Abstract
Perfluorohexanoic acid (PFHxA), a 6-carbon perfluoroalkyl (C6; CAS # 307-24-4), has been proposed as a replacement for the commonly used 8-carbon perfluoroalkyls: perfluorooctanoic acid and perfluorooctane sulfonate. PFHxA is not currently a commercial product but rather the ultimate degradation product of C6 fluorotelomer used to make C6 fluorotelomer acrylate polymers. It can be expected that, to a greater or lesser extent, the environmental loading of PFHxA will increase, as C6 fluorotelomer acrylate treatments are used and waste is generated. This article reports on a chronic study (duration 104 weeks) that was performed to evaluate the possible toxicologic and carcinogenic effects of PFHxA in gavage (daily gavage, 7 days per week) treated male and female Sprague-Dawley (SD) rats. In the current study, dosage levels of 0, 2.5, 15, and 100 mg/kg/day of PFHxA (males) and 5, 30, and 200 mg/kg/day of PFHxA (females) were selected based on a previous subchronic investigation. No effects on body weights, food consumption, a functional observational battery, or motor activity were observed after exposure to PFHxA. While no difference in survival rates in males was seen, a dose-dependent decrease in survival in PFHxA-treated female rats was observed. Hematology and serum chemistry were unaffected by PFHxA. PFHxA-related histologic changes were noted in the kidneys of the 200-mg/kg/day group females. Finally, there was no evidence that PFHxA was tumorigenic in male or female SD rats at any of the dosage levels examined.
Introduction
Perfluorinated compounds are extensively used in the manufacturing of a wide array of industrial and commercial products. Their unique surfactant properties have been used in coatings for packaging, carpets, leather products, and textiles. Selected perfluoroalkyl polymers have also been used in the manufacture of nonstick coatings on cookware (Key, Howell, and Criddle 1997). The extensive use and disposal of these perfluoroalkyl products have led to environmental contamination and have been detected in the air, water, soil, sediments, wildlife, food, human breast milk, and blood (Fromme et al. 2009; Mak et al. 2009; Naile et al. 2010; Nakata et al. 2006; Olsen et al. 2012; Pico et al. 2011; Sinclair et al. 2006; Tao et al. 2008; Zhang et al. 2011). Several studies have investigated the importance of the exposure routes including diet, drinking water, house dust, food packaging, nonstick cookware, indoor and outdoor air to perfluorinated compounds (D’Hollander et al. 2010; Fromme et al. 2009; Vestergren and Cousins 2009). Based on these investigations, the oral route of exposure has been considered the most important exposure pathway for perfluorinated compounds (Domingo 2012; Fromme et al. 2009).
Among the perfluoroalkyl acids, the 8-carbon (C8) perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) have been the most extensively studied. Their environmental persistence, bioaccumulation, and possible occupational exposure have raised concerns regarding the potential health effects of the C8 perfluoroalkyl acids in humans (Domingo 2012; Fromme et al. 2009; Grandjean et al. 2012; Melzer et al. 2010). PFOA, PFOS, and other C8 perfluorinated compounds have been reported to suppress immune response in laboratory animals and in exposed humans (reviewed by DeWitt et al. 2012). Elevated exposure to C8 perfluorinated compounds has also been reported to reduce the humoral immune response in children and be linked to thyroid disease in adults (Grandjean et al. 2012; Melzer et al. 2010). The carcinogenic potential of PFOA in rodents has been investigated in 2 dietary carcinogenicity studies in rats (Biegel et al. 2001). Carcinogenicity studies in Sprague-Dawley (SD) rats show that PFOA induces the tumor triad pattern (liver adenomas, Leydig cell tumors, and pancreatic acinar cell tumors) similar to several peroxisome proliferator–activated receptor alpha (PPARα) agonists (Biegel et al. 2001). PFOA and other structurally related perfluoroalkyl acids including perfluorononanoic acid (PFNA), perfluorodecanoic acid (PFDA), and PFOS have been shown to have tumor-promoting activity in rainbow trout (Benninghoff et al. 2012; Tilton et al. 2008). Several epidemiological studies have suggested that occupational exposure to C8 perfluorinated compounds including PFOA and PFOS produce an increased risk of bladder, prostate, and breast cancer (Alexander and Olsen 2007; Alexander et al. 2003; Bonefeld-Jorgensen et al. 2011; Gilliland and Mandel 1993; Shankar, Xiao, and Ducatman 2011). In contrast to the occupational evaluations, an epidemiologic study that examined the general population did not show any increase in tumor rates (Eriksen et al. 2009).
The relative toxicity of perfluoroalkyl acids has been suggested to be related to the bioaccumulation/elimination rate of the compound which in turn is related to carbon chain length (Kudo et al. 2006; Mulkiewicz et al. 2007; Ohmori et al. 2003). Ohmori et al. (2003) examined the relative toxicokinetics of 4 perfluorocarboxylic acids with different carbon chain length and reported that the half-lives for perfluoroheptanoic acid (PFHA, carbon perfluoroalkyl [C6]), FPOA (C8), PFNA (C9), and PFDA, C10 in male and female rats and found a correlation between urinary excretion and chain length. The shorter carbon chain compounds were more quickly excreted in the urine and thus resulted in lower concentrations of compound in the serum and liver (Kudo et al. 2001). Similarly, Mulkiewicz et al. (2007) showed a correlation between toxicity to cells in culture and carbon chain length of perfluorinated acids, with the longer the perfluorocarbon chain, the more toxic the compound (Mulkiewicz et al. 2007). With concerns over the toxicity and bioaccumulation of long carbon chain perfluorinated compounds (C8 and longer), the search and development of alternative fluorinated compounds of shorter carbon chain compounds have been performed. One such compound, the C6 PFHxA, has been developed and appears to possess many of the commercially needed properties of industrial perfluorinated compounds while having lower toxicity and greater human safety (Mulkiewicz et al. 2007). A previously performed subchronic toxicity study of PFHxA (0, 10, 50, and 200 mg/kg/day) in male and female rats showed a decrease in body weight gain, mild elevation in liver enzymes in the mid- and high-dose males rats. Centrilobular hepatocellular hypertrophy and increased liver weights were noted in the high-dose treated (200 mg/kg/day) male rats (Chengelis et al. 2009). Lower carbon chain fluoro compounds such as PFHxA have been suggested to be possible replacements for the C8 compounds (U.S. Environmental Protection Agency [USEPA] 2013).
The objective of the current study was to evaluate the possible chronic toxicologic and carcinogenic effects of orally administered PFHxA in male and female SD rats. Three doses of PFHxA were examined in male and female rats based on the results of the previously reported 90-day subchronic oral toxicity study in SD rats (Chengelis et al. 2009). The 2 high doses selected for the current study (100 mg/kg/day for male rats and 200 mg/kg/day for female rats) were expected to produce a maximally tolerated dose without compromising the survival of the rats.
Materials and Methods
Chemicals
PFHxA (98.1%; CAS number 307-24-4; C6HF11O2; molecular weight: 314.05) was provided by Miki and Co., Ltd. (Chuo-ku, Tokyo, Japan). The purity of the test material (98.1%) was considered to be 100% for dose calculation purposes. PFHxA was stored at room temperature. Previous studies have shown the compound to be stable at room temperature (Chengelis et al. 2009). PFHxA formulations for oral gavage administration to the rats were prepared in deionized water. Deionized water was used as the vehicle for the PFHxA and served as the treatment agent for the control rat groups.
Animals
All rodent treatment, toxicity assessments, and data acquisition were performed at WIL Research (Ashland, OH). The protocol was reviewed and approved by the Institutional Animal Care and Use Committees (IACUC), in compliance with the Animal Welfare Act (AWA). Male and female Crl:CD (SD) rats, approximately 26 days old, were obtained from Charles River Laboratories, Inc. (Raleigh, NC). Rats were acclimated for 14 days prior to being randomly assigned to treatment groups. Rats were individually housed during treatment in wire mesh cages suspended above a cage board. All animals were maintained in accordance with the Guide for the Care and Use of Laboratory Animals in the animal facilities at WIL Research (American Association for the Accreditation of Laboratory Animal Care [AAALAC] accredited). Rats were provided food and water ad libitum. Diet consisted of a basal rodent meal (PMI Nutrition International, LLC, and Certified Rodent LabDiet® 5002). Drinking water was delivered by an automatic watering system. Rodent room temperature and humidity controls were maintained at a temperature of 22 ± 3°C and a humidity of 50 ± 20%, respectively. A 12-hr light/12-hr dark photoperiod and a minimum of 10 fresh air changes per hour were maintained in the rodent room.
Treatment
Rats were randomly assigned by body weight stratification to 1 of the 4 treatment groups for each gender. Groups 1 to 3 consisted of 60 rats/sex and group 4 consisted of 70 rats/sex (Table 1). Group 1 males and females received deionized water (vehicle control). Male rats in groups 2, 3, and 4 received 2.5-, 15-, and 100-mg PFHxA/kg body weight/day, respectively. Female rats in groups 2, 3, and 4 received 5-, 30-, and 200-mg PFHxA/kg body weight/day, respectively. Dose selection was based on a previous subchronic toxicity study (Chengelis et al. 2009) in which 100-mg PFHxA/kg body weight/day for males and 200-mg PFHxA/kg body weight/day for females was determined to be the maximum tolerated dose (MTD; Chengelis et al. 2009). Rats in the PFHxA and vehicle control groups were administered the respective concentration of compound via gavage daily, 7 days per week for up to 104 consecutive weeks. Solutions were not buffered. During the treatment, rats were observed at least twice daily for mortality or morbidity throughout the treatment period. Clinical examinations were performed daily at the time of dosing. Physical examinations including palpable mass observations were conducted and recorded on all animals weekly.
Dose group allocation.
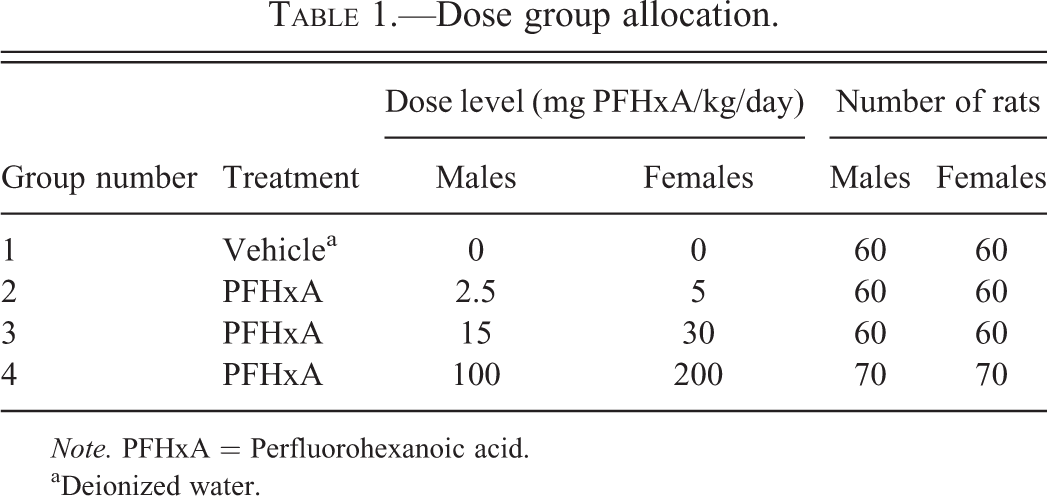
Note. PFHxA = Perfluorohexanoic acid.
aDeionized water.
Body Weight and Food Consumption
Body weights were recorded weekly beginning with the acclimation period through treatment week 14, and biweekly thereafter. Mean body weights and body weight changes were calculated and final body weights were recorded prior to each scheduled necropsy. Individual food consumption was calculated as g/animal/day and recorded weekly beginning with the acclimation period.
Functional Observational Battery and Locomotor Activity
Functional observational battery (FOB) assessment was recorded for the first 12 animals/sex/group during the 52nd week of treatment as previously described (Chengelis et al. 2009). The FOB used was based on previously developed protocols (Moser, McDaniel, and Phillips 1991; Irwin 1968; Gad 1982; Moser et al. 1988; Haggerty 1989; O’Donoghue 1989). Locomotor activity was assessed for the first available 12 animals/sex/group during the 52nd week of treatment. Locomotor activity was measured automatically using the Kinder Scientific Motor Monitor System (Kinder Scientific, LLC, Poway, CA) in a sound-attenuated room equipped with a white noise generator set to operate at 70 ± 10 dB immediately following completion of the FOB assessments. The testing of treatment groups was conducted according to replicate sequence with each animal tested separately. Data for ambulatory and total locomotor activity were tabulated. Total locomotor activity was defined as a combination of fine locomotor skills (i.e., grooming; interruption of a single photobeam) and ambulatory locomotor activity (e.g., interruption of 2 or more consecutive photobeams).
Ophthalmic Examinations
Ocular examinations were conducted on all animals prior to the initiation of dose administration study and during study weeks 51 and 103. All ocular examinations were conducted using an indirect ophthalmoscope, preceded by pupillary dilation with an appropriate mydriatic agent.
Clinical Pathology
Blood smears and blood samples for hematology and serum chemistry (except β hydroxybutyrate, free fatty acid, high-density lipoprotein [HDL], and low-density lipoprotein [LDL] cholesterol) evaluation were collected from all animals euthanized in extremis (if possible). Evaluation of serum chemistry parameters was prioritized as follows when insufficient blood samples were obtained to test for all parameters: liver parameters (alkaline phosphatase [ALP], alanine aminotransferase, aspartate aminotransferase [AST], total bilirubin, total cholesterol, and triglycerides), renal parameters (creatinine, urea nitrogen), total protein, albumin, globulin, albumin to globulin (A/G) ratio, glucose, sodium potassium, chloride, calcium, and phosphorus. Blood was collected from the retro-orbital sinus of animals anesthetized by inhalation of isoflurane at the time of necropsy.
Blood and urine samples for clinical pathology evaluations (hematology, serum chemistry, and urinalysis) were collected from the 10 animals/sex/group in groups 1 to 3 and from 20 animals/sex/group in group 4 during study weeks 25 and 51 (hematology and cholecystokinin) and during study weeks 26 and 52 (serum chemistry, urinalysis, estradiol, luteinizing hormone [LH], and testosterone). Blood smears were prepared from all animals that were euthanized in extremis and from all animals during study weeks 51 and 76. Blood samples collected during the 12th month of the study were collected after study weeks 51 or 52. Hematology parameters were evaluated from all animals at the scheduled necropsy (study week 104). The animals were fasted overnight prior to blood collection. Blood was collected from the retro-orbital sinus of animals anesthetized by inhalation of isoflurane at the time of necropsy. Urine samples were collected overnight using metabolism cages prior to the day blood samples were collected.
Hormones including LH, testosterone, and estradiol were measured in blood by immunoassay. Rat luteinizing hormone (rLH) was measured by 125I radioimmunoassay. Total testosterone and estradiol were measured using a solid-phase 125I competitive labeling radioimmunoassay.
Necropsy and Histopathological Evaluation
A complete necropsy was conducted on all animals. The necropsies included examination of the external surface, all orifices, and the cranial, thoracic, abdominal, and pelvic cavities including viscera. Tissue and organ sampling using standard necropsy protocols (Fiette and Slaoui 2011) was conducted. Organ weights were recorded and all collected organs were placed in 10% neutral buffered formalin for histopathological evaluation (Fiette and Slaoui 2011; Chengelis et al. 2009).
Fixed tissues were paraffin embedded, sectioned into 4 to 8 μm, and stained with hematoxylin and eosin. Microscopic examination was performed on all specified tissues from all animals found dead or euthanized in extremis or at the scheduled necropsy. Any evident neoplasm was graded using the method of Peto (Peto et al. 1980). For animals that were found dead or euthanized in extremis, tumors were graded according to their likelihood of having caused death or morbidity.
Statistical Analysis
Analyses were conducted using two-tailed tests (except as noted otherwise) for minimum significance levels of 1% and 5%, comparing each PFHxA-treated group to the control group by sex. Kaplan–Meier estimates (Kaplan and Meier 1958) and Wilcoxon test (Gehan 1965) were used for survival test. Body weight, body weight change, food consumption, continuous FOB, and clinical pathology data were subjected to a parametric one-way analysis of variance (ANOVA; Snedecor and Cochran 1980), followed by Dunnett’s test (Dunnett 1964). Scalar or descriptive FOB parameters were analyzed using a 2-tailed Fisher’s exact test (Steel, Torrie, and Dickey 1997). Palpable mass data were analyzed by comparing the control group to the treated groups (by sex) and analyzed using a 2-tailed Fisher’s exact test (Steel, Torrie, and Dickey 1997). Tumor incidence was analyzed using linear trend analysis by the method of Peto (Peto et al. 1980) using fixed intervals for incidental tumor analyses, that is, study weeks 0 to 50, 51 to 80, 81 to end of study, and scheduled terminal euthanasia. Each diagnosed tumor type was analyzed separately, and analysis of combined tumor types was performed as previously described (McConnell et al. 1986).
Results
Survival
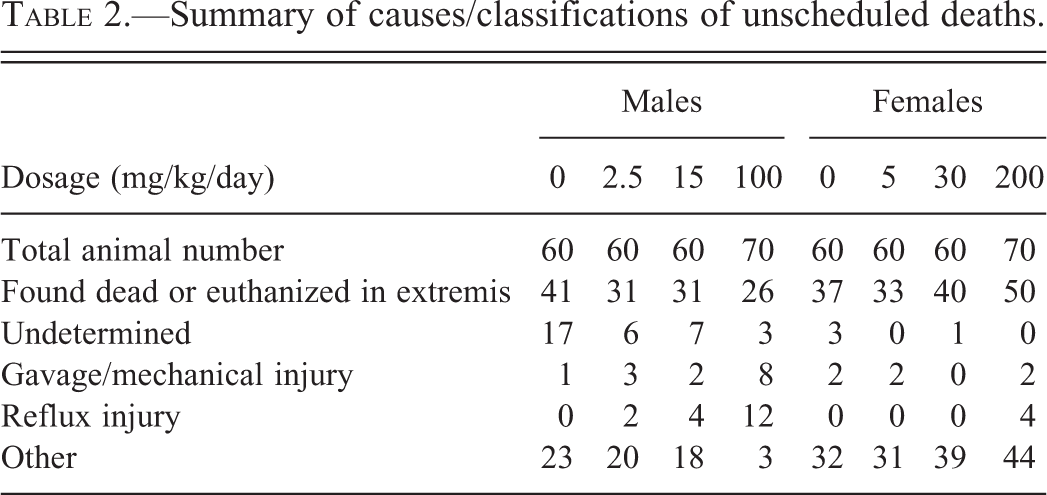
Survival data for male and female rats over the 104-week treatment period are shown in Figure 1A (males) and B (females). Several deaths occurred in rats in the highest dose groups (group 4; males [100 mg/kg/day] and females [200 mg/kg/day] prior to week 52 of the study). These early deaths were considered incidental and classified into 3 categories; reflux injury, gavage/mechanical injury, or undetermined (Table 2). Histopathological evaluations concluded that these incidental deaths were not to be related to the systemic exposure to PFHxA but were primarily due to the gavage dosing. Reflux injury was noted in some of the incidental deaths and appeared to be related to the aspiration of the compound after the gavage dosing that produced localized inflammation and/or epithelial necrosis in the larynx or pulmonary airway epithelium due to the acidic nature of the compound. While the incidence of reflux injury and gavage mechanical injury are noted separately in Table 2, these 2 categories are probably related to the gavage dosing of the animals rather than toxic properties of the compound. Additional support for this conclusion is found in the clustering of the deaths with regard to time of treatment. If the incidental deaths were directly compound related, one would have seen these deaths appear earlier in treatment and continue through the 2-year treatment period at a consistent rate. The numbers of these incidental early deaths from all causes were similar between control, low-, and mid-dose groups for male and female rats.
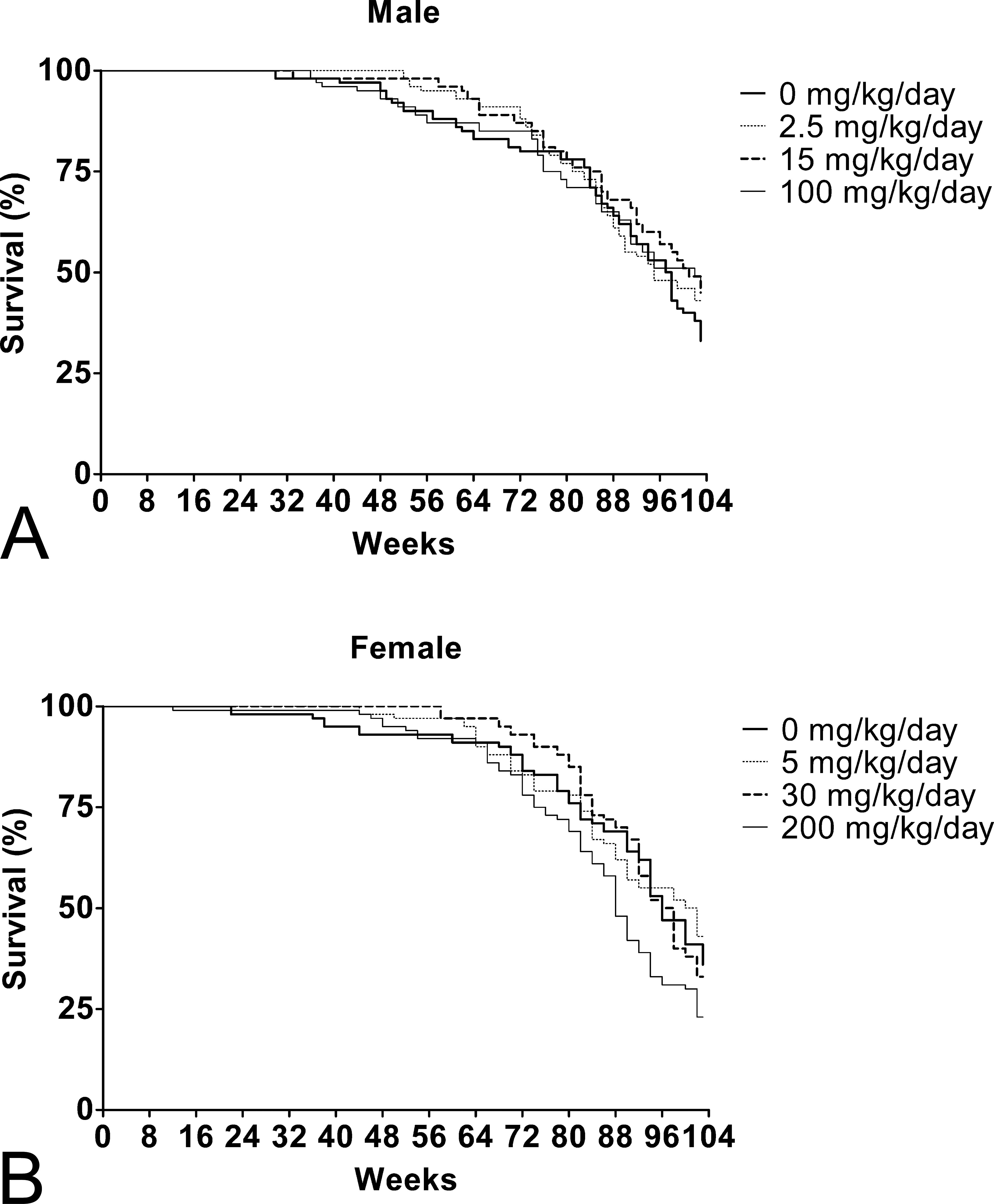
Kaplan–Meier survival curves for rats treated with PFHxA for a total of 104 weeks. (A) Survival curve of male rats treated with 0-, 2.5-, 15-, and 100-mg/kg/day PFHxA. p > .05 for all. (B) Survival curve of female rats treated with 0-, 5-, 30-, and 200-mg/kg/day PFHxA. p > .05 for all. PFHxA = Perfluorohexanoic acid.
Summary of causes/classifications of unscheduled deaths.
At the scheduled necropsy after 104 weeks of PFHxA treatment, no statistically significant difference was seen in survival rates in male rats in any of the 3 groups compared to control (Figure 1A). The survival rate of males, excluding the incidental deaths noted previously, at the end of week 104 in the control, 2.5-, 15-, and 100-mg/kg/day group was 31.0%, 43%, 43%, and 47%, respectively (Table 3). In contrast in treated female rats, a significant dose-related decrease in survival rates was seen (Figure 1B). In addition, there was a statistically significant decrease in pairwise comparisons between the control group and high-dose group. The survival rate of female rats, excluding the incidental deaths, at the end of week 104 in the control, 5-, 30-, and 200-mg/kg/day group was 36%, 43%, 33%, and 22%, respectively (Table 3).
Survival of male and female rats after PFHxA treatment.
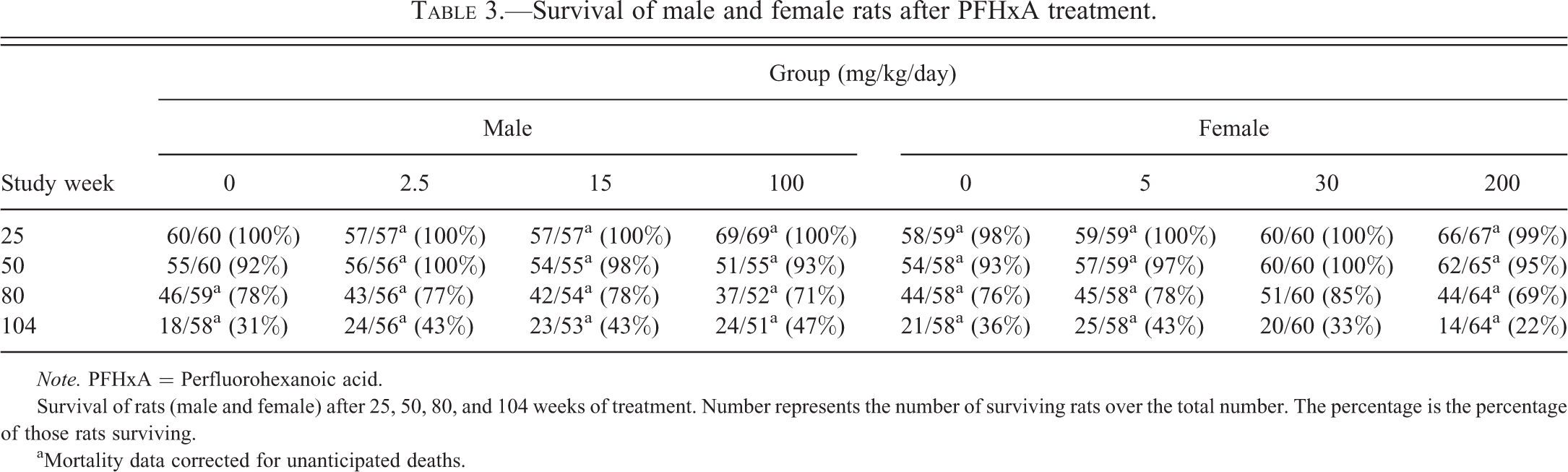
Note. PFHxA = Perfluorohexanoic acid.
Survival of rats (male and female) after 25, 50, 80, and 104 weeks of treatment. Number represents the number of surviving rats over the total number. The percentage is the percentage of those rats surviving.
aMortality data corrected for unanticipated deaths.
Clinical Observations
PFHxA-related clinical observations including rales and yellow material on the ventral trunk, anogenital, and/or urogenital area/areas were seen in the high-dose (100 mg/kg/day) males and high-dose (200 mg/kg/day) females. Very slight increased incidences of struggling during dosing were noted in these same high-dose groups. The struggling may have been related to the acidic nature of the compound since lower dose groups did not exhibit this adverse behavior. Females in the 200-mg/kg/day treatment group appeared to be slightly more sensitive to PFHxA treatment, having a higher incidence of rales and a greater persistence of yellow material at the time of dosing observation periods. This struggling during treatment may have also contributed to the incidental deaths from the gavage and reflux injury noted previously (Table 2) in the high-dose treatments.
Palpable Masses
The incidence of palpable masses was unaffected by administration of PFHxA. The number of multiple masses, the mean number of masses per animal, and the mean number of days to the first mass in the PFHxA-treated groups were similar to those in the control group (data not shown).
Body Weight and Food Consumption
No effect of PFHxA treatment on body weights (Figure 2A and B) or food consumption (data not shown) was seen in male and female rats.
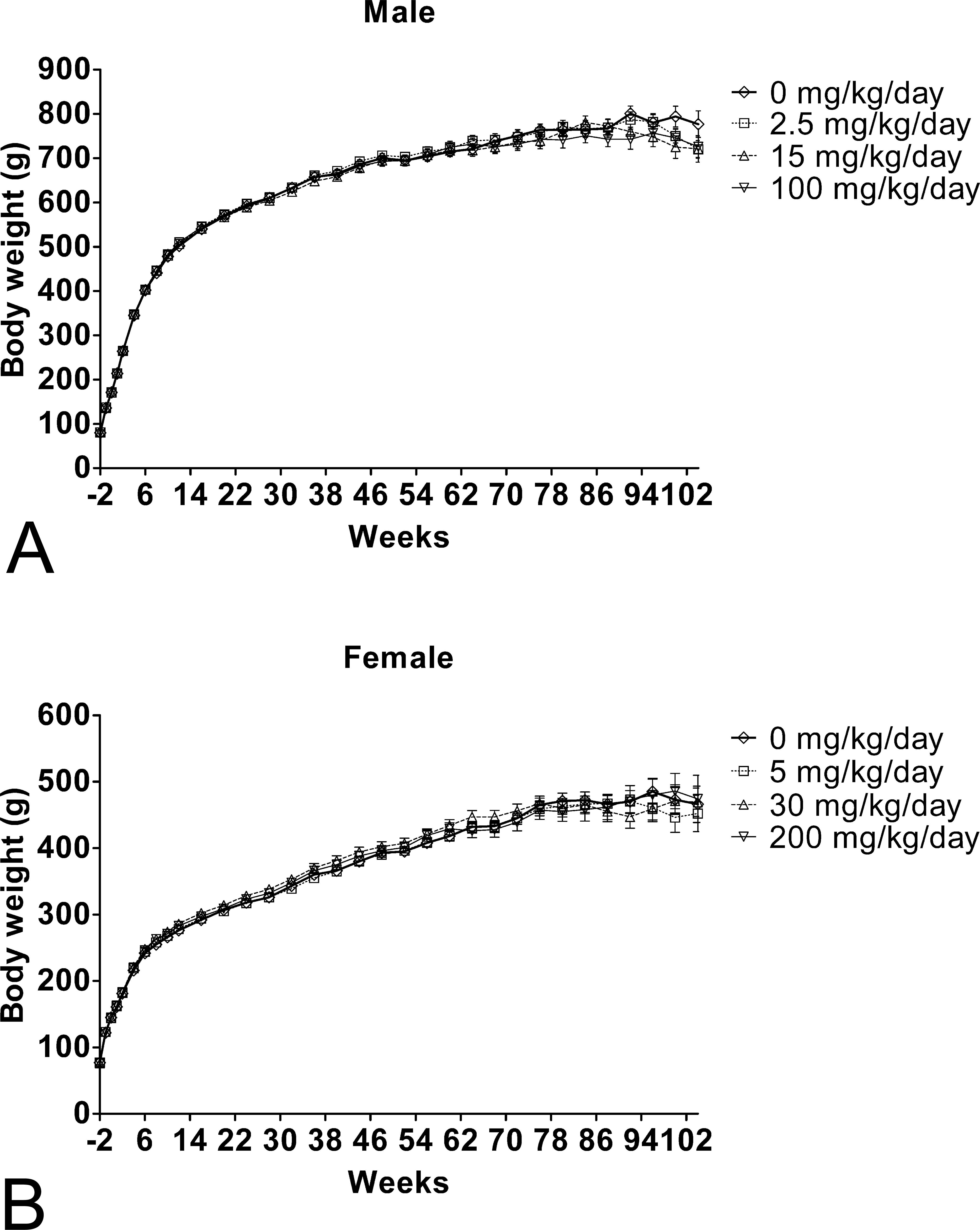
Mean body weight for rats during the 104-week toxicity study with PFHxA. Body weights were recorded weekly from 2 weeks prior to the PFHxA administration till week 14, and then biweekly thereafter till week 104. (A) Body weight of male rats treated with 0-, 2.5-, 15-, and 100-mg/kg/day PFHxA. (B) Body weight of female rats treated with 0-, 5-, 30-, and 200-mg/kg/day PFHxA. Values represent M ± SD. p > .05 for all. PFHxA = Perfluorohexanoic acid; SD = standard deviation.
FOB
FOB observations were not affected by PFHxA treatment (data not shown). No statistically significant changes in locomotor activity patterns (total and ambulatory activity counts) or in the pattern of habituation were seen with PFHxA administration. An increase in the number of male rats in the high-dose (100 mg/kg/day) group asleep or lying on their side compared to the control group was noted. In addition, lower mean grip strength was noted in the 5-mg/kg/day treated females. However, there was a lack of a dose response in these effects and no other correlating FOB or locomotor activity effects were seen. These 2 observations were therefore considered a result of normal biological variability. Hence, consistent with the aforementioned clinical/cage-side observations, it was concluded that PFHxA induced no neurobehavioral toxicity in this study.
Serum Chemistry, Hematology, Urinalysis, and Hormone analyses
At terminal sacrifice (104 weeks), no PFHxA-related changes in hematology parameters were observed (Table 4). After 51 weeks of treatment, mean red blood cell (8.1% lower) and hemoglobin (5.2% lower) values were significantly lower in the 200-mg/kg/day treated females. These red blood cell (RBC) parameters returned to control levels by the terminal sacrifice time. Reticulocyte counts were also statistically significantly higher by 26.3% and 56.3% in the high-dose (200 mg/kg/day) treated females at study weeks 25 and 51. Absolute and percentage reticulocyte counts remained slightly higher in the 200-mg/kg/day group females after 104 weeks of treatment; however, the differences were not statistically significant. The absence of correlating histologic changes and the lack of corresponding hematologic alterations in males suggested that the slight changes in female blood parameters may have been secondary to the renal effects of PFHxA or to gastric ulceration that was slightly increased in the 200-mg/kg/day group females.
Summary of selected hematology data at 25-, 51-, and 104-study week of PFHxA treatment.
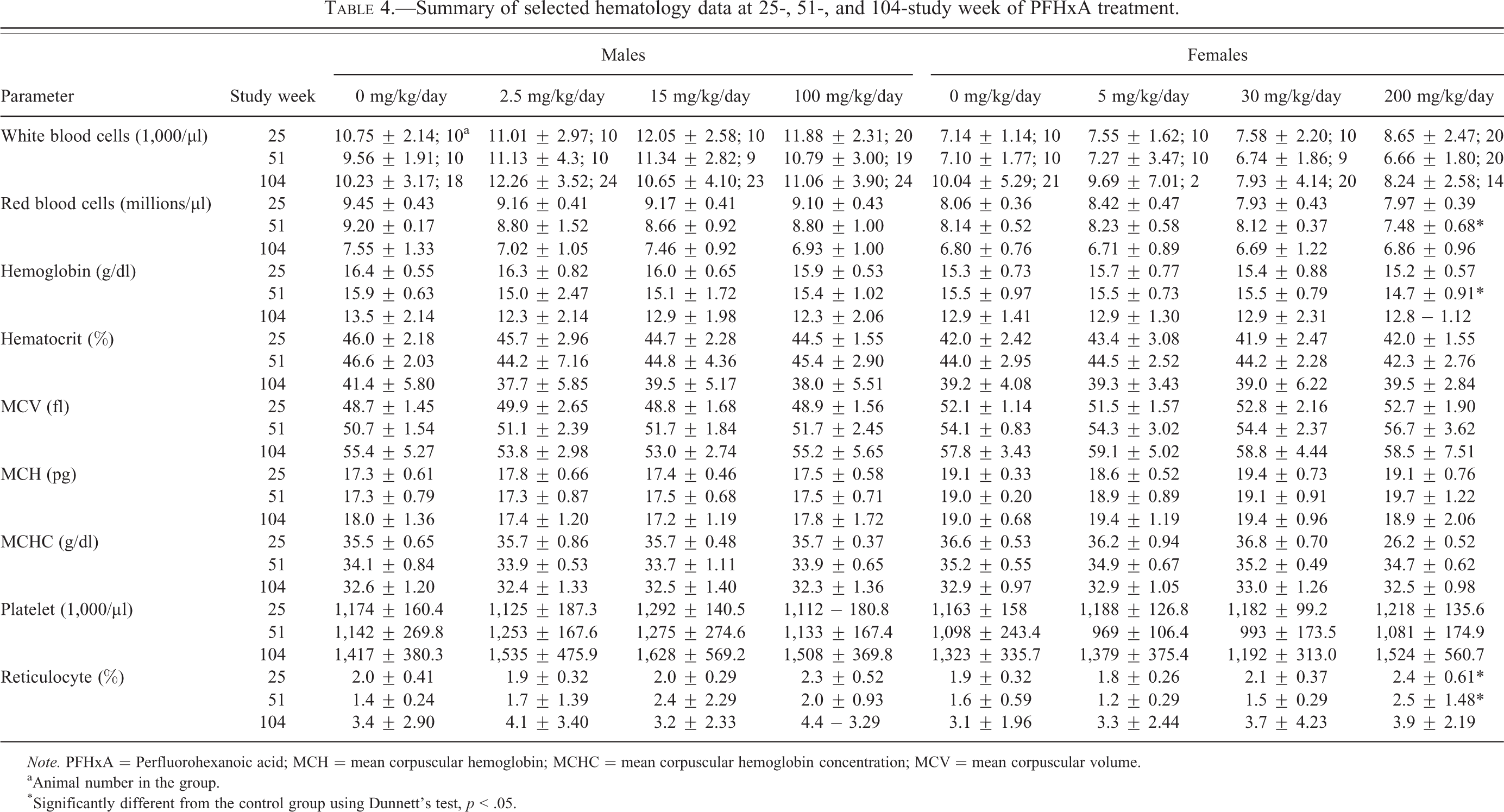
Note. PFHxA = Perfluorohexanoic acid; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume.
aAnimal number in the group.
*Significantly different from the control group using Dunnett’s test, p < .05.
In general, there were no PFHxA-related effects on serum chemistry parameters including albumin (A), globulin (G), A/G ratio, ALP, alanine transaminase (ALT), AST, and free fatty acid (Table 5). However, in study week 52, triglycerides were statistically significantly lower by 47.4% and 42.8% in the 2.5- and 100-mg/kg/day treatment group males, respectively; and free fatty acids were significantly lower by 21.1% and 19.4%, respectively, at study week 52 as compared with controls. In contrast, triglycerides were higher by 66.3% (statistically significant) and 27.2%, in the 200-mg/kg/day group females at study weeks 26 and 52, respectively. Significantly higher inorganic phosphorus levels were noted in the 15- and 100-mg/kg/day group males at study week 52 (8.5% and 6.8%, respectively), and sodium was statistically significantly higher by 0.7% in the 100-mg/kg/day group males. Similar differences were not observed in the females. Additional significant changes included lower LDL and very low-density lipoprotein (VLDL) cholesterol values in the 200-mg/kg/day group females at study week 26 (Table 5). Although these clinical chemistry parameters varied from the control group in a statistically significant manner, most alterations were sporadic in nature, were of a magnitude that would be considered to be toxicologically not important, were not dose-dependent, and/or were not associated with histologic changes. Thus, the mean differences were not considered to be PFHxA related.
Summary of selected serum chemistry data at study week 26 and 52 following PFHxA administration.
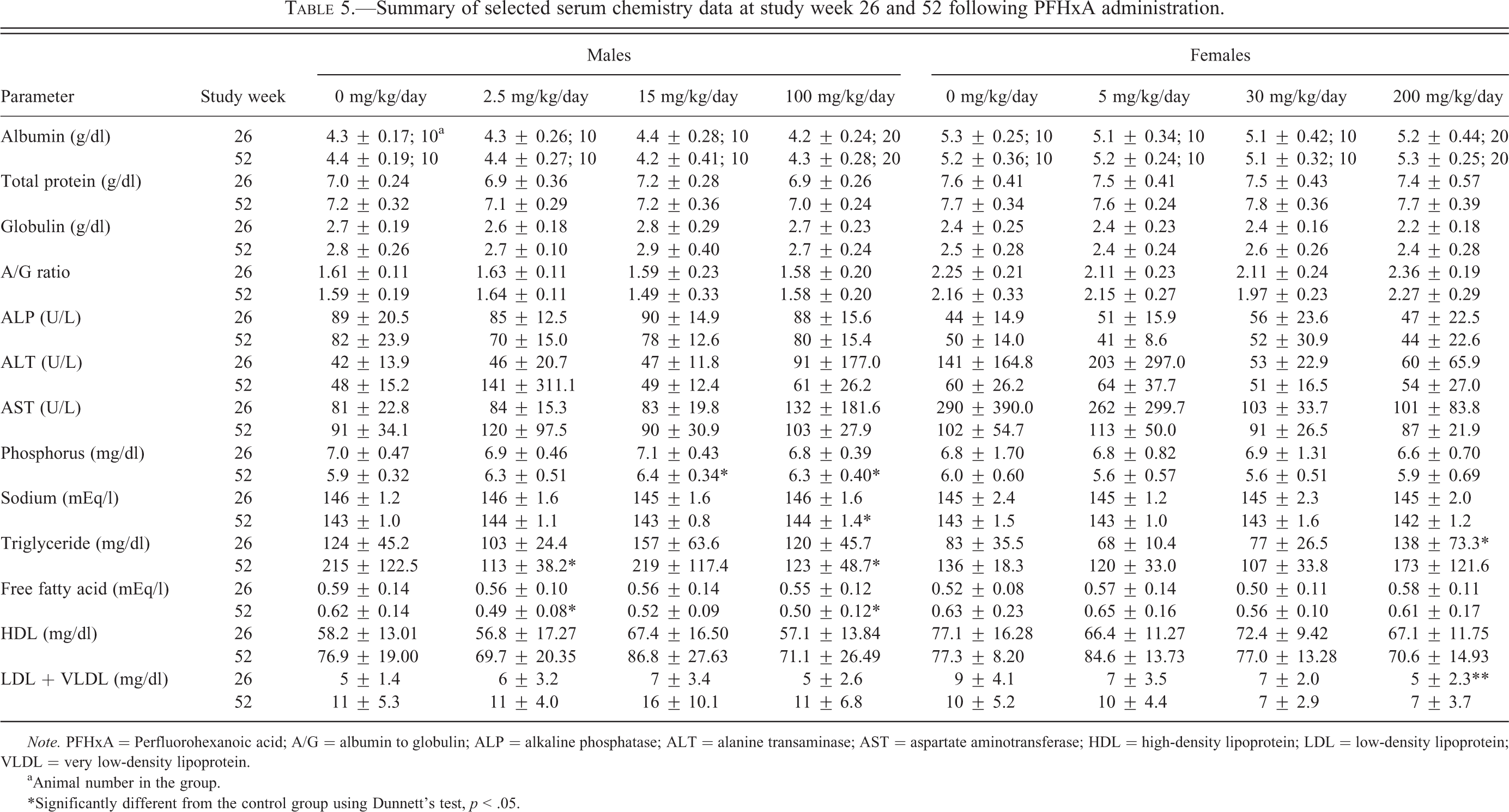
Note. PFHxA = Perfluorohexanoic acid; A/G = albumin to globulin; ALP = alkaline phosphatase; ALT = alanine transaminase; AST = aspartate aminotransferase; HDL = high-density lipoprotein; LDL = low-density lipoprotein; VLDL = very low-density lipoprotein.
aAnimal number in the group.
*Significantly different from the control group using Dunnett’s test, p < .05.
There were no PFHxA-related alterations in hormone parameters including the evaluations for estradiol, LH, and testosterone at the sampling performed after 26 or 52 weeks of dosing or cholecystokinin at the sampling performed after 27 or 53 weeks of dosing (data not shown). Although none of the hormone values varied from the control group in a statistically significant manner, testosterone and LH levels were slightly lower in the 2.5-, 15-, and 100-mg/kg/day group males after 26 weeks of dosing. These differences, however, were not dose-dependent, and by study week 52, the values had returned to near control group levels. Hormone levels in all PFHxA-treated females were similar to control group values at all evaluations.
PFHxA-related effects on urinalysis parameters consisted of higher mean urine volume and lower specific gravity in the 200-mg/kg/day group females, and lower pH values in the 100-mg/kg/day group males (Table 6). At study week 26, mean urine volume was slightly, but statistically significantly, higher, and mean specific gravity was slightly, but statistically significantly, lower in the 200-mg/kg/day group females when compared to the control group. At study weeks 26 and 52, slightly lower pH values were recorded for the 100-mg/kg/day group males. In the 200-mg/kg/day group females, alterations in urine quantitative parameters corresponded with renal tubular degeneration and/or papillary necrosis and were therefore considered to be PFHxA related. The low urine pH noted in the 100-mg/kg/day group males was attributed to the acidic nature of the test substance.
Summary of urinalysis data following treatment with PFHxA for 26 and 52 weeks.
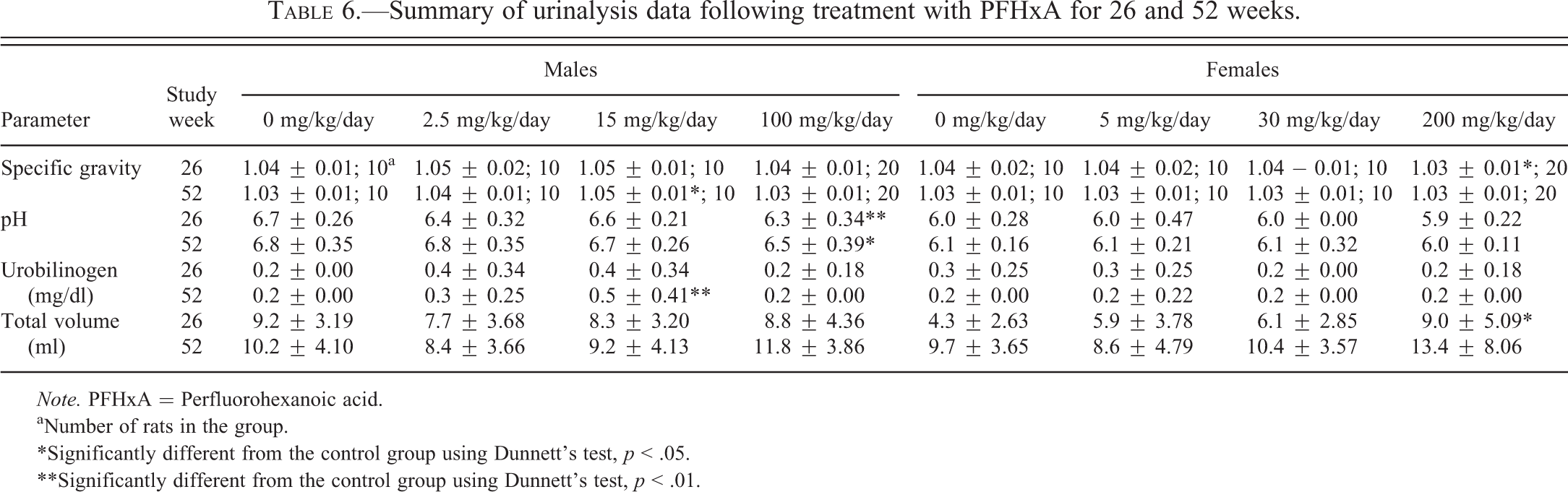
Note. PFHxA = Perfluorohexanoic acid.
aNumber of rats in the group.
*Significantly different from the control group using Dunnett’s test, p < .05.
**Significantly different from the control group using Dunnett’s test, p < .01.
Ophthalmic Evaluations
No PFHxA treatment–related ophthalmic lesions were observed at the scheduled examination and no PFHxA-related macroscopic findings were observed at the schedule necropsy (data not shown).
Histopathological Observations
Nonneoplastic findings
PFHxA-related histologic changes were noted in the kidneys of the high-dose (200 mg/kg/day) treated females. These included minimal to severe papillary necrosis and/or minimal to moderate renal tubular degeneration (Table 7). At intermediate and/or high-dose levels, both sexes exhibited several minor tissue changes that were thought to be secondary to accidental aspiration of PFHxA dosing formulations, having a low pH, and that resulted in compromised pulmonary and hepatic perfusion or mucosal irritation associated with the physical properties of PFHxA. These changes included pulmonary and stomach changes such as acute pulmonary congestion and/or hemorrhage, increased pulmonary alveolar macrophages in males and females, and erosion or ulceration of the glandular or nonglandular stomach in females (Tables 8 and 9). Liver effects were also seen including hepatocellular necrosis and congestion (Table 10). Hepatocellular necrosis was seen throughout the liver (necrosis, hepatocellular) as well as in the centrolobular region of the liver lobule (necrosis, hepatocellular, centrilobular). The necrosis seen in the liver was consistent with ischemia resulting from diminished hepatic blood flow. Most liver cell necrosis was observed in animals that died or were euthanized prior to the scheduled necropsy. While liver necrosis appeared to be greater in the female treated rats, liver congestion was increased significantly in the high-dose treated male rats.
Incidence of histopathological findings in the kidney of PFHxA-treated rats.
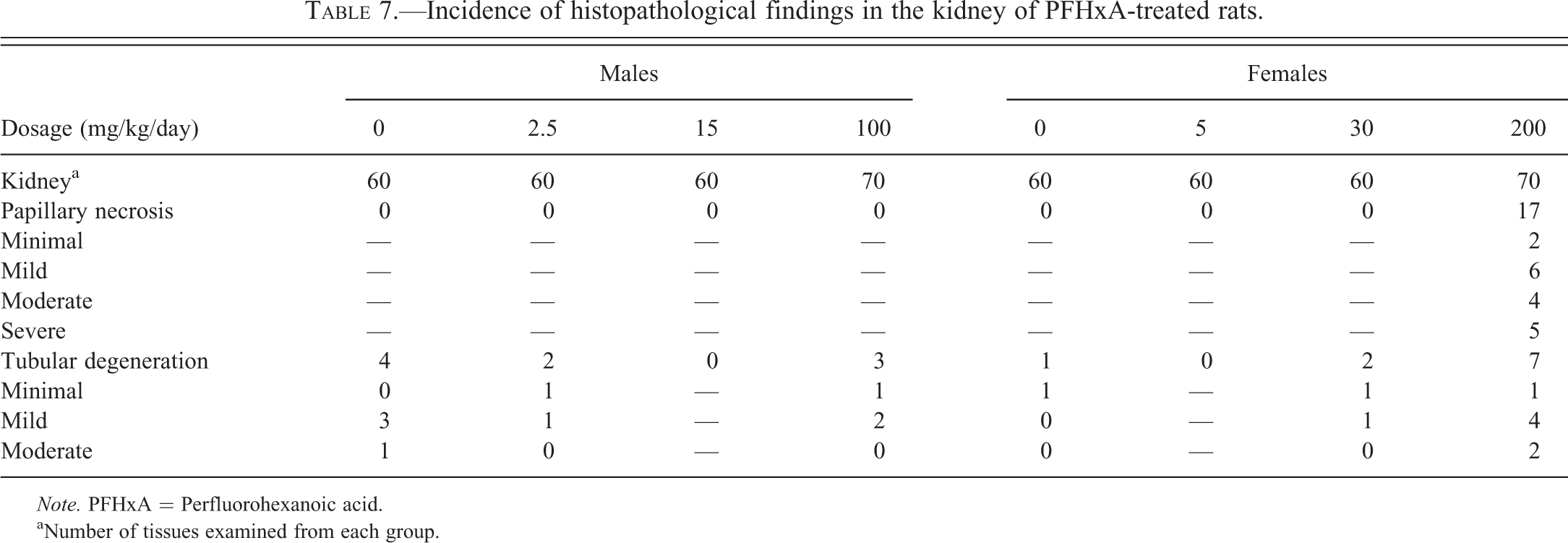
Note. PFHxA = Perfluorohexanoic acid.
aNumber of tissues examined from each group.
Incidence of histopathological findings in the lungs of PFHxA-treated rats.
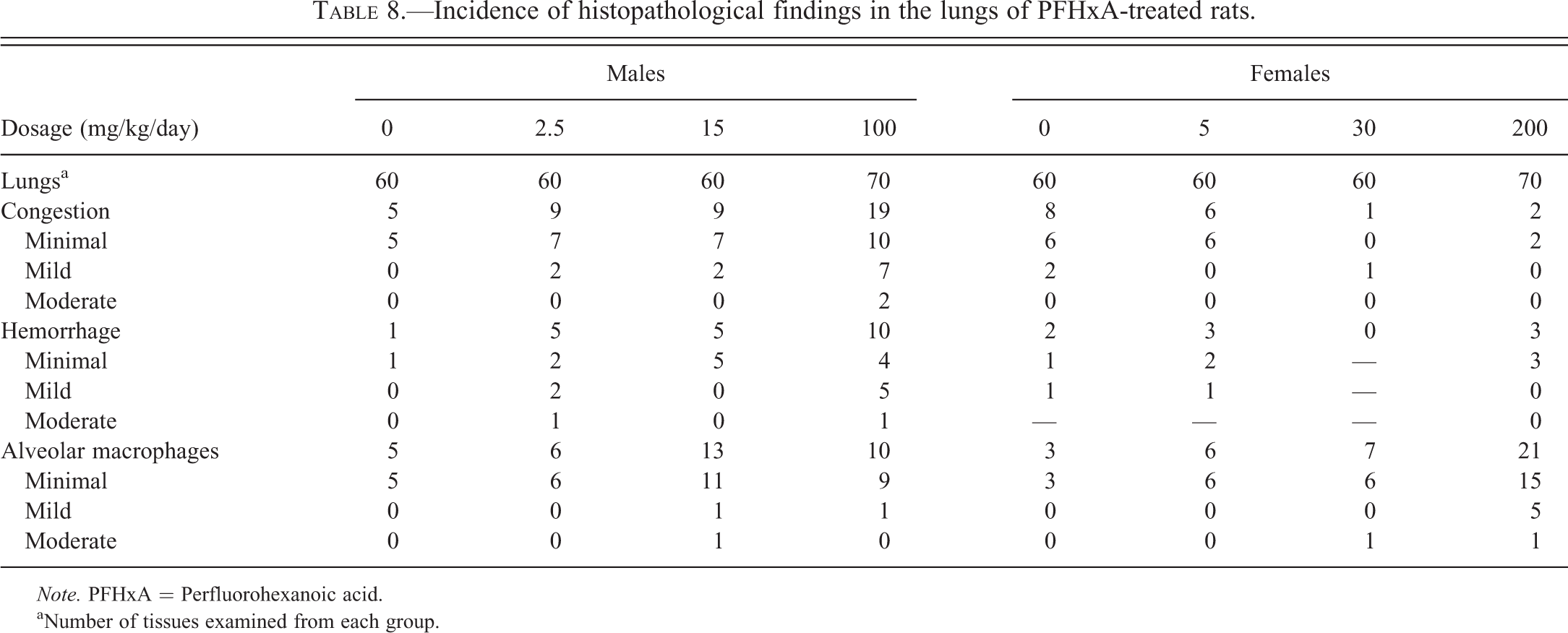
Note. PFHxA = Perfluorohexanoic acid.
aNumber of tissues examined from each group.
Incidence of histopathological findings in the stomach of PFHxA-treated rats.
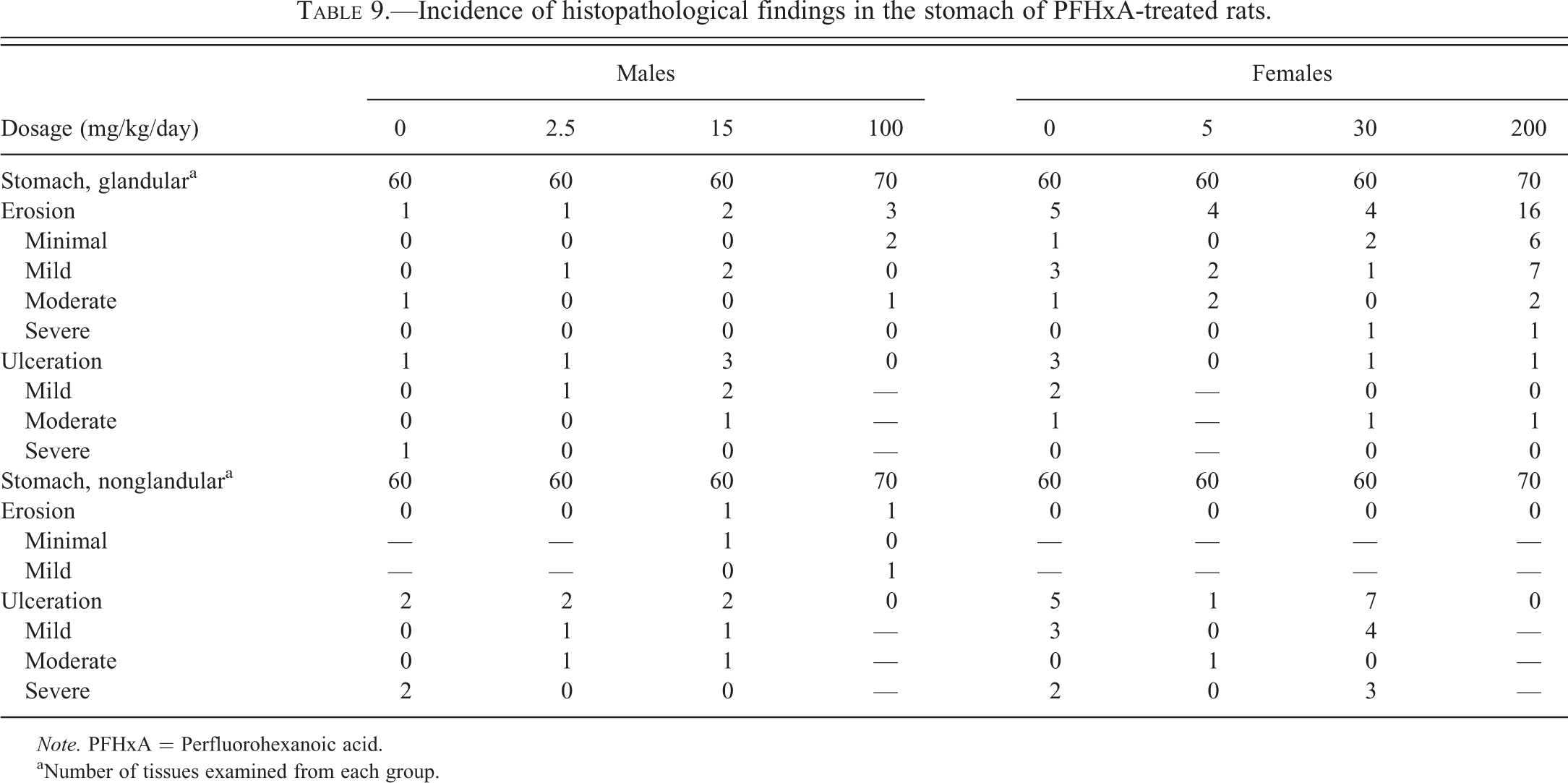
Note. PFHxA = Perfluorohexanoic acid.
aNumber of tissues examined from each group.
Incidence of histopathological findings in the liver of PFHxA-treated rats.
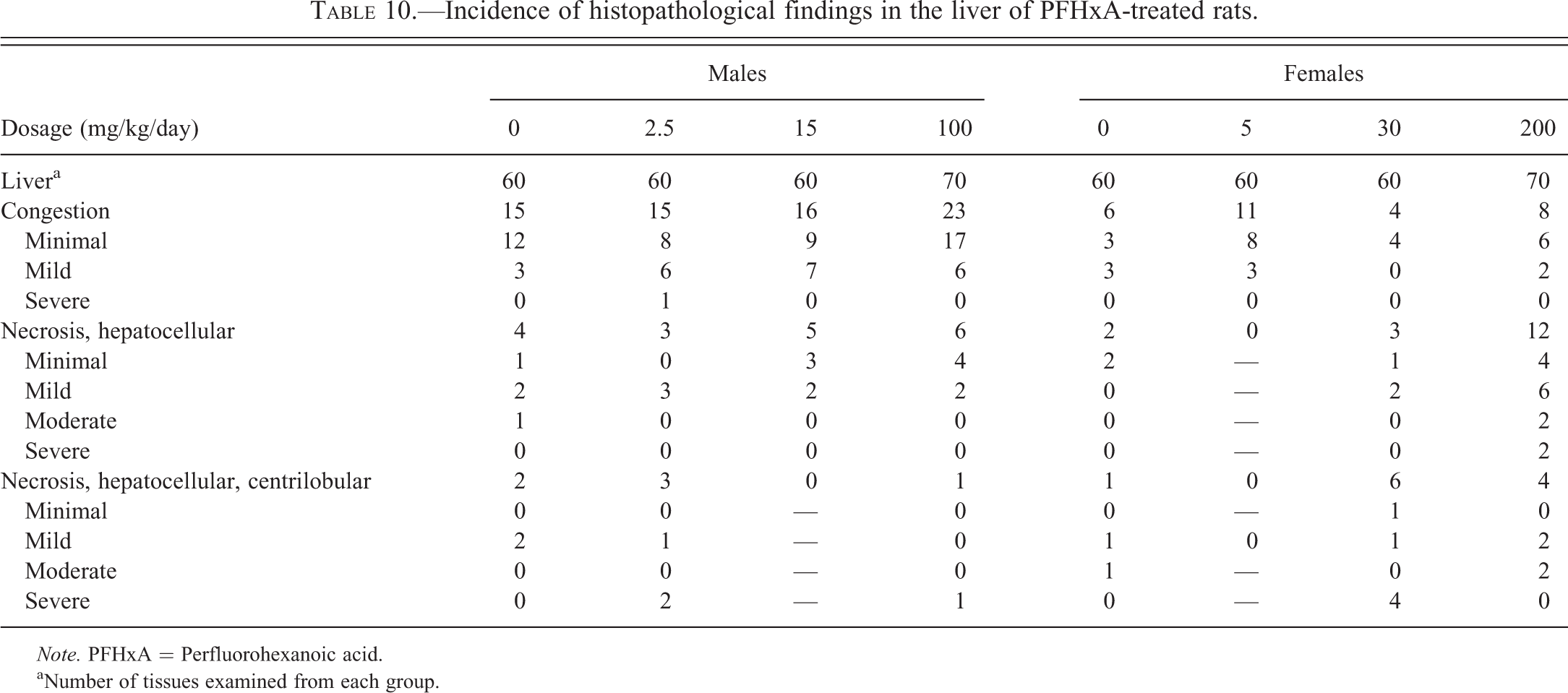
Note. PFHxA = Perfluorohexanoic acid.
aNumber of tissues examined from each group.
Neoplastic findings
No increase in neoplasms related to treatment of PFHxA at any of the 3 dosage levels examined after treatment for 104 weeks was seen in either male or female rats. There was no gross or microscopic evidence that oral gavage with PFHxA at the dosages studied resulted in an increased incidence of neoplasia in any organ. No statistically significant effects of PFHxA on tumor incidence were noted.
Discussion
PFHxA (C6) is an important compound because it has been identified by regulatory agencies as the most likely long-term environmental degrading of a new class of chemicals (C6 chemistry) that have been proposed as alternatives to traditional oil and water repellents often used on outerwear and carpets (USEPA 2013). PFHxA is currently not a commercial product and is not likely to become one in the near future. It is, rather, the ultimate degradation product of C6 fluorotelomer acrylate polymers, and the C6 acrylate monomers and C6 alcohols that are the intermediates used to make C6 fluorotelomer acrylate polymers. These are chemicals that are already being widely marketed as substitutes for C8 fluorotelomer acrylates for which there is a concern for degradation to PFOA. They have both water and oil repellent properties and are very durable. Unlike PFOA, which itself was used as an emulsifier for the polymerization of fluoropolymers, there is no current literature suggesting that PFHxA will be so used. It can be expected that, to a greater or lesser extent, the environmental loading of PFHxA will increase as C6 fluorotelomer acrylate treatments are used and waste is generated. The traditional chemistry of these products was based on C8, linear chains of 8 carbons, with 2 fluorines attached to each of the carbons and additional fluorine attached to a carbon at one end of the chain. The C8 compound, PFOA, is the primary environmental degradant of C8-based chemistry. PFOA has been shown to be highly persistent in the environment, and it has a high retention time in mammals, including humans. It is suspected of being related to various toxicity and ecotoxicity end points and has been shown to be carcinogenic in rodents. Although not generally used in the manufacture of the new class of C6-based chemicals, PFHxA is a nearly unavoidable low-volume impurity in their manufacture, and, probably, the most significant terminal environmental degradant of these chemicals. If C6 chemistry successfully replaces C8 chemistry, it must be expected that PFHxA will migrate to the environment through multiple pathways (Martin et al. 2003a, 2003b; Russell, Nilsson, and Buck 2013). As an example, apparel and carpet applications for C6 chemistry would be expected to lead to environmental releases of PFHxA through laundering and landfill disposal scenarios. So it is of considerable importance to correctly characterize the toxicology of this compound.
This report describes the findings of the 2-year study performed to evaluate the possible toxicologic and carcinogenic effects of the PFHxA when administered orally to SD rats. The dosage levels of 2.5, 15, and 100 mg/kg/day of PFHxA (males) and 5, 30, and 200 mg/kg/day of PFHxA (females) selected for this 2-year bioassay were based on a previous 13-week range-finding study (Chengelis et al. 2009). There was no evidence of carcinogenicity in either male or female rats treated with PFHxA when administered orally daily for 7 days per week for 104 weeks. Pathologic changes related to PFHxA administration were restricted to the kidneys of the high-dose, 200-mg/kg/day treatment group females and were characterized by papillary necrosis and tubular degeneration. These renal effects, however, were sporadic, and the majority of female rats from the 200-mg/kg/day group females survived to the 24-month sampling without evidence of PFHxA-related renal damage. It is unclear from this study what the mechanism was for the selective kidney effects in the female rat. Further investigation will be needed to understand this effect. Minor hematological alterations (lower red blood cell counts and higher reticulocyte counts) were also observed in the 200-mg/kg/day group females. These changes were attributed to slight blood loss from gastric erosion and ulceration, and possibly from renal hemorrhage associated with papillary necrosis, although distinct hemorrhage was not observed microscopically in the renal pelvises. Toxicologically meaningful alterations in serum chemistry parameters were not observed. Changes in urine parameters in male and female rats were likely related to the test substance possibly through the reduced ability of the high-dose (200 mg/kg/day) group females to concentrate urine and as the result of the lower urine pH seen in the high-dose (100 mg/kg/day) group males.
In summary, PFHxA when administered orally daily for 104 weeks at multiple doses was not carcinogenic to male and female rats at dosages that included maximally tolerated levels. Nonneoplastic changes of note including renal changes, papillary necrosis, and tubular degeneration in female rats and changes in urine parameters in male and female rats were likely related to PFHxA treatment. After 104 weeks of PFHxA treatment, no significant difference in survival rates in male rats was seen while the survival rate of female rats at the highest dose studied was significantly decreased. In this regard, PFHxA differed from PFOA (at doses of 300 ppm in the diet), which has been shown to be carcinogenic in SD rats, inducing the tumor triad pattern (liver adenomas, Leydig cell tumors, and pancreatic acinar cell tumors) similar to several PPARα agonists (Biegel et al. 2001). It is important to note that none of the tumor triad organ sites usually seen with PPARα activators (liver, pancreas, and Leydig cells) were modified by PFHxA chronic treatment. Similarly, no preneoplastic changes (i.e., hyperplasia) in these 3 organs were noted following PFHxA treatment. Therefore, under the treatment conditions in this chronic bioassay (daily gavage treatment with PFHxA), PFHxA was not carcinogenic.
Footnotes
Authors’ Note
This work was performed at WIL Research in Ashland, Ohio, and was supported jointly by AGC Chemicals, Asahi Glass Com., Ltd. 10 Goikaigan, Ichihara-shi, Chiba 290-8566, Japan, and Daikin Industries, Ltd. Chemicals Division EHS Department, 1-1 Hitotsuya-Nishi, Settsu, Osaka 565-8585, Japan.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
