Abstract
Polysorbate 80 (PS80) functions as a dispersing agent or solubilizer in many pharmaceuticals, and as a stabilizer in biopharmaceuticals. Topical or parenteral administration of low doses of PS80 in biopharmaceuticals has been associated with mild allergic reactions, including local injection site reactions in humans. High doses of PS80, such as levels found in traditional Chinese herbal parenteral medicines, have been linked to systemic effects consistent with anaphylactoid-type reactions, which are characterized by the direct release of histamine from mast cells (degranulation). Nonclinical safety assessments of PS80
Impact Statement
High doses of the excipient PS80, such as the levels found in injectable traditional herbal remedies, have been linked to serious, clinical systemic anaphylactoid allergic reactions. Although biopharmaceuticals contain much lower levels of PS80, their safety has come under scrutiny from regulatory agencies. We have utilized both exploratory and GLP studies to establish dose threshold levels for PS80 using routes of administration relevant to biopharmaceuticals: IV Bolus, IV infusion, and SC bolus. This analysis will allow for safe dosing of biopharmaceuticals which contain PS80 in patients.
Introduction
Polysorbate 80 (PS80; Tween 80) can improve the efficiency of drug delivery by enhancing solubility in aqueous solution. Topical or parenteral administration of PS80 has been associated with adverse allergic reactions, including both local injection site reactions, as well as systemic effects.1-7 Concentrations of PS80 in biopharmaceuticals range between 0.005% and 0.2% w/v, although higher levels are sometimes utilized, particularly in traditional Chinese herbal injectable medicines.8,9 Figure 1 depicts the peak dosage of PS80 (mg/kg) delivered by commercially approved biopharmaceuticals compared to traditional Chinese herbal injectable medicines. Biopharmaceuticals deliver less than 1 mg/kg PS80 per dose, yet some traditional medicines can deliver up to 100-fold higher dosage. The biopharmaceutical Stelara® (ustekinumab) delivers a 0.52 mg/kg dose of PS80, one of the highest intravenous (IV) PS80 level found in marketed biopharmaceutics (package insert). Comparatively, PS80 levels in Stelara are 14-fold lower than the lowest mg/kg dose of PS80 delivered in traditional Chinese injectable medicines via the same route. Exposure levels for subcutaneously (SC) administered biopharmaceuticals are even lower: the highest SC dosed biopharmaceutical, Rituxan Hycela, contains 0.2% PS80, resulting in 0.134 mg/kg PS80 per dose (package insert). Although systemic anaphylactoid reactions have only been linked to the high levels of PS80 (>1%) found in traditional Chinese medicines, these reactions have increased scrutiny of all biopharmaceutical formulations containing PS80. PS80 in some traditional Chinese medicines (TCM) is >100-fold higher than commercially approved biologics. The graph illustrates the peak PS80 dosage (mg/kg based on a 60 kg body weight) delivered by commercially approved biologics as compared to traditional Chinese herbal medicines. SM = small molecule; TCM, traditional Chinese medicine. Circled products are approved in China. Source: Review of prescribing information from marketed products.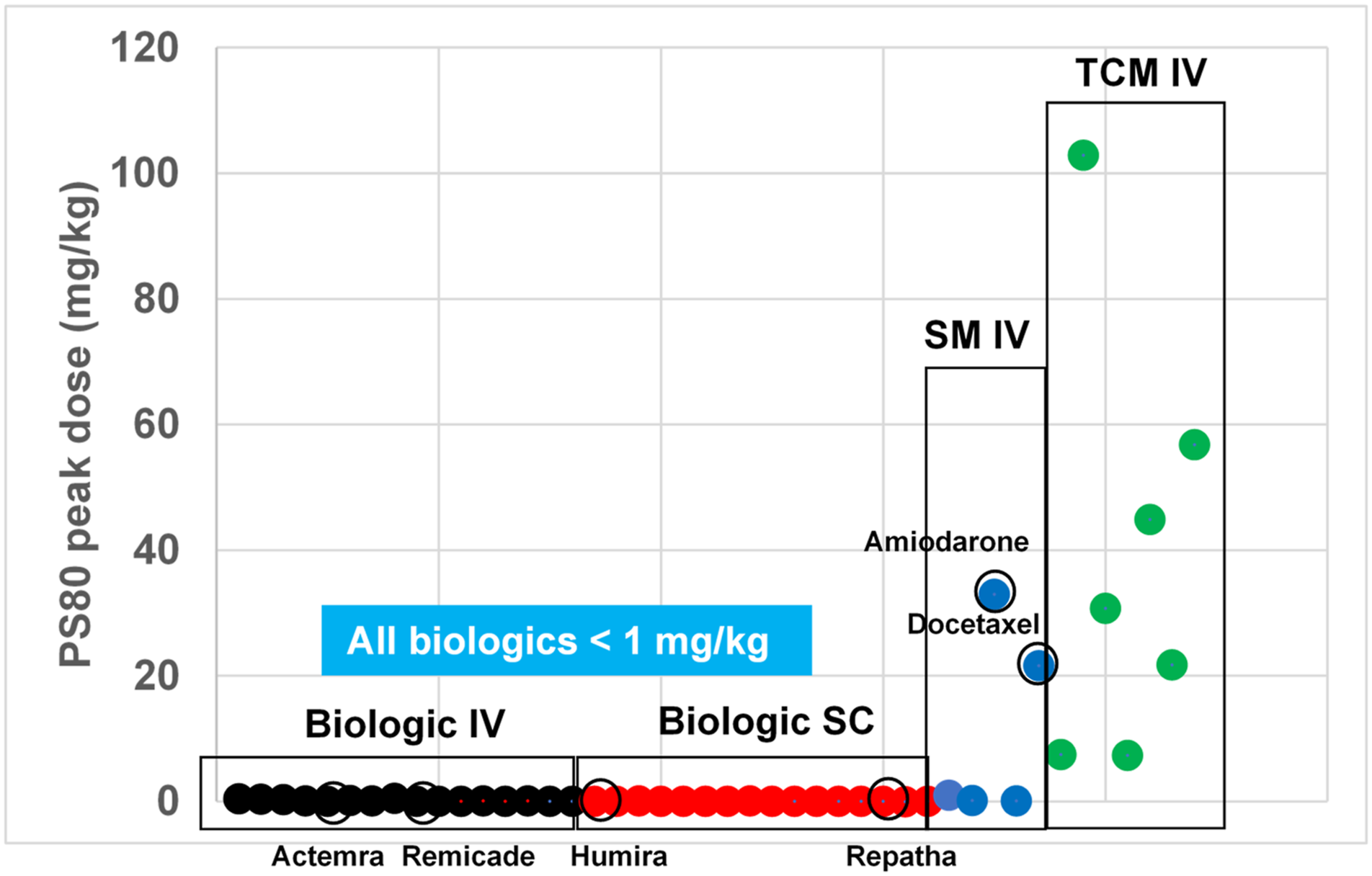
Reactions linked to PS80 are similar to immune allergic reactions that are observed following the first administration of an allergic reaction-triggering agent. However, unlike common hypersensitivity reactions, PS80 induces anaphylactoid-type reactions, which are defined by the direct release of histamine from mast cells (degranulation), leading to activation of the complement system, without the involvement of pre-existing immunoglobulin-E (IgE) antibodies.2,4,5,10–12 Characterization of PS80-related reactions
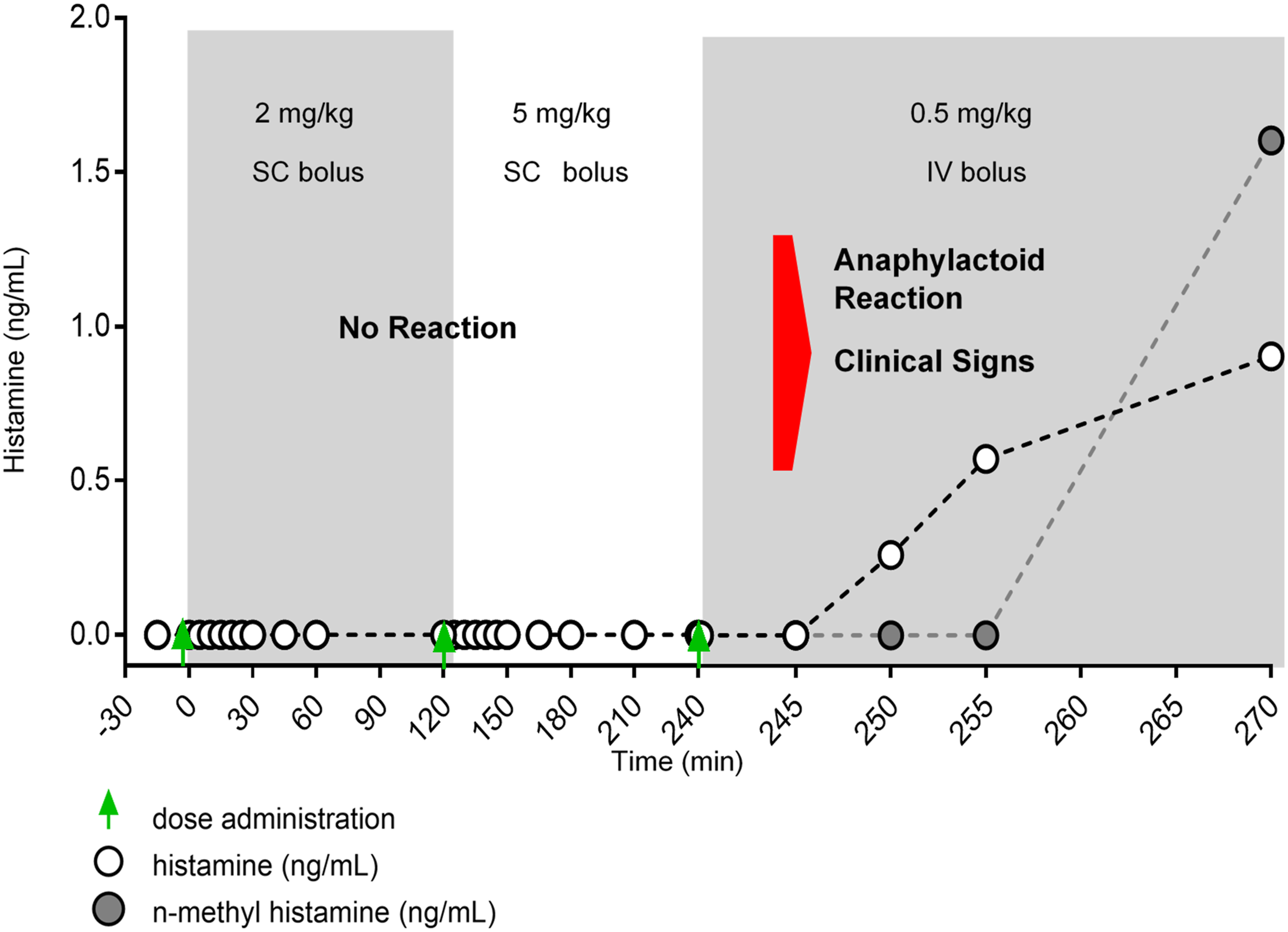
Despite the relevance of dogs as a model system for PS80 research, studies have reported conflicting evidence about the concentration of PS80 required to elicit a reaction, so the dose threshold associated with anaphylactoid reactions is difficult to determine. Differences in the observed adverse reactions from study to study may be directly related to the composition and quality of the specific PS80 used. For example, one study reported that 1 mg/kg 1% PS80 (USP-grade) administered IV to dogs did not result in a significant increase in plasma histamine or cardiovascular changes,
13
yet resulted in clinical signs. In a different study, dogs were treated with
Materials and Methods
Materials
USP-grade PS80 (CAS number 9005-65-6; Item number: 4117-04) used in this analysis was sourced from J.T.Baker/Avantor Performance Materials (Center Valley, PA) and met standard chemical specifications according to the certificate of analysis. For USP-grade PS80, oleic acid is certified to be greater than 58% of the final composition. For animal studies, PS80 was formulated to 0.25% in 0.9% sodium chloride solution (saline), filter sterilized, and stored at 4°C prior to dosing.
Animal Welfare
Canines were housed in an AAALAC, International accredited facility. Animals were cared for in accordance with the
Canines (
PS80 Administration in Anesthetized Dogs
Exploratory studies evaluated the effects of PS80, administered by IV infusion, IV bolus, or SC bolus, on hemodynamic parameters including heart rate (HR), left ventricular contractility (dP/dtmax) and mean arterial pressure (MAP) in an anesthetized beagle dog model. For each study, cardiovascular measurements were collected continuously. Cardiovascular data measurements were reduced into 30 second averages and then further reduced into 5-minute averages. A total of 2–3 animals were used for cardiovascular assessment for each route of administration. In addition, acute clinical observations of anaphylactoid responses (erythema, angioedema, and hyperemia) during dose administration were recorded. Animals were anesthetized using alpha-chloralose, instrumented with a solid-state pressure catheter in the left ventricle (LV) (contractility), fluid-filled catheter in the femoral artery (arterial pressure) and electrocardiogram (ECG) electrodes in a modified Lead II position. After cardiovascular and hemodynamic stabilization, animals received vehicle prior to doses of PS80 via IV or SC bolus or IV infusion followed by a monitoring period. If the highest planned PS80 dose chosen for any given animal did not induce an anaphylactoid reaction, a positive control dose of 1 mg/kg IV bolus was given to demonstrate that the animal could respond to PS80. Blood samples were collected throughout the study and processed to either serum or EDTA-plasma. Samples were snap-frozen in dry ice and stored at −80°C. LC-MS/MS was used to assay histamine and n-methyl histamine in serum or plasma samples. All dose formulation concentrations were within 10% of the target for solutions (1% or 0.25%) of PS80 before administration.
PS80 Administration in Conscious Dogs
A conscious dog study was completed at Charles River Laboratories (Laval, QC). Dogs (n = 10 IV bolus; n = 4 IV infusion; n = 10 SC bolus) were divided into Groups 1–5. Groups 1, 2, and 5 (n = 5 IV bolus; n = 5 SC bolus; n = 2 IV infusion, respectively) were monitored via a telemetry implant for cardiovascular changes. Arterial blood pressures were obtained from the transmitter catheter inserted into the femoral artery. Electrocardiograms were obtained from the biopotential leads, from the telemetry transmitter, in a Lead II configuration. During each treatment session, cardiovascular function (arterial blood pressure and ECG), body temperature, and physical activity were recorded continuously (except during designated study procedures or other procedures such as data backup) from at least 24 hours prior to the first dosing start until 24 hours after the last dosing start. Blood samples were collected from non-telemetered dogs in Groups 3, 4, and 5 (n = 5 IV bolus; n = 2 IV infusion; n = 5 SC bolus, respectively) to monitor histamine release and processed for plasma collection and analysis by LC-MS/MS or histamine HTRF ELISA. Animals received up to 4 doses, by IV bolus injection, 30 min IV infusion, or SC injection, each separated by a washout period of at least one day, until the maximum tolerated dose was reached. Positive dose thresholds were defined as all animals presenting a non-adverse allergic reaction to the PS80 dose based on clinical signs. Plasma samples from untreated dogs administered saline as a vehicle control or at predose timepoints were collected to establish baseline values for histamine and NMH levels.
Quantitative Plasma Histamine and NMH by Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS)
Chemical standards of histamine (H7250 from Sigma–Aldrich (St. Louis, MO)) and NMH (M7780 from Sigma–Aldrich) were spiked into blank dog serum or 40 mg/mL human serum albumin in phosphate buffered saline to generate calibration curve samples. 50 μL of serum samples or calibration curve samples were treated with 150 μL of extraction solvent which consisted of 1 μM tolbutamide (internal standard) in acetonitrile. The mixture was vortexed at 1500 rpm for 15 min and then centrifuged at 4500 rpm for 15 min. The supernatant was transferred to a new plate and subjected to liquid chromatography (LC)-selected reaction monitoring (SRM) MS analysis.
Chromatographic separation was performed using a Waters BEH HILIC column (2.1 × 150 mm; 1.7 μm; kept at 60°C) on a Shimadzu Nexera (Kyoto, Japan) high pressure liquid chromatography (HPLC) system. Solvent A contained 50% acetonitrile, 45% of water, and 5% of 100 mM ammonium formate 50:45:5 (v:v); solvent B contained 95% of acetonitrile and 5% of 100 mM ammonium formate 95:5 (v:v); both solvents were maintained at pH 3.22. The flowrate was 600 μL/min, and gradient was set as follows: 83%B from 0–0.4 min, ramped down to 0.1%B from 0.4–2 min, maintained at 0.1%B from 2–3 min and then ramped up to 83%B at 3.1 min for equilibration. Detection and quantitation of histamine and NMH was carried out on a QTRAP 6500 mass spectrometer (Sciex, Framingham, MA) equipped with an electrospray ionization source, operating in positive ionization mode.
Data was analyzed using the Quantitate module embedded in Analyst 1.7 (Sciex). Peaks were integrated and the peak area ratios of analyte to internal standard were used for quantitation. Linear regression (1/x2 weighting,
Quantitative Histamine Analysis by Homogenous Time Resolved Fluorescence (HTRF) Methodology
Preparation of the standard curve and samples was conducted according to CisBio Bioassays’ Histamine-Dynamic Assay manufacturer’s instructions. Briefly, histamine is detected in a competitive assay by using an anti-histamine antibody labeled with Europium cryptate (donor) and histamine labeled with d2 (acceptor). When the two dyes are in close proximity, the excitation of the donor with a laser or flash lamp triggers a Fluorescence Resonance Energy Transfer (FRET) towards the acceptor, which in turn fluoresces at 665 nm. If histamine is present in the sample, it competes with the binding between the two HTRF detection solutions and prevents FRET from occurring.
Results
Cardiovascular Changes Correlate with Increased Histamine Levels in Anesthetized Dogs with a Clinical Anaphylactoid Reaction
Positive anaphylactoid reactions as defined by marked cardiovascular changes, including rapid increases in heart rate and transient but inconsistent changes in MAP, were observed in all anesthetized dogs after IV administration of 1 mg/kg PS80 (Figure 2). Clinical observations consistent with an allergic response were also observed and included skin erythema of the entire ventrum; moderate skin erythema from the ventral cervical area to umbilical region and severe skin erythema in the inguinal and hypogastric regions, severe angioedema of the muzzle, periorbital region, ear pinnae, front limbs and front paws, moderate angioedema of the rear limbs and rear paws and hyperemia of the upper labia, and oral mucosa. LC-MS/MS methodology detected transient increased histamine and its degradation product, NMH, at the onset of clinical signs and cardiovascular changes (Figure 3); transient increased histamine was confirmed via HTRF analysis (data not shown). Consistent with NMH biology as a metabolite of histamine with a longer half-life, the maximum levels of NMH occurred at a later timepoint and remained increased for a longer period of time to the increase in histamine (Figure 3). Representative CV observations from PS80 research in anesthetized dogs. Increased heart rate (upper left panel) and decreased mean arterial pressure (upper right panel), systolic blood pressure (lower left panel), and diastolic blood pressure (lower right panel) are hallmarks of anaphylactoid reactions and coincide with the onset of clinical symptoms of anaphylactoid reactions (denoted by the red arrow). 1 mg/kg IV bolus was used as a known positive dose level for anaphylaxis in subsequent experiments. Representative data from n = 1 dog. Representative data showing increased histamine levels assayed via liquid chromatography mass spectrometry (LC-MS/MS) analysis. LC-MS/MS analysis was used to measure the histamine and n-methyl histamine (NMH, the degradation product of histamine) levels in dog serum or plasma, respectively. Clinical observations (onset denoted with the red arrow) consistent with an anaphylactoid reaction correlated with an increase in measured histamine and/or NMH levels. NMH levels peaked later than histamine levels, consistent with NMH biology as a histamine degradation product. Representative data from n = 1 dog.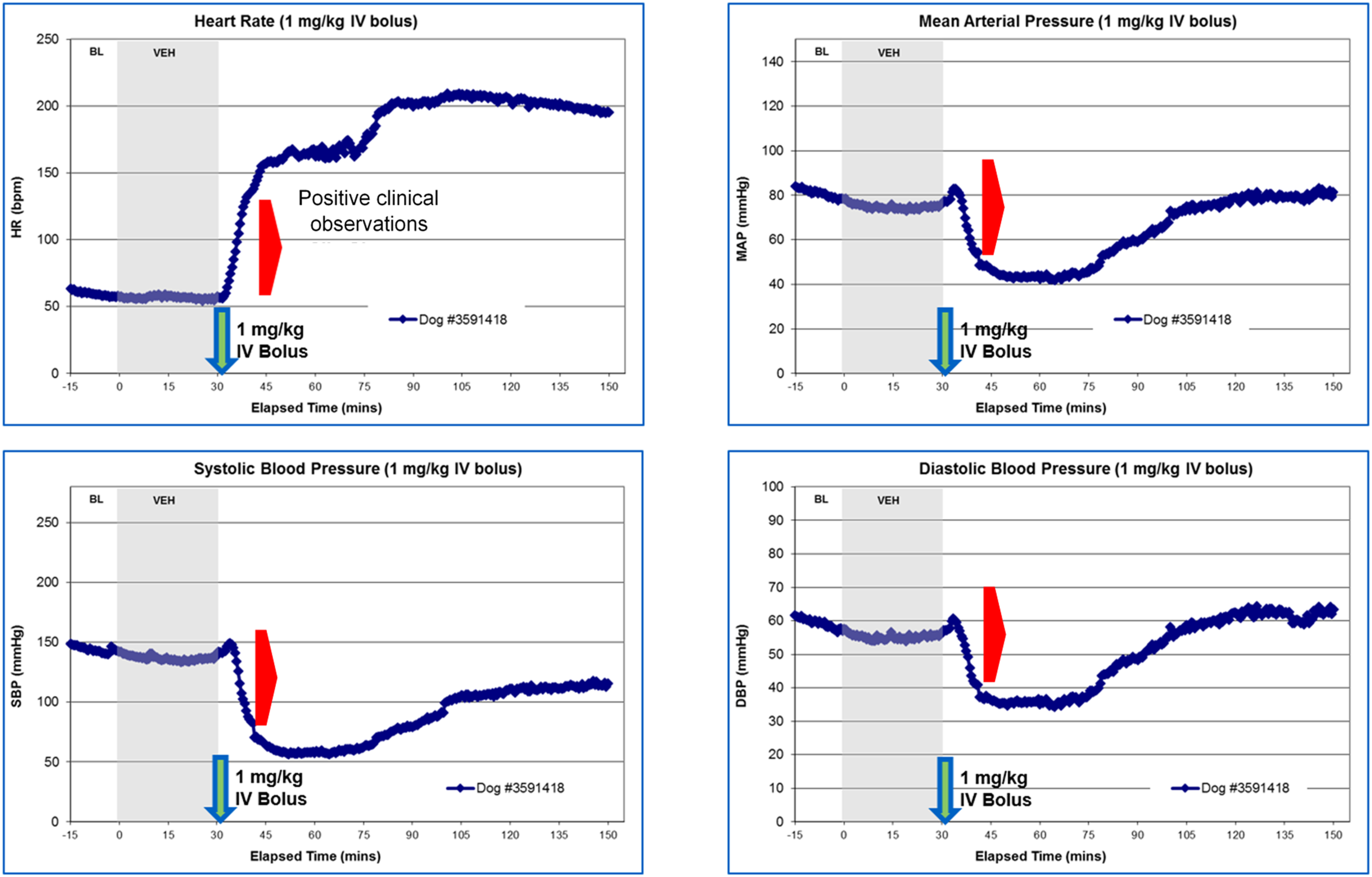
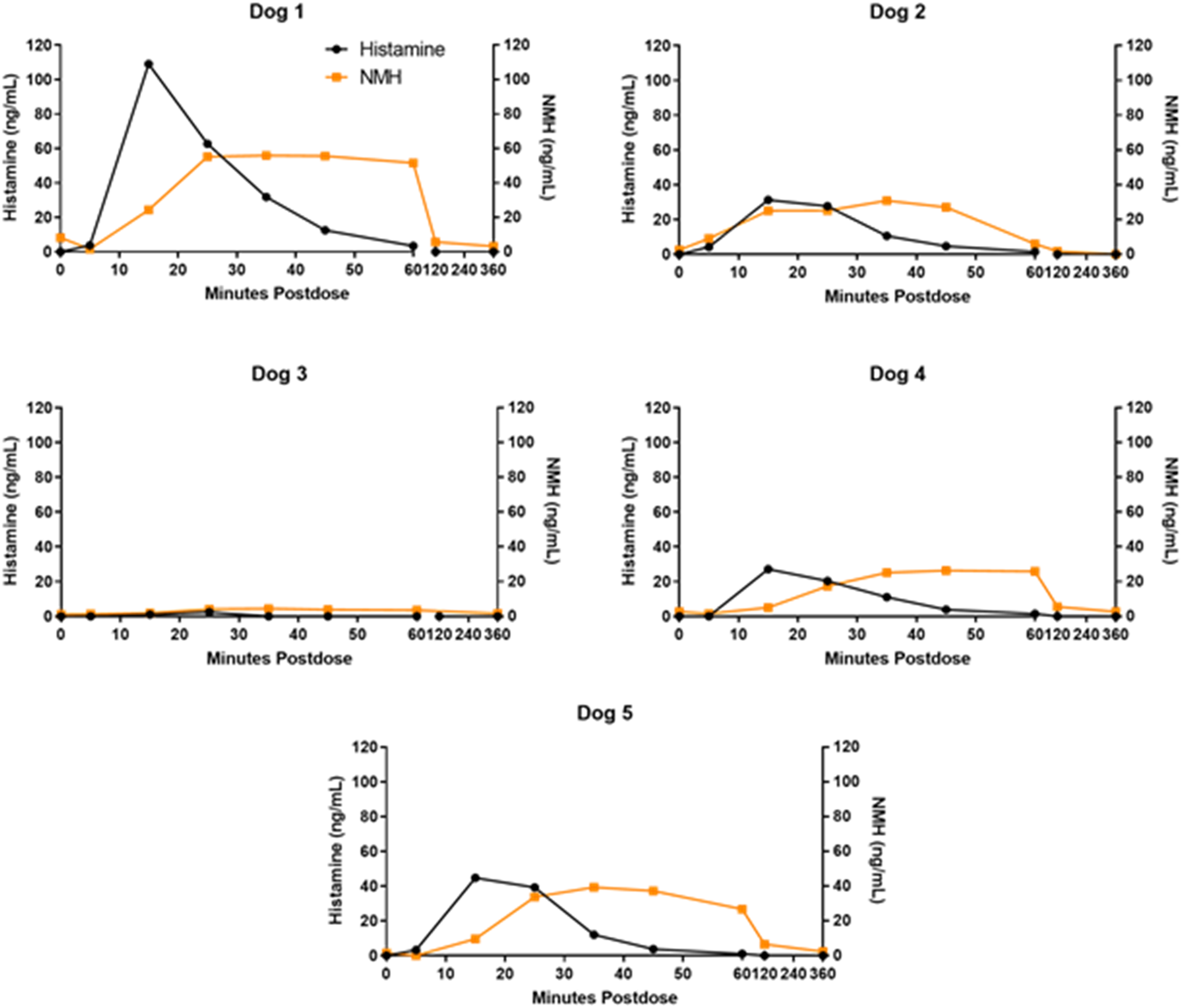
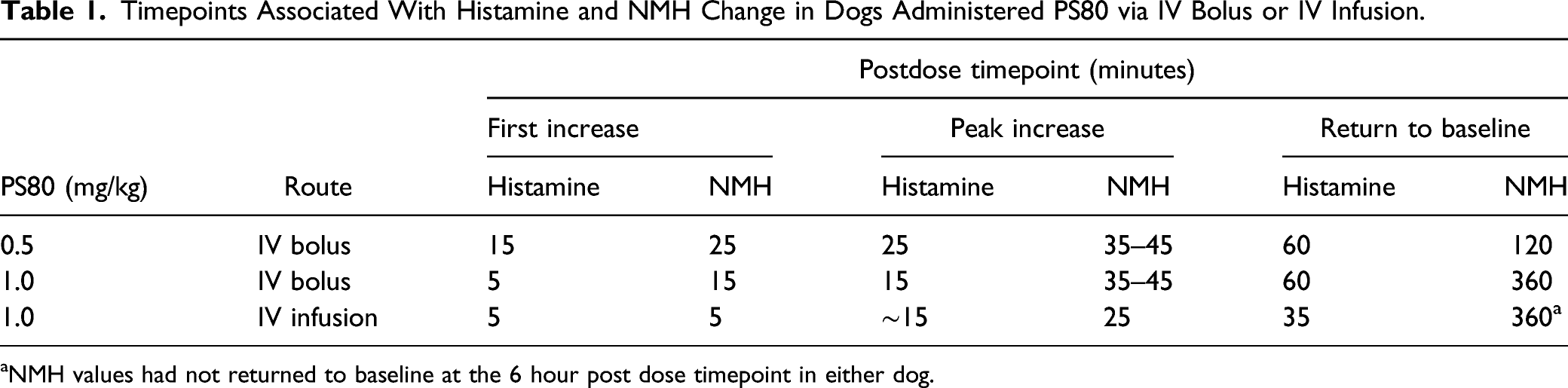
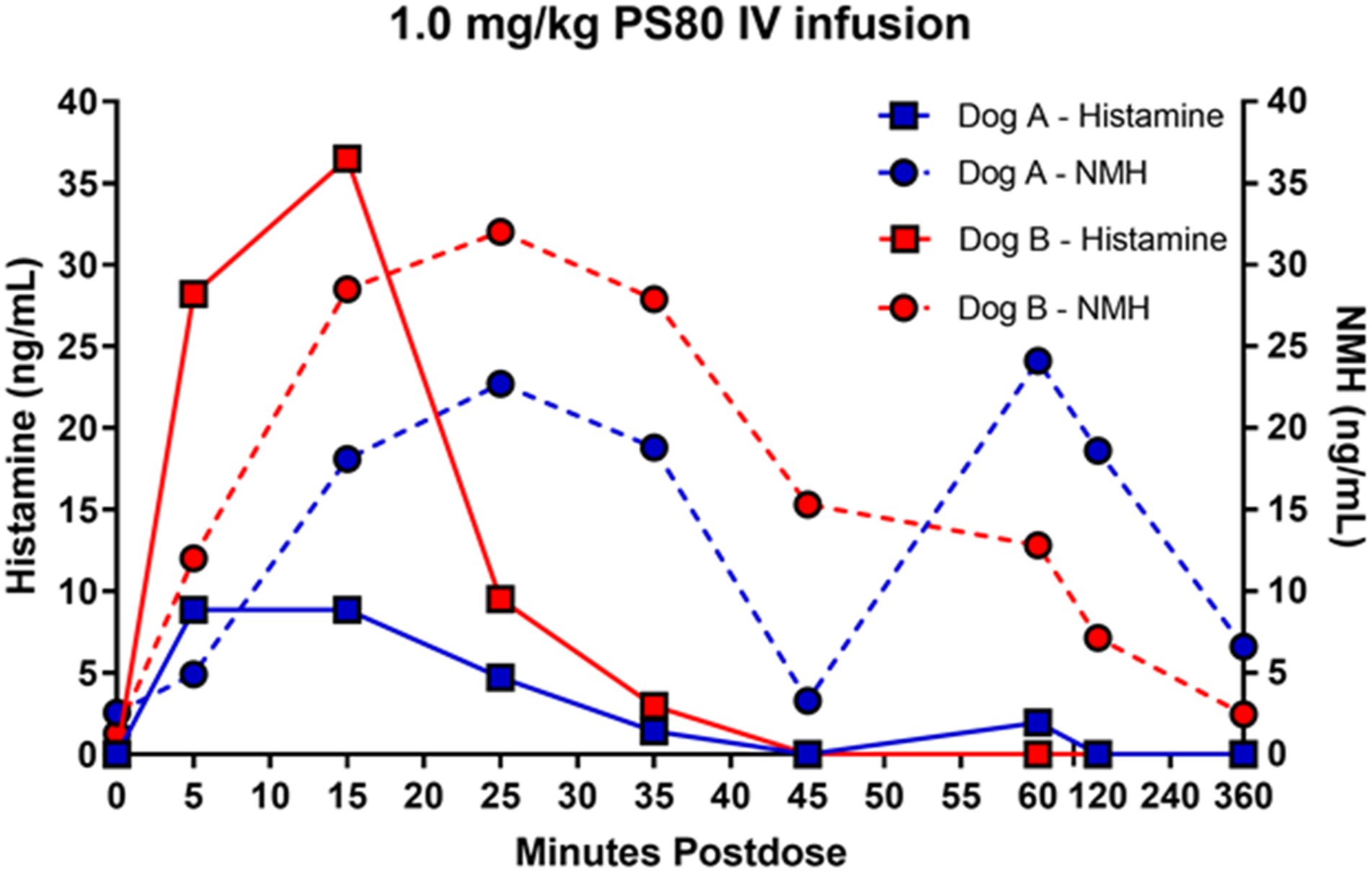
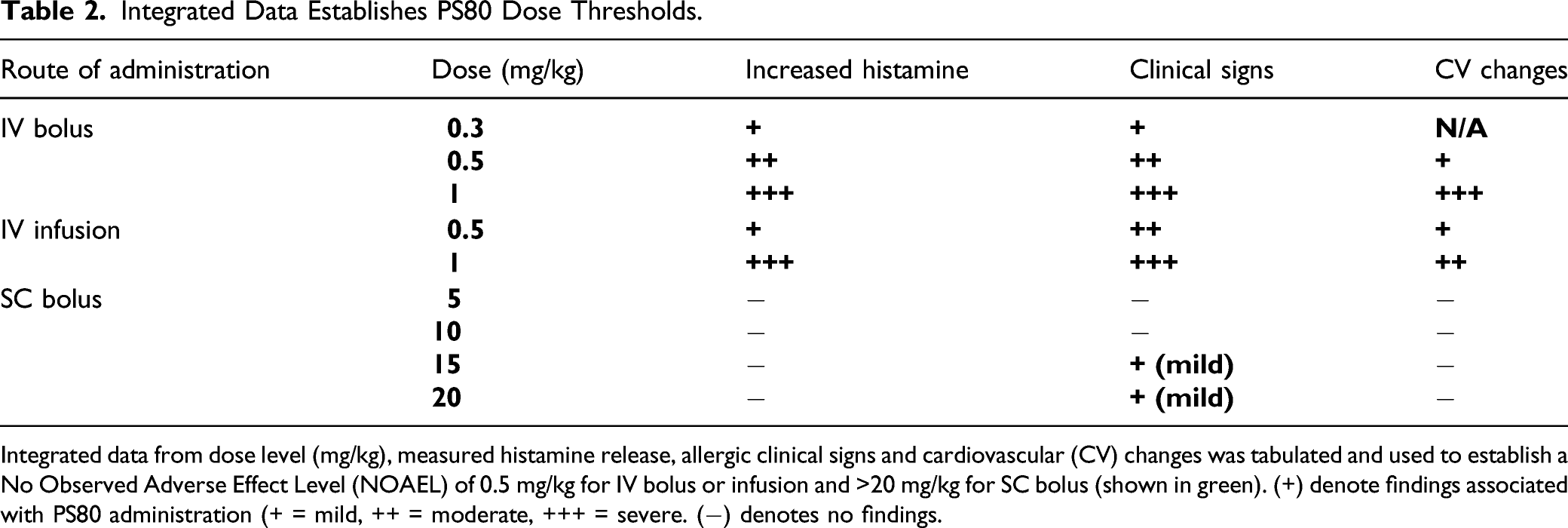
A Comprehensive Study in Conscious Dogs Defines the Threshold for Dose using Multiple Dosing Regimens
To further assess the potential for PS80-related anaphylactoid reactions, additional studies in conscious dogs were initiated to analyze a combination of cardiovascular data, clinical signs, histamine and NMH biomarkers. Plasma histamine levels were analyzed by HTRF, and increased in a dose dependent manner when PS80 was administered via IV bolus at all dose levels tested (0.3, 0.5, and 1 mg/kg) (Figure 4A-4D); these findings were correlated with analysis of plasma histamine and NMH levels by LC-MS/MS (Figure 5). Plasma histamine, measured by HTRF analysis, increased when PS80 was administered via IV infusion at 1 mg/kg, the highest dose level tested, but was comparable to baseline histamine levels at lower PS80 dose levels of 0.3 or 0.5 mg/kg (Figure 4C and 4D). There was no increased plasma histamine concentration in dogs administered PS80 via the SC route at the highest dose level tested, 20 mg/kg (Figure 4A and 4B). However, high animal variability relative to the magnitude of histamine or NMH response was observed in all five dogs at all dose levels (Figure 5). In general, histamine increased earlier, reached peak values earlier, and returned more rapidly to baseline compared to the NMH (Figure 5, Table 1). A similar pattern of histamine and NMH change was also identified in dogs administered PS80 via IV infusion (Figure 6). The time to peak histamine levels was generally 15–25 minutes after PS80 administration (Table 1). Allergic clinical signs were observed with doses of 1 mg/kg PS80 via IV bolus or IV infusion; clinical signs were not observed unequivocally for SC injections up to the highest dose tested (20 mg/kg). These clinical observations were occasionally associated with transient or delayed arterial blood and pulse pressure decreases following IV and SC bolus injections, deemed non-adverse. Based on the integrated data from both anesthetized and conscious dogs the No Observed Adverse Effect Level (NOAEL) for PS80 was defined as 0.5 mg/kg for IV bolus or IV infusion, and up to 20 mg/kg for SC bolus (Table 2). IV bolus and IV infusion of PS80, but not SC injection, resulted in increased histamine levels in conscious dogs. Histamine levels were measured by HTRF analysis after conscious dogs were treated with PS80. (A and B) show histamine levels after SC injection, which did not exhibit increases in baseline histamine release. (C and D) depict detected histamine increase after IV bolus injection (C) or after IV infusion (D). Histamine level was markedly increased after IV bolus injection of 1 mg/kg PS80. Green arrows and dashed vertical line denote the time of PS80 administration. Plasma histamine and NMH changes in conscious dogs administered PS80 via 1 mg/kg IV bolus injection. Graphs of individual histamine and NMH levels from 5 dogs show variability after administration of PS80 via IV bolus administration. For graphing purposes, all timepoints below the limit of quantification (BLOQ) were assigned a value of 0.00. Histamine levels are graphed in black; NMH levels are graphed in orange. Timepoints Associated With Histamine and NMH Change in Dogs Administered PS80 via IV Bolus or IV Infusion. aNMH values had not returned to baseline at the 6 hour post dose timepoint in either dog. Plasma histamine levels increase prior to NMH in two conscious dogs administered PS80 via IV infusion. Graph shows individual histamine and NMH levels in two dogs after a 1.0 mg/kg PS80 IV infusion. Solid lines denote histamine levels; dotted lines denote NMH levels. Integrated Data Establishes PS80 Dose Thresholds. Integrated data from dose level (mg/kg), measured histamine release, allergic clinical signs and cardiovascular (CV) changes was tabulated and used to establish a No Observed Adverse Effect Level (NOAEL) of 0.5 mg/kg for IV bolus or infusion and >20 mg/kg for SC bolus (shown in green). (+) denote findings associated with PS80 administration (+ = mild, ++ = moderate, +++ = severe. (−) denotes no findings.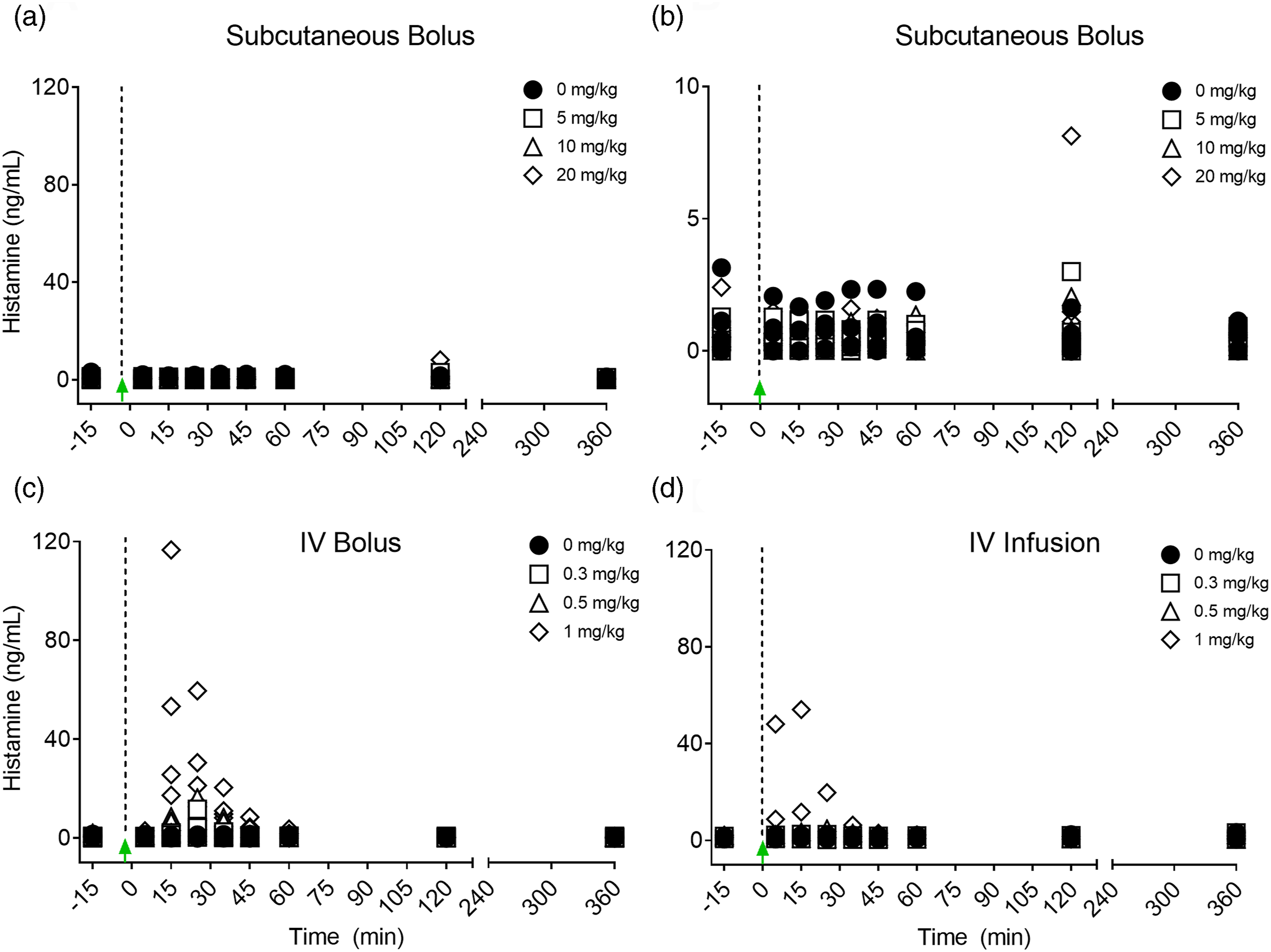
Discussion
Traditional Chinese Medicines and Biopharmaceuticals Utilize PS80 for Different Functions
All marketed biologic formulations of PS80 have <1 mg/kg exposure at peak dosage, whereas traditional Chinese herbal injectable medicines contain much higher levels of PS80, up to 100 mg/kg (Figure 1). This difference in concentration highlights that PS80 is utilized as a solubilizer in traditional herbal injectable medicines but as a stabilizer in biopharmaceuticals.
When PS80 is used in high concentrations as a solubilizer, the complexity of PS80 composition and the lack of consistency between different manufacturers of the product becomes an important factor in adverse reactions. However, it is difficult to link specific components of PS80 to adverse reactions. Because of the diverse synthetic processes used by different companies to obtain the final product, PS80 composition is not identical from manufacturer to manufacturer, 14 and this issue represents an important consideration in safety evaluations. For example, a recent study assessed the lipid composition of PS80 from seven Chinese suppliers through high resolution mass spectrometry techniques. 20 These analyses demonstrated that PS80 from different suppliers produced final products with differences in the lipid composition and proportion; further study also indicated that impurities were present in some of the samples. 20 In a different study, PS80 from Acros and Fisher suppliers were analyzed using similar high resolution mass spectrometry techniques 21 and several differences were observed in the lipid polymeric species between the two samples. However, due to the methodology used in these studies, it is difficult to quantify the range of composition difference to be expected from manufacturer to manufacturer. These studies underscore that diversity in PS80 formulation leads to difficulty in drawing broad conclusions about product safety from literature sources.
Product impurities, such as peroxide, oxirane,8,14 and residual fatty acids that are not completely consumed in the synthesis reaction have been identified, and are potential triggers of anaphylactoid reactions. For example, ten batches of PS80 solutions from various suppliers were assessed for three impurities (ethylene glycol, 2-chloroethanol, and hydrogen peroxide). 22 The study found that three specific batches and one impurity, hydrogen peroxide, induced anaphylactoid reactions in a novel microplate-based zebrafish assay. 22 The samples that induced these reactions were much higher in hydrogen peroxide residue and peroxide value. Other canine studies have also found that there is a correlation between hydrogen peroxide content and the occurrence of anaphylactoid reactions.3,10 In particular, one study highlighted that reactions to PS80 are closely related to the purity of the product. 10 This study observed that impurities including free oleic acid, hydrogen peroxide, and ethylene oxide may trigger anaphylactoid reactions. 10 Thus, there appears to be a link between adverse reactions and poor PS80 purity and controls on raw materials used in the production of PS80 are critical to reduce safety concerns. In our studies, a NOAEL was established for USP-grade PS80 administered via IV and SC routes of administration (Table 1), showing that USP-grade product is unlikely to cause concerns at the low levels of PS80 used in biopharmaceuticals.
PS80 is also commonly used as an excipient in oral drug formulations; the FDA Inactive Ingredient database lists the highest oral maximum daily exposure for PS80 as 350 mg/day. Previous research examined 0.2% PS80 (10 mg/kg) by oral gavage in rats, mice, dogs and cynomolgus monkeys, without significant clinical observations. 23 Furthermore, in preclinical research, oral studies frequently utilize concentrations of PS80 up to 1% without anaphylactoid clinical observations. In ten different studies, vehicle control dogs were dosed with 1% PS80 orally, 12/66 had clinical signs of an allergic reaction post-dosing (red skin), suggesting that 1% oral PS80 is unlikely to cause severe evidence of anaphylactoid reactions (Amgen internal data). Thus, 1% PS80 is acceptable in oral formulations for preclinical studies in dogs. Higher sensitivity thresholds observed in both SC and oral administration to PS80 are connected to pharmacokinetic relationships after administration; our data suggests that anaphylactoid reactions are driven by the peak concentration achieved (Cmax) rather than the concentration of PS80 vs time after dosage (AUC).
Conclusions
Based on the results of these studies, significant safety margins for biopharmaceutical IV and SC products were defined as up to 60-fold for marketed IV products and 24,000-fold marketed SC products (Figure 7). The level of PS80 in biotherapeutics is generally safe, and based on the margins that we have established, there is a threshold level for PS80-induced anaphylaxis in the most sensitive species (dog) to PS80-based effects. PS80 safety margins for marketed biologic products. Graph summarizes calculated safety margins for PS80 exposure in marketed biopharmaceutical products administered by IV (black dots) or SC (red dots). The minimum safety margin was calculated for SC dosing because there was no reaction at the highest dose tested. Circled products are approved in China.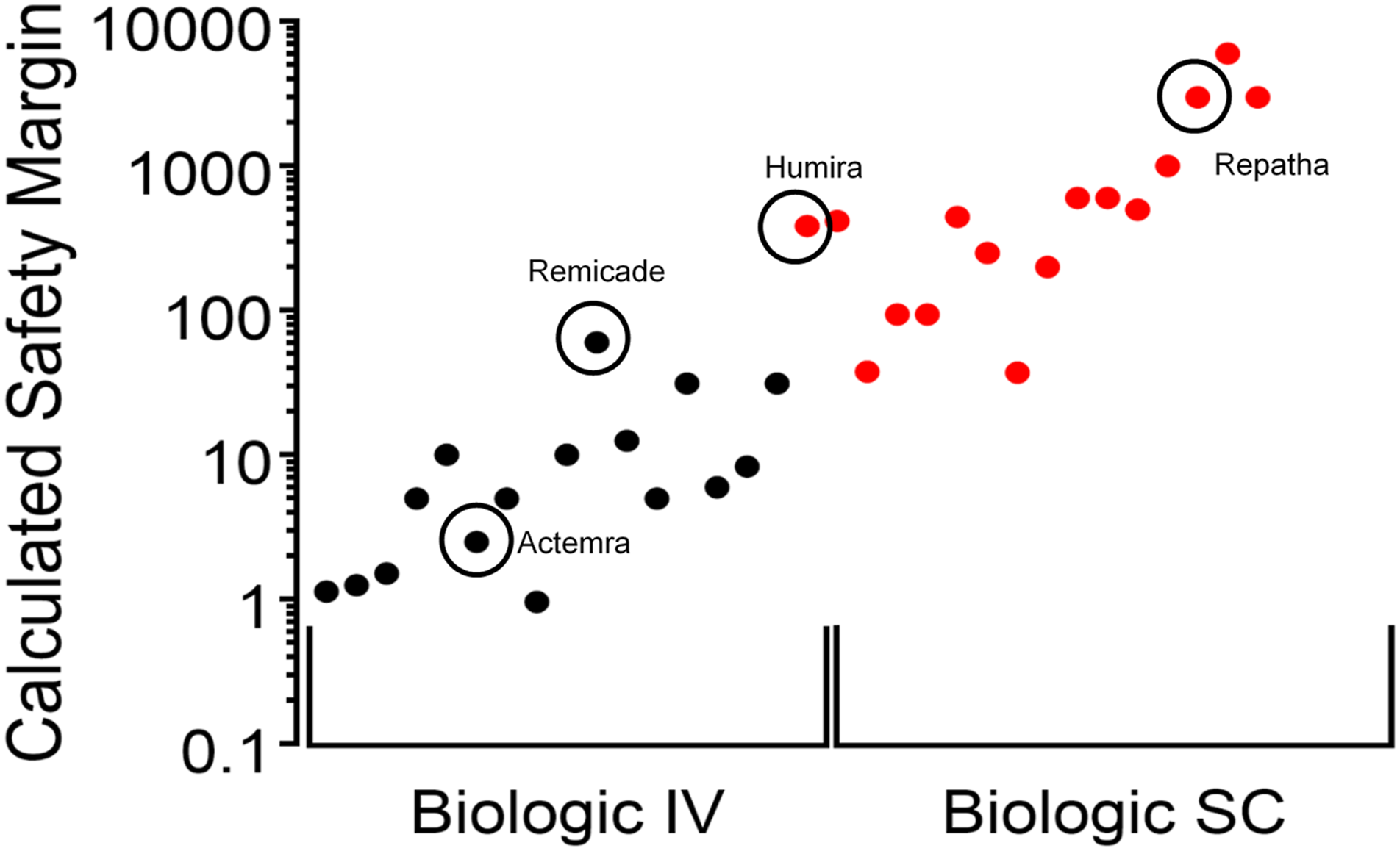
Footnotes
Acknowledgments
We would like to thank Geil Merana and Nikita Kolhatkar for contributing to literature analysis for this work.
Author Contributions
Holbrook, K. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; Andrews, D. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript; Sutherland, W. contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Coppi, A. contributed to design, contributed to analysis and interpretation, and critically revised manuscript; Barry, S. contributed to acquisition and critically revised manuscript; Escamilla, M. contributed to acquisition and critically revised manuscript; Authier, S. contributed to conception and design, contributed to interpretation, and critically revised manuscript; Miraucourt, L. contributed to interpretation and critically revised manuscript; Xie, F. contributed to acquisition and analysis and critically revised manuscript; Rock, B. contributed to design, contributed to acquisition and analysis, and critically revised manuscript; Bussiere, J. contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
