Abstract
This article presents a supplemental data analysis and evaluation of the findings from an oral (gavage) combined developmental and perinatal/postnatal reproduction toxicity study of the ammonium salt of perfluorohexanoic acid (CASRN: 21615-47-4) in Crl: CD-1(ICR) mice. The original study has been cited as supporting a lowest-observed-adverse-effects level of 175 mg/kg/d and no-observed-adverse-effects level of 35 mg/kg/d for developmental effects from perfluorohexanoic acid (PFHxA, CASRN: 307-24-4) in mice. The statistical analysis reported in 2014 was accurate in terms of quantifying statistical significance within phase 2 of the study. However, given the low incidence of findings, the purpose of this article is to extend the analysis and interpretation of findings by pooling the control group information from both phases of the same study, comparing the study findings to the incidence rates for stillbirths and postpartum viability for this species and strain of mouse observed for similar studies conducted by the same laboratory, and evaluating data on the incidence and range of spontaneous eye abnormalities reported in the literature. Based on this supplemental evaluation, the original study supports a NOAEL of 175 mg/kg/d for PFHxA in mice, which is a factor of 5-fold higher than previously reported. Furthermore, to the extent that this study may be considered in the selection of a point of departure for PFHxA in mice, it is noted that 175 mg/kg/d for maternal exposure is an unbounded NOAEL for developmental effects, meaning that the study did not establish a dose at which developmental effects may occur.
Introduction
The reproductive and developmental toxicity of perfluorohexanoic acid (PFHxA) has been investigated in several rodent studies in mice
1
and rats
2,3
exposed during pregnancy. Loveless et al
3
previously concluded sodium perfluorohexanoate (NaPFHx) is not a reproductive or developmental hazard, given that effects in rats were limited to reduced F1-generation pup weights through postpartum day 21 (preweaning period) in the high-dose treatment group of 500 mg/kg/d. The findings of greatest interest from the original mouse study,
1
which is the subject of this addendum, have included statistically significant differences between the high-dose treatment group (175 mg/kg/d) and control group (phase 2 only) for the following developmental end points in F1-generation pups: Stillbirths for n = 3 of 241 pups (1.2%) attributable to 1 F0-generation female, Mortality for n = 4 of 238 pups (1.7%) on the first day postpartum (noted as PPD 0), Corneal opacity in n = 2 of 238 pups (0.8%), observed in 1 pup in each of 2 litters, and A 12.5% reduction in average pup weight per litter, exclusively on the first day postpartum.
The original study has been cited as supporting a lowest-observed-adverse-effects level (LOAEL) of 175 mg/kg/d and no-observed-adverse-effects level (NOAEL) of 35 mg/kg/d for developmental effects from perfluorohexanoic acid (PFHxA, CASRN: 307-24-4) in mice. 4 -6 Considering the updated analyses and full weight of evidence reported herein, it is now clear that the low incidence rates of stillbirths and ocular effects reported in 2014 are actually unrelated to exposure to PFHxA during pregnancy. Furthermore, the postpartum mortality and body weight decrement of approximately 12% were limited to measurements during the first day postpartum, and there were no observed differences in survival or body weight gain of pups throughout the remainder of lactation and postweaning periods.
Accordingly, this study supports an unbounded NOAEL of 175 mg/kg/d for PFHxA in mice, which is a factor of 5-fold higher than previously reported. This means that it is likely that the NOAEL for developmental effects occurs at a maternal dose greater than 175 mg/kg/d in mice, consistent with the results in rats. 3
The results of this study on PFHxA further support what has been generally observed for other perfluoroalkyl acids (PFAAs) in that developmental toxicity has generally only been seen in the presence of maternal toxicity.
Study Design
The original study consisted of 2 phases with identical protocols except for a lower dose range in phase 2. The test substance administered was the ammonium salt of PFHxA (ie, APFHx) which, once absorbed into the bloodstream of animals (including humans), dissociates to form the PFHx− anion. The PFHxA also exists in the environment as the anion due to its low pK a (<1). Figure 1 provides the chemical structures for PFHxA and APFHx, and for expediency the results hereafter are discussed in terms of PFHxA. Doses were administered once daily via oral gavage to 20 females/group during gestation days 6 through 18. Treatment groups were higher in phase 1 (100, 350, and 500 mg/kg/d) than in phase 2 (7, 35, and 175 mg/kg/d). In phase 1, 13 of 80 (16%) of F0-generation female mice were found dead, including 3 in the control group and 6 in the low-dose group, which the authors noted were within the historical range for the testing facility (Charles River Laboratory, Horsham, PA). Furthermore, 2 of the 3 deaths in the highest dose group appeared to be due to the stress of nursing, which is known to occur in mice. 7 -11 By contrast, no mortality occurred in phase 2. Even though phase 1 mortality was largely unrelated to PFHxA exposure, the outcomes from phase 2 were considered more indicative of developmental end points of interest in F1-generation pups in the absence of maternal health effects.
Structure of (A) perfluorohexanoic acid (PFHxA; CASRN: 307-24-4) and (B) ammonium salt of PFHxA, also called ammonium perfluorohexanoate (APFHx; CASRN: 21615-47-4).
Measurement end points for this study included mortality, viability, body weight, clinical signs, abortions, premature deliveries, pregnancy and fertility, litter observations, and sexual maturity in the F1 generation. Mice from each dose group were necropsied, and major organs were examined for gross lesions. Livers were also weighed and analyzed for residual PFHxA.
The original study
1
presents results grouped into 4 developmental periods: (1) gestation, (2) lactation/preweaning (PPDs 0-20), (3) postweaning (PPDs 21-41), and (4) sexual maturation. For convenience, those reported findings are summarized in Table 1. Positive clinical findings are grouped into 2 categories: (a) related to PFHxA and (b) unrelated to PFHxA. In the original study, multiple criteria were applied when concluding a treatment group result was unrelated to PFHxA, including: Lack of dose-dependency Infrequent occurrence (low number of mice and/or number of litters) Observation only occurred in control group No statistical significance in group comparisons (treatment vs control) Differences attributable to litter size rather than test substance Tissue concentration did not correspond to clinical signs of adverse effect Observation is common in Crl: CD-1 mice.
Summary of Findings From Iwai and Hoberman for F1-Generation Mice for Phase 2.a,b
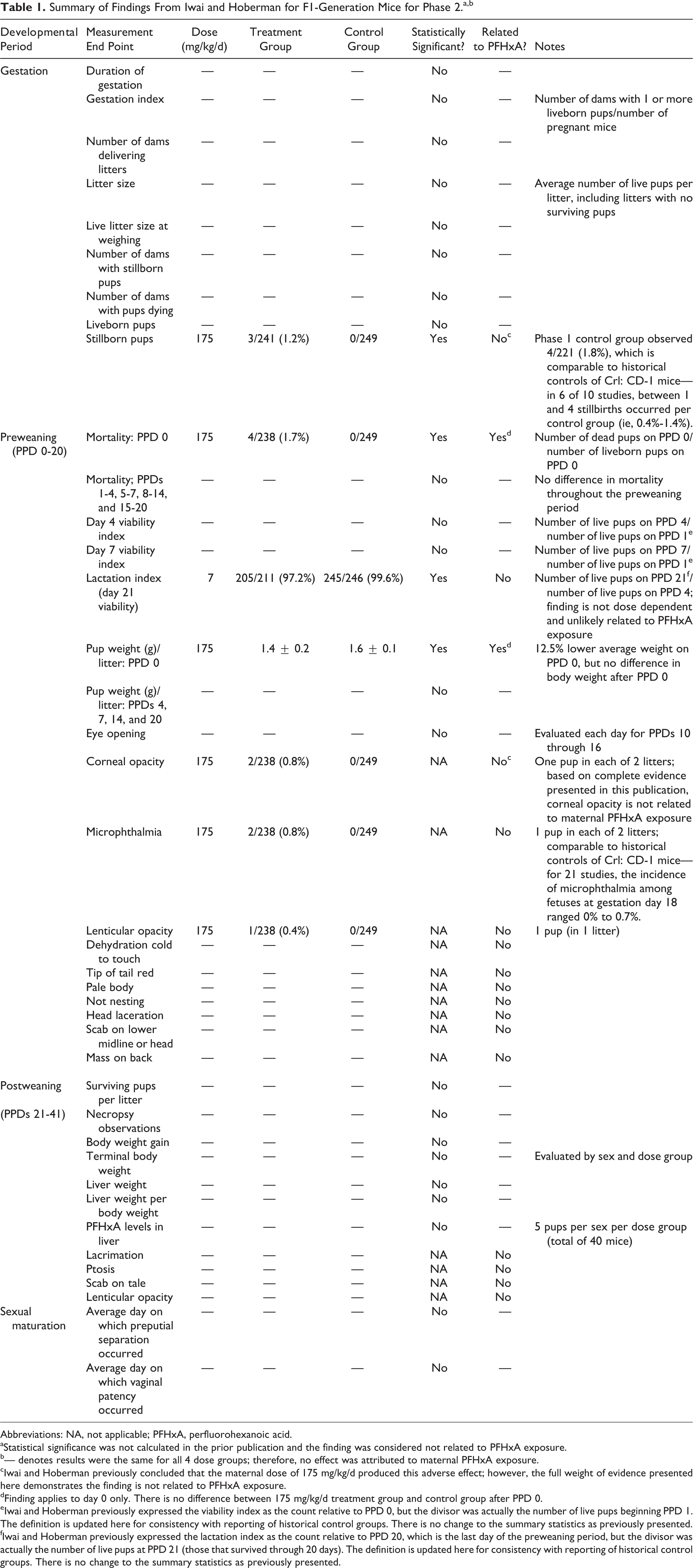
Abbreviations: NA, not applicable; PFHxA, perfluorohexanoic acid.
aStatistical significance was not calculated in the prior publication and the finding was considered not related to PFHxA exposure.
b— denotes results were the same for all 4 dose groups; therefore, no effect was attributed to maternal PFHxA exposure.
cIwai and Hoberman previously concluded that the maternal dose of 175 mg/kg/d produced this adverse effect; however, the full weight of evidence presented here demonstrates the finding is not related to PFHxA exposure.
dFinding applies to day 0 only. There is no difference between 175 mg/kg/d treatment group and control group after PPD 0.
eIwai and Hoberman previously expressed the viability index as the count relative to PPD 0, but the divisor was actually the number of live pups beginning PPD 1. The definition is updated here for consistency with reporting of historical control groups. There is no change to the summary statistics as previously presented.
fIwai and Hoberman previously expressed the lactation index as the count relative to PPD 20, which is the last day of the preweaning period, but the divisor was actually the number of live pups at PPD 21 (those that survived through 20 days). The definition is updated here for consistency with reporting of historical control groups. There is no change to the summary statistics as previously presented.
The utility of considering multiple criteria is to reduce the chance of misinterpreting a link between chemical exposure and adverse effect on the basis of statistical significance alone.
Stillbirth and Postpartum Mortality End Points
The study yielded the following data from which to assess pup mortality attributable to developmental toxicity: (a) incidence of stillbirths, (b) mortality within 24 hours postpartum, and (c) overall pup viability during later development stages (eg, preweaning and postweaning). The distinction in when pup mortality occurs may have important implications for the data analysis and interpretations of statistical significance, particularly when the incidence rates for each measurement end point are low in both control and treatment groups. In this study, pups were evaluated for vital status within 24 hours postpartum. A pup that was delivered and observed to be dead was classified as either stillborn or liveborn based on the condition of the removed lungs. Pups with lungs that sink when immersed in water were considered stillborn, whereas pups with lungs that float were considered liveborn and to have died shortly after birth. The latter category contributes to the mortality rate within 24 hours postpartum.
Reanalysis of Stillbirth End Point
The number of F1-generation pups delivered, number of stillbirths, and percentage of stillbirths were previously reported in table 4 of the original study for phase 1 and table 7 for phase 2 (reproduced in Table 2 for convenience). In phase 2 of the study, 3 of 241 delivered pups (1.2%) were reported as stillborn for the high-dose group (175 mg/kg/d). There were no stillbirths in the 2 lower dose groups and the control group. However, in the phase 1 control group, 4 of 221 delivered pups (1.8%) were stillborn. This occurrence of stillbirths in the control group, at a higher incidence rate than the phase 1 treatment group reported for the same study, indicates that the observation of stillbirths in phase 2 high-dose group is unlikely to be attributable to in utero PFHxA exposure.
Summary Statistics Relevant to Reanalysis of the Stillbirth End Point (From Iwai and Hoberman).
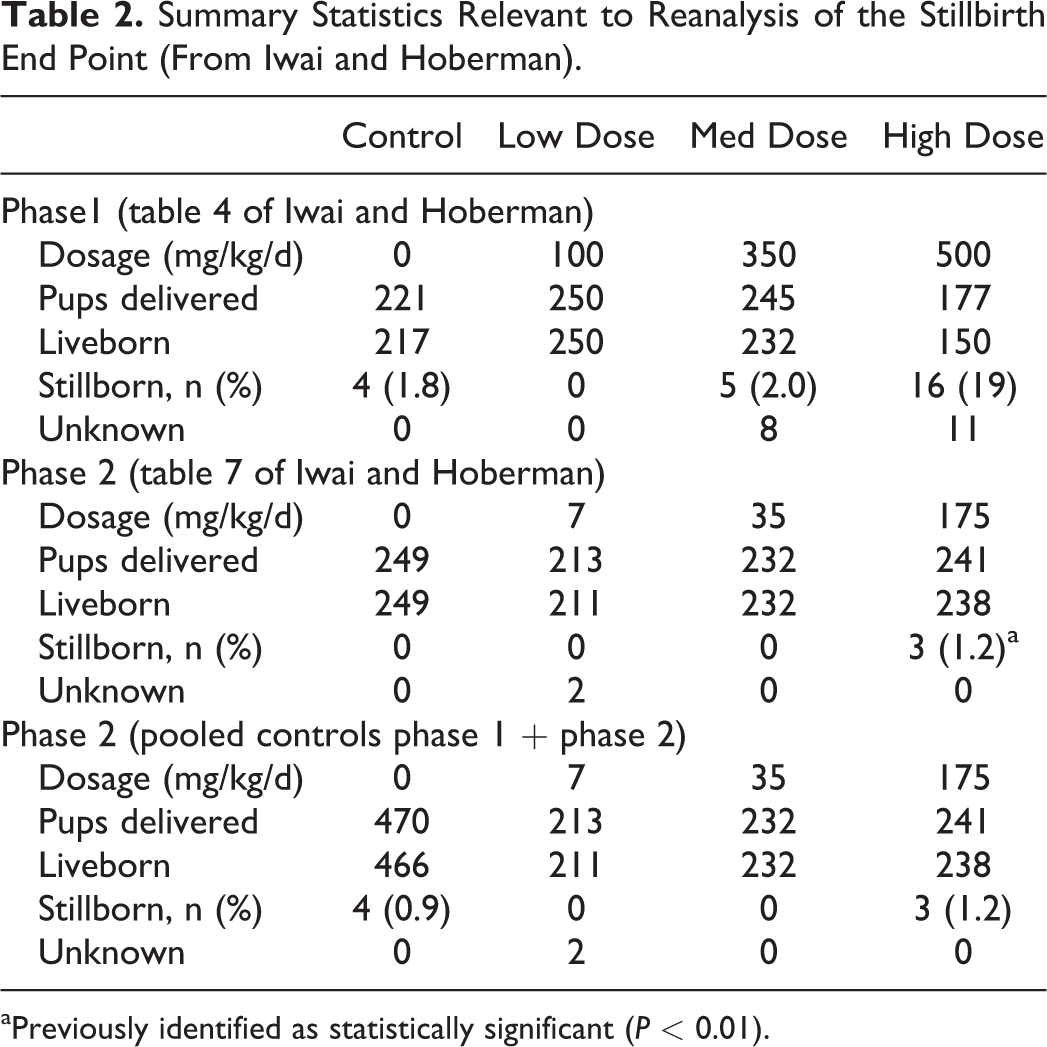
aPreviously identified as statistically significant (P < 0.01).
Fisher Exact Test
To further assess if the occurrence of 1.2% stillbirths in the phase 2 high-dose group is statistically significant (ie, greater than would be expected by chance alone) from controls, the statistical analysis was repeated for phase 2, but with pooled controls (ie, phases 1 and 2 combined). The Fisher exact test of independence is employed to detect significant differences in proportional data. This test is often applied for group comparisons in toxicological studies and was used in the original study for cases when more than 75% of the scores were tied. When pooling the control data, results indicate that the difference in stillbirths (ie, 0.9% for controls and 1.2% for the 175 mg/kg/d treatment group) is not statistically significant (P = 0.169).
Historical Control Data Report
Although it is common to focus on findings from one specific study, since most toxicity studies are conducted in genetically homogeneous animal strains, historical control data (HCD) from previous studies can be helpful in interpreting results, particularly if the findings occur infrequently and the studies are all conducted by the same testing laboratory. 12,13
This study was conducted at Charles River Laboratories. Dosing of the F0-generation Crl: CD-1 mice was initiated in December 2009 and the final scheduled sacrifice of F1-generation mice occurred in February 2010. In March 2018, Charles River prepared a report summarizing HCD in their Crl: CD-1 mice. Here, we compile the results for studies conducted within a 5-year interval before late 2009 to after early 2010 (ie, 2004 through 2015), which includes control groups from a total of 11 studies with final reports dated between December 2004 and August 2015. 14 Each toxicity study was compliant with Good Laboratory Practice and conducted with similar sample sizes as the present study (ie, approximately 20 F0-generation females producing litters with an average of 250 pups per control group). Control groups were untreated or received an appropriate vehicle.
Figure 2 summarizes the distribution of stillbirth counts and incidence rates across the 11 studies. The historic data reveal that a low incidence rate of stillbirths is relatively common and unrelated to administration of a test substance. At least 1 stillbirth was observed in 6 of 11 (55%) control groups, and 3 or more stillbirths were observed in 4 of 11 (36%). Incidence rates for individual studies varied between 0.6% and 1.8%. Therefore, the incidence rates in this study for the 175 mg/kg/d treatment group (ie, 3 stillbirths of 241 pups delivered, 1.2%) and the control group (pooling phases 1 and 2; ie, 4 stillbirths of 470 pups delivered, 0.9%) are both within the historic range for Crl: CD-1 mice and are consistent with the expected incidence rate for at least 1 of every 3 studies.
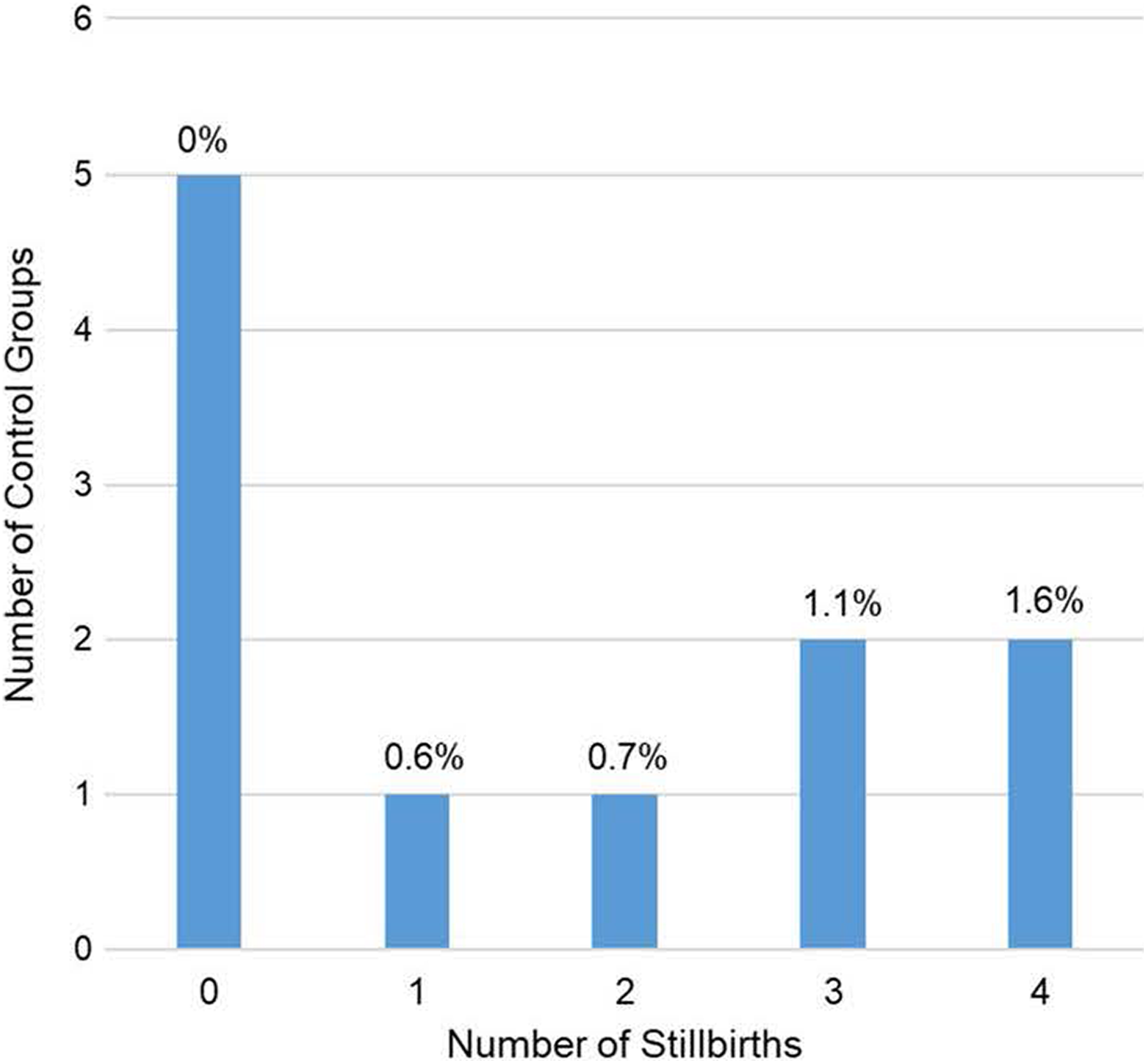
Incidence of stillbirths in Crl: CD-1 mice among 11 historical control groups of toxicity studies conducted by Charles River Laboratories between December 2004 and August 2015. The average litter size is 225 pups per control group. The average percentage of stillborn pups (ie, number of stillbirths per number of pups delivered) per incidence category (number of stillbirths observed in a control group) is shown above each bar. The percentage of pups that are delivered stillborn in Crl: CD-1 mouse control groups ranges from 0% to 1.8%, and the average (including studies with no stillbirths) is approximately 0.6%. The probability of observing 2 or more stillbirths (ie, the number observed in this study) is 45% (5 of 11 studies); the probability of observing at least 1 stillbirth is 55% (6 of 11 studies).
Reanalysis of Postpartum Mortality End Point
With the exception of the first day postpartum, there was no difference in pup survival throughout the preweaning period (PPDs 1-20). Among the litters of dams administered 175 mg/kg/d, 1.7% (4 of 238 pups) were found dead or presumed cannibalized on the first day postpartum. This finding is statistically significant (P ≤ 0.01), given that no pups were dead or missing in control groups of phases 1 and 2. For the 350 mg/kg/d treatment group, 3 of 232 (1.3%) liveborn pups were found dead or cannibalized on the first day; however, this incidence rate was not statistically significant. Among the control groups in the HCD database discussed above, mortality on the first day postpartum (recorded as PND 1 rather than PND 0 in some studies) was reported for 9 studies. In 3 of the 9 studies (33%), between 1 and 10 pups were dead or presumed cannabilized, equivalent to incidence rates ranging from 0.3% to 3.9%. Therefore, similar to the reevaluation of incidence rates for stillbirths discussed above, the incidences of pup mortality on the first day postpartum for the 175 and 350 mg/kg/d treatment groups are within the historic range for Crl: CD-1 mice, and one might expect similar incidences to occur, unrelated to exposure to a test substance, in at least 1 of every 2 studies.
For this study, PNDs 1-4 was also measured, and the results were previously reported in table 4 (phase 1) and table 7 (phase 2) of Iwai and Hoberman. 1 Dead pups were observed in the control groups (2 of 217 or 0.9% for phase 1; 3 of 249 or 1.2% for phase 2) and the 175 mg/kg/d treatment group (3 of 234 or 1.3%). These incidence rates are comparable to that of 9 studies from the HCD database, for which the mean is 2.1% and the maximum observed in 2 control groups is 3.9%. Similarly, the day 4 viability index (number of live pups on PPD 4 divided by the number of liveborn pups) for controls in this study is 99.1% (215 of 217) for phase 1 and 98.8% (246 of 249) for phase 2, compared with 97.0% (231 of 238) for the 175 mg/kg/d treatment group. Likewise, there was no difference in pup survival based on the day 7 viability index nor on any subsequent day during the preweaning period. Therefore, the incidence of pup deaths for the 175 mg/kg/d treatment group does not appear to be related to maternal PFHxA exposure.
Corneal Opacity and Microphthalmia
Corneal opacity was reported for 2 of 238 pups (0.8%), each from a different litter from the 175 mg/kg/d treatment group. Similarly, microphthalmia (undersized eye) was reported for 2 of 238 pups from different litters from the same dose group. These preweaning clinical findings were not observed in any other dose group and therefore can be characterized as spontaneous and variable, rather than dose dependent. The high incidence of pup mortality in the 350 and 500 mg/kg/d dose groups of phase 1 precludes a firm conclusion about the absence of dose response between maternal PFHxA exposure and ocular effects in newborn pups.
Corneal opacity in mice can appear as a unilateral or bilateral area of white or gray opacification, haziness, or translucency. 15 -17 Morphological alterations that contribute to this effect include lesions or alterations in the corneal epithelium, stroma, or endothelium, which interferes with the normal transmission of light through these structures. 15 The most frequent reported microscopic appearance of corneal opacification in the mouse is mineralization of the anterior corneal stroma and the basement membrane zone immediately beneath the corneal epithelium. 15
A study published in 1991 reported that spontaneous corneal opacities were observed in 4.3% of CD-1 mice, inclusive of adults. 16 The suspected etiology in young mice was ammonia produced in the environment from urinary urea by urease-positive fecal bacteria. Frequent (twice-weekly) cage cleaning reduced the incidence of corneal opacity to 0.8%, the same rate observed for the 175 mg/kg/d dose group. Although this mechanism is likely not a factor for newborn pups, and the rate of spontaneous alterations is expected to change in even the same strain of test species over a period of decades, this study demonstrates how environmental conditions can contribute to anomalies observed during the preweaning phase.
Mukaratirwa et al 18 summarize the incidence and range of spontaneous lesions of the eye in Crl: CD-1(ICR)BR mice used in toxicity studies at Charles River Laboratories. Data were collected from control dose groups of 4-, 13-, 80-, and 104-week toxicity studies conducted between 2005 and 2013. While the 4-week studies would be most comparable to this study, the sample size (a total of 250 mice from 17 studies) is essentially equivalent to that of a single phase of the current study. No incidences of corneal opacity were observed among the 4-week (n = 250), 13-week (n = 430), or 80-week (n = 510) control groups, for a total of 0 of 1,190 mice. However, in the 104-week study, the incidence rate for spontaneous alterations of the cornea was 81 of 2,266 (3.6%). The most common lesion affecting the cornea was keratitis (ie, infiltration with neutrophils and stromal edema as acute, or thickening and keratinization of the corneal epithelium and stromal fibrosis as chronic), followed by mineral deposits and corneal ulceration.
Charles River Laboratory reviewed historic study data for 21 studies consisting of a total of 441 control group litters of CD-1 mice between 2008 and February 2018. Eye anomalies (including corneal opacity and microphthalmia) were reported for a single pup in 3 of 21 (14%) studies (each from different control group litters). The 2018 HCD report 14 discussed above (see “Stillbirth End Point” section) provides an additional summary of the incidence rate of microphthalmia specifically. The incidence rate is reported as ranging from 0% to 5% per study, based on an evaluation of control groups for 21 studies consisting of a total of 453 litters and 3,176 fetuses examined following cesarean delivery on gestation day 18.
Gilger et al 17 recently compiled data on spontaneous ocular abnormalities in 5300 CD-1 mice ages 1 to 12 weeks. The test animals were raised in the same facilities that served as the source of mice used in studies by Charles River Laboratories. Spontaneous ocular lesions were observed in approximately 10% of test animals. The incidence of “small globe,” recorded as phthisis bulbi and often indistinguishable from microphthalmia during clinical examination, was 0.33%.
In conclusion, the low incidence of ocular effects in this study is unlikely related to maternal PFHxA exposure. This conclusion is supported by the HCD for CD-1 mice, which demonstrates that for any given study, there is greater than a 1 in 10 chance that an eye anomaly such as corneal opacity and microphthalmia will occur spontaneously in at least 1 pup in the absence of exposure to test material. The observations in this study are restricted to a single treatment group (175 mg/kg/d), and the overall incidence rate of 0.8% for both corneal opacity and microphthalmia is consistent with low reference ranges reported in the literature and HCD reports for Crl: CD-1 mice.
Body Weight
Pup body weights were reduced on the first day postpartum in all dose groups, but persisted in only the 350 and 500 mg/kg/d groups of phase 1. Therefore, the study supports a conclusion that body weights are reduced at birth for the 175 mg/kg/d dose group, but body weight gains of the F1-generation mice throughout lactation and postweaning periods are unaffected by maternal doses of PFHxA as high as 175 mg/kg/d. The potential for adverse outcomes associated with an early but short-term and reversible deficit in body weight is unclear, given that postnatal functional parameters are not evaluated in this type of study protocol.
Discussion
When phase 1 and phase 2 control groups are pooled, the n = 4 stillbirths represent 0.9% of pups delivered, compared with n = 3 stillbirths representing 1.2% of pups delivered for the phase 2 high-dose (175 mg/kg/d) group. This difference is not statistically significant (P < 0.05, Fisher exact test). This result changes the conclusion for the original study with respect to the designation of a lowest-observed-adverse-effects level of 175 mg/kg/d and NOAEL of 35 mg/kg/d for PFHxA due to F1 stillbirths. Instead, the study supports a conclusion that the phase 2 high-dose group of 175 mg/kg/d is an unbounded NOAEL for this end point. The results of this supplemental statistical analysis of the pooled controls, together with the HCD for 11 studies using the same mouse strain and testing laboratory, provide strong evidence that there is greater than a 50% chance of observing a low percentage of stillbirths in Crl: CD-1 mice and that this probability is no different for mice exposed to PFHxA during pregnancy in the dose range 7 to 175 mg/kg/d. Therefore, stillbirths observed in this study are not an appropriate basis for classifying PFHxA as a developmental toxicant in mice.
There are notable differences in the relevance of developmental effects end points when comparing PFAAs of different carbon chain lengths. In studies of CD-1 mice, the developmental toxicity induced by exposure to perfluorooctanoic acid (PFOA) throughout gestation included dose-related full litter resorptions, reduced postnatal survival, delayed eye opening, growth deficits, and sex-specific alterations in pubertal maturation. 19 By contrast, exposure to the C4 PFAA (perfluorobutyric acid or PFBA) during pregnancy in mice did not produce the same adverse developmental effects, although some subtler effects were still detectable. 20 Similar to PFBA exposure, this study with PFHxA demonstrates that exposure during pregnancy does not affect survival, growth, or sexual maturation of F1-generation pups throughout lactation and postweaning developmental periods at doses as high as 175 mg/kg/d.
The difference in outcomes between PFOA and lower carbon chain PFAAs is likely attributable to differences in elimination kinetics and possible differences in mechanism of action, although how PFOA exposure may result in developmental toxicity has not been elucidated. Differences between PFOA and lower carbon chain PFAAs have been well-documented. 2,3 In this study, the body burden of PFHxA in F1-generation mice after weaning was measured. In phase 2, there was no detectable amount of PFHxA found in the liver upon killing (PPD 41). Iwai and Hoberman 1 also characterized pharmacokinetics of PFHxA in female mice when administered once by oral gavage at dose levels of 35, 175, and 350 mg/kg. The PFHxA was rapidly absorbed (C max reached within 30 minutes) and in general was not quantifiable at 24 hours after dose administration. The terminal elimination half-life of PFHxA ranged from 0.9 to 1.2 hours and was dose independent. In a mouse multiple-dose study, 13 daily doses of orally administered PFHxA ammonium salt were followed by a single oral administration of [14C]-PFHxA ammonium salt. 21 Excretion was rapid, with mean recoveries of over 90% of the administered dose (mean values >95% of the ultimately recovered material) at 24 hours after dosing, irrespective of gender. Thus, for the original study, by 48 hours postweaning (ie, PPD 22), most of the PFHxA would be eliminated, even if one assumes a preferential accumulation of PFHxA in the liver (like PFOA). 1
For PFHxA, systemic toxicity is likely to be a more sensitive end point than developmental effects in rodents. In other words, establishing a point of departure protective of systemic toxicity will also be protective of developmental toxicity. The findings from this study are consistent with two 90-day repeated-oral dose studies in rats of PFHxA and related salts, which demonstrated changes in liver weights accompanied by minimal hepatocellular hypertrophy and slightly higher hepatic peroxisomal β-oxidation activity. The studies support an NOAEL of 50 mg/kg/d for PFHxA (acid) in male rats, based on liver weight increases and hepatocellular hypertrophy, 2 and 20 mg/kg/d for perfluorohexanoate (NaPFHx), based on nasal lesions in male rats. 3 The NOAEL for developmental toxicity in rats is 100 mg/kg/d, based on reduced F1-generation pup weights at 500 mg/kg/d. Therefore, in rats, exposure to PFHxA supports a 2- to 5-fold difference in the NOAEL, with systemic toxicity being a more sensitive end point than developmental toxicity.
Conclusions
Table 3 presents a side-by-side summary of the findings from the original study as reported by Iwai and Hoberman
1
and the findings based on the supplemental analysis presented in this addendum. The supplemental analysis supports the following conclusions regarding the potential for developmental toxicity associated with maternal exposure to PFHxA: Based on a reanalysis of stillbirths and the recent HCD, stillbirths observed in this study are unlikely to be related to in utero PFHxA exposure, and therefore, the stillbirth end point is not indicative of developmental toxicity in mice; Corneal opacity and microphthalmia observed in this study are unlikely to be related to in utero PFHxA exposure and therefore are not indicative of developmental toxicity in mice; Mortality on the first day postpartum is statistically significant at 175 mg/kg/d, but not at 350 mg/kg/d; the incidence rate is within the observed range for approximately one-third of the historic control groups and there are no sustained differences in viability indices throughout lactation and postweaning periods; A decrement in average pup weight per litter on the first day postpartum is statistically significant at 175 mg/kg/d, yet there are no sustained effects on growth observed throughout lactation and postweaning periods; The study supports an unbounded NOAEL of 175 mg/kg/d for maternal PFHxA exposure based on an array of developmental toxicity end points; and As with other PFAAs, developmental toxicity in mice is not attributable to in utero PFHxA exposure when doses are well below levels that may contribute to maternal toxicity.
Summary of Updates to Findings From Iwai and Hoberman.

Abbreviations: LOAEL, lowest-observed-adverse-effects level; NOAEL, no-observed-adverse-effects level; PFHxA, perfluorohexanoic acid.
Footnotes
Author Contributions
H. Iwai and E. Mendelsohn contributed to acquisition, contributed to analysis or interpretation of data, and critically revised the manuscript for important intellectual content. A. Hoberman substantially contributed to conception or design; contributed to acquisition, analysis, or interpretation of data; and critically revised the manuscript for important intellectual content. P. Goodrum substantially contributed to conception or design; contributed to acquisition, analysis, or interpretation of data; drafted the manuscript; and critically revised the manuscript for important intellectual content. J. Anderson substantially contributed to conception or design and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the FluoroCouncil. The authors thank members of the FluoroCouncil Panel for their insightful comments on this addendum. The funders were given the opportunity to review the draft addendum; the purpose of such review was to allow input on the clarity of the science presented but not on interpretation of the research findings. The researchers’ scientific conclusions and professional judgments were not subject to the funders’ control; the contents of this addendum reflect solely the view of the authors. Furthermore, as noted in Iwai et al, the research for the original article was conducted entirely at Charles River Laboratories under the sponsorship of Daikin Industries Ltd.
