Abstract
Reduction in sensitivity in terms of cytotoxicity is responsible for therapy failure in patients undergoing chemotherapy with first-line anticancer drug molecules. A plethora of literature evidence points out that increased O-linked β-N-acetylglucosamine transferase (OGT) enzyme level/hyper-O-GlcNAcylation has direct implications in development of cancer and interferes with clinical outcomes of chemotherapy via interaction with oncogenic factors. The aim of this research was to evaluate the combination approach of anticancer drugs with an OGT inhibitor (OSMI-1) as an alternative way to resolve issues in the treatment of prostate cancer and assess the benefits offered by this approach. Effect of combination of doxorubicin and docetaxel with OSMI-1 on drug-induced cell death and synergism/antagonism was investigated using resazurin assay. Reduction in OGT enzyme level was evaluated using ELISA kit. Caspase-3/7 fluorescence assay was performed to detect apoptosis induction in PC-3 cells after treatment with the combinations of doxorubicin and OGT inhibitor to further understand the mechanism of cell death by concomitant treatment. Studies reveal that combination approach is indeed effective in terms of reducing the half-maximum growth inhibition value of doxorubicin when concomitantly treated with OSMI-1 and has synergistic effect in prostate cancer cells. PC-3 cells exhibited elevated levels of OGT enzyme in comparison to WPMY-1, and OSMI-1 has potential to inhibit OGT enzyme significantly. Data show that OSMI-1 alone and in combination with doxorubicin reduces OGT enzyme level significantly accompanied by increased apoptosis in prostate cancer cells. Combination of doxorubicin with OSMI-1 reduced the elevated OGT level which led to a drastic increase in sensitivity of PC-3 cells toward doxorubicin in comparison to doxorubicin alone. This finding provides important insight regarding alternative treatment strategies for effective management of cancer.
Introduction
Prostate cancer (PCa) is the second most common type of cancer worldwide and the fifth leading cause for cancer-related deaths among men. 1 Globally, estimated numbers of PCa diagnosed in the year 2018 were close to 1.3 million, which was 7.1% of all cancers diagnosed. 1 Depending on the stage or classification (androgen dependent or castrate resistant), different treatment options such as androgen deprivation therapy (ADT), radiation therapy, or chemotherapy may be considered for PCa management. 2 Once ADT becomes ineffective, due to the progression to castration resistance prostate cancer (CRPC), systemic chemotherapy becomes the main treatment option for management of the disease. 3 Mitoxantrone was the first approved chemotherapeutic agent for CRPC treatment. 4 In the year 2004, docetaxel (DOC) was approved in combination with prednisolone and became the first-line treatment for CRPC. 5 Although this combination offers modest survival of 7 to 8 months, patients who were pretreated with mitoxantrone and prednisolone show resistance to DOC therapy. 5 -7 More recently, cabazitaxel was approved for metastatic CRPC in patients previously treated with DOC and provides an average survival advantage of 2.4 months. 8 Other chemotherapy treatment options include paclitaxel, carboplatin, and doxorubicin as neoadjuvant chemotherapy. 9 Doxorubicin is less frequently used due to its dose-dependent toxicity and side effects on cardiovascular system. 9 However, the use of chemotherapy in PCa is limited by the development of chemoresistance and significant toxicities. 10 -12 Reports show that there are numerous intrinsic and extrinsic factors involved in chemoresistance but among them P-glycoprotein drug efflux, 13 inhibition of apoptosis, 14 hyperglycaemia, 15 and activation of oncogenic factors through alternative signaling pathways 16 play pivotal role in CRPC progression. Clinical management of PCa is challenging due to existence of advanced, metastatic, castration resistant, and chemoresistant PCa. There is an urgent need of not only newer therapeutic options but also novel combination strategies.
A growing body of evidence demonstrates that hyper-O-GlcNAcylation has become a general trait of cancer progression and metastasis. 17 -19 O-GlcNAcylation is a reversible post-translational modification in which addition of O-linked-β-D-N-acetylglucosamine (O-GlcNAc) from sugar donor uridine diphosphate N-acetylglucosamine (UDP-GlcNAc) onto the serine/threonine of numerous proteins is catalyzed by O-linked β-N-acetylglucosamine transferase (OGT) enzyme. 20 Kanwal et al reported that O-GlcNAcylation protects the human breast cancer derived Michigan Cancer Foundation-7 (MCF-7) cells from 4-OH-tamoxifen via reduction in expression of estrogen receptor α, which finally results in drug resistance. 21 Liu et al have shown in their study that activation of hexosamine biosynthetic pathway led to an increased level of O-GlcNAc, which enhanced the resistance of cancer cells toward anticancer molecules. This study also focused on the evaluation of combination of inhibitor of OGT enzyme and chemotherapeutic agent to enhance cytotoxicity and found that combination does effectively increases cell death in breast cancer cells. 22 Recently, Liu et al have reported that high level of O-GlcNAcylation results in reduced effectiveness of bortezomib in breast cancer cells, and a combination approach of O-GlcNAc inhibitor increased its effectiveness significantly. 23 OSMI-1 is a cell permeable molecule which potently inhibits OGT enzyme (half maximal inhibitory concentration [IC50] = 2.7 µM) and in turn leads to an effective reduction in O-GlcNAcylation. 24
Activity of OGT is essential for the stabilization of various oncogenic factors such as c-MYC which is one of the predominant factors contributing to the progression of malignancy in PCa. The OGT inhibitor disrupts this interaction by inhibition of OGT enzyme leading to suppression of cell cycle progression in PCa cells. 25,26 However, these studies primarily focused on the role of OGT inhibitor(s) on oncogenic factors and the signaling pathways involved but did not explore the benefits of the combination approach which is the prime focus of our research work. A combination of a chemotherapeutic agent with an enzyme inhibitor disrupts the key signaling pathways exploited by cancer cells for their proliferation and survival and presents a promising alternative approach to combat cancer by enhancing their cytotoxic potential. 12 Hence, the aim of this current study was to evaluate the effect of OGT enzyme inhibition on DOC- and doxorubicin-induced cytotoxicity in PCa cells in vitro. To achieve this, we pretreated PC-3 and WPMY-1 cells with the OGT enzyme inhibitor, OSMI-1, and then exposed these cells to various concentrations of chemotherapeutic agents. Lastly examined if any effects were correlated to changes in OGT levels.
Materials and Methods
Cell Culture and Reagents
PC-3 and WPMY-1 cells were obtained from American Type Culture Collection. PC-3 and WPMY-1 cells were grown in Ham F-12K medium and Dulbecco modified Eagle medium (Life Technologies), respectively. Each medium was supplemented with 0.1% gentamicin (Sigma-Aldrich) and 10% fetal bovine serum (Scientifix). Cells were incubated at 37 °C with 5% CO2 and 95% humidity. Doxorubicin HCl (DOX; cat# D558000) and DOC (cat# 2232-10) were purchased from Sapphire Bioscience (Redfern); OSMI-1, dimethyl sulfoxide (DMSO), and resazurin were purchased from Sigma-Aldrich.
Drug Formulations
Doxorubicin and DOC were dissolved in DMSO to make 10 mM stock solution. Stock solutions were stored in several aliquots at −20 °C to avoid repetitive freeze–thaw cycles. Doxorubicin HCl and DOC stock solutions were serially diluted using respective media to prepare drug working solutions. Working solutions were diluted 100 times with cell-culture media before treatment.
Resazurin Assay
The PC-3 and WPMY-1 cells were seeded at a density of 5 × 103 cells per well in a 96-well plate 1 day before treatment. Then it was treated with serially diluted concentrations (0.00001-100 µM) of doxorubicin, DOC, OGT inhibitor (OSMI-1), combination of both drug with OSMI-1 (5, 10, and 20 µM), and DMSO control (0.1% final vehicle concentration) and incubated for a period of 24 and 72 hours, respectively. Following incubation, treatment with resazurin solution (44 µM) was done and incubated further for another 4 hours at 37 °C with 5% CO2. Fluorescence intensity was measured using a Tecan Infinite 200Pro Microplate reader (excitation λ: 530 nm, emission λ: 590 nm; Tecan). Half-maximum growth inhibition (GI50) value was determined for all treatments using GraphPad Prism 7.02 Software.
Combination Index Analysis
PC-3 and WPMY-1 cells were seeded at a density of 5 × 103 cells per well in a 96-well plate. Fixed dose of OSMI-1 (5, 10, and 20 µM) was added in combination with doxorubicin or DOC (0.1, 1, 10, and 100 µM) simultaneously and incubated for 72 hours at 37 °C with 5% CO2. Cell viability was determined using resazurin assay. The percentage growth was calculated from growth inhibition graph. Values of fraction affected (FA) for each selected concentration for individual candidates were calculated using the formula, FA = 1 − (% growth/100). Calculated FA values were entered, and combination index (CI) was calculated using nonfixed ratio model with the help of Compusyn software (ComboSyn Inc) using the mass-attraction equation (Chou-Talalay method). 27
OGT ELISA Assay
PC-3 and WPMY-1 cells were seeded at a density of 1 × 105 cells per well in a 6-well plate. The next day cells were treated with doxorubicin (1 µM), OSMI-I, or a combination of both in addition to DMSO control (0.1% final vehicle concentration) for 24 and 72 hours, respectively. Lysates were prepared by lysing the cells with cell extraction buffer (cat# FNN0011; Life Technologies). The OGT enzyme level was measured using human OGT ELISA kit (Aviva System Biology) as per the manufacturer protocol. Absorbance was measured at 450 nm using a microplate reader (Tecan Infinite 200Pro, Tecan). Concentrations of enzyme were calculated from standard calibration curve.
Caspase-3/7 Fluorescence Assay
In brief, PC-3 and WPMY-1 cells were seeded at a density of 2 × 104 cells per well in a 96-well plate and incubated overnight. The next day cells were treated with doxorubicin (1 µM), OSMI-1, and a combination of both, followed by incubation for 72 hours at 37 °C with 5% CO2. Following incubation, caspase-3/7 fluorescence assay kit (Cayman chemical company) was used as per manufacturer protocol to determine the number of cells undergoing apoptosis. Fluorescence intensity was measured using a Tecan Infinite 200Pro-Microplate reader (excitation λ: 485 nm; emission λ: 535 nm; Tecan).
Statistical Analysis
All data are represented as the mean ± standard deviation, N = 3. One-way analysis of variance was conducted followed by Tukey multiple comparison test using the GraphPad Prism 7.02 Software. Differences between groups were considered statistically significant if P value was < 0.05. The statistical significance is denoted by asterisks (*P < 0.05; **P < 0.01; ***P < 0.001).
Results
Combination of Doxorubicin With OGT Inhibitor Enhances the Sensitivity of PC-3 Cells Toward Doxorubicin
Doxorubicin is the most frequently used chemotherapeutic agent for treatment of range of solid tumors. Its clinical utility is limited due to dose-dependent cardiotoxicity and chemoresistance. Enhancing efficacy of existing clinical drugs when used in combination with other agents is a rational approach to overcome the challenges involved in the clinical management of cancer. In this regard, aberrant O-GlcNAcylation is one of the various mechanisms responsible for resistance in cell death in PCa, and inhibition of OGT enzyme poses an effective strategy to overcome that resistance. To investigate this potential role of OGT inhibition, the combined antiproliferative effect in terms of GI50 of therapeutic agents (doxorubicin and DOC) and OSMI-1 as well as single agents was assessed using resazurin assay in PC-3 and WPMY-1 cells, respectively. It was found that doxorubicin alone has a GI50 value of 0.50 µM and 0.17 µM upon 72-hour treatment in PC-3 and WPMY-1 cells, respectively (Figure 1A and B). In PC-3 and WPMY-1 cells, DOC as a single agent has a GI50 value of 0.0005 µM after 72-hour treatment (Figure 1C and D). It was observed that there was a significant reduction in the GI50 values of doxorubicin when PC-3 cells were treated with different concentrations of OGT inhibitor (5, 10, and 20 µM OSMI-1) in a dose-dependent manner in comparison to doxorubicin alone as shown in Figure 1A. Combination of doxorubicin and OSMI-1 demonstrated almost 2.5-fold (0.183 µM for 20 µM OSMI-1) increase in the efficiency of cell death, whereas doxorubicin when treated in combination with 10 and 5 µM OSMI-1 for 72 hours reduced doxorubicin’s GI50 value to 0.233 and 0.353 µM, respectively, in PC-3 cells (Table 1). The GI50 values for the same combinations were also evaluated after 24-hour treatment (Supplemental Figure S1A) but no significant reduction in cell proliferation was seen. The GI50 values for the combination of DOC with OSMI-1 (5, 10, and 20 µM) were found to be 0.0227, 0.0216, and 0.0.0004 µM after 72-hour treatment, respectively (Table 1), whereas these combinations did not exhibit any significant reduction in cell proliferation at 24 hours in PC-3 cells (Supplemental Figure S1C). These data provide evidence that the combination approach with DOC was showing increase or no significant change in GI50 when compared to DOC alone. OSMI-1 showed GI50 value of >100 and 40 µM after 24 and 72-hour (Figure 1A and Supplemental Figure S1A) time point, respectively, in PC-3, whereas in WPMY-1 cells it was found to be ≥100 and ≈20 µM after 24 and 72 hours, respectively (Figure 1B and Supplemental Figure S1B). As shown in Supplemental Figure S2, almost all the selected concentrations of OSMI-1 demonstrate a GI50 >100 µM after 24-hour treatment in PC-3 cells, indicating that OSMI-1 alone has no significant antiproliferative effect when used alone. When the treatment time is prolonged to 72 hours, 10 and 20 µM of OSMI-1 demonstrate ≈12% and up to 33% reduction in cellular proliferation. These results provide evidence that OSMI-1 enhances the sensitivity of PC-3 cells toward doxorubicin, whereas that is not the case with DOC. Therefore, further experiments were focused on investigating the combination effects of OSMI-1 and doxorubicin.
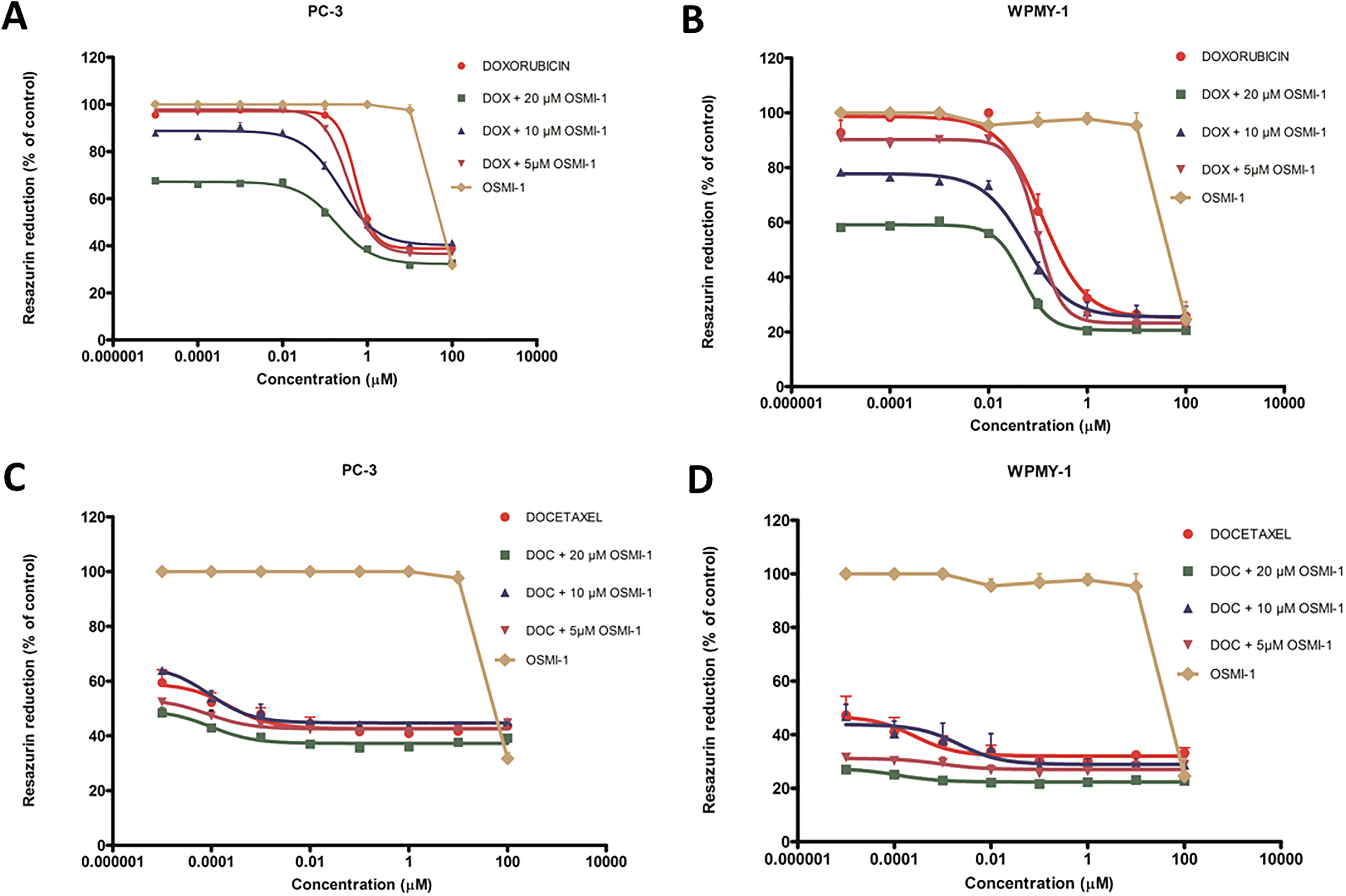
Combination of doxorubicin with OSMI-1 reduces GI50 and increases its cell killing efficiency. A and B, PC-3 and WPMY-1 cells were treated with doxorubicin alone (0.00001-100 µM), OSMI-1 alone, and in combination for 72 hours, respectively. Cell viability was assessed using the resazurin assay. C and D, PC-3 and WPMY-1 cells were treated with docetaxel alone, OSMI-1 alone, and in combination for 72 hours, respectively. Cell viability was assessed using the resazurin assay. Data represent the mean ± SD, N = 3, *P < 0.05, ***P < 0.0001. GI50 indicates half-maximum growth inhibition; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor; SD, standard deviation.
GI50 of Doxorubicin and Its Combination With OSMI-1 at 72 hours in PC-3.
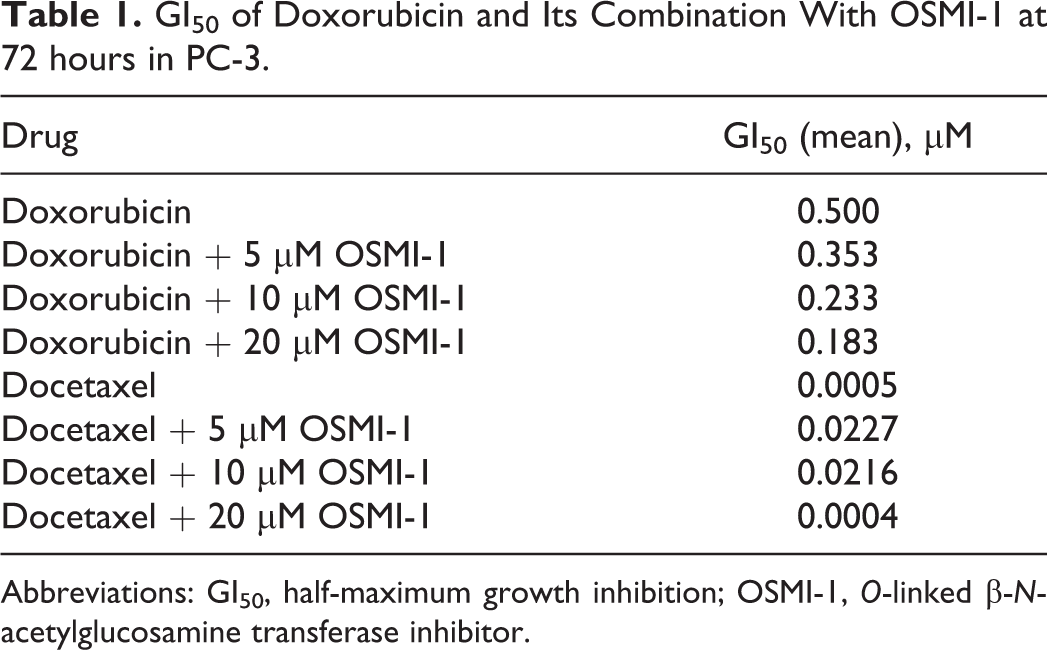
Abbreviations: GI50, half-maximum growth inhibition; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor.
Evaluation of CI and Synergism for the Combination of Doxorubicin With OGT Inhibitor
Combination of doxorubicin and OSMI-1 (at 5 and 10 µM) demonstrated a significant reduction in GI50 without much effect on the cell viability in PC-3 cells. The increased sensitization of PC-3 cells toward the combination of doxorubicin and OSMI-1 prompted us to determine whether the effect is synergistic, additive, or antagonistic based on the assessment of CI (CI >1, 1, <1, respectively). 28 As 20 µM of OSMI-1 had a significant effect on the cell viability, combination of 5 and 10 µM OSMI-1 and doxorubicin at 0.1, 1, 10, and 100 µM was selected for further evaluation in PC-3 cells. As seen in Table 2, all the combinations of doxorubicin with 5 µM OSMI-1 exhibit a synergistic effect on cell killing. 27 Additionally, it is also observed that as the ratio of drug to OGT inhibitor increases (0.02-20), the CI value also increases (0.02-0.87). A similar trend was noted for 10 µM OSMI-1 and with the increment of the dose of doxorubicin by 10×, the CI value increased proportionally (Table 2). Data from Table 2 represent that at low dose of doxorubicin, combination with OSMI-1 exhibit a better effect in comparison to higher concentrations. The CI value for the combination of DOC with OSMI-1 demonstrates antagonistic effect in PC-3 cells (Table 3) at high concentrations. Combination of doxorubicin with OSMI-1 also shows synergism in normal stromal cells (Table 4). To strengthen that the combination of doxorubicin and OSMI-1 would have added benefit, CI values were calculated and strong synergy was observed along the magnitude of cell killing in PC-3 cells, indicating the potential application of combination approach.
Combination Index of Doxorubicin and OSMI-1 in PC-3 Cells.
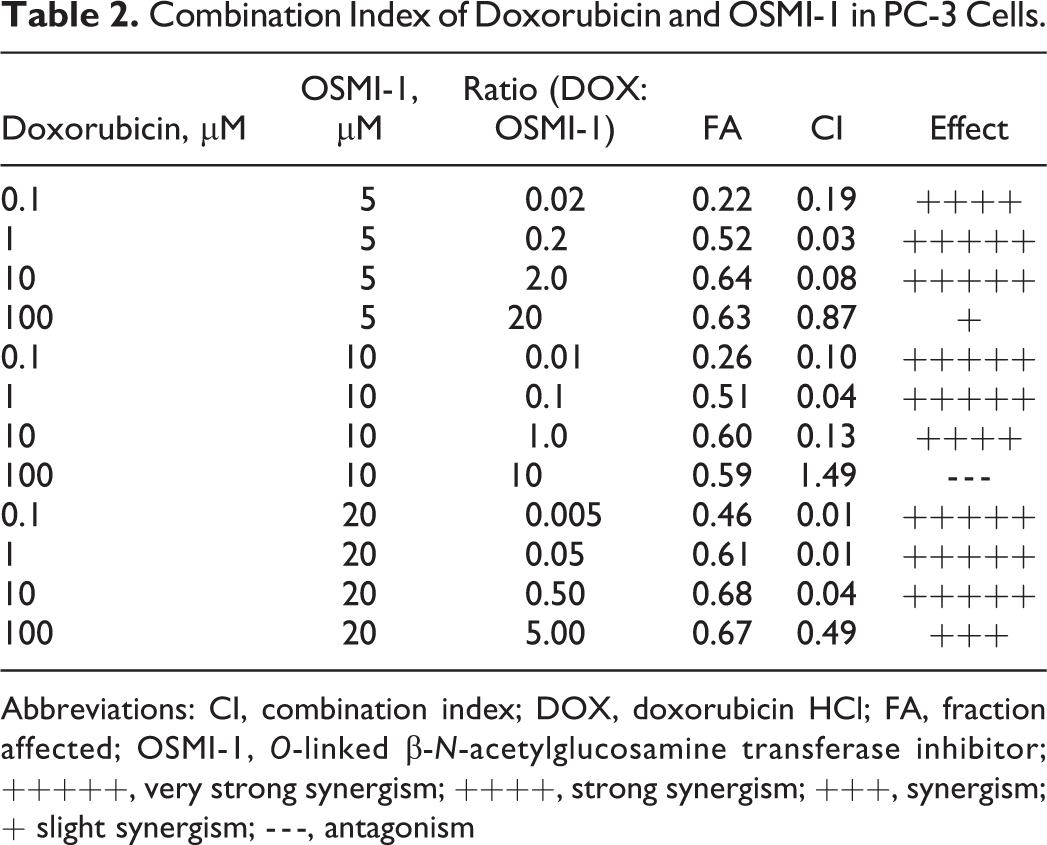
Abbreviations: CI, combination index; DOX, doxorubicin HCl; FA, fraction affected; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor; +++++, very strong synergism; ++++, strong synergism; +++, synergism; + slight synergism; - - -, antagonism
Combination Index of Docetaxel and OSMI-1 in PC-3 Cells.
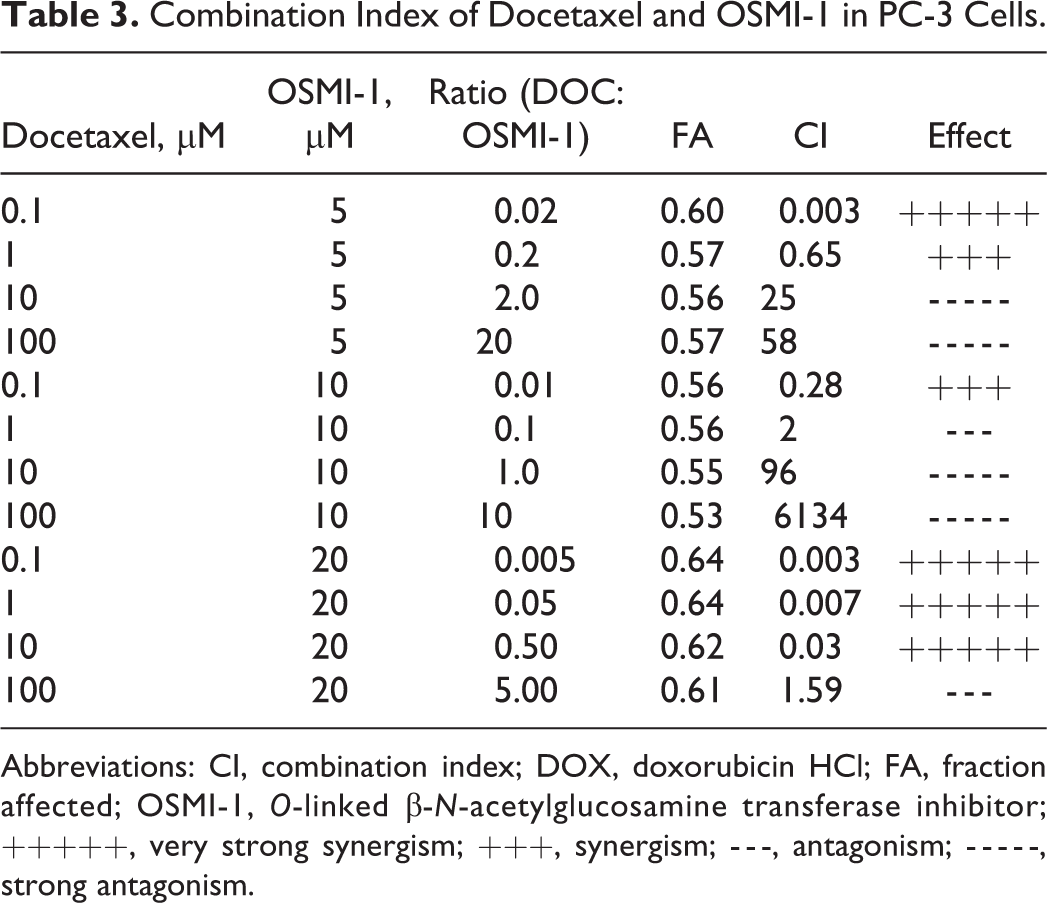
Abbreviations: CI, combination index; DOX, doxorubicin HCl; FA, fraction affected; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor; +++++, very strong synergism; +++, synergism; - - -, antagonism; - - - - -, strong antagonism.
Combination Index of Doxorubicin and OSMI-1 in WPMY-1 Cells.
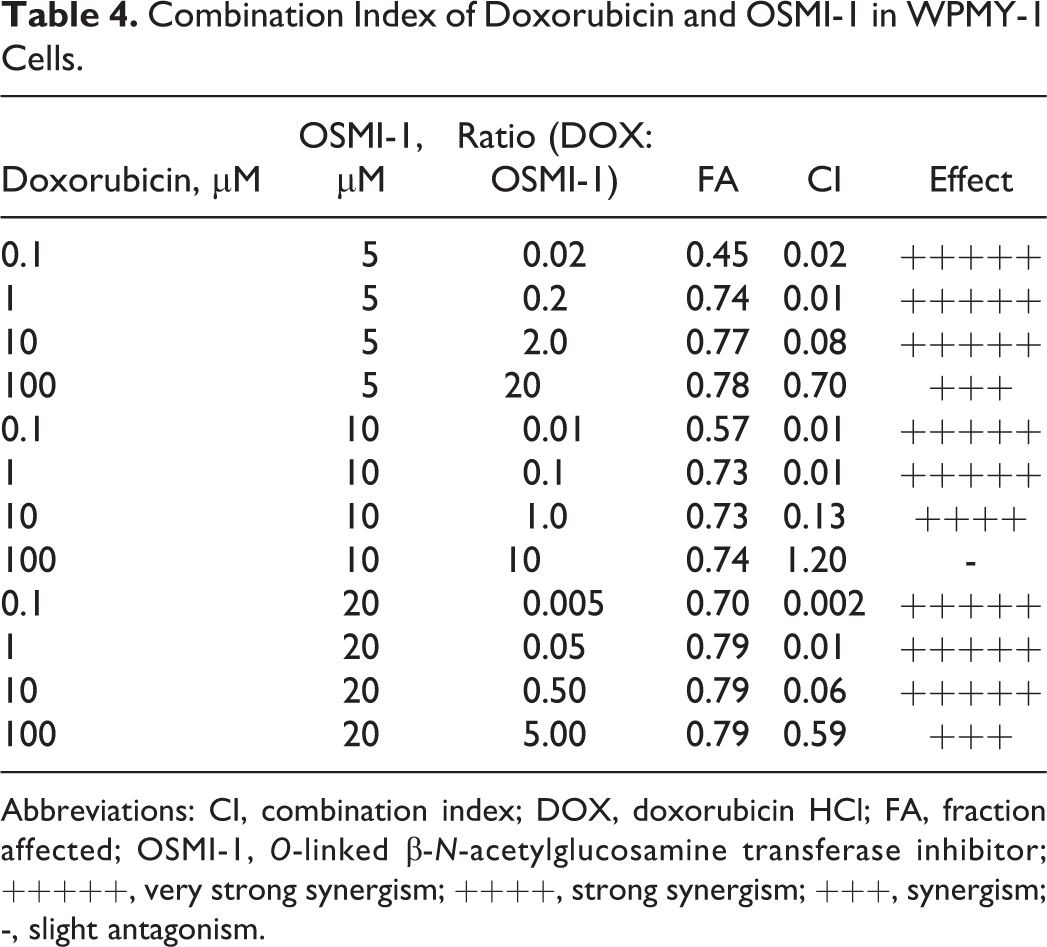
Abbreviations: CI, combination index; DOX, doxorubicin HCl; FA, fraction affected; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor; +++++, very strong synergism; ++++, strong synergism; +++, synergism; -, slight antagonism.
Effect of OGT Inhibitor OSMI-1 Alone and Its Combination With Doxorubicin on OGT Enzyme Level
A combination of doxorubicin with OGT inhibitor demonstrated a significant reduction in growth inhibition value and exhibited strong synergistic effect which led us to further investigate its effect on OGT enzyme levels. PC-3 and WPMY-1 cells were treated with doxorubicin alone, OSMI-1 alone, and in combination with OSMI-1 and doxorubicin for 24 and 72 hours, respectively. After 24-hour treatment with 10 and 20 µM OSMI-1 alone, in PC-3 cells OGT level was measured to be 0.14 and 0.08 ng/mL, whereas respective combination with doxorubicin and control showed 0.12, 0.10, and 0.13 ng/mL, respectively. The OGT level in WPMY-1 cells after 24 hours was found to be 0.08 ng/mL for control, which indicates that there is a significant difference in OGT enzyme level in cancerous cells (PC-3) and noncancerous prostate cells (WPMY-1). No significant difference in OGT enzyme level was observed between control and different treatment combinations at 24 hours in both the cell types (Figure 2A and B). After treatment for 72 hours, OGT level was found to be 0.11, 0.08, and 0.08 ng/mL at 5, 10, and 20 µM dose of OSMI-1, respectively, in PC-3 cells. Combination of doxorubicin and OSMI-1 (5, 10, and 20 µM) reduced OGT level to 0.10, 0.08, and 0.07 ng/mL, respectively, in comparison to control value of 0.22 ng/mL, which highlights the fact that there is a significant drop in enzyme level upon treatment with the aforementioned combinations. Figure 2C depicts a concentration-dependent increase in OGT enzyme inhibition in comparison to control in PC-3 cells. OSMI-1 and all of its combinations did not show any significant difference in OGT levels against control in WPMY-1 cells even after 72-hour treatment (Figure 2D). Results of this study suggests that combination of doxorubicin with OGT inhibitor significantly reduces OGT enzyme level and sensitizes the cancer cells toward the cytotoxic effects of chemotherapeutic agents.
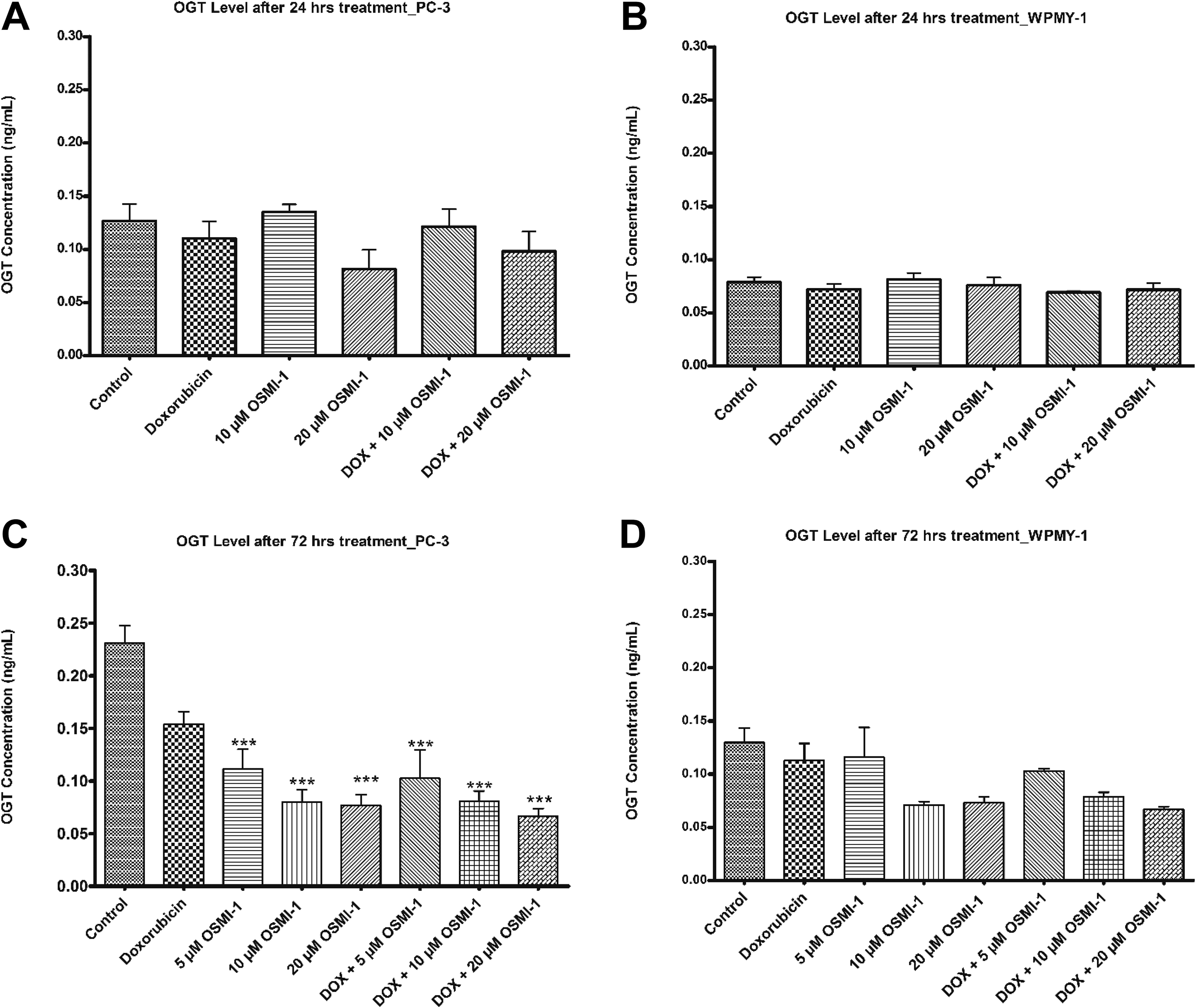
OSMI-1 and its combination with doxorubicin significantly reduce OGT enzyme level. A and B, PC-3 and WPMY-1 cells were treated with doxorubicin (GI50), OSMI-1 (10 and 20 µM), and combination of doxorubicin and OSMI-1 for 24 hours, respectively. C and D, PC-3 and WPMY-1 cells were treated with doxorubicin (GI50), OSMI-1 (5, 10, and 20 µM), and combination of doxorubicin and OSMI-1 for 72 hours. Cells were lysed using lysis buffer. Lysates were analyzed using OGT ELISA kit (Aviva system biology). The data present the mean ± SD, N = 3, **P < 0.01, ***P < 0.0001. GI50 indicates half-maximum growth inhibition; OSMI-1, O-linked β-N-acetylglucosamine transferase inhibitor.
Concomitant Treatment of Doxorubicin With OGT Inhibitor Induces Apoptosis in PC-3 Cells
Caspase 3/7 was used as an index of apoptosis in this study. Here, we demonstrate that the combination of OSMI-1 with doxorubicin in PC-3 cells induced significant apoptosis after 72-hour treatment (P values <0.0001 and <0.001 compared to control and doxorubicin shown in Figure 3A), whereas doxorubicin alone failed to achieve the same amplitude of caspase 3/7 activation. Among the combinations tested, increment in the dose of OGT inhibitor resulted in a greater induction of caspase 3/7 activation in the PC-3 cells. As shown in Figure 3B, combination of OSMI-1 with doxorubicin also increased the number of cells undergoing apoptosis in WPMY-1 cells. This finding highlights the fact that combination with OGT inhibitor versus doxorubicin alone leads to a greater number of cancer cells committing to apoptosis.
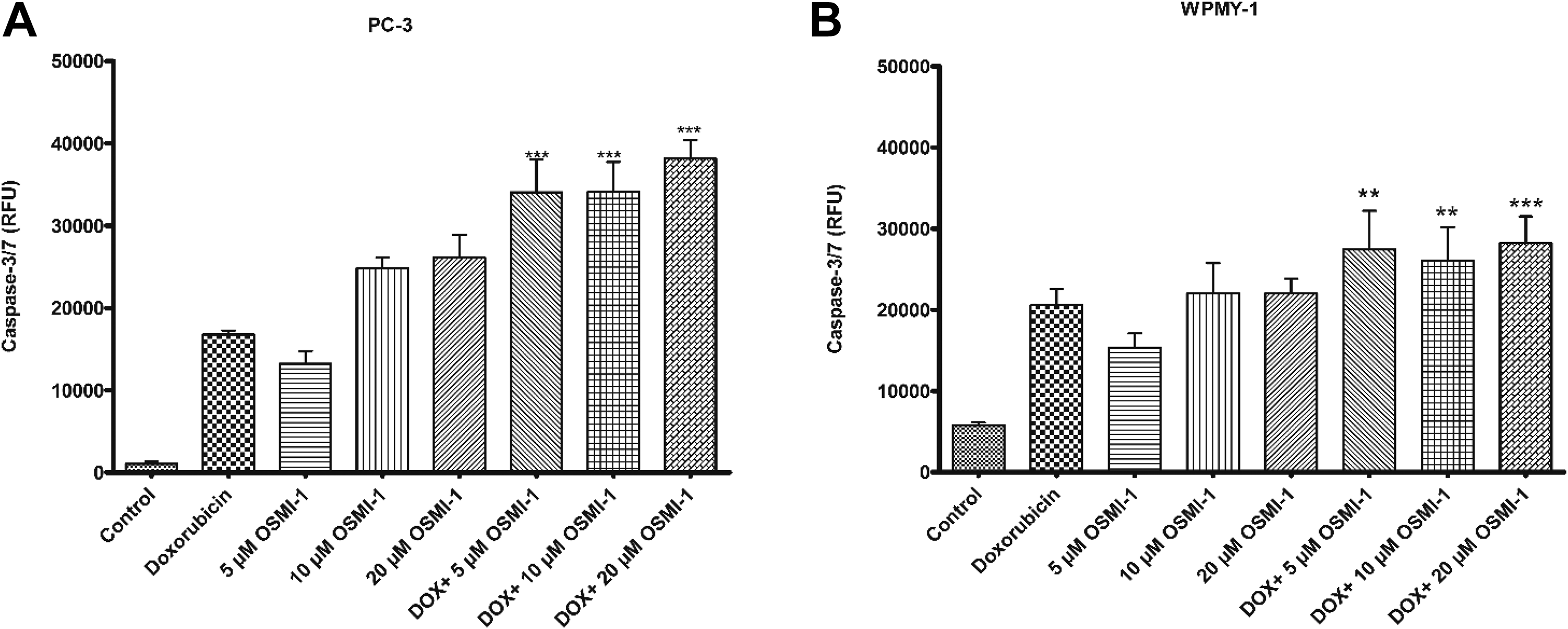
Combination of doxorubicin with OGT inhibitor induces significant apoptosis in prostate cancer cells. A and B, PC-3 and WPMY-1 cells were treated with doxorubicin (GI50), OSMI-1 (5, 10, and 20 µM), and combination of doxorubicin and OSMI-1 for 72 hours, respectively. Activation of caspase 3/7 was analyzed using caspase 3/7 fluorescence kit (Cayman chemicals) as per manufacturer protocol. The data present the mean ± SD, N = 3, *P < 0.05, **P < 0.01, ***P < 0.0001. GI50 indicates half-maximum growth inhibition; OGT, O-linked β-N-acetylglucosamine transferase inhibitor; RFU, relative fluorescence units; SD, standard deviation.
Discussion
Reduction in cytotoxicity is a major factor limiting the effectiveness of chemotherapy in the treatment of PCa. 10 Therefore, there is an urgent need to identify novel strategies to overcome this limitation and improve the effectiveness of current treatments. The aim of our study was to evaluate the impact of OGT enzyme inhibition on DOC- and doxorubicin-induced cytotoxicity in PCa cells and to explore the benefits of this combination approach. Our study demonstrated that PC-3 cells were less sensitive toward treatment with doxorubicin in comparison to DOC. However, DOC with OSMI-1 was not as effective as doxorubicin when combined and has not exhibited clear synergistic trend at selected concentrations. This might be due to the direct or indirect interference of mechanistic pathways involving any oncogenic factor or high concentration level and remains to be further elucidated. In case of DOC, lowering of concentration range to nanomolar might provide more information with respect to interaction of DOC and OSMI-1. Combination of OGT inhibitor (OSMI-1) with doxorubicin significantly reduced its GI50 value which might reduce the dose-dependent cardiotoxicity. Combination index was analyzed for the proposed combinations, and a strong synergistic effect was seen on the antiproliferative activity of doxorubicin when it was concomitantly treated with OSMI-1 in PC-3 and WPMY-1 cells. This results also point toward the lack of selectivity of the combinations tested toward PC-3 cells over normal cells. To understand the impact OGT inhibition has on the reduction of GI50 value of doxorubicin, OGT level was measured in PC-3 and WPMY-1 cells. Upon measurement of OGT enzyme, it was observed that cancerous cells showed significantly high level of OGT enzyme in comparison to normal prostate cells. 25 Our experimental findings suggest that OSMI-1 is capable of significantly reducing OGT level alone as well as in combination with doxorubicin in PC-3 cells in a dose- and time-dependent manner. We also revealed that combination of doxorubicin with OSMI-1 enhanced the induction of apoptosis in PC-3 cells in comparison to doxorubicin alone in a dose-dependent manner. The observation of this study agrees with the findings of Liu et al, where they have reported that combination of doxorubicin with OGT inhibitor has significant impact on cell death with improved effectiveness in MCF-7 breast cancer cells. 22 Different OGT inhibitors have shown significant effect on restoring sensitivity of cancer cells toward chemotherapeutic agents in numerous cancer types. 21 -23 Hyper-O-GlcNAcylation/elevated OGT level is considered as characteristic hallmark for cancer development and progression. 19,29,30 Reduced level of OGT enzyme either by limiting gene expression (RNAi) or by the use of a pharmacological inhibitor of OGT enzyme leads to reduction in c-MYC level in PCa cell lines. 26 Inhibition of OGT activity destabilizes c-MYC and thus supports metabolic reprogramming in prostate tumor cells. 31 Our findings are in consistent with literature that aberrant high levels of OGT enzyme induce resistance in cancer cells toward anticancer agents and provides protective mechanisms against drug-induced cell death via apoptosis.
Conclusion
In a nutshell, our research work identifies the effective combinations of doxorubicin with OSMI-1 that has synergistic effect in PCa cells. This might prove to be an effective strategy for effective management of pan-cancers. However, further in-depth investigation on underlying mechanism is warranted.
Supplemental Material
Supplemental_Material - Investigating the Impact of OGT Inhibition on Doxorubicin- and Docetaxel-Induced Cytotoxicity in PC-3 and WPMY-1 Cells
Supplemental_Material for Investigating the Impact of OGT Inhibition on Doxorubicin- and Docetaxel-Induced Cytotoxicity in PC-3 and WPMY-1 Cells by Vivek Makwana, A/Prof Shailendra-Anoopkumar Dukie and Santosh Rudrawar in International Journal of Toxicology
Footnotes
Author Contributions
S. Rudrawar contributed to conception and design, contributed to analysis, drafted manuscript, and critically revised manuscript; S. Dukie contributed to conception and design, contributed to analysis and interpretation, drafted manuscript, and critically revised manuscript; V. Makwana contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support to SR from an Australian Research Council—Discovery Early Career Research Award (DE140101632) is gratefully acknowledged. VM is supported PhD scholarship (GUIPRS and School of Pharmacy and Pharmacology).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
