Abstract
Polysorbate 80 (PS80) is commonly used in pre-clinical formulations. The dose threshold for cardiovascular (CV) changes and hypersensitivity reaction in the dog was assessed and compared to other species. PS80 was administered by intravenous (IV) bolus (.5, 1 mg/kg), IV infusion (.3, .5, 1, 3 mg/kg), subcutaneous (SC) injection (5, 10, 15 mg/kg) and oral gavage (10 mg/kg) to dogs with CV monitoring. Monkeys and minipigs received PS80 by IV infusion at 3 mg/kg. Plasma histamine concentration was measured following PS80 IV infusion and with diphenhydramine pre-treatment in dogs only. In dogs, PS80 was not associated with CV changes at doses up to 15 mg/kg SC and 10 mg/kg oral, but decreased blood pressure and increased heart rate with IV bolus at ≥ .5 mg/kg and IV infusion at ≥ 1.0 mg/kg and decreased body temperature with IV infusion at 3 mg/kg was observed. Transient edema and erythema were noted with all administration routes, in all three species including doses that were devoid of CV effects. In monkeys and minipigs, PS80 did not induce CV, cutaneous or histamine concentration changes. These results suggest that mild, transient skin changes occur following PS80 administration at doses that are not associated with CV effects in the dogs. In dogs, the cardiovascular effect threshold was <.5 mg/kg for IV bolus, .3 mg/kg for IV infusion, 15 mg/kg for SC injection, and 10 mg/kg for oral administration. Monkey and minipig were refractory to PS80-induced histamine release at 3 mg/kg by IV infusion over 15 minutes.
Keywords
Introduction
Polysorbate 80 (PS80) (also known as Tween® 80 or polyoxyethylene-sorbitan-20-monooleate) is a synthetic non-ionic surfactant widely used in drug formulation vehicle as a solubilizing or dispersing agent.1,2 Various pharmaceutical products for topical, oral and parental routes of administration are formulated in PS80, including chemotherapies, vaccines, vitamins, analgesics and antiarrhythmics. 3 PS80 is also a widely used vehicle in pre-clinical studies for oral and parental administration of poorly water-soluble compounds. Although the use of PS80 as an excipient is considered generally safe and is approved by the U.S. Food and Drug Administration (FDA) for intravenous drug delivery, recent studies have suggested its implication in clinical adverse effects including acute hypersensitivity and allergic reactions.3-5 In dogs, recognized as the most sensitive species, intravenous PS80 has been associated with severe non-immunologic anaphylactoid reactions (eg erythema, swelling), hypotensive effects and depression of myocardial contractility.6-11 Our study aimed to determine the dose threshold for reactivity to PS80 in the dog and associated changes in cardiovascular parameters and histamine blood levels, and to compare these effects with other nonrodent species commonly used in pre-clinical studies such as the cynomolgous monkey and the minipig.
Methods
Statement on Use and Care of Animals
During the study, care and use of animals were conducted in accordance with principles outlined in the current Guide to the Care and Use of Experimental Animals published by the Canadian Council on Animal Care and the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 2010). Citoxlab (now Charles River Laboratories) North America’s facility is AAALAC accredited. All procedures were conducted as per Standard Operating Procedures (SOPs).
Animal Housing
Seven (4 males and 3 females) cynomolgus monkeys (Macaca fascicularis), 7 (4 males and 3 females) göttingen minipigs (Sus scrofa), and 27 (22 males and 5 females) beagle dogs (Canis familiaris) were selected for this study. Monkeys were between 2 and 5 years old and weighed between 2.1 and 3.9 kg. Minipigs were between 4 and 5 months old and weighed between 11.0 and 12.6 kg. Dogs were between 9 and 19 months old and weighed between 6.8 and 12.1 kg. The animal room environment was controlled (temperature 21 ± 3°C, humidity 30–70%, 12 h light, 12 h dark, 10–15 air changes per h) with temperature and relative humidity being monitored continuously. A standard certified commercial chow (Monkeys: Certified Hi-Fiber Primate Diet #7195CTM, Minipigs: Certified Miniswine Diet #7037CTM, Dogs: Certified 25% Lab Dog Diet #8727CTM, Envigo Teklad, Madison, WI, USA) was provided twice daily for monkeys and minipigs and daily for dogs.
Animal Preparation
Animals were surgically prepared with telemetry transmitters (Model D70-PCT or M11, Data Sciences International, St-Paul, MN, USA) for arterial blood pressure, heart rate and body temperature monitoring. Prophylactic antibiotics (Penicillin G Procaine, Vetoquinol, Lavaltrie, QC, Canada, 30 000 UI/kg; Enrofloxacin, Bayer Health Care, ON, Toronto, Canada, 5 mg/kg) and analgesic (Buprenorphine, Champion Alstoe, Whitby, ON, Canada, .3 mg/mL, .4 mL) were administered intramuscularly prior to surgery and for 2 days post-surgery. A non-steroidal anti-inflammatory (Meloxicam, Boehringer Ingelheim, Burlington, ON, Canada, .2 mg/kg) was administered once after surgery. A local anesthetic (Bupivacaine .25%, Hospira, Montreal, QC, Canada, .4 mL; Lidocaine 20 mg/mL, Vetoquinol, Lavaltrie, QC, Canada, .4 mL) was administered subcutaneously in 6–10 sites distributed over the surgical sites during the surgery. Animals were placed on a heating pad and inhaled a mixture of oxygen (O2) and isoflurane (AErraneTM, Baxter Corporation, Mississauga ON, Canada). Respiratory rate was maintained between 8 and 20 breaths/min with an inspiratory airway pressure between 18 and 25 cm H2O using a mechanical ventilator (Hallowell EMC, Pittsfield, MA, USA). Heart rate, hemoglobin oxygen saturation (SpO2), and body temperature were monitored continuously during anesthesia. The telemetry transmitter was implanted between the aponeurosis of the transverse abdominis and the internal abdominal oblique muscles. The pressure catheter was inserted in the carotid or femoral artery. A recovery period of at least 4 weeks was allowed between the surgery and the start of cardiovascular recordings.
Experimental Methods
Treatments
For threshold assessment in dogs, a first cohort of animals received PS80 NF (Tween® 80, J.T. Baker Chemical Co, Phillipsburg, NJ, USA) by subcutaneous (SC) injection at 5, 10, and 15 mg/kg (2, 4 and 6 mL/kg respectively, 2.5 mg/mL), by intravenous (IV) bolus at .5 and 1 mg/kg (.2 and .4 mL/kg respectively, 2.5 mg/mL) or by a 30-min IV infusion at .3, .5, and 1 mg/kg (.12, .2 and .4 mL/kg respectively, 2.5 mg/mL) in a dose escalation design. The vehicle (.9% sodium chloride for injection USP, Baxter, Mississauga, ON, Canada) was administered as a control for each route of administration. A second cohort of dogs received PS80 NF (Tween® 80, J.T. Baker Chemical Co, Phillipsburg, NJ, USA) at 10 mg/kg by oral gavage (5 mL/kg, .2% w/v Tween® 80 and .6% methylcellulose A15 in reverse osmosis water). All dogs administered PS80 by oral gavage were pre-treated with an anti-emetic (Maropitant citrate, Zoetis, Kalamazoo, MI, USA, 1 mg/kg, SC). For species comparison, a second cohort of dogs, and a cohort of monkeys and minipigs received PS80 (Tween® 80, Sigma-Aldrich, Saint Louis, MO, USA) by a 15-min IV infusion at 3.0 mg/kg (2 mL/kg). In dogs, an anti-histaminic (diphenhydramine hydrochloride, Fresenius Kabi Canada Ltd, Toronto, ON, Canada, 1 mg/kg, SC) was administered to a subgroup of animals to prevent anaphylactoid reactions.
Cardiovascular Data Monitoring
Hemodynamic parameters (heart rate and mean, systolic, diastolic, and pulse blood pressure) and body temperature were recorded continuously in freely moving animals from at least 24 hours prior to dosing to at least 24 hours post-dosing completion using a computerized data acquisition system (Ponemah version 6.32, Data Sciences International, St-Paul, MN, USA) or in anesthetized animals receiving isoflurane at 1.5–2% for species comparisons (Figures 5 and 6). For telemetry data acquisition, at least one receiver for telemetry signals (Data Sciences International, St-Paul, MN, USA) was installed on the cage for each animal.
Measurement of Histamine Concentrations
Blood samples (a target of 2 mL each) were collected prior to dosing and at 10, 20, 25, 35, 45, 60, and 75 minutes post-dosing. Samples were collected into tubes containing K2 EDTA as anticoagulant and kept on wet ice pending centrifugation. Samples were centrifuged under refrigeration (set to 4°C at 1550 g RCF) for 10 minutes and placed on dry ice until storage in a freezer set to −70°C pending analysis. Histamine plasma concentrations were measured by ELISA (ab213975 histamine ELISA kit, Abcam, Canada, Range: .098 ng/mL–25 ng/mL, Sensitivity .03 ng/mL).
Statistical Analysis
SAS version 9.3 (SAS Institute, Cary, NC, USA) was used for the statistical analysis. The analysis of cardiovascular data was performed using a repeated measures analysis of variance (ANOVA). Each repeated measure analysis was defined by using the treatment as repeated factor, the covariance structures and the Kenward and Roger’s method. Pairwise comparisons of each dose level with the vehicle were conducted using t-test on least-squares means. Probabilities of P ≤ .05 were considered statistically significant.
Results
Effects of PS80 on Clinical Signs and Cardiovascular Function in Dogs
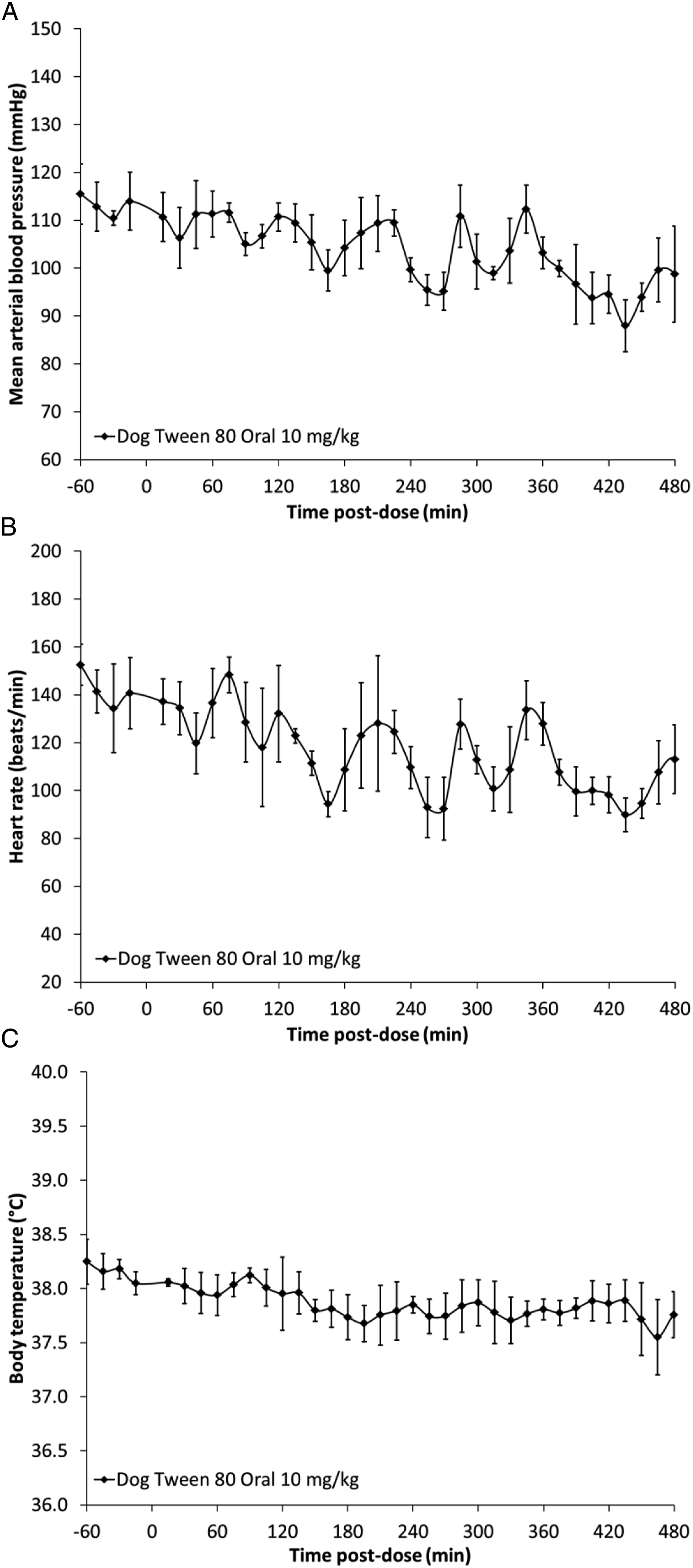
In the dog, administration of PS80 by SC injection at 5, 10 and 15 mg/kg and by oral gavage at 10 mg/kg was not associated with any effects on cardiovascular function (Figures 1 and 2). However, mild and transient clinical signs of hypersensitivity reaction including erythema, edema, and scratching were observed from approximately 20 to 60 min post-dose in 2/5 animals at 10 mg/kg SC, 4/5 animals at 15 mg/kg SC, and 1/3 animals at 10 mg/kg oral (Table 1). Mean arterial blood pressure (a), heart rate (b) and body temperature (c) in beagle dogs (n = 5) administered PS80 by subcutaneous injection at 5, 10 and 15 mg/kg. Data are presented as mean ± standard error of mean. Mean arterial blood pressure (a), heart rate (b) and body temperature (c) in beagle dogs (n = 5) administered PS80 by oral gavage at 10 mg/kg. Data are presented as mean ± standard error of mean. Anaphylactoid skin reaction and cardiovascular changes in dogs, non-human primates and gottingen minipigs with polysorbate 80 at different doses and routes of administration. CV: cardiovascular, SC: subcutaneous, IV: intravenous.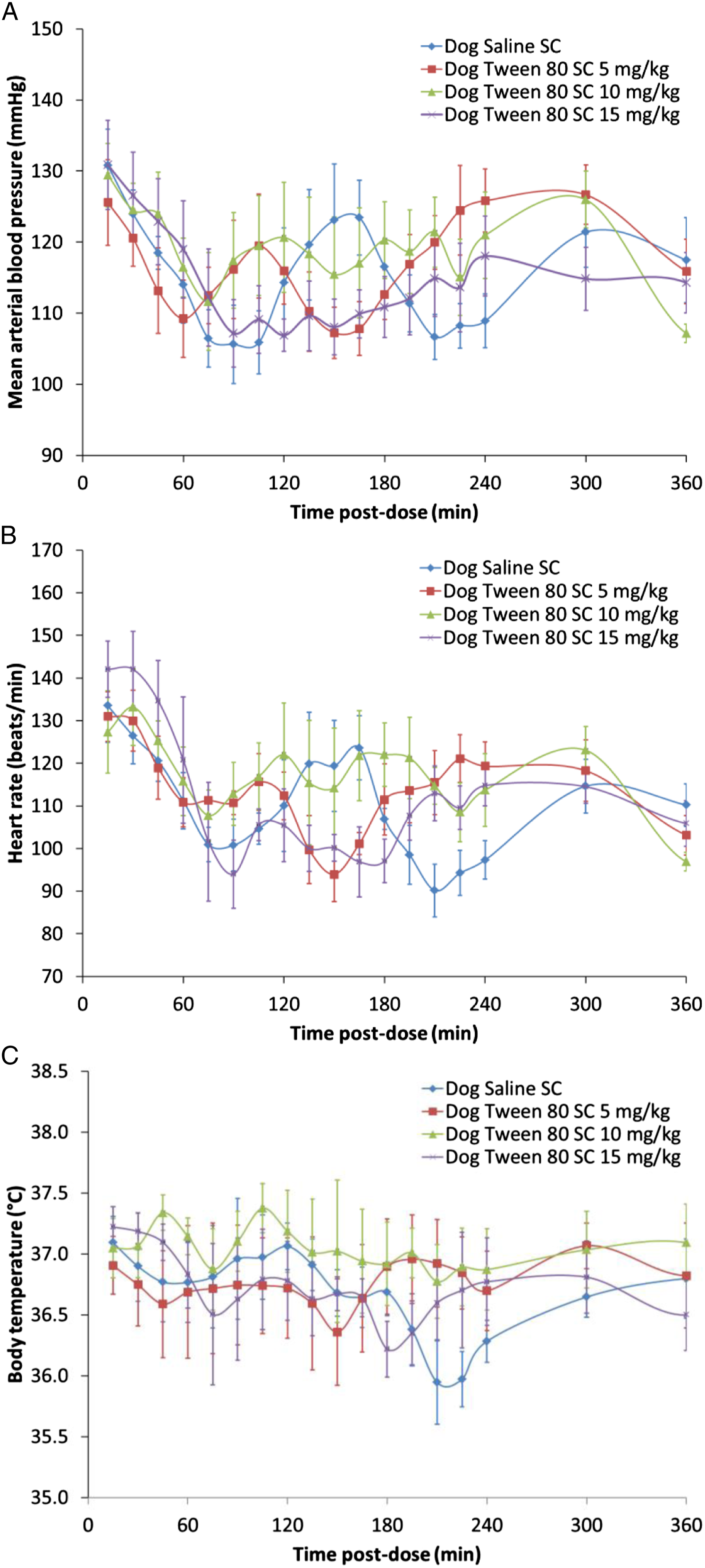
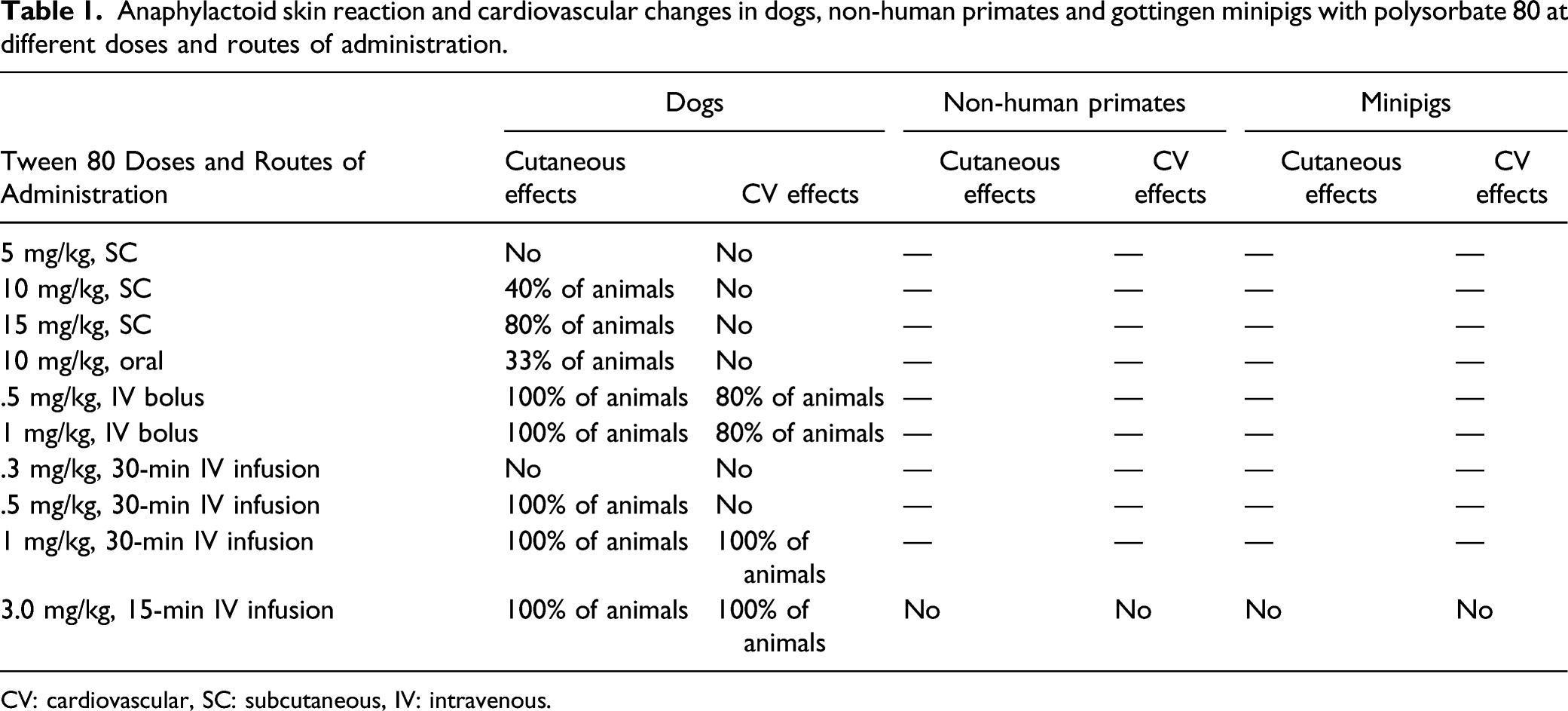
When PS80 was administered by IV bolus at .5 and 1 mg/kg, blood pressure was decreased, and heart rate was increased in a dose-dependent fashion up to 120 minutes post-dose (Figure 3). At nadir, mean arterial blood pressure was decreased by 5% (−6 mmHg) and 12% (−15 mmHg, P ≤ .01) while heart rate was increased by 41% (+40 bpm) and 43% (+51 bpm) at .5 and 1 mg/kg, respectively. There was a large inter-individual variation in the cardiovascular effects following IV bolus administration, with 2 out of 5 animals presenting more severe decreases in arterial blood pressure (up to −43 mmHg) and one animal without any changes at 1 mg/kg (Table 1). All animals showed adverse clinical signs starting at approximately 15 to 30 minutes post-dosing at both dose levels including facial edema, erythema and excessive scratching although the severity did not seem to correlate with the magnitude of cardiovascular changes. Mean arterial blood pressure (a), heart rate (b) and body temperature (c) in beagle dogs (n = 5) administered polysorbate 80 by intravenous bolus injection at 0.5 and 1 mg/kg. Data are presented as mean ± standard error of mean.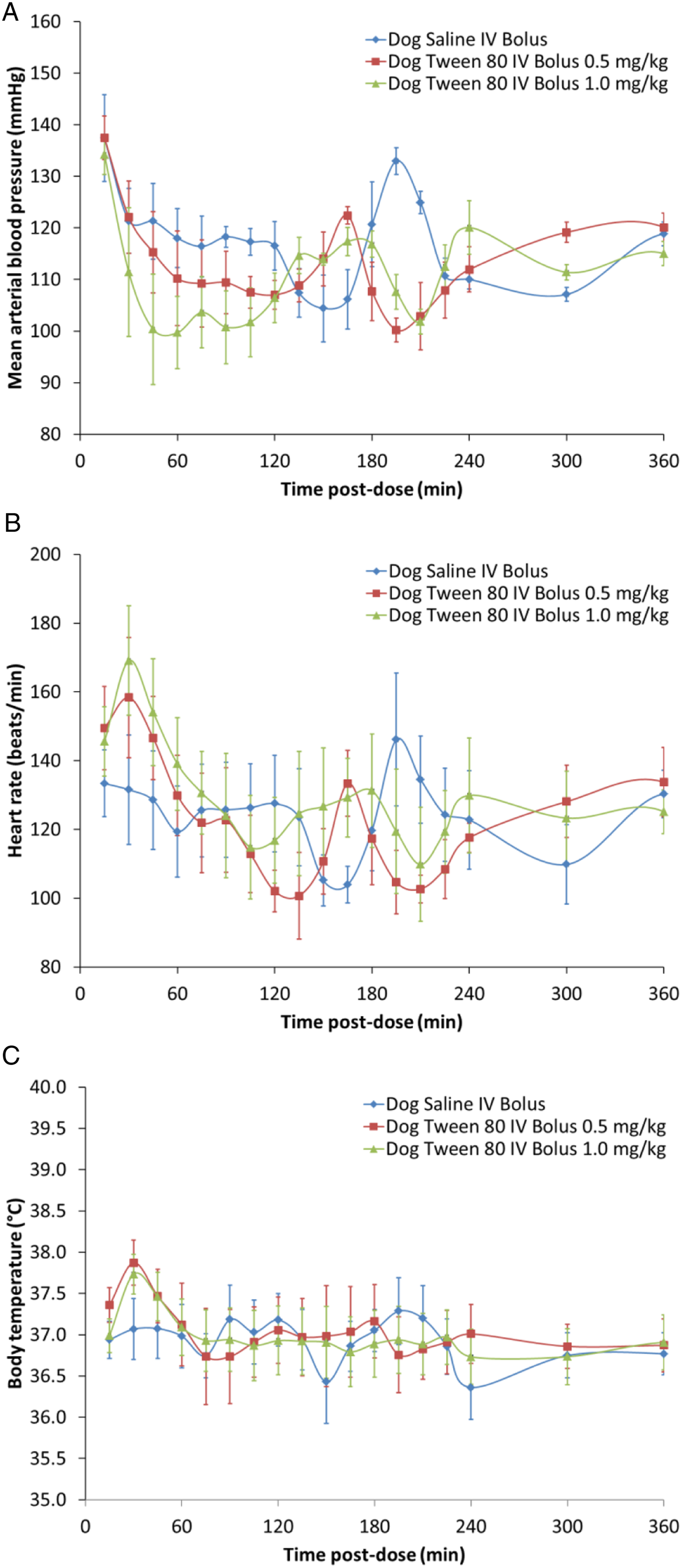
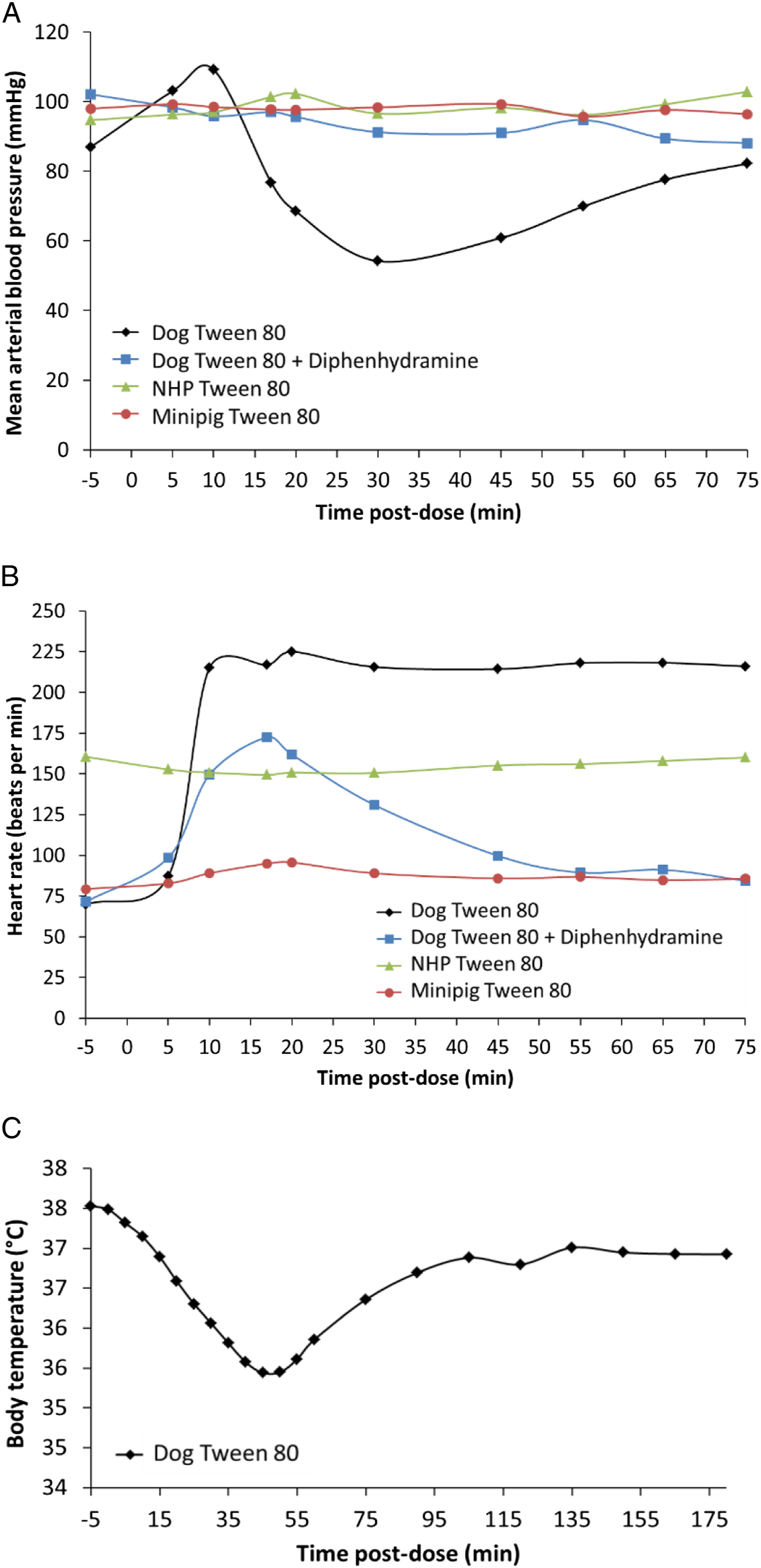
Doses of .5 and 1 mg/kg administered by a 30-min IV infusion were associated with transient cutaneous reaction in all dogs from approximately 15 to 120 minutes following the end of the infusion with a trend toward slightly more severe clinical signs at the highest dose. Cardiovascular effects were only seen at 1 mg/kg where mean arterial blood pressure was decreased by −11% (−14 mmHg) and heart rate was increased by 64% (+ 22 bpm) at 3 hours and 1 hour post-dose respectively (Figure 4). At 3 mg/kg IV (15-min infusion), body temperature was noted to decline with a −2°C nadir achieved by 45 min post-dose and recovery to baseline comparable values by 105 min post-dose. Mean arterial blood pressure was decreased by 38% (−33 mmHg) and heart rate was increased by 222% (+155 bpm) at 20 and 30 min post-dose respectively (Figure 5). All animals presented anaphylactoid cutaneous reaction including bilateral symmetrical erythema, pruritus and edema (Table 1). These cardiovascular and cutaneous effects induced by PS80 were attenuated with diphenhydramine. Mean arterial blood pressure remained similar to baseline in dogs pre-treated with diphenhydramine while heart rate was reduced by 30% compared to dogs without diphenhydramine (Figure 5). Cutaneous changes (erythema and facial edema) after diphenhydramine pre-treatment were absent to mild compared to moderate to severe without diphenhydramine. Mean arterial blood pressure (a), heart rate (b) and body temperature (c) in beagle dogs (n = 2) administered polysorbate 80 as a 30-min intravenous infusion at 0.3, 0.5 and 1 mg/kg. Data are presented as mean ± standard error of mean. Mean arterial blood pressure (a), heart rate (b) and body temperature (c) in anesthetized beagle dogs (n = 4), non-human primates (n = 4) and göttingen minipigs (n = 4) following a 15-min intravenous infusion of polysorbate 80 at 3 mg/kg, and with diphenhydramine pre-treatment (1 mg/kg, SC) in Beagle dogs (n = 4).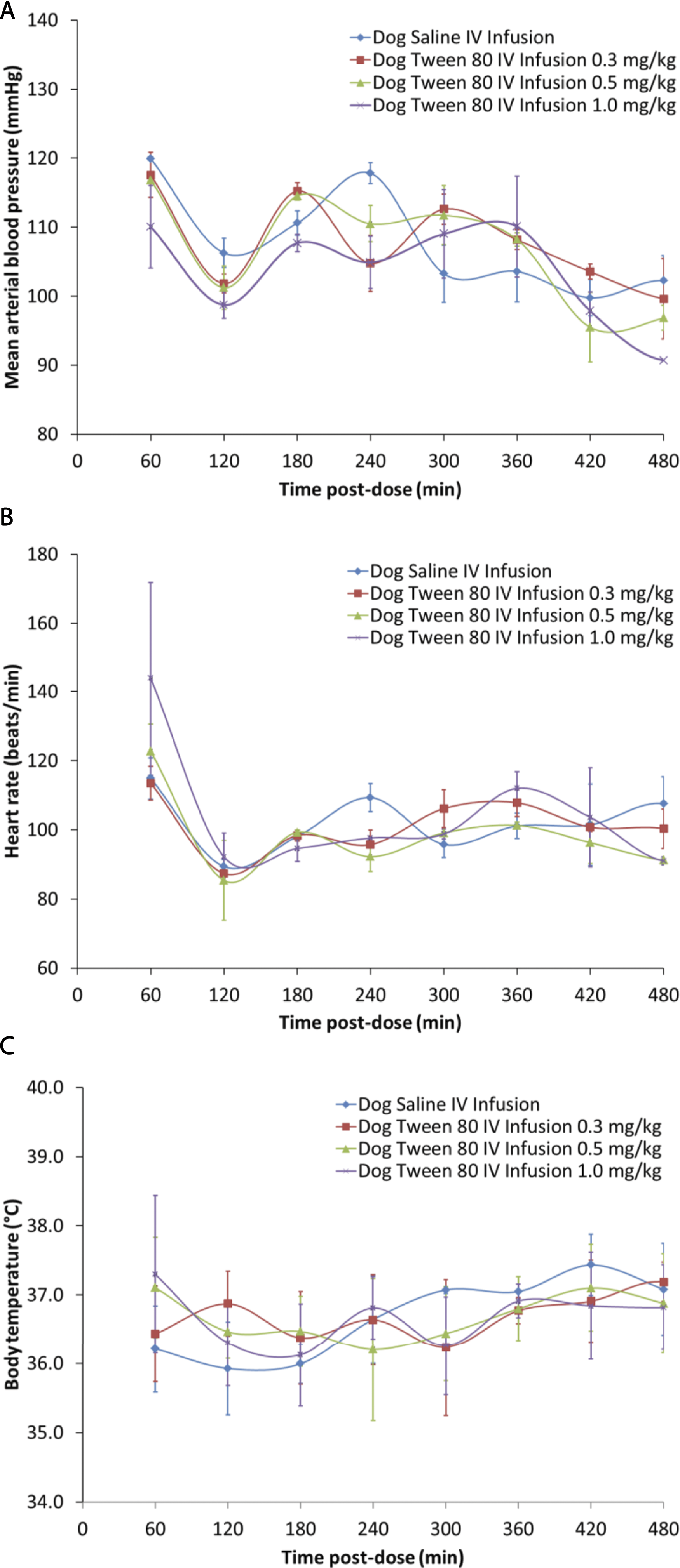
Effects of PS80 on Clinical Signs and Cardiovascular Function In Monkeys and Minipigs
As opposed to dogs, which were very sensitive to PS80, a 15-min intravenous infusion at 3 mg/kg did not cause any changes in heart rate, blood pressure or body temperature and did not induce any cutaneous reaction in the anesthetized monkey or minipig.
Histamine Concentrations in Dogs, Monkeys and Minipigs
In dogs, intravenous infusion of polysorbate 80 at 3 mg/kg induced a significant increase in histamine levels (Figure 6). Increases from baseline were noted starting at 10 minutes following infusion start. Peak effects were observed 20 minutes following infusion start where plasma levels were increased by 1720% from baseline. Administration of diphenhydramine as a pre-treatment in dogs receiving PS80 was associated with a 3-fold reduction in histamine release. In comparison, the same dose of PS80 administered intravenously had no significant effect on histamine levels in the monkey or minipig. Percentage change in plasma histamine concentration in beagle dogs (n = 4), non-human primate (n = 3) and göttingen minipigs (n = 3) following a 15-min intravenous infusion of polysorbate 80 at 3 mg/kg, and with diphenhydramine pre-treatment (1 mg/kg, SC) in Beagle dogs (n = 4). Data are presented as mean ± standard deviation.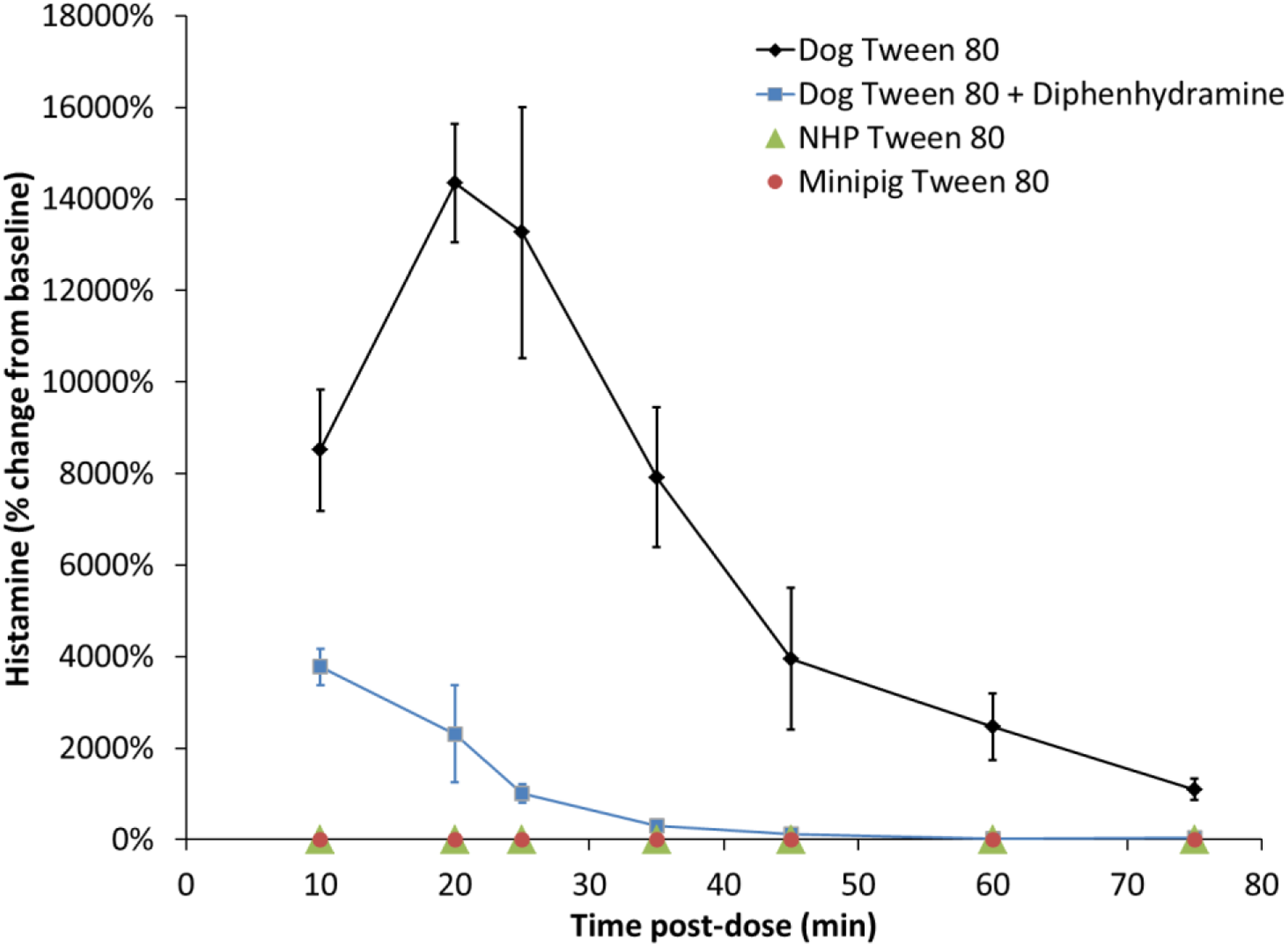
Discussion
Anaphylaxis is traditionally induced by the cross-linking of antigen specific IgEs that have bound to the IgE receptor (FcεRI) on mast cells and basophils. 12 This binding consequently induces a signaling cascade which results in the release of cytokines, immune mediators such as histamine and proteases, as well as lipid-mediators including the platelet-activating factor (PAF). 12 Anaphylactoid reactions differ from traditional anaphylaxis in terms of their inducibility by non-IgE mediated reactions, and rather occur through non-immune mediated release of mediators or from the activation of the complement pathway. 13 This form of hypersensitivity also involves mast cells which respond to stimulants by releasing cytokines and other mediators into the extracellular space. 14
As an amphipathic, non-ionic compound composed of sorbitan polyoxyethylene (POE) fatty acid esters, 15 PS80 is commonly used in medical and biopharmaceutical formulations including chemotherapies, vaccines, vitamins, analgesics, and antiarrhythmics 3 mostly due to its solubilizing properties at low concentrations and low toxicity levels. 16 Specifically, formulation processing can be associated with protein aggregation, which in turn may potentially induce undesired immunological response with antibodies directed towards protein aggregates. 15 Excipients such as PS80 may be used to mitigate protein adsorption and interface-induced protein aggregation.15,17 Variations in PS80 composition between suppliers or products can lead to changes in physico-chemical properties and need to be considered when evaluating the potential of PS80 to induce anaphylactoid reactions. 18
Histamine is a mediator released primarily from mast cells and basophils with an influence on T lymphocyte regulation during pathogenesis of allergic diseases. 19 Specifically, multiple receptors expressed at the surface of mast cells including FcεRI and FcγRI, receptors for complement components such as C3aR and C5aR as well as nerve growth factor (NGF) Trk A, all contribute to the release of histamine upon binding of their respective ligand. 19 Specific for histamine, the H1 receptor is distributed in neurons, smooth muscle cells of the airways and blood vessels 20 and stimulates vasodilation, cellular migration and bronchoconstriction. 21 Immediate hypersensitivity reactions which are typically considered as signs of hypersensitivity type I and including edema, erythema, and pruritus are observed following activation of H1R. Instead, H2, H3 and H4 receptors are involved in vascular permeability and airway constriction 22 neuro-inflammatory disease 23 and release of inflammatory signaling cascades consisting of cytokines and cell chemotaxis.19,24 Like humans, H1 and H2 receptors are also distributed throughout various tissues in dogs, including all sections of the gastrointestinal (GI) tract,25,26 bronchial and tracheal smooth muscle, 27 and atrioventricular tissue. 28
Histamine release mediates an increase in vascular permeability and vasodilation via the activation of histamine receptors including H1 and H2, however the response varies between the 2 receptor subtypes. 29 Specifically, H1 receptors reside primarily in endothelial cells where their activation results in production of the vasodilator, nitric oxide (NO). 29 NO subsequently diffuses from endothelial cells to vascular smooth muscle cells, resulting in an increase in cyclic guanine monophosphate (cGMP) and relaxation in vascular smooth muscle.29-31 Similarly, H2 receptors located in vascular smooth muscle cells induce cyclic adenosine monophosphate (cAMP)-mediated vasodilation. 29
Beyond vascular effects described above, histamine has also been associated with immunological effects. The binding and activation of histamine receptors leads to several downstream events including the differentiation of T cells, B cells, monocytes and dendritic cells in lymphatic organs and tissues. 20 Specifically, during inflammation, high levels of the H1 receptor in T helper (Th1) cells results in Th1 activation, leading to increased synthesis of IFN-γ and upregulation in cytokine cascades. 32 Moreover, in Th2 cells, H2 receptor levels are more prevalent than H1 receptors leading to the suppression of histamine and decrease in cytokine release. 32
Results that are summarized in the current manuscript provide evidence that dogs are highly sensitive to histamine-mediated anaphylactoid reactions following PS80 administration by various routes. The data also indicate that cutaneous changes occur before cardiovascular changes, although the effects may be mediated by the vasoactive properties of histamine. Interestingly, the data provided from NHPs and minipigs indicate that these 2 species were refractory to PS80-mediated histamine release and anaphylactoid reactions at PS80 doses and route that were used in this study.
Histamine receptor activities can be pharmacologically altered receptor antagonists such as diphenhydramine. Specifically, antihistamine drugs bind to histamine receptors (H1 or H2) resulting in an inactive state, and thus a decrease in histamine response. 33 The use of histamine receptor antagonists has been considered for mechanistic investigations of PS80-related effects in nonclinical studies, mostly when using dogs. Administration of diphenhydramine reduced plasma histamine concentrations possibly through antagonist activity on H1 receptors located on mast cells, the main source of histamine. 19
In conclusion, the current study characterizes the effects of PS80 including cardiovascular and cutaneous effects in dogs as a function of the dose delivered by different routes. Administered dogs displayed signs of anaphylactoid reaction manifested in the form of erythema, edema and signs of pruritus that occurred at lower doses but with cardiovascular effects (increased heart rate and decreased blood pressure) as the PS80 dose was increased. The threshold for cardiovascular effects was <.5 mg/kg for intravenous bolus, .3 mg/kg for intravenous infusion, 15 mg/kg for SC injection and 10 mg/kg for oral administration. These results may inform decisions related to PS80 vehicle composition in toxicology studies, particularly when using dog as an animal model.
Footnotes
Author Contributions
Myléne Pouliot contributed to conception, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Jeanine Bussiere contributed to conception, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Aldo Coppi contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Kristen Holbrook contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Amy Shelton contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Samantha Sparapani contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Jonathan Maher contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Tanja S Zabka contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Simon Authier contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
