Abstract
This is a safety assessment of glycerin as used in cosmetics. Glycerin functions as a denaturant, fragrance ingredient, hair conditioning agent, humectant, oral care agent, oral health-care drug, skin protectant, skin conditioning agent—humectant, and viscosity-decreasing agent. The Cosmetic Ingredient Review Expert Panel (Panel) reviewed relevant animal and human data. The Panel concluded that glycerin is safe as a cosmetic ingredient in the practices of use and concentration described in this safety assessment.
Introduction
This is a review of the available scientific literature and submitted unpublished data relevant to assessing the safety of glycerin as used in cosmetics. Glycerin is reported to function in cosmetics as a denaturant, fragrance ingredient, hair conditioning agent, humectant, oral care agent, oral health-care drug, skin protectant, skin conditioning agent—humectant, and viscosity-decreasing agent. 1
Much of the information in this safety assessment was obtained from summary information made available by the Organization for Economic Cooperation and Development Screening Information Data Set, the European Commission-European Chemicals Bureau, and the European Chemicals Agency. 2 -4
Chemistry
Definition and Structure
Glycerin (CAS no. 56-81-5) is the polyhydric alcohol that conforms generally to the structure in Figure 1. 1 The molecular formula is C3H8O3. Glycerin (also referred to as glycerol in the literature) is a simple triol, that is, it has 3 hydroxyl groups.
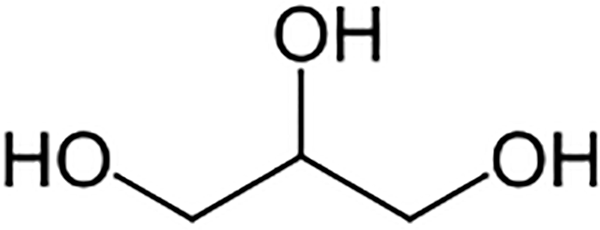
Glycerin.
Glycerin is naturally occurring in all animals and plant matter in combined form as glycerides in fats and oils or in intracellular spaces as lipids. 3 While the chemicals are identical, there is naturally occurring glycerin derived from plants and animals and synthetic glycerin obtained from nontriglyceride sources. 5
Physical and Chemical Properties
Glycerin is a clear, syrupy liquid (Table 1). 2,6 It can be in a crystallized state, but seldom is because of its tendency to supercool and the pronounced effect of small amounts of water in depressing the freezing point. 3
Chemical and Physical Properties of Glycerin.
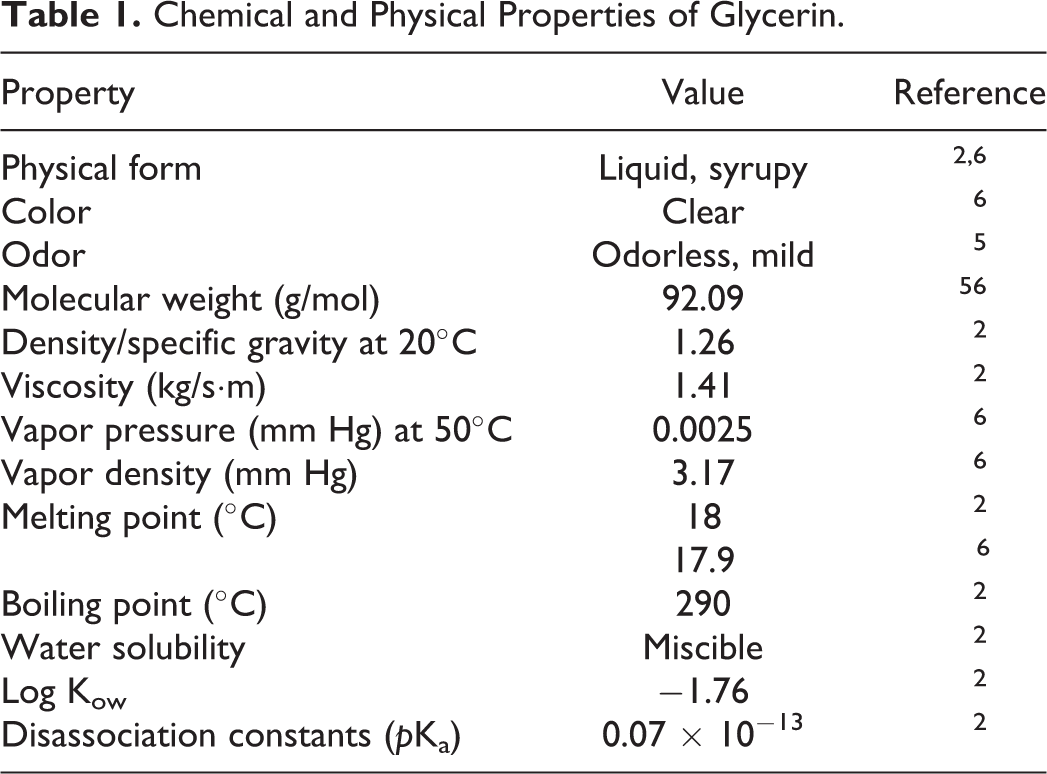
Glycerin has solvent properties similar to those of water and simple aliphatic alcohols. 5 The chemical is completely miscible with water, methanol, ethanol, and the isomers of propanol, butanol, and pentanol. Glycerin is also fully miscible with phenol, glycol, propanediols, amines, and heterocyclic compounds containing a nitrogen atom in the ring (eg, pyridine, quinoline). Glycerin is less soluble in acetone, diethyl ether, and dioxane and is almost insoluble in hydrocarbons, long-chain aliphatic alcohols, fatty oils, and halogenated solvents such as chloroform.
Method of Manufacture
Natural glycerin is obtained as a byproduct in the hydrolysis of fats and oils. 5 Multiple methods exist for the synthetic manufacture of glycerin. The starting materials for synthetic production of glycerin include allyl chloride, acrolein, propylene oxide, sugars, certain polyalcohols, fats, or epichlorohydrin. 5
In one method, allyl chloride is oxidized with hypochlorite to produce dichlorohydrin, which is then converted, without isolation, to epichlorohydrin by ring closure with calcium hydroxide or sodium hydroxide. 5 Epichlorohydrin is hydrolyzed to yield glycerin by heating to 80°C to 200°C with a 10% to 15% aqueous solution of sodium hydroxide or sodium carbonate at atmospheric pressure or overpressure. The yield of glycerin, calculated from allyl chloride, is 98%, obtained as a dilute (10%-25%) solution containing 5% to 10% sodium chloride and <2% other impurities. The aqueous glycerin solution is concentrated in a multistage evaporation plant under vacuum to produce a glycerin concentration of >75%, after separating precipitated sodium chloride. The glycerin solution is then distilled under high vacuum and the co-distilled water is separated by fractional condensation. The glycerin is treated further to remove color impurities and odorous material; this can be accomplished, for example, using activated carbon.
A second method involves the oxygenation of propene to acrolein, which is then reduced under Meerwein-Ponndorf-Verley conditions to yield allyl alcohol. 5 The allyl alcohol is then epoxidated with hydrogen peroxide and the resulting glycidol is hydrolyzed to produce glycerin.
Impurities
The US Pharmacopeia-National Formulary standards state that the amount of any individual impurity in glycerin cannot exceed 0.1% and that the total for all impurities, including diethylene glycol and ethylene glycol, must not exceed 1%. 7
The US Food and Drug Administration (FDA) notes that glycerin is a byproduct of biodiesel fuel produced from the Jatropha species of plant. 8 There is a possibility that toxic impurities, including phorbol esters, may be present in glycerin produced this way. Conventional impurity tests may not detect these toxins, and glycerin from this source should therefore not be used in human and animal food, medical products, cosmetics, and other FDA-regulated products. The FDA advises industry to be aware of the potential for substitution or use of oils, glycerin, and proteins derived from the Jatropha plant.
A cosmetics raw material supplier reported that glycerin is 95% to 99.5% pure. 2 Impurities are water and trace levels of polyglycerol.
Use
Cosmetic
Glycerin is reported to function in cosmetics as a denaturant, fragrance ingredient, hair conditioning agent, humectant, oral care agent, oral health-care drug, skin protectant, skin conditioning agent—humectant, and viscosity-decreasing agent. 1
The FDA collects information from manufacturers on the use of individual ingredients in cosmetics as a function of cosmetic product category in its Voluntary Cosmetic Registration Program (VCRP). In 2014, glycerin was the third most frequently reported ingredient in the VCRP database (after water and fragrance). Glycerin was reported to be used in 15,654 cosmetic products; 10,046 are leave-on products, 5,441 are rinse-off products, and 167 products are diluted for the bath (Table 2). These uses include 862 products for use near the eye, 160 lipsticks, 369 hair dyes and colors, 1,259 bath soaps and detergents, 7,756 skin care products, and 244 suntan preparations. 9 Glycerin is reported to be used in 125 baby products. Two uses for anhydrous glycerin (in an eye makeup preparation and a moisturizer) were reported in the VCRP; these uses were merged with the glycerin uses.
a Totals = rinse-off + leave-on + diluted for bath product uses.
b Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
c Aerosol hair spray 0.11% to 10%; pump hair spray 0.11% to 30%; spray face and neck products 0.5% to 10%; spray body and hand product 0.006% to 5%; spray moisturizers 3.3%; aerosol suntan products 6%; pump spray suntan products 4.1% to 10%.
d It is possible these products may be sprays, but it is not specified whether the reported uses are sprays.
e Not specified whether a powder or a spray, so this information is captured for both categories of incidental inhalation.
f It is possible these products may be powders, but it is not specified whether the reported uses are powders.
g Not spray.
h Aerosol spray 0.019% to 0.05%; pump spray 2% to 4%.
A survey was conducted by the Personal Care Products Council (Council) of the maximum use concentrations for this ingredient. 10 Glycerin is reported to be used at concentrations up to 79.2% in leave-on products, 99.4% in rinse-off products, and 47.9% in products diluted for the bath. It is used at up to 21% in baby products, 40.6% in eye lotions, 25% in perfumes, 47.3% in hair grooming aids, 68.6% in oral hygiene products, 99.4% in skin cleaning products, and 17.9% in suntan preparations.
Glycerin was reported to be used in aerosol/spray products that include hair sprays at concentrations at up to 30% (in propellant spray products at concentrations up to 10% and in pump spray products up to 30%), pump deodorants at up to 4%, face and neck products at up to 10%, body and hand products at up to 5%, moisturizing products at up to 3.3%, and suntan products at up to 10% (in propellant spray products at up to 6% and in pump spray products up to 10%). These propellant/pump spray products could possibly be inhaled. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles below 10 µm compared with pump sprays. 11 -14 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) in any appreciable amount. 11,13 There is some evidence indicating that deodorant spray products can release substantially larger fractions of particulates having aerodynamic equivalent diameters in the range considered to be respirable. 11 However, the information is not sufficient to determine whether significantly greater lung exposures result from the use of deodorant sprays, compared to other cosmetic sprays.
Noncosmetic
Code of Federal Regulations (CFR) listings concerning glycerin are provided in Table 3. For example, glycerin is considered generally recognized as safe (GRAS) by the FDA for its use in food packaging and it is a multiple-purpose GRAS food substance when used in accordance with good manufacturing practices [21CFR182.90; 21CFR182.1320]. Also, glycerin is approved for use in over-the-counter drugs, such as anorectal drug products, dermal protectants (up to 45%), in ophthalmic drug products (up to 1%), and in oral health-care products [21CFR346.14; 21CFR347.10; 21CFR349.12].
Code of Federal Regulations Listings Concerning Glycerin From FDA, EPA, and ATF.

Abbreviations: ATF, Alcohol and Tobacco Tax and Trade Bureau; CFR, Code of Federal Regulations; EPA, Environmental Protection Agency; GRAS, generally recognized as safe; USP, US Pharmacopeia.
Glycerin functions as a humectant, solvent, cake icing component, confectionary component, bodying agent, and plasticizer for foods. 3
Glycerin has been administered orally and/or intravenously to reduce intracranial pressure caused by various medical conditions. 15 Glycerin has been used to reduce brain volume for neurosurgical procedures. It is also used as the active ingredient in laxative products (ie, glycerin suppositories).
Glycerin is used in paints, lacquers, and varnishes; polymers; tobacco products; absorbents and adsorbents; adhesives and binding agents; antifreezing agents; cleaning agents and disinfectants; explosives; heat transferring agents; pesticides; and softeners. 3 It is an intermediate and monomer in resins, polyols, and polyurethanes. 2
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Glycerin is rapidly absorbed from the intestine and the stomach, distributed throughout the extracellular space, and excreted renally. 2,16,17 Glycerin is phosphorylated to α-glycerophosphate by glycerol kinase, predominantly in the liver (80%-90%) and kidneys (10%-20%), and incorporated via the standard metabolic pathways to form glucose (gluconeogenesis) and glycogen. 16,18 Glycerin kinase is also found in intestinal mucosa, brown adipose tissue, lymphatic tissue, lung, and pancreas. Glycerin may also combine with free fatty acids in the liver to form triglycerides (lipogenesis) that can be distributed to adipose tissues. The glycerin turnover rate is directly proportional to plasma glycerin levels. 19
Free glycerin is naturally present in human plasma. 20 Normal serum levels in adult humans range from 0.05 to 0.1 mmol/L. Urinary glycerol excretion is associated with plasma glycerol concentrations >0.327 ± 0.190 mmol/L.
Dermal/percutaneous
Data on dermal absorption, distribution, metabolism, and excretion of glycerin were not found in the published literature nor were unpublished data provided.
Oral
Orally administered glycerin is rapidly absorbed from the gastrointestinal tract, and peak serum concentrations occur within 60 to 90 minutes. 15 Glycerin is distributed throughout the blood. Glycerin generally does not appear in ocular fluids; however, it may enter the orbital sac when the eye is inflamed. It is not known if glycerin is distributed into milk. The elimination half-life of glycerin is approximately 30 to 45 minutes. Most orally administered glycerin is incorporated into body fat or brought into glycolysis or gluconeogenesis pathways (principally in the liver) via glycerokinase-catalyzed phosphorylation and oxidation to dihydroxyacetone phosphate, with ultimate conversion to carbon dioxide and water in the former pathway, and glucose and glycogen synthesis in the latter. Glycerin can also combine with free fatty acids to form triglycerides. Approximately 80% of glycerin metabolism takes place in the liver and approximately 10% to 20% in the kidney. The metabolism of glycerin to carbohydrate produces 4.3 cal/g glycerin. Most of an oral dose of glycerin is metabolized within 2.5 hours. Approximately 7% to 14% of an oral dose of glycerin is excreted unchanged in urine during this time.
Orally administered glycerin elevates the osmotic pressure of the plasma to such an extent that water from the extravascular spaces is drawn into the blood. The osmotic effect of glycerin produces a decrease in intraocular pressure (IOP) by reducing the volume of intraocular fluids in a manner completely independent of the normal ocular fluid inflow and outflow mechanisms. The extent of IOP reduction varies with the dose of glycerin and the etiology and degree of the increased pressure. Reduction in IOP reaches its maximum within 30 minutes to 2 hours and may persist for 4 to 8 hours. In general, reduction in IOP is greatest when the pretreatment IOP is high. The osmotic effect of glycerin may also produce tissue dehydration and a decrease in cerebrospinal fluid pressure. Glycerin produces only very slight diuresis in healthy individuals receiving a single oral dose of 1.5 g/kg or less. 15
Acute ingestion of glycerin (1 mL/kg in water) in human male patients led to an increase in plasma glycerides. In human female patients, the oral administration of glycerin (1 mL/kg in water) resulted in no change in plasma glyceride concentrations. When glycerin (1 mL/kg/d in 3 doses) was orally administered for 42 days, increased serum glyceride concentrations were observed in both sexes; however, the increase was greater in men. 21
Toxicological Studies
In a survey of 4 laboratories of the use of vehicles for in vivo experiments, the highest no-observed-adverse-effect levels (NOAELs) of various routes of administration were assembled. 22 The oral NOAELs for glycerin were 15 g/kg for rats and 500 mg/kg for guinea pigs for 1 month and 500 mg/kg for 90 days for mice (Table 4). The highest subcutaneous NOAELs were 10 mg/kg in rats and mice.
Highest NOAEL Reported in a Survey of 4 Research Organizations for Glycerin Used as a Vehicle. 22
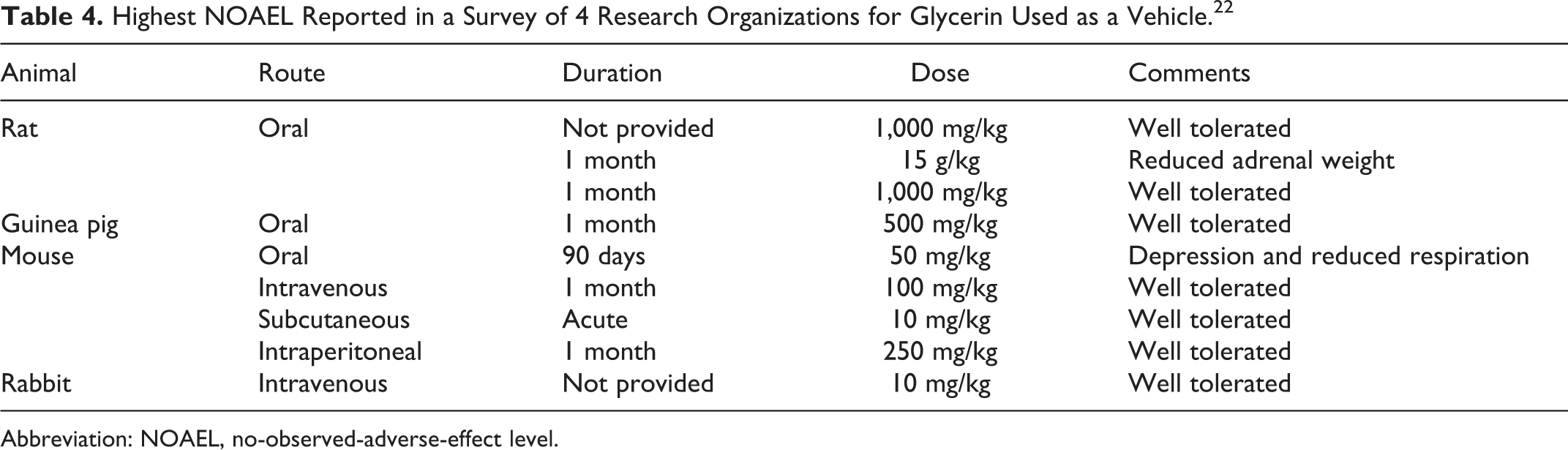
Abbreviation: NOAEL, no-observed-adverse-effect level.
Acute Toxicity
Nonhuman
Reported oral median lethal dose (LD50) values of glycerin ranged from 2,530 to 58,400 mg/kg in rats; there were no deaths at 24,000 mg/kg in one study (Table 5). 3,18,23 -27 Reported oral LD50 values of glycerin were 4,090 to 38,000 mg/kg in mice, 27,000 mg/kg in rabbits, and 77,500 mg/kg in guinea pigs. 3,18,23,24,26 -29 The dermal LD50 value of glycerin was reported to be >21,900 mg/kg in rats and >18,700 mg/kg in rabbits. 3,26 The approximate value for the median lethal time (time until death) after exposure (Lt50) for rats was determined to be 423 minutes for exposure to glycerin vapors at 11.0 mg/L. 4 Reported intraperitoneal LD50 values of glycerin were 4,420 to 10,100 mg/kg in rats and 8,600 to 9,500 mg/kg in mice. 3 Reported subcutaneous LD50 values of glycerin were 100 mg/kg in rats and 91 to 100 mg/kg in mice. 3,29 Reported intravenous LD50 values of glycerin were 5,200 to 6,600 mg/kg in rats, 4,250 to 6,700 mg/kg in mice, and 53,000 mg/kg in rabbits. 3
Acute Toxicity Studies of Glycerin.

Oral—Human
The oral lowest lethal dose (LDLO) of glycerin was reported to be 1,428 mg/kg for humans. 3 There were no signs of toxicity when patients (n = 10 men, 4 women) were orally administered glycerin (30 mL; 95% purity administered in orange juice) after each of 3 meals in 1 day. 2
When used as a drug, it was reported that the adverse effects following the oral administration of glycerin (dose not provided) include mild headache, dizziness, nausea, vomiting, thirst, and diarrhea. 15 Headache may result from cerebral dehydration, which may be prevented or relieved by having the patient lie down during and after treatment. Hypotonic fluids relieve thirst and headache caused by the dehydrating action of glycerin.
Repeated Dose Toxicity
Oral—Nonhuman
Repeated dose toxicity studies are summarized in Table 6. Undiluted glycerin caused a dose-dependent increase in the number of animals showing hyperemia, petechial hemorrhage, and erosions in the gastrointestinal tract. 30 In short-term feeding experiments, glycerin at 20% for 4 weeks in feed produced no adverse effects, but at 53.4%, an increase in kidney weights and increased liver enzymes were observed. 4,30 -32 The NOAEL was between 115 and 2,300 mg/kg when administered in water to rats for 44 days. 33 Calcified masses were observed in kidney tubules between the cortex and medulla in 3 of 5 rats administered either natural or synthetic glycerin (3,335 mg/kg/d) in drinking water for 6 months. 28 When glycerin was administered in the diet for 2 years, feed consumption was slightly increased in males fed synthetic glycerin versus those fed naturally sourced glycerin. There were no treatment-related effects in organ weights and gross pathology. 26
Repeated-Dose Toxicity Studies.
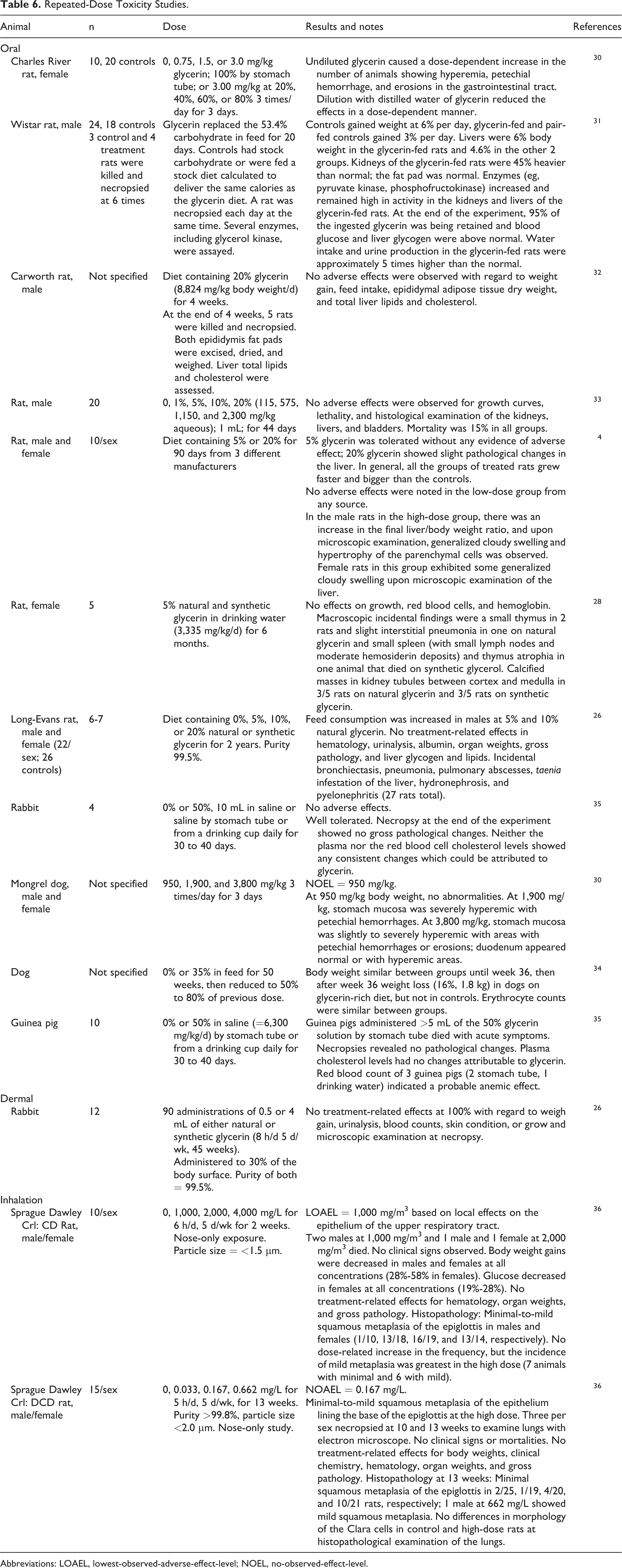
Abbreviations: LOAEL, lowest-observed-adverse-effect-level; NOEL, no-observed-effect-level.
The no-observed-effect-level (NOEL) in mongrel dogs was 950 mg/kg/d when glycerin was orally administered for 3 days (Table 6). At 3,800 mg/kg/d, the mucosa of the stomach was severely hyperemic with petechial hemorrhages. 34 Mongrel dogs experienced weight loss after 36 weeks when glycerin (35%) was incorporated into their feed. The weight loss continued when the glycerin content was reduced by 50% to 80% for the remainder of a 50-week study. 30
There were no pathological changes in guinea pigs (n = 10) orally administered glycerin (6,300 mg/kg/d) for 30 to 40 days (Table 6). 35
Oral—Human
There were no signs of toxicity or effects on blood or urine production when patients (n = 10 males, 4 females) were orally administered glycerin (approximately 1.3-2.2 g/kg/d; glycerin in orange juice with meals) for 50 days. 3 The NOAEL was ≥2.2 g/kg/d. No further information was provided. There were no adverse effects observed in patients (n = 14) administered glycerin (30 mL, neat) 3 times daily with each meal for 50 days. 16
Dermal—Nonhuman
There were no treatment effects when glycerin (100%; 0.5-4 mL) was administered to 30% of the body surfaces of rabbits 5 d/wk for 45 weeks (Table 6). 26
Inhalation—Nonhuman
The inhalation lowest-observed-adverse-effect-level (LOAEL) was 1,000 mg/L for glycerin administered nose only 6 h/d, 5 d/wk for 2 weeks in Crl: DCD Sprague Dawley rats, based on local effects on the epithelium of the upper respiratory tract (Table 6). 36
The inhalation NOAEL was 0.167 mg/L for glycerin-administered nose only for 5 h/d, 5 d/wk for 13 weeks in Crl: DCD Sprague Dawley rats (Table 6). 36 There was minimal squamous metaplasia of the epiglottis in 2 of 25, 1 of 19, 4 of 20, and 10 of 21 rats at 0, 33, 167, and 662 mg/L, respectively; 1 male in the high-dose group showed mild squamous metaplasia.
Reproductive and Developmental Toxicity
In a 2-generation reproductive study in rats (n = 10 per sex), the administration of glycerin (0%, 20%; ∼2,000 mg/kg/d in drinking water) for 8 weeks before mating until weaning of pups produced no adverse effects on the reproductive efficiency of the parents (F0 generation), or the growth, fertility, or reproductive performance of the untreated F1 generation. 2 No histological changes occurred in the tissues of either the F1 or F2 generations. The onset of estrus cycles, weight gain, and microscopic observations of the endocrine organs were comparable to those of the controls in both the F1 and the F2 generation. In the F0 generation, all 10 females became pregnant with similar litter size as controls (9.0 vs 8.1). In the F1 generation, 9 of 10 females became pregnant.
When glycerin (13.1, 60.8, 282, and 1,310 mg/kg/d) was administered by gavage to Wistar rats (n = 25-28) on days 6 through 15 of gestation, there were no adverse effects observed in the dams. 37 The NOAEL for maternal toxicity and teratogenicity was 1,310 mg/kg/d. The numbers of pregnancies were 23 of 25, 24 of 25, 22 of 28, and 22 of 25 for 13.1, 60.8, 282, and 1,310 mg/kg/d, respectively, and 21 of 25 for controls. The number of implantations, resorptions, litter sizes, weights, and sex ratio were similar among groups, as were the incidences of external, visceral, and skeletal abnormalities.
When glycerin (12.8, 59.4, 276, and 1,280 mg/kg/d) was administered by gavage to CD-1 mice (n = 25) on days 6 through 15 of gestation, there were no adverse effects observed in the dams. 37 The NOAEL for maternal toxicity and teratogenicity was 1,280 mg/kg/d. The numbers of pregnancies were 14 of 15, 12 of 15, 10 of 18, 13 of 20, and 13 of 15 for controls, 12.8, 59.4, 276, and 1,280 mg/kg, respectively. The number of implantations, resorptions, litter sizes, weights, and sex ratio were similar among groups as were external, visceral, and skeletal abnormalities.
When glycerin (11.8, 54.8, 254.5, and 1,180 mg/kg/d) was administered by gavage to Dutch-belted rabbits (n = 25) on days 6 through 18 of gestation, there were no adverse effects found in the dams. 37 The NOAEL for maternal toxicity and teratogenicity was 1,180 mg/kg/d. The numbers of pregnancies were 22 of 25, 23 of 25, 20 of 25, 22 of 25, and 21 of 25 for controls, 11.8, 54.8, 254.5, and 1,180 mg/kg/d, respectively. The number of implantations, resorptions, litter sizes, weights, and sex ratio were similar among groups, as were external, visceral, and skeletal abnormalities.
Male Fertility—Nonhuman
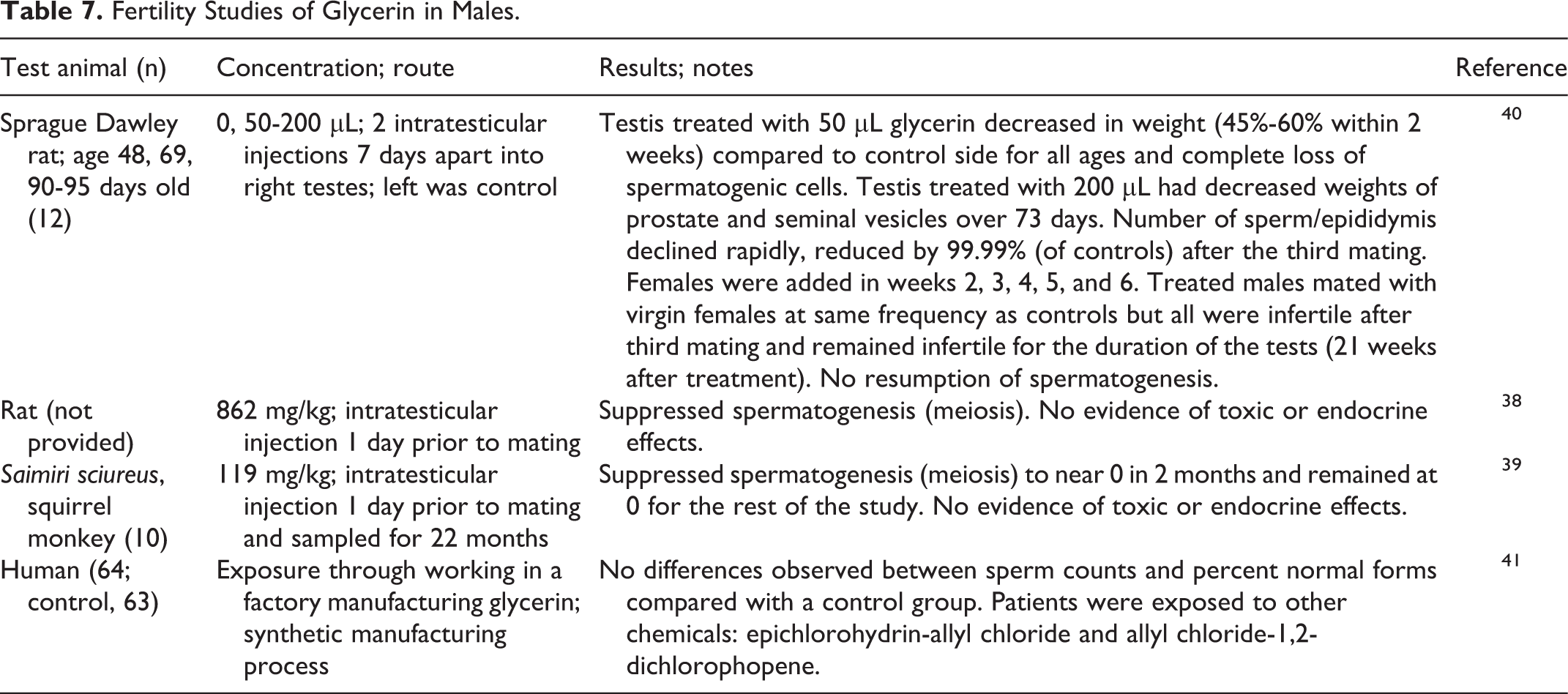
Glycerin injected into the testes of rats (50-200 µL and 862 mg/kg body weight) and monkeys (119 mg/kg body weight) suppressed spermatogenesis (Table 7). 38 -40
Fertility Studies of Glycerin in Males.
Male Fertility—Human
In a fertility study of male employees (n = 64) who manufacture synthetic glycerin, there were no differences observed in sperm counts and percentage normal forms compared with a control group (n = 63) who did not work with glycerin (Table 7). 41
Genotoxicity
In vitro
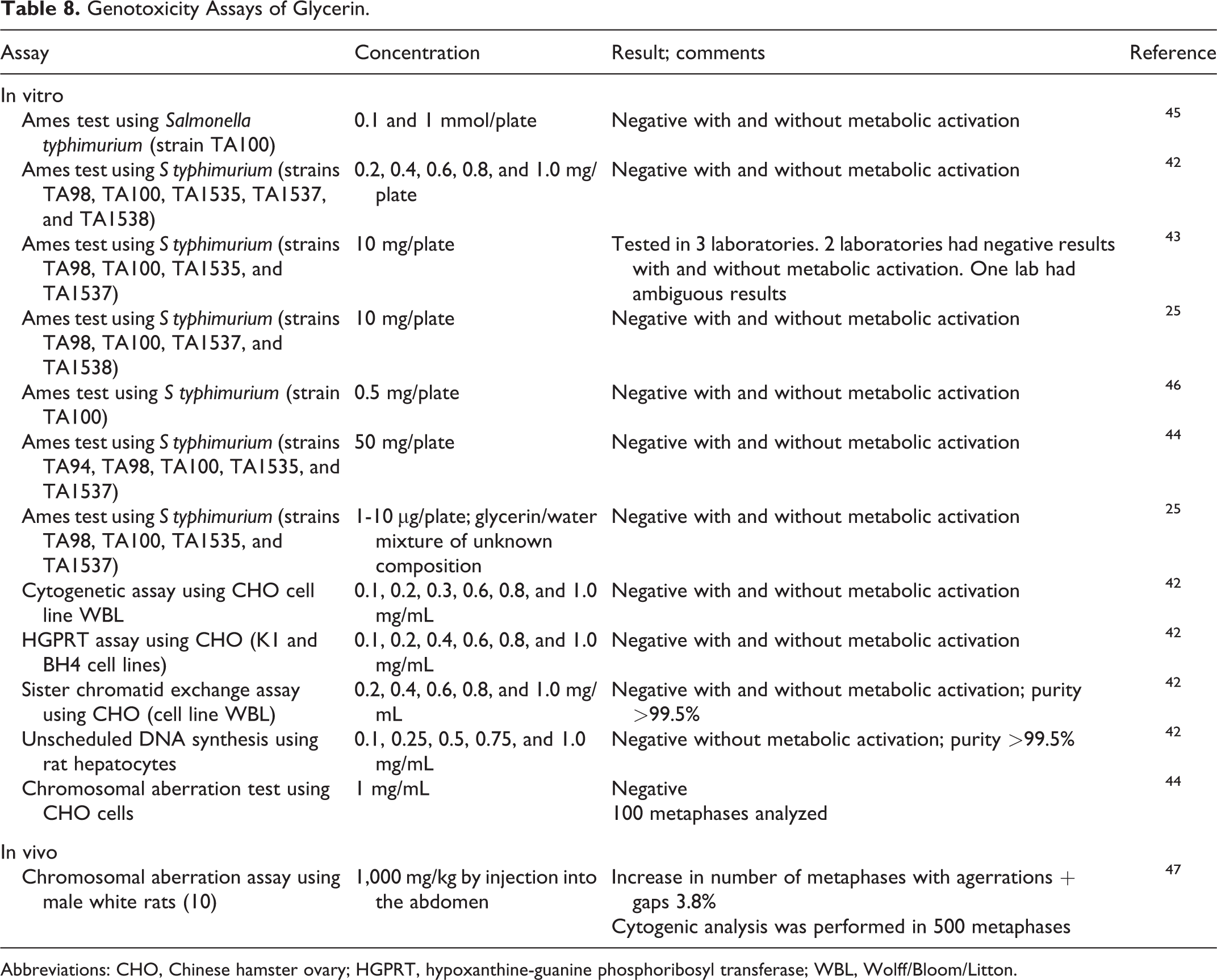
Glycerin was not genotoxic in multiple Ames tests using multiple strains of Salmonella typhimurium up to 50 mg/plate (Table 8). 25,42 -46 It was not genotoxic in a cytogenetic assay, X-linked hypoxanthine-guanine phosphoribosyl transferase (HGPRT) assay, sister chromatid exchange assay using Chinese hamster ovary (CHO) cells, unscheduled DNA synthesis assay using rat hepatocytes, or chromosome aberration test using CHO cells; up to 1.0 mg/mL was tested in these studies. 42,44
Genotoxicity Assays of Glycerin.
Abbreviations: CHO, Chinese hamster ovary; HGPRT, hypoxanthine-guanine phosphoribosyl transferase; WBL, Wolff/Bloom/Litton.
In Vivo
In a bone marrow chromosomal aberration assay, glycerin was not genotoxic when administered by injection into the abdomens of rats at 1,000 mg/kg (Table 8). 47
Carcinogenicity
Glycerin administered in the feed of rats at concentrations up to 20% for 1 year or up to 10 g/kg for 2 years did not increase the incidence of tumors (Table 9). 26
Carcinogenicity Studies of Glycerin.

Abbreviation: 4NQO, 4-nitroquinoline 1-oxide.
Glycerin administered in drinking water, up to 5% in as little as 4 weeks, had a synergistic effect with 4-nitroquinoline 1-oxide (4NQO) in mice (Table 9). 48 -50 There was an increased number of pulmonary tumor-bearing mice in the treated mice compared to controls.
Irritation and Sensitization
Irritation
Dermal—Nonhuman
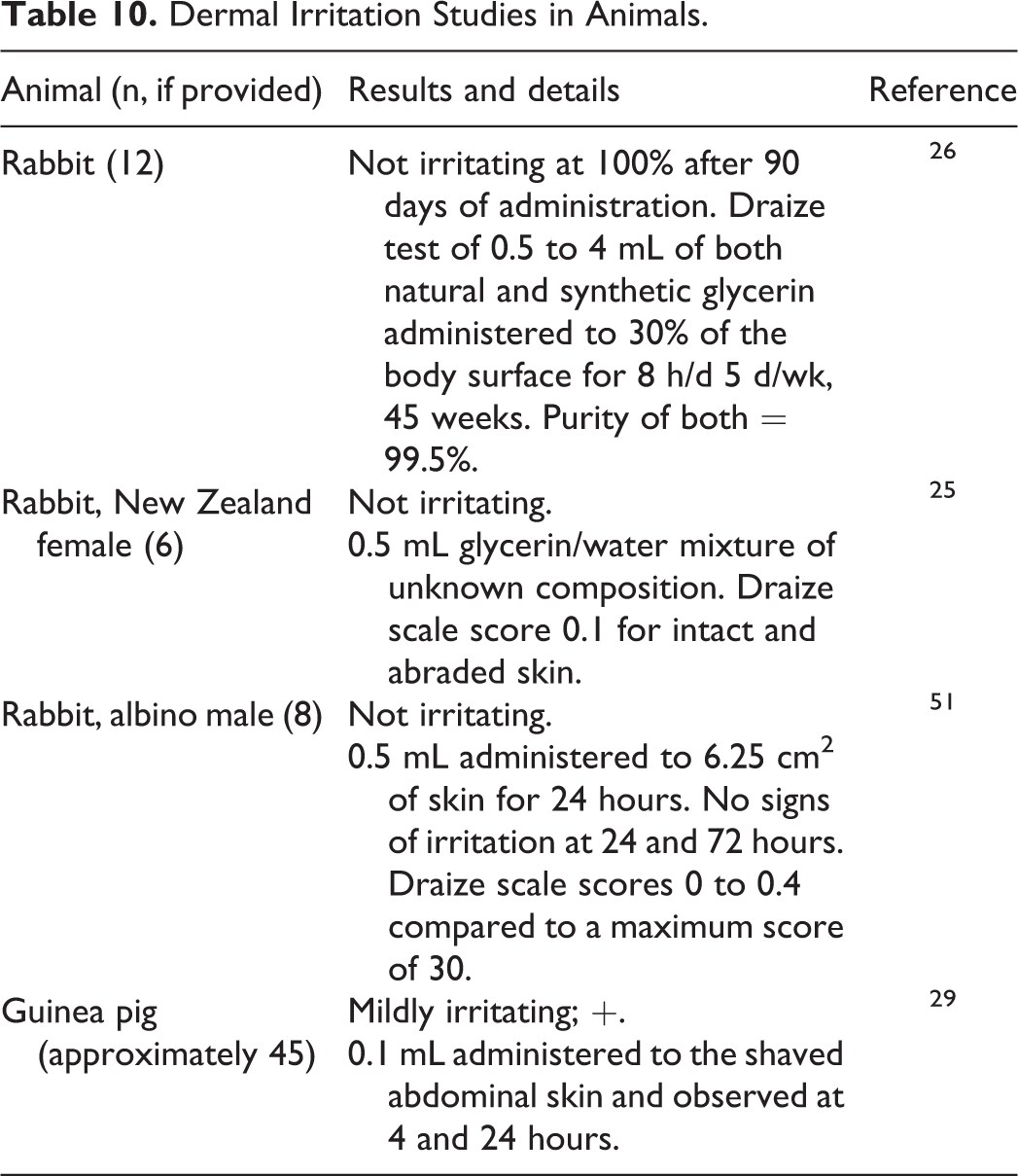
Glycerin was not dermally irritating in rabbits at concentrations up to 100% (Table 10). 25,26,51 Glycerin was a mild dermal irritant at 100% in guinea pigs. 29
Dermal Irritation Studies in Animals.
Dermal—Human
Glycerin (50% in water) was not irritating to patients with dermatitis (n = 420) when administered for 20 to 24 hours under occlusion. 52 One patient had a positive reaction. She reported using a mixture of glycerin (1 part) and 70% ethanol (9 parts) applied on the hands after washing with soap and water. She was tested with glycerin (1%, 5%, and 10% in water) and her glycerin–ethanol mixture (100%), resulting in +++ reactions for both test substances 48 and 72 hours after exposure. There were negative results in a routine test series. The eczema resolved after she stopped using the ethanol–glycerin mixture.
Glycerin (10%; 0.05 mL) was slightly irritating in a 48-hour occlusive patch test. 3 The irritation score was 4 of 9 on day 14 of observation. No further information was provided.
In a patch test, glycerin (25%; 0.2 mL) was not an irritant when administered to patients (n = 33) for 24 hours. 4
Ocular—Nonhuman
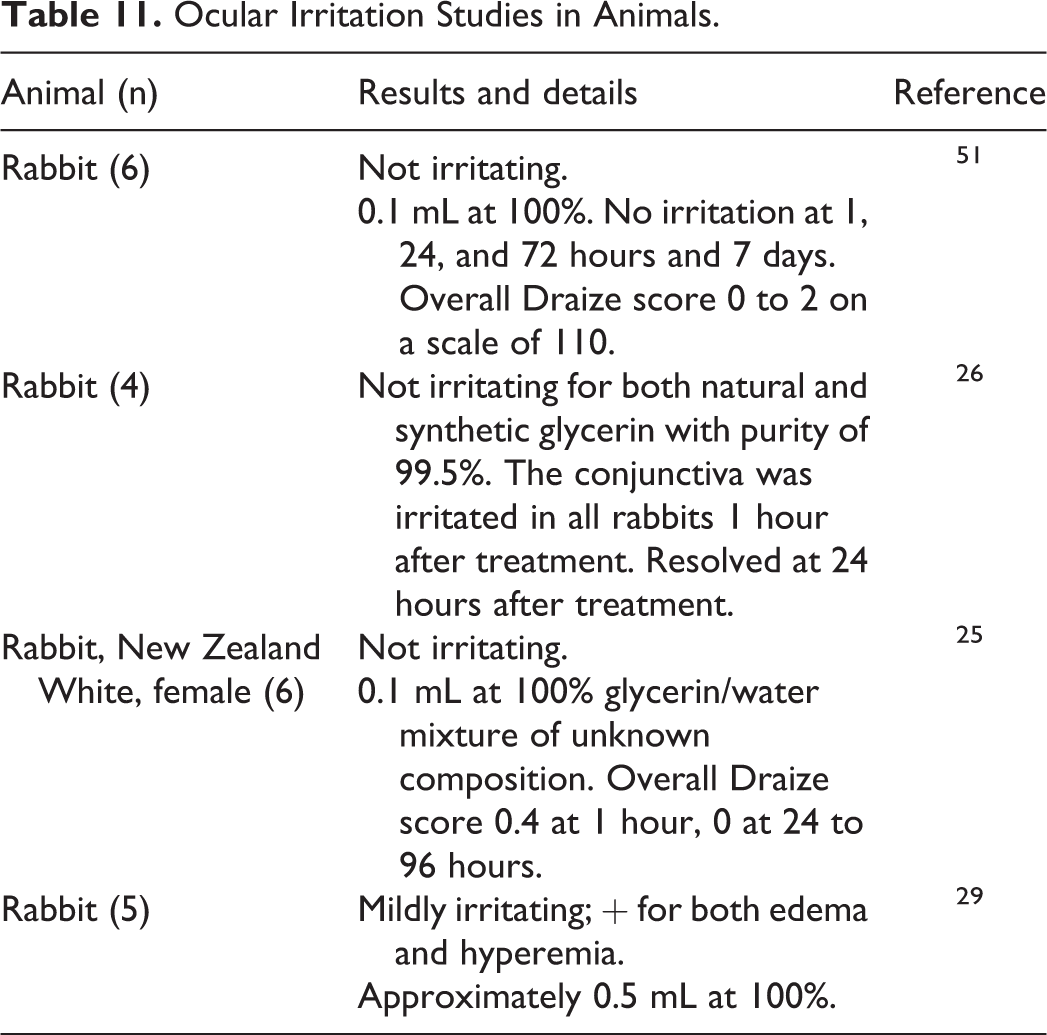
Glycerin was not irritating to the eyes of rabbits at concentrations up to 100% (Table 11). 25,26,29,51
Ocular Irritation Studies in Animals.
Ocular—Human
Topical administration of anhydrous glycerin to the eyes of human patients with edema of the superficial layers of the cornea resulted in reduced edema and improved visualization. 15 Pain and/or irritation have occurred following administration of glycerin to the eye. No further information was provided.
Glycerin (100%) was reported to be nonirritating when administered to the eyes of human patients (n not specified). 3 There was a strong burning and stinging sensation, with tear production, but no injury was observed. No further information was provided.
Sensitization
Dermal—Nonhuman
Natural and synthetic glycerin were not sensitizing to white male guinea pigs (n = 12). 26 The induction phase consisted of 10 injections of 0.1 mL of a 0.1% solution every other day. The challenge phase consisted of an injection of 0.05 mL of the 0.1% solution after a 2-week resting phase.
Dermal—Human
A moisturizer containing glycerin (65.9%) was not sensitizing in a modified Draize test (n = 48). 53 There were no reactions during either the induction phase or the challenge phase. The test substance was administered 10 times under occlusion for 48 or 72 hours (over the weekend). The challenge patch was in place for 48 hours. The test site was observed at removal and 48 hours after removal.
Patients (n = 15) who worked in a foam rubber factory and were regularly exposed to glycerin were not sensitized to glycerin when patched tested at 100% for 48 hours. 54
Case Reports
A 29-year-old woman presented with a 7-month history of patchy eczema on her eyelids, face, neck, scalp, and axillae. 55 She was patch tested using the European standard series, and bases, cosmetics, and hairdressers series. She was also patch tested with her own cosmetics and toiletries. She had a + positive reaction on day 4 to dimethylaminopropylamine (1% aqueous) and to her own hand moisturizing cream. Further testing of the ingredients of this cream had a + positive reaction on day 4 to glycerin (1% aqueous). Her eczema resolved when she avoided glycerin-containing cosmetics.
Summary
This is a safety assessment of glycerin, a polyhydric alcohol. Glycerin is reported to function in cosmetics as a denaturant, fragrance ingredient, hair conditioning agent, humectant, oral care agent, oral health-care drug, skin protectant, skin conditioning agent—humectant, and viscosity-decreasing agent.
Impurities were reported to be water, polyglycerol, and diethylene glycol. Phorbol esters may be present if the source material is the Jatropha plant.
Glycerin is reported to be used in 15,654 cosmetic products: 10,046 are leave-on products, 5,441 are rinse-off products, and 167 are products that are diluted for the bath. Glycerin is reported to be used at concentrations up to 78.5% in leave-on products, 68.6% in rinse-off products, and 47% in products diluted for the bath.
Glycerin is considered to be GRAS by the FDA for food packaging and as a multiple-purpose food substance. Glycerin is also used as an active ingredient in some over-the-counter drugs.
Glycerin is rapidly absorbed in the intestine and the stomach and distributed throughout the extracellular fluids through much of the body. It is metabolized primarily by the liver and kidneys, with the remainder excreted in urine. Free glycerin is naturally present in humans, primarily in plasma.
The reported oral LD50 of glycerin ranged from 2,530 to 58,400 mg/kg in rats, 4,090 to 38,000 mg/kg in mice, 27,000 mg/kg in rabbits, and 77,500 mg/kg in guinea pigs. The dermal LD50 of glycerin in rats was reported to be >21,900 mg/kg and >18,700 mg/kg in rabbits. The approximate Lt50 for rats was determined to be 423 minutes for exposure to glycerin vapors at 11.0 mg/L.
The oral LDLO of glycerin was reported to be 1,428 mg/kg for humans. There were no signs of toxicity when humans were orally administered 30 mL glycerin. When orally administered as a drug, adverse effects in humans included mild headache, dizziness, nausea, vomiting, thirst, and diarrhea.
In short-term feeding experiments using rats, 20% glycerin for 4 weeks in feed had no adverse effects, but at 53.4%, the kidneys weights were increased and liver enzymes were elevated. When glycerin was administered in the diet for 2 years, feed consumption was slightly increased in males fed synthetic glycerin versus those fed naturally sourced glycerin. There were no treatment-related effects in organ weights and gross pathology. The NOEL in mongrel dogs was 950 mg/kg/d when orally administered for 3 days. At 3,800 mg/kg/d, the mucosa of the stomach was severely hyperemic with petechial hemorrhages. Mongrel dogs experienced weight loss after 36 weeks when 35% glycerin was incorporated into their feed. There were no pathological changes in guinea pigs orally administered 6,300 mg/kg/d glycerin for 30 to 40 days.
There were no signs of toxicity or effects on blood or on urine production when human patients were orally administered approximately 1,300 to 2,200 g/kg/d glycerin for 50 days. The NOAEL was ≥2,200 mg/kg/d.
There were no treatment effects when 100% glycerin was topically applied daily to 30% of the body surfaces of rabbits for 45 weeks.
The inhalation LOAEL was 1,000 mg/m3 for glycerin administered 6 h/d, 5 d/wk for 2 weeks in rats. The inhalation NOAEL was 0.167 mg/L for glycerin administered for 5 h/d, 5 d/wk for 13 weeks in rats.
No adverse effects were observed in rats administered 20% glycerin in drinking water throughout gestation and nursing of pups. The F1 generation reproduced normally. The oral NOAEL for maternal toxicity and teratogenicity for rats was 1,310 mg/kg/d. The NOAEL for maternal toxicity and teratogenicity in mice was 1,280 mg/kg/d. The NOAEL for maternal toxicity and teratogenicity in rabbits was 1,180 mg/kg/d.
Glycerin injected into the testes of rats (50-200 µL and 862 mg/kg body weight) and monkeys (119 mg/kg body weight) suppressed spermatogenesis.
Glycerin was not genotoxic in multiple Ames tests using multiple strains of S typhimurium at concentrations up to 50 mg/plate. It was not genotoxic in a cytogenetic assay, X-linked HGPRT, sister chromatid exchange assay, unscheduled DNA synthesis assay, and chromosome aberration test at concentrations up to 1.0 mg/mL.
In 2 chromosomal aberration assays, glycerin was not genotoxic when administered orally to rats at 1 mg/kg or by injection into the abdomen at 1 g/kg. In a dominant lethal gene assay using rats, the results were ambiguous.
Glycerin administered in the feed of rats at doses up to 20% in feed for 1 year or up to 10 g/kg for 2 years did not increase the incidence of tumors. Orally administered glycerin, in concentrations up to 5%, had a potentiating effect on the carcinogenicity of 4NQO in mice.
Glycerin was not dermally irritating to rabbits when applied at concentrations up to 100% to up to 30% of the body surface 8 h/d, 5 d/wk for 45 weeks. Glycerin was a mild dermal irritant at 100% in guinea pigs.
Glycerin at 50% was not irritating to patients with dermatitis. Glycerin at 25% was not an irritant when administered to patients in a patch test.
Undiluted glycerin was not irritating when administered to the eyes of human patients. Strong burning and stinging sensation with tear production was reported, but no injury was observed.
Natural and synthetic glycerin were not sensitizing to white male guinea pigs at 0.1%. A moisturizer containing 65.9% glycerin was not sensitizing to humans.
Discussion
When considering the safety of glycerin, the Cosmetic Ingredient Review Expert Panel (Panel) noted that it is naturally occurring and abundant in animal and human tissues, including the skin and blood. The available data demonstrated low systemic oral and dermal toxicity for multiple animal species and humans, in both acute and long-term studies. There were no reproductive or developmental effects observed in oral studies using rats, mice, and rabbits. Glycerin was not genotoxic in multiple in vitro tests and it was not carcinogenetic to rats in a long-term feeding study. This ingredient was not a dermal or ocular irritant and was nonsensitizing to guinea pigs and humans. The Panel also noted the high frequency of use that is reported for glycerin and the low instances of reports of toxicity, irritation, or sensitization in the literature and the fact that glycerin is GRAS.
The source materials and intermediate forms of glycerin (eg, epichlorohydrin) should be completely consumed and/or eliminated in the manufacturing process. The Panel noted FDA’s warning that companies should monitor and audit their naturally derived ingredients because of the potential presence of phorbol esters if the source material is the Jatropha plant.
The Panel discussed the issue of incidental inhalation exposure from hair sprays (up to 10% in aerosol sprays and 30% in pump sprays), deodorants (up to 4% in a pump spray), face and neck products (up to 10% in aerosol sprays), face powders (up to 15%), body and hand sprays (up to 5% in aerosol sprays), moisturizing products (up to 3.3% in aerosol sprays), and in suntan products (up to 6% in aerosol sprays and 10% in pump sprays). Because the results of a 2-week (up to 1,000 mg/L) and a 13-week (up to 662 mg/L) inhalation study demonstrated little or no toxicity, the Panel concluded that there was little potential for respiratory effects at reported use concentrations. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredient is used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
Conclusion
The Panel concluded that glycerin is safe in cosmetics in the present practices of use and concentration described in this safety assessment.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Executive Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Author Contributions
L. Becker contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, P. Snyder, and L. Gill contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. B. Heldreth contributed to analysis and interpretation and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.