Abstract
This study aimed to evaluate the in vitro cytotoxic effects of locally produced processed natural coral (PNC) using human osteoblasts (HOS). Cytotoxicity was not observed when HOS cells were cultured with PNC, as assessed by (3-(4,5-dimethylthiazol-2-yl)-2-5-diphenyl tetrazolium bromide; MTT) and Neutral Red (NR) assays at concentration up 200 mg/mL for up to 72 hours. Flow cytometry (FCM) analysis showed that PNC (200 mg/mL) did not decrease viability of HOS cells after 48 and 72 hours of treatment. In a cell attachment study, the HOS cells attached to the edge of the PNC disc, and later grew into the pores of the PNC disc. All results from these studies indicate that locally produced PNC material is noncytotoxic and favors the growth of HOS cells.
Introduction
Virtually every operative day, orthopedists, neurosurgeons, maxillocraniofacial surgeons, and periodontists need to fill the defects in bone or augment deficient bone. When the defect is small, an autologous bone graft is the best solution, but in larger defects, the addition of nonautologous bone graft and biomaterials are a necessity. Recent advances in orthopedic and maxillocraniofacial surgery can be attributed to the revolution in biomaterials. During the last decade, a large number of biomaterials have been proposed as artificial bone filler for repairing bone defects. 1 Bone formation requires a physical structure to which osteoblasts can adhere. Therefore, the concept of using porous devices composed of biocompatible materials was conceived. Osteogenesis, osteoinduction, and osteoconduction are the essential elements of bone regeneration, along with the final bonding between host bone and grafting material which is called osteointegration. 2
Biomaterials or scaffolds for osteogenesis should mimic bone morphology, structure, and function in order to optimize integration into surrounding tissue. Bone is a structure of hydroxyapatite (Ca10(PO4)6(OH)2) crystals deposited within an organic matrix in which 95% is type 1 collagen. The morphology is composed of trabecular bone, which creates a porous environment with 50% to 90% porosity. Scaffolds for bone regeneration should meet certain criteria to serve the skeletal functions including mechanical properties similar to those bones at the repair site, biocompatibility, and biodegradability appropriate with remodelling. 3 Scaffolds serve primarily as osteoconductive moieties, because new bone is deposited by creeping substitution from adjacent living bone.
Research on natural coral as a bone substitute has been reported in many experimental studies. It has been proven to be biocompatible, biodegradable, and has not been found to cause any inflammatory responses. 4 Coral is made of calcium carbonate (98%-99%) in the form of aragonite, with the trace elements and amino acids. 5 It has been used as a biomaterial for bone replacement for several reasons; the material simplifies the surgical procedure, does not require harvesting of autologous bone, and transmission of infections such as AIDS and hepatitis can be avoided with certainty. Furthermore, coral has porous architecture, high-compression breaking stress, and resorbability. It has been reported that the porosity and 3-dimensional structure of coral implant encourages bony ingrowth. 6
Natural coral is a bone graft substitute, which has been widely used in maxillofacial, orthopedic, otorhinolaryngology, and periodontal surgery. The capacity of coral to disappear and be substituted by new bone distinguishes it from nonresorbable materials extensively used in these surgeries. An optimal clinical utilization of coral requires a thorough knowledge of factors influencing resorption, particularly regarding the interface between implant and connective tissue, which is larger than the surface in contact with the bone.
7
Coral mineral has had considerable success considering its porous structure (which ranges from 150-500 μm), its similarity to cancellous bone, and also because it is one of the limited number of materials that will form chemical bonds with bone and soft tissue in vivo.
8
Studies indicate that a favorable pore size and microstructural composition are important factors facilitating in-growth of fibrovascular tissue or bone from the host and pore interconnection sizes are of utmost importance when hard and soft tissue in-growth are involved.
8
The structure of the commonly used natural coral
All newly developed biomaterials must fulfill stringent criteria laid out by government authorities and international agencies, such as the United States Food and Drug Administration (FDA) and International Organization for Standardization (ISO), before receiving approval for clinical application. 11 In practice, standard cell-based toxicity assays are performed in vitro and high-risk materials are removed at this early stage. This method has the advantages of simplicity, good sensitivity and reproducibility, and is widely used in the initial evaluation of biocompatibility of biomaterials. 11 In this study, an in vitro cytotoxicity testing was carried out on PNC using a human osteoblast (HOS) cell line.
Materials and Methods
Biomaterial
The biomaterial used in this study was dead coral of
Cell Line
The cell line used in this study was HOS (CRL-1543) obtained from the American Type Culture Collection (ATCC, USA). The cells were cultured in Dulbecco’s-Modified Eagle’s Medium (DMEM; GIBCO, USA) containing 10% fetal bovine serum (FBS; GIBCO, USA), and 1% penicillin-streptomycin (GIBCO, USA). The cultures were incubated at 37°C with 5% CO2 in 25 cm2 flask (NUNC, Denmark). The cells were cultured until they reached confluence as observed under inverted microscope (Carl Zeiss, Germany). In general, cells from 3 to 5 passages were used in the all experiments.
Cytotoxicity
In the present study, in vitro cytotoxicity tests were carried out on the processed natural coral (PNC) material using extraction/indirect and direct methods as per ISO 10993-5. 12 This is carried out by extracting the material in growth medium with serum by incubation in a 5% CO2 incubator at 37°C for a minimum period of 24 hours. Then, the extraction medium is introduced to the cells for further cytotoxicity evaluation. The indirect test is carried out based on the compounds possibly released from the material, 13 whereas, for the direct method, the cells are evenly distributed over the surface of the material and incubated in a 5% CO2 incubator at 37°C for a minimum of 24 hours. The cytotoxicity is evaluated along with the cells attached to the material. Extraction/indirect and direct methods allow both qualitative and quantitative assessment of cytotoxicity. 12
Cytotoxicity via Indirect Method
Sterilized PNC material in the form of granules (size, 0.5-1 mm) and powder (1-50 μm) were added into separate 50-mL bottles containing 10 mL of growth medium. According to ISO 10993-12, 14 the concentration of the material for the extraction method should range from 100 to 200 mg/mL. Hence, the maximum concentration of 200 mg/mL was used in the current study, which was serially diluted to 6.25 mg/mL. The 50-mL bottles were then incubated in a 5% CO2 incubator at 37°C for 48 hours without agitation. At the end of the incubation period, PNC materials were removed and the extracts were used. Cells treated with 70% ethanol served as a positive control, and the negative control consisted of cells without addition of materials for both the (3-(4,5-dimethylthiazol-2-yl)-2-5-diphenyl tetrazolium bromide; MTT) and the Neutral Red (NR) assays.
3-(4,5-dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium bromide; MTT Assay
After incubation for 48 hours, extraction medium was filtered through a 0.22-μm millipore membrane filter (Schleicher, Germany) and 100 μL of extraction medium (200 mg/mL) was serially diluted up to 6.25 mg/mL in a 96 well-plate (Falcon, USA) containing 1 × 104 HOS cells/well. Cells were cultured in 96-well plates and incubated in a 5% CO2 incubator for 72 hours at 37°C. Then, 10 μL of MTT (Calbiochem, Germany) at a concentration of 5 mg/mL was added to the culture medium and further incubated for 4 hours in a 5% CO2 incubator at 37°C to allow the uptake of dye by the surviving cells. After this, the MTT-containing media was removed and the cells were lysed with 100 μL of dimethyl sulfoxide (DMSO). Absorbance at 640 nm (A640) was measured with Enzyme-Linked Immunosorbent Assay (ELISA) reader (Tecan, Switzerland).
Neutral Red Assay
After incubation for 48 hours, extraction medium was filtered through a 0.22-μm millipore membrane filter (Schleicher, Germany) and 1 mL of extraction medium (200 mg/mL) was serially diluted to 6.25 mg/mL in a 24-well plate (Falcon, USA) containing 3 × 104 HOS cells/well. Cells were cultured in 24-well plates and incubated in a 5% CO2 incubator for 72 hours at 37°C. The cells were incubated without changing the growth medium. After incubation, 1 mL of NR (Sigma, USA) (0.02%) was added and further incubated for 2 hours in a 5% CO2 incubator at 37°C to allow the uptake of dye by the surviving cells. Then, the NR-containing media was removed and the cells were rinsed with 1 mL of phosphate-buffered saline (PBS). One mL of 1% sodium dodecyl sulfate (SDS) was added to lyse the cells. Absorbance at 540 nm (
Apoptosis Study
Extraction medium was filtered through a 0.22-μm millipore membrane filter (Schleicher, Germany) and 1 mL of extraction medium (200 mg/mL) was added into the 24-well plate (Falcon, USA) containing 3 × 104 HOS cells/well. Cells grown without any addition of material were used as the negative control. The cultures were incubated in a 5% CO2 incubator for 72 hours at 37°C. After the incubation period, the cells were washed twice with PBS and then 2 mL of trypsin was added to detach the cells. The cells were then removed and placed in 10-mL centrifuge tubes containing 5 mL of fresh growth medium and centrifuged at 1000 rpm for 5 minutes. The supernatant was removed and the cells were counted using hemocytometer chamber (Neubauer, Germany). The cell pellet (1 × 106 cells) was resuspended in 100 μL of Annexin-V-FLOUS labeling solution (Roche, Germany) and incubated at room temperature in the dark for 10 minutes. The cells were then subjected to flow cytometry (FCM) analysis using 488 nm excitation and a 515 nm bandpass filter for fluorescein detection. There were 3 replicates for a single concentration (200 mg/mL), and the experiments were repeated twice to calculate the mean value of viable and apoptotic cells.
Cytotoxicity via Direct Method
Processed natural coral discs (10 mm diameter and 1 mm thickness) were placed into 24-well plates, and 1 mL of growth medium was added and incubated for 1 hour to humidify the material. Then, 1 mL of growth medium containing 3 × 104 HOS cells was added. Cultured cells without any material added were used as a negative control, and cultured cells with rubber latex were used as a positive control. The cultures were incubated in a 5% CO2 incubator at 37°C for 1, 24, 72, and 168 hours without changing the growth medium. Cells were observed under an inverted light microscope (Carl Zeiss, Germany). After incubation, 1 mL of NR (0.02%) was added and further incubated for 2 hours in a 5% CO2 incubator at 37°C to allow the uptake of dye by the surviving cells. Then, the NR was removed and the cells were rinsed with 1 mL PBS. Sodium dodecyl sulfate (1%; 1 mL/well) was added to lyse the cells. Absorbance at 540 nm (
Cell Attachment Using Inverted Microscope
Processed natural coral discs (10 mm diameter and 1 mm thickness) were placed into 24-well plates and 1 mL of growth medium was added and incubated for 1 hour to humidify the material. After 1 hour of incubation, the PNC discs were seeded with 3 × 104 HOS cells. Cultured cells without any addition of material were used as a negative control, and cultured cells with rubber latex were used as the positive control. The cultures were incubated in a 5% CO2 incubator for 72 hours at 37°C without changing the growth medium. The test was carried out in 3 replicates and the whole experiment was repeated twice. The experiments were evaluated based on the adherence of cells to the material observed under inverted light microscope (Carl Zeiss, Germany).
Cell Attachment Using Scanning Electron Microscopy
The adherence of cells to the PNC material was also observed under scanning electron microscopy (SEM). Processed natural coral discs (10 mm diameter and 1 mm thickness) were placed into 24-well plate and 1 mL of growth medium was added and incubated for 1 hour to humidify the material. The excess medium was removed and the PNC discs were seeded with 3 × 104 HOS cells/well and incubated in a 5% CO2 incubator for 72 hours. Media were then removed and the cells were washed twice with PBS. The cells were then fixed in formaldehyde and buffered osmium tetroxide solution (1%) and subsequently dehydrated in ethanol. The PNC disc and Thermanox film (negative control) were coated with gold (Sputter coater, Germany) and examined under SEM.
Data Analysis
Statistical analysis was carried out using SPSS software version 12.0.1 and analyzed using one-way analysis of variance (ANOVA). Statistical significance was defined as
Calculation of EC50
The EC50 for the PNC material and positive control (70% ethanol) was calculated by nonlinear regression analysis using GraphPad Prism 5 software.
Results
Cytotoxicity Study by Extraction Method
In order to evaluate the cytotoxicity effect of PNC material, tests on extract/indirect contact were carried out as per ISO 10993-5. 12 The proliferation rate was evaluated using MTT, NR assay, and FCM analysis.
Proliferation Study on Different Particle Sizes of PNC by MTT Assay
The cell proliferation percentage of HOS cells on 2 different particle sizes of PNC, positive control (70% ethanol), and negative control (cells without material added) using MTT assay is shown in Table 1 . The highest percentage of cell proliferation for both the PNC granules (100.6) and the PNC powder (98.5) were noted at 200 mg/mL concentration, whereas the lowest percentage of cell proliferation was seen at a concentration of 25 mg/mL for both the PNC granules (78.1) and the PNC powder (85.9). The EC50 for PNC could not be determined even at the highest concentration of 200 mg/mL, whereas, for the positive control (70% ethanol), the EC50 was calculated to be 25.27 mg/mL. The results revealed that both PNC granules and PNC powder were not cytotoxic by MTT assay.
Cell Proliferation Percentage of Human Osteoblasts (HOS) Cells Exposed to the Extracts of Processed Natural Coral (PNC) Granules, PNC Powder, Positive and Negative Controls Using (3-(4,5-dimethylthiazol-2-yl)-2-5-diphenyl tetrazolium bromide (MTT) Assay
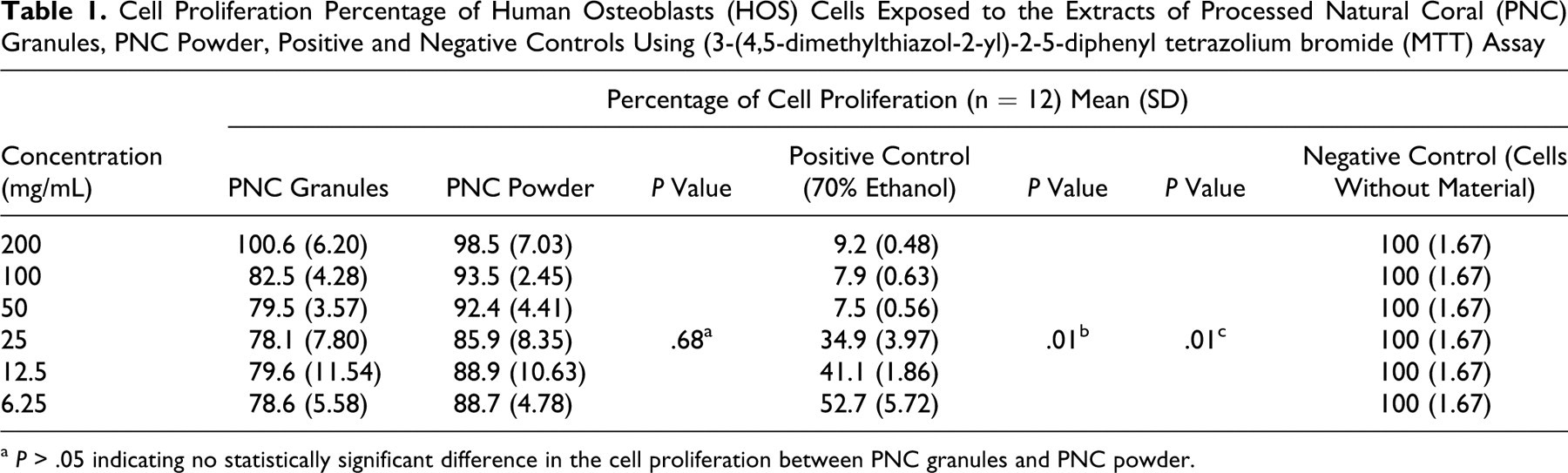
a
Proliferation of HOS Cells on 2 Different Particle Sizes of PNC by NR Assay
The cell proliferation percentage of HOS cells on 2 different particle sizes of PNC, positive control (70% ethanol), and negative control (cells without material added) using NR assay is shown in Table 2 . The highest percentage of cell proliferation for the PNC granules (103.2) was noticed at a concentration of 200 mg/mL, whereas for the PNC powder (97.0), it was noticed at a concentration of 6.25 mg/mL. The lowest percentage of cell proliferation for the PNC granules (80.1) was noticed at a concentration of 25 mg/mL and for the PNC powder (86.1) at 50 mg/mL. The EC50 for the positive control (70% ethanol) was calculated to be 12.80 mg/mL. Even at the highest concentration of 200 mg/mL of PNC, the EC50 could not be determined. This test showed that both PNC granules and powder were not cytotoxic to the HOS cells.
Cell Proliferation Percentage of Human Osteoblasts (HOS) Cells Exposed to the Extract of Processed Natural Coral (PNC) Granules, PNC Powder, Positive and Negative Controls by Neutral Red Assay
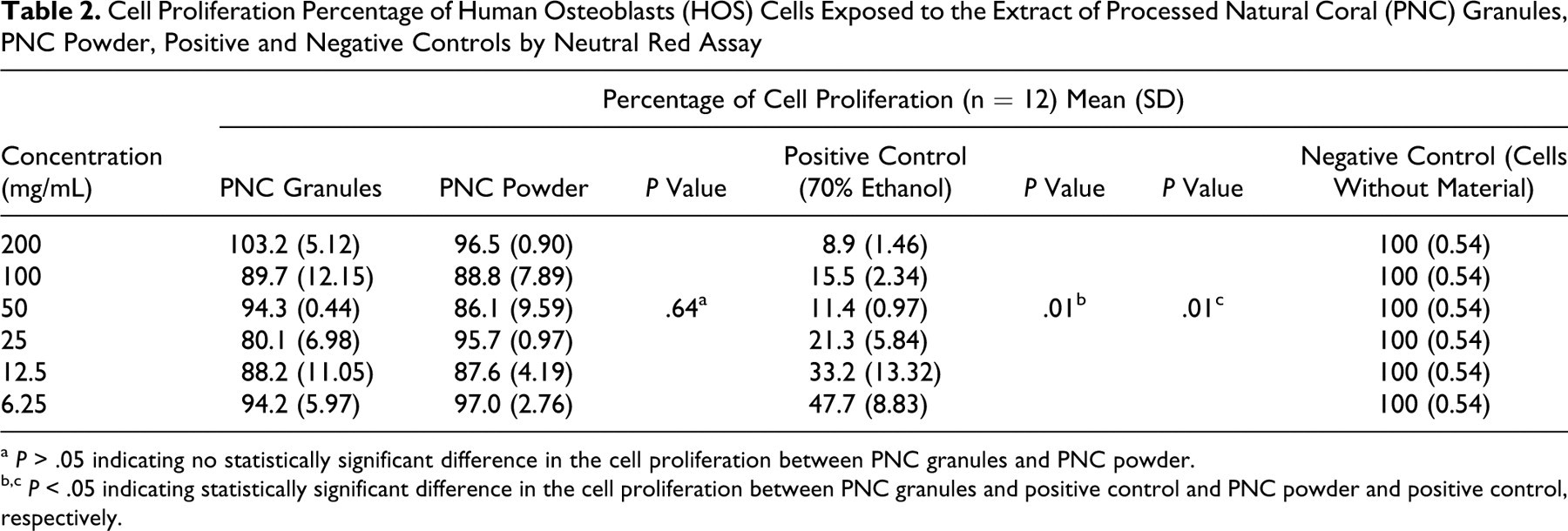
a
b,c
Apoptosis Study Using FCM
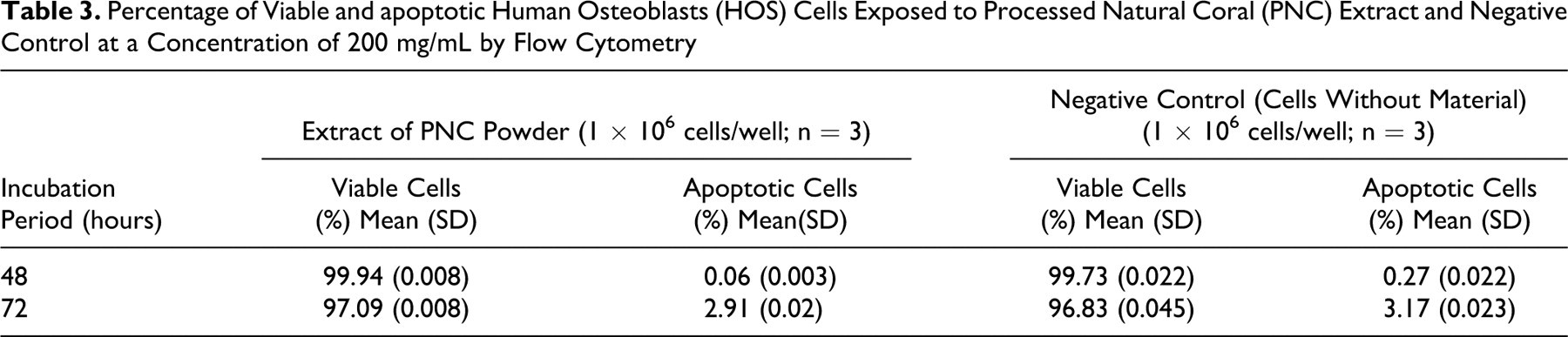
The percentage of viable and apoptotic HOS cells exposed to the extract of PNC powder and negative control (cells without material added) is presented in Table 3 . The percentage of viable cells was highest at an incubation period of 48 hours (99.94) with the extract of PNC powder compared to the incubation period of 72 hours (97.09).
Percentage of Viable and apoptotic Human Osteoblasts (HOS) Cells Exposed to Processed Natural Coral (PNC) Extract and Negative Control at a Concentration of 200 mg/mL by Flow Cytometry
Cytotoxicity Study by Direct Method
In order to evaluate the cytotoxicity effect of PNC material, tests on direct contact were carried out as per ISO 10993-5. 12 The cell proliferation was evaluated using NR assay and microscopic analysis.
Proliferation Study of Cells at Different Incubation Periods Using NR Assay
Table 4 shows the proliferation rate of HOS cells at different incubation periods of PNC material, rubber latex (positive control), and negative control. The proliferation percentage of HOS cells was highest (99.9) after 1 hour of incubation for both PNC and positive control and gradually decreased with longer incubation periods. In this case, there was no cytotoxicity until an incubation period of 72 hours (cell proliferation of 97.6%), whereas the extended incubation period of 168 hours induced cytotoxicity (cell proliferation of 39.3%).
Proliferation Percentage of HOS Cells at Different Incubation Periods for PNC Powder, Positive, and Negative Controls Using Neutral Red Assay—Direct Method (n = 6)
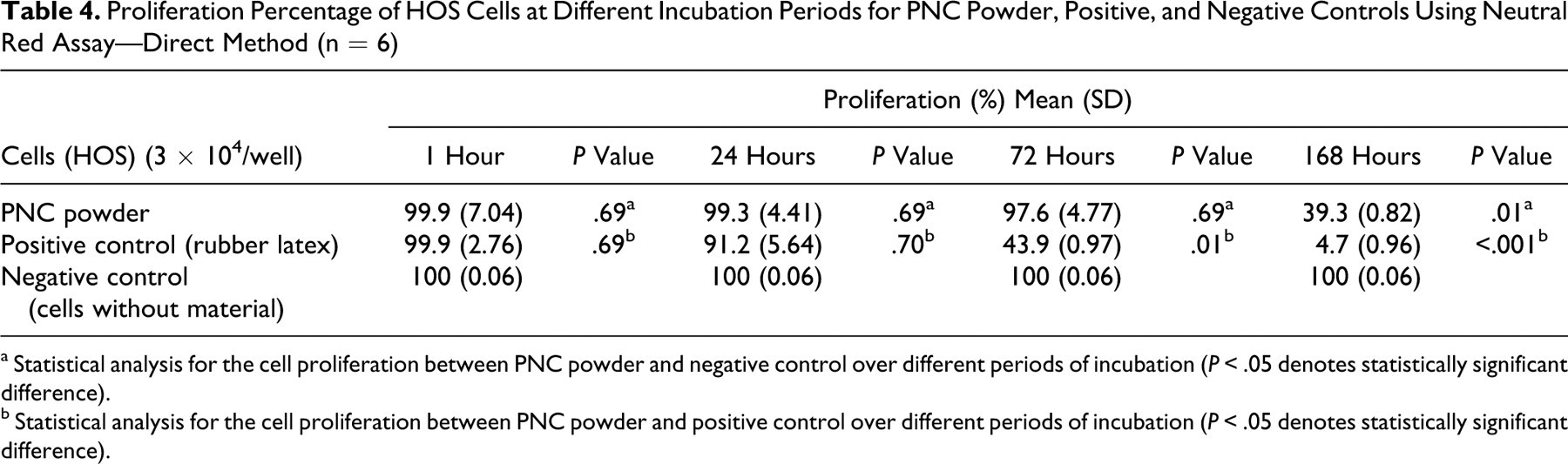
a Statistical analysis for the cell proliferation between PNC powder and negative control over different periods of incubation (
b Statistical analysis for the cell proliferation between PNC powder and positive control over different periods of incubation (
Attachment of HOS Cells Under Inverted Microscope
Figures 1A and 1B shows the attachment of HOS cells on to the edge of the PNC disc at a magnification of ×200 and ×400, respectively, whereas Figure 1C shows the absence of growth of cells on the rubber latex (positive control) after 72 hours of incubation.

A and B, Human osteoblasts (HOS) cells attached at the edge of the processed natural coral (PNC) disc (as indicated by arrows) after 72 hours of incubation at ×200 and ×400, respectively. C, Absence of growth of HOS cells on the rubber latex (positive control) after 72 hours of incubation. D, Absence of spread of HOS cells onto thermanox plastic disc (negative control) after 72 hours of incubation. E, HOS cells spread onto the PNC disc (as indicated by arrows) after 72 hours of incubation. F, Growth and spread of HOS cells into the PNC disc pore (as indicated by arrow) after 72 hours of incubation.
HOS Cells Viewed Under SEM
Figure 1E shows the attachment and spread of the HOS cells on the PNC disc. The cells also grew into the pores of the PNC disc (Figure 1F) after 72 hours of incubation period, whereas, on the thermanox plastic disc (negative control) the cells only grew on the disc but did not spread on the surface (Figure 1D).
Discussion
The applications of natural coral scaffolds for tissue engineering have been evaluated earlier
15
and reported as a favorable carrier for osteogenetically competent cells to attach and remain viable.
16
In the present study, PNC from the
The results from this study (Table 1) show that the minimum and maximum proliferation rates were at the concentrations of 25 and 200 mg/mL for the PNC granules and powder, respectively. Neither PNC granules nor powder is cytotoxic as per the MTT assay. In the case of positive control (70% ethanol), the EC50 was calculated to be 25.27 mg/mL, which indicates that the procedure is competent in observing the effect. In the case of negative control (HOS cell line not treated with any material), the proliferation rate was 100%.
The NR assay (3-amino-
In this study, NR assay was used to study the proliferation of PNC granules and powder by extraction method and also to study different incubation periods of HOS cells by direct method. The results (Table 2) demonstrated that the minimum and maximum proliferation rates were at the concentrations of 25 and 200 mg/mL for the PNC granules and powder, respectively. The negative control, cells not treated with any material, showed 100% proliferation rate.
Gregoire et al 18 reported that the particle size of materials influences the viability of cells. In this study, both the granules and the powder form of PNC did not show any cytotoxicity overall though there have been differences in the proliferation rate between both the particle sizes at the same respective concentrations. Two possible explanations for the size effect were suggested by Yina et al 19 . First, the surface energy of small particles is higher than that of large particles. The different surface energies may have different effects on the surfactant adsorption and conformation, which may alter chemical reactivity of surfactant per unit surface area of particles. In this way, the same surfactant may behave differently when it interacts with cells. Another possible explanation involves consideration of different interaction areas of large and small particles. The effective interaction area that a large particle can access the cell is greater than that for a smaller particle. Within this specific area, there are more function groups on individual large particle. Thus, each large particle exerts a stronger stimulus on the cells.
The use of FCM has become an exceptionally powerful tool for investigating the characteristic features of individual cells in a heterogeneous population without the need of physical separation. In a study done by Ferraz et al 20 , to study the biocompatibility of glass-reinforced hydroxyapatite as a scaffold using FCM, they found that the increase in the number of viable cells was the same or elevated following incubation on the preimmersed HA and glass-reinforced HA coatings compared with nonimmersed materials. In the current study, Annexin-V-FLOUS staining was used to detect and quantify the viable and apoptotic host cells after treatment with PNC powder. Table 3 shows that the viable cells were higher (99.94%) after 48 hours of incubation period compared to 97.09% for an incubation period of 72 hours. Similarly, the apoptotic cells were 0.06% and 2.91% for incubation periods of 48 and 72 hours, respectively. In the case of negative control, the percentage of viable cells showed highest (99.73) after 48 hours of incubation and lowest (96.83) after 72 hours of incubation. The tests with extract of PNC material and negative control show that the viable cells were more than 90% for both the periods of incubation. In this study, the proliferation rates of HOS cells of the PNC disc (Table 4) were seen highest at 1 hour of incubation (99.9%) and gradually decreased with corresponding increase in incubation periods. It was lowest for the incubation period of 168 hours (39.3%). In the case of positive control (rubber latex) also, the proliferation rate was highest at 1 hour of incubation (99.9%) and lowest (4.7%) at 168 hours of incubation. In the case of negative control, the proliferation rate was 100% for HOS cells, which was not treated with any material. These results indicate that the PNC material did not induce any cytotoxicity until an incubation period of 72 hours but exhibited cytotoxicity at 168 hours. For the positive control, the rubber latex induced cytotoxicity at 72 hours of incubation. The results demonstrated that without changing the medium, the PNC material was not toxic for up to 72 hours of incubation period but became toxic at 168 hours. This can be attributed to the leeching of the elements from the PNC and rubber latex which could have induced the cytotoxicity at their respective incubation periods. At the same time, there was no indication of any cytotoxicity in the negative controls even at 168 hours. Hence, the depletion of nutrients and amino acids present in the medium cannot be taken as a criteria for the induction of cytotoxicity in the cases of PNC material and positive control. To the best of our knowledge, this is the first study carried out at different incubation periods of coral extract on cell lines. Shaari and Samsudin 21 evaluated in vitro cytotoxicity on value-added hydroxyapatite as bone replacement using MRC-5 cell line at 1, 3, 24, 72, and 168 hours. They found that the test material started to exhibit cytotoxicity at 72 hours of incubation period with a percentage of killed cells of 9.44% and later increased to 46.27% at 168 hours on incubation.
Shors et al
22
studied the biocompatibility and osteoconduction of porous hydroxyapatite of
In this study, HOS cells were cultured on the PNC discs and observed under an inverted microscope after 72 hours of incubation. The HOS cells were found attached on the edge of the PNC disc, whereas, the cells were not attached on the edge of rubber latex. This shows that PNC material favors the attachment and growth of HOS cells, and hence the PNC is biocompatible. Scanning electron microscope study on the PNC disc seeded with HOS cells showed that the HOS cells attached on the surface of the PNC disc (Figure 1E) and later grew into the pores of the PNC disc (Figure 1F), whereas, in the case of thermanox plastic (Figure 1D), the cells only attached on the surface but did not spread on the surface. Moreover, the HOS cells not only attached well on the PNC disc but also maintained their typical appearance and retained their spherical configuration. This shows that the PNC material acts as a good scaffold for the cells to grow. Zhang et al 15 reported that since natural coral scaffold is porous, it provides a large internal surface for cell adhesion and migration. According to Edward et al 25 and Wiedmann et al, 26 the attachment of cells on the scaffold is influenced by the scaffold structure and cell substrate biocompatibility, leading to subsequent cell spreading, cell migration, and often cell differentiation function. Morphologically, they maintained their typical appearance with filopodia, attached well to the scaffold, and retained their spherical configuration, suggesting a well-differentiated phenotype. 27
The results of the above study show that the locally produced PNC material by Health Campus, Universiti Sains Malaysia, is noncytotoxic and favors the growth of HOS cells, thus holding a promise of being used as a scaffold material in future.
Footnotes
Acknowledgments
The authors thank the staff of Craniofacial Science Laboratory, School of Dental Sciences, and Staff of Tissue Bank, School of Medical Sciences, Universiti Sains Malaysia for their assistance in conducting this study.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
This research was financially supported by a short-term grant (304/PPSG/6131202) of Universiti Sains Malaysia, Malaysia.
