Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of methylisothiazolinone (MI), which functions as a preservative. The Panel reviewed relevant animal and human data provided in this safety assessment and in a previously published safety assessment of MI and concluded that MI is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a quantitative risk assessment.
Introduction
In 2010, the Panel published a final report of the safety assessment of methylisothiazolinone (MI) with the conclusion that “MI is safe for use in cosmetic formulations at concentrations up to 100 ppm (0.01%).” 1 At the March 2013 Cosmetic Ingredient Review (CIR) Expert Panel meeting, the Panel reviewed newly provided clinical data indicating a higher than expected frequency of individuals who have allergic reactions to the preservative MI. In some cases, comparative data were available indicating a higher frequency of positive reactions than currently seen with the combination preservative, methylchloroisothiazolinone (MCI)/MI. The Panel reopened this safety assessment to gather and evaluate additional data.
In June 2014, the Panel reviewed the results of quantitative risk assessments (QRAs) performed by Cosmetics Europe and the CIR Science and Support Committee (CIR SSC). The results supported the safety of the use of MI in rinse-off product categories at concentrations up to 100 ppm. However, the QRAs indicated that MI use in many leave-on product categories would be safe only at lower concentrations.
The Panel previously reviewed the safety of the mixture MCI/MI (sold at a ratio of 3:1) with the conclusion that the mixture “may be safely used in ‘rinse-off’ products at a concentration not to exceed 15 ppm and in ‘leave-on’ products at a concentration not to exceed 7.5 ppm.” 2
Extensive data from the original MI safety assessment report, which was finalized in 2008 and published in 2010, were considered by the Panel during the review of this amended safety assessment. Because those data are included in the report that was published in 2010 1 (and can be found on the CIR website [https://www.cir-safety.org/ingredients]), only new information will be included in this report. However, notes have been added in text, referring to the original safety assessment, to identify the types of data that were available in that original report.
Chemistry
The definition, physical and chemical properties, method of manufacturing, and impurities of MI were described in the original safety assessment (Figure 1). 1
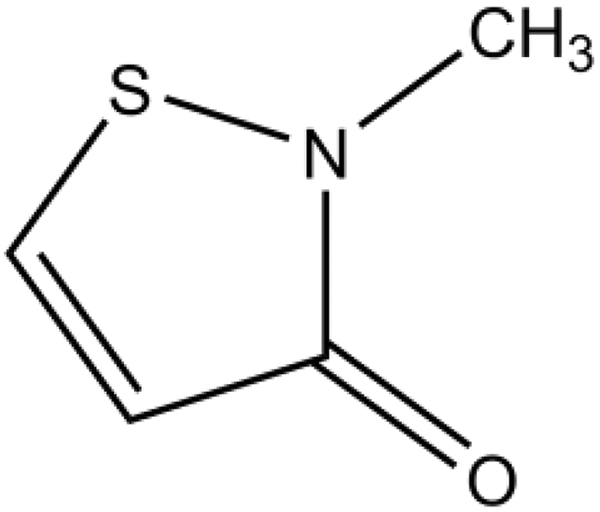
Methylisothiazolinone.
Use
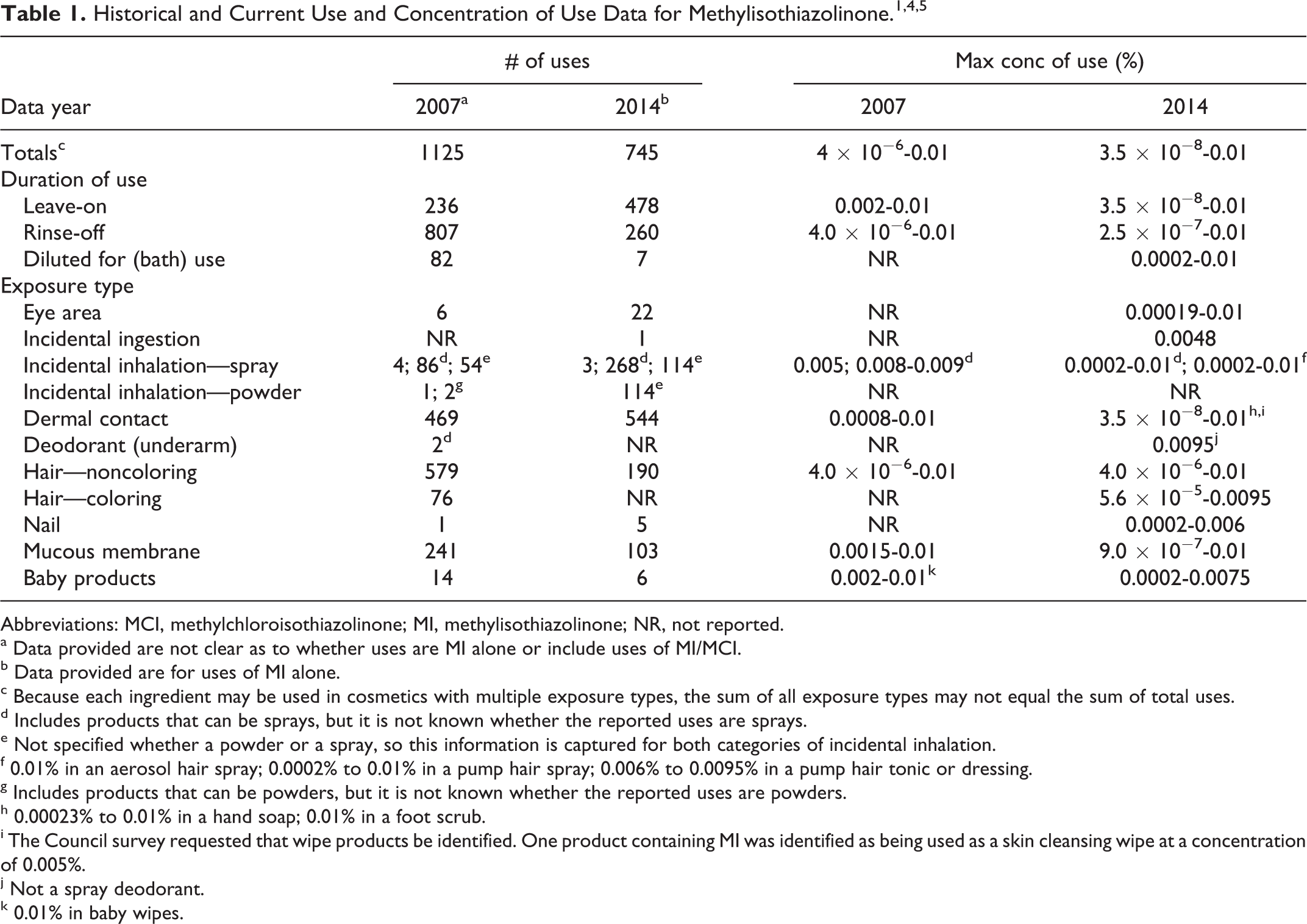
Cosmetic
Table 1 presents the historical and current product formulation data for MI. Methylisothiazolinone functions as a preservative in cosmetic products. 3 According to information from the Food and Drug Administration (FDA) Voluntary Cosmetic Registration Program (VCRP) database in 2007, MI had 1,125 reported uses, with the majority of the uses reported in noncoloring hair conditioners and shampoos. 1 It should be noted that the information from the VCRP in 2007 did not clearly distinguish cosmetic products in which MI was used in combination with MCI from products in which MI was used without MCI. This safety assessment addresses the use of MI in cosmetic products that do not also contain MCI. In 2008, industry reported the maximum use concentration range to be 4 × 10−6% to 0.01%, with 0.01% reported in both leave-on and rinse-off baby, noncoloring hair, and dermal contact products. 1 In 2014, the VCRP database indicated that MI is used as an ingredient in 745 cosmetic products that do not also contain MCI, with the majority of the uses reported in leave-on products such as skin moisturizers. 4 A survey of use concentrations conducted by the Personal Care Products Council (Council) in 2014 reported a maximum concentration of use range of 3.5 × 10−8% to 0.01%, with 0.01% reported in multiple product categories including eye makeup remover, hair shampoos and conditioners, and skin care products (both leave-on and rinse-off). 5
Abbreviations: MCI, methylchloroisothiazolinone; MI, methylisothiazolinone; NR, not reported.
a Data provided are not clear as to whether uses are MI alone or include uses of MI/MCI.
b Data provided are for uses of MI alone.
c Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
d Includes products that can be sprays, but it is not known whether the reported uses are sprays.
e Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
f 0.01% in an aerosol hair spray; 0.0002% to 0.01% in a pump hair spray; 0.006% to 0.0095% in a pump hair tonic or dressing.
g Includes products that can be powders, but it is not known whether the reported uses are powders.
h 0.00023% to 0.01% in a hand soap; 0.01% in a foot scrub.
i The Council survey requested that wipe products be identified. One product containing MI was identified as being used as a skin cleansing wipe at a concentration of 0.005%.
j Not a spray deodorant.
k 0.01% in baby wipes.
Methylisothiazolinone was reported to be used in noncoloring hair sprays and hair tonics or dressings that may be aerosolized or become airborne and could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 6 –9 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount. 7,8
The European Union’s Scientific Committee on Consumer Safety (SCCS) recently released an updated opinion on the use of MI. 10 It states that, in leave-on cosmetic products (including “wet wipes”), no safe concentration has been adequately demonstrated for induction or elicitation of contact allergy. In rinse-off cosmetic products, the SCCS has recommended that concentrations up to 0.0015% (15 ppm) MI are safe, in terms of the potential for induction of contact allergy, but stated that there is no information available to evaluate the potential for this ingredient to elicit contact allergy. Furthermore, the SCCS opinion states that MI should not be added to cosmetic products that contain MCI/MI. Cosmetics Europe, the personal care products industry trade association in Europe, has recommended the discontinuation of MI specifically in leave-on skin products, including wet wipes. 11
Noncosmetic
The noncosmetic uses of MI include use in water-based paints, which has been noted in a number of case studies of sensitization reactions (eg, see Table 2). The uses of MI in paints and other noncosmetic products were described in the original safety assessment of MI that was published in 2010. 1
Quantitative Risk Assessment of Methylisothiazolinone (MI) at Highest Maximum Use Concentration (100 ppm) in Cosmetic Products.a,28
Abbreviations: AEL, acceptable exposure levels; CEL, consumer exposure level; NESIL, no expected sensitization induction level; QRA, quantitative risk assessment.
a Shaded rows indicate the ratio of AEL × CEL−1 is less than 1.
b Exposure values assumed for each product category were from the IFRA RIFM QRA Information Booklet (2011) 50 and Api et al. (2008). 51
c Based on NESIL of 15 µg/cm2/d.
d Note that this product category may be diluted prior to application.
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Absorption, distribution, metabolism, and excretion studies are summarized in the original safety assessment of MI that was published in 2010. 1
Toxicological Studies
Acute Toxicity
Acute oral and dermal toxicity studies are summarized in the original safety assessment of MI that was published in 2010. 1
Repeated Dose Toxicity
Oral repeated dose toxicity studies are summarized in the original safety assessment of MI that was published in 2010. 1
Reproductive and Developmental Toxicity
Reproductive and developmental toxicity studies are summarized in the original safety assessment of MI that was published in 2010. 1
Carcinogenicity
Carcinogenicity studies of the sole ingredient MI were not discovered in the published literature, and unpublished data were not submitted to CIR. 1
Genotoxicity
In vitro genotoxicity studies are summarized in the original safety assessment of MI that was published in 2010. 1
Neurotoxicity
Neurotoxicity studies are summarized in the original safety assessment of MI that was published in 2010. 1
Irritation and Sensitization
Irritation
Nonhuman and human dermal irritation studies, and nonhuman ocular irritation studies, are summarized in the original safety assessment of MI that was published in 2010. 1
Sensitization
Nonhuman
A letter to the editor reporting the reevaluation of published local lymph node assay (LLNA) data indicated that MI should be categorized as a strong sensitizer and not a moderate sensitizer, in contrast to previous reports. 12 The earlier reports incorrectly reported 1.9% as the EC3 for MI; the correct value is 0.4%, which is the lowest EC3 estimated from multiple LLNAs using, for example, an acetone/oil vehicle.
Human
Methylisothiazolinone was named the Allergen of the Year for 2013 by the American Contact Dermatitis Society because of the increasing frequency of use of this preservative in consumer products and the increasing incidences of contact allergy reported to be associated with exposures to MI, especially in the European Union. 13 –16 The standard series of patch testing includes exposures to 100 ppm MCI/MI mixture (3:1 ratio). This test may miss up to 40% of subjects with contact allergy to MI, alone, because of the relatively low MI concentration in the MCI/MI mixture tested (approximately 25 ppm MI in a 100 ppm MCI/MI test solution). 17,18 Recommendations have been made to test for contact allergy to MI alone, although there currently is no consensus about the concentration of MI that should be used in such testing. 13,19 –24
The dose–response relationship of contact allergy to MI was investigated in 11 MI-allergic patients. 25 The patients were patch tested with 2 dilution series of 12 doses of MI (Neolone 950 9.7% active ingredient) in 10% ethanol and 90% aqua and 12 doses of MI with 9.26 µg phenoxyethanol/cm2 in 10% ethanol and 90% aqua. (Phenoxyethanol may increase antimicrobial efficacy of MI and was tested to determine whether it influenced reactivity to MI.) The MI doses with and without phenoxyethanol were 0.0105, 0.105, 0.147, 0.21, 0.441, 1.47, 2.94, 4.41, 8.82, 15, 30, and 60 μg MI/cm2. Controls (n = 14) who were not MI-allergic patients were patch tested with 60 µg MI/cm2 and 9.26 µg phenoxyethanol/cm2. Each test site received 15 µL of each dilution applied by filter disc in a Finn Chamber and were occluded for 2 days. Readings were performed on days 2, 3 or 4, and 7. The subjects also underwent a repeated open application test (ROAT) with a cream that contained 0, 0.0105, 0.105, or 0.21 µg MI/cm2 (0, 5, 50, or 100 ppm MI) with phenoxyethanol in 10% ethanol and 90% water. The patients applied 20 µL of the test solution from 4 different bottles twice a day to four 3 cm2 areas of the volar forearm. Sites were read on days 2, 3 or 4, 7, 14, and 21, with additional reading if a reaction occurred between visits. In the patch test, results showed that phenoxyethanol had no influence on reactions to MI. The lowest eliciting dose in the patch test was 1.47 µg MI/cm2 (49 ppm). No reactions were observed at 0.441 µg MI/cm2 (15 ppm) or lower, nor were there any reactions in the control subjects. In the ROAT, 7 patients (64%) reacted to 0.105 and 0.21 µg MI/cm2 and 2 patients (18%) reacted to 0.0105 µg MI/cm2. The authors of this study recommended that the permitted amount of MI in cosmetics be reduced from 100 ppm.
In a human repeated insult patch test (HRIPT) of 226 subjects performed in accordance with the International Contact Dermatitis Research Group criteria for MI, 56 subjects received 100 ppm MI alone and the remaining 170 subjects received 100 ppm MI in combination with various glycols that are used as preservative boosters. 26 No evidence of induced allergic contact dermatitis was observed in any of the subjects, with or without glycols. The study concluded that 100 ppm MI does not cause a risk in cosmetic products when applied on uncompromised skin in the general population. Additional nonhuman and human sensitization studies are summarized in the original safety assessment of MI that was published in 2010. 1
Quantitative Risk Assessment
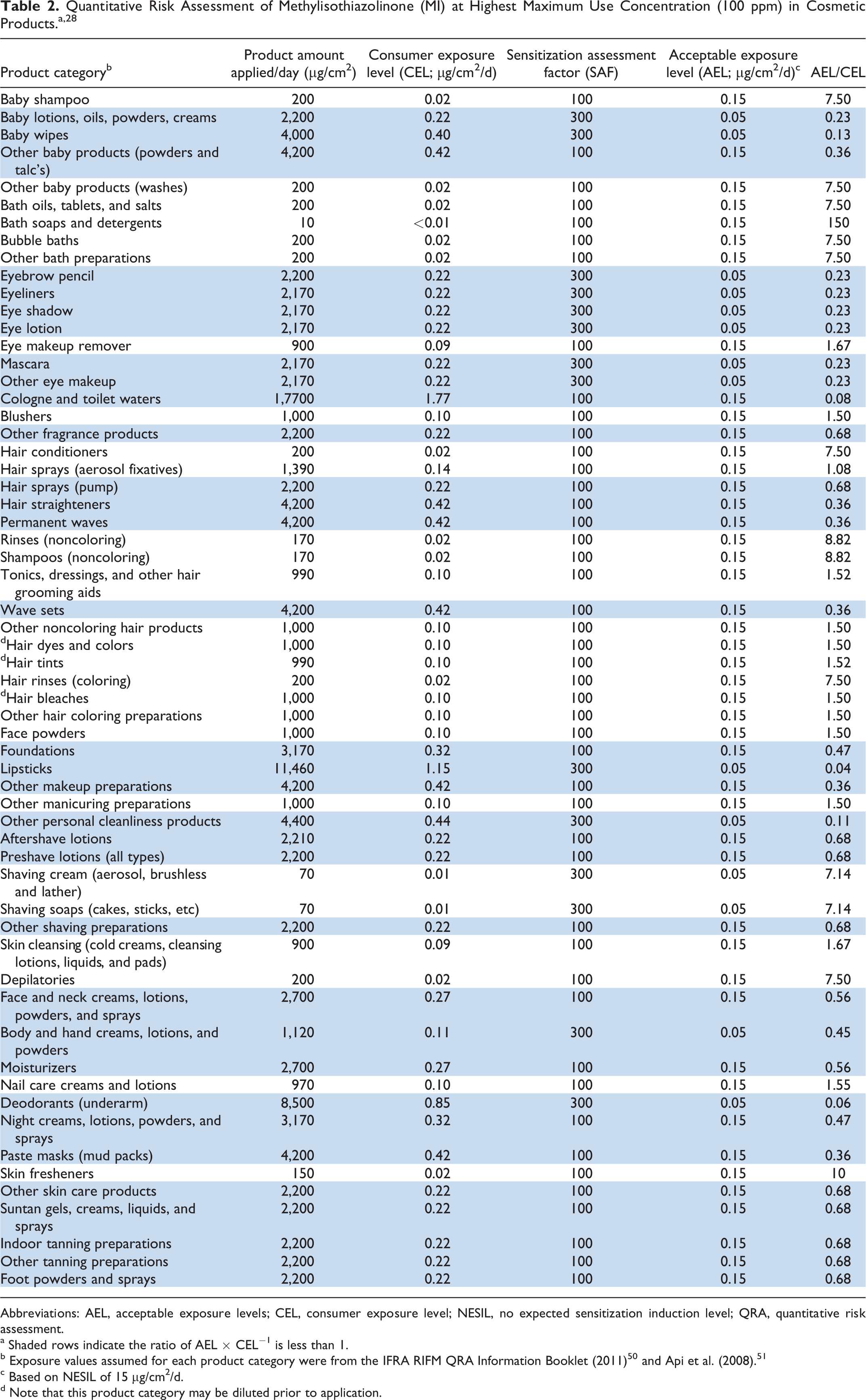
Both Cosmetics Europe and the CIR SSC conducted QRAs, assuming 100 ppm (0.01%) MI in many categories of cosmetic products, in response to the increased incidences of contact sensitization to MI in Europe. 27,28 Both of these QRAs were conducted using the same no expected sensitization induction level (NESIL = 15 µg/cm2/d) and sensitization assessment factors (SAFs).
Table 2 summarizes the QRA conducted by the CIR SSC. A conservative NESIL of 15 µg/cm2/d was derived for MI based on a weight-of-evidence (WoE) evaluation of data from 5 HRIPTs and 4 LLNAs. The NESIL was then used to calculate acceptable exposure levels (AELs) for the potential for the induction of sensitization from dermal exposure to MI in cosmetic products, assuming the maximal use concentration of 100 ppm MI and product category–specific SAFs. The ratio of the AEL and the consumer exposure level (CEL) was then calculated for each of many cosmetic product categories, ranging from hair conditioners (CEL = 0.02 µg/cm2/d) to lipsticks (CEL = 1.15 µg/cm2/d). The concentration of an ingredient is considered to be acceptable in a product when AEL/CEL ≥1 (ie, AEL ≥ CEL).
According to the Cosmetics Europe calculations, the lowest estimated CEL to MI was 0.0011 µg/cm2/d for shower gel, and the highest estimated exposure was 2.27 µg/cm2/d for a nail varnish. The AEL/CEL ratios indicated that concentrations of MI up to 100 ppm (0.01%) would be acceptable for 20 of the 42 categories assessed by Cosmetics Europe and for 27 of the 60 categories assessed by the CIR SSC.
Phototoxicity
Nonhuman and human phototoxicity and photosensitization studies are summarized in the original safety assessment of MI that was published in 2010. 1
Clinical Use
Case Reports
A sampling of case reports and retrospective and multicenter studies reporting MI allergy are summarized in Tables 3 and 4, respectively. Numerous reports of contact allergy, particularly to toilet wipes and water-based wall paint containing MI, have been reported. 29 –37 Incidences of contact allergy to MI, tested separately from MCI/MI, appear to be increasing in Europe in recent years. 38 –49 Additional case reports are summarized in the original safety assessment of MI that was published in 2010. 1
Case Studies.

Abbreviations: BIT, benzisothiazolinone; DMDM, 1,3-Dimethylol-5,5-dimethyl; MCI, methylchloroisothiazolinone; MI, methylisothiazolinone.
Retrospective and Multicenter Studies.

Abbreviations: MCI, methylchloroisothiazolinone; MI, methylisothiazolinone.
Summary
In 2010, the Panel published the final report of the safety assessment of MI with the conclusion that “MI is safe for use in cosmetic formulations at concentrations up to 100 ppm (0.01%).” At the March 2013 CIR Expert Panel meeting, the Panel reopened this safety assessment to gather and evaluate newly provided clinical data indicating a higher than expected frequency of individuals who have allergic reactions to the preservative MI. This summary only contains newly identified information on the MI. The original report should be consulted for the information that was previously reviewed by the Panel.
According to the FDA’s VCRP database in 2007, MI had 1,125 reported uses, with the majority of the uses reported in noncoloring hair conditioners and shampoos. Industry reported the maximum use concentration range to be 4 × 10−6% to 0.01%, with 0.01% reported in leave-on and rinse-off baby, noncoloring hair, and dermal contact products. The information obtained from the VCRP in 2007 did not clearly distinguish cosmetic products in which MI was used in combination with MCI from cosmetic products in which MI was used without MCI. This safety assessment addresses the use of MI in cosmetic products that do not also contain MCI. In 2014, the VCRP database indicated that MI was used as an ingredient in 745 cosmetic products that do not also contain MCI, with the majority of the uses reported in leave-on products such as skin moisturizers. A survey of use concentrations conducted by the Council in 2014 reported a maximum concentration of use range of 3.5 × 10−8% to 0.01%, with 0.01% reported in multiple product categories including eye makeup remover, hair shampoos and conditioners, and skin care products (both leave-on and rinse-off).
The European Union’s SCCS has a recently updated opinion on the use of MI and has found that in leave-on cosmetic products (including “wet wipes”) no safe concentration has been adequately demonstrated for induction or elicitation of contact allergy. In rinse-off cosmetic products, the SCCS has concluded that concentrations up to 0.0015% (15 ppm) MI are safe, in terms of induction of contact allergy, but recognized that there is no information available to evaluate the potential for this ingredient to elicit contact allergy. Furthermore, the SCCS states that MI should not be added to cosmetic products that contain MCI/MI. A reevaluation of the LLNA results reported in the published literature in an editorial article indicates that MI should be categorized as a strong sensitizer, and not a moderate sensitizer as previously reported.
Methylisothiazolinone was named Allergen of the Year for 2013 by the American Contact Dermatitis Society due to the rise of use of the preservative and the increased incidences of contact allergy being reported, especially in the European Union. A standard series of patch testing includes the mixture MCI/MI, which may miss 40% of contact allergy to MI alone due to the relatively low concentration of MI in the mixture. Recommendations have been made to test for MI contact allergy separate from the MCI/MI, although there currently is no consensus of about the concentration of MI that should be tested.
In sensitization studies conducted in 11 MI-allergic patients, the lowest eliciting dose in a patch test was 1.47 µg MI/cm2 (49 ppm). No reactions were observed at 0.441 µg MI/cm2 (15 ppm) or lower, nor were there any reactions in the controls. In a ROAT, 7 (64%) patients reacted to 0.105 and 0.21 µg MI/cm2 and 2 (18%) patients reacted to 0.0105 µg MI/cm2. In a HRIPT of 100 ppm MI, with or without various glycols, no evidence of induced allergic contact dermatitis was observed in any of the subjects.
Numerous reports of contact allergy, particularly to toilet wipes and water-based wall paint containing MI, have been reported. Incidences of contact allergy to MI, tested separately from MCI/MI, appear to be increasing in Europe in recent years.
Cosmetics Europe and the CIR SCC conducted QRAs of MI in response to the increased incidences of contact sensitization to MI in Europe. The QRA, which used a conservative NESIL of 15 µg/cm2/d that was derived based on a WoE evaluation of data from 5 HRIPTs and 4 LLNAs, predicted that consumer exposures to 100 ppm MI in skin leave-on products and cosmetic wet wipes could induce skin sensitization, while exposures to the same concentration in rinse-off products and hair care leave-on products would not induce skin sensitization.
Discussion
The Panel noted the numerous reports of contact allergy to MI in Europe and the increased incidences of contact allergy to MI observed in their own clinical experience. The Panel also noted that MI was named Allergen of the Year for 2013 by the American Contact Dermatitis Society because of the increasing incidence of contact allergy associated with the increasing use of this ingredient as a preservative in cosmetics. The Panel reviewed the results of QRAs performed by Cosmetics Europe and the CIR SSC using an appropriate NESIL (ie, 15 µg/cm2/d) selected based on a WoE evaluation of EC3 values from LLNAs and the results of HRIPTs. The results supported the safety of the use of MI in rinse-off product categories at concentrations up to 100 ppm. However, the QRA indicated that MI use in many leave-on product categories would be safe only at concentrations lower than 100 ppm. As shown in Table 3, for example, the AEL/CEL calculated for 100 ppm (0.01%) MI in baby wipes was 0.13, which the Panel recognizes to be consistent with the reports of increasing incidence of contact allergy associated with the use of MI in wet wipes.
Based on the QRA results, the Panel felt that the current limitation of 100 ppm supported the safety of MI in rinse-off products. Nonetheless, they felt that leave-on products should be formulated to contain MI concentrations below 100 ppm and to be nonsensitizing, as demonstrated, for example, by QRA estimates of safe exposures (typically expressed in µg/cm2/d) for the relevant cosmetic product category.
The risk of inducing sensitization depends on the dose of MI per unit area of the skin exposed (eg, expressed in units of µg/cm2/d). One type of cosmetic product will differ from another in the potential to cause sensitization at a given MI concentration if they differ substantially in application rate, which depends on the amount of product applied per day and the total surface area of the skin to which the product is applied. This helps to explain why the risks associated with MI in rinse-off products are less than those associated with leave-on products and, for instance, why the risks associated with exposures to MI in leave-on hair conditioners would likely be substantially lower than those associated with MI in wipes.
It is important to note that appropriate exposure assumptions used in a QRA can vary depending on factors such as differences in regional habits and practices, properties of the formulation, and degree to which conservative default assumptions and exposure scenarios may be refined based on specific exposure data. The Panel stressed the importance of clearly identifying and justifying the exposure assumptions, and the sources of the assumptions, used in any QRA that might be conducted to predict concentrations of MI unlikely to induce sensitization from the use by consumers of a specific cosmetic product or product category.
The Panel determined that the maximum MI concentration should never exceed 100 ppm (0.01%) in any hair product, leave-on product, or rinse-off product, based on the potential for inducing sensitization and concentrations greater than 100 ppm.
The Panel’s recommendations for MI in rinse-off and leave-on cosmetic products are intended to prevent the induction of sensitization to MI. The Panel cautioned that following these recommendations may not necessarily prevent the elicitation of allergic reactions in individuals who are already allergic to MI. Individuals sensitized to MI should avoid products that contain MI.
The Panel discussed the issue of incidental inhalation exposure to MI in noncoloring hair sprays and hair tonics or dressings. There were no chronic inhalation toxicity data identified or provided. Methylisothiazolinone reportedly is used at concentrations up to 0.01% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles produced in cosmetic aerosols would not be respirable to any appreciable amount. Coupled with the small actual exposures expected in the breathing zone and the absence of significant signs of toxicity in subchronic, chronic, and reproductive and developmental animal studies reviewed previously by the Panel, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at https://www.cir-safety.org/cir-findings.
Conclusion
The CIR Expert Panel concluded that MI is safe for use in rinse-off cosmetic products at concentrations up to 100 ppm and safe in leave-on cosmetic products when they are formulated to be nonsensitizing, which may be determined based on a QRA.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
C. L. Burnett contributed to conception, design, acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. I. Boyer contributed to analysis and interpretation. W. F. Bergfeld, D. V. Belsito, R. A. Hill, C. D. Klaassen, D. C. Liebler, J.G. Marks, R. C. Shank, T. J. Slaga, P. W. Snyder, and L. J. Gill contributed to conception, design, analysis, and interpretation, critically revised the manuscript, and gave final approval. B. Heldreth contributed to analysis and interpretation, critically revised the manuscript, and gave final approval. All authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.