Abstract
Bovine respiratory syncytial virus (BRSV) is associated with severe respiratory disease in cattle. BRSV infection frequently leads to the death of young infected animals. The presence of BRSV in postmortem specimens is routinely detected using indirect immunofluorescence (IIF). However, this technique requires special equipment and considerable expertise. The present paper describes the development of a 1-step ELISA for rapid (1.5 hours) detection of BRSV antigen in organ homogenates. The performance of the new 1-step ELISA was evaluated using bovine postmortem specimens (n = 108) in comparison with 3 other BRSV diagnostic techniques: indirect immunofluorescence, the Clearview respiratory syncytial virus (RSV) test, and real-time reverse transcriptase polymerase chain reaction (RT-PCR). The relative sensitivity, specificity, and the kappa coefficient of 1-step ELISA, the Clearview RSV electroimmunoassay (EIA), and IIF were calculated, using real-time RT-PCR as the reference test. The new 1-step ELISA was the most sensitive and specific of the 3 tests. Thus, the new 1-step ELISA is a reliable test for detecting BRSV antigen in organ homogenates.
Introduction
Bovine respiratory syncytial virus (BRSV) is a pneumovirus of the Paramyxoviridae family. 5 This virus is transmitted by aerosols and infects respiratory tract mucosal cells. The clinical signs of BRSV infection are associated with pulmonary localization of the virus, which is mainly in the cranio-ventral lobe of the lung. 16 , 20 BRSV infection causes severe respiratory signs in young cattle and frequently leads to the death of the infected animal. 2 , 11 , 17 , 19 , 20 BRSV infection is widespread and has a major economic impact. The isolation of BRSV in clinically affected animals using conventional cell cultures is challenging. Postmortem diagnosis of BRSV infection is done either by virus isolation on cell monolayers using organ homogenates or, more frequently, by using indirect immunofluorescence (IIF) on acetone-fixed organ cryosections. 15 BRSV is labile even in optimally stored samples; thus, viral isolation attempts are often unsuccessful. 10 , 18 On the other hand, IIF on organ cryosections is less dependent on transport or storage conditions. However, because the IIF technique uses only a small portion of the entire organ, the infected area can be missed. Furthermore, IIF requires special equipment (cryostat and fluorescent microscope) and considerable expertise. 12 , 18 Diagnostic methods such as ELISA, virus isolation, or real-time reverse transcriptase polymerase chain reaction (RT-PCR) that use organ homogenates extend the analysis to larger parts of the sample.
Recently, a real-time RT-PCR assay was developed for BRSV diagnosis and has yielded very good results. 1 , 3 , 7 , 8 However, at present, some veterinary diagnostic laboratories are not yet equipped to perform real-time PCR routinely. Thus, there is a need for sensitive and rapid BRSV diagnostic methods for routine use on postmortem specimens. ELISA is simple to perform and to interpret. The cost, reliability, and ease-of-use of ELISA have made it a widely used method in both veterinary and human diagnostics. 4 Unfortunately, most of the commercially available BRSV ELISA kits involve a long and complex procedure or lack sensitivity. 12 The purpose of this study was to develop and validate a new 1-step ELISA method for BRSV diagnosis using postmortem specimens.
Material and methods
Antibodies. AK13A2 is a mouse monoclonal antibody (MAb) of IgG1 isotype that recognizes the F protein of human respiratory syncytial virus (HRSV). The antibody was obtained by immunizing Balb/c mice with the Long strain (LS) subtype A of HRSV, as previously described. 14 AK13A2 reacts with the F protein of several RSV species, including BRSV strain RB94. 14 The MAb was purified on a protein G-sepharose column. Ten milligrams of the purified antibody were covalently linked to horse radish peroxidase (HRPO), as previously described 9 and another 10 mg of the purified antibody were linked to biotin using a biotinylation procedure according to the manufacturer's instructions a . The peroxidase-linked and biotinylated conjugates produced were designated AK13A2 HRPO and AK13A2 bi, respectively.
Viral strains used in this study.

Cells and viruses. Vero cells (ATCC CCL-81) and Madin Darby Bovine Kidney cells (ATCC CCL-22) were grown at 37°C, 5% CO2 in Dulbecco Eagle's minimal medium supplemented with 2.5 mM L-glutamine, 1% sodium hydrogenocarbonate, 0.5 mM mercaptoethanol, 50 U/ml penicillin, 50 μg/ml streptomycin, and 10% (V/V) of heat-inactivated fetal calf serum. To culture BRSV and HRSV, subconfluent Vero cell monolayers were inoculated at a multiplicity of infection of 0.1 and grown in Dulbecco Eagle's minimal medium supplemented with 2.5 mM L-glutamine, 1% sodium hydrogenocarbonate, 0.1% mercaptoethanol, 50 U/ml penicillin, and 50 μg/ml streptomycin. To culture parainfluenza-3 virus (PI3V), bovine viral diarrhea virus (BVDV, strain NADL), bovine herpesvirus-1 (BHV-1), and bovine coronavirus, subconfluent MDBK cell monolayers were inoculated under the same conditions. After inoculation, the cells were incubated at 37°C, 5% CO2 until an extensive cytopathic effect was observed. Prior to use, the virus cultures were stored at −70°C. Following thawing and clarification, the titer of the RSV preparations was determined using the Spearman and Kärber method and expressed as tissue culture infectious dose 50 (TCID50) 13 Four RSV strains representing the different subtypes were used in this study (Table 1). As BRSV virions appear as membrane bound particles in cell culture, 5 the virus preparations and uninfected Vero cell lysates (positive and negative controls, respectively) were incubated at 4°C for 16 hr with 1% (W/V) Zwittergent b in order to free the BRSV particles properly from the cell membranes. The treated cultures were centrifuged at 2,000 × g for 10 min and frozen (-20°C) in aliquots until use.
Clinical samples. Two sets of diagnostic specimens (A and B) consisting of various organs (lung, trachea, spleen, kidney, liver, bowel, gut, and cotyledon) were collected from dead cattle and submitted to the Virology Diagnostic Laboratory of the Veterinary and Agrochemical Research Center (VAR), Ukkel, Belgium. Routinely, all the samples coming from animals with respiratory diseases were analyzed for BRSV, BHV-1, and PI-3V by IIF, even organs that were not a target of these viruses. Set A (150 samples) was collected from 2000 to 2001. Set B (108 samples from 58 animals) was collected during 2003. Samples of set A were used for the development and the analysis of the diagnostic specificity and samples of set B for the validation of the 1-step ELISA.
For each sample, approximately 2 cm3 of the specimen were used to prepare a homogenate. The tissue was minced and homogenized in 15 ml of PBS (NaCl 8 g/liter, Na2HPO4.2H2O 1.15 g/liter, KH2PO4 0.2 g/liter, and KCl 0.2 g/liter) with a tissue homogenizer c . The homogenates were centrifuged at 2,000 × g for 10 min and the supernatants were used for ELISA (sets A and B), Clearview RSV EIA (set B), and real-time RT-PCR analyses (set B). Another portion of the specimen was frozen and used to prepare the cryosections for IIF (sets A and B).
BRSV 1-step ELISA procedure. The BRSV 1-step ELISA was designed as a simultaneous double-antibody sandwich ELISA. To establish the optimal AK13A2bi and AK13A2HRPO conjugate dilutions in the mix, a checkerboard titration was performed twice with constant virus concentration (undiluted virus culture). The relative proportion and dilution of the two conjugates giving the highest signal to noise ratio were retained to prepare the RSV conjugate mix.
In each well, 100 μl of sample or controls, along with 100 μl of RSV conjugate mix were added to avidin-coated ELISA plates (maxisorb d 96-well plates coated with a 10 μg/ml avidin e in PBS). The plates were covered and incubated for 1 hr on a shaker f at room temperature. At the end of this incubation step, the plates were washed 5 times with phosphate buffered saline (PBS) containing 0.1% Tween 20 (PBST) and 3 times with PBS. Then, 100 μl of substrate chromogen mix (50 mM sodium citrate buffer pH 4.25 containing 0.4% (W/V) tetramethyl benzidine (TMB) and 0.043% H2O2) were added to each well. The reaction was allowed to develop for 2–4 min at room temperature before being stopped by the addition of 100 ml of a 1M H3PO4 solution. The optical density of each well was read at 450 nm using a spectrometer g .
Well-to-well variation (standard deviation of the OD values of 8 replicates) was calculated using 2 positive and 2 negative samples.
Determination of the cut-off value for the 1-step ELISA. The cut-off value of the 1-step ELISA was determined using homogenates of 93 BRSV-negative samples from set B (n = 108). The BRSV-negative status of these samples was established using real-time RT-PCR. The cut-off value was set at the mean OD value of this negative population (n = 93) plus 3 SD. 6
Clearview RSV enzyme immunoassay (EIA) h . The Clearview RSV test is a lateral flow immunogold assay that allows direct visual detection of RSV antigen in clinical samples. It is based on the detection of RSV F protein by a specific monoclonal antibody. Homogenates of set B were tested using the Clearview test according to the manufacturer's instructions. The supernatant of a BRSV RB94 virus culture and an uninfected Vero cell culture supernatant were used as positive and negative controls, respectively. The test was read independently by 3 technicians. Samples that were positive on at least 2 of the 3 readings were scored as BRSV Clearview positive.
Samples used in this study.

Indirect immunofluorescence (IIF). Five-micron cryosections were performed on frozen organ samples of sets A and B. The sections were fixed in acetone for 15 min at room temperature and washed 3 times in PBS. Cryosections of each sample were incubated overnight with biotin-labeled a bovine polyclonal field antibodies specific for either BRSV, PI3V, BVDV, BHV-1 (the absence of cross-react with other viruses was evaluated), at room temperature, then washed 3 times with PBS and air dried. Finally, the sections were incubated for 1 hr at 37°C with fluorescein-isothiocyanate (FITC)-conjugated streptavidin i , washed again as previously described, then dried and examined using an epifluorescence microscope j at 488 nm.
Real-time RT-PCR. The RNeasy mini tissue kit k was used to extract RNA from 100 μl of homogenates from set B (n = 108), according to the manufacturer's instructions. Real-time RT-PCR was performed on the viral RNA. First, RNA was reverse transcribed to cDNA with random hexanucleotide and the First cDNA synthesis kit according to the manufacturer's instructions. l The real-time RT-PCR was performed as previously described, 3 using primers and probes targeting sequences of a conserved region of the RSV nucleoprotein gene. The amplification mixture (25 μl) was composed of 2.5 μl of reverse transcription product, 0.2 μM RSV forward primer, 0.2 μM RSV reverse primer, 0.2 μM probe, 12.5 μl iQ Supermix m containing dNTP, MgCl2, the polymerase buffer, and iTaq DNA polymerase in 20 mM Tris-HCl, pH 8.4 buffer. The amplification was performed on an iCycler m with the following parameters: 2 min at 50°C, 10 min at 95°C, 45 cycles of 15 sec at 95°C and 1 min at 59°C. Relative fluorescent units (RFU) for the FAM probe were monitored during each cycle at 490 nm. Samples that had a Ct value lower than 45 were scored as BRSV real-time RT-PCR positive. To estimate the reproducibility of the results, each sample was analyzed in triplicate.
Detection limit of 1-step ELISA, real-time RT-PCR, and Clearview RSV EIA. The supernatant of zwittergent-treated virus cultures of known titer was used for determining detection limits of the one step ELISA. Serial 2-fold dilutions of the 4 RSV strains (virus) and Vero cell lysate (blank) were done. One hundred microliters of each dilution were analyzed in duplicate using the 1-step ELISA following the procedure previously described. The mean of the corrected OD values (OD virus-OD blank) was calculated. A plot of the corrected OD values was made as a function of virus titer in TCID50/ml. The detection limit of the ELISA was calculated using Biolise software n and expressed as the virus titer that yielded an OD value 0.2 units.
Furthermore, the BRSV RB94 dilutions were analyzed using real-time RT-PCR and the Clearview RSV EIA. For both tests, the lowest virus titer that gave a positive result established the detection limit of the test.
Validation of the 1-step BRSV ELISA. The validation of the 1-step ELISA was done on autopsy specimens of set B (n = 108). This sample set was tested using the 1-step ELISA, IIF, real-time RT-PCR, and the Clearview RSV EIA as previously described. Cryosections of these samples were analyzed by IIF immediately after being received. The 1-step ELISA, Clearview RSV test, and real-time RT-PCR were done on the supernatant of homogenates after 2 freezing/thawing cycles. Test results were expressed as BRSV positive or negative according to the criteria defined for each test. The relative sensitivity, specificity, and Cohen's kappa coefficient were calculated for 1-step ELISA, IIF, and the Clearview RSV EIA, using real-time RT-PCR as the reference method. The different parameters were calculated based on contingency tables. 15 , 21
Results
Determination of the cut-off value of the 1-step ELISA. When the 108 samples from set B were tested by real-time RT-PCR, 93 were negative (or doubtful that were considered to be negative) and 15 were positive. The mean of the OD value of the 93 real-time RT-PCR-negative samples was 0.07 OD units with an SD of 0.037 OD units. The calculated cut-off value of the 1-step ELISA was fixed at 0.181 (= mean + 3 SD). This cut-off value was rounded off to 0.2 OD units. The mean of the corrected OD of 15 real-time PCR-positive samples was 1.003 OD units with a standard deviation (SD) of 1.148 OD units (range 0.052–3.04).
Detection limit of 1-step ELISA, Clearview RSV EIA, and real-time RT-PCR (Table 3). The limit of detection of the 1-step ELISA for 2 bovine and 2 human RSV strains belonging to different subtypes was calculated. Polynomial analysis of the fitted curve (Biolise) yielded limits of detection of 103.9 TCID50/ml for BRSV strains, and 4–8 times higher values for HRSV strains (Table 3). The Clearview RSV EIA had a detection limit of 105 TCID50/ml for BRSV RB94, which is comparable to the detection limit for HRSV indicated by the manufacturer (105.3 PFU/ml or 105.1 TCID50/ml because 1 PFU = 0.7 TCID50/ml). Of the 3 tests, the real time RT-PCR had the lowest detection limit, because it detected BRSV RB94 virus at a titer lower than 10 TCID50/ml.
Detection limits for 1-step BRSV ELISA, Clearview RSV and real-time RT-PCR.
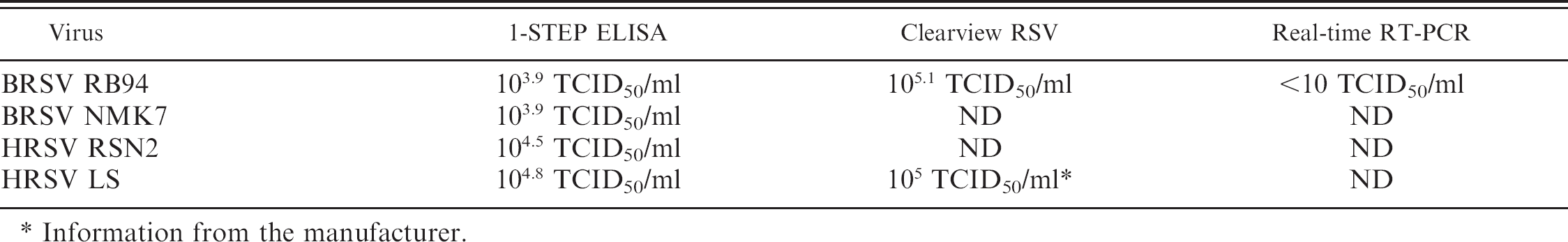
Information from the manufacturer.
Specificity of the 1-step ELISA. When virus cultures of other bovine respiratory viruses (BHV-1, BVDV, PI-3V, and bovine coronavirus) were used to assess cross-reactivity of the 1-step BRSV ELISA, the test was uniformly negative. Regarding clinical samples (set A), 6 samples had positive IIF results for PI-3V, 14 samples had positive IIF results for BHV-1, and 20 samples had positive IIF results for BVDV. All these samples, negative for BRSV by IIF, were also negative in the 1-step BRSV ELISA. Whereas 15 out of 19 BRSV IIF positive samples were also positive in the 1-step ELISA.
Validation of the 1-step ELISA. Because real-time RT-PCR had the highest analytical sensitivity (lowest limit of detection), it was used as the reference method to calculate the relative sensitivity, specificity, and Cohen's kappa value for the other 3 methods, using data from sample set B (n = 108). The results are presented in Table 4. For some samples with high Ct values (close to 45), real-time RT-PCR generated discordant results on different test runs; the BRSV status of these samples were considered doubtful (+/-). For statistical analyses, their BRSV status was considered to be negative.
For analysis of the results (set B), the samples were classified as being from the respiratory tract or other tissues. BRSV is known to be a strictly respiratory pathogen, the nonrespiratory tract tissues were therefore considered as relevant negative controls. For the respiratory tract tissue samples, 5 samples had positive and 37 samples negative BRSV results on all 4 tests. 1-step BRSV ELISA showed a low well-to-well variation (standard deviation <5%). The ELISA was the only test for which all positive and negative results were confirmed by at least 1 other test method. For the nonrespiratory tract tissues, most samples were negative on all 4 tests; however, 8 of these samples had doubtful real-time RT-PCR results, and 2 other samples had IIF positive results. Some samples were different organ specimens taken from the same animal. In general, similar results were obtained with these specimens. However, 2 exceptions must be noted. In one animal, the lung, spleen, and kidney tissue, and in another animal, the trachea and lung tissue were analyzed. In these animals, real-time RT-PCR, 1-step ELISA, and Clearview RSV EIA gave negative results for all organs. In IIF, the spleen and the kidney of one animal (Table 4 †) and the trachea of the other animal (Table 4 *) were BRSV positive, whereas both lung cryosections were negative.
Table 5 presents the relative sensitivity, relative specificity, and Cohen's kappa values for IIF, Clear-view EIA, and 1-step ELISA, using the real-time RT-PCR as the reference method. The 1-step ELISA had the highest relative sensitivity (60%) compared to IIF and Clearview RSV (47% for both). Furthermore, the new 1-step ELISA was the only BRSV diagnostic method that had no false-positive results and thus had a relative specificity of 100% when compared to real-time RT-PCR. Regarding the kappa values, the results of the 1-step ELISA were in good agreement with the real-time RT-PCR results (kappa = 0.72), whereas the agreement of the 2 other BRSV diagnostic methods with the real-time RT-PCR results was notably weaker (IIF, kappa = 0.50 and Clear-view RSV EIA, kappa = 0.57).
Pattern of the BRSV test results of set B samples by 1-step ELISA, IIF, Clearview RSV EIA and real-time RT-PCR.

Trachea.
Spleen and kidney.
Relative sensitivity (Sens), specificity (spec) and correlation coefficient (Kappa, κ) for IIF, Clearview RSV and 1-step BRSV ELISA results, using real-time RT-PCR (Real) as the reference test.

Discussion
Enzyme immunoassays are preferred diagnostic tests because they are inexpensive and easy to use. However, most of the commercially available RSV ELISAs suffer from either poor sensitivity or a time-consuming 3-step procedure. 12 In the present study, a new quick, reliable antigen detecting ELISA was developed for BRSV diagnosis. The new 1-step ELISA is economical, user-friendly, and can be easily performed using the existing equipment in veterinary diagnostic laboratories. When performed according to the described procedure, the 1-step ELISA had low background staining (mean OD of 93 negative samples = 0.07) and low well-to-well variation. The antigen (F protein) present in BRSV-infected clinical samples that is recognized by the 1-step ELISA is stable over a long period of time (>3 years) at −20°C, even under poor storage conditions with several freezing-thawing cycles, as shown by the results from sample set A.
With respect to diagnostic specificity, the 1-step ELISA reacts with the 2 HRSV strains of subtype A and B. However, the 2 tested HRSV had to be present at titers at least 4 to 8 times higher than the BRSV to generate positive test results. These results are suprising, but might be explained by that the optimization of the 1-step ELISA was performed with BRSV samples. No cross reactivity was observed for PI-3V, BHV-1, bovine coronavirus, and BVDV.
Laboratory diagnosis of BRSV infection is commonly done by indirect immunofluorescence (IIF). 12 , 15 However, the data generated in this report indicates that false-positive IIF results are likely to occur. Indeed, in 2 animals, real-time RT-PCR, 1-step ELISA, and Clearview RSV EIA gave negative results for all organs, whereas spleen and kidney of 1 animal and the trachea of the other animal were IIF-positive but lung sections of the same animals were IIF negative. Given that the spleen and kidneys have not been described as target organs for BRSV, these IIF results are likely to be false-positives.
In this study, the Clearview RSV EIA, developed for human diagnosis, was efficient for diagnosing BRSV in bovine autopsy specimens. These results corroborate a previous study using the Clearview EIA for BRSV diagnosis 15 and are not surprising in view of the high homology between the F protein (target of this test) of BRSV and HRSV. 14 However, the Clearview RSV test had 3 (2 negative and 1 positive) BRSV results that did not match the results of the other 3 tests.
Recently, a real-time RT-PCR was developed for BRSV diagnosis. 3 , 8 Given the detection limit established with the RB94 BRSV strain, the real-time RT-PCR was the most sensitive method among the 4 tests evaluated in this paper. The higher sensitivity of realtime RT-PCR could explain the positive results obtained in the 6 samples that were negative with the other 3 tests. However, real-time RT-PCR requires complex sample preparation, high level of skill and experience, and costly equipment and consumables. Furthermore, real-time RT-PCR gave many discordant results for clinical samples with high Ct values, notably for 8 nonrespiratory tissue samples.
Because of the high sensitivity of real-time RT-PCR, it was chosen as the reference method to calculate the relative sensitivity, specificity, and Cohen's kappa values for the other 3 methods. The IIF had the lowest relative sensitivity and specificity of the 3 immunological methods. A kappa value of 0.50 indicated a moderate correlation between the IIF results and those of real time RT-PCR. The Clearview RSV EIA had only slightly better sensitivity, specificity, and kappa value than the IIF test. The new 1-step ELISA was the only BRSV diagnostic method that had no false positive results and, thus, had a relative specificity of 100% compared to real-time RT-PCR. Furthermore, the 1-step ELISA had a relative sensitivity that was clearly the highest, and the kappa value indicated a good correlation between the results of 1-step ELISA and real-time RT-PCR. These findings paralleled the detection limit of the different methods (Table 3).
On the basis of its higher relative sensitivity, specificity and Cohen's kappa value when compared to IIF and Clearview ELISA, the new 1-step ELISA developed in this study proved to be a rapid and improved alternative to the existing BRSV diagnostic methods. The 1-step ELISA is a robust test that gives highly reliable results for diagnosing BRSV in organ homogenates.
Acknowledgements
The authors thank Delphine Marchal, Sandrine Henin, Yves Etienne, Pierre Mouton, Hugues Heine, Ann Geeninckx-Lambert, and Bruno Detry for their excellent technical assistance. This work was funded by the Province du Luxembourg of Belgium.
Footnotes
a.
D-biotinoyl-ε-aminocaproic acid N-hydroxysuccinimide ester, Roche Diagnostics Corp. Indianapolis, IN.
b.
Zwittergent, Calbiochem, VWR, Leuven, Belgium.
c.
Ultra Turrax T25 basic homogenizer, IKA Labortechnik, Staufen, Germany.
d.
Nunc MaxiSorp™ F96, VWR International, Leuven, Belgium.
e.
Neutravid, Belovo, Bastogne, Belgium.
f.
Titertek shaker, Flow Laboratories, Meckenheim, Germany.
g.
Multiskan RC spectrometer, Labsystem, Life Science International, Zellik, Belgium.
h.
Clearview RSV kit, Oxoid N.V./S.A., Ukkel, Belgium.
i.
Streptavidin-FITC, Sigma-Aldrich N.V./S.A., Bornem, Belgium.
j.
Leitz Fluovert FS, CA Greiner & Söhne GmbH & Co KG, Nürtenberg, Germany.
k.
RNeasy mini tissue kit, QIAGEN GmbH, Hilden Qiagen, Germany.
l.
First cDNA synthesis kit, Roche Diagnostics Belgium NV, Vilvoorde, Belgium.
m.
Bio-Rad Laboratories S.A.-N.V., Nazareth Eke, Belgium.
n.
Biolise software, version 2.0, Life Science International, Zellik, Belgium.
