Abstract
Risk assessment of chemical mixtures has emerged as a focus of research efforts, but traditional toxicology testing in mammals is costly, time-consuming, and subject to ethical scrutiny in the context of recent trends to reduce reliance on animal testing. In this review, which is a summary of presentations given at a workshop in Havana, Cuba, in April 2019, we survey the utility of zebra fish as an alternative laboratory model in whole-mixture and component-based testing, as well as in vitro modeling in 3-dimensional organotypic cultures from primary human cells cultured at the air–liquid interface and organ-on-a-chip platforms. Finally, we discuss the complexities of assessing the dynamics and delivery of multispecies liquid aerosol mixtures along the human respiratory tract, with examples of alternative and computational approaches to aerosol dosimetry. The workshop contributed to the professional development of Cuban toxicologists, an underserved segment of the global scientific community, delivering a set of tools and recommendations that could potentially provide cost-effective solutions for scientists with limited research resources.
Introduction
Environmental chemicals are typically found in mixtures and not singly. Industrialization has led to the constant synthesis and release of new and complex chemical mixtures into the air, soil, and water. Assessing the potential risk of these mixtures has become an area of major focus for researchers in toxicology and public health. 1 Evaluating the risk can involve a whole-mixture approach or a component-based approach, and the evaluation proceeds from hazard identification and dose–response assessment to exposure quantification and risk characterization. Selecting an appropriate approach to the quantitative assessment of the health hazard from chemical mixtures depends on the availability of toxicity data for a given mixture, data on a similar mixture, or data on the individual components of the mixture. 2,3
Challenges in assessing the potential risk from chemical mixtures involve limited time and resources for the thousands of mixtures that should be evaluated, and the influence of ambient conditions on the evolution of mixtures must also be considered. The recent trend to reduce reliance on animal testing has promoted the use of alternative animal models and in vitro and in silico methods. These methods are increasingly being leveraged in hazard identification and facilitate controlled testing conditions, a high level of standardization, and a reduction in interexperiment variability. They are also low cost, use only small amounts of material, and reduce the need for animal testing. Advanced alternative methods are also used for safety assessments of consumer products. 4 Protocols for testing mixture toxicity are being developed for organisms such as zebra fish, Drosophila, and Caenorhabditis elegans for cell cultures and organ-on-a-chip systems and for computational modeling and prediction of the pharmacokinetics and pharmacodynamics of these mixtures.
This review is a summary of some presentations given at the workshop “Alternatives to Animal Use in Risk Assessment of Mixtures,” which took place in April 2019 in Havana, Cuba. The workshop was hosted by the toxicology special interest group of the Cuban Society of Laboratory Animal Science.
Zebra Fish (Danio rerio) as an Alternative Model for Studying the Toxicity and Safety of Mixtures
Zebra fish assays are rapid, cost-effective, and enable automated high-content and high-throughput screening in aquatic risk assessment. 5 This alternative animal model is used regularly in toxicity and safety studies because of its characteristics and the ease of cultivation, 6 and only a small quantity of the exposure agent or mixture is required for screening a large number of zebra fish and comparing the effects. Image analysis software packages can capture developmental phenotypes and provide objective scoring, allowing for comparison of toxicant effect patterns. 7 These assays can test for numerous toxic end points of single or multiple chemical agents. Zebra fish are well suited to mixture assessment because of the ease with which they can be exposed to multiple agents. 8 Comparative testing can also be conducted in zebra fish by first assessing the toxicity of each component and then the toxicity of the mixture as a whole to determine the types of interactions, if any, between components. These could be classified as less than additive (antagonism, inhibition, or masking) or more than additive (synergism or potentiation).
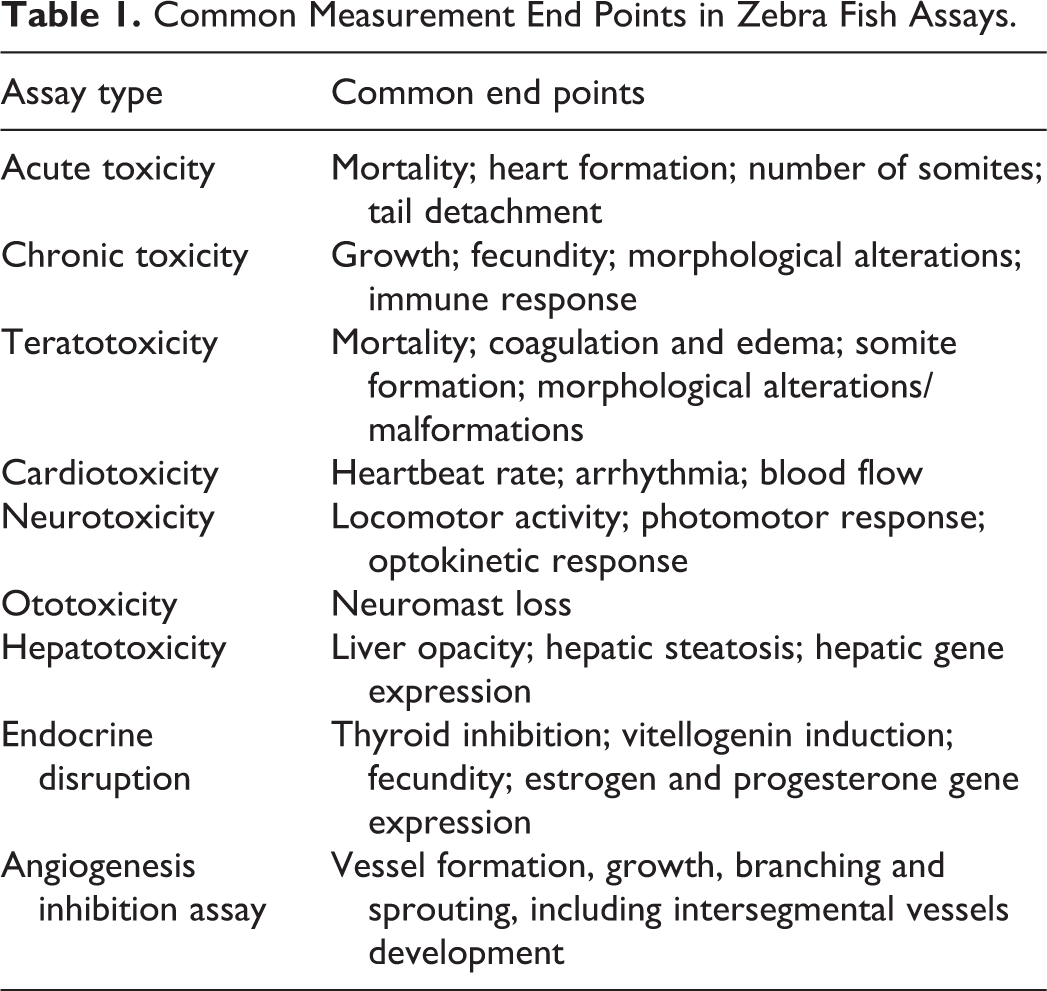
Zebra fish have been used to evaluate the mixture toxicity of chemical classes such as polycyclic aromatic hydrocarbons, 9 pesticides, 10 surfactants, 11 metals, 12 or a combination of pollutants from multiple classes. 13 Table 1 lists some specific end points that are commonly measured in zebra fish assays.
Common Measurement End Points in Zebra Fish Assays.
Mixture toxicity testing in zebra fish is relevant to understanding the hazardous effects of environmental chemicals on both humans and aquatic organisms. One major recent topic of research in these populations is the effect of pharmaceuticals and personal care products that are excreted or washed off into wastewater but remain viable after municipal water purification and are then discharged into open bodies of water. For instance, Galus and colleagues used zebra fish to test the effects of a 6-week exposure to a mixture of the drugs acetaminophen, carbamazepine, gemfibrozil, and venlafaxine. They reported impaired oocyte development, apoptosis, and renal toxicity in adult zebra fish and increased mortality and developmental abnormalities in embryos. 14 Such findings of adverse effects of chemical mixtures in aquatic ecosystems have implications for the health of multiple species, including humans.
Advanced In Vitro Approaches for Comparative Assessment of Heated Tobacco Products
Smoking tobacco causes a number of serious diseases, such as chronic obstructive pulmonary disease and lung cancer, and increases the risk of early death. Philip Morris International is developing novel products with the potential to reduce the individual risk and population harm caused by cigarettes. Tobacco products are complex mixtures of compounds: many chemicals known to cause harm are generated by the high temperatures of combusting tobacco and heat-not-burn products, such as heated tobacco products, heat the tobacco at lower temperatures, thus producing an aerosol with lower levels of the harmful and potentially harmful compounds found in cigarette smoke. Recently, a study stated that electronic cigarettes could be a unique harm reduction to prevent smoking relapse and that vaping can be a promising substitute for cigarette smoking that reduces the harm from tobacco smoking. 15 However, the safety of electronic cigarettes is highly debated. Extensive and rigorous scientific testing is carried out to determine whether these products reduce the risk of smoking-related diseases as compared to continued smoking. Testing the toxicity of these complex mixtures must take into account the effects on the entire organism while reducing the cost and time required by leveraging high-throughput experimental technologies, integrating laboratory and computational sciences, and describing biological phenomena using mathematical models.
The field of systems toxicology integrates classic toxicology with a quantitative analysis of the molecular and functional changes that occur across multiple levels of biological organization to achieve a detailed mechanistic and dynamic understanding of toxicological processes. Systems toxicology is a tool for analyzing transcriptome and proteome data and elucidating mechanistic pathways by computing response profiles and identifying perturbed biological networks and probable tissue-level effects. 16 This enables interspecies and intersystem translation, which is essential for effective risk assessment. 17 Philip Morris International uses systems toxicology to quantify the risk reduction potential of modified risk tobacco products 18 in vitro and in vivo; develop methods for comparing the biological impact of cigarette smoke, smoking cessation, and aerosol from reduced risk products; and obtain insights into the mechanisms underlying cigarette smoke-induced diseases. This involves a detailed study of the biological processes that govern molecular changes, cellular and tissue alterations, and the consequent pathophysiological changes. Biological networks that model biochemical, transcriptional, epigenomic, and metabolomics processes, as well as protein–protein interactions, are leveraged to investigate exposure effects from mixture characterization to quantitative assessment by computing systems response profiles, identifying perturbed biological networks, and calculating the product’s biological impact (Figure 1). 16
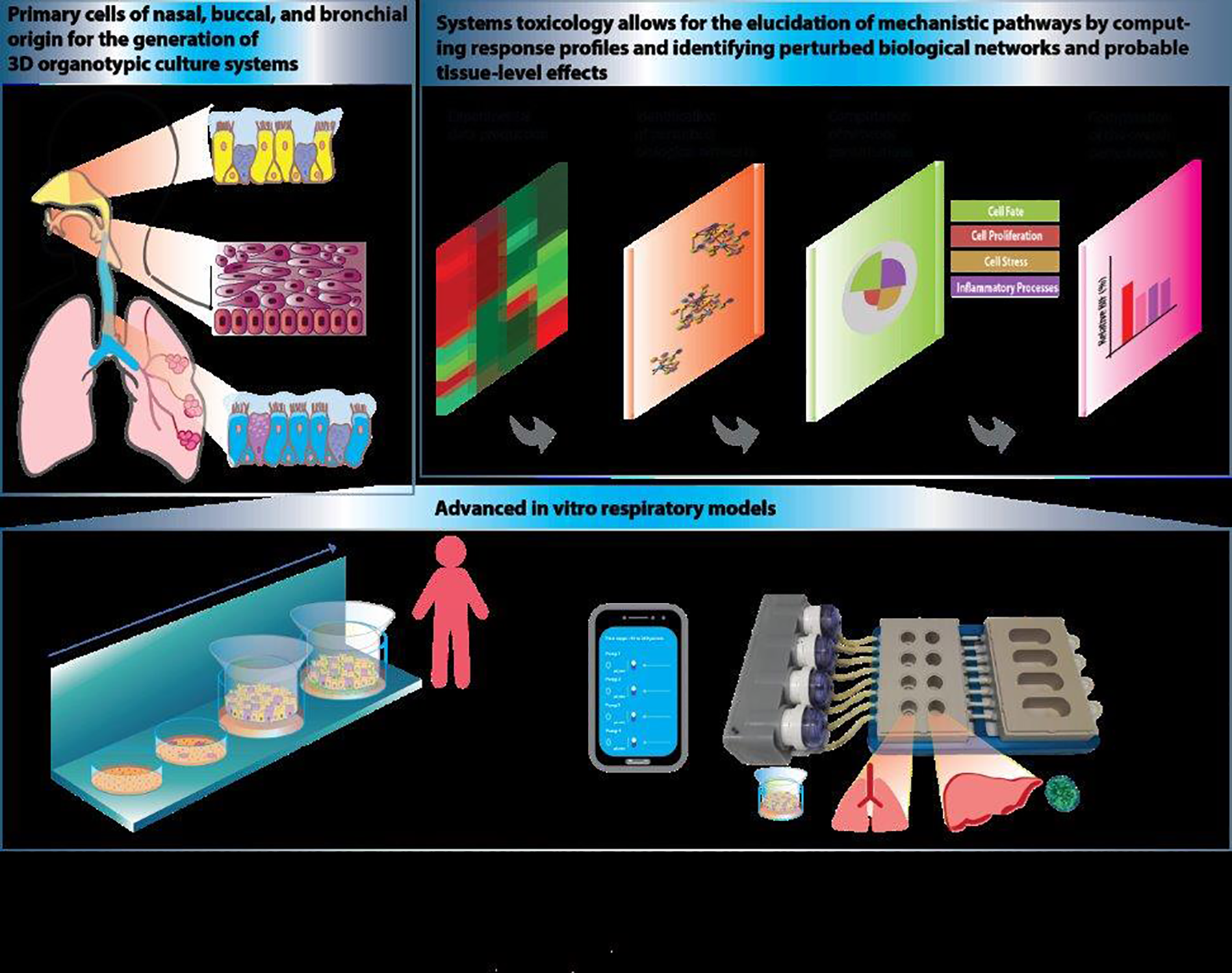
Inhalation exposure studies leveraging advanced in vitro respiratory models and systems toxicology. Primary epithelial cells of nasal, buccal, and bronchial origin can be used to generate 3-dimensional organotypic airway epithelial cultures that are grown at the air–liquid interface (upper left panel). Different from simple 2-dimensional monolayer epithelial cultures grown submerged in medium, the apical side for organotypic airway cultures is open to air, thus amenable for a direct exposure to aerosols (bottom panel). Other alternative test systems for assessing the toxicity of complex mixtures are organ-on-a-chip devices. We have developed a combined lung/liver-on-a-chip platform involving continuous medium circulation to enable the study of organ–organ cross talk (lower panel). Identification of perturbed biological impact can be facilitated by a systems toxicology approach (upper right panel). We use the approach to analyze transcriptome and proteome data, to elucidate mechanistic pathways by computing response profiles and identifying perturbed biological networks, and finally to calculate the product’s biological impact.
Human 3-dimensional organotypic airway epithelial cultures are derived from primary cells and can be used to study the impact of exposure to complex aerosols in vitro. Unlike 2-dimensional monolayer epithelial cultures that are grown submerged in medium, organotypic airway epithelial cultures are grown at the air–liquid interface and thus can be directly exposed to the aerosol of interest on the apical side. 19 Nasal, buccal, and bronchial epithelial cultures have been used to characterize exposures and compare the biological effects of cigarette smoke and aerosol from a heat-not-burn product, the Tobacco Heating System 2.2 (THS2.2). Exposing the cultures to aerosol from THS2.2 results in the generation of fewer harmful and potentially harmful constituents, reduced induction of inflammatory mediators, and fewer differentially expressed genes than exposure to cigarette smoke at comparable nicotine concentrations. Moreover, morphological tissue damage was not observed in nasal, buccal, or bronchial cultures exposed to THS2.2 aerosol, 19 unlike the tissue damage observed in these cultures following exposure to cigarette smoke.
Another alternative testing method for assessing the toxicity of complex mixtures harnesses organ-on-a-chip devices. These are in vitro systems of 3-dimensional human tissues that are cultured with a continuous medium circulation to mimic physiological conditions. Organ-on-a-chip platforms provide human-relevant physiological exposure response data using in vitro methods. Several models can be connected on one device, enabling the study of organ–organ cross talk following a chemical exposure. 20 We previously constructed a combined lung/liver-on-a-chip platform with liver spheroids and normal human epithelium cultured at the air–liquid interface and composed of multiple cell types (Figure 1). The lung tissues remained stable for up to 6 months in culture and exhibited active ciliary beating, while the liver spheroids displayed strong and stable xenobiotic metabolism for up at 6 weeks and secreted albumin and transferrin. On the chip, both tissues exhibited stable characteristics for at least 28 days. The platform was validated against bronchial tissues cultured without liver spheroids and showed that liver-dependent metabolism affected the toxicity of aflatoxin to bronchial tissue. 20
Challenges remain in the toxicity assessment of electronic cigarettes. There is a lack of standards in selecting chemicals to monitor, as the list of harmful and potentially harmful constituents found in cigarettes may not be applicable to electronic cigarettes. 21 Analytical methods are not standardized, and some of the assays used may not offer the sensitivity and reproducibility required for allowing comparisons between studies. Moreover, the flavoring ingredients added to some products may be considered safe for ingestion, but their inhalational effects are unknown. 22,23 Finally, there are no universal standards on the protocol for aerosol generation; the puffing regimen and coil temperature are known to affect the generation of chemicals such as carbonyls, and the vaping topography is heterogeneous. When designing studies to assess the risk of complex mixtures, investigators must select physiologically relevant doses and chemicals, control the exposure conditions, and take into account variations in the chemical and physical characteristics of particles in mixtures.
Investigating the Dynamics and Delivery of Multispecies Liquid Aerosol Mixtures Along the Human Respiratory Tract
In vitro and in silico experimental dosimetry methods are gaining popularity as low-cost, efficient approaches for assessing the toxicity of complex aerosols. Information on the deposition of inhaled particles in the respiratory tract contributes to assessing the risk of liquid aerosol mixtures, such as those from electronic cigarettes. 24,25 Different approaches are available for characterizing the properties of liquid aerosol mixtures and their components and their deposition in the respiratory tract following inhalation. For liquid aerosols, calculations must take into account aerosol properties (including chemical composition, phase partitioning, particle size distribution, hygroscopicity, and individual species volatility). Furthermore, chemical composition as a superposition of individual species modulates the mixture properties (eg, vapor–liquid equilibrium) as a whole, causing difficulties in generalization of the obtained assessment data. Current research challenges in the field of inhaled aerosol dosimetry include the limited availability of detailed anatomical data and aerosol deposition data sets linked with aerosol chemistry, lack of direct links of exposure and deposition models to health end points, and a paucity of guidelines for validating dosimetry models in hazard and risk assessment. 26
Aerosol generation, administration, and inhalation processes affect and are affected by the physicochemical properties of the liquid mixture used to form the aerosol particles as well as thermodynamic conditions. In the process of aerosolization (particularly for thermally driven aerosols generated from supersaturated vapors via the consecutive nucleation and condensation/evaporation processes), the composition of the initial liquid mixture can be affected by the formation of additional products of thermal degradation (eg, carbonyls). The generation of liquid aerosols involves considerations of mixture component solubility, stability, and volatility. Aerosol particle evolution occurs during transport and deposition in the respiratory tract; this is often governed by spatial and temporal thermodynamics of evaporation/condensation, coalescence/breakup, and finally, drift processes. Accurate dose calculation must account for aerosol physics, inhalation topography, and lung morphology and take into account aerosol particle filtration (losses), such as those that occur in the oral cavity. 25 The exposure measure, mathematically expressed by multiplication of the exposure duration, aerosol concentration, and respiratory minute volume as an inhalation sampling parameter, is not sufficient for calculating the delivered dose. Apart from aerosol evolution along the respiratory tract, multiple mechanisms are responsible for the aerosol deposition, including impaction, sedimentation, interception, electrostatic precipitation, and diffusion. These mechanisms and their spatial and temporal interactions along the respiratory tract add to the complexity of aerosol dosimetry to calculate the effective lung deposition efficiency.
Computational models for describing the deposition of inhaled aerosols are typically categorized as simplified airway models, that is, whole-lung models (eg, multiple-path particle dosimetry [MPPD] model) or computational fluid dynamics-based models (eg, AeroSolved) that account for the geometrical complexity of the airways (Figure 2). 26 The MPPD model is based on the structure of the bronchial tree but is limited by the lack of correlating empirical data and was developed for nonevolving particles. On the contrary, the computational fluid dynamics models can reach a much detailed level of complexity but still suffer from the limited computational feasibility for the real-life scenario simulations.
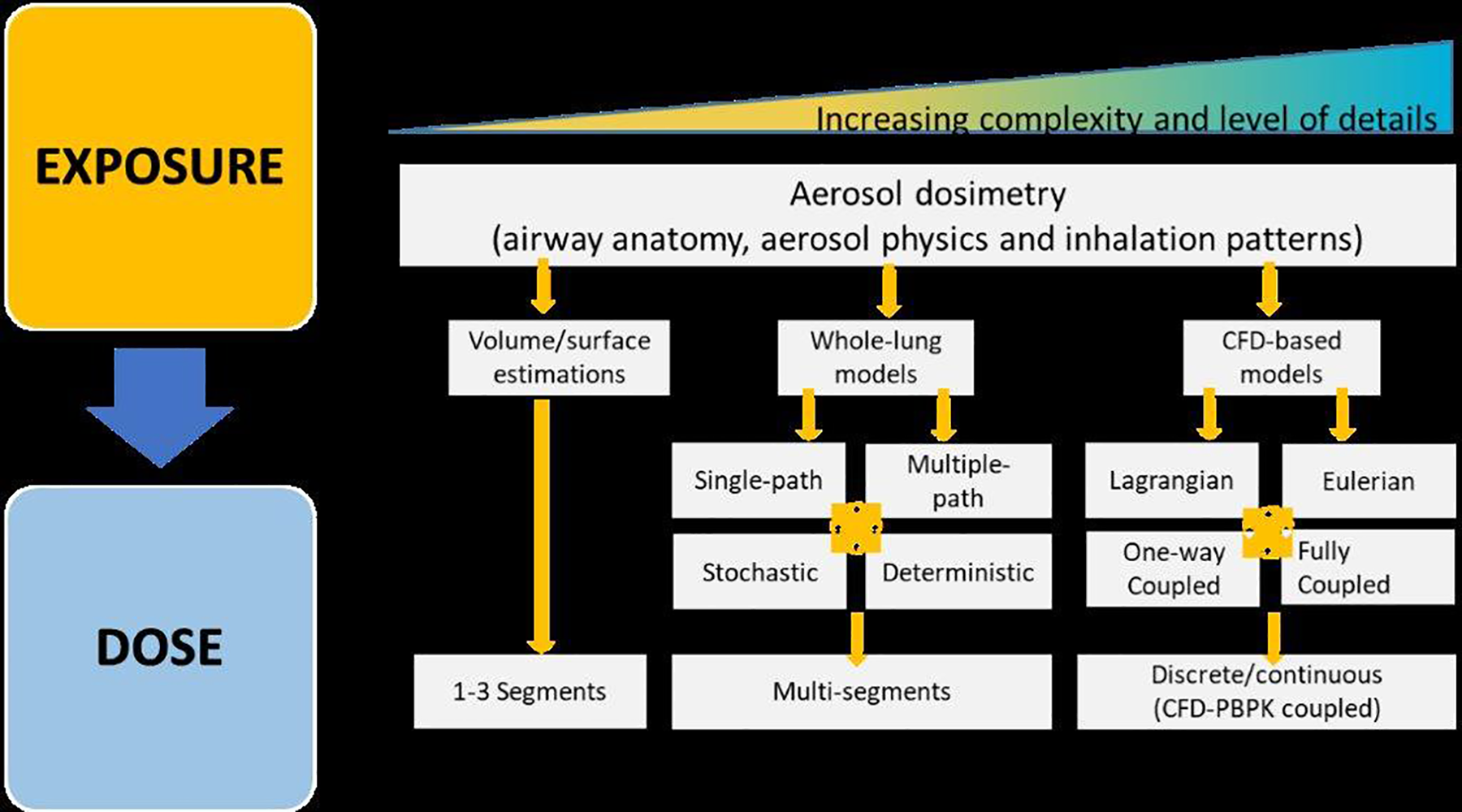
Dosimetry considerations for inhaled aerosols in the respiratory tract. Classification of in silico approaches for exposure to dose predictions at various levels of complexity and granularity in delivering the regional aerosol dosimetry data: general methods classified into (1) volume/surface estimations, (2) whole-lung models, and (3) computational fluid dynamics models.
Computational in vivo dosimetry approaches, based on whole-lung modeling, have focused on animal lung morphometry and respiratory physiology but still suffer from limited knowledge, validation, and data concerning upper respiratory tract filtration. The generated findings are not directly translatable to human-relevant exposures (eg, rodents are obligate nose-breathers). Segmented cast models are useful in determining the regional deposition of aerosol if the aerosol constituents are not highly volatile, but they exist for only a limited number of animal strains and are often poorly validated because of a lack of experimental data. Computational models based on the modeling of aerosol flows (transport, evolution, and deposition) can be used to refine the whole-lung modeling approaches and are the subject of ongoing development and research. These models incorporate higher levels of aerosol complexity and details and are often used to develop, characterize, and validate inhalation devices, aerosol generators, and aerosol delivery and exposure systems. However, their application as mentioned earlier is still limited because it requires extensive computational capabilities to simulate even single-case scenarios.
Organotypic respiratory cultures offer an in vitro approach for assessing the risk of exposure to the multispecies aerosols and mixtures from which they are formed. Biomarkers of exposure, response, and disease can be studied in bronchial epithelium, and airway basal cells have been shown to differentiate to specific cell types when cultured at the air–liquid interface. 27 Aerosol exposure systems for cell cultures offer continuous, uniform aerosol dilution and constant unidirectional aerosol flow but cannot control aerosol evolution and particle losses during transport to mimic human inhalation dynamics. These systems are a useful tool in fundamental research into biological responses, but the findings cannot be easily translated into real-life case scenarios.
Overall, aerosol dosimetry is complex and, typically, cannot account for variations in breathing patterns; findings in rodents cannot be translated to human-relevant recommendations. Scaling of aerosol and airway diameters is critical, and the deposited dose and its localization may vary over time. Dosimetry calculations may be improved by further development of whole-lung modeling approaches in parallel with supporting experimental validation and computational fluid dynamics models. Great care must be taken in assessment of toxicological exposure of evolving liquid mixtures as often their properties affecting dose are significantly modulated along the route from the aerosol generation site to the aerosol delivery.
Needs for Continuous Training and Development of Alternative Methods and Teaching Protocols in Cuba
TOXCAL is a special interest group of the Cuban Society of Laboratory Animal Science, a sister organization of the Society of Toxicology and the Hispanic Organization of Toxicologists. Its mission is to add knowledge and skills related to laboratory animal science, improve the expertise of toxicologists and/or laboratory animal science personnel, contribute to the excellence of safety research, and facilitate the mutual recognition of results within the framework of current ethical standards (Figure 3).

The TOXCAL team with our Cuban colleagues in Havana, Cuba, 2019.
TOXCAL comprises approximately 183 specialists in different research areas, located in all the national institutions, faculties, and research centers in Cuba. Approximately 75% hold a Master’s or Doctorate degree. The main strength of TOXCAL is this highly specialized and qualified membership, which promotes the spread of knowledge and current approaches for safety evaluations via formal collaborative agreements among universities and research institutions.
This program was developed to overcome the limitations and lack of knowledge in this field. A current objective of TOXCAL is to support training related to alternative methods to laboratory animal science. Among the differently accepted methods, the most important for immediate implementation as part of a long-term vision for toxicity testing are alternative models (zebra fish toxicity assays), in vitro assays (mainly tests of irritability, cell viability, and phototoxicity), in silico methods, and alternative tools.
The workshop “Alternatives to Animal Use in Risk Assessment of Mixtures,” hosted by TOXCAL, brought together professionals and academicians in this field to discuss the latest developments and emerging risks in occupational and environmental toxicology. The underserved laboratory animal research and toxicology communities in Cuba have many disadvantages, including a lack of financial support, unavailability of cell lines and specialized equipment, and a lack of certified animal facilities, specialized literature, and specialist training. The workshop was a great opportunity because it allowed members to review the technical basis for using animals for research purposes, particularly for toxicology studies, as well as new methods and alternative models to animal studies. Such methodologies for risk assessment are also essential elements in integrative national environmental policies regarding hazardous substances. The “Alternatives to Animal Use in Risk Assessment of Mixtures” workshop provided an excellent opportunity to assess the Cuban context within the toxicology field, facilitating discussion on the scope and challenges of alternative methods and models to the use of laboratory animals. As a result of this workshop, we anticipate initiatives in which different institutions, universities, and industry work together toward a common purpose of encouraging scientific exchange and collaboration within specific animal laboratory science and toxicology areas; collaboration within specific areas of focus throughout the toxicology community, providing opportunities to involve new members; establishment of TOXCAL as a reference group stimulating scientific exchange and better training, support, and knowledge among the toxicology community in Cuba, leading to a diversified portfolio of toxicological testing approaches level; and improved dissemination and application of information and expertise.
Summary
Assessing the hazard and risk of chemical mixtures and reducing the reliance on animal testing are 2 topics at the center of current optimization efforts in toxicological research. Nonmammalian organisms, in vitro studies, and computational algorithms to predict toxicity and component interaction have emerged as viable tools in these efforts. These are also cost-effective solutions for scientists with limited research resources. The workshop, which was hosted by the Cuban Society of Laboratory Animal Science, contributed to the professional development of Cuban toxicologists, who are an underserved segment of the global scientific community. The presentations delivered at the workshop were designed to disseminate knowledge and equip Cuban toxicologists with the skills and information necessary to contribute qualified and competent expertise in their professional capacity. The workshop also served to foster collaboration and partnership opportunities for researchers who lack financial support, as well as access to pertinent insights to establish alternative test methods for toxicological testing.
Footnotes
Authors’ Note
The information in this review was previously presented at the Alternatives to Animal Use in Risk Assessment of Mixtures Workshop, Havana, Cuba, on April 12, 2019.
Acknowledgments
The authors thank Edanz Medical Writing for providing writing support, which was funded by Philip Morris International. The authors are also grateful to Sorange Hernandez Fernandez for her contributions to this paper.
Author Contributions
A.W. Hayes, A. Muriana, A. Alzualde, D. B. Fernandez, A. Iskandar, M. C. Peitsch, A. Kuczaj, and J. Hoeng substantially contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Hayes served as a paid consultant to Philip Morris International.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing support was funded by Philip Morris International.
