Abstract
The Tg.rasH2 mouse was developed as an alternative model to the traditional 2-year mouse bioassay for pharmaceutical carcinogenicity testing. This model has found extensive use in support of pharmaceutical drug development over the last few decades. It has the potential to improve quality and timeliness, reduce animal usage, and in some instances allow expedient decision-making regarding the human carcinogenicity potential of a drug candidate. Despite the increased use of the Tg.rasH2 model, there has been no systematic survey of current practices in the design, interpretation of results from the bioassay, and global health authority perspectives. Therefore, the aim of this work was to poll the pharmaceutical industry on study design practices used in the dose range finding and definitive 6-month studies and on results relative to the ongoing negotiations to revise The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use S1 Guidance. Twenty-two member companies of International Consortium for Innovation and Quality in Pharmaceutical Development DruSafe Leadership Group participated in the survey, sharing experiences from studies conducted with 55 test compounds between 2010 and 2018. The survey results provide very useful insights into study design and interpretation. Importantly, the results identified several key opportunities for reducing animal use and increasing the value of testing for potential human carcinogenicity using this model. Recommended changes to study designs that would reduce animal usage include eliminating the requirement to include positive control groups in every study, use of nontransgenic wild-type littermates in the dose range finding study, and use of microsampling to reduce or eliminate satellite groups for toxicokinetics.
Introduction
The Tg.rasH2 mouse model was developed as a short-term testing alternative to the traditional 2-year mouse carcinogenicity study (lifetime bioassay). The aim of both tests is to assess the carcinogenic potential of chemicals. In addition to the traditional or Tg.rasH2 mouse study, such an assessment generally includes tests for genotoxic potential, a carcinogenicity study (lifetime bioassay) in the rat and additional informative data such as histopathology results from other toxicology studies, and structure and pharmacological class-related information. The investment to complete such an assessment can be significant in terms of animal numbers, resources, and time such that often the carcinogenicity assessment is often on the critical path to pharmaceutical registration. Alternative animal models, such as the Tg.rasH2 have the potential to improve the quality and timeliness of the assessment and reduce the number of animals required. 1,2
The Tg.rasH2 mouse, developed in the early 1990s, harbors multiple copies of the human H-ras protooncogene with its own promoter region. 3 The H-ras gene is a member of a family of ras genes that encodes a 21-kDa protein, which is a key molecule in the signal transduction pathway for cell proliferation and inhibition of cell death by apoptosis. 4,5 This mouse model, which is slightly smaller in body weight than the wild-type mouse, typically develops tumors spontaneously after approximately 6 months of age. 4 Mutations or initiating events in the activating codon of the H-ras transgene result in enhanced expression of H-ras genes and increased tumor incidence. Mutations in the promoter region can be caused by genotoxic agents, but presumably also by spontaneous mutations that result from enhanced cell proliferation as can be induced by nongenotoxic carcinogens. Thus, primary mutation of the transgene by a genotoxic chemical as an initiating event is not a prerequisite for tumor development, and overexpression followed by spontaneous mutation of the transgene is a probable mechanism for tumor formation. Similarly, mutation of the endogenous mouse ras gene and/or mutation of other cooperating cancer driver genes are possible mechanistic causes of enhanced tumorigenesis. 5
Numerous reports on the responsiveness of the Tg.rasH2 mouse model to genotoxic and nongenotoxic carcinogens indicate that this model is an effective alternative to the traditional mouse carcinogenicity study and has been acknowledged as such by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), the Committee for Medicinal Products for Human Use (CHMP), and the US Food and Drug Administration (FDA). 6 -10 The model was originally validated against mutagenic carcinogens 4 and subsequently tested in a large collaborative evaluation of 21 substances including nonmutagenic carcinogens. 8 The model generally gives negative results for nongenotoxic non-carcinogens, is positive for most genotoxic carcinogens, and is negative for nongenotoxic rodent carcinogens. 11 However, the model, as is the case for all rodent models of carcinogenicity, does not have 100% sensitivity or specificity for known human carcinogens and non-carcinogens. Nevertheless, the Tg.rasH2, when justified as a suitable alternative to the traditional 2-year mouse model, offers many advantages. Therefore interest in and use of the model has increased substantially over the last 20 years.
In conjunction with a 2-year carcinogenicity study in rat, the predictivity of the rodent carcinogenicity test for human cancer risk increases. However, in certain circumstances, the outcome of the 2-year rat study itself can be predicted based on knowledge of the pharmacological mode of action and histopathology and other data from subchronic and chronic rat studies, in addition to standard genetic toxicology profiling. 12,13 Thus, ICH has published a Regulatory Notice Document that considers a change toward an integrated holistic approach to address the risk of human carcinogenicity of small molecule pharmaceuticals. In addition to the predictive knowledge described above, data from a transgenic mouse carcinogenicity study such as the Tg.rasH2 model could add value to the prediction of human carcinogenic potential. 14 Of interest is whether exposure thresholds can be defined for the Tg.rasH2 mouse similar to that for traditional rat and mouse 2-year carcinogenicity studies. For traditional 2-year carcinogenicity studies, drug exposures that exceed 25X the human exposures achieved at the maximum recommended human dose (aka the “25X rule”) are considered adequate to test the carcinogenic potential of a drug. 15 To date, this approach has not been applied to the Tg.rasH2 model where testing to a maximum tolerated dose (MTD) or maximum feasible dose (MFD) is typical.
Several reviews of the use of the Tg.rasH2 model have appeared in the literature. These reviews have focused on responsiveness to genotoxic and nongenotoxic compounds, 2,16 the use of positive controls in typical study designs, dose selection criteria, and interpretative factors such as the influence of body weight parameters on spontaneous tumor incidence. 17 -25 Despite the increased use of the Tg.rasH2 model, there has been no systematic survey of current practices in the design and interpretation of results from the bioassay. Therefore, the aim of this work was to poll the industry on Tg.rasH2 study design practices used in the dose range finding and definitive 6-month studies, and on results relevant to the ongoing negotiations toward revisions of ICH S1 in order to identify opportunities for refining the study design in ways that reduce animal usage and increase the value of study results for assessing human carcinogenic risk of pharmaceuticals.
Materials and Methods
The Tg.rasH2 survey was administered by the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ), Washington, DC. International Consortium for Innovation and Quality in Pharmaceutical Development is a technically focused organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators, and the broader R&D community. International Consortium for Innovation and Quality in Pharmaceutical Development members include many of the largest pharmaceutical companies [iqconsortium.org]. Within IQ is the DruSafe Leadership Group, focused primarily on common scientific and regulatory issues in nonclinical safety testing in drug development, which occasionally conducts surveys of its membership in order to gain cross-industry learnings. The current survey was sent to all 36 member companies of the DruSafe Leadership Group. Each responding company submitted a single response to the survey. The survey was distributed October 2018 and information submitted in response to all questions was restricted to the 8.5-year period of January 2010 to June 2018. Per IQ guidelines, the IQ Secretariat was responsible for uploading the survey into Survey Monkey, blinding the responses with regard to the respondent’s company affiliation, and distributing the blinded responses to the survey team. Following quality control review by the IQ secretariat and preliminary results review by the survey team, several follow-up questions were submitted to selected companies in order to clarify responses.
The original survey can be found in Supplemental Data. Overall, there were 65 questions covering the following topics: Number of compounds tested in conventional mouse models versus Tg.rasH2, and the rationale for model choice Use of positive controls Design of the dose range finding study and the definitive Tg.rasH2 (eg, test group size, frequency of toxicokinetic collections and blood collection technique) Dose selection rationale and achieved exposure multiples Interpretation, clinical, and regulatory implications Nontypical spontaneous tumors Use of Tg.rasH2 for testing therapeutics other than small molecules
Results
Survey responses were received from 22 of 36 companies. All but one of the 22 companies had completed at least 1 carcinogenicity study in any mouse model during the specified time frame. Among those companies, 55 compounds were tested using the Tg.rasH2 mouse model, whereas 31 were tested in a conventional mouse model (Figure 1). Among the companies giving a reason for not testing a compound in the Tg.rasH2 model, one indicated it was company policy not to use Tg.rasH2, and that company also cited lack of sufficient historical control data (HCD) as one of the reasons. Among those companies that used both models, 27% responded that the decision of which model to use depended on the therapeutic modality. Thirteen percent indicated that, as a general practice, they preferred the standard 2-year mouse model over Tg.rasH2 in order to be able to use the 25X rule for high dose selection. Ten percent of the respondents indicated that they have actually experienced specific cases where they opted to test their compound in a 2-year CD-1 mouse study in order to use the 25X rule and avoid the high doses needed to achieve the MTD in a Tg.rasH2 study. Seven percent indicated that the decision depended on whether or not the Tg.rasH2 had an acceptable metabolite profile (ie, comparable to human). Because of the shorter study duration and time to results offered by the Tg.rasH2 mouse study compared to the traditional 2-year study, it is also sometimes used for internal decision-making rather than exclusively to meet regulatory requirements. Indeed, 20% of companies indicated the study had been used for internal decision-making.
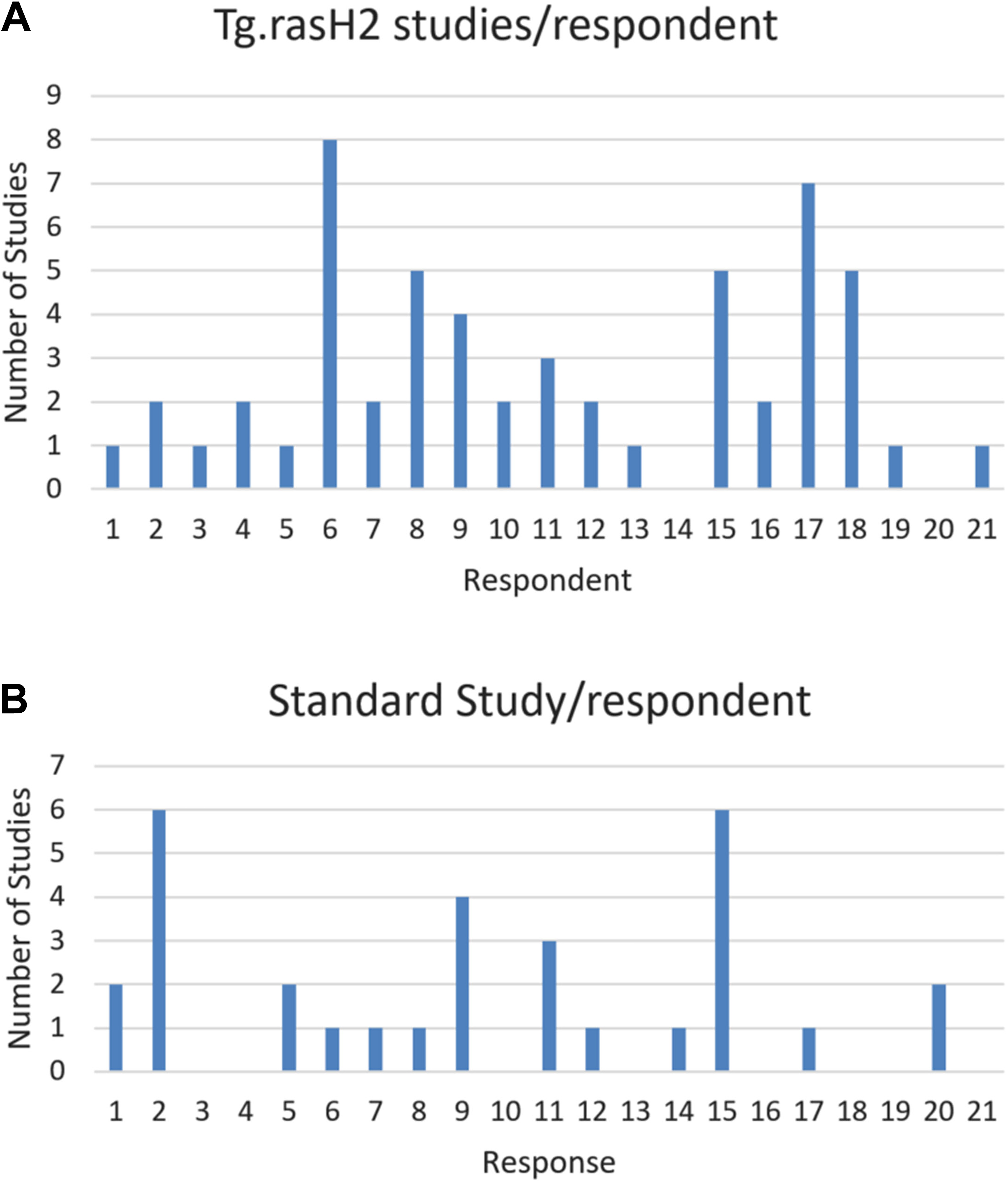
Number of studies reported as completed per survey respondent during the period of January 2010 to June 2018. A, Number of 6 month Tg.rasH2 carcinogenicity. B, Number of traditional 2-year mouse carcinogenicity studies.
An important aspect of this survey was to determine the value of including positive control groups (eg, urethane or N-nitrosomethylurea [NMU]) in the study design. Of the 55 compounds tested in Tg.rasH2 by survey participants, all included a positive control group, all positive control groups included males and females (group size averaged 19/sex, range 10-50) and the positive controls responded as expected in 100% of the studies.
It is typical for a study sponsor to submit the Tg.rasH2 protocol to the US FDA Executive Carcinogenicity Assessment Committee (ECAC) for review and comment. 26 Of the 55 studies reported in the survey, 53 of the study protocols were reviewed by the ECAC. In one case, the ECAC recommended including a positive control group where one had not been proposed in the original study design. That company then included the positive control group and commented that, as a result of that feedback, it now routinely includes a positive control group in all Tg.rasH2 mouse studies. The use of positive control groups is the result of concern from the Drug Regulatory Authorities (DRAs) about genetic drift and the possibility for loss of function of the human ras transgene. Companies were asked if they would be willing to forgo inclusion of a positive control group in future Tg.rasH2 studies if recent test facility-specific positive control data could be provided to the DRA together with genotyping data from each litter comprising the study animals. Assuming DRAs would accept those data, 89% responded “yes.”
One hundred percent of studies using Tg.rasH2 were preceded by a dose range finding (DRF) study, which is typically submitted to the ECAC as data supporting dose selection for the definitive 6-month study. During the evaluation period, companies reported a total of 66 DRF studies. All of the studies included a full list (∼40) of tissues for histopathological evaluation 27 and toxicokinetic evaluations. A total of 90% of the reported DRF studies were 4-week treatment duration. Studies of shorter (eg, 2-week) or longer (eg, 13-week) duration were also reported. A total of 79% of DRFs used the nontransgenic wild-type littermates; 13% used the Tg.rasH2 mouse; and 8% relied on data from the CD-1 mouse and stated that in those cases the CD-1 mouse was also used as the rodent species for general toxicity profiling. Only 3 respondents stated that a different toxicity profile was observed in a DRF using wild-type littermates versus definitive Tg.rasH2 studies. Those differences include one study indicating different tolerability, one indicating different target organs, and one indicating different plasma exposure. Among those companies that used the Tg.rasH2 mouse in the DRF, none of the 9 studies yielded differences in toxicity profiles compared to the definitive study. Only 1 of 19 companies responding to the question indicated that results from the DRF using wild-type littermates or Tg.rasH2 mice impacted an ongoing clinical trial, in this case prompting a self-imposed clinical hold until a follow-up study could be completed to resolve the concern raised. Similarly, non-neoplastic findings observed in the definitive Tg.rasH2 study were never reported to have had impact on an ongoing clinical trial.
Of the 66 DRF studies, 65 included satellite groups for toxicokinetic analyses and all but 1 included a full time-course profile (ie, for purposes of determining AUC and Cmax values). The group size averaged 31/sex (range 3-72). Approximately half (35 studies) included sampling for toxicokinetics on a single day, generally day 28, while half generally sampled on days 1 and day 28 (or at the end of the treatment period). A total of 70% of respondents used terminal bleeds (blood collected at necropsy) as the method for blood collection while 20% used another technique such as retro-orbital sinus bleeds, and 10% reported using microsampling. When doses studied in the definitive Tg.rasH2 study were similar as that studied in the DRF, 82% of companies repeated the toxicokinetic analyses in the definitive study generally citing as the reason that confirmatory and/or Good Laboratory Practice-compliant toxicokinetic analyses is expected for the definitive study. When not included in the definitive study, it was reported that the toxicokinetic data obtained from the DRF were sufficient and that repeated analyses were not necessary. For the definitive Tg.rasH2 study, most companies reported sampling during weeks 1 and week 26. Whereas the use of microsampling technique to collect blood from main study animals was reported infrequently (3 companies), 1 company reported its use on 7 studies suggesting proficiency with the technique.
Dose Selection and Study Outcome
A sponsor’s rationale for dose selection is generally provided to the ECAC upon request for special protocol review. As shown in Figure 2, the highest dose selected for the definitive study most frequently was based on the MTD criterion. Other bases for dose selection were consistent with ICH M3R2 guidance and included the MFD, saturation of exposure, achieving exposure multiples in excess of 25X the highest anticipated therapeutic exposure, or a combination of justifications. Using those approaches, 45%, 18%, 33%, and 4% of studies achieved exposure multiples >25x, 11X-25X, 1X-10X, or
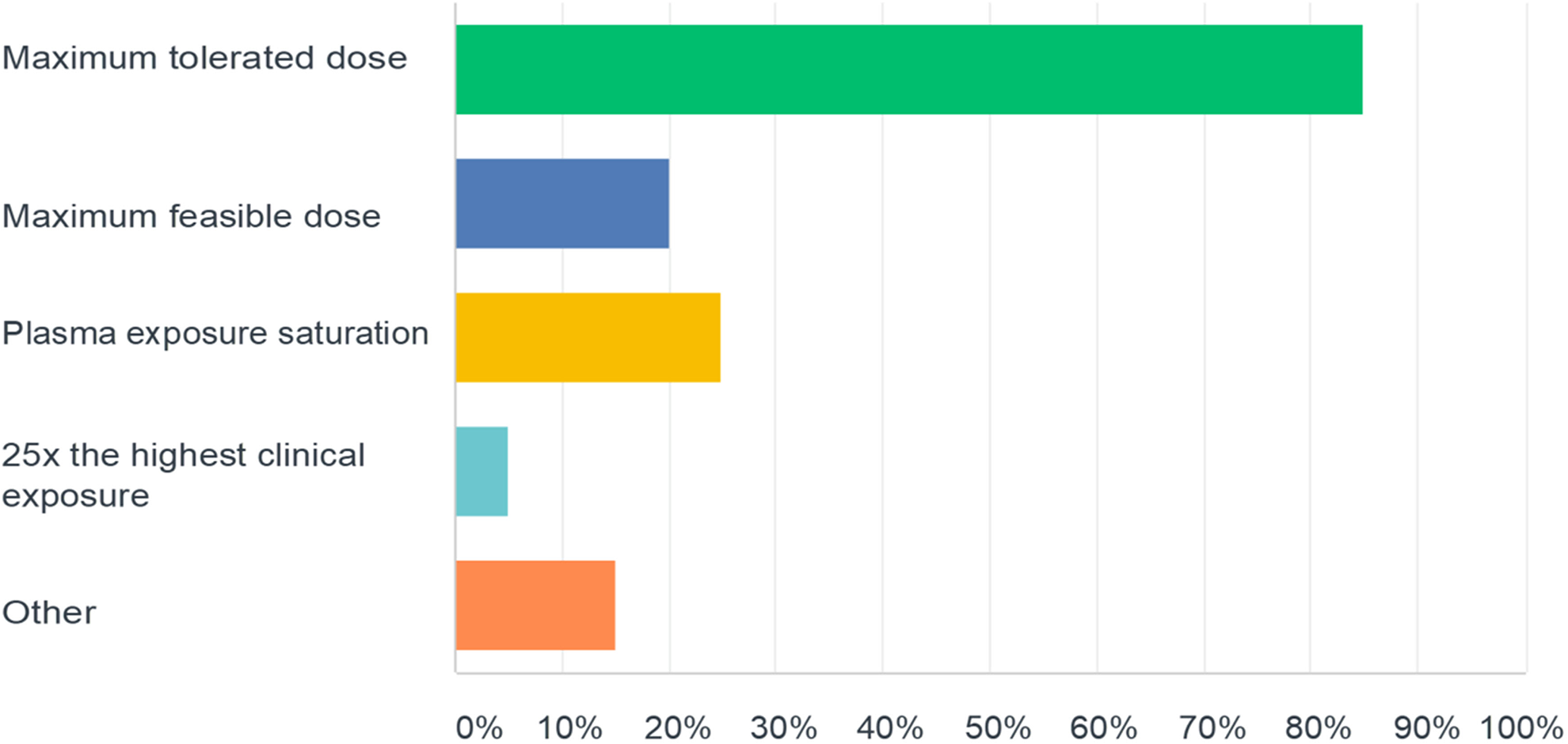
Responses to the question “For those compounds tested in the 6 month Tg.rasH2 mouse carcinogenicity study, what was the rationale for selection of the highest dose (select all that apply)?”
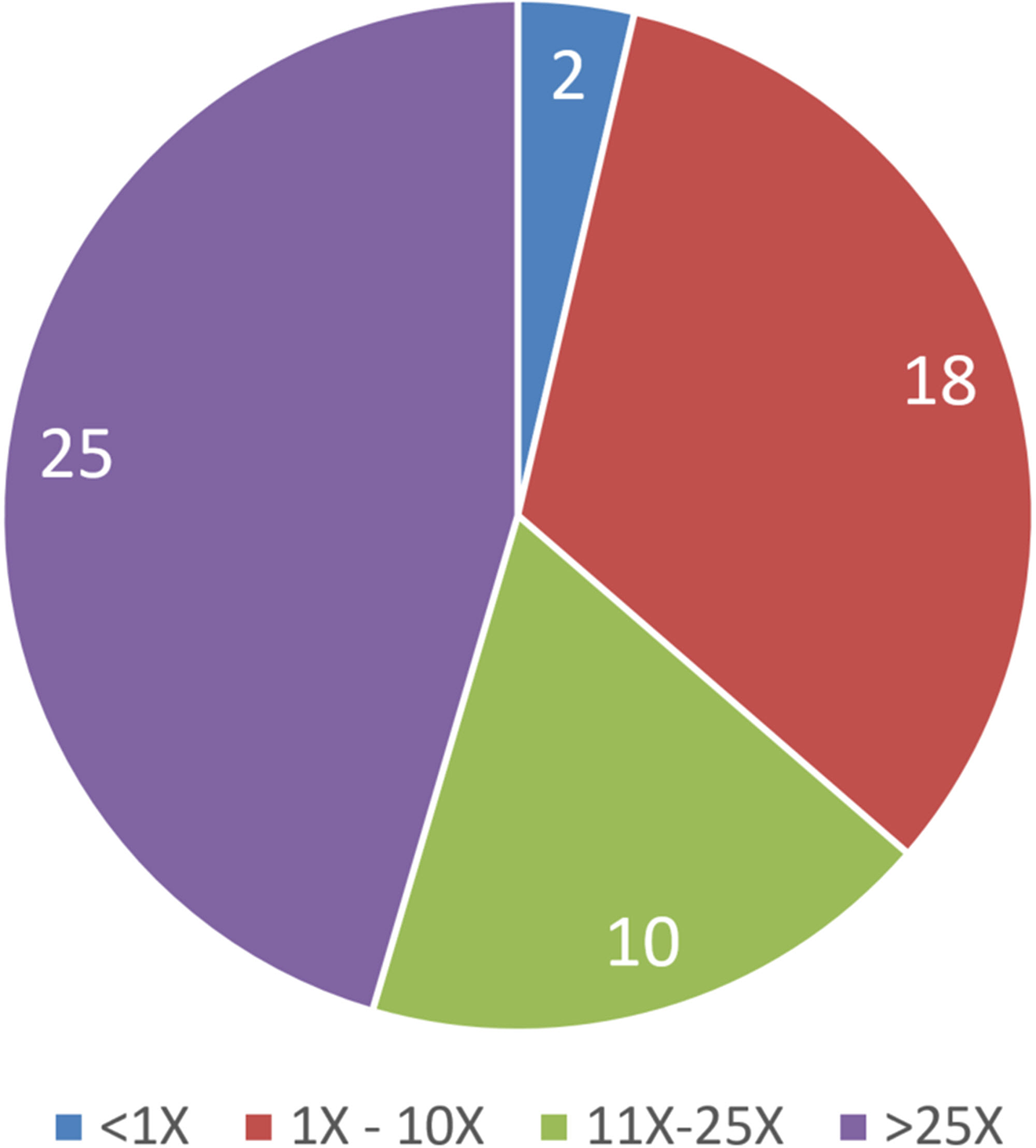
Percentage of Tg.rasH2 studies achieving multiples of the highest anticipated human therapeutic exposure achieved in the reported Tg.rasH2 carcinogenicity studies.
Of the 55 compounds reported in the survey as having been studied in the Tg.rasH2 model, 4 were concluded to be positive for carcinogenicity. One was positive at an exposure multiple (relative to the highest anticipated therapeutic exposure) of 18 000X, whereas 2 were positive between 1X and 10X, and one was positive at
Spontaneous tumors in the Tg.rasH2 model have been characterized. 17,24 In the current survey, only 16% of respondents (3 companies) reported observing a spontaneous tumor in an organ/tissue that was not listed in the HCD of the test facility where the study was conducted. For these 3 respondents, 3 to 5 additional tumors in novel organs were identified; none of the additional tumors themselves were novel. These tumors included forestomach squamous cell carcinoma (1), leiomyosarcoma/leiomyoma (1 each), uterine sarcoma (1), lymphangiosarcoma/lymphangioma (1 and 2, respectively), hepatocellular adenoma in a female mouse (1), and pituitary adenoma (1). In addition, 2 sponsors identified hemangiosarcoma in organs not previously included in the HCD; however, as these sites have been previously cited as locations for this tumor-type in Tg.rasH2 mice 17,24 this was not considered necessarily unusual. The above tumor types and sites are also those where mice of the background strain can demonstrate the observation (eg, liver, bone marrow, skeletal muscle, uterus).
Discussion
The present results are the first pharmaceutical industry-wide survey of current practices in study design and interpretation of results from the Tg.rasH2 mouse model. Established in the early 1990s 3 , the model has grown in interest as an alternative to the traditional 2-year mouse model. Much of this interest relates to the strategic flexibility and advantages offered by the model in terms of resources, timeline, and drug substance requirements compared to the traditional 2-year mouse study. Whereas the 2-year mouse study timeline is approximately 3 years from start of dosing to final report, the typical timeline for the Tg.rasH2 model is approximately 12 to 18 months. This significantly shorter timeline enables back loading of the registration-required study in high attrition-risk drug development programs where it is advantageous to delay investments. On the other hand, the study can be front loaded during a clinical program when questions of carcinogenic risk arise that have the potential to impact the safety of clinical trial participants. Fewer animals are used in the Tg.rasH2 model compared to the traditional 2-year mouse study (∼250 vs ∼600 excluding animals for toxicokinetics) thereby offering an important contribution to animal welfare goals of 3Rs (replace, reduce, and refine), although it must be recognized that the wild-type littermates not used for DRF or other experiments may be euthanized. Coupled with the shorter treatment duration (6 months vs 2 years), total drug substance requirements are approximately 90% less thereby saving significant resources, and test facility animal housing and husbandry demands are reduced by 75%.
Importantly, the Tg.rasH2 model has a low false-positive rate and, in comparison to the traditional 2-year mouse study, the Tg.rasH2 model identified known human carcinogens similarly, but produced fewer neoplastic outcomes in response to known rodent-specific carcinogens. 28 As discussed by Morton et al 28 and Storer et al, 29 the Tg.rasH2 model offers the ability to detect genotoxic and nongenotoxic chemicals of known concern for human risk, has a relatively low incidence of spontaneous tumors, produces fewer positive outcomes that are either rodent-specific or otherwise irrelevant for human cancer risk assessment, and is less likely to have spontaneous age-related morbidity, mortality, and neoplasias that are known to decrease survival and thereby confound interpretation in the 2-year rodent studies. As a result of these advantages, use of the Tg.rasH2 carcinogenicity bioassay is rapidly replacing the 2-year mouse study and, as reported by FDA authors in 2015, more than 75% of mouse carcinogenicity studies are conducted using the Tg.rasH2. 30 Similarly, participants in this survey related that 55 of 87 compounds, or 63%, were tested in the 6-month Tg.rasH2 model rather than the conventional 2-year mouse study.
Based on the wide use of the Tg.rasH2 model, the survey presented here aimed to capture current practices in the design and interpretation of data from the bioassay. Data from 22 companies comprising definitive Tg.rasH2 studies on 55 compounds and 66 DRF studies provided an excellent basis for the survey. The survey provided a reasonable representation of practices across the industry, as only 6 companies provided results on more than 3 Tg.rasH2 studies, while data on the remaining 13 studies were from companies who submitted data on
The large majority of companies used the MTD as the basis for high dose selection in the definitive Tg.rasH2 study, which confirms previous but smaller analyses by single companies. 21,22 An MTD is generally defined as a dose that results in sustained reduction in body weight gain of up to 10%, or other end points of toxicity that would not result in decreased survival over the 6-month study treatment duration. That the MTD was most often the basis for high dose selection is likely a reflection of US FDA ECAC expectations for an acceptable high dose. Thus, the doses chosen for the Tg.rasH2 study that are to be tolerated over 6 months, are usually higher than for the 2-year rat studies. Exposure multiples may also be higher, although the achieved multiples are not included in labeling by the US FDA as they are for the traditional 2-year rat study 30 and by other DRAs. This survey shows that almost half of the Tg.rasH2 studies achieved exposure multiples in excess of 25X, a multiple regarded as being sufficient to adequately test the carcinogenic potential of a chemical in traditional 2-year rat or mouse studies.
In contrast to traditional rat and mouse carcinogenicity models, no DRA has provided guidance on an upper limit of exposure considered sufficiently high to test the carcinogenic potential of a compound in Tg.rasH2. As a result, the majority of companies responding to this survey indicated that they would consider opting for a traditional CD-1 mouse study and apply the 25X rule if excessive exposure multiples were necessary to achieve an MTD in the Tg.rasH2 model. When asked to define an acceptable exposure multiple, the most frequent responses were 25X and 50X.
It has been the expectation of DRAs that positive controls be included in the definitive Tg.rasH2 study to demonstrate responsiveness of the model and to mitigate risk of a false negative due to genetic drift. However, the results of this survey call into question the continued inclusion of positive control groups. Positive control agents are typically administered by intraperitoneal injection as 3 doses of urethane (1000 mg/kg) or a single dose of NMU (75 mg/kg). Paranjpe et al 17 stated that over a decade’s time conducting Tg.rasH2 studies, the positive control group (urethane) produced tumors as expected in 100% of studies. This survey, covering an additional 5 years beyond the Paranjpe et al 17 paper, also reports the positive control group responding consistently as expected. Hemizygous rasH2 transgenic mice bred for such studies are identified by polymerase chain reaction analysis of DNA samples, and those quality control data are available upon request from the supplier (Taconic Biosciences, Inc, personal communication). Inclusion of such quality control data in study reports could offer a reasonable substitute for the in vivo positive controls. The large majority of respondents to this survey indicated a desire to eliminate the positive control group and instead supply genotyping data from the litters used on study in conjunction with facility-specific positive control data. If acceptable to DRAs, such a change would reduce animal usage substantially.
Dose-range finding studies are routinely conducted prior to the definitive 6-month Tg.rasH2 study. Range-finding studies typically include an initial 5 to 7 day tolerability study evaluating clinical signs, body weight, and gross observations with our without toxicokinetics, which then informs dose selection for a more comprehensive 4-week dose-range finding study. Our survey found that 79% of DRFs used the nontransgenic wild-type (CByB6F1) mice and in only 3 studies did respondents observe a different toxicity or exposure profile compared to that observed in the definitive Tg.rasH2 study, suggesting that use of the wild-type littermates is an acceptable surrogate for the Tg.rasH2 mouse for purposes of establishing the toxicity profile and doses for the definitive study. This result is in contrast to Paranjpe et al, 31 who proposed that the Tg.rasH2 mouse be used in the DRF studies on the basis that test compound-related sensitivity to toxicity is greater in the Tg.rasH2 versus the larger CByB6F1 littermates. Such a proposal would result in substantial unused, possibly wasted wild-type littermates.
Toxicokinetic analyses were found routinely to be included in the DRF and definitive Tg.rasH2 study. Most companies used either terminal bleeds, limited to one time point per animal, or retro-orbital sinus bleeds that, due to blood volume restrictions imposed by animal welfare concerns, are generally limited to 3 bleeds per mouse with the last collection being a terminal bleed. Even when doses used in the definitive study were the same or similar to that used in the DRF, toxicokinetic analyses were repeated in the definitive study. This result generates interesting questions with regard to the value of toxicokinetic analyses as currently applied in Tg.rasH2 and the possibility to refine study designs to use fewer animals.
One company reported use of the microsampling technique for blood collection in 7 studies. Microsampling is gaining increasing application in toxicology studies, and with sample volumes as low as 10 μL, repeated sampling is possible thus significantly reducing animal numbers in toxicokinetic satellite groups and possibly eliminating the need for satellite groups. 32 Studies to evaluate the effect of repeated microsampling on the hematological profile of Tg.rasH2 mice have not been reported and are encouraged.
The US FDA has stated that exposure margins are not considered in either dose selection or when interpreting drug-related results
30
for Tg.rasH2 studies raising the question of the value to FDA of any toxicokinetic analyses, other than confirming exposure to test article. In contrast, other DRAs do include Tg.rasH2 study exposure data in drug labeling. This highlights a need for harmonization around an exposure limit for Tg.rasH2 bioassays similar to the “25X rule” valid for traditional rodent carcinogenicity bioassays. Such an approach seems reasonable, and the current survey shows that 3 of 4 compounds that tested positive in the Tg.rasH2 study were detected at exposure multiples
Within the efforts launched to revise ICH S1 Pharmaceutical Carcinogenicity Testing Guidelines, 14 changes being considered include elimination of the need to conduct a 2-year rat study when certain weight-of-evidence criteria support the conclusion that such a study would provide no additional value. A mouse study would still be expected with few exceptions. As discussed previously, 28 any decision to conduct a 2-year mouse study under such circumstances when a 6-month Tg.rasH2 could be conducted instead, would negate the gains in timelines to complete carcinogenicity testing requirements. If such revisions are accepted, greater use of the Tg.rasH2 model, compared to the traditional 2-year mouse study, would be anticipated, as the model would serve to (1) provide greater assurances that weigh-of-evidence criteria are used to support a decision to waive the 2-year rat study and (2) provide greater assurances that the drug candidate is void of human relevant tumorigenic risk.
As mentioned previously, one advantage of the Tg.rasH2 model is a low overall spontaneous tumor rate, and background tumors have been well characterized. 17 Survey results showed a low incidence of spontaneous tumors not previously identified in testing facility HCD. A few spontaneous tumors were identified by 3 companies that had not previously been recorded in the testing facility/ies’ HCDs. When one examines the published literature, most of these tumors have been recorded previously in untreated Tg.rasH2 mice, 17,24 and for those not identified individually, Paranjpe et al 17 indicate an incidence of “other tumors combined” (ie, not identified individually) of 35 (4.90%) of 710 in male mice and 167 (8.55%) of 710 in female mice, stating that their individual incidence is low and their occurrence sporadic. Therefore, occasional cases of other spontaneous tumor types have been reported, and as the number of studies increase at other facilities, the overall spectrum of background tumors observed at each facility is likely to increase also. None of the tumors identified as potentially novel for the test facility were considered to indicate a notable shift in the tumor profile of the Tg.rasH2 mouse model. It is important to note that the majority of publically available HCD are reported in the papers of Paranjpe et al. More broad disclosure of HCD in the open literature by contract research organizations and pharmaceutical companies is encouraged.
Conclusions
With greater than 75% of mouse carcinogenicity bioassays submitted to the US FDA being conducted using the Tg.rasH2 mouse, this cancer bioassay is the predominant choice in the pharmaceutical industry as an alternative to the traditional 2-year mouse cancer bioassay. Having advantages of time, resources, low incidence of spontaneous tumors and reduced positive outcomes that are either rodent-specific or otherwise irrelevant for human cancer risk assessment, the Tg.rasH2 offers many advantages over the traditional 2-year mouse study. The current survey provides insight into current design and interpretation aspects of the study and has identified several opportunities for refining the study design in ways that reduce animal usage and increase the value of study results for assessing human carcinogenic risk of pharmaceuticals. Recommended changes to study designs that would reduce animal usage include eliminating the requirement to include positive control groups in every study, use of nontransgenic wild-type littermates in DRF and other experiments, and use of microsampling to reduce or eliminate satellite groups for toxicokinetics.
Supplemental Material
Supplemental Material, DruSafe_Tg.RasH2_Questionnaire_Final - Tg.rasH2 Mouse Model for Assessing Carcinogenic Potential of Pharmaceuticals: Industry Survey of Current Practices
Supplemental Material, DruSafe_Tg.RasH2_Questionnaire_Final for Tg.rasH2 Mouse Model for Assessing Carcinogenic Potential of Pharmaceuticals: Industry Survey of Current Practices by Matthew S. Bogdanffy, Jacob Lesniak, Raja Mangipudy, Frank D. Sistare, Karyn Colman, David Garcia-Tapia, Thomas Monticello and Diann Blanset in International Journal of Toxicology
Footnotes
Acknowledgments
The authors wish to thank the IQ Secretariat, especially Ann Marie Stanley, for their help administering the survey, collecting and blinding the results. The authors also acknowledge with gratitude the contributions of the IQ DruSafe Tg.rasH2 Working Group who are, in addition to the authors: Mark Fielden (Amgen), Frank Geoly (Pfizer), David Jacobson Kram (ToxRox), Eric Harstad (Theravance), Doug Keller (Sanofi), and Jose Lebron (Merck & Co, Inc, West Point, Pennsylvania).
Author Contributions
Bogdanffy, M. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Lesniak, J., Manigpudy, R., Sistare, F., Colman, K., Garcia-Tapia, D., and Monticello, T. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and critically revised manuscript; Blanset, D. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This survey was developed with the support of the International Consortium for Innovation and Quality in Pharmaceutical Development (IQ, ![]() ). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
). IQ is a not-for-profit organization of pharmaceutical and biotechnology companies with a mission of advancing science and technology to augment the capability of member companies to develop transformational solutions that benefit patients, regulators and the broader research and development community.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
