Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reassessed the safety of the mixture Methylchloroisothiazolinone (MCI)/Methylisothiazolinone (MI), which functions as a preservative in cosmetic products. The Panel reviewed relevant animal and human data provided in this safety assessment, and data from the previously published safety assessment of this mixture, and concluded that MCI/MI is safe in cosmetics when formulated to be nonsensitizing, based on the results of a quantitative risk assessment or similar methodology; however, at no point should concentrations exceed 7.5 ppm in leave-on products or 15 ppm in rinse-off products.
Introduction
This safety assessment is on the combination of Methylchloroisothiazolinone (MCI) and Methylisothiazolinone (MI) as used in cosmetics. In 1992, the original report on MCI/MI was published by the Expert Panel for Cosmetic Ingredient Safety (Panel) and concluded that this mixture may be “safely used in rinse-off products at a concentration not to exceed 15 ppm and in leave-on cosmetic products at a concentration not to exceed 7.5 ppm.” 1 The stated safe-for-use concentration refers to a mixture containing 76.7% MCI and 23.3% MI (roughly, 3:1). According to the Cosmetic Ingredient Review (CIR) procedures, the Panel evaluates the conclusions of previously issued reports. The Panel determined that this safety assessment should be reopened to reassess the conclusion based on the numerous sensitization studies and reports that have been published since 1992.
While defined as separate ingredients that function as preservatives in cosmetics in the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary), 2 MCI is only known to be used in concert with MI. This safety assessment does not directly address the safety of the cosmetic use of either ingredient alone; however in 2020, the Panel assessed the safety of MI formulated without MCI and concluded that MI (alone) is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm (ie, 0.01%) and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a quantitative risk assessment (QRA) or similar methodology. 3
This safety assessment includes relevant published and unpublished data that are available for each end point that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the end points that Panel typically evaluates, is provided on the CIR website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Chemistry
Definition
Methylchloroisothiazolinone (CAS No. 26172-55-4) is the heterocyclic organic ingredient that conforms to the structure shown in Figure 1. 2
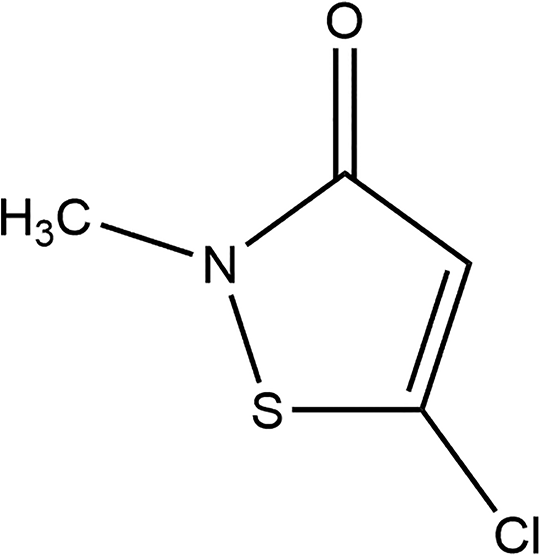
Methylchloroisothiazolinone.
Methylisothiazolinone (CAS No. 2682-20-4) is the heterocyclic organic ingredient that conforms to the structure shown in Figure 2. 2
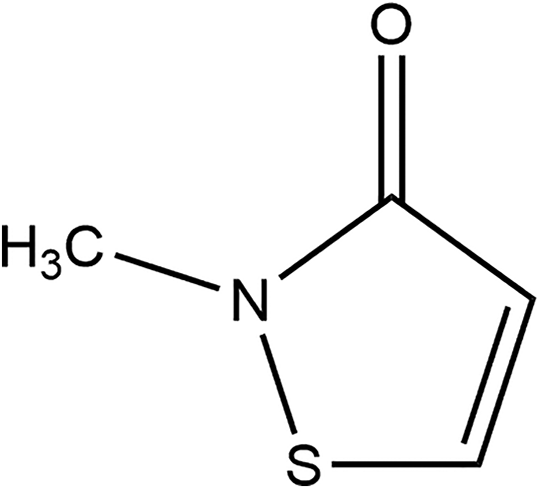
Methylisothiazolinone.
Physical Properties
MCI/MI is readily miscible in water, lower alcohols, glycols, and other hydrophilic organic solvents. 1 This mixture is a clear, light amber liquid with a specific gravity of 1.19 (at 20 °C), a pH of 3.5 (as supplied), and a freezing point of −18 to 21.5 °C.
Impurities
Dimethylnitrosamine was reported to be formed as a reaction by-product at very low concentrations. 1 To limit the presence of this impurity, methyl-3-mercaptopropionate is added during production.
Use
Cosmetic
The safety of the cosmetic ingredients included in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the cosmetics industry in response to surveys, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
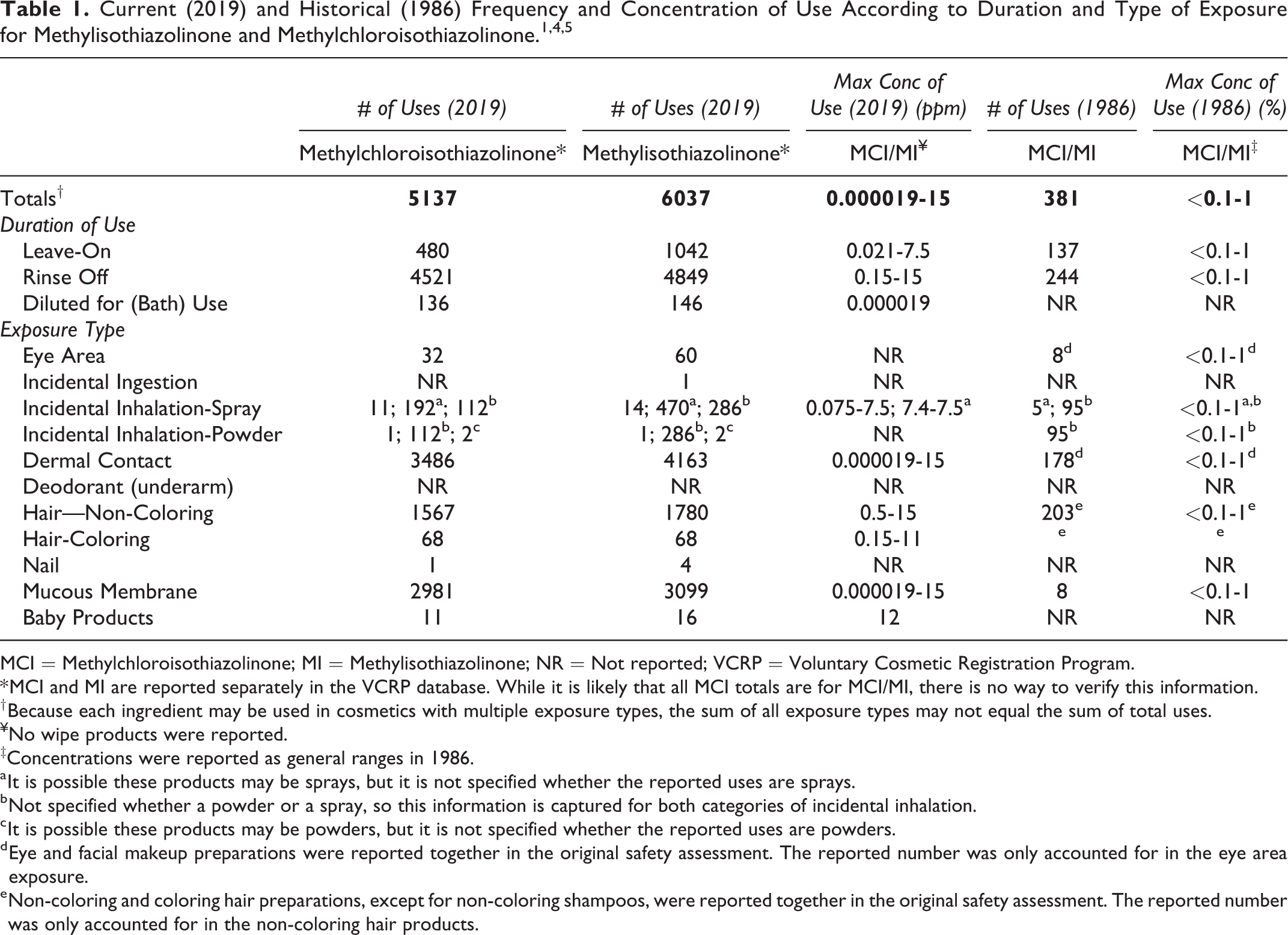
MCI and MI are reported to the VCRP separately and not as a mixture. According to 2019 VCRP survey data, the total number of uses reported for MCI is 5137; 480 of these are in leave-on products (Table 1). 4 MI has 6037 reported uses; 1042 of these are in leave-on products. The uses have increased significantly since the original report on MCI/MI was published; in 1986, the total number of uses reported for the ingredient mixture was 381. 1 In 2019, the Council reported that MCI/MI (3:1) is used at up to 7.5 ppm in leave-on products and at up to 15 ppm in rinse-off products. 5 In the original report, concentration of use was reported as a range; the concentration of use range for MCI/MI in both leave-on and rinse-off products was reported to be <0.1% to 1% (<1000-10000 ppm). 1
MCI = Methylchloroisothiazolinone; MI = Methylisothiazolinone; NR = Not reported; VCRP = Voluntary Cosmetic Registration Program.
* MCI and MI are reported separately in the VCRP database. While it is likely that all MCI totals are for MCI/MI, there is no way to verify this information.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
¥No wipe products were reported.
‡Concentrations were reported as general ranges in 1986.
a It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
c It is possible these products may be powders, but it is not specified whether the reported uses are powders.
d Eye and facial makeup preparations were reported together in the original safety assessment. The reported number was only accounted for in the eye area exposure.
e Non-coloring and coloring hair preparations, except for non-coloring shampoos, were reported together in the original safety assessment. The reported number was only accounted for in the non-coloring hair products.
MCI/MI may be used in products that can be incidentally ingested or come into contact with mucous membranes; for example, there are uses reported in lipsticks (reported in the VCRP only; concentration not reported), bath preparations (0.000019 ppm), and bath soaps and detergents (up to 15 ppm). 4 Additionally, this mixture has been reported to be used in products that may come into contact with the eyes; for example, these ingredients are reported to be used in eye makeup preparations (reported in the VCRP only; concentration not reported). Moreover, this mixture has been reported to be used in spray and powder products that could possibly be inhaled; for example, MCI and MI are reported to be used in colognes (0.075 ppm), hair sprays (7.5 ppm), and face powders (reported in the VCRP only; concentration not reported).
In the European Union, MCI/MI is listed under Annex V, the list of preservatives allowed in cosmetic products, with the restriction that the combination may be used at a maximum concentration of 0.0015% (ie, 15 ppm) in rinse-off products as a 3:1 ratio of MCI: MI. 6 The Scientific Committee on Consumer Safety (SCCS) in 2009 noted that MCI/MI is a well-recognized skin sensitizer at current conditions of use and concentration. The SCCS concluded that MCI/MI in a ratio of 3:1 does not pose a risk to the health of the consumer when used as a preservative at a maximum concentration of 0.0015% in rinse-off cosmetic products, apart from its sensitizing potential. 7 Induction and elicitation were considered less likely in a rinse-off product than when the same concentration is present in a leave-on product. In 2016, however, a proposal to amend Annex V to state that no safe concentrations for MI have been adequately demonstrated for use in leave-on cosmetic products (including “wet wipes”) was announced, which would effectively ban MCI/MI from use in leave-on products. 8
Non-Cosmetic
MCI/MI (3:1) has been determined to be safe for use in indirect food additives as adhesive, coating, and paper and paperboard components only as an antimicrobial agent or a slimicide (21CFR §175.105, §175.300, §175.320, §176.170, and §176.300).
MCI/MI is reported to be used in water-based wall paints. 9 Analysis of 60 paint samples found the concentration of MCI to range from 0.5 to 3.5 ppm while the concentration of MI ranged from 1.1 to 142.7 ppm.
Toxicokinetics
MCI/MI was absorbed after oral administration and then was excreted in the urine or feces; storage in the tissues was minimal. Up to 62% of a single percutaneous dose was bound to the site of application 24 hours after exposure. The MCI/MI bound to the skin had a 13.1-day half-life. 1
In an oral metabolism study in humans, 4 volunteers received 2 mg of labeled 3-[ 13 C]MI or 3-[3H]MCI (16.3 and 13 μmol, respectively) in 200 µL of ethanol in a glass of water, separately and at least 2 weeks apart. 10 Over a 48-hour period, consecutive and complete urine samples were collected and examined for the content of N-methylmalonamic acid (NMMA). NMMA represented 23.7% and 13.3% of the dose excreted in urine after exposure of MI and MCI, respectively, with more than 90% excreted within the first 24 hours. Excretion of NMMA was rapid with mean half-lives of 6.1 and 7.6 hours for MI and MCI, respectively.
Toxicological Studies
MCI/MI was moderately to highly toxic to rats, and highly toxic to rabbits when administered orally, and moderately toxic when applied dermally. 1 No treatment-related effects were observed in rats which received MCI/MI in oral doses up to 24.4 mg/kg/d for 2 weeks. Doses of MCI/MI up to 2.8 mg/kg/d applied dermally to rabbits, 5 d/wk for 3 weeks, produced moderate irritation at the application site but no systemic toxicity. Dermal application of MCI/MI at doses up to 0.4 mg/kg/d for 3 months produced no systemic toxicity in rabbits. No toxicologically significant treatment-related effects were observed in dietary studies of rats or dogs at doses up to 30 and 28 mg/kg/d, respectively.
Short-Term Toxicity Studies
Oral
In a 28-day repeated-dose oral study, male and female rats received MCI/MI (1.3%:0.38%) diluted in corn oil via gavage at 0, 0.26, 0.78, 2.3, and 7.0 mg/kg bw/d. 11 Water and feed consumption were monitored during the dosing period. At study end, the rats were killed, organs were weighed, and histological examinations were performed. Hematology, serum clinical chemistry, and biomarkers of inflammation were also assessed. No treatment-related effects on weight gains, organ weight, or hematological parameters were observed. A reduction of serum triglyceride levels in males and induction of hepatic phase 1 xenobiotic metabolizing enzymes in females, with subtle histological changes in the liver, were observed in the 7.0 mg/kg dose group. The authors stated that these changes were likely an adaptive, reversible response. The lowest-observed-effect-level (LOEL) was determined to be 7.0 mg/kg bw/d.
Subchronic Toxicity Studies
Inhalation
In a 13-week repeated-dose inhalation study performed in accordance with Organization for Economic Co-operation and Development (OECD) test guideline (TG) 413, groups of 16 Crl: CD(SD)BR rats per sex were exposed to 14% MCI/MI. 12 The rats were exposed whole body for 6 hours per day, 5 day per week, at aerosol concentrations of 0, 0.34, 1.15, or 2.64 mg active ingredient (ai)/m3, with an aerosol particle size of 1.1 to 1.4 µm (mean mass median diameter), which is defined as the diameter at which 50% of the particles by mass are larger and 50% are smaller). During the exposure period, the rats were observed for clinical signs of toxicity, and body weight and ophthalmologic evaluations were made. At study termination, hematology, clinical chemistry, gross pathology, and histopathologic evaluations were conducted. No statistically significant effects were observed in the hematology, gross pathology, or ophthalmologic evaluations at any concentration. At 2.64 mg/m3, rats of both sexes had signs consistent with exposure to a sensory irritant, including chromorhinorrhea, rhinorrhea, eye squint, bradypnea, and dyspnea. Decreased body weight gains, decreased male spleen weights, and decreased serum protein in females were also observed in rats exposed to 2.64 mg/m3. No treatment-related clinical signs of toxicity, body weight effects, or organ weight effects were observed in the 0.34 or 1.15 mg/m3 dose groups. Treatment-related histopathologic findings consisting of slight to moderate incidences of eosinophilic droplets in the anterior respiratory mucosa of the nasal turbinates and slight rhinitis in the lining of the anterior portion of the nasal cavity were observed in the 2.64 mg/m3 dose group. At 1.15 mg/m3, rhinitis was observed in rats of both sexes. No treatment-related histopathologic effects were observed in the 0.34 mg/m3 dose group. All histopathologic changes were minor, potentially reversible, and generally reflective of minimal tissue response to a very mild, low-grade respiratory irritant. Based on the occurrence of rhinitis, the LOEL was 1.15 mg/m3 ai. The no-observable-effect-level (NOEL) was 0.34 mg/m3 ai.
Developmental and Reproductive Toxicity Studies
MCI/MI administered by gavage to pregnant rabbits (gestation days 6 through 18) at doses up to 13.3 mg/kg/d was toxic to the dam, embryo, and fetus; the compound was not teratogenic. 1 In pregnant rats (gestation days 5 through 15) that received MCI/MI at doses up to 15 mg/kg/d, toxicity was observed in the dams, but no treatment-related effects were noted in any of the reproductive parameters of the surviving dams and fetuses and no teratogenicity was observed.
Genotoxicity
The result of genotoxicity testing of MCI/MI varied with the assay used. 1 Mutagenicity was observed in several Ames tests with and without metabolic activation, but no genotoxicity was observed in several in vitro mammalian cell assays. Results were mixed in a mouse lymphoma cell assay, with genotoxicity observed when there was no metabolic activation.
The mutagenicity of a trade name mixture containing MCI/MI (14% ai; 10% MCI: 3.4% MI) and 5 cosmetic products that contained the trade name mixture was studied in an Ames test using Salmonella typhimurium strain TA 100, with and without metabolic activation. 13 The cosmetic products were diluted in distilled deionized water and tested at up to 400 µL/plate; MCI/MI was tested at doses ranging from 0.00039 to 0.05 µL/plate. Three of the 5 products were direct acting mutagens, while the other 2 were too cytotoxic to determine mutagenicity. Metabolic activation reduced cytotoxicity but did not eliminate mutagenicity. Mutagenicity was also observed with MCI/MI, with and without metabolic activation, in a dose-dependent manner.
Carcinogenicity
Dermal application of 400 ppm of 2.67% MCI/MI in distilled water, 3 times per week for 30 months, had no local or systemic tumorigenic effect in male mice. 1
Dermal Irritation and Sensitization
The dermal irritation of MCI/MI was concentration dependent in rabbits under occlusive patches, with 560 ppm being nonirritating, 2800 ppm being moderately irritating, and 5600 ppm being severely irritating. 1 In humans, MCI/MI was irritating in a dose-dependent manner, with 100 ppm essentially nonirritating, 200 ppm slightly irritating, and 400 to 800 ppm strongly irritating. MCI/MI is a sensitizer: The concentration of MCI/MI in cosmetic products which produced sensitization varies. The available human sensitization test data at concentrations of 50 ppm and above gave mixed results. MCI/MI was not a sensitizer at a concentration of 15 ppm.
Human
In a human repeated insult patch test (HRIPT) of a hand wash containing 12 ppm MCI/MI in 50 volunteers, applications of 0.2 mL were made directly on the back as open patches on an area of approximately 2 cm2 for a dose of 1.2 µg/cm2. 14 No adverse effects were observed during the study and no irritation or sensitization was observed during induction or challenge.
Phototoxicity
MCI/MI was not a photosensitizer at a concentration of 15 ppm in human volunteers. 1
Ocular Irritation
MCI/MI in an aqueous solution was not a cumulative ocular irritant when tested at 55 ppm in rabbits; it was corrosive when tested at 1.1% (11000 ppm and higher). 1
Clinical Studies
Provocative Studies
A repeated open application test (ROAT) was performed on 15 patients with known contact allergy to 100 ppm MCI/MI and/or MI (6 patients reacted to MCI/MI only, 6 patients reacted to MI only, and 3 patients reacted to both MCI/MI and MI). 15 Each patient was given 2 sets of aqueous skin creams. One cream contained MI at 100 ppm, while the other contained paraben preservatives. The patients applied the creams twice daily for 2 weeks to the outer aspect of the upper arm on an area of 25 cm2. The sites were evaluated by dermatologists prior to the ROAT commencement; after 1 and 2 weeks, 8 patients had positive allergic responses at the test sites who received the MI-containing creams. Of the patients with the known MI allergy, 5 had positive responses. Of the patients with the known MCI/MI allergy, 6 had positive responses.
Baseline series patch tests, photopatch tests, and/or phototests were performed on a total of 10 patients with suspected photoaggravated contact dermatitis to MCI/MI or MI. 16 All 10 patients underwent the baseline patch tests: The test concentrations for MCI/MI were 0.01% or 0.02% aq., and for MI was 0.2% aq. Six patients were photopatch tested with cosmetics containing MCI/MI and/or MI (amount of test substance not reported), with one of the 2 identical patches being irradiated with 5 J/cm2 long-wavelength ultraviolet (UVA) light. Phototests were performed on 2 patients with UVA/mid-wave length ultraviolet radiation ranging between 290 and 400 nm. Seven patients had positive patch tests to both MCI/MI and MI, and 3 patients had positive patch tests to only MI. Four patients had transient photosensitivity. Photopatch tests with MCI/MI and/or MI gave stronger reactions than baseline patch tests with these ingredients, indicating photoaggravation.
Baseline and Retrospective Studies
Dermal
Numerous baseline and retrospective studies which included testing with MCI/MI have been published since the original report was issued; a sampling of these studies is presented in Table 2. The results of these studies demonstrate that sensitization to MCI/MI is found worldwide, with reported rates as low as 0.7% (out of 703 patients; United States) to as high as 15.4% (out of 635 patients; Thailand).17-50
Baseline and Retrospective Studies.
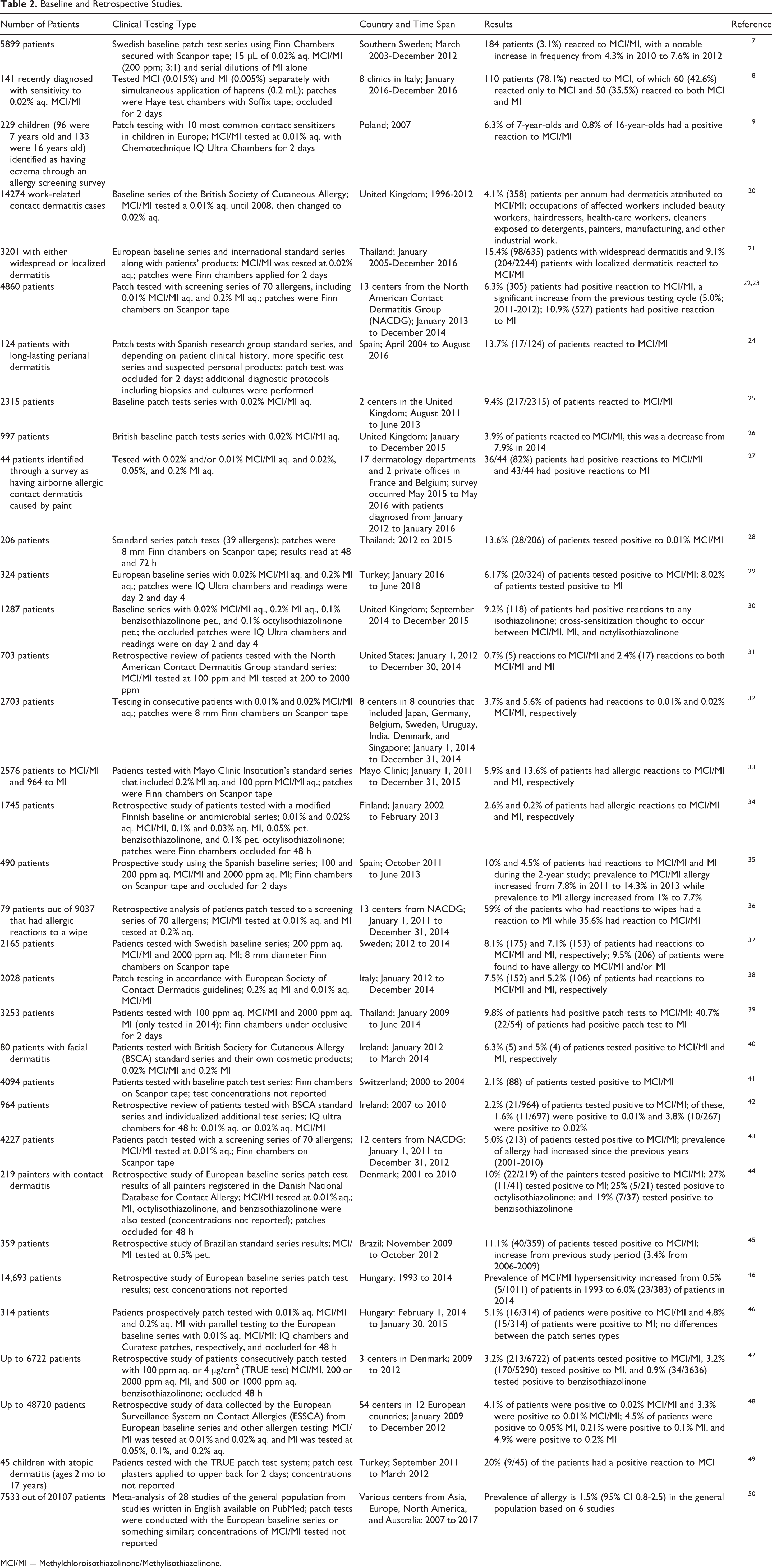
MCI/MI = Methylchloroisothiazolinone/Methylisothiazolinone.
Case Reports
Cases studies include reports of MCI/MI sensitization from a wide range of materials, including personal care products (including wet wipes), ultrasound gels, paints, glues, cleaners, and industrial biocides.51-68 Dermal sensitization from paint was hypothesized to be from airborne exposure in several patients.56,58,63,64
Airway Dysfunction
Peripheral airway dysfunction was observed in a retrospective assessment of 24 children in South Korea, with no underlying disease, who were exposed to MCI/MI as a humidifier disinfectant (HD). 69 The children were exposed to MCI/MI at high density for up to 6 months and were exposed initially as infants. Pulmonary function was assessed with impulse oscillometry. One child died at age 4 month after continuous use of the HD over 3 months.
In a related study of 530 registered patients with lung disease who were exposed to HDs in South Korea, 3 definite or probable cases of airway dysfunction were reported from use of an HD that contained MCI/MI (127 mg/L MCI:37 mg/L MI). 70 Two of these cases were in infants, and 1 was in an adult patient who died. Another 33 cases of airway dysfunction were possibly or unlikely/intermittently associated with the HD containing MCI/MI; 5 deaths were reported in these cases.
Case studies from South Korea related to exposure of an HD containing MCI/MI, resulting in lung injury, included a set of twin girls who were exposed from ages 4 to 6 months, and another girl who was exposed from age 11 to 25 months.71,72 The twin girls presented with cough, sputum, and respiratory difficulty, and were observed with pneumomediastinum on chest X-ray. 71 In the latter case, the patient presented with coughing, fever, dyspnea, and tachypnea that progressively worsened and she developed acute respiratory distress syndrome: The patient died during hospitalization. 72
Risk Assessment
A skin sensitization induction risk assessment of MCI/MI was performed with various personal care products. 73 An estimated daily consumer exposure level (CEL) for rinse-off and leave-on products was calculated using the amount of product applied per application, number of applications per day, a retention factor, the MCI/MI concentration, and body surface area values. The researchers assumed that the products contained the maximum recommended safe concentration of 15 ppm MCI/MI in rinse-off products and 7.5 ppm MCI/MI in leave-on products. The estimated CELs were compared with the no-expected-sensitization-induction-level (NESIL) for MCI/MI of 0.83µg/cm2, which was based on the value reported by the SCCS from weight-of-evidence (WoE) data from an HRIPT. The sensitization assessment factors (SAFs) were applied to calculate product-specific margins of safety (MOSs). The researchers found that the MOSs for rinse-off products ranged from 5 to 63, whereas the MOSs for leave-on products ranged from 0.03 to 1.49. An MOS of 1 or greater indicates a low likelihood of sensitization induction. The researchers concluded that the results provide evidence that some leave-on products containing the maximum recommended safe concentration of MCI/MI may increase the risk of sensitization induction due to exposure to MCI/MI, while rinse-off products were not associated with a potential increased risk of skin sensitization induction.
In another skin sensitization risk assessment of MCI/MI, the maximum safe concentration of 15 ppm MCI/MI in representative-type cosmetics (which included shampoos, conditioners, soap, lotions, hand and face cream, deodorants, wipes, and eye and face makeup) indicated the possibility of skin sensitization when an NESIL of 1.25 µg/cm2 was used in the determination. 74 However, there was no potential for skin sensitization at this concentration for just rinse-off products. In this assessment, the MOS was calculated as the acceptable exposure level (AEL)/CEL and was considered safe when the AEL/CEL ratio was 1 or more. The AEL is calculated as the NESIL/skin SAF. For the representative type cosmetics, the SAF was 300; while in rinse-off products, it was 100. The MOS for representative type cosmetics was determined to be 0.00538 and the MOS for rinse-off products was 2.14.
In a QRA performed by the CIR Science and Support Committee (SSC), a conservative NESIL of 0.83 µg/cm2 was derived for MCI/MI based on a WoE evaluation of HRIPT data and data from local lymph node assays (LLNA). 75 The NESIL was then used to calculate AELs for the potential for the induction of sensitization from dermal exposure to MCI/MI in cosmetic products, assuming the maximal use concentration of 15 ppm for rinse-off products and 7.5 ppm for leave-on products and VCRP product category-specific QRA SAFs. The SAFs include 6 component factors (interindividual, site, skin condition, matrix, occlusion, frequency and duration of exposure). Individual CELs were then calculated for numerous VCRP product categories, ranging from baby shampoo (CEL = 0.0024 µg/cm2) to skin cleansing products (CEL = 0.0135 µg/cm2). The lowest CEL to MCI/MI was 3.8 × 10−9 µg/cm2 for bubble baths, and the highest estimated exposure was 0.0315 µg/cm2 for permanent waves. By using the maximum reported MCI/MI concentration of use levels provided by the Council survey (Table 3), an adequate MOS for skin sensitization is provided for all reported uses except for permanent waves (using 7.5 ppm MCI/MI) and for skin cleansing products (ie, cold creams, cleansing lotions, liquids, and pads; using 15 ppm MCI/MI). The maximum supportable concentration of MCI/MI for permanent waves and skin cleansing products are 2 and 9 ppm, respectively. When using the exposure assumptions in this risk assessment on all reported VCRP product categories of use with the maximum recommended concentrations of use, as set by the original CIR conclusion, of 7.5 ppm in leave-on products and 15 ppm in rinse-off products (Table 4), an adequate MOS could not be assured for baby shampoo (MOS = 0.92), permanent wave (MOS = 0.13), hair tints (MOS = 0.56), skin cleansing products (0.61), or cologne and toilet waters (0.50).
Quantitative Risk Assessment of MCI/MI at the Highest Reported Maximum Use Concentrations in Cosmetic Products. 75
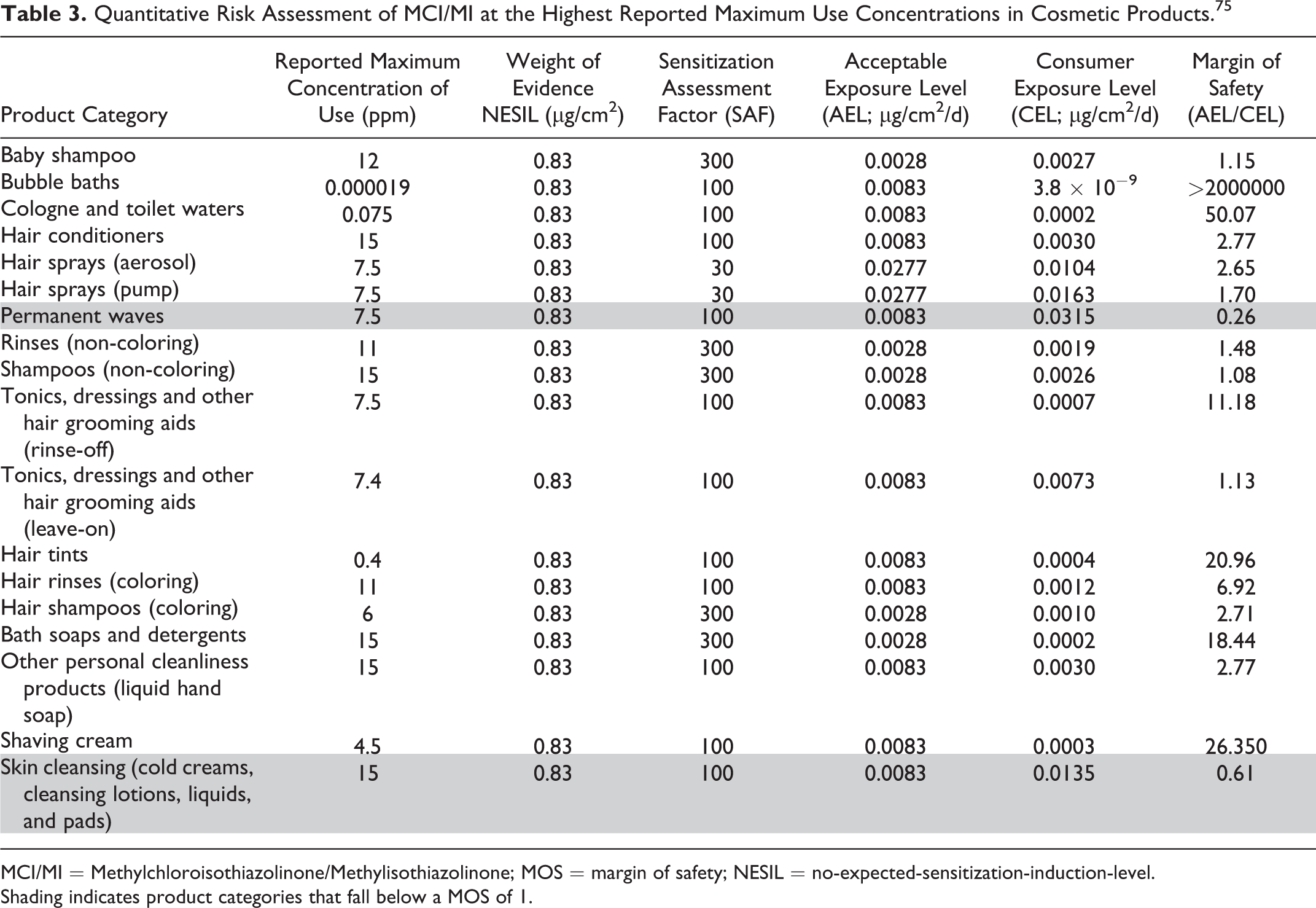
MCI/MI = Methylchloroisothiazolinone/Methylisothiazolinone; MOS = margin of safety; NESIL = no-expected-sensitization-induction-level.
Shading indicates product categories that fall below a MOS of 1.
Quantitative Risk Assessment of MCI/MI at the Maximum Recommended Use Concentrations in Cosmetic Products. 75
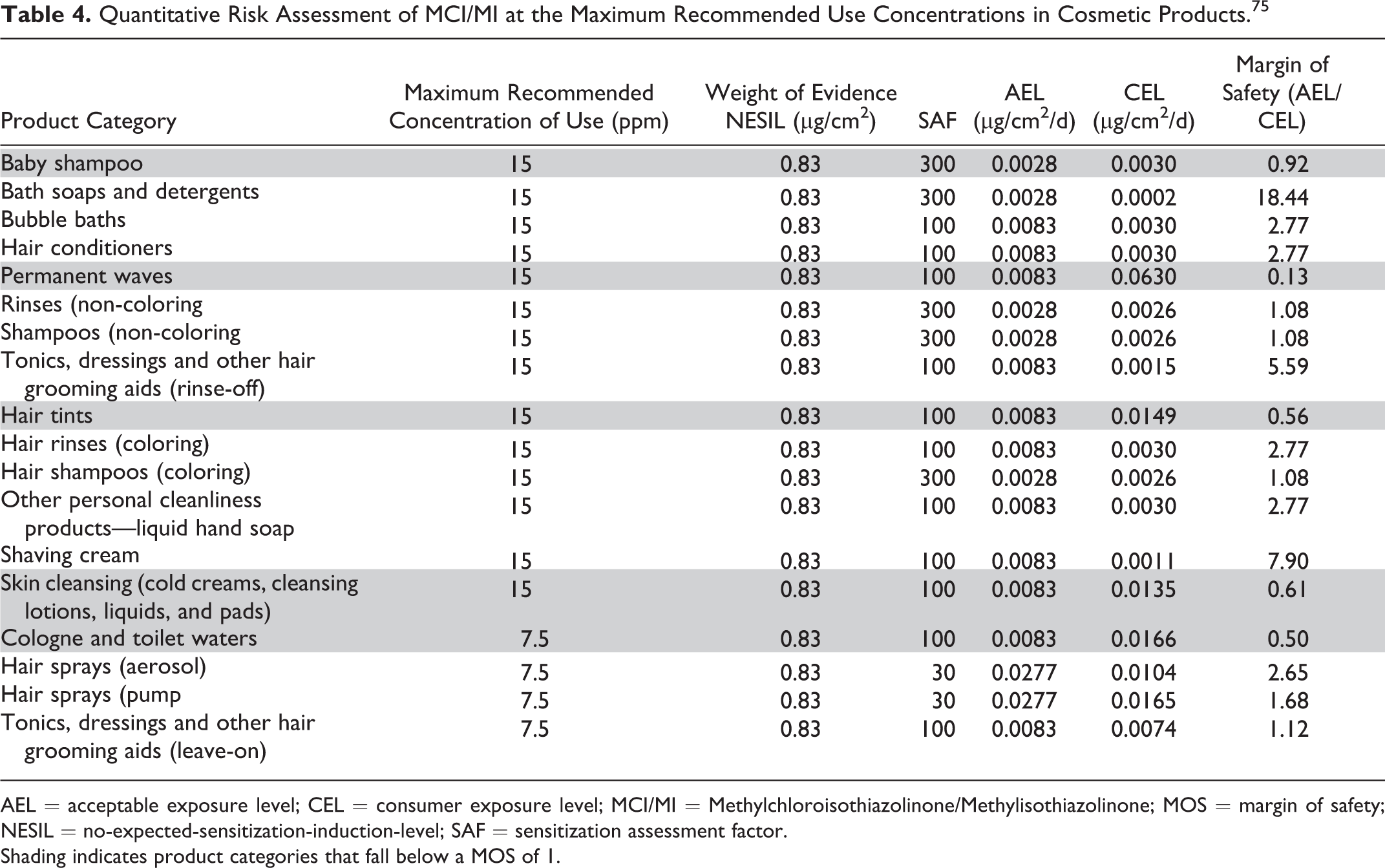
AEL = acceptable exposure level; CEL = consumer exposure level; MCI/MI = Methylchloroisothiazolinone/Methylisothiazolinone; MOS = margin of safety; NESIL = no-expected-sensitization-induction-level; SAF = sensitization assessment factor.
Shading indicates product categories that fall below a MOS of 1.
Summary
This safety assessment is for the combination of MCI and MI as used in cosmetics. Each ingredient is reported to function as a preservative in cosmetic products. In 1992, the original report on MCI/MI was published with the Panel’s conclusion that this mixture may be “safely used in rinse-off products at a concentration not to exceed 15 ppm and in leave-on cosmetic products at a concentration not to exceed 7.5 ppm.” The stated safe-for-use concentration refers to a mixture containing 76.7% MCI and 23.3% MI.
MCI and MI are surveyed separately in the VCRP and not as a mixture. According to 2019 VCRP survey data, the total number of uses reported for MCI is 5137; 480 of these are in leave-on products. MI has 6037 reported uses; 1042 of these are in leave-on products. The number of uses has increased significantly since the original report on MCI/MI was published; in 1986, the total number of uses for the ingredient mixture was 381. In 2019, the Council reported that MCI/MI (3:1) is used at up to 7.5 ppm in leave-on products and at up to 15 ppm in rinse-off products. In the original report, concentration of use was reported as a range; the concentration of use range for MCI/MI in both leave-on and rinse-off products was reported to be < 0.1 to 1%.
In the European Union, MCI/MI is listed as a preservative in Annex V; it is limited to a maximum concentration of 0.0015% (ie, 15 ppm) in rinse-off products as a 3:1 ratio of MCI: MI. The SCCS concluded in 2009 that MCI/MI in a ratio of 3:1 does not pose a risk to the health of the consumer when used as a preservative at a maximum concentration of 0.0015% in rinse-off cosmetic products, apart from its sensitizing potential. In 2016, however, a proposal to amend Annex V to state that no safe concentrations for MI have been adequately demonstrated for use in leave-on cosmetic products (including wet wipes) was announced, which would effectively ban MCI/MI from use in leave-on products. At that time, Annex V was amended to restrict the use of MI in rinse-offs to no more than 100 ppm, although the amendment to ban use in leave-ons was deferred.
MI and MCI were determined to metabolize into NMMA in humans after oral ingestion. Excretion of the metabolite through urine was rapid.
The LOEL for MCI/MI in a 28-day repeated-dose oral study in rats was 7.0 mg/kg bw/d, the highest dose that was tested. At this dose, a reduction of serum triglyceride levels was observed in males and induction of hepatic phase 1 xenobiotic metabolizing enzymes with subtle histological changes in the liver were observed in females. In a 13-week inhalation study of 14% MCI/MI in rats that followed OECD TG 413, MCI/MI was tested at up to 2.64 mg ai/m3. Based on the occurrence of rhinitis, the LOEL was 1.15 mg/m3. The NOEL was 0.34 mg/m3.
A trade name mixture containing MCI/MI (14% ai) and cosmetic products containing this mixture were mutagenic in an Ames test, with and without metabolic activation.
An HRIPT of a hand wash containing 12 ppm MCI/MI was not irritating or sensitizing in 50 volunteers. Provocative baseline patch tests and phototoxicity tests produced positive results in patients with suspected MCI/MI allergy. Numerous baseline and retrospective studies that included MCI/MI indicate that sensitization to this preservative occurs worldwide. Numerous case studies demonstrate sensitization to MCI/MI resulting from exposure to a wide range of materials, including personal care products, paints, glues, and cleaners. Peripheral airway dysfunction has been observed in patients in South Korea that were exposed to MCI/MI as an HD.
Skin sensitization induction risk assessments of MCI/MI in multiple personal care and cosmetic products using an NESIL of 0.83 µg/cm2 found that some leave-on products (eg, colognes and toilet waters) with MCI/MI at the recommended safe concentration of 7.5 ppm may increase the risk of sensitization induction. In most rinse-off products, 15 ppm MCI/MI was not associated with a potential increase risk of skin sensitization induction.
Discussion
This safety assessment is for the combination of MCI and MI as used in cosmetics. Based on the numerous sensitization studies and reports that became available since the original report was issued, this safety assessment was reopened to reassess the conclusion published in 1992.
The Panel noted the results of a QRA for skin sensitization performed by the CIR SSC. The results indicated that some leave-on products comprising MCI/MI at the recommended maximum safe concentration of 7.5 ppm may yet increase the risk of inducing dermal sensitization. In most rinse-off products, 15 ppm MCI/MI was not associated with a potential increased risk of skin sensitization induction. Individuals previously sensitized to MCI/MI should avoid products that contain this ingredient mixture.
MCI/MI is a useful and necessary preservative system in cosmetic products. The Panel is aware that the conclusion herein differs from that reached by counterparts in the European Union. In part, the differing conclusions are based on interpretation of earlier LLNA data on which the hazard assessments were determined. However, the Panel supports managing sensitization risks by the use of valid assessment tools and strategies, such as a QRA system (or similar methodology). Instead of banning ingredients that may pose a risk under certain conditions (eg, formulation, body part exposure), the Panel has proposed that such risk-mitigating tools and strategies can be applied by formulators and thus avoid exhausting available preservative systems. Such systems are necessary to protect consumers from microbial contaminations that would otherwise occur in cosmetic products.
In response to concerns of reports of adverse events observed in infants following inhalation exposure to HDs that contained the MCI/MI preservative mixture, the Panel requested, and received, an inhalation study of at least 3 months in duration that is in accordance with the OECD TG 413. The Panel determined that the data mitigated concern for the use of this ingredient mixture at the reported concentrations in cosmetic products that could be incidentally inhaled following use. The concentrations used in the HD were orders of magnitude greater than those found in cosmetics.
Conclusion
The Panel concluded that the ingredient mixture MCI/MI is safe in cosmetics when formulated to be nonsensitizing, based on the results of a QRA or similar methodology; however, at no point should concentrations exceed 7.5 ppm in leave-on products or 15 ppm in rinse-off products.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Burnett, C. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript, and critically revised the manuscript, Bergfeld, W., Belsito, D., Peterson, L., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., and Snyder, P. contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.