Abstract
Carcinogenicity studies have been performed in conventional 2-year rodent studies for at least 3 decades, whereas the short-term carcinogenicity studies in transgenic mice, such as Tg.rasH2, have only been performed over the last decade. In the 2-year conventional rodent studies, interlinked problems, such as increasing trends in the initial body weights, increased body weight gains, high incidence of spontaneous tumors, and low survival, that complicate the interpretation of findings have been well established. However, these end points have not been evaluated in the short-term carcinogenicity studies involving the Tg.rasH2 mice. In this article, we present retrospective analysis of data obtained from control groups in 26-week carcinogenicity studies conducted in Tg.rasH2 mice since 2004. Our analysis showed statistically significant decreasing trends in initial body weights of both sexes. Although the terminal body weights did not show any significant trends, there was a statistically significant increasing trend toward body weight gains, more so in males than in females, which correlated with increasing trends in the food consumption. There were no statistically significant alterations in mortality trends. In addition, the incidence of all common spontaneous tumors remained fairly constant with no statistically significant differences in trends.
Introduction
During the nonclinical phase of the drug development process, carcinogenicity studies of pharmaceuticals and other compounds are conducted in rodents. Traditionally, these 2-year studies have been conducted in rat models such as F344 and Sprague-Dawley (SD) and in mouse models such as CD-1 and B6C3F1. However, worldwide regulatory acceptance has allowed the use of short-term, 26-week carcinogenicity studies in transgenic animals, including CByB6F1-Tg(HRAS)2Jic (±hemizygous c-Ha-ras; Tg.rasH2) mice, in place of the 2-year mouse assay. The heterozygous Trp53 model is also an acceptable model for short-term carcinogenicity assessment although only for genotoxic compounds.
In the last decade, the Tg.rasH2 mouse has become a popular model for performing 26-week carcinogenicity studies, replacing more than half of all 2-year carcinogenicity studies performed in conventional mouse models. 1 -3 We recently published the largest historical control database for both neoplastic and nonneoplastic lesions in Tg.rasH2 mice. 3,4 Based on extensive experience with this assay in our laboratory, a number of improvements have been made including reducing the number of animals in the positive control group from 25 to 10 mice/sex, leading to a substantial reduction in animal use and thereby contributing to the 3Rs (replace, reduce, and refine) of animal research. 5 We also recently proposed that instead of reading a full tissue list from all animals in the study, only select tissues in the low and mid doses can be evaluated without affecting the integrity of the studies. 6 Additionally, we have recently established the relationship of body weight parameters with the incidence of common spontaneous tumors in the Tg.rasH2 mice. 7
Increasing trends in the initial body weights and body weight gains of CD-1 and B6C3F1 mice and SD and F344 rats, which are used in traditional 2-year carcinogenicity assays, have been well established. 8 -22 Increasing initial body weights and body weight gains have been directly associated with decreased survival in both rats and mice in 2-year studies, mainly due to an increase in spontaneous tumors as well as degenerative diseases. These trends have hindered the ability of the conventional 2-year studies to demonstrate true-positive dose–response relationships. 15 -17,20,23 -25 Being a relatively new model, there has been no information available on trends in bodyweight parameters, tumor incidence, and survival rates in Tg.rasH2 mice. In this article, we present trend analysis of the initial body weights, terminal body weights, the percentage of body weight gains, food consumption, mortality, and the incidence of common spontaneous tumors in male and female Tg.rasH2 mice.
Materials and Methods
Animals
CByB6F1-Tg(HRAS)2Jic (±hemizygous c-Ha-ras) mice, obtained from Taconic Farms (Germantown, New York), were used in all studies. The knock-in Tg element (human prototype c Ha-ras gene with its own promoter/enhancer) is injected into C57BL/6 × BALB/c F2 zygotes, which are crossed back to the C57BL/6J strain forming C57BL/6JJic-Tg(HRAS)2Jic. The CByB6F1-Tg(HRAS)2Jic (±hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mice with BALB/cByJJic female mice. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study. Animals were assigned to groups using a computer-generated randomization program. On the first day of treatment, animals were 6 to 10 weeks of age and weighed at least 20 or 15 g (males and females, respectively). Individual body weights for each dose group of each sex were within ±20% of the mean at the start of the study.
Housing and Environmental Conditions
Housing and environmental conditions were similar in all studies. Animals were single housed in polycarbonate cages with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had ad libitum access to water and powdered feed (Harlan TEKLAD Global Diet, Madison, Wisconsin).
Regulatory Requirements
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care-accredited facility. All procedures involving but not limited to acclimation, randomization, application of unique identification system, housing, provision of food and water, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the Good Laboratory Practice Regulations, Standard Operating Procedures, and the protocol for each study.
Retrospective Analysis
The database was constructed based on data collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had an 2 control groups), conducted at our facility. All studies followed the same study design and were placed under the same environmental conditions. The first of these studies was completed in 2004, and the last study was completed in 2013. Vehicle was administered to 810 male and 810 female mice by oral gavage (24 studies), dosed feed (3 studies), or intravenous injection (2 studies that received 8-10 doses each) for 26 weeks. Studies with all routes of administration were included in the analysis as no differences were seen that could be related to the route of administration. Predose body weights (initial body weights) were collected on day 1, and mice were weighed weekly thereafter for the next consecutive 13 weeks. After 13 weeks, the body weights were collected biweekly until termination of the study (terminal body weights). Food consumption was recorded weekly for individual animals from day 1 until termination of the study at 26 weeks. For all studies, except dosed feed studies, individual food consumption values above what is expected for mice were excluded from the group average values. At study termination, all animals in each study were humanely killed by CO2 overdose and subjected to complete necropsy. All animals that died before study termination were also subjected to complete necropsy. Protocol-required tissues were saved in 10% neutral-buffered formalin, processed routinely, stained with hematoxylin and eosin, and evaluated microscopically. The histopathology for each study was peer reviewed.
The data were analyzed for trends in the initial and terminal body weights, the percentage of body weight gains, food consumption, mortality, and incidence of the common spontaneous tumors in male and female mice assigned to different studies. The spontaneous tumors analyzed included (1) lung tumors (adenomas and carcinomas), (2) splenic hemangiosarcomas, (3) nonsplenic hemangiosarcomas, (4) combined incidence of all hemangiosarcomas (splenic and nonsplenic), and (5) Harderian gland tumors (adenomas and carcinomas). The data were analyzed using regression statistics; specifically, the estimated slope of each fitted regression line was tested for a difference from 0 (Microsoft Excel 2007, Microsoft corporation) using LINEST. Nonsplenic hemangiosarcomas included multicentric hemangiosarcomas and hemangiosarcomas of organs other than spleen such as testes, seminal vesicle, epididymis, liver, lung, nasal cavity, skin/subcutis, bone, ovary, vagina, uterus, and kidneys.
Results
The data from 29 studies with 32 control groups (3 of the studies had 2 control dose group) were analyzed for trends in initial and terminal body weights (Figures 1 and 2), percentage of body weight gains (Figure 3), food consumption (Figure 4), percentage of mortality (Figure 5), incidence of lung tumors (Figure 6), incidence of splenic hemangiosarcomas (Figure 7), incidence of nonsplenic hemangiosarcomas (Figure 8), combined incidence of all hemangiosarcomas (Figure 9), and incidence of Harderian gland tumors (Figure 10). The slope and statistics of the regression line for each parameter analyzed are summarized in Table 1.
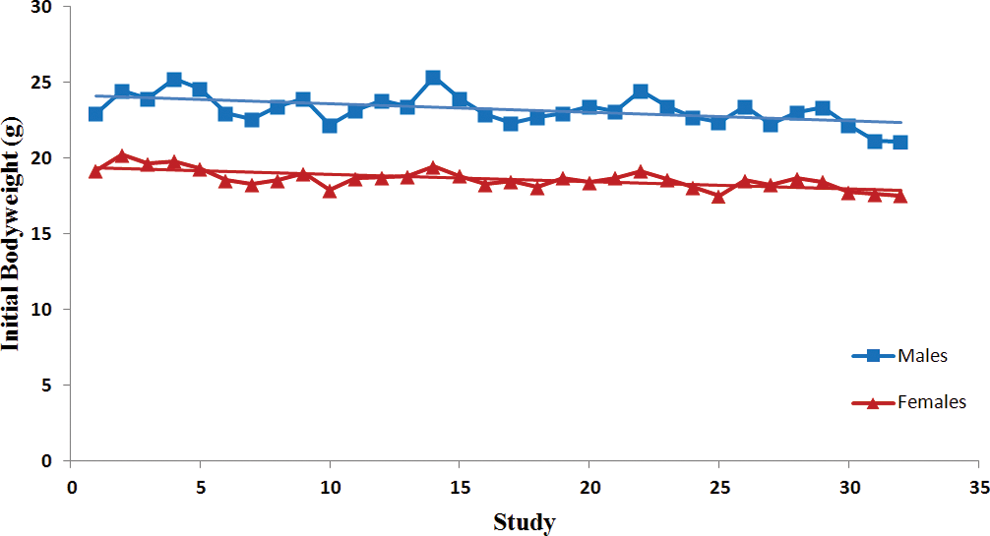
Trend analysis of initial body weights. Each point represents average data from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
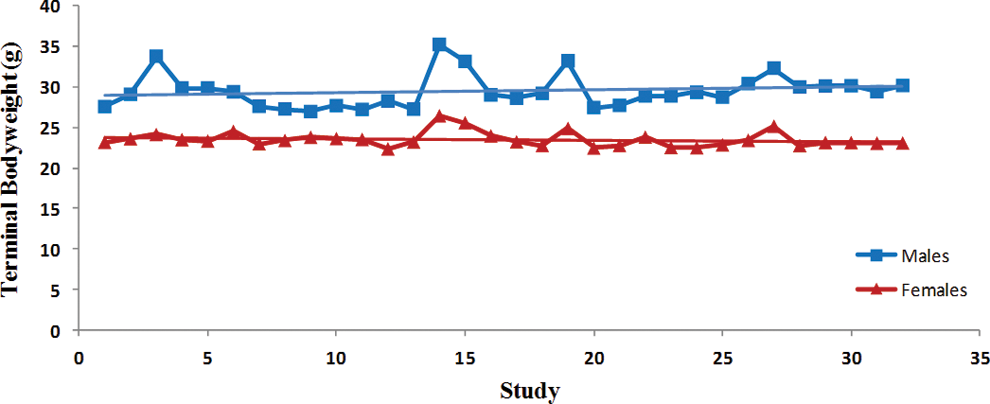
Trend analysis of terminal body weights. Each point represents average data from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
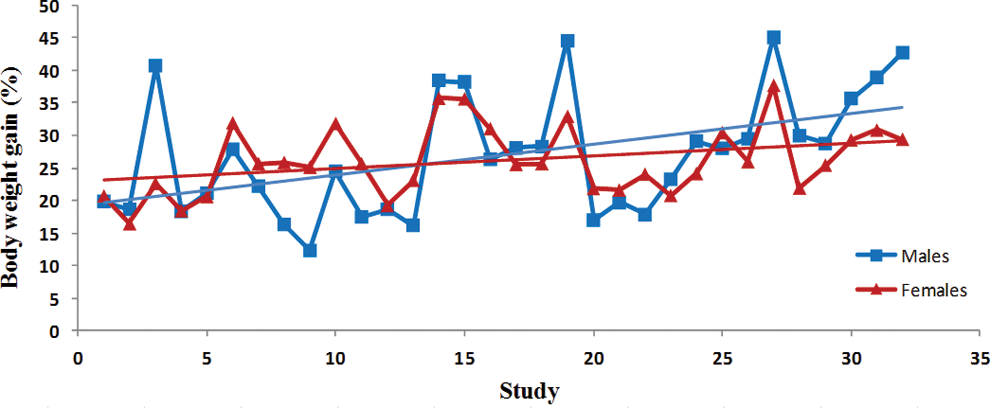
Trend analysis of body weight gains. Each point represents average data from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
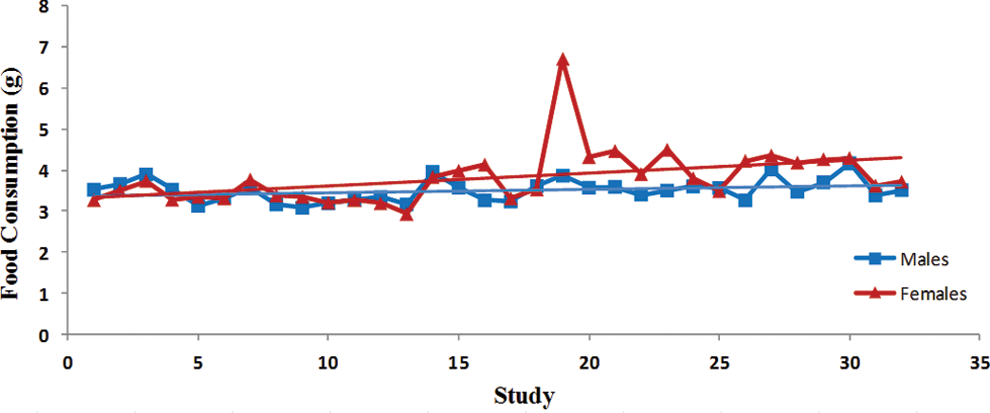
Trend analysis of food consumption. Each point represents average data from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes. For all studies, except dose fed studies, individual food consumption values above what is expected for mice, were excluded from the group average values. The study with an apparent spike in food consumption was a dose-fed study.
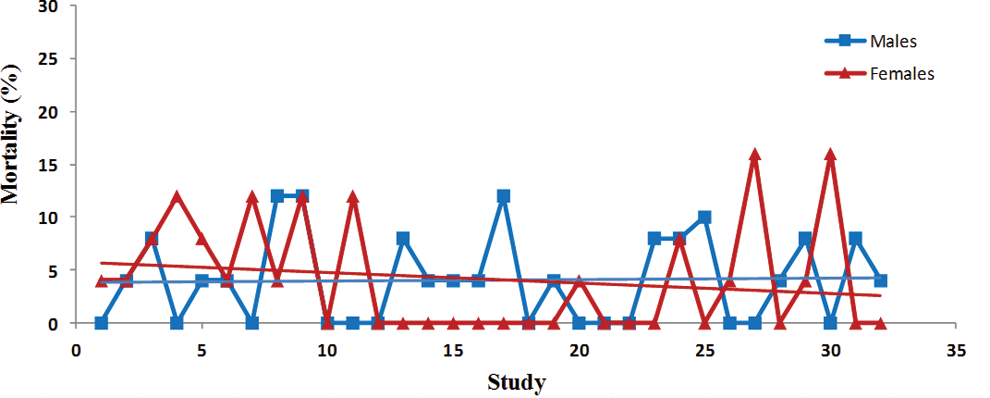
Trend analysis of mortality. Each point represents percentage of mortality from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
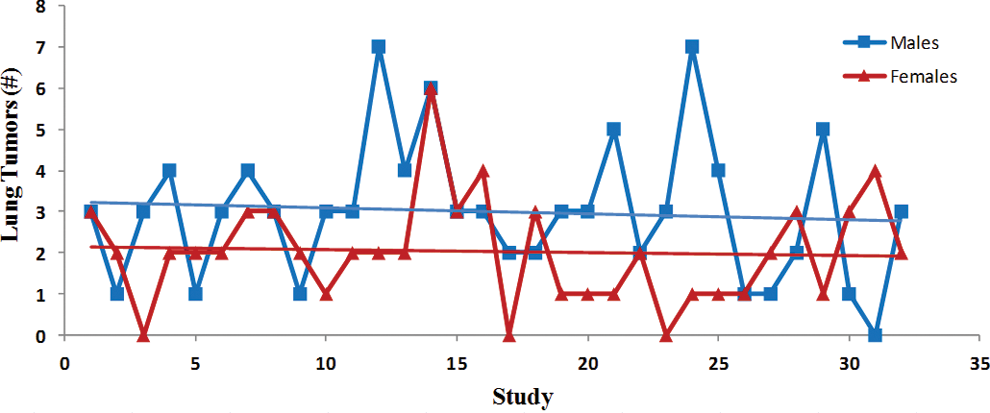
Trend analysis of the lung tumors (adenomas and carcinomas). Each point represents total tumor incidence from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
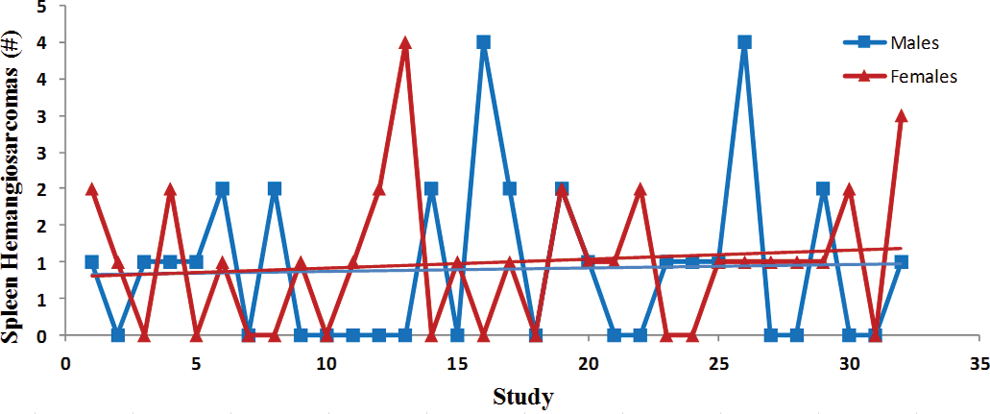
Trend analysis of the splenic hemangiosarcomas. Each point represents total tumor incidence from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
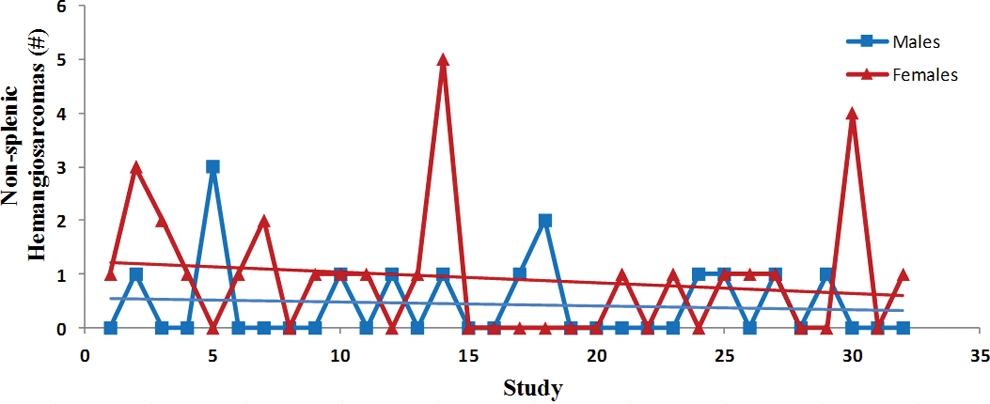
Trend analysis of the nonsplenic hemangiosarcomas. Each point represents total tumor incidence from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
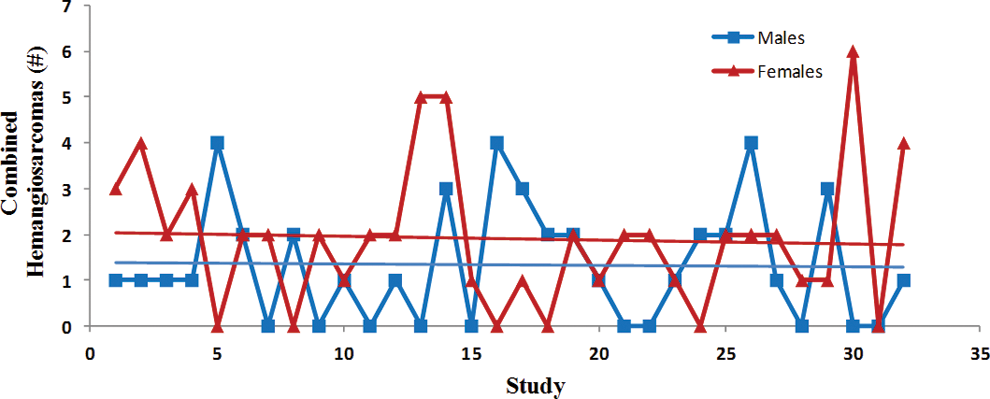
Trend analysis of the combined incidence of all hemangiosarcomas. Each point represents total tumor incidence from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
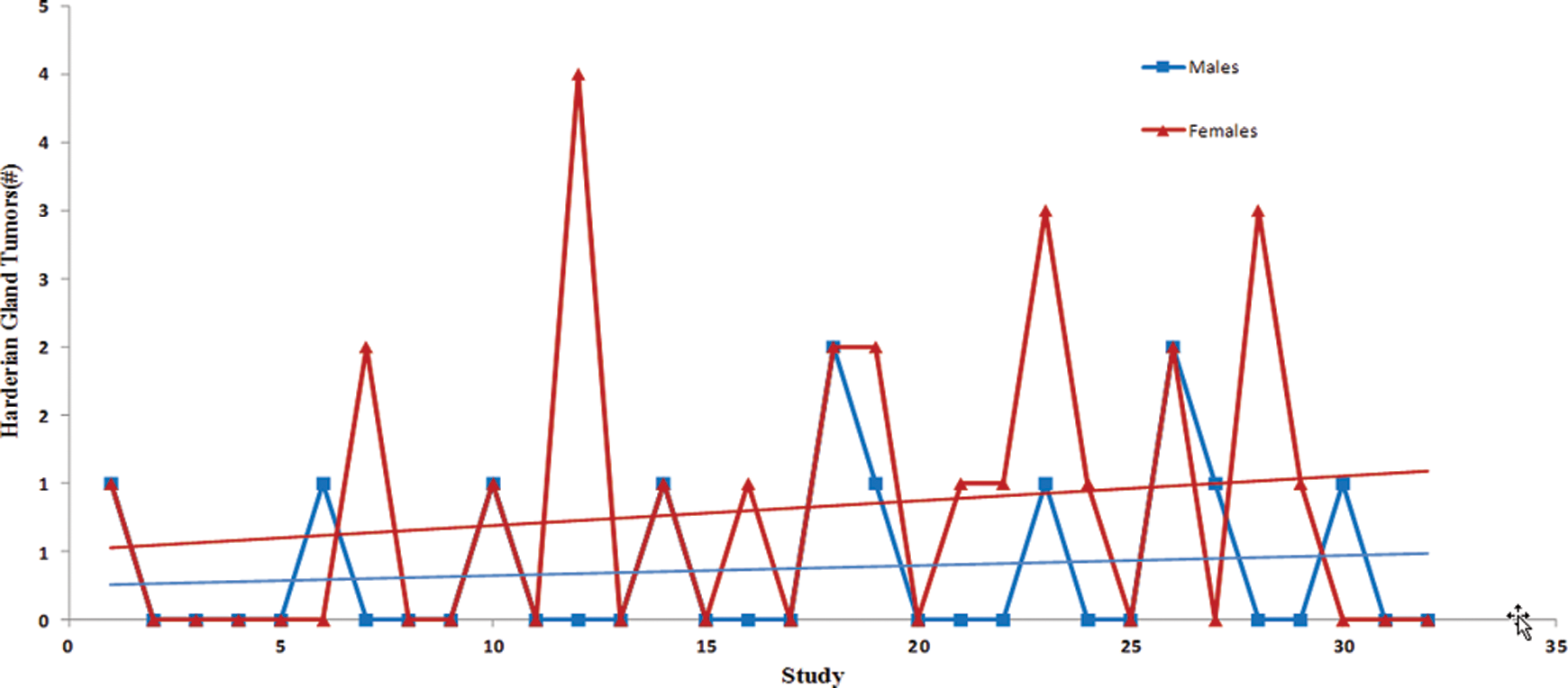
Trend analysis of the Harderian gland tumors (adenomas and carcinomas). Each point represents total tumor incidence from a single control group. Data were collected from Tg.rasH2 mice assigned to 29 studies, with 32 control groups (3 studies had 2 control groups) for a total of 810 animals examined. Trend line is applied for illustrative purposes.
The Slope and Statistics of the Regression Line for Trends in Initial Body Weights, Terminal Body Weights, Body Weight Gains, Food Consumption, Mortality, Lung Tumors, Splenic Hemangiosarcomas, Nonsplenic Hemangiosarcomas, All Hemangiosarcomas, and Harderian Gland Tumors.
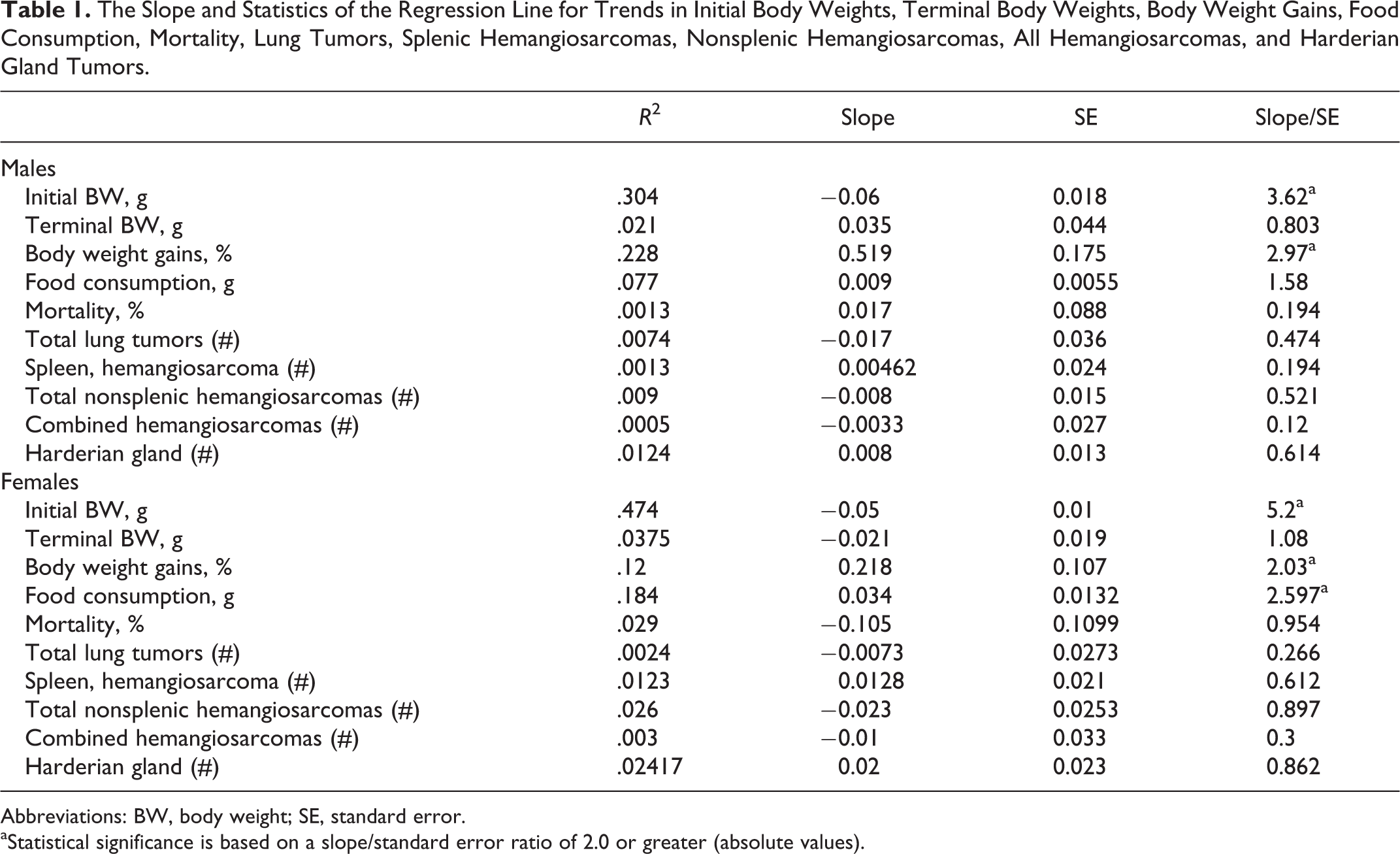
Abbreviations: BW, body weight; SE, standard error.
aStatistical significance is based on a slope/standard error ratio of 2.0 or greater (absolute values).
There was a statistically significant steady decline in the initial body weights in both sexes (Table 1). The variability in the initial body weights between studies was less pronounced in the females than the males (Figure 1). There was no statistically significant variation in the terminal body weights of either sex (Figure 2). Despite the statistically significant steady decline in the initial body weights and lack of any trends in the terminal body weights, there was a significant trend for percentage of increased body weight gains in both sexes (Table 1). However, the percentage of body weight gains varied considerably between studies (Figure 3). The increase in body weight gain correlated with a higher trend in average food consumption, which was statistically significant in females but not in males (Figure 4 and Table 1). Since apparently high food consumption values were not excluded in dosed feed studies, 1 study showed an apparent spike in food consumption. Mortality remained fairly consistent in male and female animals, with no statistically significant trends; however, the mortality in females appears to be decreasing, although it was not statistically significant (Figure 5).
The incidence of tumors was fairly consistent between studies as there were no statistically significant differences, and the differences between males and females were subtle (Figures 6 –10). There was a trend toward a decline in the incidence of lung tumors, nonsplenic hemangiosarcomas, and the combined incidence of all hemangiosarcomas in both sexes, although these declining trends were not statistically significant. On the other hand, there was a nonstatistically significant trend toward an increase in the incidence of splenic hemangiosarcomas (Figure 7) as well as Harderian gland tumors in both sexes (Figure 10).
Discussion
The survival of rodents in 2-year studies has long been an issue, and there are large variations between the strains of rats and mice and between different testing facilities. In CD1 mice, the survival rate is 27% to 73% in males and 25% to 78% in females. 27 -30 The survival rate in male and female B6C3F1 mice is 60% to 75%, and 49% to 61% in male and female F344 rats. 12,31 In addition, the incidence of spontaneous tumors reported in conventional rodent strains has also been very high. In CD1 mice and F344 rats, the incidence of tumors is close to 80% and in B6C3F1 mice the incidence is between 75% and 85%. 12,27 -31 The large variation in survival and tumor incidence in the references noted here are most likely due to variables in the breeding colonies and interlaboratory differences. In comparison, the average mortality in Tg.rasH2 mice has remained very low at 3.94% in both sexes, and the incidence of spontaneous tumors has remained below 25%. 3
Over the past 3 decades, there have been increasing trends in the initial body weights of conventional rodents such as CD1 and B6C3F1 mice and SD and F344 rats. The causes of increasing trends are many but primarily include changes in breeding considerations, particularly selection of breeding stocks for faster growth and earlier reproduction to satisfy user preferences for larger rodents and lower costs. Other possible causes include improved diet formulations and changes in housing and environmental conditions, particularly changes in housing protocols from group to single housing. 8 -22 Increases in initial body weights and body weight gains have been directly associated with decreased survival in both rats and mice in 2-year studies, mainly due to an increase in spontaneous tumors as well as degenerative diseases. 15 -17,20,23 -25 There has also been a reduction in the survival of male F344 rats (from 66% to 50%) due to leukemia, pituitary tumors, and nephropathy. 15 -17,20,23 -25 The incidence of leukemia has increased from 27% to 51% in male F344 rats and from 18% to 28% in female rats; mammary gland fibroadenomas have increased from 28% to 41%. 15 -17,20,23 -25 Similarly in B6C3F1 mice, liver tumors have increased from 30% to 42% in males and 8% to 24% in females. 10,11,12,19,26 Insufficient survival for the duration of the study and excessively high incidence of background tumors has prevented adequate interpretation of the data and demonstration of dose–response relationships. 15 -17,20,23 -25
Herein, we have presented trend analysis of body weight parameters, mortality, and incidence of spontaneous tumors from the data collected from control animals of 26-week studies in Tg.rasH2 mice between 2004 and 2013. Since all studies were performed in the same facility, differences that could have been caused by changes in environmental conditions, husbandry practices, water supply, diet, and diagnostic criteria are mitigated. Contrary to the increasing trends in the initial body weights observed in 2-year chronic carcinogenicity studies, there are no increasing trends in the initial body weights in Tg.rasH2 mice. On the other hand, there is a statistically significant steady decline in the initial body weights in both sexes of Tg.rasH2 mice. The lack of increasing trends in initial body weights in Tg.rasH2 mice is likely due to consistent breeding practices employed in the production of Tg.rasH2 mice in comparison with conventional 2-year rodent models in which there is more selective breeding. Despite decreasing trends in initial body weights and lack of any trends in terminal body weights, there was a significant trend toward increasing body weight gains. These body weight gains correlated with the increasing food consumption, which was only statistically significant in females, but not in males. In spite of an increasing trend in body weight gains, there were no trends toward increasing mortality or increasing incidence of spontaneous tumors that were statistically significant. On the contrary, survival in females appears to be increasing, and the incidence of spontaneous lung tumors, nonsplenic hemangiosarcomas, and combined incidence of all hemangiosarcomas appear to be declining in both sexes. On the other hand, spontaneous splenic hemangiosarcomas and Harderian gland tumors appear to be increasing in a nonstatistically significant manner in both sexes, likely due to increased survival. Since there were no statistically significant trends in tumor incidence, we do not feel that the apparent trends in the initial body weights and body weight gains have any impact on tumor formation. In addition, the Tg.rasH2 mice respond consistently to the positive control, urethane, as we have seen in previously published articles. 3,5
Thus, the retrospective analysis of the data compiled from studies conducted between 2004 and 2013 shows that despite declining initial body weights and increasing trend in body weight gains, there are no increasing trends in mortality and the incidence of spontaneous tumors. Due to lack of drifts in the mortality and incidence of spontaneous tumors, the Tg.rasH2 model appears to be genetically stable and the interpretation of the Tg.rasH2 studies can be made with greater confidence.
Footnotes
Author Contributions
M. G. Paranjpe contributed to conception, acquisition, analysis, and interpretation, drafted the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. M. D. Denton contributed to analysis, drafted the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. T. Vidmar, contributed to analysis and interpretation, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. R. H. Elbekai contributed to conception and design, acquisition, analysis, and interpretation, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
