Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) assessed the safety of 47 Citrus peel-derived ingredients, which are most frequently reported to function in cosmetics as skin conditioning agents. The Panel reviewed the available data to determine the safety of these ingredients. Because final product formulations may contain multiple botanical ingredients, each containing similar constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should use good manufacturing practices to limit impurities that could be present in botanical ingredients. The Panel concluded that Citrus peel-derived ingredients are safe in the present practices of use and concentration in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen (5-MOP).
Keywords
Introduction
Citrus peel-derived ingredients are most frequently reported to function in cosmetics as skin conditioning agents, according to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary) (Table 1).
1
This report assesses the safety of the following 47 ingredients: Citrus Aurantifolia (Lime) Peel Citrus Aurantifolia (Lime) Peel Extract Citrus Aurantifolia (Lime) Peel Powder Citrus Aurantifolia (Lime) Peel Water Citrus Aurantium Amara (Bitter Orange) Peel Citrus Aurantium Amara (Bitter Orange) Peel Extract Citrus Aurantium Amara (Bitter Orange) Peel Powder Citrus Aurantium Bergamia (Bergamot) Peel Water Citrus Aurantium Dulcis (Orange) Peel Extract Citrus Aurantium Dulcis (Orange) Peel Powder Citrus Aurantium Dulcis (Orange) Peel Wax Citrus Aurantium Sinensis Peel Extract Citrus Aurantium Tachibana Peel Extract Citrus Depressa Peel Extract Citrus Depressa Peel Powder Citrus Grandis (Grapefruit) Peel Citrus Grandis (Grapefruit) Peel Extract Citrus Grandis (Grapefruit) Peel Powder Citrus Hassaku/Natsudaidai Peel Powder Citrus Iyo Peel Extract Citrus Iyo Peel Water Citrus Jabara Peel Extract Citrus Jabara Peel Powder Citrus Jabara Peel Water Citrus Junos Peel Extract Citrus Junos Peel Powder Citrus Junos Peel Water Citrus Limon (Lemon) Peel Citrus Limon (Lemon) Peel Extract Citrus Limon (Lemon) Peel Powder Citrus Limon (Lemon) Peel Water Citrus Limon (Lemon) Peel Wax Citrus Natsudaidai Peel Extract Citrus Nobilis (Mandarin Orange) Peel Extract Citrus Nobilis (Mandarin Orange) Peel Powder Citrus Paradisi (Grapefruit) Peel Extract Citrus Reticulata (Tangerine) Peel Extract Citrus Reticulata (Tangerine) Peel Powder Citrus Shunkokan Peel Extract Citrus Sunki Peel Extract Citrus Tachibana/Reticulata Peel Powder Citrus Tangelo Peel Powder Citrus Tangerina (Tangerine) Peel Citrus Tangerina (Tangerine) Peel Extract Citrus Unshiu Peel Extract Citrus Unshiu Peel Powder Citrus Unshiu Peel Water Definitions and Functions of Citrus Peel-Derived Ingredients.
1

The Panel has previously reviewed the safety of Citrus-derived peel oils and Citrus fruit-derived ingredients in separate assessments and concluded that 14 Citrus-derived peel oil ingredients and 80 Citrus fruit-derived ingredients are safe for use in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen (5-MOP).2,3 Of note, the Panel concluded that Citrus-derived peel oils could potentially promote tumors if the formulation produces irritation. 3 The safety of Citrus flower- and leaf-derived ingredients and Citrus plant- and seed-derived ingredients are published in separate reports.
Some of the Citrus peels that are used to derive the ingredients described in this safety assessment are consumed as food. The U.S. Food and Drug Administration (FDA) determined that the use of some Citrus peels as direct food substances are generally recognized as safe (GRAS). Additionally, essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus peels are GRAS for their intended use in foods for human and animal consumption. Daily consumption of these GRAS foods would result in much larger systemic exposures than what is expected from use in cosmetic products, even if there was 100% absorption from cosmetics. Thus, the systemic toxicity potential of Citrus peel-derived ingredients via oral exposure is not addressed further in this report. The primary focus of this safety assessment is the review of the safety of topical exposure.
Citrus Ingredients That Potentially Function Solely as Fragrance Ingredients.

Botanical ingredients are complex mixtures of many constituents, some of which have the potential to cause toxic effects; for example, bergapten (also known as 5-methoxysporalen or 5-MOP) is a naturally occurring phototoxic furanocoumarin (psoralen) in some Citrus ingredients. In this assessment, the Panel is reviewing the potential toxicity of each Citrus peel-derived ingredient as a whole, complex substance. Except for specific constituents of concern that have been identified, the Panel is not reviewing the potential toxicity of the individual constituents of the Citrus peels from which the ingredients in this report are derived.
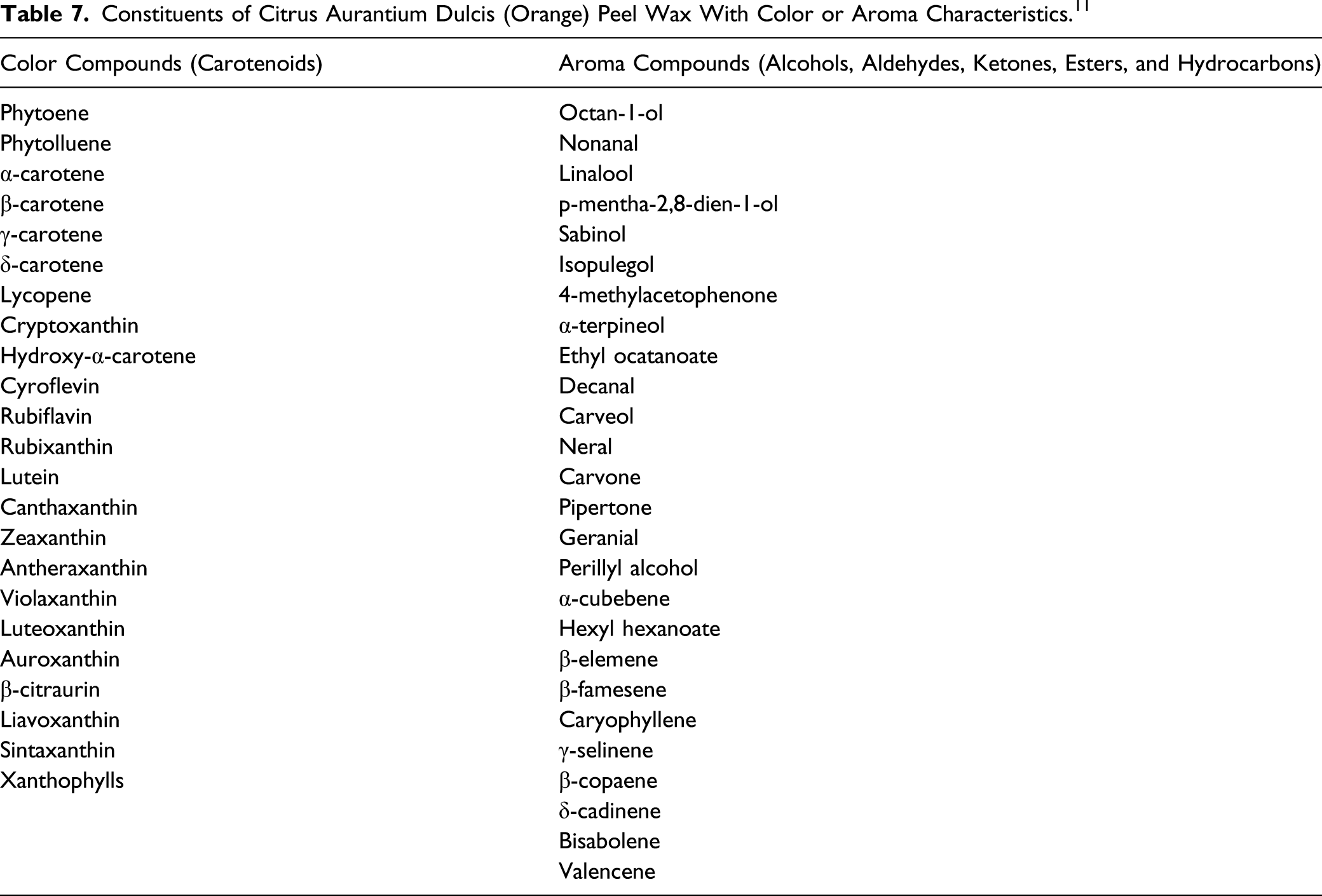
Review of Citrus Genus Species Names. 4
Chemistry
The definitions and functions of the citrus-derived ingredients included in this report are provided in Table 1. The definition indicates the part(s) of the plant from which an ingredient is obtained. In some cases, the definition provides insight on the method(s) of manufacture.
Physical and Chemical Properties
Physical and Chemical Properties of Citrus Aurantium Dulcis (Orange) Peel Wax.

Method of Manufacturing
According to the Dictionary, waters are prepared from leaves, stems, flowers, bark, roots, or other parts of a plant or the whole plant. 5 Waters, and essential oils, are prepared by a number of processes, but the most widely used method is steam distillation. The condensate from steam distillation produces two distinct fractions that contain the volatile ingredients from the plant. The water insoluble fraction contains the “oil.” The water soluble fraction includes the term “water” in the INCI name.
Citrus Aurantium Amara (Bitter Orange) Peel Extract
A supplier reported that its Citrus Aurantium Amara (Bitter Orange) Peel Extract products are produced by extracting dried raw peels from Citrus aurantium amara with an ethanol solution. 6 The resultant materials then undergo filtration, concentration, sedimentation, and adjustment before packaging. One product (a powdered form) has anhydrous sodium sulfate added as a vehicle prior to packaging.
Another supplier reported that its products are produced by extracting ripe pericarp from Citrus aurantium Linne (Rutaceae) with either an ethanol or a 1,3-butylene glycol solution and then filtering the extract. 7
Citrus Aurantium Dulcis (Orange) Peel Wax
According to data provided by a supplier, Citrus Aurantium Dulcis (Orange) Peel Wax is a by-product from orange essential oil and orange juice production. 8 Citrus Aurantium Dulcis (Orange) Peel Wax is obtained by distillation of citrus terpenes and orange essential oil from orange fruit peels. The crude wax is processed by physical methods only, and is further refined with various absorbents and filtration. The deodorization process removes all terpenes and most of the essential oil components.
Citrus Reticulata (Tangerine) Peel Extract
A supplier reported that Citrus Reticulata (Tangerine) Peel Extract is produced through the hydroalcoholic extraction of tangerine peel, which is then concentrated until it contains at least 98% of the flavonoid luteolin. 9 The resultant product is a powder.
Another supplier reported that its products are produced by extracting ripe peels of Citrus reticulata Blanco (Rutaceae) with either an ethanol solution or 1,3-butylene glycol solution and then filtering. 7
Citrus Unshiu Peel Extract
According to a supplier, Citrus Unshiu Peel Extract is obtained by maceration of fine-cut Citrus unshiu peel in water and ethanol. 10 The resultant product is filtered and dried. Another supplier reports that its Citrus Unshiu Peel Extract products are produced by extracting dried raw peels with either an ethanol solution or a 1,3-butylene glycolic solution. 6 The resultant materials undergo various forms of filtration, concentration, sedimentation, and adjustment before packaging. One product (a powdered form) has anhydrous sodium sulfate added as a vehicle prior to packaging, while another has squalene added.
Constituents/Composition
The Citrus ingredients are complex botanicals made of numerous constituents.
Citrus Aurantifolia (Lime) Peel Extract
Citrus Aurantium Amara (Bitter Orange) Peel Extract
A supplier reported that their Citrus Aurantium Amara (Bitter Orange) Peel Extract products contain flavonoids, sugar, and/or hesperidin. 6
A supplier of a product containing 1.55% Citrus Aurantium Amara (Bitter Orange) Peel Extract, 25.81% alcohol, and 72.64% water stated that the product contained 2.8 ppm 5-MOP. 7 No other analysis was performed on this product. The same supplier has a product containing 2.0% Citrus Aurantium Amara (Bitter Orange) Peel Extract, 29.4% butylene glycol, and 68.6% water.
Citrus Aurantium Dulcis (Orange) Peel Wax
Based on data provided by a supplier, Citrus Aurantium Dulcis (Orange) Peel Wax is a water-free substance unlikely to be contaminated by microorganisms (bacteria, yeast, or fungi) because of the high temperature, filtration, and absorbents used during processing. 8
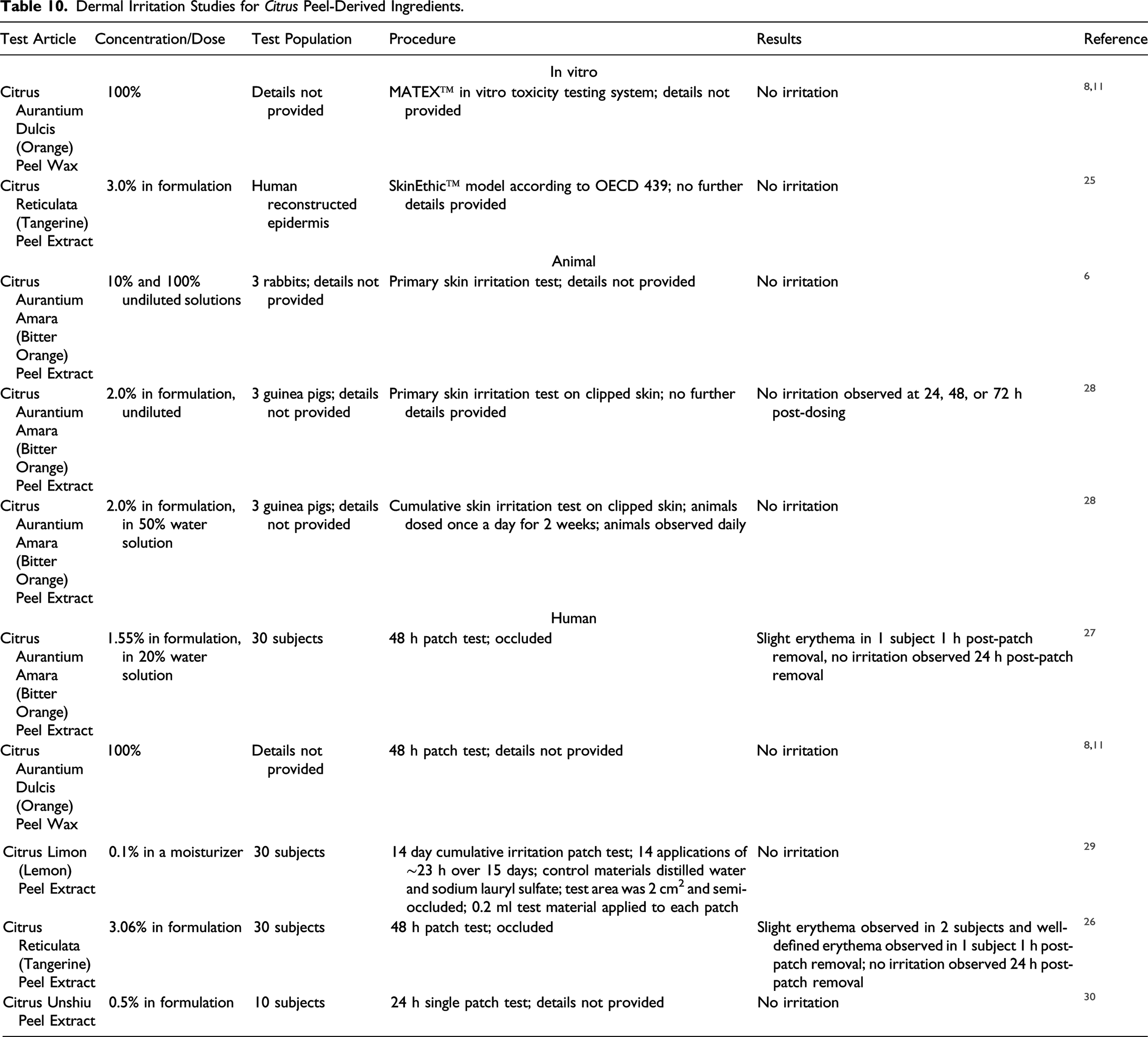
The provided data indicate that Citrus Aurantium Dulcis (Orange) Peel Wax consists of approximately 60% esters (C42-C60), 18% phytosterols (beta-sitosterol, stigmasterol), 3% sterol esters, 8% free fatty acids, 5% hydrocarbons, and 4% free fatty alcohols. 8 Approximately 50% of Citrus Aurantium Dulcis (Orange) Peel Wax consists of unsaturated monoesters of unsaturated fatty acids and long-chain alcohols, with the fatty acids consisting mostly of linoleic, oleic, linolenic, arachidic, and erucic acids. 11 The fatty alcohol portion of the ester is mostly dotriacontanol (C32) and tetratricontanol (C34).
Primary Chemical Composition of Citrus Aurantium Dulcis (Orange) Peel Wax by Percent. 11

Constituents of Citrus Aurantium Dulcis (Orange) Peel Wax With Color or Aroma Characteristics. 11
Citrus Reticulata (Tangerine) Peel Extract
A supplier of a product containing 3.06% Citrus Reticulata (Tangerine) Peel Extract, 25.41% alcohol, and 71.53% water reported that its product did not contain furanocoumarins. 7 Another product of this supplier contained 3.0% Citrus Reticulata (Tangerine) Peel Extract, 29.1% water, 67.9% butylene glycol, and no furanocoumarins. No further composition data were provided on these products.
Citrus Unshiu Peel Extract
According to a supplier, Citrus Unshiu Peel Extract is composed of pectin, peptides and amino acids, essential oils, phenolic acids, flavonoids (flavonols, flavones, and flavonones), carotenoid pigment, and tocopherol analogues. 10 Another supplier reports that its Citrus Unshiu Peel Extract products contain flavonoids, sugar, and/or hesperidin. 6 One product was reported to contain essential oil component (no further details provided).
Impurities
Citrus Aurantium Amara (Bitter Orange) Peel Extract
A supplier reports that its Citrus Aurantium Amara (Bitter Orange) Peel Extract products contain no more than 20 ppm heavy metals (one product not more than 10 ppm) and no more than 2 ppm arsenic. 6
Citrus Aurantium Dulcis (Orange) Peel Wax
According to data provided by a supplier of Citrus Aurantium Dulcis (Orange) Peel Wax, 1,4-dioxane, ethylene oxide, solvents (eg, benzol), nitrosamines, and free amines were not present in this product. 8 Heavy metals, pesticides, and polycyclic aromatic hydrocarbons were absent or present at very low concentrations (detail not provided). Low concentrations of fragrance allergens were present (detail not provided).
Citrus Unshiu Peel Extract
According to a supplier, a commercial product containing 0.5% Citrus Unshiu Peel Extract (dry) contains <2.5 ppm (detection limit) allergens, <1 ppm heavy metals, <5 ppm (detection limit) formaldehyde, <0.04 ppm (detection limit) pesticides, and <10 ppm (detection limit) ethanol. 10 An analysis of just the ingredient Citrus Unshiu Peel Extract found allergens and ethanol to be less than detection limits. The allergens under analysis were not specified.
Another supplier reports that its Citrus Unshiu Peel Extract products contain no more than 20 ppm heavy metals (a few products were no more than 10 ppm) and no more than 2 ppm arsenic (one products was no more than 1 ppm). 6
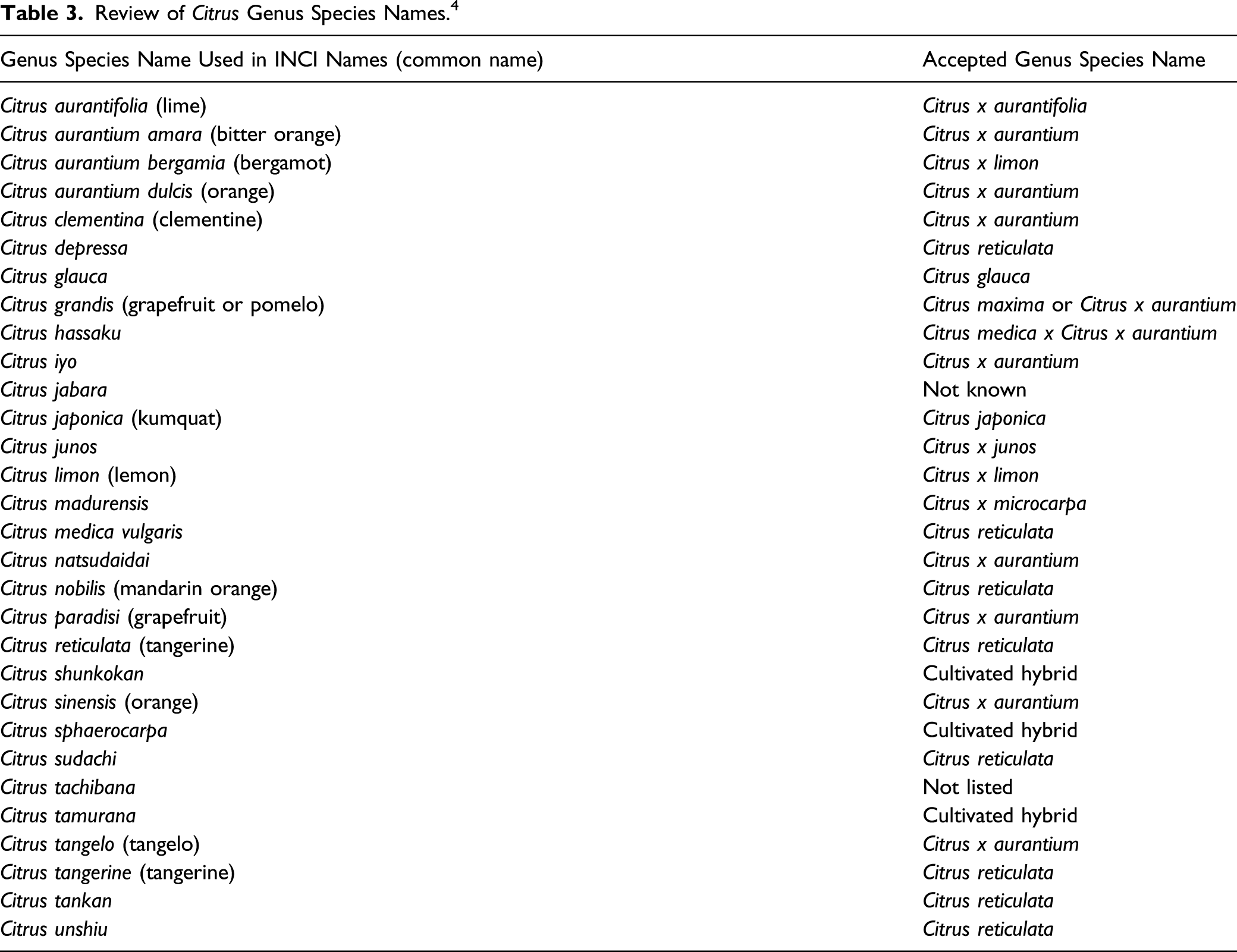
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the U.S. FDA and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s VCRP database. Use concentration data are submitted by Industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
NR = Not reported. NS = Not surveyed.
†Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
dListed as Citrus Aurantium (Bitter Orange) in the VCRP database.
eIncludes uses listed under Citrus Sinensis (Sweet Orange) Peel Extract in the VCRP database.
fListed as Citrus Sinensis (Sweet Orange) Peel Wax and Orange Peel Wax in the VCRP database.
gListed as Citrus Tachibana (Tachibana Orange) Peel Extract and Citrus Tachibana Peel Extract in the VCRP database.
hListed as Citrus Grandis (Pomelo) Peel Extract in the VCRP database.
iListed as Citrus Junos (Xiaang Cheng) Peel Extract in the VCRP database.
jListed as Lemon Peel in the VCRP database.
kListed as Citrus Reticulata (Mandarin Orange) Peel Extract in the VCRP database.
lListed as Citrus Unshiu (Satsuma Orange) Peel Extract in the VCRP database.
mNot in the INCI dictionary; included because of similarity.
Ingredients That Are Not Reported to Be in Use.

In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided in the survey. For example, Citrus Paradisi (Grapefruit) Peel Extract is reported to be used in 61 formulations, but no use concentration data were provided. In other cases, no uses were reported to the VCRP, but a maximum use concentration was provided. For example, Citrus Aurantium Amara (Bitter Orange) Peel was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to 0.2%. It should be presumed that Citrus Aurantium Amara (Bitter Orange) Peel is used in at least one cosmetic formulation.
Some of these ingredients may be used in products that can be incidentally ingested or come into contact with mucous membranes. For example, Citrus Aurantium Dulcis (Orange) Peel Wax is used at 1.9% in a lipstick and Citrus Aurantium Amara (Bitter Orange) Peel is used at 0.2% in personal cleanliness products. Additionally, some of these ingredients were reported to be used in fragrance preparations, hair sprays, skin care preparation sprays, and face powders and could possibly be inhaled. For example, Citrus Aurantium Dulcis (Orange) Peel Extract was reported to be used in a moisturizing product at a maximum concentration of 0.15% and Citrus Grandis (Grapefruit) Peel Extract was reported to be used in face powders at up to 0.1%. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays.14-17 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.15,16 Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.18-20
The Citrus ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the European Union (EU); however, furocoumarins are prohibited from use in cosmetics except for normal content in natural essences and in sun protection and bronzing products where the content shall be below 1 mg/kg. 21
The International Fragrance Association (IFRA) has issued standards for Citrus oils and other furocoumarin-containing essential oils. 22 Finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015% or 15 ppm 5-MOP. This equates to a concentration of 0.0075% or 75 ppm in a fragrance compound when used at 20% in a consumer product that is applied to the skin. If the level of 5-MOP has not been determined, limits specified for individual oils should be observed, and when such oils are used in combination with other phototoxic-constituent containing ingredients, the potential for an additive effect should be considered and use concentrations should be reduced accordingly.
An IFRA standard also has been issued for 7-methoxycoumarin, which is prohibited for use in fragrance compounds. 23 Based on established maximum levels of this substance from commercially available natural sources (like essential oils, extracts and absolutes), IFRA has determined that exposure to 7-methoxycoumarin from the use of these oils and extracts is acceptable if the level of 7-methoxycoumarin in the finished product does not exceed 100 ppm.
Non-Cosmetic
The essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from the following Citrus plant sources are GRAS for their intended use in foods for human consumption: Citrus aurantifolia (lime); Citrus aurantium (bergamot); Citrus aurantium (bitter orange; the flowers and peel); Citrus limon (lemon); Citrus paradisi (grapefruit); Citrus reticulata (tangerine); Citrus reticulata blanco (mandarin); Citrus sinensis (orange; the leaf, flowers, and peel) and citrus peels (species not specified) (21CFR182.20). These essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of these Citrus plant sources are GRAS for their intended use in animal drugs, feeds, and related products (21CFR582.20).
Citrus aurantium amara (bitter orange) and extracts of its dried fruit and peel have been used in traditional Western medicines and in Chinese and Japanese herbal medicines. 24
Toxicokinetics
No relevant published toxicokinetic studies on Citrus peel-derived ingredients were identified in a literature search for these ingredients and no unpublished data were submitted; these types of data were not expected to be found because botanical ingredients are mixtures of many constituents.
Toxicological Studies
Acute Toxicity
Some of the Citrus ingredients in this assessment are foods, and daily exposure from consumption would result in much larger systemic exposures than those resulting from use in cosmetic products. Also, as noted earlier, essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from some Citrus peels are GRAS for their intended use in foods for human and animal consumption according to the FDA. Thus, the systemic toxicity potential of Citrus peel-derived ingredients via oral exposure is not addressed further in this report. The safety assessment is focused on the potential for irritation and sensitization from topical exposure to the Citrus ingredients used in cosmetic products.
Repeated Dose Toxicity
No relevant published repeated dose toxicity studies on Citrus peel-derived ingredients were identified in a literature search, and no unpublished data were submitted.
Reproductive and Developmental Toxicity
No relevant published reproductive and developmental studies on Citrus peel-derived ingredients were identified in a literature search, and no unpublished data were submitted.
Genotoxicity
Citrus Reticulata (Tangerine) Peel Extract
A formulation containing 3% Citrus Reticulata (Tangerine) Peel Extract was not genotoxic in a reverse mutation assay (no further details provided). 25
Carcinogenicity
No relevant published carcinogenicity studies on Citrus peel-derived ingredients were identified in a literature search, and no unpublished data were submitted.
Irritation and Sensitization
Dermal Irritation
Dermal Irritation Studies for Citrus Peel-Derived Ingredients.
Ocular Irritation
Citrus Aurantium Amara (Bitter Orange) Peel Extract
The ocular irritation potential of an undiluted formulation containing 2.0% Citrus Aurantium Amara (Bitter Orange) Peel Extract was studied in 3 albino rabbits. 28 The test material was instilled into the conjunctival sac of one eye and the other eye served as a control. Eyes were observed for irritation at 0, 1, 24, 48, and 72 h post-instillation. Redness was observed in the conjunctiva immediately after instillation, but not at later time points. No inflammatory signs were observed in the iris or cornea. The test material was considered almost non-irritating.
Citrus Limon (Lemon) Peel Extract
In an Epiocular™ tissue equivalent in vitro assay, a moisturizer containing 0.1% Citrus Limon (Lemon) Peel Extract tested neat at pH 5.5 was not predicted to be an ocular irritant. 31
Citrus Reticulata (Tangerine) Peel Extract
In an in vitro assay using the neutral red release method on Statens Seruminstitut rabbit cornea (SIRC) cell lines, 3.0% Citrus Reticulata (Tangerine) Peel Extract in formulation was classified as having relatively low cytotoxicity. 25 This suggests that this ingredient is likely not an ocular irritant.
Sensitization
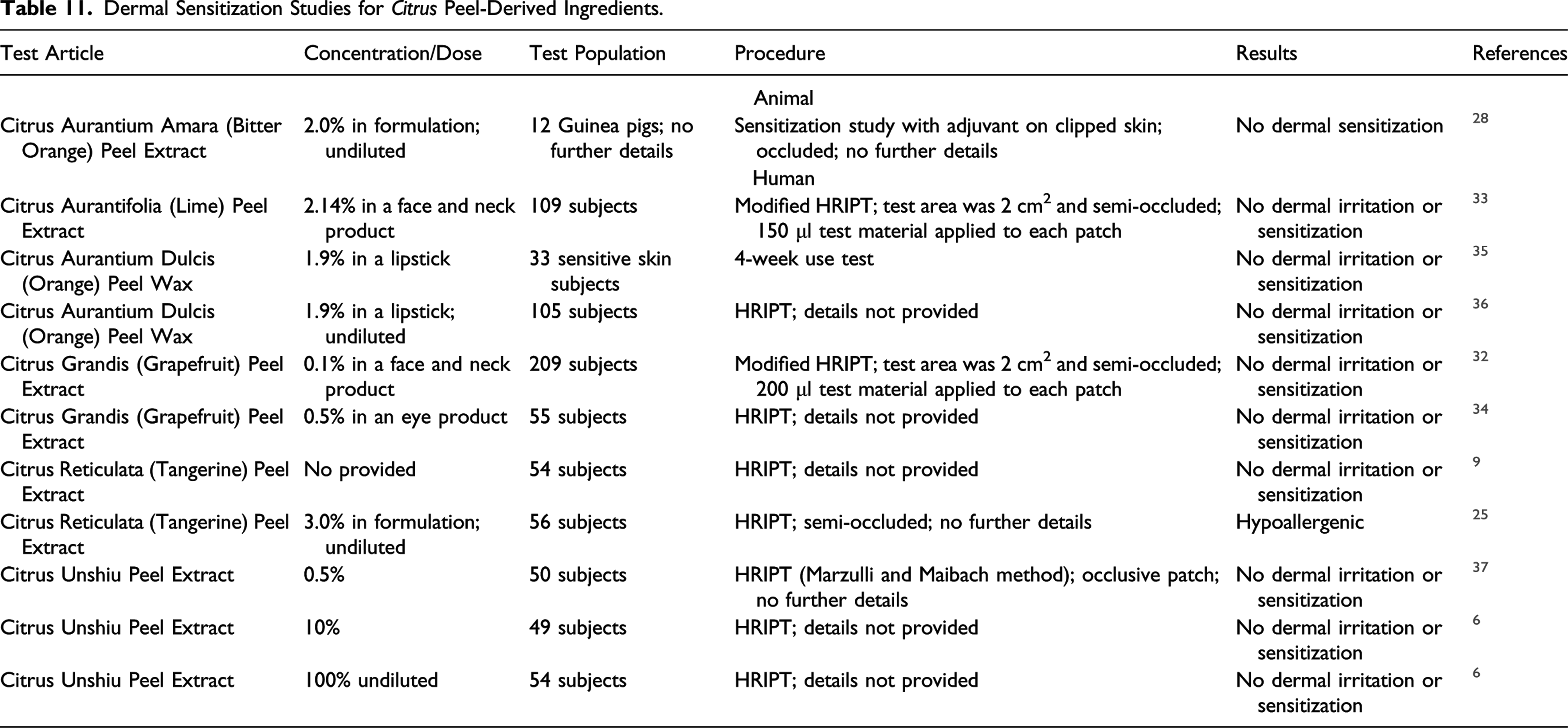
Dermal Sensitization Studies for Citrus Peel-Derived Ingredients.
Phototoxicity and Photosensitization
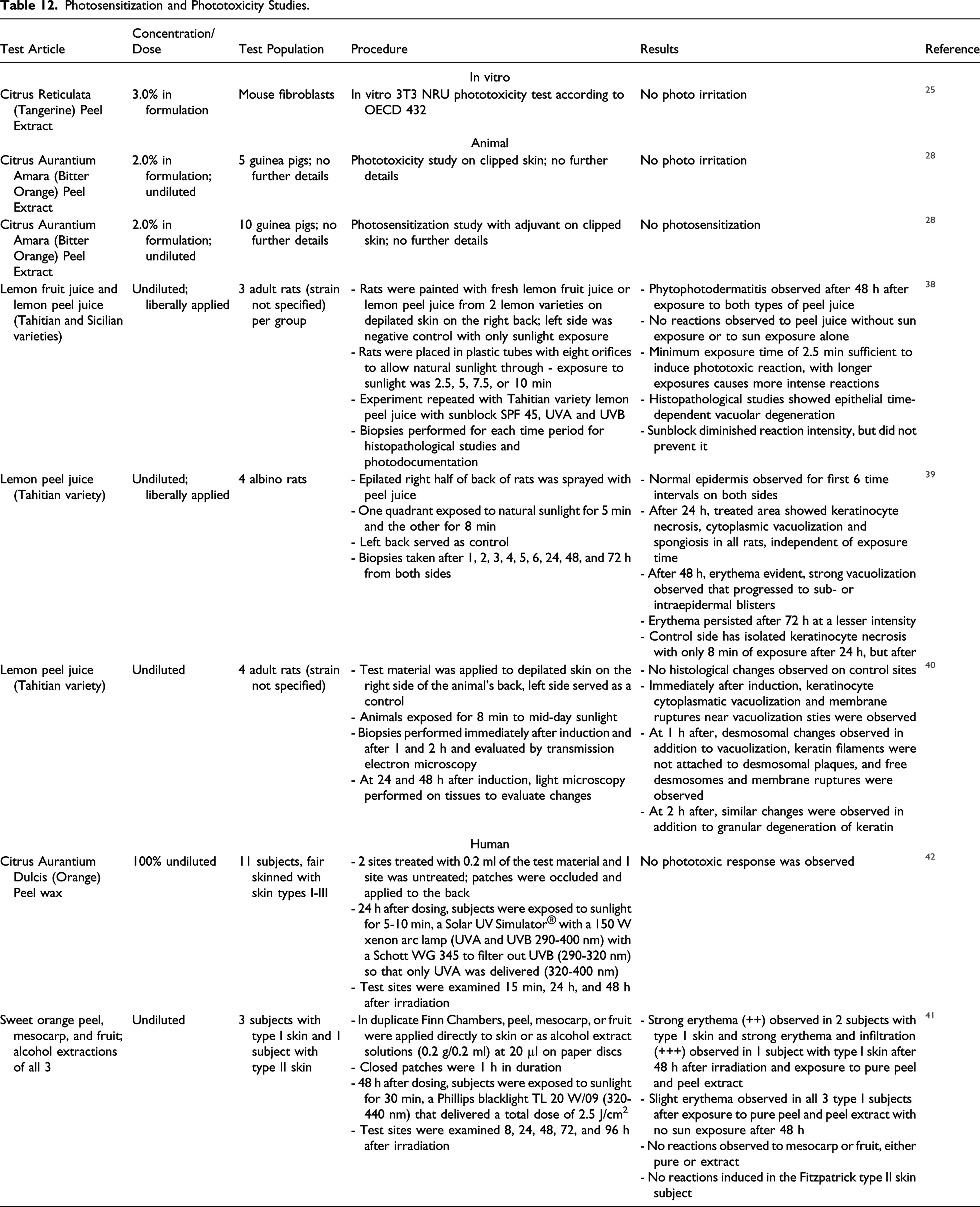
Photosensitization and Phototoxicity Studies.
Occupational Exposure
In a retrospective study (2001-2010) of professional food handlers in Denmark, 8.5% (16/188) of the patients had positive skin prick test reactions to orange peel and 7.9% (15/191) of the patients had positive skin prick test reactions to lemon peel. 43
Summary
The 47 Citrus peel-derived ingredients described in this report are reported to function in cosmetics primarily as skin conditioning agents. Botanical ingredients such as those derived from the genus Citrus are composed of hundreds of constituents, some of which have the potential to cause toxic effects; for example, bergapten (also known as 5-methoxypsoralen or 5-MOP) is a naturally occurring, phototoxic furanocoumarin (psoralen) in Citrus. The Panel reviewed the information available on the potential toxicity of each Citrus peel-derived ingredient as a whole, complex substance. Except for specific constituents of concern that have been identified, the Panel is not reviewing the potential toxicity of the individual constituents of the Citrus peels from which the ingredients in this report are derived.
Citrus Limon (Lemon) Peel Extract has the most reported uses of the cosmetic ingredients in this report, with a total of 150; more than half of the uses are in rinse-off preparations (eg, non-coloring hair conditioners, hair shampoos, and skin cleansing preparations). Citrus Paradisi (Grapefruit) Peel Extract has the second greatest number of overall uses reported, with a total of 61; more than half of the uses are in skin care preparations. The results of the concentration of use survey conducted in 2016 by the Council indicate Citrus Aurantium Dulcis (Orange) Peel Powder has the highest reported maximum concentration of use; it is used at up to 2% in skin cleansing preparations. The highest reported maximum concentration of use in a leave-on product is 1.9% in a lipstick for Citrus Aurantium Dulcis (Orange) Peel Wax.
The Citrus ingredients described in this safety assessment are not restricted from use in any way under the rules governing cosmetic products in the EU; however, furocoumarins are prohibited from use in cosmetics, except for normal content in natural essences and in sun protection and bronzing products where the content shall be below 1 mg/kg. IFRA also has issued standards for Citrus oils and other furocoumarin-containing essential oils. Finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015% or 15 ppm 5-MOP. If the level of 5-MOP has not been determined, limits specified for individual oils should be observed, and when these oils are used in combination with other phototoxic ingredients, the potential additive effect should be taken into consideration and use levels in the final formulation should be carefully monitored.
Some of the Citrus ingredients in this assessment are found in foods, and daily exposure from food use would result in much greater systemic exposures than those from cosmetic products. Essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from some Citrus peels are GRAS for their intended use in foods for human and animal consumption, according to the FDA.
A formulation containing 3% Citrus Reticulata (Tangerine) Peel Extract was not genotoxic in a reverse mutation assay (no further details provided).
No irritation potential was observed for Citrus Aurantium Dulcis (Orange) Peel Wax (100%) or Citrus Reticulata (Tangerine) Peel Extract (3.0% in formulation) in in vitro tests. Citrus Aurantium Amara (Bitter Orange) Peel Extract was not irritating when tested up to 2.0% in formulation in rodents and 100% in rabbits. In human subjects, no irritation was observed after topical exposure to Citrus Aurantium Dulcis (Orange) Peel Wax (100%), Citrus Limon (Lemon) Peel Extract (0.1% in a moisturizer), and Citrus Unshiu Peel Extract (0.5% in formulation). Any irritation observed in tests with Citrus Aurantium Amara (Bitter Orange) Peel Extract (1.55% in formulation; 20% water solution) and Citrus Reticulata (Tangerine) Peel Extract (3.06% in formulation) was resolved within 24-h of exposure.
In in vitro assays, Citrus Limon (Lemon) Peel Extract (0.1% in a moisturizer) and Citrus Reticulata (Tangerine) Peel Extract (3.0% in formulation) did not predict ocular irritation. An undiluted formulation containing 2.0% Citrus Aurantium Amara (Bitter Orange) Peel Extract was almost non-irritating to the eyes of 3 albino rabbits.
No sensitization was observed in guinea pigs exposed to Citrus Aurantium Amara (Bitter Orange) Peel Extract (2.0% in formulation). Citrus Aurantifolia (Lime) Peel Extract (2.14% in a face and neck product), Citrus Aurantium Dulcis (Orange) Peel Wax (1.9% in a lipstick), Citrus Grandis (Grapefruit) Peel Extract (up to 0.5% in formulation), Citrus Reticulata (Tangerine) Peel Extract (up to 3.0% in formulation), and Citrus Unshiu Peel Extract (up to 100%) were not dermal irritants or sensitizers in HRIPTs.
No photo irritation was observed in an in vitro study of Citrus Reticulata (Tangerine) Peel Extract (3.0% in formulation). Citrus Auratnium Amara (Bitter Orange) Peel Extract (2.0% in formulation) did not induce photo irritation or photosensitization in guinea pigs. Undiluted lemon peel juice produced phototoxic reactions in several rat studies. In humans, Citrus Aurantium Dulcis (Orange) Peel Wax (100%) was not phototoxic, but phototoxic reactions were observed in 3 subjects (out of 4) with Fitzpatrick type I skin exposed to undiluted sweet orange peel.
In a retrospective study of professional food handlers in Denmark, 8.5% (16/188) of the patients had positive skin prick test reactions to orange peel and 7.9% (15/191) of the patients had positive skin prick test reactions to lemon peel.
No relevant published studies on the toxicokinetics, repeated dose toxicity, reproductive and development toxicity, or carcinogenicity of Citrus peel-derived ingredients were discovered and no unpublished data were submitted to address these topics.
Discussion
The Citrus ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from the use of cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some Citrus peels are GRAS in foods and animal feeds. Additionally, volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are used as flavoring agents. Consequently, the primary focus of this safety assessment is on the potential for irritation and sensitization from dermal exposures to the Citrus ingredients.
Although there are many differences among Citrus peel-derived ingredients derived from different species, cultivars, growth conditions, extraction methods, and preparation techniques, the weight of the evidence indicates a consistent lack of irritation and sensitization across multiple test methods and ingredients, including testing at maximum use concentrations. However, the Panel expressed concern about the potential for constituents in Citrus peel-derived ingredients, including the furocoumarin 5-MOP, to cause phototoxicity. IFRA has issued standards for Citrus oils and other furocoumarin-containing essential oils, and the Panel agreed that adherence to the IFRA standards for such constituents will prevent phototoxicity. According to these standards, finished products that are applied to the skin, excluding rinse-off products, must not contain more than 0.0015%, or 15 ppm, 5-MOP. An IFRA standard also has been issued for 7-methoxycoumarin; based on established maximum levels of this substance from commercially available natural sources (like essential oils, extracts and absolutes), exposure to 7-methoxycoumarin from the use of these oils and extracts is regarded to be acceptable if the level of 7-methoxycoumarin in the finished product does not exceed 100 ppm.
Additionally, during the assessment of safety for the Citrus-derived peel oils, the Panel was concerned with findings of a rodent carcinogenicity study in which tumor promotion may have been caused by repeated skin irritation and resultant proliferation of 9,10-dimethyl-1,2-benzanthracene-treated basal cells. The Panel concluded that Citrus-derived peel oils could potentially promote tumors if the formulation produces irritation. While no significant skin irritation was reported following the use of Citrus peel-derived ingredients, the Panel felt that these botanical ingredients must be formulated to be non-irritating.
The Panel noted that because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients in one formulation may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of plant constituents that may cause sensitization or other adverse effects. Specific examples of constituents that could induce adverse effects are limonene, citral, and furocoumarins (such as 5-MOP and 7-methoxycoumarin).
The Panel discussed the issue of incidental inhalation exposure in fragrance preparations, hair sprays, skin care preparation sprays, and face powders. There were no inhalation toxicity data available. The Panel considered other pertinent data indicating that incidental inhalation exposures to Citrus peel-derived ingredients in such cosmetic products would not cause adverse health effects, including data characterizing the potential for these ingredients to cause ocular or dermal irritation or sensitization, and other effects. These ingredients are reportedly used at concentrations up to 0.15% in cosmetic products that may be aerosolized. The Panel noted that droplets/particles from spray and loose-powder cosmetic products would not be respirable to any appreciable amount. The potential for inhalation toxicity is not limited to respirable droplets/particles deposited in the lungs. In principle, inhaled droplets/particles deposited in the nasopharyngeal and thoracic regions of the respiratory tract may cause toxic effects depending on their chemical and other properties. However, coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Finally, the Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded the following 47 Citrus peel-derived ingredients are safe in the present practices of use and concentration in both rinse-off and leave-on cosmetic products when formulated to be non-sensitizing and non-irritating, provided that leave-on products do not contain more than 0.0015% (15 ppm) 5-MOP. Citrus Aurantifolia (Lime) Peel* Citrus Aurantifolia (Lime) Peel Extract Citrus Aurantifolia (Lime) Peel Powder Citrus Aurantifolia (Lime) Peel Water* Citrus Aurantium Amara (Bitter Orange) Peel Citrus Aurantium Amara (Bitter Orange) Peel Extract Citrus Aurantium Amara (Bitter Orange) Peel Powder Citrus Aurantium Bergamia (Bergamot) Peel Water Citrus Aurantium Dulcis (Orange) Peel Extract Citrus Aurantium Dulcis (Orange) Peel Powder Citrus Aurantium Dulcis (Orange) Peel Wax Citrus Aurantium Sinensis Peel Extract* Citrus Aurantium Tachibana Peel Extract Citrus Depressa Peel Extract Citrus Depressa Peel Powder* Citrus Grandis (Grapefruit) Peel* Citrus Grandis (Grapefruit) Peel Extract Citrus Grandis (Grapefruit) Peel Powder* Citrus Hassaku/Natsudaidai Peel Powder* Citrus Iyo Peel Extract* Citrus Iyo Peel Water* Citrus Jabara Peel Extract Citrus Jabara Peel Powder* Citrus Jabara Peel Water* Citrus Junos Peel Extract Citrus Junos Peel Powder Citrus Junos Peel Water* Citrus Limon (Lemon) Peel Citrus Limon (Lemon) Peel Extract Citrus Limon (Lemon) Peel Powder Citrus Limon (Lemon) Peel Water* Citrus Limon (Lemon) Peel Wax Citrus Natsudaidai Peel Extract* Citrus Nobilis (Mandarin Orange) Peel Extract Citrus Nobilis (Mandarin Orange) Peel Powder* Citrus Paradisi (Grapefruit) Peel Extract Citrus Reticulata (Tangerine) Peel Extract Citrus Reticulata (Tangerine) Peel Powder* Citrus Shunkokan Peel Extract* Citrus Sunki Peel Extract* Citrus Tachibana/Reticulata Peel Powder* Citrus Tangelo Peel Powder* Citrus Tangerina (Tangerine) Peel* Citrus Tangerina (Tangerine) Peel Extract Citrus Unshiu Peel Extract Citrus Unshiu Peel Powder Citrus Unshiu Peel Water*
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.