Abstract
COVID-19 is a rapidly spreading disease, posing a huge hazard to global health. The plasmid vaccine pTK1A-TPA-SpikeA (named COVID-
Introduction
COVID-19 has rapidly spread since its outbreak in Wuhan, China, in 2019, and with increasing numbers of confirmed cases and almost 4 million deaths (https://www.worldometers.info/coronavirus/), it poses a major hazard to global public health and a huge economic burden. 1 Since it has been proven extremely difficult to control severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) transmission, due to its highly infectious properties and asymptomatic carriers, vaccination is probably the best approach to control the epidemic. 2,3 Indeed, a large number of vaccine candidates are in development, and several of them have been approved and showed positive results. 4,5
The TKSB01 plasmid vaccine (pTK1A-TPA-SpikeA, named COVID-
It was developed for intramuscular injection followed by a short electrical stimulation, a method termed electro-gene-transfer (EGT) or electroporation (EP). 7 This method aims to improve DNA vaccine immunogenicity by enhancing the passage of cell and nuclear membranes, thus facilitating DNA delivery. 8 - 11 Additionally, the associated local inflammation results in activation of the adaptive immune system. 12 -15
Since the SARS-CoV-2 vaccines are developed as preventive measures to be administered to a healthy population, it is of utmost importance to properly evaluate the safety of such vaccines in well-designed and GLP-compliant preclinical toxicity studies. 16 -18 Therefore, the aim of this study was to assess the systemic toxicity and local tolerance of the TKSB01 plasmid in Sprague Dawley (SD) rats, delivered intramuscularly and followed by EP.
Materials and Methods
Animal Husbandry and Maintenance
The in vivo part of this study was carried out in the GLP-certified Italian facility European Research Biology Center (ERBC) S.r.l. located in Pomezia, Rome, Italy. A total of 130 Hsd: SD rats (65 males and 65 females), 6 to 7 weeks old and with body weight of approximately 175 to 200 grams for males and 150 to 175 grams for females, were used (supplied by Envigo RMS srl, San Pietro al Natisone, UD, Italy). The animals were housed in a limited access rodent facility in ERBC, which is fully accredited by Association for Assessment and Accreditation of Laboratory Animal Care International. Animal room controls were set to maintain temperature and relative humidity at 22 °C ± 2 °C and 55% ± 15%, respectively. The animals were housed up to 5 of one sex in a cage. Drinking water and a commercially available laboratory rodent diet (4 RF 21, Mucedola S.r.l.) were offered ad libitum throughout the study except for the overnight fasting applied for clinical pathology investigation. The SD rat was the species and strain of choice because it had been proven to be a relevant model in terms of immunogenic response in previous preliminary studies. 6 Furthermore, it is accepted by many regulatory authorities and there are ample experience and background data on this species and strain. Procedures and facilities were compliant with the requirements of the Directive 2010/63/EU on the protection of animals used for scientific purposes. The national transposition of the Directive is defined in Decreto Legislativo 26/2014. Aspects of the protocol concerning animal welfare have been approved by the animal welfare body. The study plan is in line with regulatory indications reported in the World Health Organization guideline relevant for toxicity studies of vaccines. 19
Experimental Design
The animals were randomized into 4 groups: groups 2 to 4 comprised 20 male and 20 female rats. The first 10 females and 10 males of each group of 20 were killed in day 30, 2 days after the last administration (early sacrifice). The remaining 10 animals/sex were allowed a 4-week recovery period and were sacrificed on day 57 (late sacrifice). Group 2 animals acted as procedure control group being subjected to administration with PBS plus EP. An additional control group (group 1) of 5 animals per sex were administered with PBS without EP and were killed in day 30 (early sacrifice). The intramuscular dosing was delivered approximately every 2 weeks for a total of 3 administrations (days 1, 14, and 28) alternating left and right thigh. As indicated in Table 1 and Figure 1, the left side was always treated twice while the right side received only one dose.
Study Design.

Abbreviations: EP, electroporation; PBS, phosphate-buffered saline.
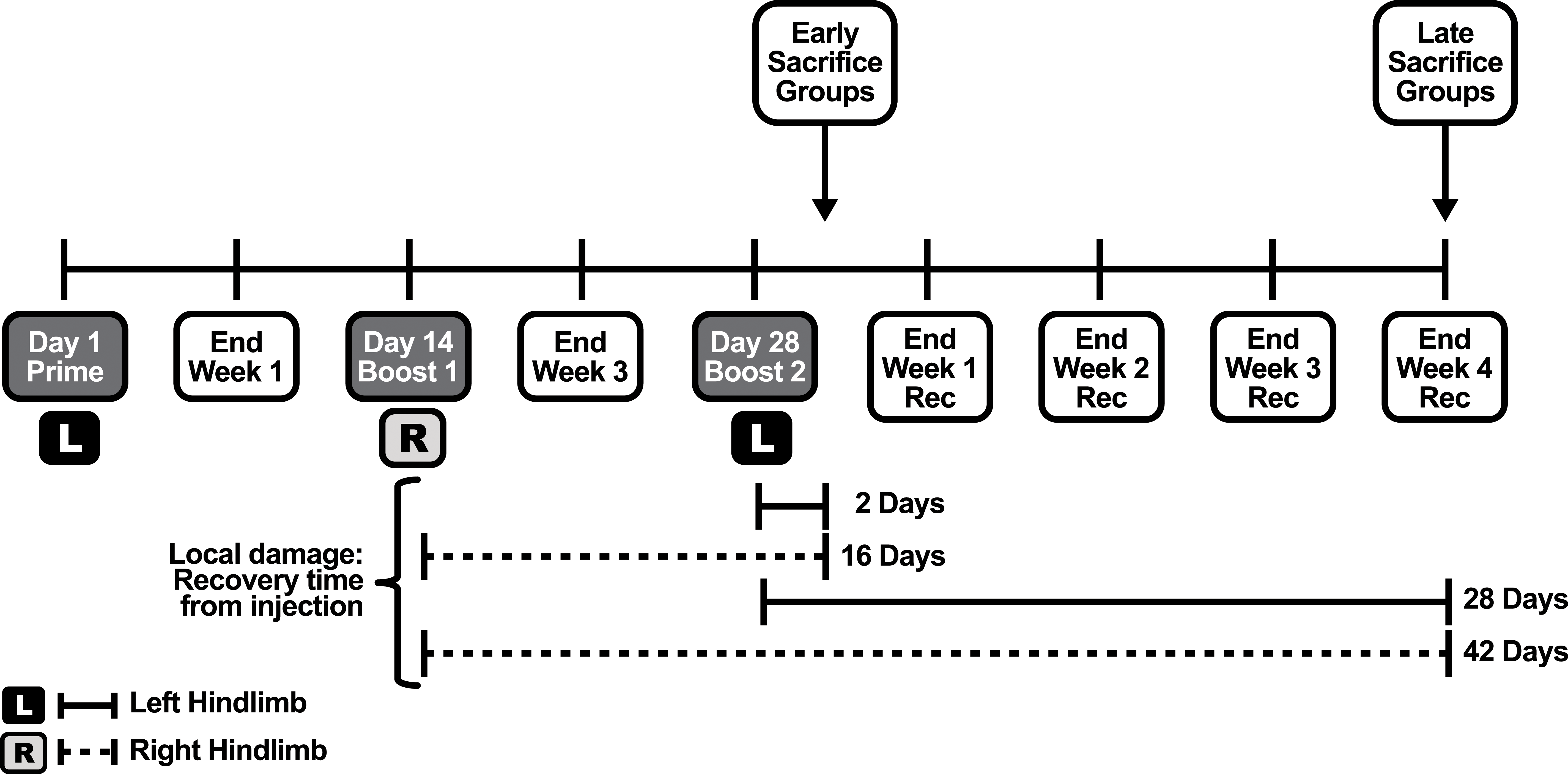
Study design scheme.
The test item (TKSB01 Plasmid, pTK1A-TPA-SpikeA) was manufactured by Biomay (Vienna Competence Center). The intramuscular dosing was performed by using the DNA vaccine delivery system, IGEA Cliniporator [Supplied by IGEA S.r.l., Carpi (MO), Italy], which is composed of a power unit connected to an electrode for intramuscular application and managed by a console running the Cliniporator software. This has facilitated the injection of the plasmid in a specific position of the IGEA’s needles geometry and allowed the EP of the volume between the needles. To guide proper dosing and, at termination, correct tissue sampling, prior to the first administration, and as necessary during the course of the study, the thigh was shaved, and the treatment area was marked with a permanent marker pen. The COVID-
The following EP conditions were used: 8 pulses, 110 V, corresponding to 275 V/cm, 20 ms length, 120 ms pause length, total duration: 1 second. These conditions were selected to maximize the intracellular exposure of the animals to the plasmid and to study the local toxicity and its reversibility under the worse possible EP conditions.
The doses have been selected between the maximum ethically feasible dose (high dose level, 420 µg/rat) and a dose of 105 µg/rat that induces a significant immune response in this animal species. Overall, the high dose is 1/5th of the highest dose of 2 mg to be used in the first in-human study. When expressed per unit of body weight, this dose equates approximately to 2 mg/kg, which is 50-fold higher than the highest dose of 0.04 mg/kg (i.e., 2 mg, assuming a 50 kg body weight). The repeat-dose GLP toxicology study was conducted with 2 dose levels of 105 and 420 µg of COVID-
Dose and Volume of Administration.

Observations and Examinations
In-life monitoring of body weight, food consumption, body temperature, daily clinical observations, clinical pathology, and ophthalmoscopy was carried out during the study.
All online data were collected and statistically analyzed by the computerized system Pristima, Version 6.4.1. On each day of dosing, the clinical observations were performed at the same time intervals after dosing: session 1—soon after dosing, session 2—0.5 to 1 hours post dose. The injection site was examined at predose, 24 and 48 hours after each administration, once a week during the recovery period, and on the day of necropsy. The body temperature was measured before each administration and at approximately 6, 24, and 48 hours post dose.
Three sessions of ophthalmoscopy were carried out: before the start of treatment, at the completion of the 3 injections (study day 29), and the end of the recovery period (study day 56). The ophthalmoscopy was performed by means of an ophthalmoscope and by a slit-lamp microscope, after the instillation of 0.5% Tropicamide (Visumidriatic, Visufarma S.p.A.). The integrity of the fundus of the eyes and the transparency (and integrity) of the crystalline lens, uveal tract, iris, and cornea were assessed.
The day of each scheduled necropsy (study days 30 and 57), individual overnight urine samples were collected from all male and female animals and analyzed for turbidity, specific gravity, pH, protein, glucose, and ketones content. The sediment was also microscopically examined for the presence of cellular components, crystal, spermatozoa, and precursors.
As part of each sacrificial procedure, blood samples were collected from the abdominal vena cava of all animals, overnight deprived of food, under profound isoflurane anesthesia, and analyzed for hematology, coagulation (prothrombin time, activated partial thromboplastin time, fibrinogen by Instrumentation Laboratory ACL Elite PRO), and clinical chemistry. The hematological analysis provided standard red and white blood cell counts including large unstained cells (LUCs) and hemoglobin concentration (Siemens Advia 120).
The clinical chemistry parameters were analyzed by Siemens Advia 1800 and included alkaline phosphatase, alanine transaminase, aspartate aminotransferase, blood urea nitrogen, creatine kinase, urea, creatinine, glucose, gamma-glutamyl transferase, triglycerides, lactate dehydrogenase, total bile acids, total bilirubin, total protein, albumin, globulin, A/G ration, and electrolytes. The serum sample was also assayed for α-2 macroglobulin (A2M) by enzyme-linked immunosorbent assay (ELISA) method as acute inflammation marker.
All animals were killed by exsanguination under isoflurane anesthesia at the completion of the scheduled test period and subjected to necropsy. A detailed
A full list of tissues was obtained for histopathology and included the following tissues/organs: abnormalities, adrenal glands, aorta, bone marrow (from sternum) brain, cecum, colon, duodenum, epididymides, eyes, Harderian glands, heart, ileum, injection site (left and right quadriceps), injection site (left and right overlying skin), jejunum (including Peyer’s patches), femur with joint, kidneys, larynx, liver, lungs (including mainstem bronchi), lymph nodes (cervical, inguinal, iliac, mesenteric), mammary area, nasal cavity, esophagus, optic nerves, ovaries, oviducts, pancreas, parathyroid glands, pituitary gland, prostate gland, rectum, salivary glands, sciatic nerve, seminal vesicles, skeletal muscle, skin, spinal column, spinal cord, spleen, stomach, testes, thymus, thyroid gland, tongue, trachea, ureters, urinary bladder, uterus—cervix, and vagina. Samples of all the tissues were fixed and preserved in 10% neutral buffered formalin (except eyes, optic nerves, Harderian glands, testes, and epididymides, which were fixed in Modified Davidson’s fluid and preserved in 70% ethyl alcohol).
Tissue processing was carried out in Propath UK Ltd., Hereford, United Kingdom, while histopathology evaluation was performed in Envigo CRS Israel Limited, Ness-Ziona, Israel. After dehydration and embedding in paraffin wax, sections of the tissues were cut at 5-µm thickness and stained with hematoxylin and eosin. With reference to the injection site, the quadriceps muscle from both sides and from all animals were divided into 3 portions (i.e., cranial, central, distal) and embedded in paraffin. This trimming procedure followed the recommendation of Sellers et al, 18 in order to achieve optimal sampling of the site of injection. From the overlying skin, a single sample was processed, corresponding to the central portion of the skin injection site. One slide per block was prepared to produce a total of 3 sections per each muscle and 1 section per each skin on each side.
Histopathological evaluation was performed on all tissues from animals killed on day 30 from groups 2 to 4. For group 1, only the injection site (quadriceps muscle and the overlying skin) was evaluated. In addition, all abnormalities from all animals (groups 1-4) killed on day 30 were evaluated. For the late sacrifice animals, only the injection sites were evaluated.
Histopathological changes were described and scored by the study pathologist, using semiquantitative grading of 5 grades (0-4), taking into consideration the severity of the changes 20 :
Grade 0: No changes observed.
Grade 1: Minimal changes.
Grade 2: Mild changes.
Grade 3: Moderate changes.
Grade 4: Severe changes.
Immunogenicity
As part of this repeated dose toxicity study, humoral immune–mediated responses to the vaccine were investigated to prove the relevance of the rat species in terms of immunological responsiveness. Prior to necropsy on days 30 and 57, blood samples were collected from the abdominal vena cava of all animals in tubes without anticoagulant. Serum was separated and stored frozen at −80 °C until transfer to Takis immunology laboratories where the bioanalyses were carried out.
The method applied consisted of an ELISA sandwich format Antibody titer was calculated starting from the optical density (OD) readings from the ELISA experiments. In brief, MaxiSorp ELISA plates (Nunc) were coated with 1 µg/mL of the recombinant RBD (protein produced by the Sponsor) in PBS for overnight adsorption at 4 °C. Plates were washed in PBS/Tween (0.05% vol/vol) and wells blocked using 3% bovine serum albumin (A2153 Sigma-Aldrich) in PBS-Tween20 (0.05%). Serum samples serially diluted in PBS-Tween20 (0.05%) containing 1% of BSA were added and incubated overnight at 4 °C.
At the completion of the incubation period, the plates were washed and the secondary goat anti-Rat immunoglobulin G (IgG; whole molecule; Sigma-Aldrich A8438 Lot.SLBW6479), labeled with alkaline phosphatase was added at 1:2000 dilution for 1 hour at room temperature. After washing p-nitrophenyl phosphate (Liquid Substrate System for Elisa, Sigma P7998) was added. The OD values for each well were measured at 405 nm.
Each plate included 6 replicates of diluent (blank). The average value of blanks was calculated and subtracted to sample values. Each serum sample was tested in duplicate at different dilutions within the same plate. Optical density mean value of the 2 individual readings was calculated and subtracted from the blank mean plate value.
The threshold for positivity was set at 3× the OD value of the negative control (serum obtained from animals before the start of the experiment, i.e., the basal values) or 0.2, whichever was higher, for the calculation of the IgG end point values.
Based on results, end point titers (EPTs) were calculated according to Van Doremalen et al, 21 slightly modified, as follows: the OD against log10 sample dilution was plotted and linear regression analysis of this curve allowed calculation of the EPT with a threshold for positivity set at 3 times the background or 0.2 OD, whichever was higher (experimental end point). All animals with coefficients of correlation (r) below 0.9 or having the first sample dilution OD lower than the experimental EP were excluded from the analysis. The ELISA method was not formally validated, but amply applied with the same experimental conditions in all pharmacology experiments carried out in mice. 6
Statistical Analysis
The statistical analysis was applied by comparing each test item treated group with the control group 2 that received PBS as vehicle followed by EP. Standard deviations were calculated as considered appropriate by the validated computerized system Pristima, Version 6.4.1. For continuous variables, the significance of the differences among groups was assessed by analysis of variance. Differences between each treated group and the control group 2 were assessed by Dunnett’s test using a pooled error variance. The homogeneity of the data was verified by Bartlett’s test before Dunnett’s test. If the data were found to be inhomogeneous a Modified
Results
Mortality, Clinical Signs, and Body and Organ Weights
No premature deaths occurred in the study and no clinical signs of toxicological significance were recorded throughout the study. Careful observations of the injection sites did not reveal signs of redness, swelling, scab, or other macroscopic alterations during the in-life phase. No limping or difficulty in ambulation was recorded. Some statistically significant increases in rectal body temperature were obtained at 6, 24, or 48 hours post injection in treated males and/or females compared to the control group (group 2). However, these differences were minimal and within the normal body temperature range in rats.
Body weight was unaffected by treatment throughout the study and the food consumed was comparable between groups throughout the study. No treatment-related lesions were recorded during the ophthalmoscopy evaluations.
Clinical Pathology and Organ Weights
At the end of the dosing phase, 2 days after the last administration, LUCs were statistically significantly higher in treated animals compared to the control group (Table 3). This cell population represents activated lymphocytes, and LUCs may be a valuable marker of activation and innate immune system stimulation. 22
Selected Hematology Parameters at the End of the Dosing Phase, 2 Days After the Last Administration.
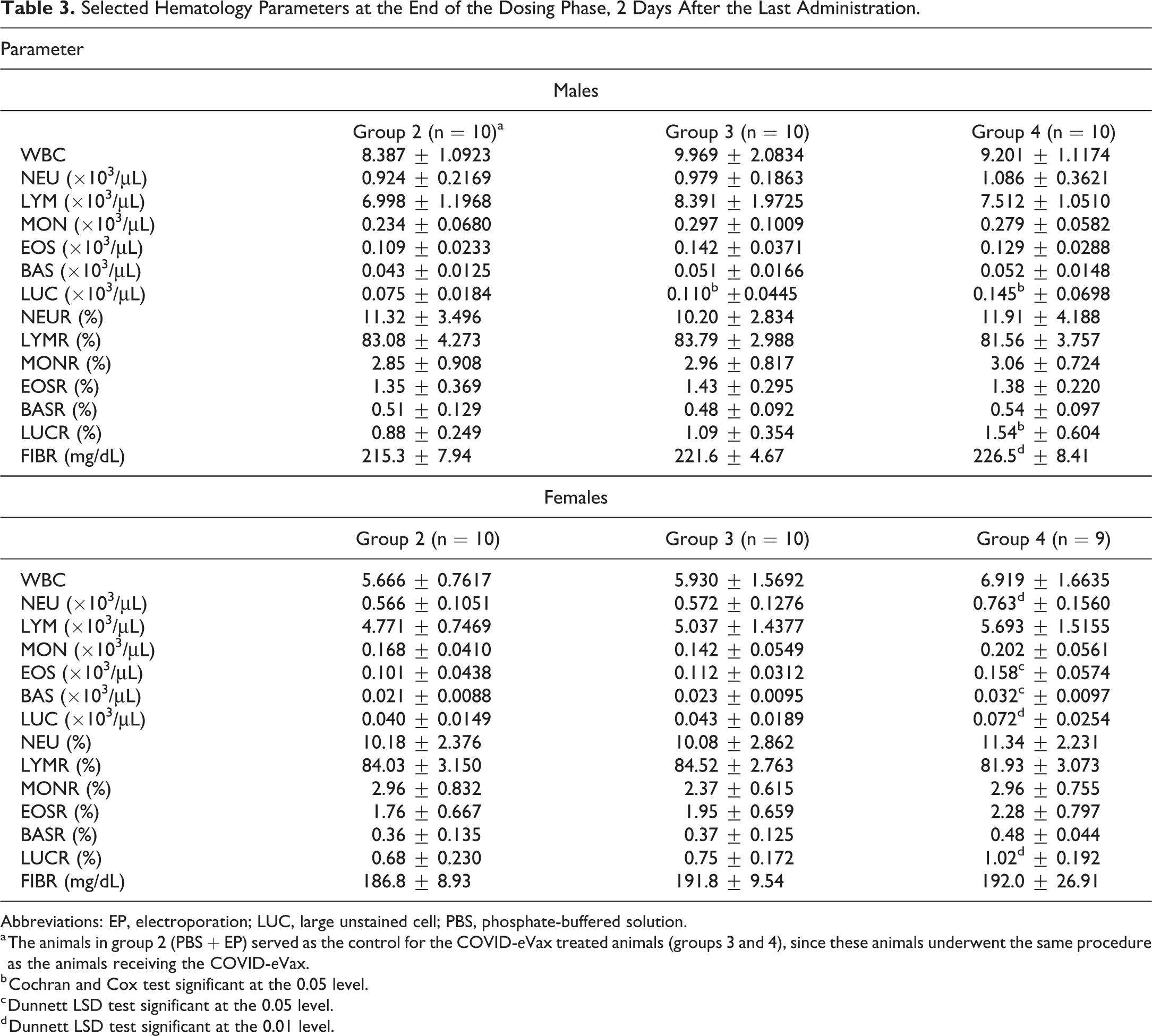
Abbreviations: EP, electroporation; LUC, large unstained cell; PBS, phosphate-buffered solution.
a The animals in group 2 (PBS + EP) served as the control for the COVID-
b Cochran and Cox test significant at the 0.05 level.
c Dunnett LSD test significant at the 0.05 level.
d Dunnett LSD test significant at the 0.01 level.
Several females dosed at 420 μg/animal showed an increase of the other white blood cell subpopulations (Table 3), such as neutrophils, eosinophils and basophils. All findings recorded at the end of the dosing phase showed reversibility at the end of the recovery phase. A statistically significantly increase of fibrinogen was observed in males dosed at 420 μg/animal. No changes were recorded at the end of the recovery phase.
No changes of toxicological significance were found in the clinical chemistry evaluations, or measurement of α-2-macroglobulin, a major acute-phase protein that responds in rats similar to the c-reactive protein in humans. 23 The translational value of A2M or α-1 acid glycoprotein as additional inflammatory biomarkers in rat toxicity studies was also recently highlighted by Reagan et al. 24
No significant signs of acute inflammation were reported in the animals killed 48 hours after the last injection (day 30) and those killed after 4 weeks of recovery (day 57). No changes were observed in the urinalysis tests. No differences in the organ weights were recorded among groups on day 30 and on day 57.
Immunogenicity
Enzyme-linked immunosorbent assay results and RBD-binding IgG EPT calculations showed that the intramuscular injections of COVID-
At sacrifice of the main groups (i.e., 2 days after the last administration of COVID-
On the contrary, all COVID-
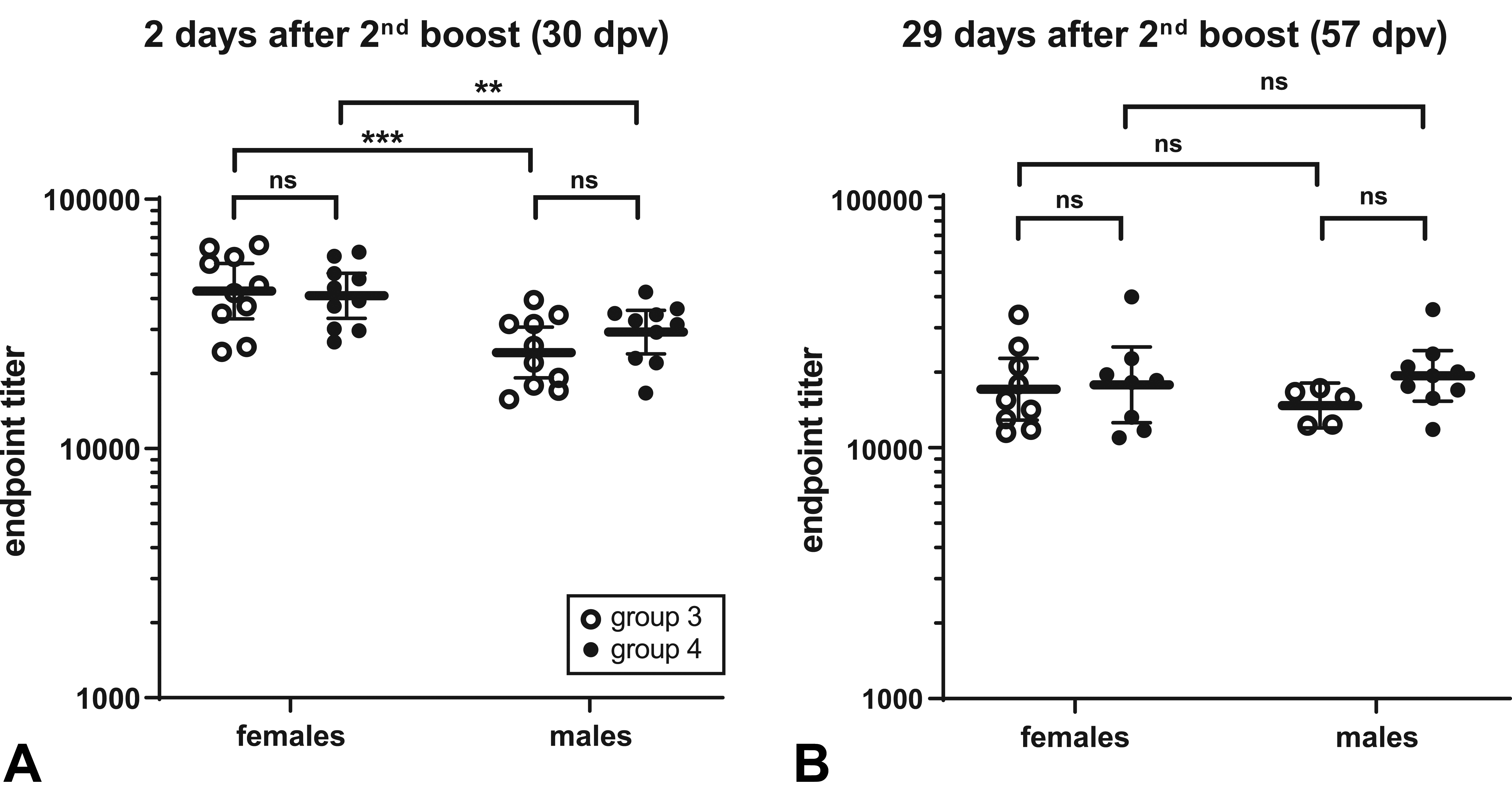
Immunoglobulin G (IgG) titer in female and male rats treated with COVID-
In the recovery group lower but still relevant EPTs have been calculated. In this case, the difference between the 2 sexes is no longer evident (Figure 2B). These data point toward a sustained immunity after COVID-
Macroscopic Observations
Dosing phase (day 30)
Treatment-related changes were seen at the injection sites (i.e., skeletal muscle) of all treated groups, as well as in the regional lymph nodes (iliac and inguinal lymph nodes; Table 4).
Macroscopic Findings in the Injection Site and Regional Lymph Nodes.
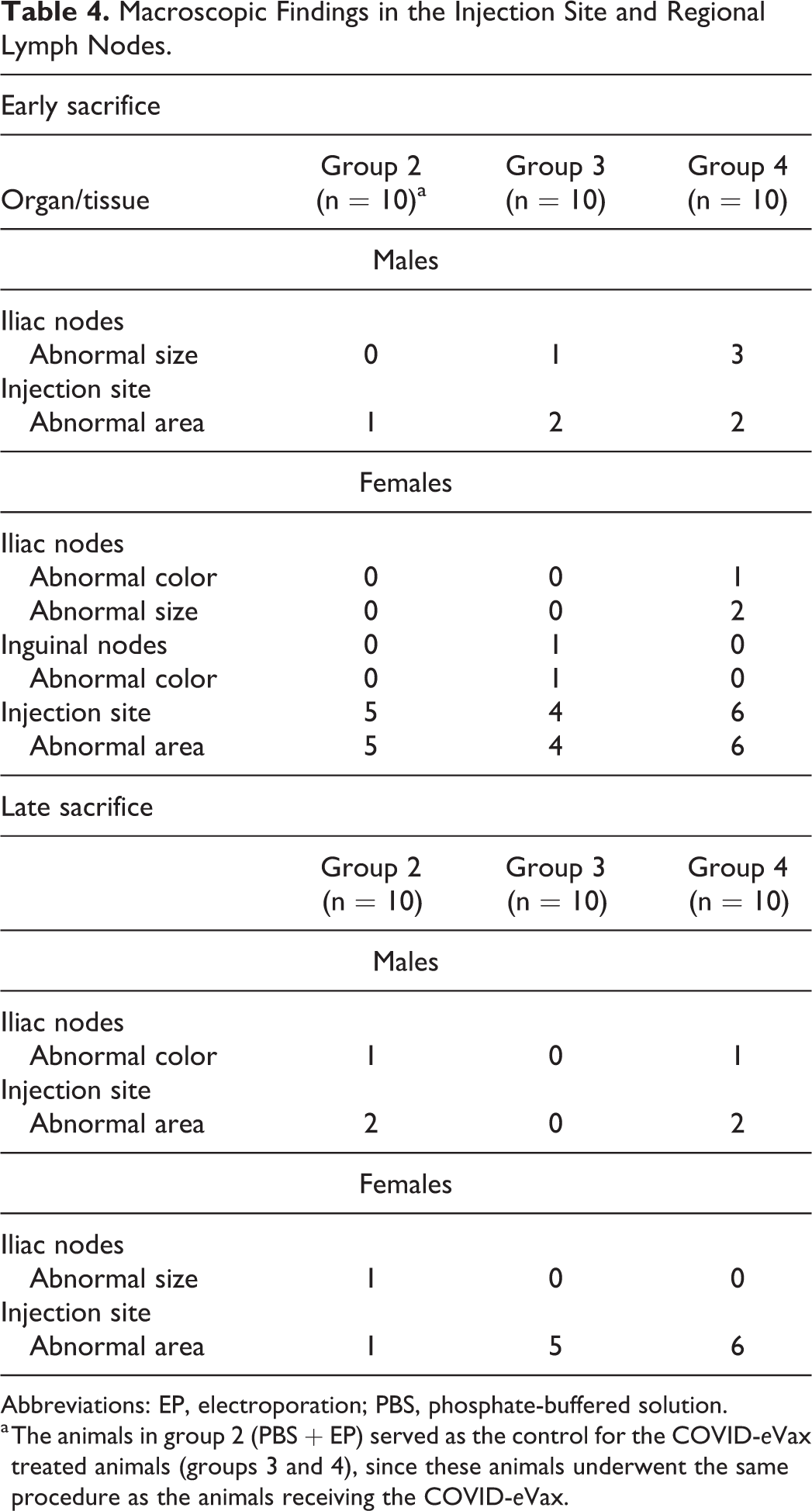
Abbreviations: EP, electroporation; PBS, phosphate-buffered solution.
a The animals in group 2 (PBS + EP) served as the control for the COVID-
The treatment-related macroscopic lesions in the iliac and inguinal lymph nodes consisted of sporadic cases of abnormal size and/or color. The treatment-related macroscopic lesions in the injection site consisted of sporadic cases of abnormal color. The changes were comparable in incidence, severity, and nature in all treated groups (i.e., group 2, 3, and 4).
Recovery phase (day 57)
Treatment-related changes were seen at the injection sites (i.e., skeletal muscle). Changes were comparable in incidence, severity, and nature in all treated groups (i.e., group 2, 3, and 4).
Microscopic Findings
Dosing phase (day 30)
Treatment-related changes were seen at the injection sites (i.e., skeletal muscle), in the overlying skin of all treated groups, as well as in the regional lymph nodes (iliac and inguinal lymph nodes; Table 5).
Selected Microscopic Findings.a
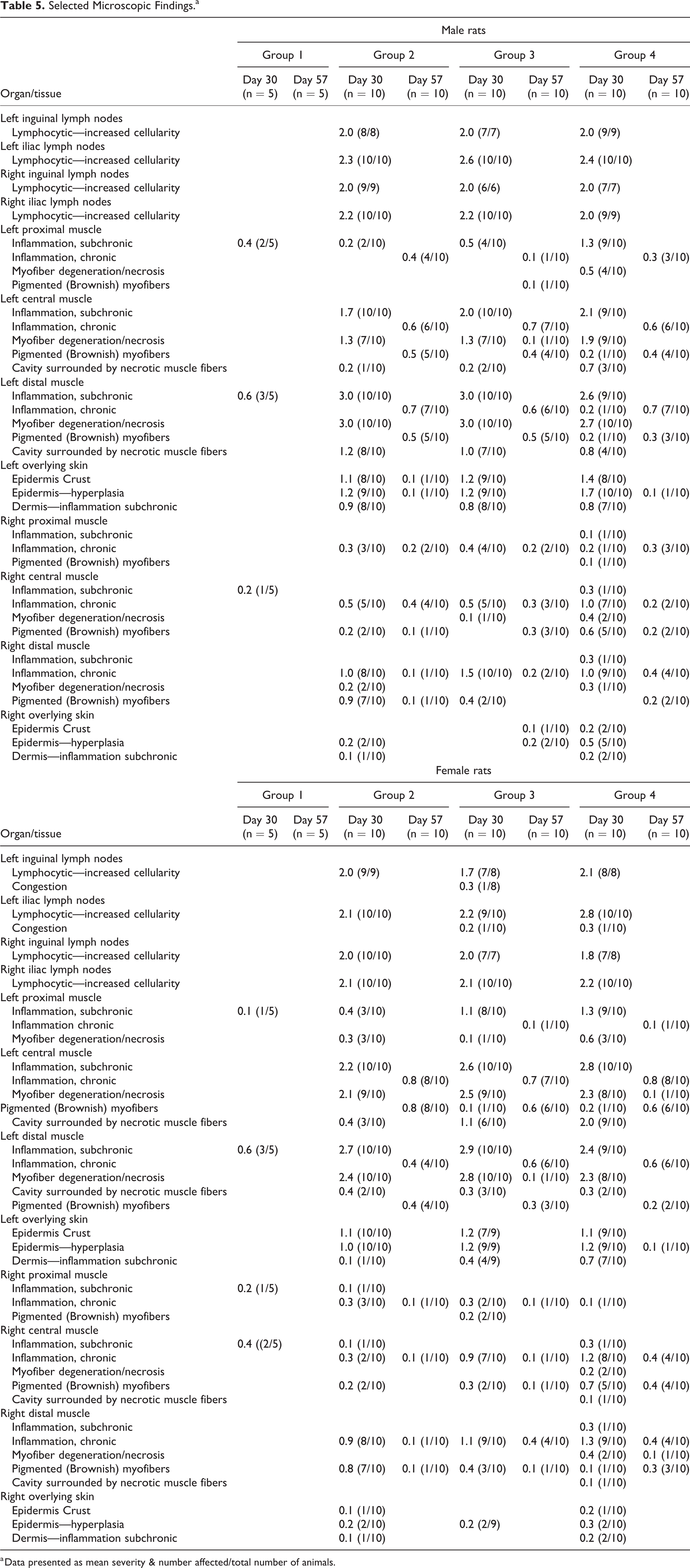
a Data presented as mean severity & number affected/total number of animals.
Skeletal muscle
Relatively more severe lesions and of acute nature were seen in the skeletal muscle of left injection sites (i.e., samples collected 2 days post last dosing session, an injection was done on day 1 and day 28 of the experiment), when comparing to the samples collected from the right injection site (i.e., samples collected 16 days post last dosing session, injection done on day 14 of the experiment), in which the lesions were chronic in nature, indicating a clear trend for recovery.
The changes in the left injection site consisted of focal muscle fiber necrosis (i.e., coagulative necrosis surrounding center of lytic necrosis) surrounded by subchronic inflammation (i.e., mixed mononuclear cell infiltration, predominantly macrophages). The lesions were mostly scored as mild to moderate, and there were no apparent changes in severity of the lesions when comparing between the groups (Figure 3A-E).
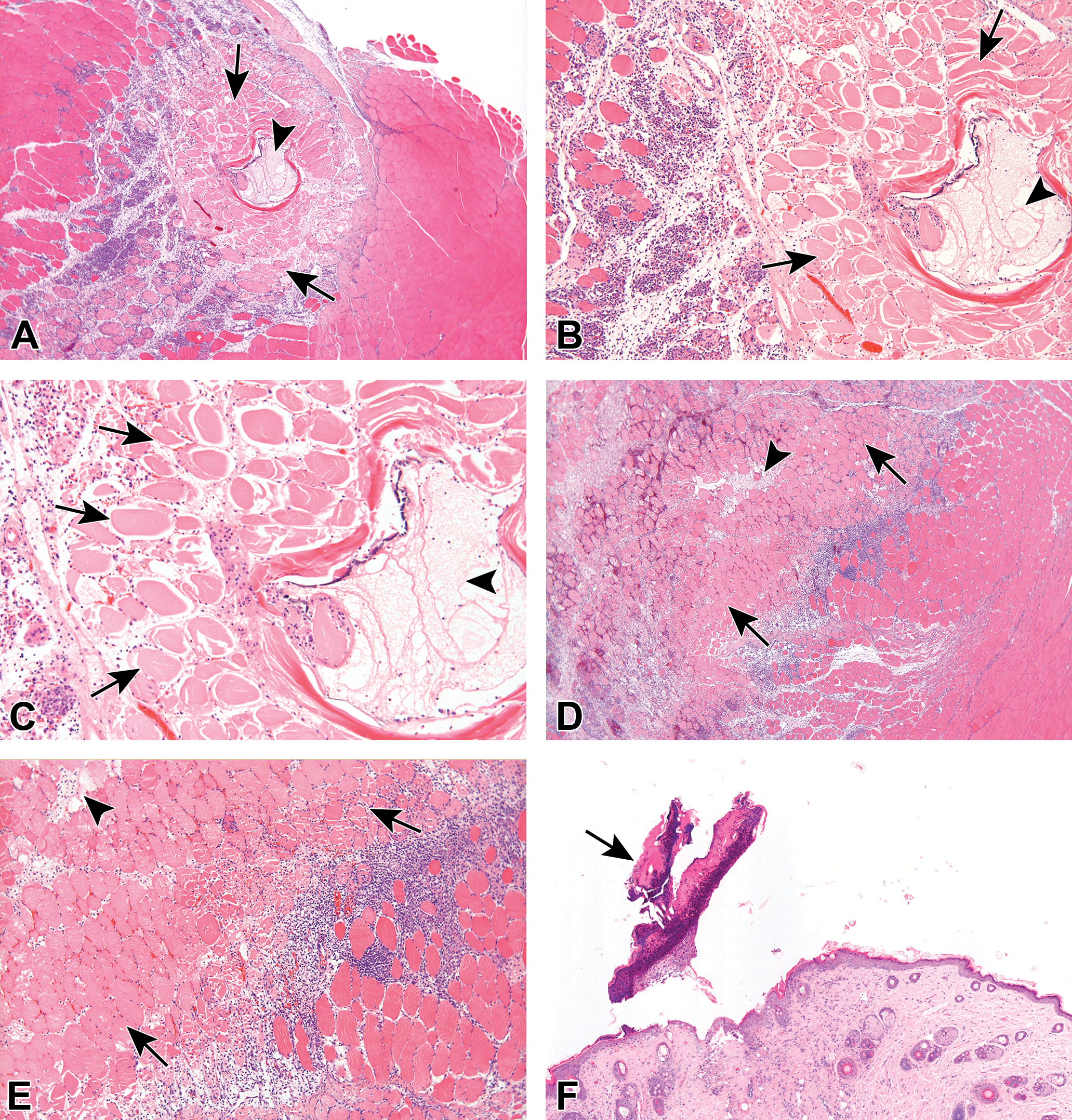
A, Histological section of the left injection site of a male rat from group 2, injected with PBS + EP (vehicle control), terminated 2 days after the last injection. The section shows muscle fiber necrosis (i.e., coagulative necrosis surrounding center of lytic necrosis) surrounded by acute to subchronic inflammation (i.e., polymorphonuclear cells, mixed mononuclear cell infiltration, predominantly macrophages). The lesions were mostly scored as mild to moderate. Arrows indicate necrotic muscle fibers, surrounded by inflammatory reaction. Arrowhead indicates cavity formation within the necrotic focus. B, Higher magnification of A. Arrows indicate necrotic muscle fibers, surrounded by inflammatory reaction. Arrowhead indicates cavity formation within the necrotic focus. C, Higher magnification of B. Arrows indicate necrotic muscle fibers, surrounded by an inflammatory reaction. In the center of the coagulative necrosis, the myofibers undergo lysis. Arrowhead indicates cavity formation within the necrotic focus. D, Histological section of the left injection site of a male rat from group 4, injected with COVID-
The changes in the right injection site consisted of chronic inflammation (i.e., mostly fibroblastic proliferation with collagen deposition and relatively minor component of mixed mononuclear cell aggregation), as well as brownish pigmented muscle fibers and macrophages. At this stage, the lesions were mostly scored as minimal to mild, and there were no apparent changes in the severity of the lesions when comparing between the groups.
Skin overlying the injection sites in the skeletal muscle
Relatively more severe lesions were seen in the skin overlying the left injection sites (i.e., samples collected 2 days post last dosing session, injection was done on day 1 and day 28 of the experiment), when comparing to the samples collected from the skin overlying the right injection site (i.e., samples collected 16 days post last dosing session, injection done on day 14 of the experiment), indicating a clear trend for recovery.
The changes in the skin overlying the left injection site consisted of focal epidermal hyperplasia, covered with minimal to mild crust, and subchronic inflammation in the underlying dermis (Figure 3F-G). The changes were scored from minimal to mild, and there were no apparent changes in severity of the lesions when comparing between the groups. Similar changes were seen in the skin overlying the right injection site; however, the lesions were always of minimal severity and much less frequent, indicating a clear trend of time-related recovery, without complications.
Regional lymph nodes, iliac and inguinal lymph nodes
The changes consisted of increased lymphocytic cellularity, scored as mild to moderate, comparably scored in groups 2, 3, and 4 (Table 5), reflecting reactive hyperplasia of the lymphoid tissue, secondary to the changes in the injection site related to the electroporation (Figure 3H-K). 25 No treatment-related changes were observed in any of the other tissues examined.
Recovery phase (day 57 of study)
Treatment-related changes were seen at the injection sites (i.e., skeletal muscle and overlying skin) of all treated groups. The changes were comparable in incidence, severity, and nature in all treated groups (i.e., group 2, 3, and 4). The changes in the injection site demonstrated a clear trend for time-related recovery, when comparing to the 30 days time point, and generally, the severity of the lesions was scored only as of minimal degree.
The changes in the injection site–skeletal muscle consisted of minimal chronic inflammation associated with brownish pigmented muscle fibers and macrophages (Figure 3L-Q). Only sporadic cases of minimal fiber degeneration/necrosis were noted. The changes in the skin overlying the injection site consisted of sporadic cases of minimal focal epidermal hyperplasia, covered with crust.
Discussion
New delivery technologies have recently accelerated the rapidly growing sector of genetic vaccines. One of these vaccination tools is the EGT technology. Electro-gene-transfer is a powerful method of DNA delivery currently employed for several medical applications, such as DNA vaccination and gene therapies. Due to its safety, efficacy, and ease of application, EGT is the ideal candidate vaccine platform as a substitute for viral gene delivery methods.
11,13,15
This procedure consists of exposing a cell or a tissue to an external electric field that increases cell membrane permeability to molecules that otherwise would cross the plasma membrane with low efficiency, and for this reason, it has been widely used in different biomedical applications.
26
In addition, EGT may enhance immune responses through increased protein expression, secretion of inflammatory chemokines and cytokines, and recruitment of antigen-presenting cells at the site of EP.
12,27
COVID-
This study summarizes the toxicological evaluation of COVID-
In the injection sites, muscle fiber necrosis associated with subchronic inflammation was evident, accompanied by focal coagulative necrosis surrounding center of lytic necrosis within the muscle, surrounded by necrotic muscle fibers. These changes are expected and are part of the normal reaction to EP, as was observed also in previous animal studies with EP. 28- 30 The presence of inflammation at the injection site, characterized by the presence of different immune cells, is considered a desirable reaction since it is hypothesized to enhance an immune reaction to the injected antigen. 31
The presence of inflammation in the animals undergoing EP was also mirrored in the hematology evaluations, showing higher LUC levels, which represent activated lymphocytes, 22 being the result of the plasmid-induced immune activation, in addition to additional inflammatory markers, such as elevated fibrinogen levels.
The fact that all changes were comparable between the groups that received the COVID-
The presence of inflammation in the injection sites of vaccine safety studies is considered a normal finding, even when the reaction is robust, as long as there are no other adverse clinical signs unrelated to injection site reactions and if there is evidence for reversibility. 18 Based on these recommendations, the reactions observed in the current study are to be interpreted as nonadverse.
In summary, based on the results obtained in the study, it can be concluded that the intramuscular EP-assisted DNA vaccine, TKSB01 plasmid (pTK1A-TPA-SpikeA), showed an excellent safety profile in SD rats under these experimental conditions, and support its further development for use in humans.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Isabella Andreini is a consultant to Rottapharm Biotech; Emanuele Marra, Laura Luberto, Daniela Stoppoloni, Maria Lucrezia Pacello and Luigi Aurisicchio are employees of Takis Biotech; and Gianfranco Caselli is an employee of Rottapharm Biotech.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Takis Biotech, Rome, Italy. Takis Research activities are supported in part by the Italian Ministry of Economic Development through grants F/050298/02/X32, F/090033/01-04/X36 and F/190180/01/X44. The authors are also grateful to Lazio Innova for the funding provided through the grant A0376-2020-0700050 Prog. T0002E0001 “Emergenza Coronavirus ed oltre.”
