delivered litters, all of the natural delivered pups appeared normal at birth and their viability was 99.3% or higher (PND1-PND21) and 92.4% (PND4-PND35) during the preweaning period for rats and rabbits, respectively.
There were no latent effects of maternal RSVPreF3 treatment on the postnatal growth and development of rat and rabbit offspring prior to weaning (Table 7). This is supported by mean body weights and tibia measurements (rats only) that were similar to or higher than control pups during this period. In addition, there were no RSVPreF3-related effects on reflex and physical development, as the average postnatal ages in which 50% of rat and/or rabbit pups in each group achieved testing criterion for hair growth, incisor eruption, surface/air righting, pinna unfolding, eye opening, acoustic (auditory) startle response, forelimb grip reflex, and pupil constriction response were similar to control, except for incisor eruption in rats. Incisor eruption among rat pups in the RSVPreF3 vaccine group occurred on PND11.4 in 66.2% of the population compared with 67.6% of control pups that did not reach testing criterion until the following day on PND12.2. Although this finding was statistically significant (P ≤ 0.01), it was not considered related to the RSVPreF3 vaccine because there were no differences from control either in the time it took for incisors to erupt in all remaining pups in each group or in the timing of any other developmental end point evaluated during the preweaning period.
During the postweaning period, an assessment of growth and development of juvenile rats was performed frequently via daily checks for clinical signs, periodic weekly body weight measurements, attainment of physical sexual maturity, and functional neurobehavioral testing. There were no noteworthy differences from control in any of these end points. However, a statistically significant decrease in mean body weight gain (data not shown) was noted for F1 male offspring in the RSVPreF3 group during PND50 to PND57 (0.94X of control; P ≤ 0.05); a similar finding was not observed at any of the other six intervals evaluated for males prior to PND50 or after PND57 and was not evident among F1 female offspring at any time during the study. Thus, the finding was not considered toxicologically relevant. There were no RSVPreF3-related effects on sexual maturation as the age of attainment for balanopreputial separation for male rats (44.2 vs 44.7 days for the control) and vaginal patency in female rats (30.6 vs 30.4 days for the control) were similar to those of control.
On PND70, just before euthanasia, the F1 rats were subjected to a battery of functional neurobehavioral tests. Reported values for home cage and open-field behaviors, response to routine handling, tactile, auditory, and visual stimulation were all normal in both groups. The animals had the appearance of being well groomed with no abnormal eye or oral secretions and no alterations in body temperature, respiration, or mobility; and no abnormalities in various reflexes or grip strength were noticed in the RSVPreF3 vaccine-treated animals, when compared with controls.
Upon necropsy at scheduled euthanasia, no differences were noted between RSVPreF3-treated animals and controls. The absolute brain weights (male: 1.89 vs 1.91 g for the control; female: 1.82 vs 1.85 g for the control) or the ratio of brain weights to terminal body weights in the F1 generation males or females were not statistically distinguishable.
Discussion
An RSV vaccine candidate containing the prefusion antigen RSVPreF3 was evaluated in a repeated dose toxicity study in rabbits and DART studies in rats and rabbits. The repeated dose study consisted of three IM administrations at 2-week intervals in New Zealand white rabbits. The rabbit was used since this species is recommended by European Medicines Agency and WHO for these types of toxicity studies. 45,46 In addition, the rabbit can be injected with a full human dose volume of the candidate vaccine (ie, 0.5 mL). The RSVPreF3 antigen was evaluated unadjuvanted, with AS01 since this adjuvant was used with RSVPreF3 in clinical trials in older adults, or was administered concomitantly with Boostrix, a vaccine potentially given at the same time as nonadjuvanted RSVPreF3 vaccine to pregnant mothers in the third trimester of pregnancy.
The administration of any of the three RSVPreF3 vaccine presentations (ie, unadjuvanted, adjuvanted with AS01, or coadministered with Boostrix) to males and female rabbits did not impact body weight, body temperature, or food consumption. Findings related to vaccine-induced inflammatory reaction included transient increases in neutrophil counts, fibrinogen, and CRP levels on the day following vaccine administration. The CRP levels were the highest in the AS01-adjuvanted RSVPreF3 group and were not unexpected as similar findings have been observed with another AS01-adjuvanted vaccine. 47 Additionally, CRP elevation occurred when AS01 was injected by itself in rabbits without the presence of an antigen 47 and in an earlier repeated dose toxicity study, CRP elevations of the same magnitude as in the present study occurred after administration of a lower dose of RSVPreF3 (ie, 120 µg) adjuvanted with AS01 (data not shown). These observations provide evidence to the contribution AS01 plays in the induction of CRP upon vaccination with the adjuvanted RSVPreF3 vaccine.
In the present repeated dose toxicity study, vaccine-induced inflammatory reactions were noted upon microscopic examination, which included the presence of inflammatory cell infiltrates at the injection site and increased weight of the draining iliac lymph nodes, which were evident three days after the third injection. All findings were transient and were either resolved or showed evidence of reversibility by the end of the 4-week treatment-free period. For these observations, the effects were minimal or absent in the animals given the nonadjuvanted RSVPreF3 vaccine. Additionally, no interference in the immune response was noted between the RSVPreF3 vaccine and Boostrix when the two vaccines were coadministered. However, the animals that received the coadministration of the RSVPreF3 vaccine and Boostrix showed signs of increased immune/inflammatory responses following the vaccines coadministration, which is expected since two immunogenic vaccines were administered instead of one.
Since pregnant women in their third trimester and possibly women who intend to become pregnant could receive RSVPreF3 vaccination to facilitate passive transfer of antibodies to their offspring, the potential effects of RSVPreF3 administration on pre- and postnatal development of the offspring are of concern. Potential risks of immunization during pregnancy could include adverse developmental effects caused by the biological activity of the vaccine antigen or any of its components or as a result of maternal immune response. Any component of a vaccine formulation (adjuvant, excipients) may potentially directly exert deleterious effects on the development of the embryo/fetus. In addition, indirectly, it remains possible that vaccine-elicited antibodies cross-react with endogenous tissue through molecular mimicry, which may be critical if the specific off-target interaction plays an active role in fetal development. 48 Therefore, to support the clinical development of the RSVPreF3 vaccine, GSK carried out DART studies in rats and rabbits in which the potential effects of vaccine administration on female fertility, pregnancy/lactation, and pre- and postnatal development of the offspring following exposure from time of implantation through the end of pregnancy and continuing with postnatal assessment of offspring were studied. These species were chosen because they are widely accepted by regulatory agencies for such studies due to the extensive historical background data that exist on embryo-fetal development. 49 Serology results showed that placental antibody transfer was effective, with high levels of anti-RSVPreF3 antibodies detected in both fetuses and pups.
Treatment of females with RSVPreF3 for approximately one month prior to cohabitation with untreated males did not affect mating performance or fertility. Further, the growth, development, and survival of embryos were unaffected by RSVPreF3 exposure during gestation, based on the absence of embryo-fetal loss and alterations to external, visceral, and skeletal development, as well as absence of effects on normal physical growth and neurobehavioral development of offspring exposed to RSVPreF3 through lactation via mothers’ milk.
In conclusion, under the conditions of these studies, repeated administration of the investigational RSVPreF3 vaccine, either unadjuvanted, adjuvanted with AS01, or co-administered with Boostrix, was locally and systemically well-tolerated, with only transient signs of inflammatory reactions. No interference or any signs of potential synergistic effects were noted between the two vaccines, RSVPreF3 and Boostrix, that would prevent them from being administered concomitantly. Finally, injections of RSVPreF3 with a dose representing 80-fold (rats) or 20-fold (rabbits) the highest tested maternal immunization dose (ie, 120 µg) relative to body weight did not adversely affect female fertility, natural delivery or lactation, or pre- or postnatal development in rats and rabbits. These data support the continued clinical investigations of the RSVPreF3 vaccine as a maternal immunization and for administration to older adults.
Footnotes
Acknowledgments
We thank Pascal Cadot (GSK, Rixensart, Belgium) who provided scientific writing services in the manuscript development. Robert Lin (GSK, Rockville) coordinated the manuscript development. The authors would like to thank Ronan Rouxel, Rachel Thornley, and Corey Mallett for critical review of the manuscript.
Author Contributions
Stokes, A. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Franklin, K. contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Fisher, D. contributed to design, contributed to analysis and interpretation, and critically revised manuscript; Posobiec, L. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Binazon, O. contributed to design, contributed to analysis and interpretation, and critically revised manuscript; Tripathi, N. contributed to analysis and interpretation and critically revised manuscript; Ringenberg, M. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Charlap, J. contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Ziejewski, M. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Vemireddi, V. contributed to analysis and interpretation and critically revised manuscript; Weiss, P. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Majundar, R. contributed to acquisition, analysis, and interpretation and critically revised manuscript; Bouzya, B. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Donner, M.-N. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Rodriguez, L.-A. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Baumiester, J. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AHS, MAR, BB, M-ND, MKZ, JB, LP, L-AR, and OB are, or were at the time of the study, employees of the GSK group of companies. AHS, L-AR and MAR report ownership of GSK stocks. KF, NT, VV, PWK, and RM are/were employees of Covance Laboratories, Inc, a Contract Research Organization contracted by GSK in the context of the repeated dose toxicity study. DEF and JC are employees of the Charles River Laboratories, a Contract Research Organization contracted by GSK in the context of the developmental and reproductive toxicity studies. Boostrix is a trademark of the GSK group of companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored and financially supported by GlaxoSmithKline Biologicals, SA. The costs associated with the development and publishing of the manuscript were also covered by GlaxoSmithKline Biologicals SA.
