Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reassessed the safety of Methylisothiazolinone, which functions as a preservative in cosmetics. The Panel reviewed relevant animal and human data provided in this safety assessment, and data from the previously published safety assessments of Methylisothiazolinone, and concluded that Methylisothiazolinone is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm (ie, 0.01%) and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a quantitative risk assessment or similar methodology.
Introduction
Methylisothiazolinone is reported to function in cosmetics as a preservative, according to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary). 1 In 2019, the Expert Panel for Cosmetic Ingredient Safety (Panel) published an amended safety assessment of Methylisothiazolinone with the conclusion that “Methylisothiazolinone is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a quantitative risk assessment (QRA).” 2 This conclusion superseded the findings of the Panel’s earlier safety assessment that was published in 2010. 3 At the September 2019 Panel meeting, during the re-evaluation of the mixture Methylchloroisothiazolinone/Methylisothiazolinone (MCI/MI), the Panel reopened the amended safety assessment of Methylisothiazolinone to consider additional newly available data, with particular regard to inhalation toxicity.
In 2019, the Panel issued an amended safety assessment of the mixture MCI/MI (supplied as a ratio of 3:1), with the conclusion that the mixture “is safe in cosmetics when formulated to be nonsensitizing, based on the results of a QRA or similar methodology; however, at no point should concentrations exceed 7.5 ppm in leave-on products or 15 ppm in rinse-off products.” 4 In response to concerns of reports of adverse events observed in infants following inhalation exposure to humidifier disinfectants that contained the preservative mixture MCI/MI, the Panel moved to reopen the safety assessment of Methylisothiazolinone later that same year.
Data from the original Methylisothiazolinone safety assessment that was published in 2010 and the amended safety assessment that was published in 2019 are also summarized in appropriate sections of this report.2,3
This safety assessment includes relevant published and unpublished data that are available for each end point that is evaluated. Published data are identified by conducting an exhaustive search of the world’s literature. A listing of the search engines and websites that are used and the sources that are typically explored, as well as the end points that the Panel typically evaluates, is provided on the Cosmetic Ingredient Review (CIR) website (https://www.cir-safety.org/supplementaldoc/preliminary-search-engines-and-websites; https://www.cir-safety.org/supplementaldoc/cir-report-format-outline). Unpublished data are provided by the cosmetics industry, as well as by other interested parties.
Much of the data included in this safety assessment was obtained from the European Chemicals Agency (ECHA). 5 These data summaries are available on the ECHA website, and when deemed appropriate, information from the summaries has been included in this report.
Chemistry
Definition and Structure
Methylisothiazolinone (CAS No. 2682-20-4) is the heterocyclic organic compound that conforms to the structure depicted in Figure 1. 1
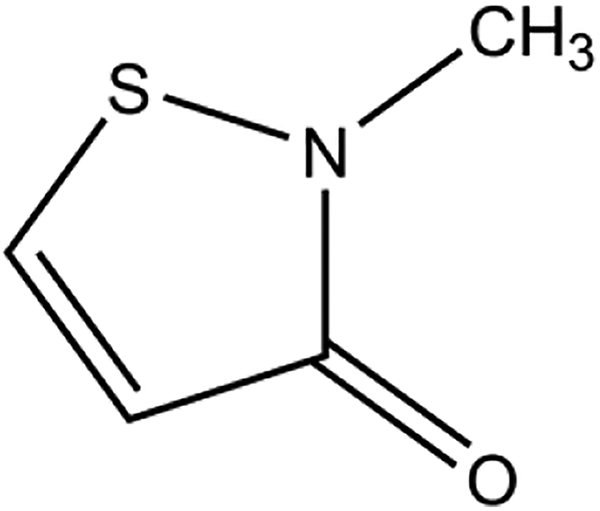
Methylisothiazolinone.
Physical and Chemical Properties
Methylisothiazolinone has a molecular weight of 115.2 Da and a density of 1.02 g/mL at 25 °C. 3 The ultraviolet/visible spectrum for a trade name Methylisothiazolinone product had peak wavelengths at 274 nm for a neutral solution, 266 nm for an acidic solution, and 274 nm for a basic solution. Additional properties are described in the original safety assessment.
Method of Manufacturing
Methylisothiazolinone is produced by the controlled chlorination of dimethyl dithiodipropionamide in solvent. 3 Methylisothiazolinone is then neutralized and extracted into water followed by a solvent strip.
Composition and Impurities
The composition of technical grade Methylisothiazolinone was 96.8% Methylisothiazolinone, 0.1% 5-chloro-2-methyl-4-isothiazoline-3-one, 0.1% 4,5-dichloro-2-methyl-4-isothiazolinone-3-one, 0.2% N, N′-dimethyl-3,3′-dithiodipropionamide, 0.5% N, N′-dimethyl-3,3′-trithiodipropionamide, 0.1% N-methyl-3-chloropropionamide, 0.3% ammonium chloride, 0.2% water, 0.1% ethyl acetate, 0.1% acetic acid, and 1.5% unknown compounds. 3 Impurities of a trade name Methylisothiazolinone product (9.5% active ingredient [ai]) included 79 to 103 ppm N, N′-dimethyl-3,3′-trithiodipropionamide, 44 to 79 ppm 5-chloro-2-methyl-4-isothiazolin-3-one, and 490 ppm N, N′-dimethyl-3,3′-dithiodipropionamide.
Use
Cosmetic
The safety of the cosmetic ingredient addressed in this assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetics industry on the expected use of this ingredient in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in the FDA Voluntary Cosmetic Registration Program (VCRP) database. Data are submitted by the cosmetic industry in response to a survey, conducted by the Personal Care Products Council (Council), of maximum reported use concentrations by product category.
According to 2019 VCRP survey data, Methylisothiazolinone (when not used with MCI) is used in a total of 915 formulations; the majority of the uses are in bath soaps and detergents (Table 1). 6 These uses have increased since the last review where 745 uses were reported; the majority of the uses reported then were in noncoloring hair conditioners and shampoos. 2 The maximum concentration of use range for Methylisothiazolinone in 2020 was reported to be 0.000002% to 0.00975% (0.02-97.5 ppm), with 0.00975% reported in hair conditioners and 0.009% used in leave-on hair products. 7 In the amended safety assessment published in 2019, the maximum concentration of use range was reported to be 3.5 × 10−8% to 0.01% (0.00035-100 ppm), with 0.01% reported in multiple product categories, including eye makeup remover, hair shampoos and conditioners, and skin care products (both leave-on and rinse-off).
Frequency and Concentration of Use According to Duration and Type of Exposure for Methylisothiazolinone.
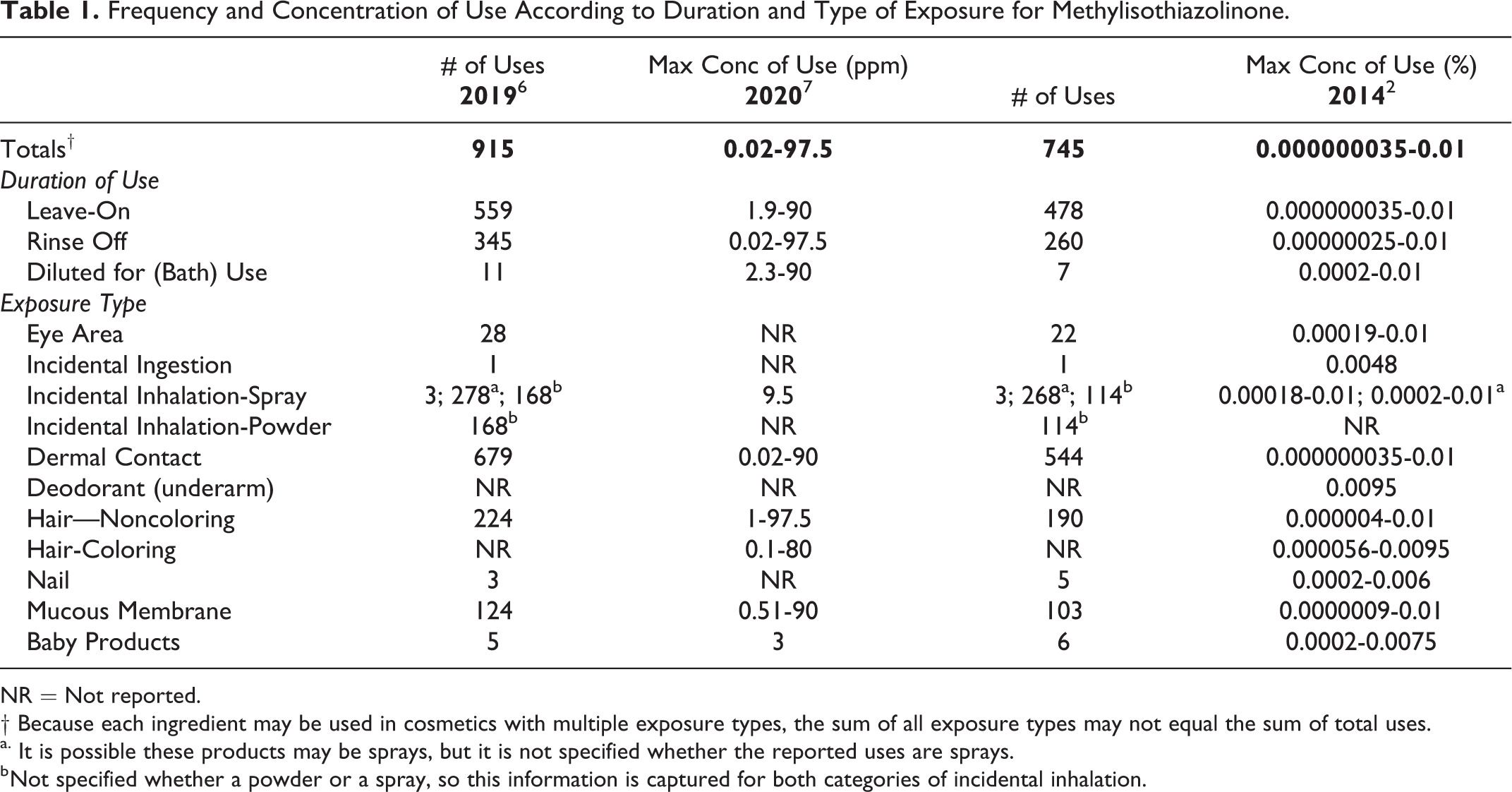
NR = Not reported.
† Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
a. It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
b Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
Methylisothiazolinone may be used in products that can come into contact with the eyes or mucous membranes; for example, it is reported to be used in bath soaps and detergents at up to 0.00755% (75.5 ppm) and in bath oils, tables and salts at up to 0.0090% (90 ppm). 7 Additionally, Methylisothiazolinone is used in cosmetic sprays and could possibly be inhaled; for example, it is reported to be used in hair sprays at up to 0.00095% (9.5 ppm). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters > 10 µm, with propellant sprays yielding a greater fraction of droplets/particles < 10 µm compared with pump sprays.8,9 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and thoracic regions of the respiratory tract and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.10,11
Under regulations governing the use of cosmetic ingredients in the European Union, Methylisothiazolinone is listed under Annex V, the list of preservatives allowed in cosmetic products, with the restriction that it may only be used in rinse-of products at up to 0.0015% (15 ppm). 12 The most recent opinion on Methylisothiazolinone by the European Union’s Scientific Committee on Consumer Safety (SCCS) has found that in leave-on cosmetic products (including “wet wipes”), no safe concentration has been adequately demonstrated for induction or elicitation of contact allergy. 13 In rinse-off cosmetic products, the SCCS has concluded that concentrations up to 0.0015% (15 ppm) Methylisothiazolinone are safe, in terms of induction of contact allergy, but recognized that there is no information available to evaluate the potential for this ingredient to elicit contact allergy. Furthermore, the SCCS states that Methylisothiazolinone should not be added to cosmetic products that already contains MCI/MI.
Non-Cosmetic
The uses of Methylisothiazolinone in paints and other non-cosmetic products were described in the original safety assessment.2,3
There is the potential for residential and occupational exposure when Methylisothiazolinone is used to preserve materials such as paints, cleaners, and plastics. In April 2020, the US Environmental Protection Agency (EPA) released a draft risk assessment for MCI/MI. 14 Included were data and analyses of residential and occupational handler risks to inhalation of spray products containing Methylisothiazolinone and Methylisothiazolinone-preserved paints. Inhalation risks to these 2 groups were assessed using the Methylisothiazolinone maximum application rate of 400 ppm by weight. The human equivalent concentrations for MCI/MI, derived from a no-observed-adverse-effect-concentration (NOAEC) of 0.34 mg/m3 (inhalation) in rats, are calculated to be 0.11 and 0.038 mg/m3, based upon an 8-hour and 24-hour time weighted average exposure period, respectively. The inhalation margins of exposure (MOEs) for residential Methylisothiazolinone aerosol and vapor exposures range from 1.0 to 14000, and the inhalation MOEs for occupational Methylisothiazolinone aerosol and vapor exposures range from 0.5 to 5800. Toxicological concern was noted when these values were less than the level of concern (LOC) of 10. Scenarios for residential handlers applying paint and occupational inhalation of paint vapors assuming long exposure durations had MOEs that had LOC below 10. Analyses of paint exposure are not relevant to the assessment of cosmetic safety due to the exposure durations and concentrations of application being magnitudes greater than those of cosmetic use.
The EPA also assessed incidental oral and dermal postapplication exposure for Methylisothiazolinone in textile and household cleaning products. 14 The induction point of departure (POD) for Methylisothiazolinone is based on the dermal sensitization induction threshold of 210 μg/cm2, while the elicitation POD is 0.0105 μg/cm2. In textile and household cleaning products, the chronic total dietary exposures do not show any risks; however, the dermal MOEs for elicitation are all of concern. As mentioned above, these analyses of exposures to textile and household cleaning products are not considered relevant to the assessment of cosmetic safety.
Toxicokinetic Studies
Absorption, Distribution, Metabolism, and Excretion
The percutaneous absorption of [14C]Methylisothiazolinone (99.88% radiochemical purity) was determined using rat skin mounted on diffusion cells. 3 Over a 24-hour period, the rate of absorption was 0.0059, 0.0277, and 0.0841 µg equivalents/cm2/h for 25, 75, and 150 ppm groups, respectively, and the mean amount of total applied radioactivity absorbed was 21.4%, 33.7%, and 51.2% for 25, 75, and 150 ppm dose groups, respectively. The total dose absorbed of aqueous solutions containing radiolabeled Methylisothiazolinone (96.90% radiochemical purity) in human epidermis was 29.8%, 38.0%, and 54.7% for 52.2, 104.3, and 313 µg Methylisothiazolinone/mL dose groups. The rate of absorption was 0.037 μg/cm2/h over a 24-hour exposure. In the same study, the total dose absorbed from shampoo, body lotion, and facial cream formulations containing 100 µg Methylisothiazolinone/mL was 29.5%, 8.98%, and 19.6%, respectively. The rates for absorption of Methylisothiazolinone in the formulations over a 24-hour exposure ranged from 0.007 to 0.026 μg/cm2/h. After oral dosing of 100 mg/kg radiolabeled Methylisothiazolinone (96.70% radio purity) in mice, total radioactive residues (TRRs) were highest in the liver and lowest in the bone 1 hour postdosing. At 24 hours postdosing, TRR declined significantly in all tissues and the tissue-to-plasma ratio showed that the radiolabel partitioned preferentially from plasma to tissues. Blood had the highest tissue-to-plasma ratio at 48 hours. TRR was higher in male tissues than female tissues overall. Most radiolabeled metabolites of Methylisothiazolinone (99.08% radio purity) were excreted in urine and feces by rats within 24 hours of oral dosing. Tissue sampling at 96 hours postdosing found 1.9% to 3.6% of the radiolabel, mainly in blood. Total mean recovery of the radiolabel was 92% to 96%. Major metabolites in urine were N-methyl malonamic acid (NMMA), 3-mercapturic acid conjugate of 3 thiomethyl-N-methyl-propionamide, and N-methyl-3-hydroxyl-propamide. Another metabolism study of radiolabeled Methylisothiazolinone (96.90% radio purity) conducted on bile duct-cannulated rats had an 88% recovery of the dose at 24 hours postoral dosing. The majority of the radiolabel was found in bile, urine, and feces. No intact Methylisothiazolinone was recovered, and the main metabolites were NMMA and 3-mercapturic acid conjugate of 3-thiomethyl-N-methyl-propionamide.
Toxicological Studies
Acute Toxicity
Methylisothiazolinone at 97.5% was slightly toxic in rats in an acute dermal toxicity study. 3 The substance was corrosive to the skin. The LD50 was calculated to be 242 mg/kg body weight (bw). In another acute dermal toxicity study, 9.69% Methylisothiazolinone was corrosive to rat skin, but no deaths occurred during the study. The LD50 was greater than 484.5 mg/kg bw.
In acute oral toxicity studies, Methylisothiazolinone was slightly toxic in rats in concentrations ranging from 9.69% to 99.7%. 3 At 9.69%, the LD50s for male and female rats were 274.6 and 105.7 mg/kg bw, respectively. Rats that died during these studies had reddened intestines and/or stomach mucosa, clear or red/yellow fluid in the intestines and/or stomach; blackened intestines and distended stomachs. Studies in rats on body lotion, shampoo, and sunscreen formulations containing 100 ppm Methylisothiazolinone found no treatment-related effects and an LD50 greater than 2000 mg formulation/kg bw. Slight toxicity, including gastrointestinal changes, was observed in mice that orally received 97.5% Methylisothiazolinone. The LD50 was 167 mg/kg bw. An acute oral toxicity study of the metabolite NMMA in rats found the substance slightly toxic. The calculated oral LD50s for NMMA in males and females were 3550 and 4100 mg/kg bw, respectively.
Acute inhalation toxicity studies in rats found that 53.52% and 97.8% Methylisothiazolinone were slightly toxic after 4 hours of exposures. 3 The LC50s were 0.35 and 0.11 mg/L, respectively. Rats that died during these studies had reddened lungs and distended gastrointestinal tracts. Mice exposed to 10 minutes of atomized 98.6% Methylisothiazolinone had up to 47% decrease in respiratory rates that equated to moderate responses for sensory irritation. 3
Acute toxicity studies are summarized in Table 2. In a dermal study in rats, the LD50 for 49.0% Methylisothiazolinone was greater than 2000 mg/kg bw. 5 In oral studies, the LD50 for a 1% solution of Methylisothiazolinone in rats was 148.0 mg/kg, while the LD50 for a 50% solution of Methylisothiazolinone in rats was 232 to 249 mg/kg in males and 120 mg/kg in females. The LC50 of aerosolized 49.8% Methylisothiazolinone in rats was 0.422 mg/L in males and 0.354 mg/L in females.
Acute Toxicity of Methylisothiazolinone.
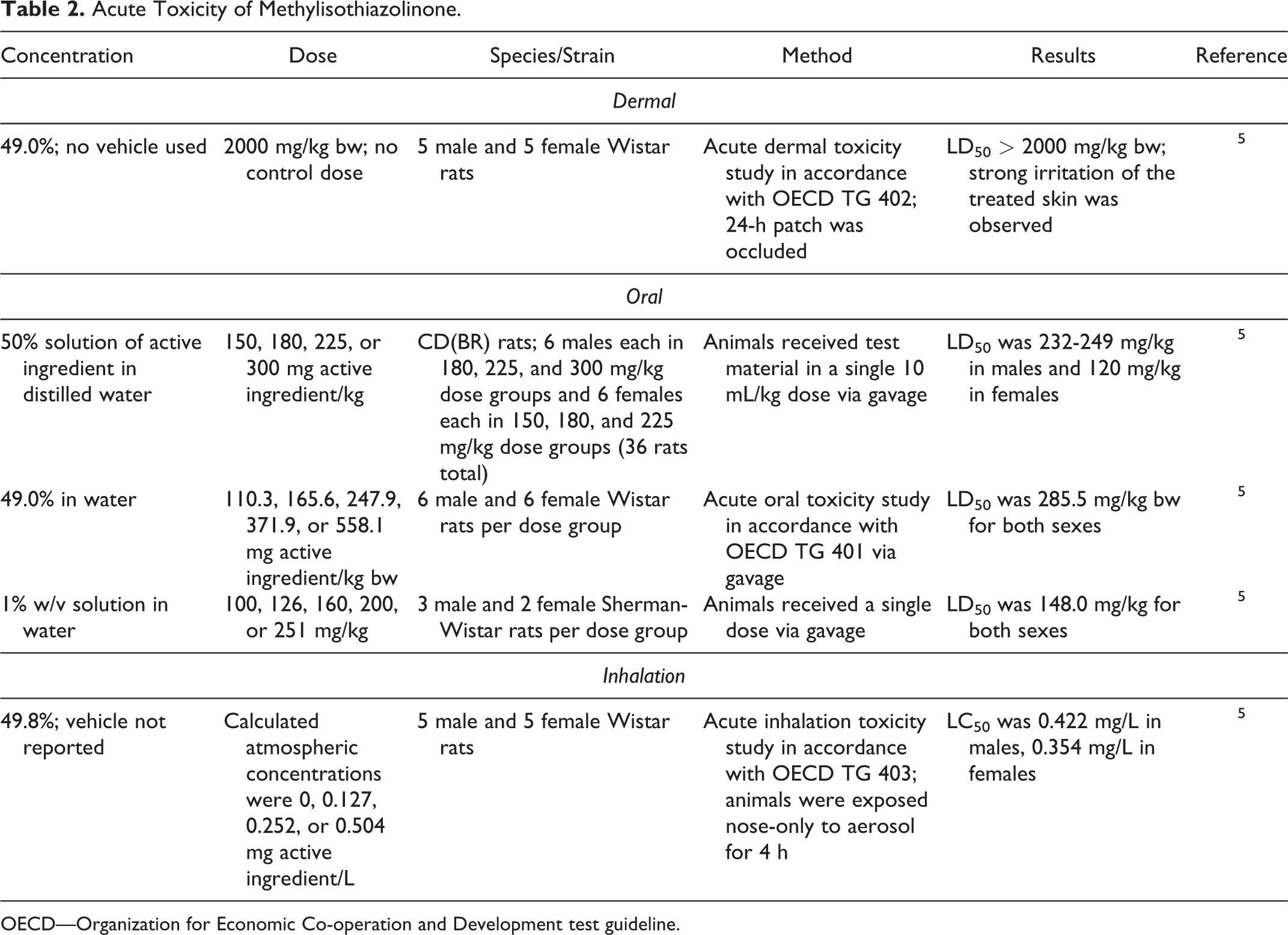
OECD—Organization for Economic Co-operation and Development test guideline.
Short-Term Toxicity Studies
Oral
In a 28-day oral toxicity study performed in accordance with Organization for Economic Co-operation and Development (OECD) test guideline (TG) 407, groups of 5 male and 5 female Wistar rats received 0, 10.03, 28.59, or 71.21 mg/kg bw Methylisothiazolinone in water daily via gavage. 5 The study included high-dose and control recovery groups that were observed for an additional 14 days following completion of the dosing period. Terminal studies included measuring organ weight and relative organ weight, and performing gross pathological and histopathological assessments. The number of mortalities was not reported. In males, the absolute and relative weights of the prostate in the low- and high-dose group, and the heart in the mid-dose group were significantly reduced when compared to the control group. However, no lesions were found in the prostate. Absolute weight of the testes and epididymides was significantly less (P < 0.05) in the high-dose recovery group when compared to the control recovery group; however, the relative weight of these organs was comparable to the control recovery group. Relative weight of the liver in the mid- and high-dose groups was significantly increased as compared to the control group; however, there was no significant variation in the high-dose recovery group and no treatment-related lesions were observed in the liver. In females, the absolute weights of the organs in the treated animals were comparable to the controls, but there were statistically significant increases in relative weight of the kidneys in the low- and mid-dose groups. These observations were considered incidental as the high-dose group and high-dose recovery group were comparable to the control groups. While pathological and histopathological changes were observed, the study summary did not detail the differences between the control and dose groups. The no-observed-adverse-effect level (NOAEL) was 28.6 mg/kg bw/d in males and females based on the combined assessment of clinical signs, mortalities, and pathological and histopathological findings; the lowest-observed-adverse-effect level (LOAEL) was 71.2 mg/kg bw/d in males and females based on lethargy and mortality. No further details were provided.
Subchronic Toxicity Studies
Oral
No toxic effects were observed when 97.5% Methylisothiazolinone was administered to rats in drinking water for 13 weeks at concentrations of 0, 75, 250, or 1000 ppm. 3 Dogs that were fed diets prepared with 51.4% Methylisothiazolinone for 3 months had an NOAEL of 1500 ppm. In a subchronic study, rats fed the metabolites NMMA and malonic acid (MA), up to 220 and 44 ppm in the diet, respectively, for 3 months showed no effects in body weight, food consumption, hematology, clinical chemistry, urinalysis, ophthalmology, or gross pathologic changes. Beagle dogs that received up to 500 ppm NMMA and 100 ppm MA in their diets for 3 months had no systemic toxicity.
In a 90-day oral toxicity study performed in accordance with OECD TG 408, groups of 10 male and 10 female Wistar rats received 0, 7.52, 15.05, or 30.09 mg/kg bw Methylisothiazolinone in water daily via gavage. 5 The study included high-dose and control recovery groups that were observed for an additional 28 days following completion of the dosing period. The animals were observed for mortalities, clinical signs of toxicity, ophthalmological changes, and feed consumption. Hematology values and clinical chemistry measurements were taken. Sperm were analyzed for motility, number, and morphology (results reported in the section below). Terminal studies included measuring organ weight and relative organ weight, and performing gross pathological and histopathological assessments. No treatment-related mortalities, clinical signs of toxicity, ophthalmological changes, or changes in feed consumption were observed. There were no significant treatment-related changes in hematological values or clinical chemistry. No significant adverse effects were reported in terminal studies. The NOAEL was 30.09 mg/kg bw/d in males and females based on no treatment-related mortality or clinical signs of toxicity.
Inhalation
While there are no published inhalation data on Methylisothiazolinone, a 13-week repeated-dose inhalation study on MCI/MI was performed in accordance with OECD TG 413. 15 Groups of 16 Crl: CD(SD)BR rats per sex were exposed to 14% MCI/MI (11% MCI/3% MI). The rats were exposed whole body for 6 h/d, 5 d/wk, at aerosol concentrations of 0, 0.34, 1.15, or 2.64 mg ai/m3, with an aerosol particle size of 1.1 to 1.4 µm (mean mass median diameter, which is defined as the diameter at which 50% of the particles by mass are larger and 50% are smaller). During the exposure period, the rats were observed for clinical signs of toxicity, and body weight and ophthalmologic evaluations were made. At study termination, hematology, clinical chemistry, gross pathology, and histopathologic evaluations were conducted. No statistically significant effects were observed in the hematology, gross pathology, or ophthalmologic evaluations at any concentration. At 2.64 mg/m3, rats of both sexes had signs consistent with exposure to a sensory irritant, including chromorhinorrhea, rhinorrhea, eye squint, bradypnea, and dyspnea. Decreased body weight gains, decreased male spleen weights, and decreased serum protein in females were also observed in rats exposed to 2.64 mg/m3. No treatment-related clinical signs of toxicity, body weight effects, or organ weight effects were observed in the 0.34 or 1.15 mg/m3 exposure groups. Treatment-related histopathologic findings consisting of slight to moderate incidences of eosinophilic droplets in the anterior respiratory mucosa of the nasal turbinates and slight rhinitis in the lining of the anterior portion of the nasal cavity were observed in the 2.64 mg/m3 dose group. At 1.15 mg/m3, rhinitis was observed in rats of both sexes. No treatment-related histopathologic effects were observed in the 0.34 mg/m3 dose group. All histopathologic changes were minor, potentially reversible, and generally reflective of minimal tissue response to a very mild, low-grade respiratory irritant. Based on the occurrence of rhinitis, the LOEL was 1.15 mg/m3 ai. The NOAEC was 0.34 mg/m3 ai.
Reproductive and Developmental Toxicity
In a teratogenicity study, Methylisothiazolinone was administered by daily single oral doses to pregnant rats at doses of 5, 20, or 60 (reduced to 40) mg/kg bw/d on gestation days 6 to 19. Females in the high-dose group had clinical signs of rales, gasping, and labored breathing and at necropsy had red areas in the glandular portion of the stomach and lungs. No treatment-related effects were observed in the fetuses. The maternal and developmental NOAELs were 20 and 40 mg/kg/d, respectively. In a teratogenicity study of Methylisothiazolinone in rabbits, pregnant females received daily single oral doses of 3, 10, or 30 mg/kg/d Methylisothiazolinone on gestation days 6 to 28. Maternal effects in the 30 mg/kg/d group included decreased defecation and dark red areas in the stomach. The maternal NOAEL was 10 mg/kg/d. No treatment-related effects were observed in the fetuses, and the developmental NOAEL was determined to be 30 mg/kg/d. A 2 generation reproduction toxicity test found that Methylisothiazolinone in drinking water at concentrations up to 1000 ppm was not a reproductive toxicant. 3
In the 90-day oral toxicity study described above, no adverse effects were observed on the male rat reproductive system after Wistar rats received up to 30.09 mg/kg bw Methylisothiazolinone in water. 5
The teratogenic potential of 49.8% Methylisothiazolinone was studied in Wistar rats in accordance with OECD TG 414. 5 Groups of 25 pregnant rats received 33.4, 49.8, or 74.7 mg/kg of the test material in water via gavage once daily on days 6 through 15 of gestation. Slight maternal toxic effects, including depressed body weight gains and feed consumption, were observed at 49.8 mg/kg and 74.7 mg/kg. A significant increase in the number of visceral anomalies were observed at 74.7 mg/kg, which were likely due to maternal toxicity. No teratogenic effects on fetuses attributed to the test material could be verified. The NOAEL and LOAEL for maternal toxicity were 33.4 mg/kg bw/d and 49.8 mg/kg bw/d, respectively; the NOAEL and LOAEL for embryotoxicity were 49.8 mg/kg bw/d and 74.7 mg/kg bw/d, respectively.
Genotoxicity
Methylisothiazolinone (up to 1000 µg/plate) and the metabolite NMMA (up to 5000 µg/plate) were not mutagenic in the Ames test when tested with and without metabolic activation. In a Chinese hamster ovary (CHO) cell assay, 97.5% pure Methylisothiazolinone was nonmutagenic when tested with and without metabolic activation (0.5-40.0 μg/mL). However, another CHO assay that studied Methylisothiazolinone at 97.5% ai (0.0785-5000 μg/mL) found significant increases in cells with chromosome aberrations, with and without metabolic activation. The aberrations were accompanied by significant cytotoxicity, which may have caused a false positive in this assay. Methylisothiazolinone was nonmutagenic in an unscheduled DNA synthesis assay and in a micronucleus test. 3
Genotoxicity studies are summarized in Table 3. Methylisothiazolinone (49.0%-49.8%) was not mutagenic in an Ames study, chromosome aberration study, or in a mammalian cell gene mutation assay, nor was it mutagenic in an in vivo micronucleus assay in mice. 5
Genotoxicity Studies of Methylisothiazolinone.
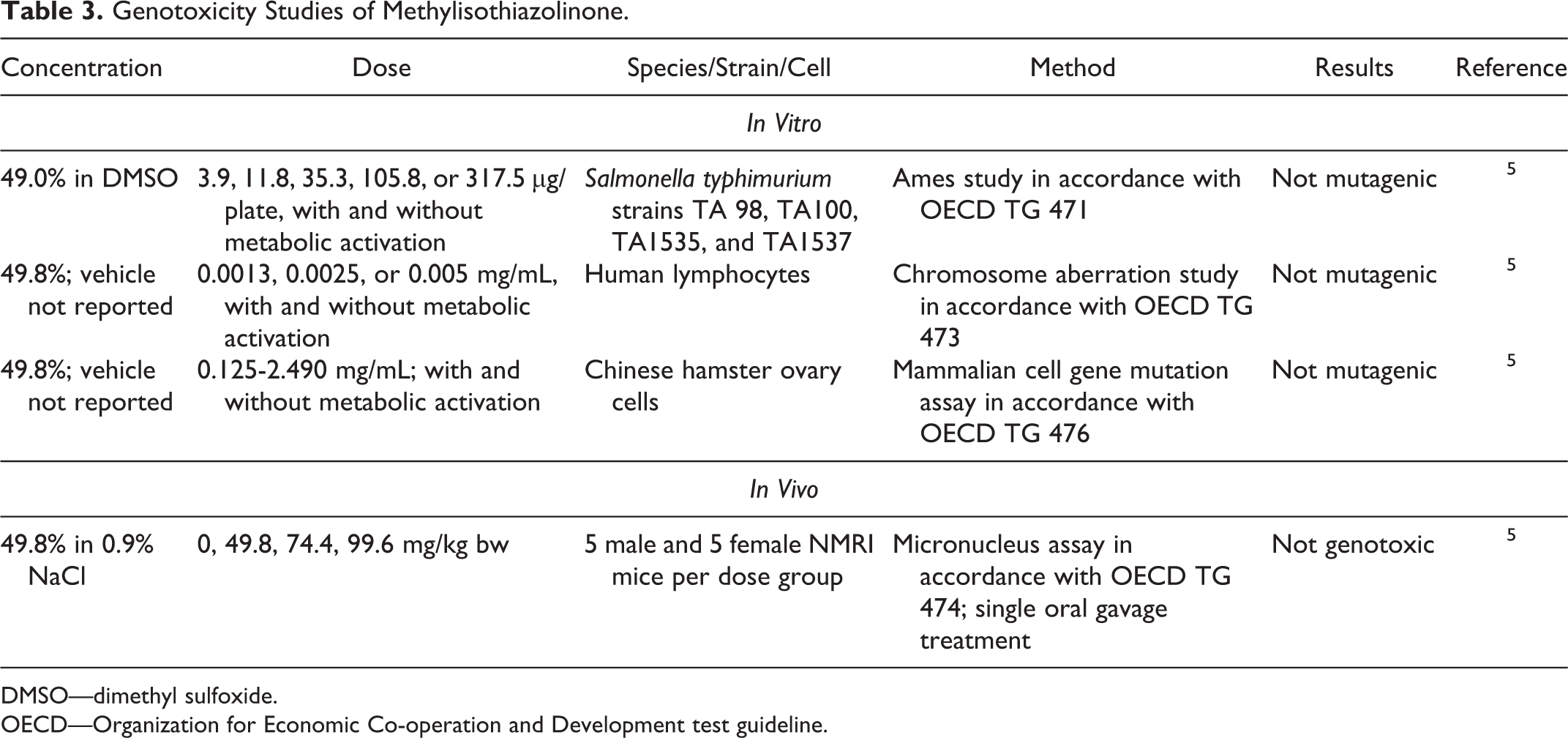
DMSO—dimethyl sulfoxide.
OECD—Organization for Economic Co-operation and Development test guideline.
Carcinogenicity
Studies of the carcinogenicity of the sole ingredient Methylisothiazolinone were not available; however, a 2-year drinking water study in rats concluded that the mixture MCI/MI tested up to 300 ppm was not a carcinogen. 3
Other Relevant Studies
Neurotoxicity
An acute in vitro neurotoxicity study of Methylisothiazolinone (up to 300 µM) in embryonic rat cortical neurons and glia observed widespread neuronal cell death within 24 hours in the cortical cultures. Glial toxicity was low. A 14-hour in vitro neurotoxicity study of Methylisothiazolinone (up to 3.0 µM) from the same laboratory concluded that prolonged exposure to Methylisothiazolinone and related isothiazolinones may damage developing nervous systems. However, no evidence of neurotoxicity has been observed in vivo. 3
Dermal Irritation and Sensitization
In EpiDerm skin constructs, 1.7% Methylisothiazolinone applied for 3 or 60 minutes was noncorrosive. 3 In the same study, 51.5% Methylisothiazolinone was noncorrosive in the 3-minute exposure but corrosive at the 60-minute exposure. Undiluted 97.8% Methylisothiazolinone was corrosive to intact rabbit skin after an exposure period of 1 hour. Rabbit dermal irritation studies of Methylisothiazolinone at 9.69% and 10% concluded the chemical was nonirritating. A single 24-hour application of 100 ppm Methylisothiazolinone in 40 volunteer subjects did not produce skin irritation. Respective skin irritation studies in body lotion, shampoo, and sunscreen formulations containing 100 ppm Methylisothiazolinone also found Methylisothiazolinone to be nonirritating.
In a guinea pig maximization test, 0.076% w/v Methylisothiazolinone was a weak sensitizer and a follow-up study found that 0.015% Methylisothiazolinone produced no sensitization.
3
An investigation using the Buehler method found that 99.8% Methylisothiazolinone was a sensitizer at concentrations
A re-evaluation of the LLNA results reported in the published literature in an editorial article indicates that Methylisothiazolinone should be categorized as a strong sensitizer and not a moderate sensitizer as previously reported. 2
In a cumulative irritation/sensitization study of Methylisothiazolinone in 80 subjects, the sensitization threshold was determined to be at or around 1000 ppm. 3 A human repeated insult patch test (HRIPT) in 98 subjects tested with 100 ppm Methylisothiazolinone concluded that Methylisothiazolinone did not induce skin sensitization in humans. A series of HRIPTs evaluating the sensitization of 50% Methylisothiazolinone at concentrations of 200, 300, 400, 500, or 600 ppm concluded that Methylisothiazolinone up to 600 ppm was not a dermal sensitizer.
In sensitization studies conducted in 11 Methylisothiazolinone-allergic patients, the lowest eliciting dose in a patch test was 1.47 µg Methylisothiazolinone/cm2 (49 ppm). No reactions were observed at 0.441 µg Methylisothiazolinone/cm2 (15 ppm) or lower, nor were there any reactions in the controls. In an HRIPT of 100 ppm Methylisothiazolinone, with or without various glycols, no evidence of induced allergic contact dermatitis was observed in any of the subjects. 2
Dermal irritation and sensitization studies are summarized in Table 4. In a rabbit irritation study, 49.0% Methylisothiazolinone in water was corrosive. 5 Methylisothiazolinone was sensitizing in a guinea pig maximization test and in an LLNA when tested at up to 10.0%; however, it was not a sensitizer in another LLNA at up to 4.5%. In human sensitization studies, dose-dependent sensitization was observed to Methylisothiazolinone at up to 2500 ppm in a cumulative irritation study and HRIPTs.
Irritation and Sensitization Studies of Methylisothiazolinone.
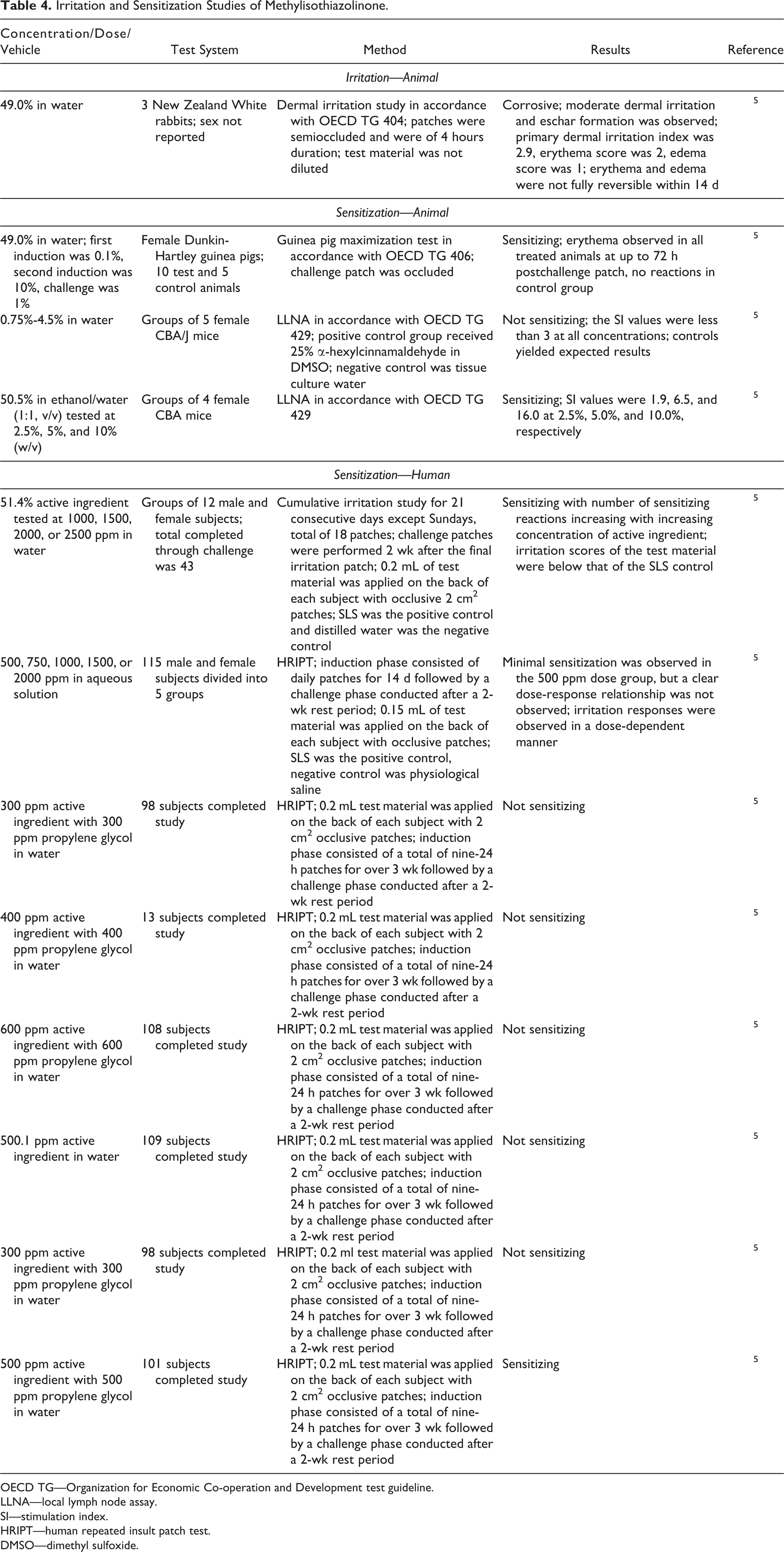
OECD TG—Organization for Economic Co-operation and Development test guideline.
LLNA—local lymph node assay.
SI—stimulation index.
HRIPT—human repeated insult patch test.
DMSO—dimethyl sulfoxide.
Phototoxicity
Methylisothiazolinone at 100 ppm was not phototoxic or photosensitizing in guinea pig studies. No phototoxic effects were observed in a study of 200 ppm Methylisothiazolinone in 12 female subjects. 3 A photosensitization study of 200 ppm Methylisothiazolinone in 32 subjects did not produce photoallergic reactions.
Ocular Irritation Studies
A bovine cornea study classified Methylisothiazolinone (neat) as mildly irritating. Ocular irritation studies in body lotion, shampoo, and sunscreen formulations containing 100 ppm Methylisothiazolinone found the formulations nonirritating in rabbit eyes. 3
Human
In an ocular irritation study, 12 human subjects received 100 ppm Methylisothiazolinone in buffered physiological saline as a single 10 µL drop in the eye on 5 consecutive days. 5 An ophthalmologist performed eye examinations and the subjects subjectively rated the irritation. Mild pink in the bulbar and palpebral conjunctiva and slight lacrimation were noted 30 to 60 seconds after instillation of the test material, but not after 60 minutes, and the results were comparable to the control subjects. No more than slight/mild stinging/burning/pain were reported for both the test material and the control. Three adverse events were reported by 2 subjects: 1 subject reported mild bilateral ocular discharge and stinging, which were possibly related to the test material; and the other subject reported mild bilateral ocular discharge which was unlikely related to the test material. The test material was considered safe and well tolerated in this study.
Clinical Studies
Retrospective and Multicenter Studies
In a clinical study of 22 patients tested with fractions isolated from a trade name mixture of MCI/MI, 2 patients had positive reactions to Methylisothiazolinone. 3 Sensitization may have been due to cross-reactions to MCI. Methylisothiazolinone was determined to be a weak sensitizer in a study of 12 patients. Eighty-five patients with predetermined sensitization to MCI/MI were tested epicutaneously to 500 or 1000 ppm Methylisothiazolinone. The results show that at high concentrations of Methylisothiazolinone (500-1000 ppm), 32% of the subjects with known sensitivity to MCI/MI reacted to Methylisothiazolinone. In a repeat open application test, 7 patients (64%) reacted to 0.105 and 0.21 µg Methylisothiazolinone/cm2 and 2 patients (18%) reacted to 0.0105 µg Methylisothiazolinone/cm2.
Incidences of contact allergy to Methylisothiazolinone, tested separately from MCI/MI, appear to be increasing in Europe since the start of the use of Methylisothiazolinone as a stand-alone ingredient. 2
Methylisothiazolinone was named Allergen of the Year for 2013 by the American Contact Dermatitis Society due to the rise of use of the preservative and the increased incidences of contact allergy being reported, especially in the European Union. 2 A standard series of patch testing includes the mixture MCI/MI, which may miss 40% of contact allergy to Methylisothiazolinone alone due to the relatively low concentration of Methylisothiazolinone in the mixture. Recommendations have been made to test for Methylisothiazolinone contact allergy separate from the MCI/MI, although there currently is no consensus of about the concentration of Methylisothiazolinone that should be tested.
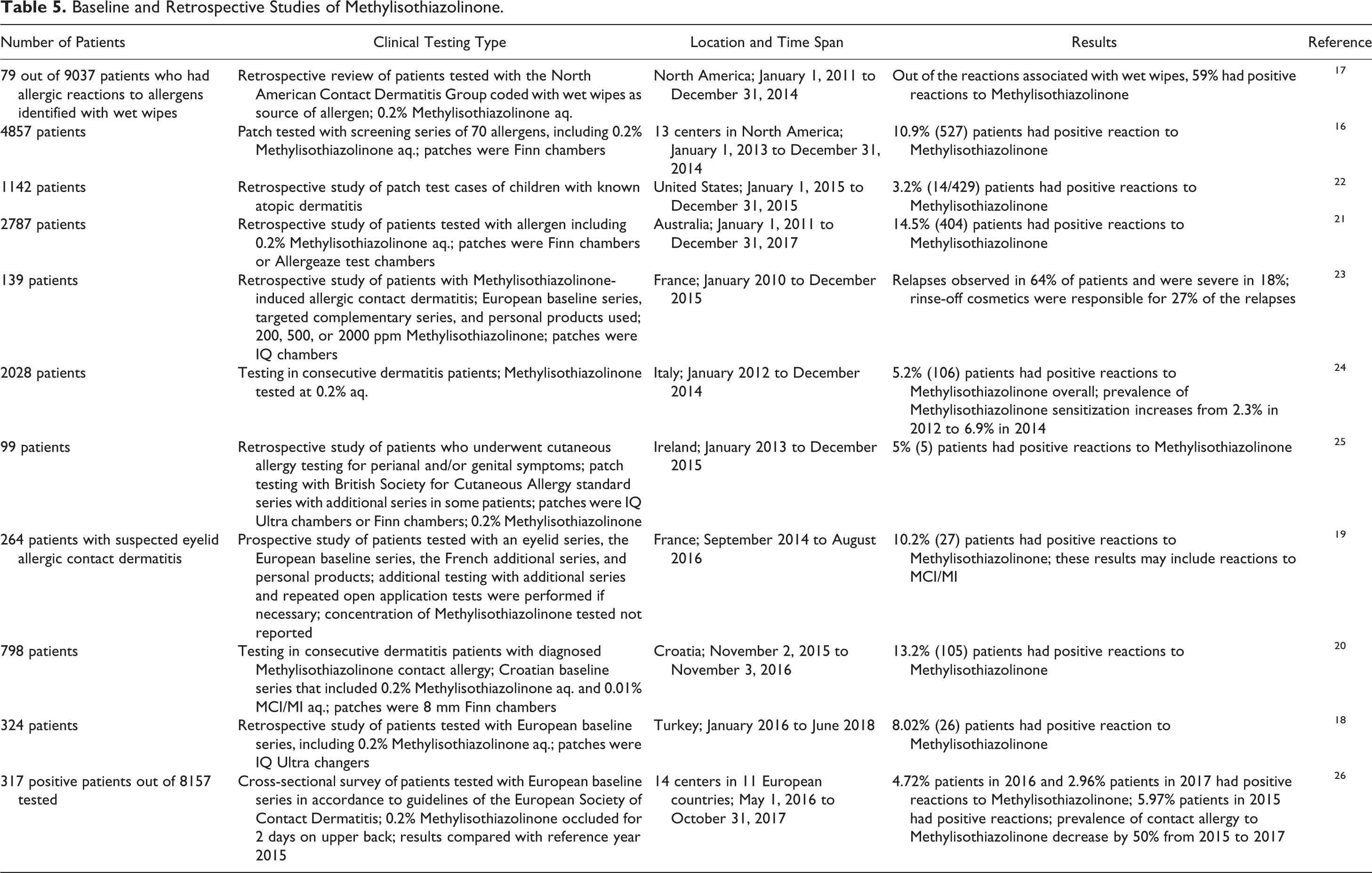
A selection of the numerous baseline and retrospective studies on Methylisothiazolinone that have become available in the published since 2014 are summarized in Table 5. These studies show that sensitization to Methylisothiazolinone is still found worldwide.16-26 In a study from 14 centers in 11 European countries, the prevalence of contact allergy to Methylisothiazolinone decreased by 50% from 2015 to 2027. 26 Of note, the share of cosmetic products (leave-on in particular) eliciting allergic contact dermatitis is decreasing.
Baseline and Retrospective Studies of Methylisothiazolinone.
Case Studies
Three cases of allergic contact dermatitis were reported in patients who had come into contact with coolant solutions containing biocides. 3 Patch testing in 2 of the patients revealed ++ and +++ reactions to Methylisothiazolinone, respectively. An investigator in this study developed eczematous dermatitis while isolating coolant components and had a ++ reaction to Methylisothiazolinone during patch testing. Another case study reported hand eczema in a diesel mechanic that was exacerbated with the use of moist toilet paper. The diesel oil and the toilet paper each contained trade name mixtures of MCI/MI biocides. Positive reactions to Methylisothiazolinone were observed with patch testing. Two cases of occupational contact allergy and dermatitis were reported in patients exposed to compounds containing the biocide Methylisothiazolinone. Patch testing revealed +++ reactions to Methylisothiazolinone. Four of 14 workers at a Danish paint factory were observed with contact dermatitis after exposure to paint additives containing 7% to 10% Methylisothiazolinone. Positive reactions were observed in all 4 patients during patch testing. Numerous other reports of contact allergy, particularly to toilet wipes and water-based wall paint containing Methylisothiazolinone, have been reported. 3
A sampling of case studies that report adverse effects to Methylisothiazolinone from various exposures is summarized in Table 6. Cases include reports of Methylisothiazolinone sensitization from a wide range of materials, including personal care products, paints, photographic processing agents, glues, eye glass frames, and cleaners.27-36
Case Reports.
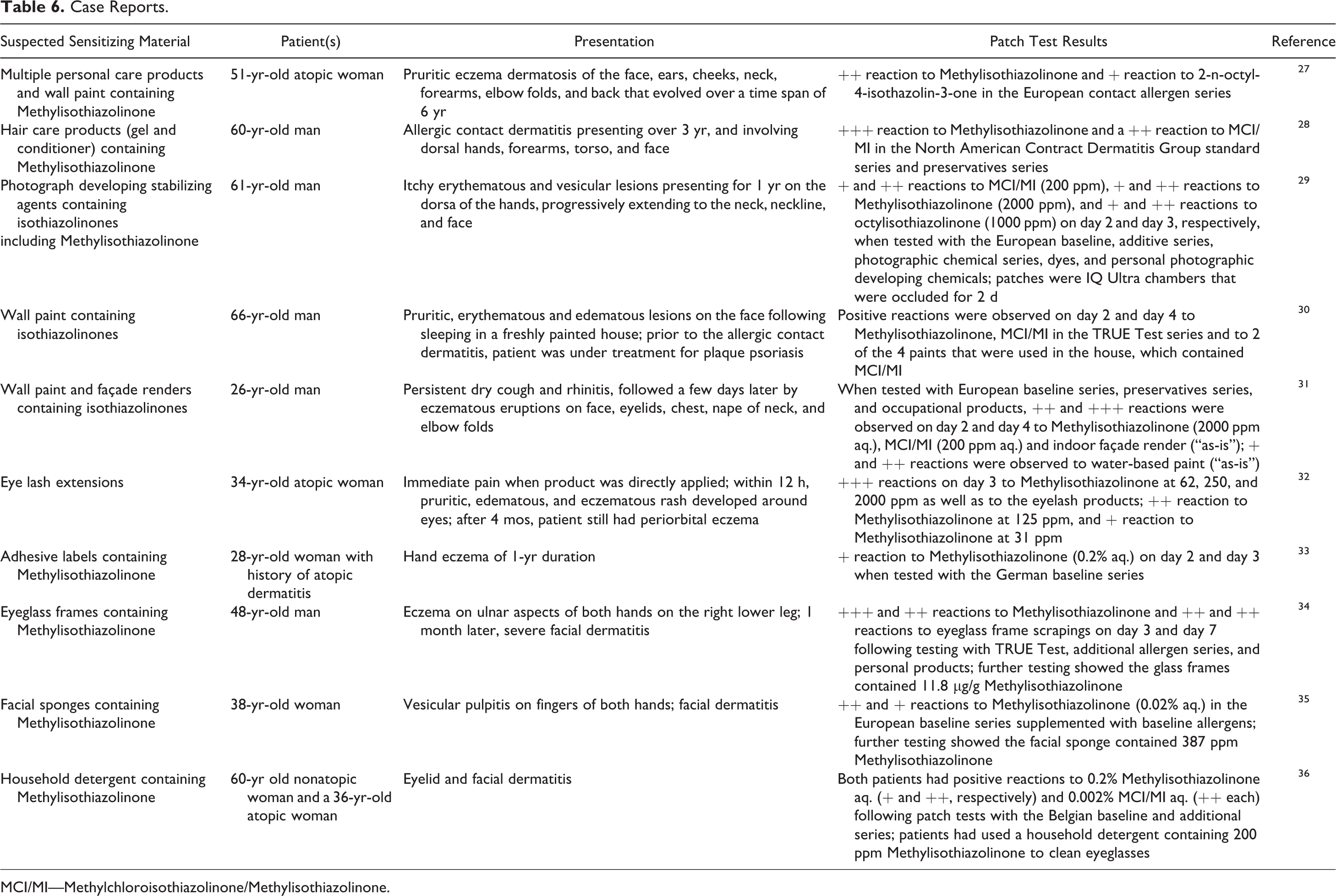
MCI/MI—Methylchloroisothiazolinone/Methylisothiazolinone.
Quantitative Risk Assessment
Cosmetics Europe and the CIR Science and Support Committee (SCC) conducted QRAs of Methylisothiazolinone in response to the increased incidences of contact sensitization to Methylisothiazolinone in Europe. 2 The QRA, which used a conservative no expected sensitization induction level of 15 µg/cm2/d that was derived based on a weight of evidence evaluation of data from 5 HRIPTs and 4 LLNAs, predicted that consumer exposures to 100 ppm Methylisothiazolinone in skin leave-on products and cosmetic wet wipes could induce skin sensitization, while exposures to the same concentration in rinse-off products and hair care leave-on products would not induce skin sensitization.
Summary
In 2019, the Panel published an amended safety assessment of the preservative Methylisothiazolinone with the conclusion that this ingredient “is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a QRA.” This conclusion superseded the findings of the Panel’s earlier safety assessment that was published in 2010. At the September 2019 Panel meeting during the re-evaluation of the mixture MCI/MI, the Panel reopened the amended safety assessment of Methylisothiazolinone to consider additional newly available data, with particular regard to inhalation toxicity.
According to 2019 VCRP survey data, Methylisothiazolinone (when not used with MCI) is reported to be used in a total of 915 formulations; the majority of the uses are in bath soaps and detergents. Use of Methylisothiazolinone (without MCI) has increased since 2014, where 745 uses were reported; the majority of the uses reported then were in noncoloring hair conditioners and shampoos. The maximum concentrations of use for Methylisothiazolinone in 2020 is reported to range from 0.000002% to 0.00975%, with 0.00975% reported in hair conditioners and 0.009% used in leave-on hair products. In the amended safety assessment published in 2019, the maximum concentration of use range was reported to be 3.5 × 10−8% to 0.01%, with 0.01% reported in multiple product categories including eye makeup remover, hair shampoos and conditioners, and skin care products (both leave-on and rinse-off).
The US EPA has released a draft risk assessment for MCI/MI that included analysis of residential and occupational handler risks to inhalation of spray products containing Methylisothiazolinone and Methylisothiazolinone-preserved paints. The inhalation MOEs for residential aerosol exposures ranged from 15 to 14000 and were not of toxicological concern because the values were greater than the LOC of 10. The MOEs for occupational aerosol exposures ranged from 4.4 to 5800; certain exposure scenarios were of toxicological concern when the LOC was below the value of 10. Scenarios for residential handlers applying paint and occupational inhalation of paint vapors assuming long exposure durations had MOEs that had LOC below 10. The US EPA also assessed incidental oral and dermal exposure in textile and household cleaning products and found that exposures across routes are not aggregated. These analyses of exposures to paints and textile and household cleaning products are not considered relevant to the assessment of cosmetic safety.
In a dermal study in rats, the LD50 for 49.0% Methylisothiazolinone was greater than 2000 mg/kg bw. In oral studies, the LD50 for a 1% solution of Methylisothiazolinone in rats was 148.0 mg/kg, while the LD50 for a 50% solution of Methylisothiazolinone in rats was 232 to 249 mg/kg in males and 120 mg/kg in females. The LC50 of aerosolized 49.8% Methylisothiazolinone in rats was 0.422 mg/L in males and 0.354 mg/L in females.
In a 28-day oral toxicity study in rats tested with 0, 10.0, 28.6, or 71.2 mg/kg bw Methylisothiazolinone, the NOAEL was 28.6 mg/kg bw/d and the LOAEL was 71.2 mg/kg bw/d based on lethargy and mortality. When Methylisothiazolinone was tested at up to 30.09 mg/kg bw in a 90-day oral toxicity study in rats, the NOAEL was 30.09 mg/kg/d based on no treatment-related mortality or clinical signs of toxicity.
In the 90-day oral toxicity study, no adverse effects were observed on the male rat reproductive system after rats received up to 30.09 mg/kg bw Methylisothiazolinone in water. In a study that investigated the teratogenic potential of 49.8% Methylisothiazolinone in rats, no teratogenic effects on fetuses attributed to the test material could be verified. The NOAEL and LOAEL for maternal toxicity were 33.4 mg/kg bw/d and 49.8 mg/kg bw/d, respectively; the NOAEL and LOAEL for embryotoxicity were 49.8 mg/kg bw/d and 74.7 mg/kg bw/d, respectively. In a 13-week inhalation study of 14% MCI/MI in rats that followed OECD TG 413, MCI/MI was tested at up to 2.64 mg ai/m3. Based on the occurrence of rhinitis, the LOEL was 1.15 mg/m3. The NOEL was 0.34 mg/m3.
Methylisothiazolinone (49.0%-49.8%) was not mutagenic in an Ames study, chromosome aberration study, or in a mammalian cell gene mutation assay. Additionally, it was not mutagenic in an in vivo micronucleus assay in mice.
In a rabbit irritation study, 49.0% Methylisothiazolinone in water was corrosive. Methylisothiazolinone was sensitizing in a guinea pig maximization test and in an LLNA when tested at up to 10.0%; however, it was not a sensitizer in another LLNA at up to 4.5%. In human sensitization studies, dose-dependent sensitization was observed to Methylisothiazolinone at up to 2500 ppm in a cumulative irritation study and HRIPTs. Methylisothiazolinone (100 ppm in saline) was considered safe and well tolerated in an ocular irritation study of human subjects.
A sampling of the numerous baseline and retrospective studies on Methylisothiazolinone that have become available in the published literature since 2014 indicate that sensitization to Methylisothiazolinone is still found worldwide. A selection of case studies that report adverse effects to Methylisothiazolinone from various exposures included reports of Methylisothiazolinone sensitization from a wide range of materials, including personal care products, paints, photographic processing agents, glues, eye glass frames, and cleaners. In a study from 14 centers in 11 European countries, the prevalence of contact allergy to Methylisothiazolinone decreased by 50% from 2015 to 2017. Of note, the share of cosmetic products (leave-on in particular) eliciting allergic contact dermatitis is decreasing.
Discussion
This safety assessment is on the preservative Methylisothiazolinone as used in cosmetics. In response to concerns of reports of adverse events observed in infants following inhalation exposure to humidifier disinfectants that contained the preservative mixture MCI/MI, the Panel moved to reopen the safety assessment of Methylisothiazolinone in September 2019. A search of inhalation toxicity data for Methylisothiazolinone (separate from the combination of MCI/MI) did not yield any additional inhalation data; however, studies were detailed in the MCI/MI report. The Panel reviewed a 13-week repeated-dose inhalation study of MCI/MI in rats and determined that the data mitigated concern for the use of Methylisothiazolinone at the reported concentrations in cosmetic products that could be incidentally inhaled following cosmetic use. The Panel also reviewed a draft risk assessment for MCI/MI produced by the US EPA and determined that the analyses of exposures to paints, textile, and household cleaning products were not relevant to the assessment of cosmetic safety due to exposure duration and concentrations of application being magnitudes greater than those of cosmetic use.
As discussed in the previous report on Methylisothiazolinone, the Panel reviewed the results of QRAs performed by Cosmetics Europe and the CIR SCC. Those results supported the safety of the use of Methylisothiazolinone in rinse-off product categories at concentrations up to 100 ppm. However, the QRA indicated that Methylisothiazolinone use in several leave-on product categories, such as wet wipes, would be safe only at concentrations lower than 100 ppm. Using the QRA results, the Panel reaffirmed the limitation of 100 ppm Methylisothiazolinone in rinse-off products. However, they also determined that the original limitation for leave-on products needed to be modified, and that leave-on cosmetic products should be formulated to contain Methylisothiazolinone at concentrations below 100 ppm and to be nonsensitizing, as demonstrated, for example, by QRA estimates of safe exposures (typically expressed in µg/cm2/d) for the relevant cosmetic product category.
The Panel’s recommendations for Methylisothiazolinone in rinse-off and leave-on cosmetic products are intended to prevent the induction of sensitization to Methylisothiazolinone. However, the Panel cautioned that following these recommendations may not necessarily prevent the elicitation of allergic reactions in individuals who are already allergic to Methylisothiazolinone. Individuals sensitized to Methylisothiazolinone should avoid products that contain Methylisothiazolinone.
The Panel discussed the issue of incidental inhalation exposure from hair sprays and fragrance preparations. The limited data available from inhalation studies, including acute exposure data on Methylisothiazolinone and subchronic exposure data on MCI/MI, suggest little potential for respiratory effects at relevant doses. Methylisothiazolinone is reportedly used at concentrations up to 0.00095% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposures expected in the breathing zone and the absence of significant signs of toxicity in acute, short-term, subchronic, chronic, reproductive and developmental animal studies, and genotoxicity studies reviewed by the Panel, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that Methylisothiazolinone is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm (ie, 0.01%) and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a QRA or similar methodology.
Footnotes
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contribution
Burnett, C. contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; and critically revised manuscript. Bergfeld, W., Belsito, D., Peterson, L., Klaassen, C., Liebler, D., Marks, J., Shank, R., Slaga, T., Snyder, P., and Cohen, D.L. contributed to conception and design and contributed to analysis and interpretation. Heldreth, B. contributed to design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
