Abstract
The Cosmetic Ingredient Review Expert Panel assessed the safety of 14 citrus-derived peel oil ingredients and concluded that these ingredients are safe for use in cosmetic products when finished products, excluding rinse-off products, do not contain more than 0.0015% (15 ppm) 5-methoxypsoralen, and when formulated to be nonsensitizing and nonirritating. The citrus-derived peel oil ingredients are most frequently reported to function in cosmetics as fragrances and/or skin conditioning agents. The Panel reviewed the available animal and clinical data to determine the safety of these ingredients. Because final product formulations may contain multiple botanicals, each containing the same constituents of concern, formulators are advised to be aware of these constituents and to avoid reaching levels that may be hazardous to consumers. Industry should use good manufacturing practices to limit impurities that could be present in botanical ingredients.
Keywords
Introduction
Citrus-derived peel oils are widely used as cosmetic ingredients and are most frequently reported to function in cosmetics as fragrances and/or skin conditioning agents (Table 1). This report assesses the safety of the following 14 citrus-derived peel oils:
Citrus aurantifolia (lime) peel oil
Citrus aurantium amara (bitter orange) peel oil
Citrus aurantium currassuviensis peel oil
Citrus aurantium dulcis (orange) peel oil
Citrus clementina peel oil
Citrus grandis (grapefruit) peel oil
Citrus iyo peel oil
Citrus junos peel oil
Citrus limon (lemon) peel oil
Citrus medica vulgaris peel oil
Citrus nobilis (mandarin orange) peel oil
Citrus reticulata (tangerine) peel oil
Citrus tachibana/reticulata peel oil
Citrus tangerina (tangerine) peel oil
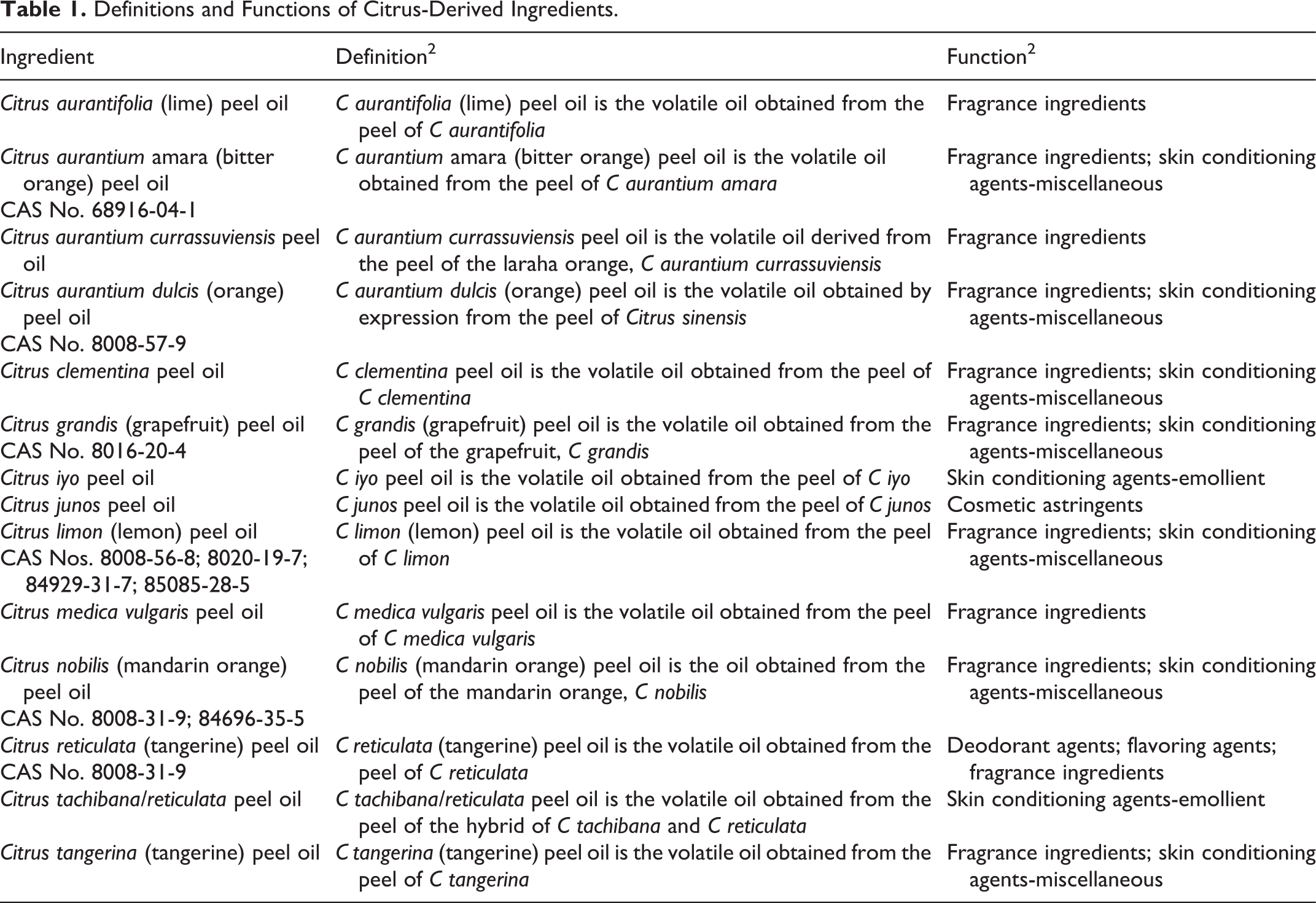
Definitions and Functions of Citrus-Derived Ingredients.
The citrus ingredients in this assessment are found in foods, and daily exposure from food use would result in much larger systemic exposures than those from use in cosmetic products. Additionally, essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some citrus fruits are generally recognized as safe (GRAS) for their intended use in foods for human and animal consumption according to the US Food and Drug Administration (FDA). Volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are described as flavoring agents in the US Pharmacopeia (USP) Food Chemicals Codex. 1 Thus, the systemic toxicity potential of citrus-derived peel oils via oral exposure is not addressed further in this report. The primary focus of this safety assessment of these citrus ingredients as used in cosmetics is on the potential for irritation and sensitization from dermal exposure.
The Cosmetic Ingredient Review (CIR) does not review ingredients that function only as fragrance ingredients because, as fragrances, the safety of these ingredients is evaluated by the Research Institute for Fragrance Materials (RIFM). Three of the citrus-derived peel oils in this report function only as fragrance ingredients, according to the International Cosmetic Ingredient Dictionary and Handbook (see Table 2). 2 However, RIFM has not confirmed that these 3 ingredients function only as fragrances and therefore within its purview; thus, CIR is reviewing the safety of these ingredients.
Citrus Ingredients That Potentially Function Solely as Fragrance Ingredients.

Botanicals such as those derived from citrus contain hundreds of constituents, some of which have the potential to cause toxic effects. For example, bergapten (also known as 5-methoxypsoralenor [5-MOP]) is a naturally occurring phototoxic furanocoumarin (psoralen) in citrus peel oils. In this assessment, CIR is reviewing information available to evaluate the potential toxicity of each of the citrus-derived peel oils as a whole, complex substance. Except for specific constituents of concern, CIR is not reviewing information that may be available to assess the potential toxicity of the individual constituents of which the citrus-derived ingredients are composed. Cosmetic Ingredient Review requested information on the concentrations (including ranges, means, upper 95%confidence limits, detection limits, etc) of individual constituents in citrus-derived peel oils used in cosmetics to facilitate the safety assessment of these ingredients. Such information on constituents that have been identified as constituents of concern by the Panel in previous safety assessments, or by other recognized scientific expert review bodies, is especially important.
Some toxicological data on lemon oil and sweet orange oil (synonyms: lemon, ext. and orange, sweet, extract, respectively) reviewed in this safety assessment were obtained from robust summaries of data submitted to the European Chemical Agency (ECHA) by companies as part of the REACH chemical registration process. These data are available on the ECHA website. 3,4
Note that, in many of the published studies included in this assessment, the information provided is not sufficient to determine how well the substance being tested represents the cosmetic ingredient. In this safety assessment, if a substance tested in a study is not clearly a cosmetic ingredient, because of lack of information on the genus and species from which the substance was derived and/or the method of extraction used, the test substance will be referred to by a common name (eg, lime oil). If the substance is clearly a cosmetic ingredient, the International Nomenclature of Cosmetic Ingredients (INCI) name will be used (eg, “C aurantifolia [lime] peel oil”). In some instances, the part of the plant from which the oil was expressed is not known but, based on the method of manufacture, the oil could have been expressed from the peels of the citrus fruit. Additionally, some inconsistencies were noted in both taxonomic and INCI naming conventions. For example, this report includes the sweet orange ingredient described as C aurantium dulcis (orange) peel oil in the International Cosmetic Ingredient Dictionary and Handbook (Dictionary). 2 In contrast, most of the published literature and the FDA refer to this ingredient as Citrus sinensis (sweet orange) peel oil. Another example of a naming inconsistency is C grandis (grapefruit) peel oil; C grandis is generally considered a name for a pummelo and is more commonly called Citrus maxima. The INCI Committee of the Personal Care Products Council (Council) is working to correct some of these errors.
Chemistry
Definition and General Characterization
The definitions and functions of the citrus-derived peel oils included in this report are provided in Table 1. In some cases, the definition provides insight on the method(s) of manufacture. It should be noted that essential oils are hydrophobic, liquid, and volatile aroma compounds from plants. These are typically mixtures of small molecules, but their chemical structures can vary widely. The volatile nature of essential oils makes them likely to be useful as fragrances but does not preclude other functions for them in cosmetics.
There are numerous Citrus varieties distributed throughout the world; most citrus fruits are grown in the temperate and tropical zones in both the northern and southern hemispheres. 5 Citrus can be propagated and new varieties can be produced by cultivating asexual nuclear or chance seedlings or by crossing or mutation.
Physical and Chemical Properties
Physical and chemical properties of the citrus-derived peel oils are provided in Table 3.
Physical and Chemical Properties of Citrus-Derived Peel Oils.

Abbreviation: UV, ultraviolet.
Method of Manufacturing
A suitable antioxidant can be added to citrus oil during preparation. 1
Bitter orange oil and orange oil are obtained using a cold-pressed extraction method from the fresh peel of the fruit of Citrus aurantium and C sinensis, respectively. 1 Citrus grandis oil 6 and C junos oil 7 are also extracted using a cold-press method. Tangerine oil (cold-pressed) is obtained from the peels of the ripe fruit of the Dancy tangerine, C nobilis or C reticulata, and from some other closely related varieties. 1
Lemon oil and lime oil, expressed, are produced by pressing the outer rind of the ripe fruit manually or by machine. 8 More economical processes involve an integrated juice-oil procedure. These oils can also be produced by distillation of expressed oils or direct distillation of fruit; distilling (rectifying) removes terpenes. Steam distillation removes nonvolatile furocoumarins.
Citrus iyo oil is produced by removing the peel from the fruit, homogenization of the peel with distilled water, and lyophilization. 9 The oil is purified using vacuum distillation.
Mandarin peel oil, expressed (identified in the literature as C reticulata), is prepared by expression of the peels of the ripe fruit of the mandarin orange. 10
Constituents/Composition
The citrus-derived peel oils are complex botanicals composed of numerous constituents; there is great variation among citrus species and cultivars because of frequent bud mutations, interspecific and intergeneric hybridization, and apomixis (ie, 1 or more of several types of asexual reproduction). 11 The composition of citrus oils will vary based on the location where the plant is grown, the maturity of the plant, and storage conditions. 5 The method of extraction will also affect the composition.
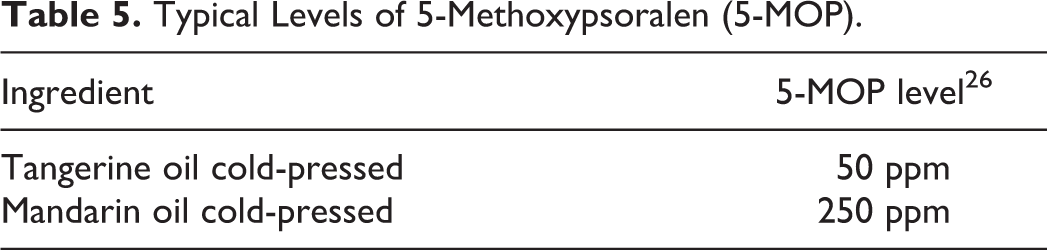
Citrus oils contain large amounts of monoterpene hydrocarbons. Limonene is the constituent present in the greatest amount, often comprising greater than 90% of the oil, and the amount present can vary within the oil; for example, limonene is reported to compose 38.1% to 95.8% C limon (lemon) peel essential oils, cold-pressed. 12,13 Citrus oils also contain sesquiterpene hydrocarbons, which are responsible for the characteristic flavors of these oils. 12 Table 4 lists the chemical compositions of cold-pressed, 6,7,11 -16 hydrodistilled, 17 -21 lyophilization/vacuum distillation, 9 and steam-distilled 22 peel oils sorted by citrus plant species; Table 5 presents the typical levels of 5-MOP found in some of the oils; and Table 6 provides the levels of major coumarins and furocoumarins in lemon and lime oil. Table 7 lists citrus constituents that are established contact allergens, according the European Commission’s Scientific Committee on Consumer Safety.
Chemical Composition (%) of Citrus Peel Oils.a
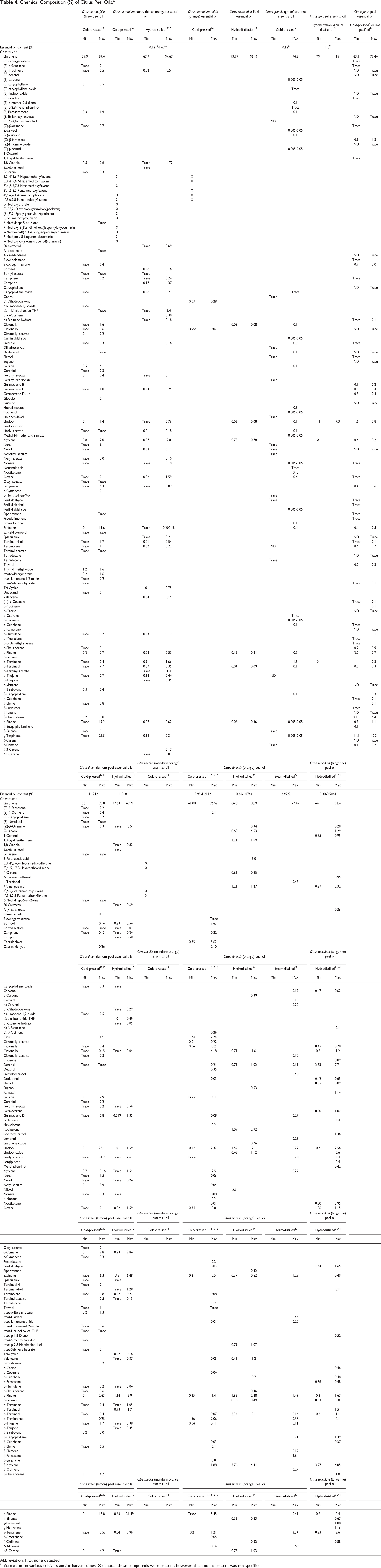
Abbreviation: ND, none detected.
aInformation on various cultivars and/or harvest times. X denotes these compounds were present; however, the amount present was not specified.
Typical Levels of 5-Methoxypsoralen (5-MOP).
Levels of Major Coumarins and Furocoumarins in Lemon Oil and Lime Oil.
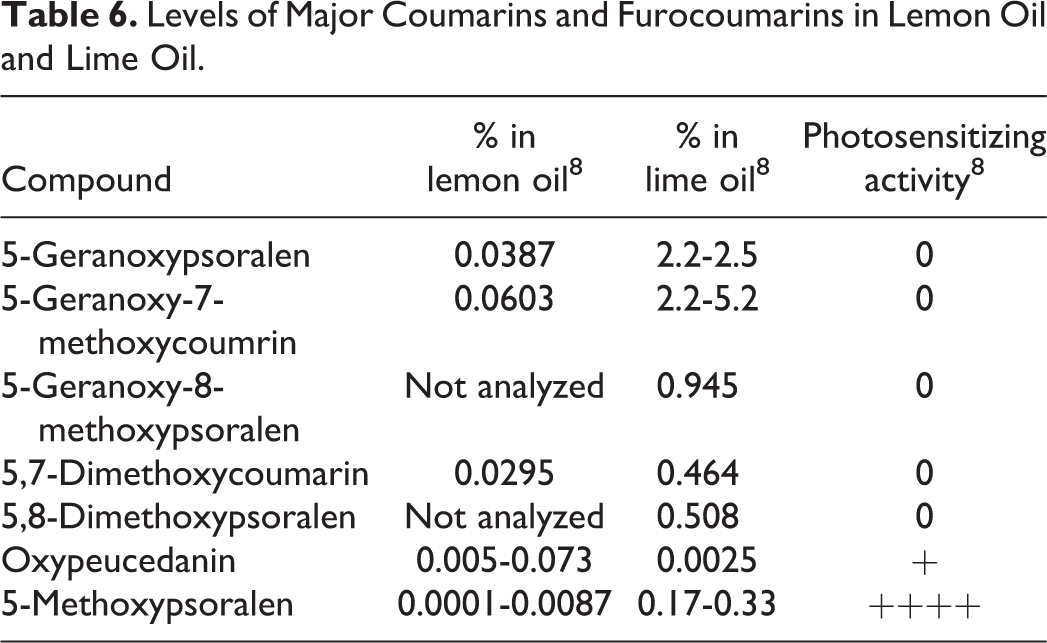
Constituents That Are Established Contact Allergens in Humans, According to the SCCS.
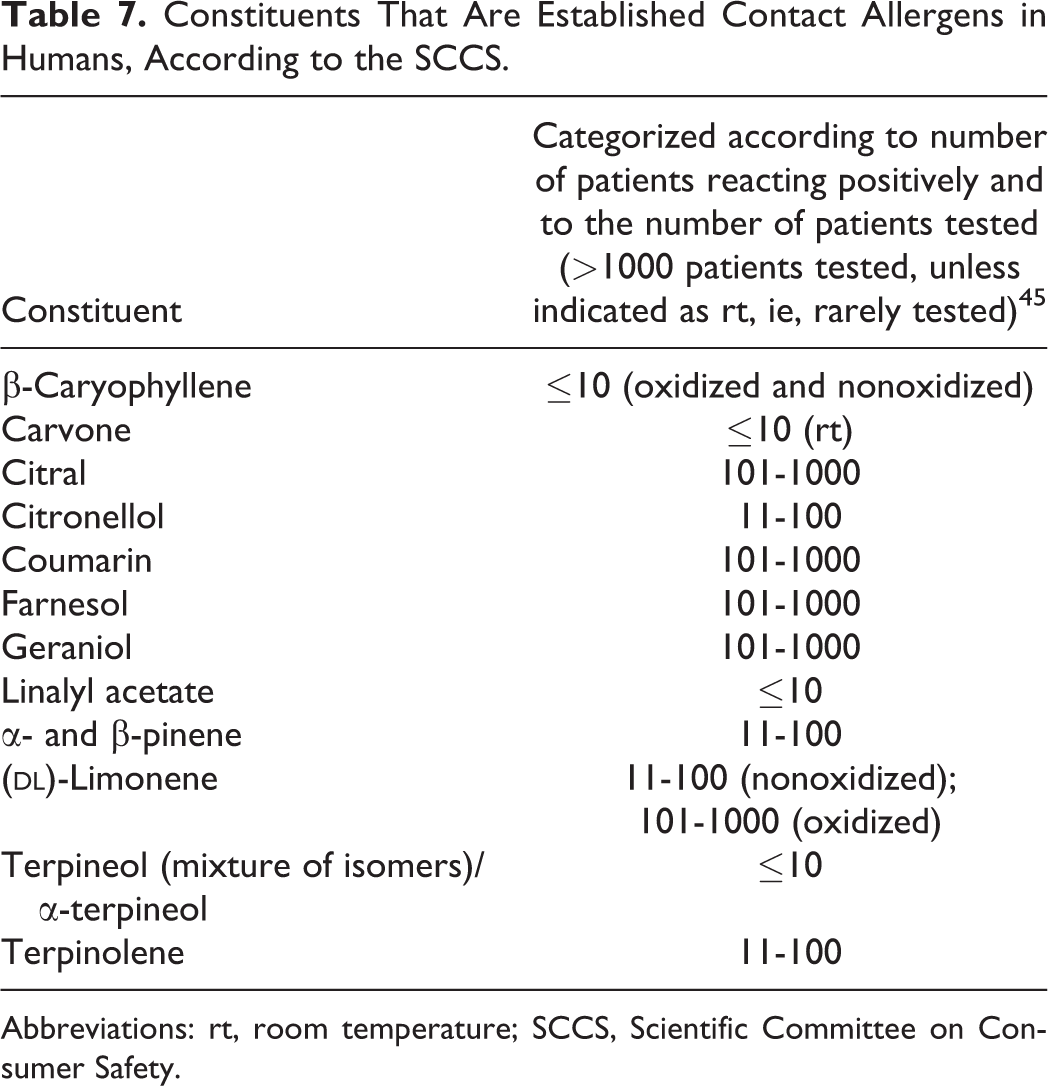
Abbreviations: rt, room temperature; SCCS, Scientific Committee on Consumer Safety.
Use
Cosmetic
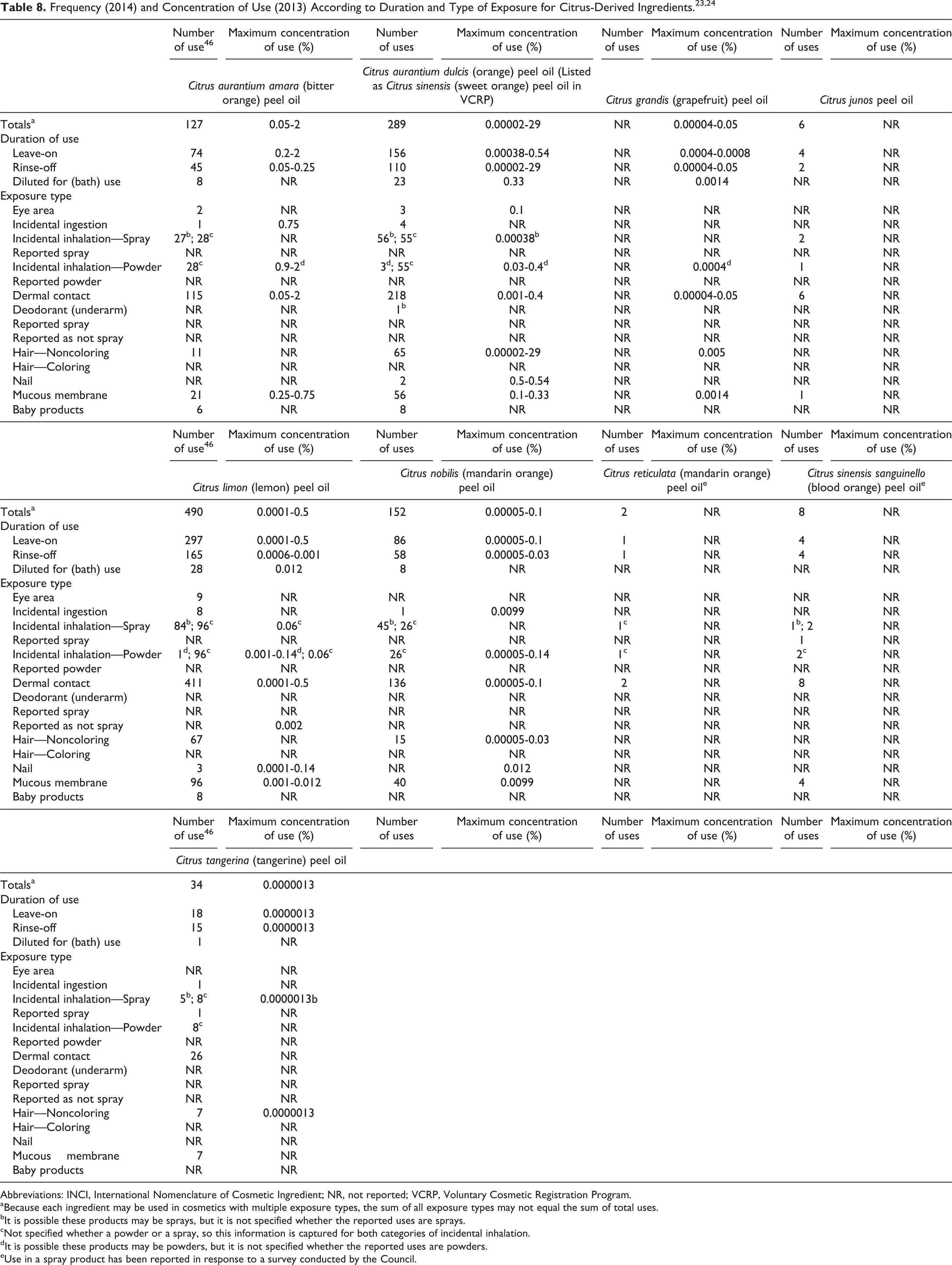
Table 8 presents the current product-formulation use data for citrus-derived peel oils. These ingredients are most frequently reported to function as fragrances and/or skin conditioning agents-miscellaneous. 2
Abbreviations: INCI, International Nomenclature of Cosmetic Ingredient; NR, not reported; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIt is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
cNot specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
dIt is possible these products may be powders, but it is not specified whether the reported uses are powders.
eUse in a spray product has been reported in response to a survey conducted by the Council.
According to information supplied to the FDA by industry as part of the Voluntary Cosmetic Registration Program (VCRP), C limon (lemon) peel oil has the most reported uses in cosmetic and personal care products, with a total of 490; more than half of the uses are in leave-on skin care preparations. 23 Citrus aurantium dulcis (orange) peel oil (reported as C sinensis [sweet orange] peel oil by the VCRP) has the second greatest number of overall uses reported, with a total of 289; about half of those uses are in leave-on skin care preparations.
In a use concentration survey conducted by the Council, C limon (lemon) peel oil had a maximum use concentration range of 0.0001% to 0.5%, with 0.5% reported in “other” skin care preparations. 24 Citrus aurantium dulcis (orange) peel oil had a maximum use concentration range of 0.00002% to 29%, with 29% reported in noncoloring hair conditioners.
In some cases, reports of uses were received from the VCRP, but no concentration of use data were provided. For example, C junos peel oil is reported to be used in 6 formulations, but no use concentration data were available. In other cases, no reported uses were identified by the VCRP, but a maximum use concentration was provided in the industry survey. For example, C grandis (grapefruit) peel oil was not reported in the VCRP database to be in use, but the industry survey indicated that it is used at concentrations up to 0.05%. It should be presumed that C grandis (grapefruit) peel oil is used in at least 1 cosmetic formulation.
Table 9 lists the 7 citrus-derived peel oils not indicated to be in use based on the VCRP data and the results of the Council concentration of use survey.
Ingredients That Are Not Reported to Be in Use.

Under the rules governing cosmetic products in the European Union, citrus-derived ingredients must have furocoumarin content below 1 mg/kg in sun protection products and in bronzing products. 25 The International Fragrance Association (IFRA) has issued standards for citrus oils and other furocoumarin-containing essential oils. 26 Thus, finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015%, or 15 ppm, 5-MOP. This equates to 0.0075%, or 75 ppm, in a fragrance compound used at 20% in a consumer product that is applied to the skin. If the level of 5-MOP has not been determined, limits specified for individual oils should be observed, and when such oils are used in combination with other phototoxic ingredients, the potential for an additive effect should be considered and use levels should be reduced accordingly. Restrictions for furocoumarin-containing essential oils have been recommended for bitter orange oil expressed, grapefruit oil expressed, lemon oil cold-pressed, and lime oil expressed.
An IFRA standard also has been issued for 7-methoxycoumarin, which is prohibited for use in fragrance compounds. 27 Based on established maximum levels of this substance from commercially available natural sources (like essential oils, extracts, and absolutes), exposure to 7-methoxycoumarin from the use of these oils and extracts is regarded to be acceptable if the level of 7-methoxycoumarin in the finished product does not exceed 100 ppm. An example of a maximum concentration based on this standard is 0.1% for lime cold-pressed oil.
Additionally, IFRA has set a standard stating that
The IFRA has also set limits on the amounts of some citrus-derived oils in finished products. For leave-on products applied to skin areas exposed to direct sunlight, these limits include 1.25% bitter orange peel expressed, 30 4% grapefruit oil expressed, 31 2% lemon oil cold-pressed, 32 and 0.7% lime oil expressed. 33 There are no restrictions for any of these oils in rinse-off products and products that are not applied to the skin. International Fragrance Association specified that if combinations of phototoxic fragrance ingredients are used, the use levels must be reduced accordingly, so that the sum of the concentrations of all phototoxic fragrance ingredients, expressed as a percentage of their respective recommended maximum levels, shall not exceed 100% in the consumer product. Additionally, the IFRA general standard described above for “Citrus oils and other furocoumarins-containing essential oils” must be considered.
Noncosmetic
The essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from the following citrus fruits are GRAS for their intended use in foods for human consumption: C aurantifolia (lime), C aurantium (bitter orange; the flowers and peel), C limon (lemon), C reticulata (tangerine), Citrus reticulata blanco (mandarin), C sinensis (orange; the leaf, flowers, and peel), and citrus peels (species not specified; 21CFR182.20). These essential oils, oleoresins (solvent-free), and natural extractives (including distillates) of these citrus fruits are GRAS for their intended use in animal drugs, feeds, and related products (21CFR582.20).
Citrus essential oils are used in the pharmaceutical industry as flavoring agents to mask the unpleasant taste of drugs. 18 Citrus aurantium amara (bitter orange) and extracts of its dried fruit and peel have been used in traditional Western medicines and in Chinese and Japanese herbal medicines. 34
Toxicological Studies
As noted earlier, the citrus ingredients in this assessment are found in foods, and daily exposures from food use would result in a much larger systemic exposure than those from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some citrus fruits are GRAS for their intended use in foods for human and animal consumption according to the FDA. Volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are described as flavoring agents in the USP Food Chemicals Codex. 1 Therefore, the systemic toxicity potential of these ingredients is not addressed further in this report. The primary focus of this safety assessment is on the potential for irritation and sensitization from dermal exposure to these citrus ingredients as used in cosmetic products.
Acute Toxicity
Dermal—Nonhuman
Lemon oil
The dermal LD50 of lemon oil was greater than 10 g/kg in rabbits. 3 An occlusive patch of undiluted oil was applied to the skin of 6 animals for 24 hours. One animal died during the observation period.
Mandarin oil—Expressed (C reticulata)
The dermal LD50 of mandarin peel oil (C reticulata) was greater than 5 g/kg in rabbits. 10 An occlusive patch of undiluted oil was applied to the skin of seven animals for 24 hours.
Orange oil
The dermal LD50 of sweet orange oil was greater than 5 g/kg in female New Zealand White rabbits. 4 An occlusive patch of undiluted oil was applied to the skin of 10 animals for 24 hours. Signs of skin irritation were reported, including moderate redness in 10 of 10 animals, slight edema in 3 of 10 animals, and moderate edema in 5 of10 animals.
Reproductive and Developmental Toxicity
No published reproductive and developmental studies on citrus-derived peel oils were identified in a literature search for these ingredients, and no unpublished data were submitted.
Genotoxicity
Genotoxicity studies for in vitro assays are summarized in Table 10. No genotoxic effects were observed for lemon oil or sweet orange oil in bacterial reverse mutation assays, mouse lymphoma cell mutation assays, and Chinese hamster chromosome aberration assays. 3,4
Genotoxicity.
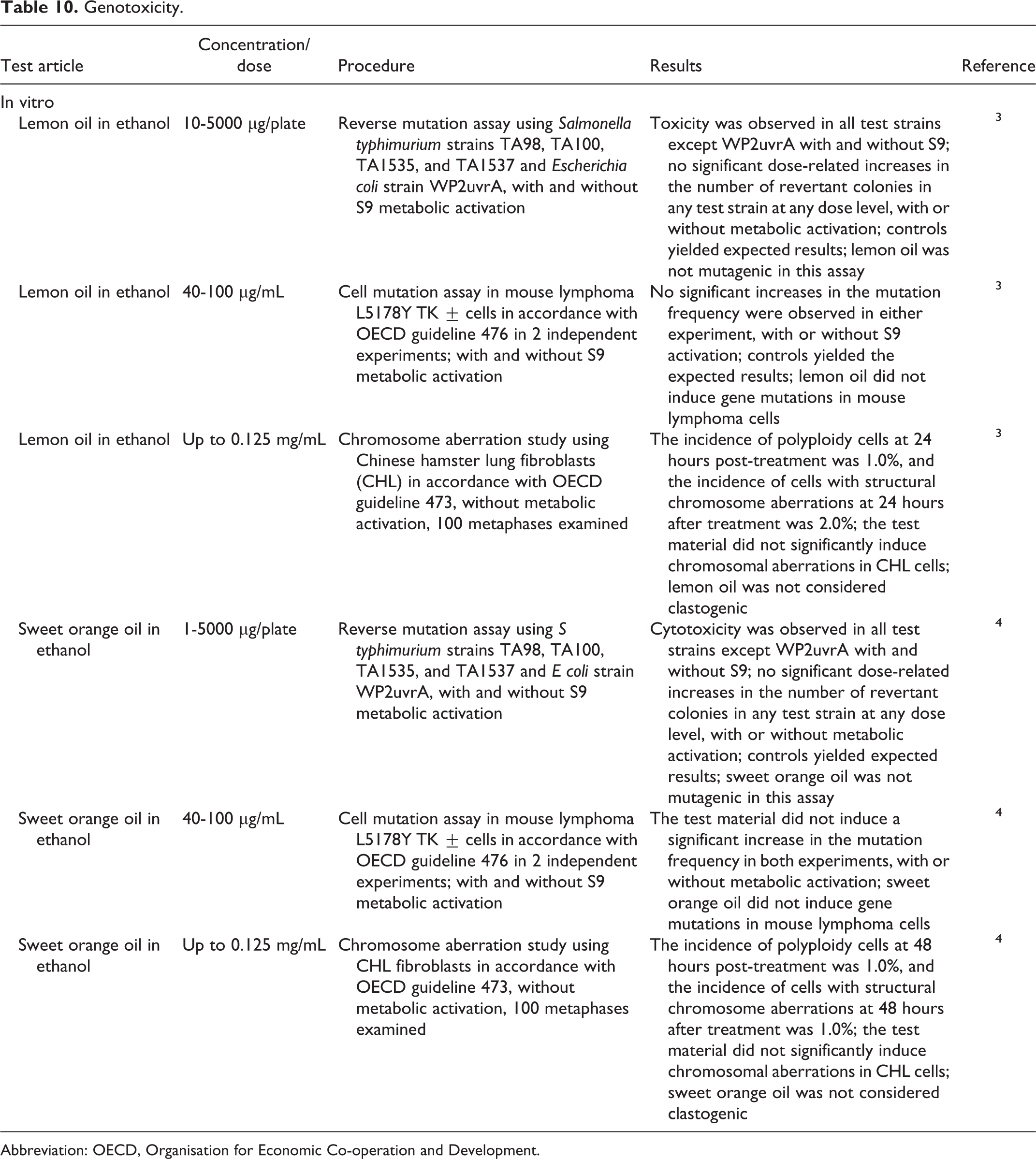
Abbreviation: OECD, Organisation for Economic Co-operation and Development.
Carcinogenicity
Orange Oil, Lemon Oil, Grapefruit Oil, and Lime Oil
Tumor-promoting activity was observed in mouse skin exposed to essential oils of orange (sweet), lemon, grapefruit, or lime. 35 Chemical constituents of these oils were not fully identified in this study, although the terpene and nonterpene fractions were separated. Groups of 10 male and 10 female strain 101 mice received a single application of 9,10-dimethyl-1,2-benzanthracene (DMBA) in acetone (300 µg in 0.2 mL in 4 groups, 225 µg in 0.15 mL in a fifth group). Group 1 was a control group that received no further treatments. Groups 2 through 5 received weekly applications of 0.25 mL of the test substances 3 weeks after the application of DMBA.
By the fifth week, papillomas were observed in group 3 (lemon oil), group 4 (grapefruit oil), and group 5 (lime oil). Papillomas were observed in group 2 (orange oil) by the 12th week. After 33 weeks, 10 of 20 mice in the lemon oil and lime oil treatment groups and 13 of 20 mice in the grapefruit oil and orange oil groups had papillomas. Only 1 mouse in the control group had papillomas after 33 weeks, and the affected site was not the treated skin. Additionally, one female mouse of the lemon oil group developed a sebaceous-gland tumor of the nipple. No malignant skin tumors were observed in the orange oil group: Treatment for this group was stopped after 42 weeks. Squamous cell carcinomas of the skin were observed in 2 mice from the lemon oil group and2 mice of the grapefruit oil group between weeks 36 and 55. One malignant skin tumor was observed in the lime oil group at week 34; however, the mouse was found dead and a proper histological examination was not possible. No malignant skin tumors were observed in the control group. Nondermal tumors during the treatment period were observed in one mouse of the orange oil group (a hemangioma of the subcutaneous tissue starting at week 7) and in one mouse of the grapefruit oil group (a spindle cell sarcoma of the subcutaneous tissues). No tumors of the internal organs were observed. The survival of all the mice, including the controls, in this experiment was poor because of a very high incidence of renal disease. 35
Orange Oil
Tumor-promoting activity was observed in mouse skin exposed to orange (sweet) oil. 35 In the study, groups of 10 male and 10 female strain 101 mice received a single application of DMBA in acetone (300 µg in 0.15 mL). One group (15 mice of each sex) was a control group that received no further treatments. Two groups received weekly applications of 0.25 mL of 40% orange oil in acetone or 80% orange oil in acetone 3 weeks after the initial application of DMBA. The applications continued for 37 weeks.
Papillomas were observed in both groups treated with orange oil starting on the 12th week. After 33 weeks, 5 of 10 mice treated with 40% orange oil and 10 of 10 mice treated with 80% orange oil had papillomas, and at study end, only 1 tumor of each group was found outside the treated area. Four mice in the control group had papillomas by week 33, but these tumors were outside the treated area of the skin. Malignant tumors were observed in one mouse of each treatment group, arising from the preexisting papilloma. Both tumors were squamous cell carcinomas, infiltrating the panniculus muscle. Additionally, tumors of the urethral orifice were observed in 4 female mice of the 40% orange oil group. The survival of all the mice in this experiment was poor due to a very high incidence of renal disease.
In the same study, tumor-promoting activity was observed in mice exposed to undiluted orange oil after pretreating the mice with either dermal or intraperitoneal injections of urethane. The effect was weak compared to the effects observed after DMBA induction.
A similar experiment performed in the same study tested the carcinogenic effects of orange oil without pretreatment with DMBA or urethane. The mice were treated once weekly with 0.25 mL of 40% or 80% orange oil in acetone or undiluted orange oil for 38 (diluted) or 46 (undiluted) weeks. This study found no evidence of direct tumorigenic effects on the treated mouse skin. Urethral orifice tumors were observed in one female mouse of the 40% orange oil group and in one female mouse of the 80% orange oil group. A papilloma was observed on the head of a mouse (outside the treatment site) that was treated with 80% orange oil. 35
Irritation and Sensitization
Dermal Irritation
Dermal irritation studies are summarized in Table 11. Lemon oil, orange oil, and mandarin peel oil all produced some reaction in irritation studies in animals, but in human patients, no irritation was observed after topical exposure to lemon oil (up to 20%) or mandarin peel oil (8%). 3,4,10,36
Dermal Irritation Studies for Citrus-Derived Ingredients.
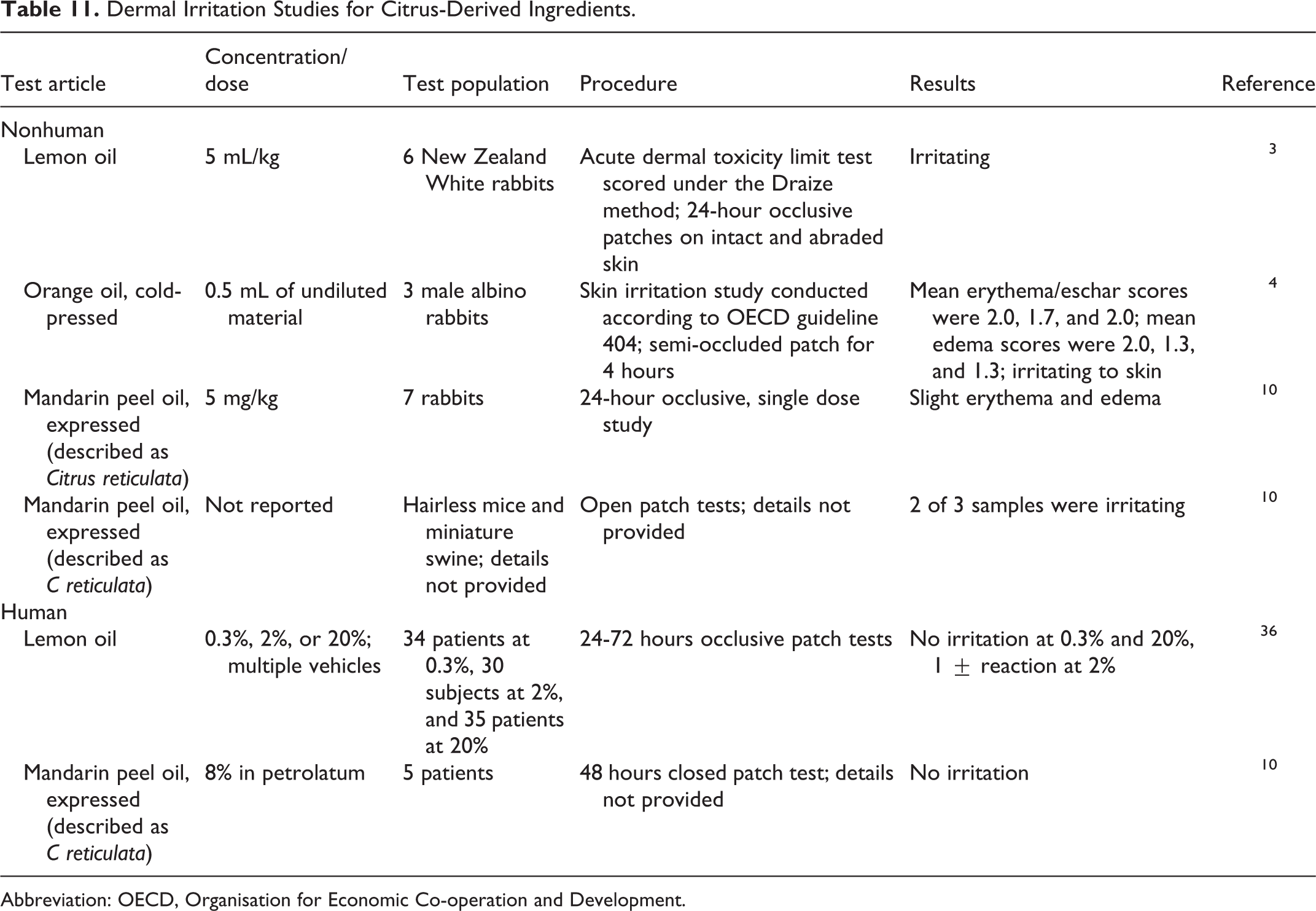
Abbreviation: OECD, Organisation for Economic Co-operation and Development.
Ocular Irritation
Lemon oil
Lemon oil tested at 5% was not irritating to the eyes of 3 albino rabbits. 3 Each rabbit had 0.1 mL of the test material instilled into the right eye with no further treatment. The left eye served as the control. Eyes were examined every 24 hours for 4 days and then again on the seventh day. No corneal opacity or iris congestion was observed. An intense conjunctival irritation involving chemosis and discharge occurred. Treated eyes were normal on the seventh day.
Orange oil
Orange oil tested undiluted did not induce significant or irreversible damage to the eyes of 3 male New Zealand White rabbits. 4 The test was performed in accordance with Organisation for Economic Co-operation and Development Test Guideline 405. The test material (0.1 mL) was instilled into one eye of each rabbit. The mean scores calculated for the 3 animals across 3 scoring times were 0.0 for corneal opacity and iris lesions and 1.0 for reddening of the conjunctivae.
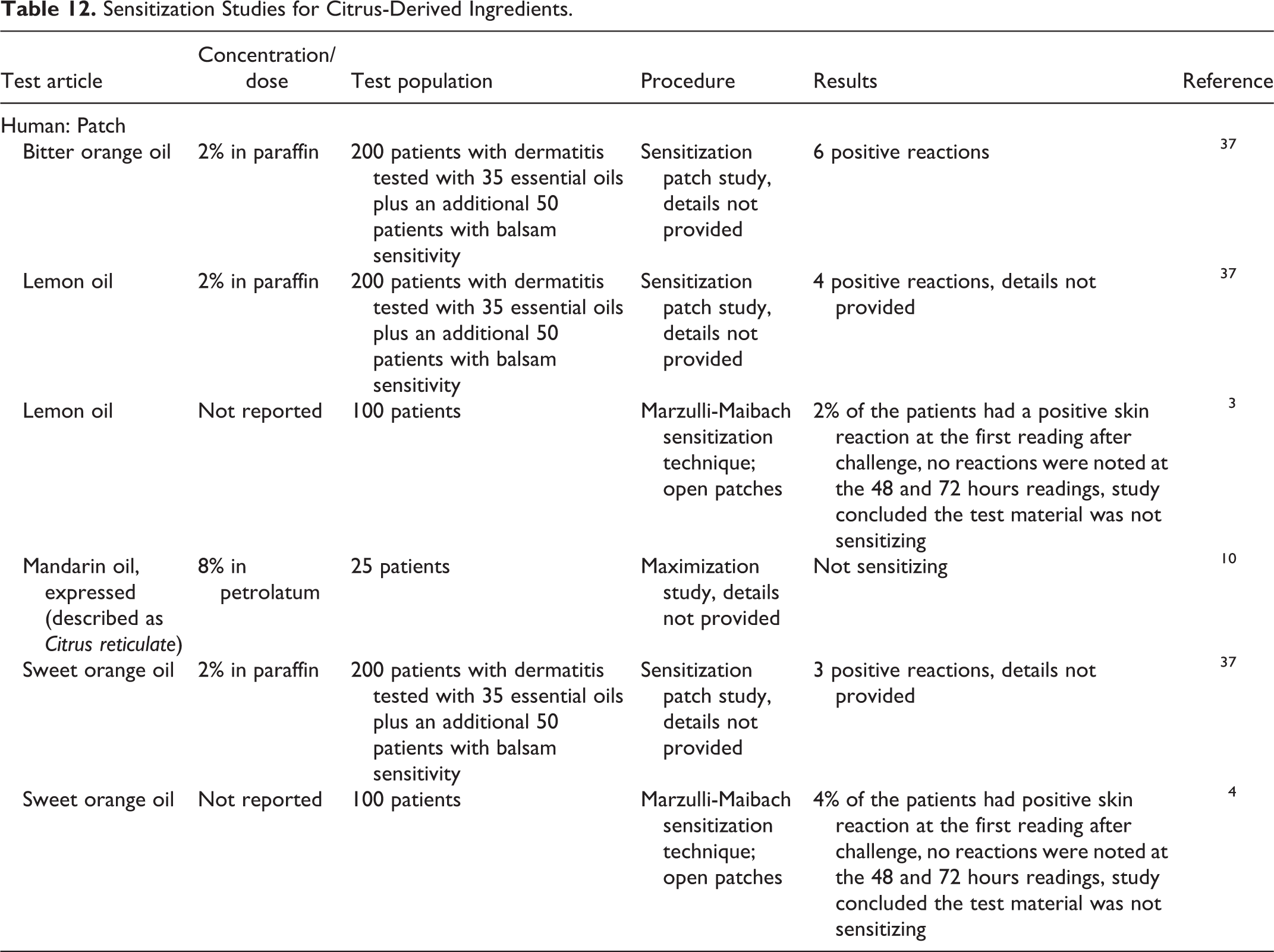
Sensitization
Sensitization studies are presented in Table 12. Mandarin peel oil (8% in petrolatum) was not sensitizing in human maximization tests. In studies of 250 patients with dermatitis, less than 2.5% had positive reactions to bitter orange oil, lemon oil, or sweet orange oil tested at 2% in paraffin. 3,4,10,37
Sensitization Studies for Citrus-Derived Ingredients.
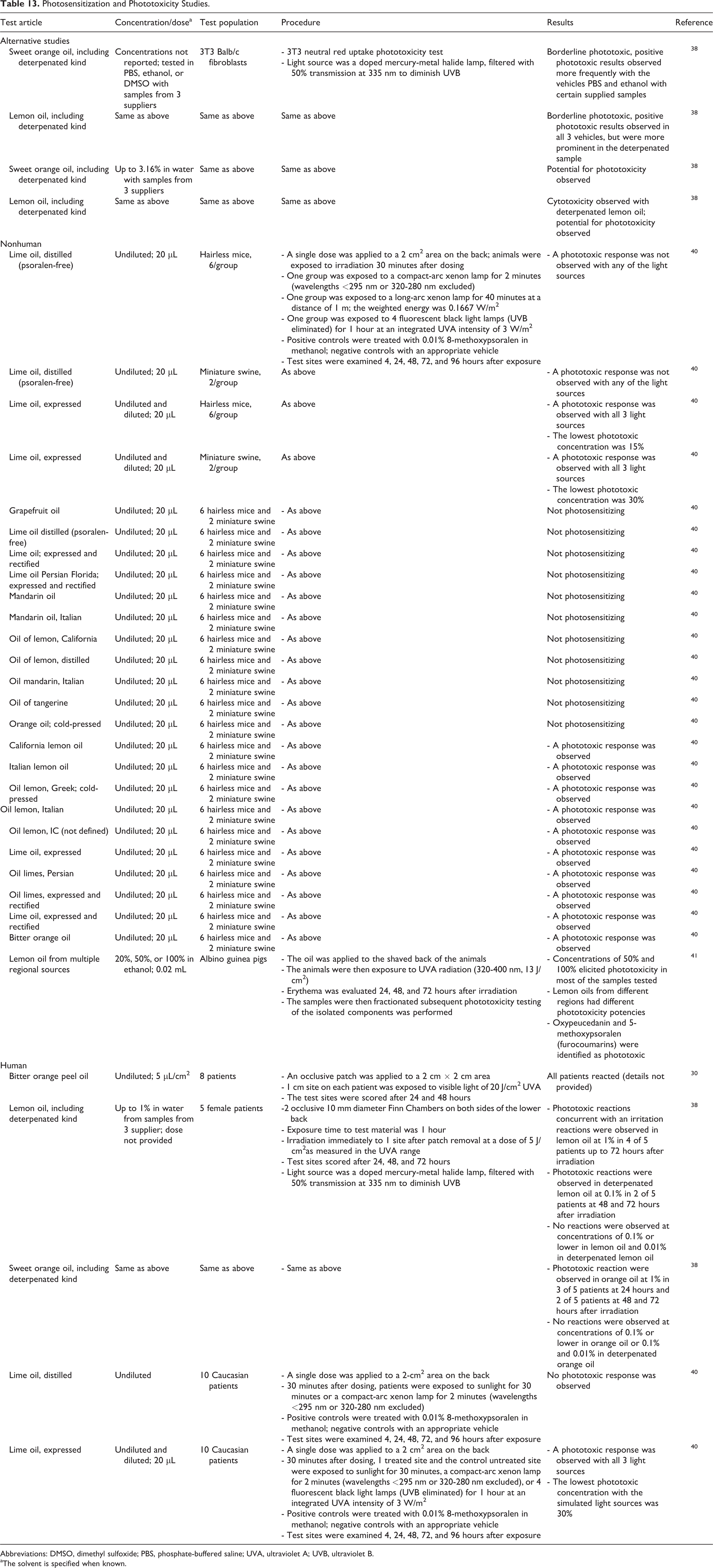
Phototoxicity and Photosensitization
Phototoxicity and photosensitization studies are presented in Table 13. Phototoxic potential was observed for sweet orange oil and lemon oil in in vitro studies. No phototoxic responses were noted in some animal studies of lime oil, lemon oil, grapefruit oil, mandarin oil, or tangerine oil; however, signs of phototoxicity were observed in response to undiluted lime oil and lime oil diluted to concentrations of 15%, undiluted bitter orange oil, and undiluted lemon oil and lemon oil diluted to concentrations of 50%. In human studies, phototoxic reactions were observed in response to undiluted bitter orange oil, lemon oil (1%), sweet orange oil (1%), and undiluted and diluted expressed lime oil (30%). Many of the citrus-derived peel oils contain constituents that are photoactive agents, although those noted to be furocoumarin free tended not to induce photosensitization. 19,30,38 -41
Photosensitization and Phototoxicity Studies.
Abbreviations: DMSO, dimethyl sulfoxide; PBS, phosphate-buffered saline; UVA, ultraviolet A; UVB, ultraviolet B.
aThe solvent is specified when known.
Occupational Exposure
In a retrospective study (2001-2010) of professional food handlers in Denmark, 8.5% (16 of 188) of the patients had positive skin prick test reactions to orange peel and 7.9% (15 of 191) of the patients had positive skin prick test reactions to lemon peel. 42
Summary
The 14 citrus-derived peel oils described in this report function primarily as skin conditioning agents-miscellaneous and fragrance. Botanicals such as citrus are composed of hundreds of constituents, some of which have the potential to cause toxic effects; for example, bergapten (as known as 5-MOP) is a naturally occurring, phototoxic furanocoumarin (psoralen) in citrus peel oils. Presently, CIR reviewed the information available on the potential toxicity of each of the citrus peel oil–derived ingredients as a whole, complex substance; CIR did not review the potential toxicity information on the individual constituents of which the citrus-derived ingredients are comprised. Cosmetic Ingredient Review requested information on the concentrations (including ranges, means, upper 95% confidence limits, detection limits, etc) of individual constituents in the citrus peel oil–derived ingredients used in cosmetics, to facilitate the safety assessment of these ingredients. Such information on constituents that have been identified as of concern by the Panel in previous safety assessments, or by other recognized scientific expert review bodies, is especially important.
Citrus oils contain large amounts of monoterpene hydrocarbons; limonene is present in the greatest amount, composing 38.1% to 95.8% of the oils. Citrus oils also contain sesquiterpene hydrocarbons, which are responsible for the characteristic flavor of these oils.
Citrus limon (lemon) peel oil has the most reported uses in cosmetics and personal care products, with a total of 490; more than half of the uses are in leave-on skin care preparations. The range of highest maximum use concentrations for C limon (lemon) peel oil is 0.0001% to 0.5%, with 0.5% reported in “other” skin care preparations. Citrus aurantium dulcis (orange) peel oil (reported as C sinensis [sweet orange] peel oil to the VCRP) has the second greatest number of overall uses reported, with a total of 289; about half of those uses are in leave-on skin care preparations. Citrus aurantium dulcis (orange) peel oil had a highest maximum use concentration range of 0.00002% to 29%, with 29% reported in noncoloring hair conditioners.
Under the rules governing cosmetic products in the European Union, citrus-derived ingredients must have furocoumarin content below 1 mg/kg in sun-protection and bronzing products. International Fragrance Association also has issued standards for citrus oils and other furocoumarin-containing essential oils. Finished products that are applied to the skin, excluding rinse-off products like bath preparations and soaps, must not contain more than 0.0015% or 15 ppm 5-MOP. If the level of 5-MOP has not been determined, limits specified for individual oils should be observed, and when such oils are used in combination with other phototoxic ingredients, the potential additive effect should be taken into consideration and use levels should be reduced accordingly. Restrictions for furocoumarin-containing essential oils and limits on the amounts of citrus-derived oils in finished products have been recommended for bitter orange oil expressed, grapefruit oil expressed, lemon oil cold-pressed, and lime oil expressed.
The European Union also set standards for limonene, stating that limonene must be included in the list of ingredients when its concentration exceeds 0.001% in leave-on products and 0.01% in rinse-off products.
The citrus peel oils in this assessment are found in foods, and the daily exposure from food use would result in a much larger systemic dose than that resulting from use in cosmetic products. Essential oils, oleoresins (solvent-free), and natural extractives (including distillates) derived from some citrus fruits are GRAS for their intended use in foods for human and animal consumption.
The dermal LD50 of undiluted mandarin peel oil (C reticulata) and undiluted sweet orange oil was greater than 5 g/kg in rabbits. In undiluted lemon oil, the dermal LD50 was greater than 10 g/kg in rabbits.
No genotoxic effects were observed in lemon oil or sweet orange oil in bacterial reverse mutation assays, mouse lymphoma cell mutation assays, and Chinese hamster chromosome aberration assays. Tumor-promoting activity was observed in mouse skin exposed to undiluted essential oils of orange (sweet), lemon, grapefruit, or lime after pretreatment with DMBA. Related studies of 40%, 80%, or 100% orange oil following pretreatment with DMBA or urethane also reported tumor-promoting activity, although the effect was weaker in the mice induced with urethane. No tumorigenic effects were observed in mice tested with orange oil without pretreatment with DMBA or urethane. Survival rates of the mice, including controls, in these experiments were poor because of a very high incidence of renal disease.
Irritation was observed in animals treated with unspecified concentrations of mandarin peel oil. In human patients, no irritation was observed after topical exposure to lemon oil (up to 20%) or mandarin peel oil (8%).
In rabbits, lemon oil tested at 5% was not irritating and orange oil tested undiluted did not induce significant or irreversible damage to the eyes. Mandarin peel oil (8% in petrolatum) was not sensitizing in human maximization tests. In studies of 250 patients with dermatitis, less than 2.5% had positive reactions to bitter orange oil, lemon oil, or sweet orange oil tested at 2% in paraffin.
Phototoxic potential was observed for sweet orange oil and lemon oil in in vitro studies. No phototoxic responses were noted in some animal studies of lime oil, lemon oil, grapefruit oil, mandarin oil, or tangerine oil; however, phototoxic reactions were observed in response to undiluted lime oil and lime oil diluted to concentrations of 15%, undiluted bitter orange oil, and undiluted lemon oil and lemon oil diluted to concentrations of 50%. In human studies, phototoxic reactions were observed in response to undiluted bitter orange oil, lemon oil (1%), sweet orange oil (1%), and undiluted and diluted expressed lime oil (30%). Many of the citrus-derived peel oils contain constituents that are photoactive agents, although those noted to be furocoumarin free tended not to induce photosensitization.
A retrospective occupational study of food handlers noted positive reactions to orange and lemon peels. No published studies on reproductive and development toxicity of citrus-derived peel oils were discovered and no unpublished data were submitted to address these topics.
Discussion
The citrus ingredients in this assessment are found in foods, and daily exposures from the consumption of foods can be expected to yield much larger systemic exposures to these ingredients than those from the use of cosmetic products. Essential oils, oleoresins (solvent-free), and natural extracts (including distillates) derived from some citrus fruits are GRAS in foods and animal feeds. Additionally, volatile oils of limes, lemons, grapefruits, bitter oranges, oranges, and tangerines are used as flavoring agents. Consequently, the primary focus of this safety assessment is on the potential for irritation and sensitization from dermal exposures to the citrus ingredients.
Most of the reports that the Panel reviewed were not sufficiently detailed to enable determining how well the substances tested represent the cosmetic ingredients included in this safety assessment. For example, the genus and species of the plants from which the test substances were derived, the cultivation methods used to grow the plants, and the methods of extraction are not specified in many of these studies.
The Panel expressed concern about the potential for constituents in citrus-derived peel oils, including the furocoumarin 5-MOP, to cause phototoxicity. International Fragrance Association has issued standards for citrus oils and other furocoumarin-containing essential oils, and the Panel agreed that adherence to the IFRA standards for such constituents will prevent phototoxicity. According to these standards, finished products that are applied to the skin, excluding rinse-off products, must not contain more than 0.0015%, or 15 ppm, 5-MOP. An IFRA standard also has been issued for 7-methoxycoumarin; based on established maximum levels of this substance from commercially available natural sources (like essential oils, extracts, and absolutes), exposure to 7-methoxycoumarin from the use of these oils and extracts is regarded to be acceptable if the level of 7-methoxycoumarin in the finished product does not exceed 100 ppm.
Additionally, based on the findings of a rodent carcinogenicity study in which tumor promotion activity may have been caused by repeated skin irritation and resultant proliferation of DMBA-treated basal cells, the Panel concluded that citrus-derived peel oils could potentially act as tumor promoters if formulated to reach irritant levels. Thus, these botanical ingredients must be formulated to be nonirritating.
The Panel noted that, because botanical ingredients are complex mixtures, there is concern that multiple botanical ingredients may each contribute to the final concentration of a single constituent. Therefore, when formulating products, manufacturers should avoid reaching levels in final formulation of plant constituents that may cause sensitization or other adverse effects. Specific examples of constituents that could induce adverse effects are limonene, citral, and other monoterpenes, furocoumarins (such as 5-MOP and 7-methoxycoumarin).
Finally, the Panel expressed concern about pesticide residues and heavy metals that may be present in botanical ingredients. They stressed that the cosmetics industry should continue to use current good manufacturing practices to limit impurities.
Conclusion
The CIR Expert Panel concluded the citrus-derived peel oils are safe for use in cosmetic products, excluding rinse-off products, do not contain more than 0.0015% (15 ppm) 5-MOP, and when formulated to be nonsensitizing and nonirritating.
C aurantifolia (lime) peel oil*
Citrus aurantium amara (bitter orange) peel oil
Citrus aurantium currassuviensis peel oil*
Citrus aurantium dulcis (orange) peel oil
Citrus clementina peel oil*
Citrus grandis (grapefruit) peel oil
Citrus iyo peel oil*
Citrus junos peel oil
Citrus limon (lemon) peel oil
Citrus medica vulgaris peel oil*
Citrus nobilis (mandarin orange) peel oil
Citrus reticulata (tangerine) peel oil*
Citrus tachibana/reticulata peel oil*
Citrus tangerina (tangerine) peel oil *
*Not reported to be in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
Burnett, C. contributed to conception and design; acquisition, analysis, and interpretation; drafted manuscript; and critically revised manuscript. Fiume, M., Gill, L., and Heldreth, B. contributed to analysis and interpretation and critically revised manuscript. Bergfeld, W., Belsito, D., Hill, R., Klaassen, C., Liebler, D., Marks, J. Shank, R., Slaga, T., and Snyder, P. contributed to conception and design; analysis and interpretation; and critically revised manuscript; and gave final approval. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.