Abstract
Subacute and subchronic studies were conducted to assess the toxicity of 2,4-dinitroanisole (DNAN) and to provide information important for protecting the health of military and civilian personnel. In the subchronic study, male and female Sprague-Dawley rats were dosed with DNAN via oral gavage at 0, 1.25, 5, 20, and 80 mg/kg/d. Likely owing to its conversion to 2,4-dinitrophenol, an inhibitor of energy homeostasis, DNAN caused an apparent increase in metabolism, leading to reduced feed efficiency ratios and body mass gains in males. Anemia, splenic enlargement, hemosiderosis, and extramedullary hematopoiesis indicated blood as a target organ, with females more sensitive than males. The DNAN was a testicular toxicant, causing decreased mass of testes and epididymides, as well as degeneration and atrophy of testicular seminiferous tubules and epididymal aspermia. Stereotypical behavior in males, gait irregularities, and cerebellar lesions indicated that DNAN is neurotoxic. Splenic enlargement, anemia, testicular toxicity, and neurotoxicity occurred only at or near lethal doses in the subchronic study.
Introduction
The 2,4-Dinitroanisole (DNAN) is a tan powder with a wax-like consistency that is practically insoluble in water. 1 It is classified as a flammable solid and is being investigated as a less-sensitive replacement for 2,4,6-trinitrotoluene (TNT) in melt-cast insensitive munitions formulations. DNAN is used industrially in the synthesis of dyes and has been used in the past by the US Military as an insecticidal ingredient in MYL louse powder. 2,3 The use of DNAN as an energetic material in explosive formulations dates back to World War II when it was used as the main ingredient in Amatol 40 for various warheads. At the time, the use of DNAN as an ingredient in explosive formulations was based primarily on the scarcity of higher performance materials, such as TNT. Renewed interest in the energetic properties of DNAN has been fueled by the need to develop munitions that are less prone to inadvertent initiation during transport and routine handling. The reduced sensitivity to environmental stimuli and nearly equal performance during testing make DNAN-based formulations desirable replacements for currently fielded munitions. 2 Despite its long historic use, information on the toxicity of DNAN is limited.
The DNAN is moderately acutely toxic, with an oral LD50 of 199 mg/kg in the rat. 4 The DNAN was reported to cause slight skin and eye irritation with reversibility in 24 to 48 hours but did not cause dermal sensitization in the modified Buehler test. 4 The DNAN was evaluated in the Ames Salmonella test (TA98, TA100, TA102, TA1535, and TA1537), with and without metabolic activation (S9). The DNAN was mutagenic only in strain TA100 without activation. 4 Evaluation using Chinese hamster ovary cells (AS52/XPRT) indicated no mutagenic induction at concentrations ranging from 0.0625 to 1.0 mg/mL. 4 The DNAN was judged to have caused no chromosomal damage and to be nonmutagenic in the in vivo mouse bone marrow micronucleus assay at doses of 10 to 90 mg/kg in both male and female mice. 4 Four-hour LC50 values of >2400 mg/m3 and >3000 mg/m3 have been reported for acute inhalation testing in the rat for DNAN 5 and DNAN aerosolized in acetone. 6 In a 14-day inhalation study using DNAN aerosolized in acetone at 150, 500, and 1500 mg/m3, all rats in the 1500 mg/m3 group and 8 of 10 animals in the 500 mg/m3 group were found dead or euthanized during the exposure period. 7 Clinical signs of toxicity observed prior to euthanasia included decreased food consumption, prostration, irregular gait, lethargy, head bobbing, poor condition, pale, backwards walking, labored breathing, and red nasal discharge. Animals exposed to 500 mg/m3 gained less weight and consumed less feed during the first week of exposure than the acetone controls. Females in the 150 mg/m3 group had statistically significant decreases, relative to the acetone control group, in mean hemoglobin (HGB) concentrations, mean corpuscular volume, and mean corpuscular HGB and increases in mean absolute monocytes (MONO) and liver weight. The urine of both male and female rats exposed to 150 mg/m3 was darker than acetone controls. The only reported compound-related microscopic finding was nonspecific minimal metaplasia of laryngeal epithelium in rats exposed to 150 mg/m3. 7 No reports of repeated dose oral testing of DNAN were identified. To ensure its safe use by military personnel and production employees handling the material on a daily basis, the subacute and subchronic oral toxicity of DNAN were investigated.
Materials and Methods
Test Substance
Neat DNAN (CAS # 119-27-7; lot#BAE10H281-008; purity: 100%) was produced by BAE Systems, Ordnance Systems, 4509 West Stone Drive, Kingsport, Tennessee.
Test Animals
Young adult male and female Sprague-Dawley (Crl: CD(SD) CD IGS) rats were obtained from Charles River Laboratories, Wilmington, Massachusetts and were acclimatized for a minimum of 14 days upon arrival. For the 14-day study, 50 male (274.1 ± 5.47 g) and 50 female (201.1 ± 7.72 g) rats (8 weeks old) were distributed, using a stratified random procedure based on body mass, into 7 DNAN treatment groups and a vehicle control group (corn oil) consisting of 6 rats per sex per group. For the 90-day study, 50 male (297.1 ± 10.88 g) and 50 female (214.1 ± 9.14 g) rats (8 weeks old) were similarly distributed into 4 DNAN treatment groups and a control group of 10 rats per sex per group. In both studies, males and females were each then divided into 2 experimental start dates, with treatment groups evenly distributed among days, to facilitate scheduling of necropsies. Housing conditions were maintained at 64°F to 79°F, 30% to 70% humidity, and with a 12-hour light/dark cycle. A certified pesticide-free rodent chow (Harlan Teklad, 8728C Certified Rodent Diet, Indianapolis, IN) and drinking quality water were available ad libitum. Rats were housed individually in suspended polycarbonate cages with ALPHA-dri bedding (Shepherd Specialty Papers, Milford, NJ). Animal use procedures were approved by the Animal Care and Use Committee at the Army Public Health Center (APHC). Animal care and use was conducted in accordance with the principles stated in the Guide for the Care and Use of Laboratory Animals and in accordance with all applicable regulations. 8 The APHC animal care program is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. These studies were conducted in compliance with Good Laboratory Practice standards as described in Title 40, Code of Federal Regulations, Part 792.
Test Article Administration
Dosage levels for the 14-day study were set at 1.5, 3, 6, 12.5, 25, 50, and 100 mg/kg/d and were set at 1, 5, 20, and 80 mg/kg/d for the 90-day study. Vehicle control animals were dosed with corn oil at the same volume per body mass (1.56 and 5 mL/kg in 14- and 90-day, respectively) as the DNAN-exposed animals. The DNAN solution/suspensions and corn oil control were administered at approximately the same time daily, 7 d/wk, using a stainless steel 16 gauge × 2 inch gavage needle.
The concentration of each dosing solution preparation was verified analytically. Samples were diluted in acetone and isoamyl acetate and analyzed using a gas chromatograph equipped with an electron capture detector and a DB-210 column. Homogeneity of the suspensions was verified by determining the concentration of samples taken from the top, middle, and bottom of the highest concentration (64 mg/mL) suspension. In addition, samples were collected from a representative suspension (5 mg/mL) at weekly intervals prior to the 14-day study to determine the stability of the dosing suspensions. The homogeneity and stability studies determined that suspension concentrations were nearly uniform throughout the sample container and throughout a 1-month sampling period.
Observations, Body Mass, and Food Consumption
Observations for mortality and signs of toxic effects were made at least twice daily, except on weekends when observations occurred only in the morning. Additionally, each animal was removed from its cage daily and given a clinical examination.
Animals were weighed twice prestudy and weekly during the study. Terminal body mass was obtained the morning of necropsy following overnight fasting. Feed consumption was determined on a weekly basis.
Neurobehavioral Observations
Potential neurotoxic effects of DNAN were evaluated using the functional observation battery (FOB) and motor activity assessment. The FOB protocol evaluated agitation, convulsions, tremors, posture, mutilation, lacrimation, salivation, piloerection, palpebral closures, exophthalmos, pupillary status, ease of removal from cage, reactivity to handling, grooms, arousal, urine and fecal boli, fecal description, gait, slow approach response, auditory startle response, tail pinch, pinna response, pupillary response, righting reflex, aerial righting, landing foot splay, forelimb grip strength, hind limb grip strength, basic movement, immobility, X and Y ambulation, and nose pokes following the methods described in the study of McDaniel and Moser. 9 Animals from the 90-day study were divided into 2 subsets for each sex, using a stratified random procedure based on dose group. The FOB was conducted on each animal prior to initiation of dosing and weekly thereafter, with 1 subset of animals being assessed per day. The order of animals evaluated each day was randomly determined prior to study initiation. The FOB was performed at the same time each morning prior to dosing. Each rat was removed from its cage and held by the observer to conduct the handheld observation of reactivity and appearance. The rat was then placed on a cart to conduct the open arena observations of gait, arousal, rears, and excretions. Home cage observations were performed on the same day each week on all animals. During week 11 of dosing, sensorimotor responses were tested after the open arena observations. Observations and FOB were performed by the same evaluator throughout the study; the evaluator was blind to the treatment groups. Motor activity was measured after week 11 of dosing using an open field chamber with automated detection devices (SmartFrame Open Field Activity System, Hamiton-Kinder, Poway, CA, and MotorMonitor software, version 4.14).
Ophthalmoscopic Examinations
All study animals in the 90-day study were examined prestudy, and animals from the control and high-dose (80 mg/kg/d) groups were examined during the last week of the study. The fundus and anterior chamber of the eye were examined using a Welch Allyn ophthalmoscope (Welch Allyn, Skaneateles Falls, NY) after instillation of tropicamide ophthalmic solution (1%).
Urinalysis
During the last 2 weeks of the 90-day study, each animal was placed in a metabolism cage capable of separating urine and feces for 1 overnight period of approximately 12 hours during which free-catch urine was collected. Animals were fasted during this period, but water was available ad libitum. Urine samples were transferred to clear graduated conical centrifuge tubes, and the volume, color, and appearance of each sample were recorded. The color of each sample was determined based on comparison with a urine color chart with 9 colors ranging from pale yellow/straw to dark amber. 10 Specific gravity was tested using a refractometer. Multistix 7 Reagent strips (Siemens, Erlangen, Germany) were used to conduct chemical analyses including pH, protein, glucose (GLU), ketones, bilirubin, blood, urobilinogen, nitrites, and leukocytes.
Necropsy
Rats that died during the course of this study were submitted for gross necropsy. Tissues that were not grossly autolytic were submitted for histopathological evaluation. All surviving rats were anesthetized with carbon dioxide (CO2), blood was collected by intracardiac puncture, and rats were euthanized using CO2. Necropsy order was randomized across treatment groups. A macroscopic examination was conducted on all terminal animals, noting all lesions and abnormal observations. The adrenals, brain, heart, kidneys, epididymides, liver, ovaries, spleen, testes, thymus, and uterus were removed, trimmed in a uniform manner, and weighed. Paired organs were weighed together.
Histopathology
Tissues were preserved in 10% buffered formalin and selectively trimmed. Tissues were routinely processed and paraffin embedded. All processed and embedded tissues were microtomed at 5-µm thick and routinely stained with hematoxylin and eosin and cover slipped. A board-certified pathologist examined slides for compound-induced histopathologic changes via light microscopy. The prevalence and severity of findings were graded compared to controls. Findings were assigned as none, minimal, mild, moderate, or severe/marked.
Sperm analysis
Cauda epididymal sperm counts were determined in males from the 90-day study using a computer-assisted sperm analyzer (TOX IVOS-CASA, Hamilton Thorne Research, Beverly, Massachusetts). After removal, trimming, and weighing, 1 epididymis was further trimmed to select the cauda portion, weighed, placed in 200 µL Roswell Park Memorial Institute-1640 medium (RPMI-1640; Sigma-Aldrich, St. Louis, Missouri) at 37°C, and minced using fine scissors followed by a gentle grinding with a pestle to release sperm. A chamber of a standard count analysis slide (Leja, Spectrum Technologies, Healdsburg, California) was loaded with the semen suspension, and the slide loaded into the semen analyzer. The number of sperm, number of motile sperm, and number of progressive sperm were determined in duplicate for each animal.
Clinical Chemistry and Hematology
Blood was obtained from CO2 anesthetized animals via intracardiac puncture at the termination of the study. Blood for clinical chemistry analyses was transferred to collection tubes free of anticoagulant, allowed to clot for at least 20 minutes, and centrifuged to obtain serum. Blood for hematology analyses was transferred immediately to tubes containing tripotassium ethylenediaminetetraacetic acid (K3EDTA). Animals were fasted overnight prior to blood collection.
The evaluated clinical chemistry parameters included the following: albumin, alkaline phosphatase, alanine aminotransferase (ALT), aspartate aminotransferase, blood urea nitrogen (BUN), calcium, cholesterol (CHOL), creatinine, GLU (fasting), globulin, lactate dehydrogenase, inorganic phosphorous, total bilirubin, total protein, sodium (Na), potassium (K), and chloride (Cl). Results were determined using the VetTest 8008 chemistry analyzer and the VetLyte Na, K, Cl analyzer (IDEXX Laboratories, Inc, Westbrook, Maine) on all valid serum samples.
The evaluated hematology parameters included the following: white blood cell (WBC) count, WBC differential (% neutrophils, % lymphocytes, % monocytes (MONO %M), % eosinophils, % basophils), red blood cell (RBC) count, HGB, hematocrit (HCT), mean cell volume (MCV), mean cell hemoglobin (MCH), mean cell hemoglobin concentration (MCHC), red blood cell distribution width (RDW), platelets (PLTs), and mean platelet volume (MPV). Results were determined using the Cell-Dyn 3700 Hematology Analyzer (Abbott Laboratories, Abbott Park, Illinois) on all valid samples.
Statistical Analyses
For continuous experimental variables measured at the end of the study, dose groups were compared using a 1-factor analysis of variance (ANOVA). Organ-to-brain and organ-to-body mass ratios were calculated and analyzed similarly to other parameters measured at the end of the study. If the ANOVA was significant, post hoc tests were used to compare pairs of dose groups, a Tukey multiple comparison test if the variance of groups were similar, and a Dunnett T3 test if the variances were unequal. Variance equality was determined by a Levene test. For body mass changes and food consumption over time, a repeated measures ANOVA with time as the repeated factor and dose group as between-group factor was used to assess changes in times and dose groups.
Neurobehavioral evaluations generated 2 types of data, continuous/count variables and categorical variables. The continuous/count variables were either measurements or counts of a specified action. The categorical variables were either the presence or absence of a response or a severity of occurrence. Due to the low frequency of grooms, this count variable was converted to a categorical (presence/absence) variable for analysis. For the motor activity data, the 15 interval recordings were averaged to obtain a single number per rat. The nose poke response was calculated by totaling the 9 nose poke recordings per interval and then taking an average over the 15-minute interval. For continuous data, an ANOVA was used to test for differences between treatment groups separately for each sex. If the ANOVA revealed significant differences, a Dunnett C test was used if variances were homogenous and a Dunnett T3 test if variances differed between treatment groups. Levene test was used to test the homogeneity of variance among treatment groups. For categorical data, Fisher exact test was used to test for differences between treatment groups at each week for each sex. If significant differences were observed, then a Mann-Whitney test was conducted to compare pairs of treatment groups. The SPSS version 16.0 and SAS version 9.2 were used for all statistical analyses. Statistical significance was defined as
Results
Analytical Chemistry
Analytical concentrations of DNAN were 98% to 115% and 84% to 120% of nominal concentrations in the 14- and 90-day studies, respectively. All results were within the 70% to 130% recovery limits for the analysis. As such, all results were reported using the nominal concentrations.
Mortality and Clinical Observations
In the 14-day study, no mortalities were observed. Clinical signs of toxicity, including lethargy, prostrate posture, rapid respiration/labored breathing, dark urine, orange feces, and barbering, were observed in the 100 mg/kg/d dose groups. Lethargy, dark urine, congested breathing, and barbering were noted in the 50 mg/kg/d groups. Clinical signs in lower dose groups were limited to barbering.
In the 90-day study, mortality or morbidity was observed in the 80 mg/kg/d group in 3 males found dead on days 51, 64, and 79, and 1 female euthanized on day 19. Clinical signs of toxicity, including lethargy, prostrate or recumbent posture, rapid/labored respiration, ataxia, irregular gait patterns (walking on toes, creeping, dragging hind end, hind end raised, pulling legs up, stiff legs, limping, dragging leg, walking backwards in circles, hind end wobbling, movements jerky, and stiff/locked muscle/limb), partial hind limb paralysis, complete front limb paralysis, dark urine, squinting, hunched posture, twitching ears, twitching tail, body twitching, head shaking, leaning to left, straub tail, curled tail, barbering, rough hair coat, piloerection, low arousal, red discharge from nose, and chromodacryorrhea, were noted in the 80 mg/kg/d group. Although the overall signs noted for males and females were similar, there was an apparent gender difference in the pattern of clinical signs. Males were consistently observed in dorsal or lateral recumbency starting approximately 3 hours after DNAN was administered and lasting approximately 3 hours. Males in this posture were alert and responsive, immediately righting themselves when disturbed. Females were observed in this posture infrequently. Males and females both demonstrated gait irregularities; however, males tended to creep with lowered hind quarters, whereas females tended to walk on their toes and high step while walking; both genders had difficulty moving the hind limbs due to what appeared to be muscle stiffness/tetany. All male DNAN dose groups exhibited dark urine, curled tail, dorsal and lateral recumbency, irregular gait (creeping, hind end lowered, and stiff muscles), chromodacryorrhea, and barbering. Lethargy, prostrate posture, congested breathing, and labored breathing were also noted in males in the 20 mg/kg/d dose group. Clinical signs in females in the 20 mg/kg/d group included dark urine, low arousal, irregular gait (walking on toes), and lateral recumbency. Clinical signs in females in lower dose groups were limited to barbering, alopecia, and congested breathing, with the exception of 1 female in both the 1 and 5 mg/kg/d groups with hind limb ataxia.
Body Mass and Food Consumption
In both the 14- and 90-day studies, with the exception of fasting during urinalysis, body mass generally increased with time for all dose groups throughout the study. Body mass of male rats given 100 mg/kg/d DNAN in the 14-day study was reduced relative to the corn oil control (5%-9%) throughout the study, whereas body mass of male rats given 50 mg/kg/d was reduced (8%-9%) at days 7 and 13 (Figure 1). Body mass of male rats given 80 mg/kg/d DNAN in the 90-day study was reduced relative to the corn oil control (5%-9%) from week 1 through week 13 (Figure 2). Body mass of female rats did not differ between treated and control groups at any time during either the 14- or 90-day studies.
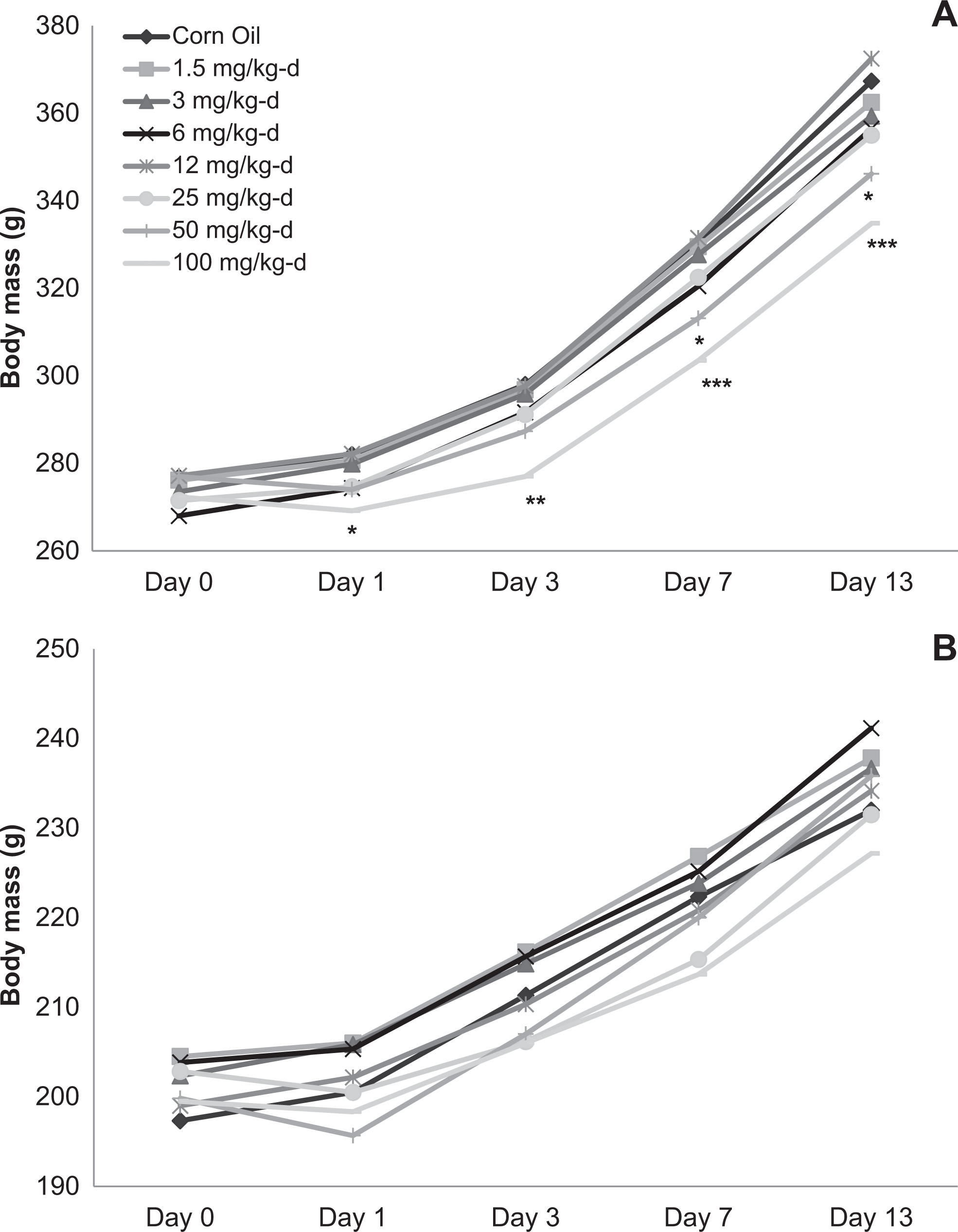
Body mass of male (A) and female (B) Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 14 days. *Significantly different from control at
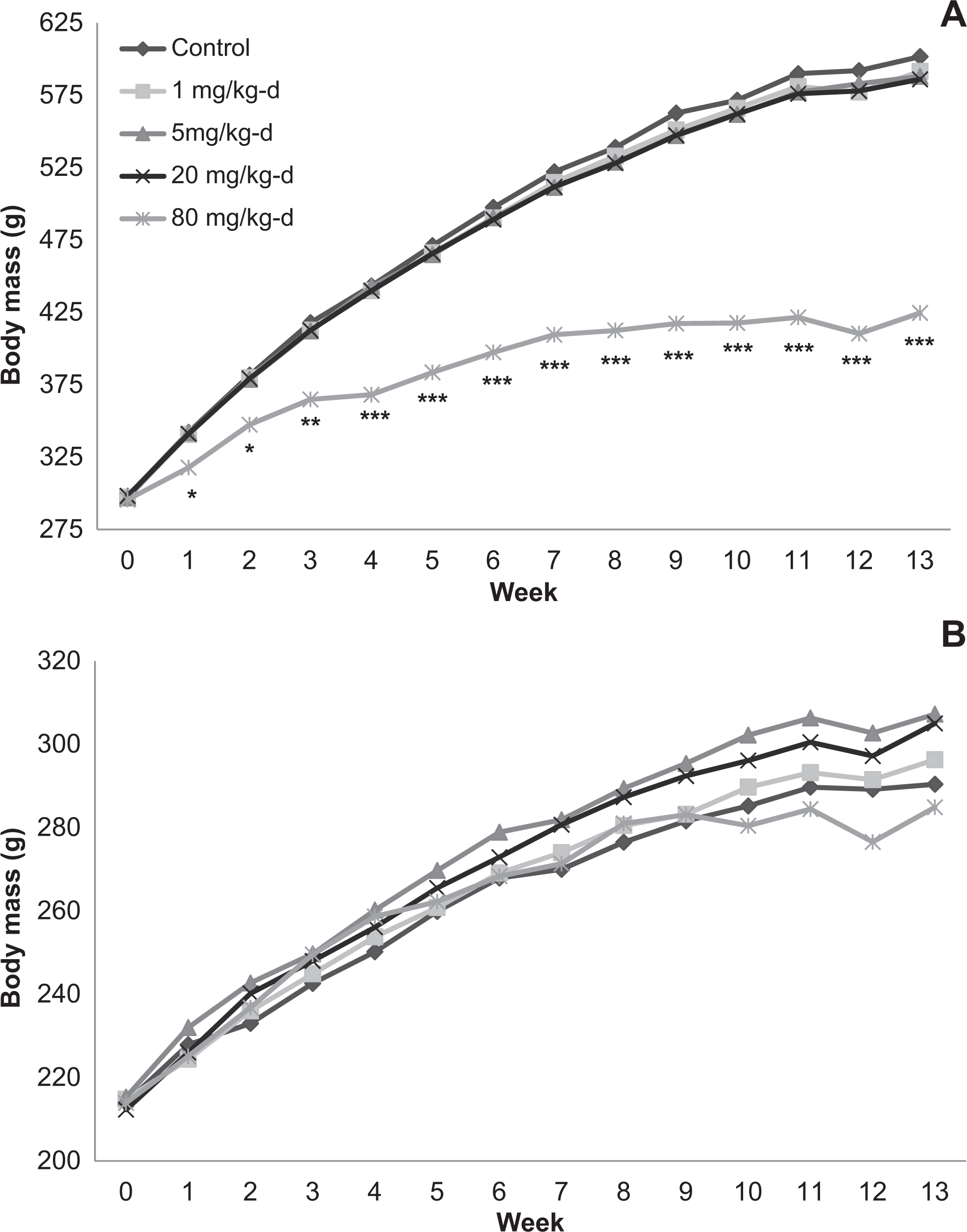
Body mass of male (A) and female (B) Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. *Significantly different from control at
The DNAN did not affect food consumption in the 14-day study (data not shown). In the 90-day study, food consumption was increased in males given 20 mg/kg/d and females given 20 and 80 mg/kg/d; however, this increase was only significant in females in the 80 mg/kg/d group (Figure 3). Males in the 80 mg/kg/d dose group in the 90-day study initially exhibited an increase in food consumption similar to that observed in the 20 mg/kg/d group; however, this increase dissipated by week 4.
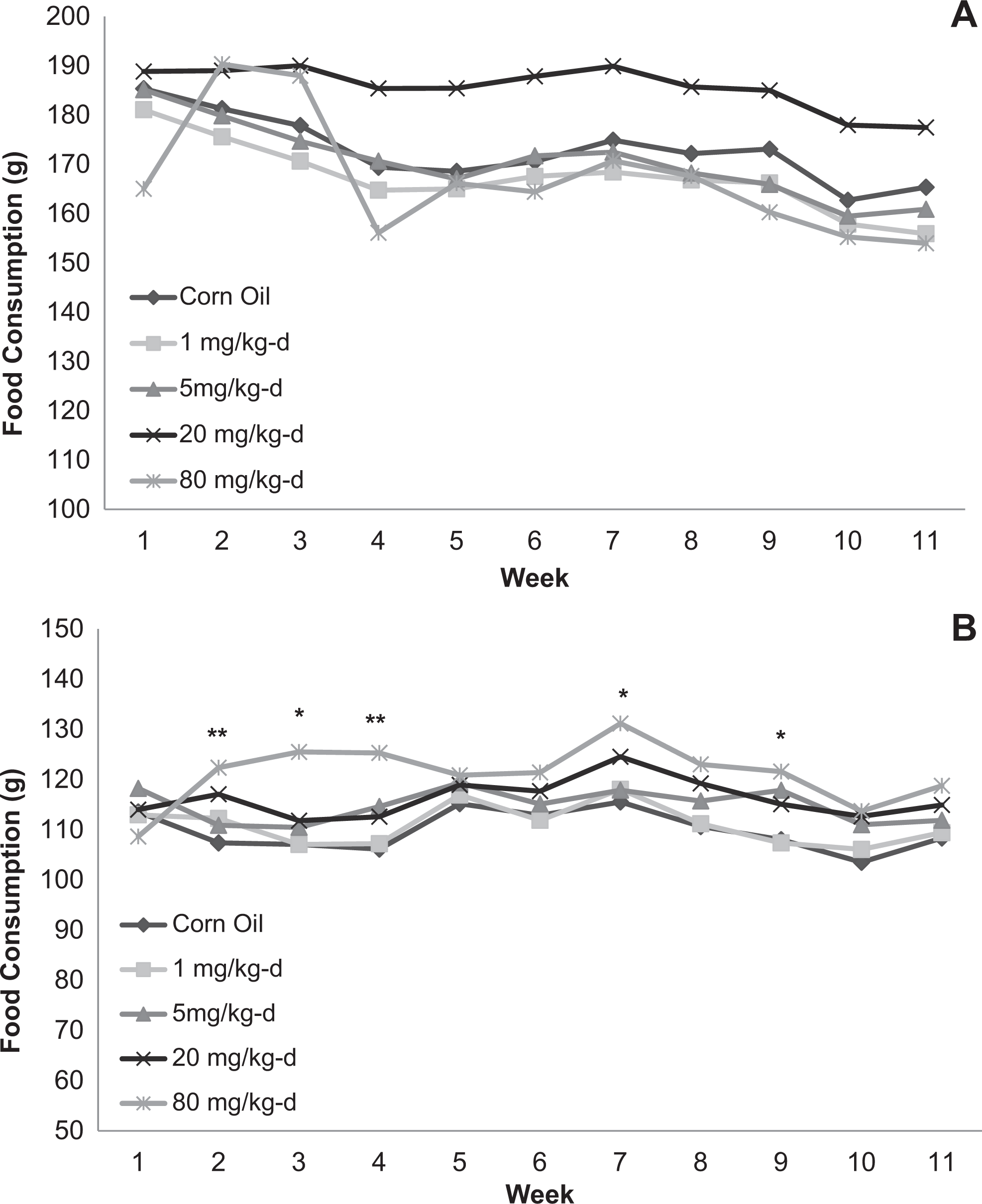
Food consumption for male (A) and female (B) Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. *Significantly different from control at
Food efficiency ratios were reduced in male rats in the 14- and 90-day studies. In the 14-day study, food efficiency ratios were reduced in males given 50 or 100 mg/kg/d (Figure 4). Food efficiency ratios were reduced in male rats given 80 mg/kg/d in the 90-day study (Figure 5). Food efficiency ratios did not differ among treated and control groups for female rats in the 90-day study.
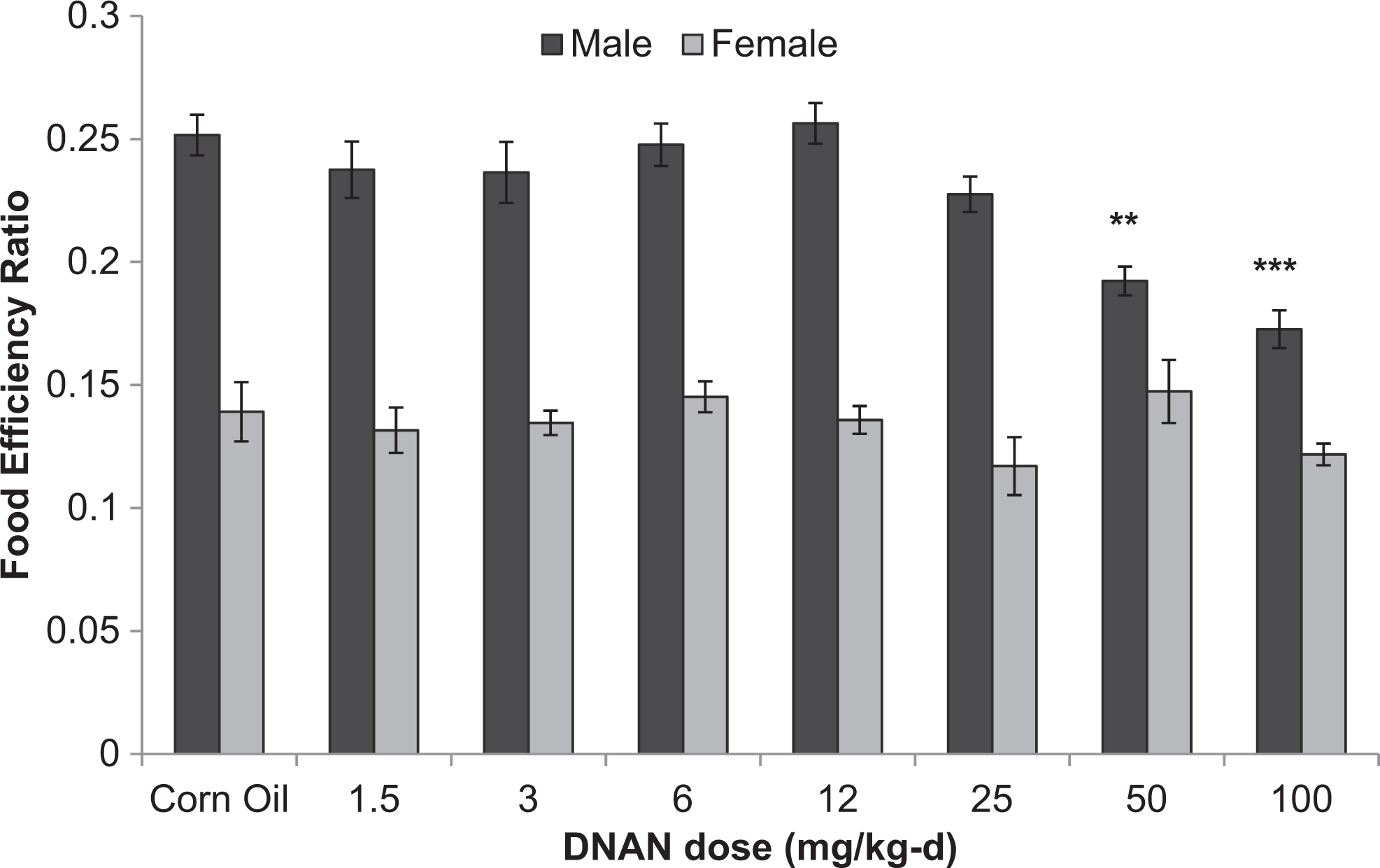
Food efficiency ratios for male and female Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 14 days. **Significantly different from control at
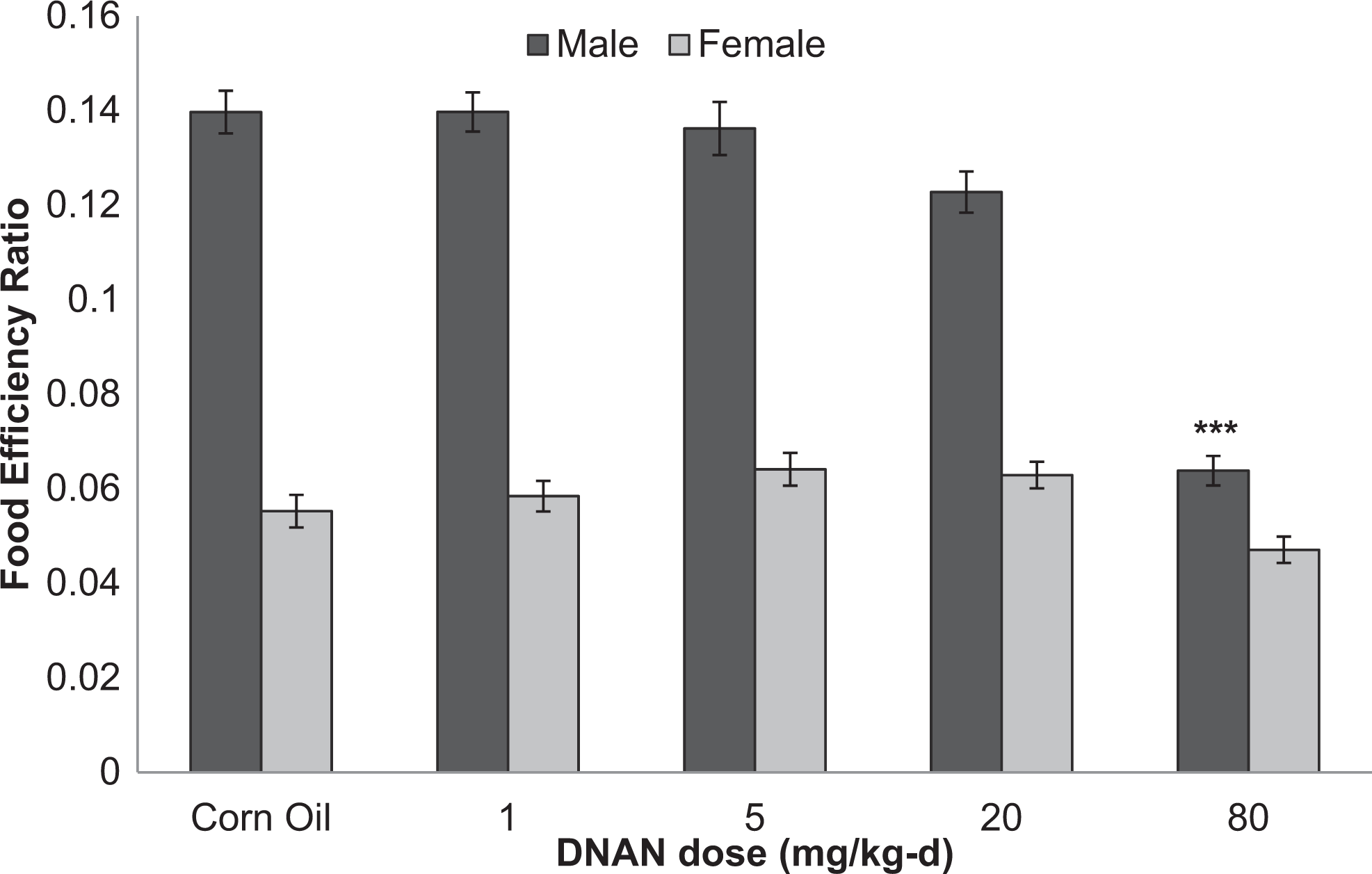
Food efficiency ratios for male and female Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. ***Significantly different from control at
Neurobehavioral Evaluations
In the neurobehavioral evaluations in the 90-day study, there were no differences between controls and DNAN groups in any of the home cage parameters: agitation, convulsions, tremors, posture, mutilation, and the area mutilated for males or females (data not shown).
There were no differences among treatment groups for lacrimation, salivation, piloerection, palpebral closures, exophthalmos, and pupillary status for male or female rats (data not shown). In male rats, fewer animals in the 80 mg/kg/d dose group than in the control group were classified as very easy to remove from the cage at weeks 1, 5, 7, and 8 (Figure 6). Reactivity to handling scores were higher in males in the 80 mg/kg/d group than the control group at weeks 2, 3, 6, 7, 9, and 10 (Figure 7). Ease of removal and reactivity to handling did not differ among treatment groups for female rats (data not shown).
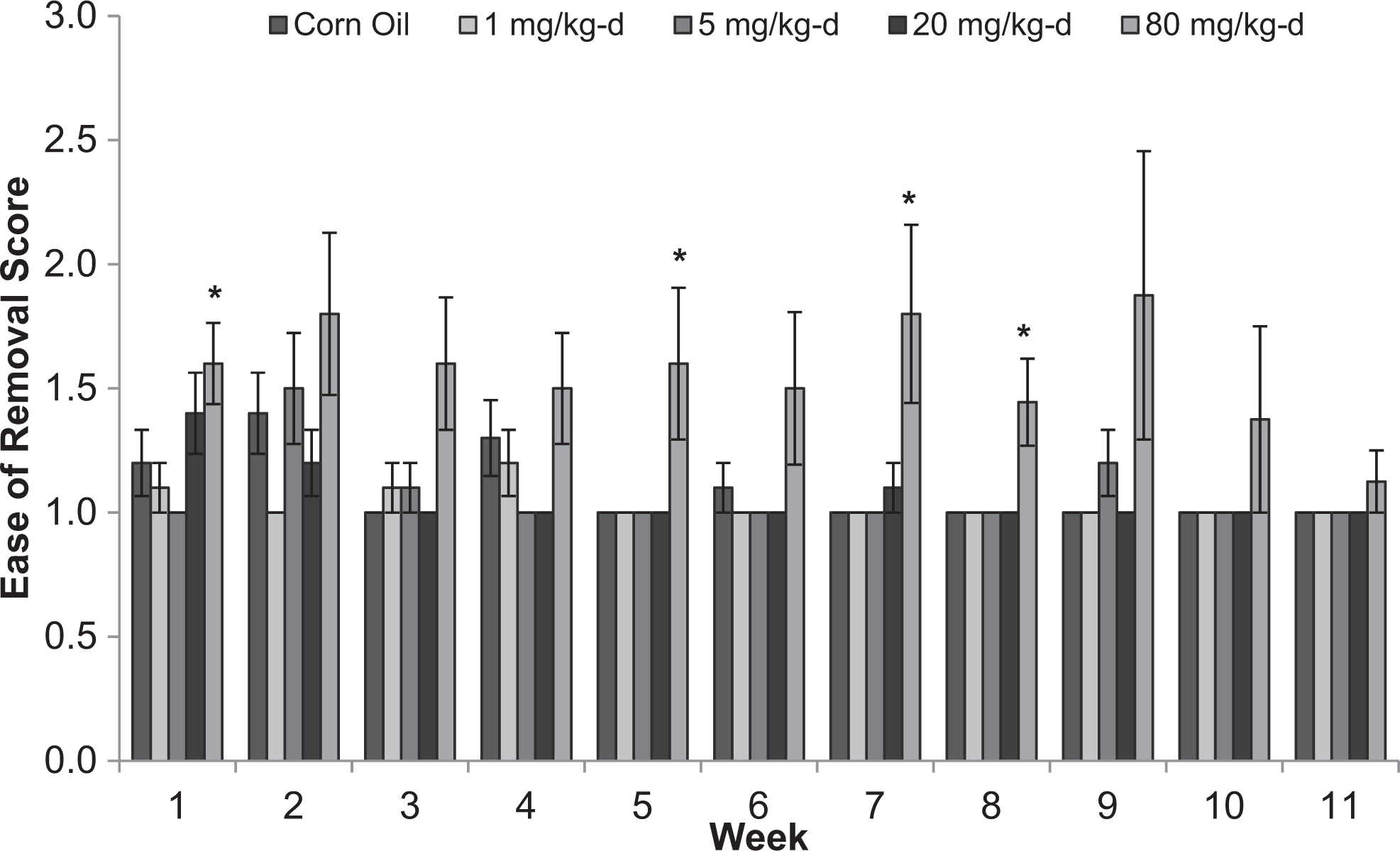
Ease of removal from cage scores for male Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. 1, very easy; 2, easy; 3, moderately difficult. *Significantly different from control at
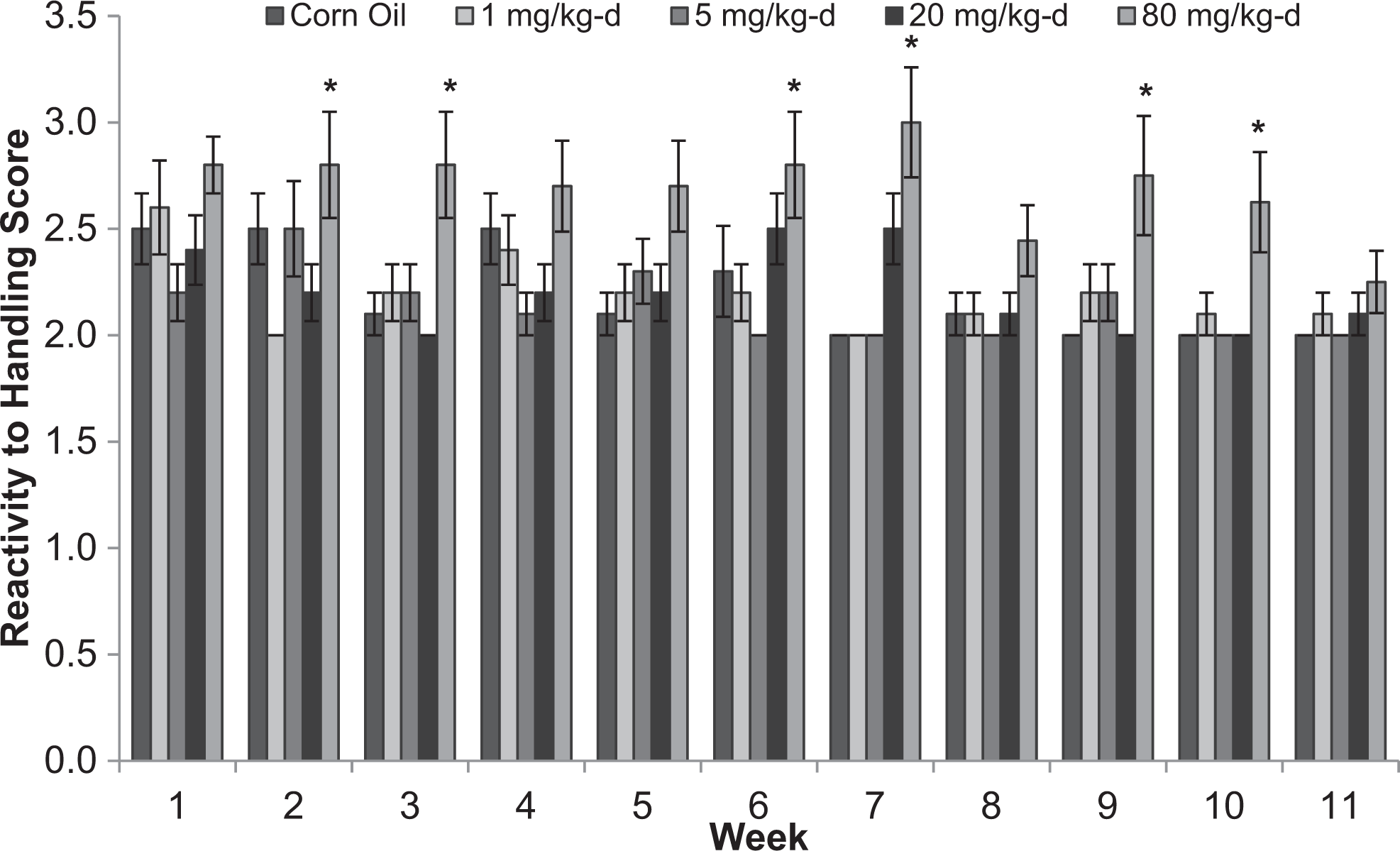
Reactivity to handling scores for male Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. 1, very low; 2, low; 3, moderately low; 4, moderately high. *Significantly different from control at
There were no differences among treatment groups in the grooms, arousal, fecal boli, fecal description, and urine parameters for male or female rats (data not shown). In males, the 80 mg/kg/d group had fewer normal gait observations than other dose groups at weeks 5 (
There were no differences between controls and DNAN groups in any of the sensory motor responses for male rats: approach, auditory startle response, tail pinch, pinna response, pupillary response, righting reflex, aerial righting, landing foot splay, forelimb grip strength, and hind limb grip strength (data not shown).
In females, there were no differences for auditory startle response, pinna response, pupillary response, righting reflex, aerial righting, landing foot splay, forelimb grip strength, and hind limb grip strength (data not shown). Tail pinch and approach differed among DNAN dose groups for females (
There were no differences among treatment groups in basic movement, immobility, and X and Y ambulation for male or female rats (data not shown). Mean number of nose pokes was lower in the 80 mg/kg/d group than the other dose groups for both males (
Ophthalmoscopic Examinations
No abnormalities were noted in terminal ophthalmoscopic examinations in the 90-day study.
Urinalysis
Urine color, volume, specific gravity, and protein concentration differed between DNAN treated and control groups in both males and females in the 90-day study (Table 1). Urine was darker in the 20 and 80 mg/kg/d dose groups than in control groups in males and females. In males, urine color was dark yellow in controls compared to gold and brown in the 20 and 80 mg/kg/d groups, respectively. Urine color in females was yellow in controls and dark yellow and gold/amber in the 20 and 80 mg/kg/d groups, respectively. Urine volume increased in a dose-dependent manner in females and was greater (3.2-fold) in the 80 mg/kg/d group than the control group. Urine volume was also greater (1.7-fold) in the 20 mg/kg/d group; however, this increase was not statistically significant. In males, urine volume was greater (2.0-fold) in the 20 mg/kg/d group than in the control. Urine volume was also increased (1.7-fold) in the 80 mg/kg/d group; however, this increase was not statistically significant. Specific gravity decreased in a dose-dependent manner in female rats and was reduced in the 80 mg/kg/d group relative to the control group. In males, specific gravity was lower in the 20 mg/kg/d dose group than in the control group. Protein concentration was lower in males in the 20 mg/kg/d group than in the control groups. Bilirubin concentration was increased (2.7-fold) in males in the 80 mg/kg/d group relative to the control group. Urine appearance, GLU, ketones, pH, urobilinogen, and leukocytes did not differ between DNAN treated and control groups.
Urinalysis Parameters in Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 90 Days.
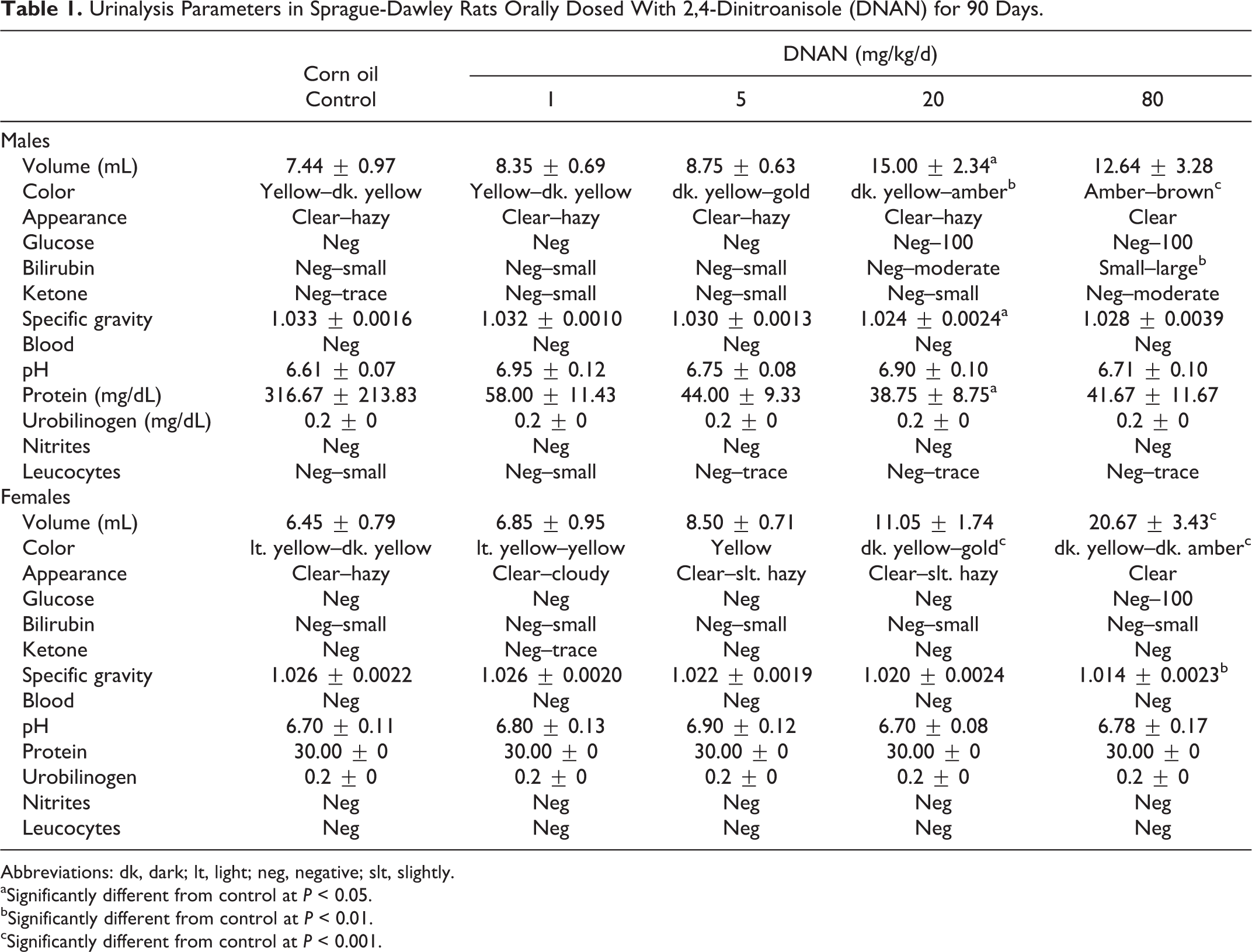
Abbreviations: dk, dark; lt, light; neg, negative; slt, slightly.
aSignificantly different from control at
bSignificantly different from control at
cSignificantly different from control at
Organ Mass and Ratios
Mean kidney, liver, and spleen absolute and/or relative organ masses differed between DNAN treated and control groups in both males and females. Mean kidney-to-body mass ratios were increased in both the 14- and 90-day studies. In the 14-day study, mean kidney-to-body mass ratios were increased in the 50 and 100 mg/kg/d groups, relative to the control, in males (Table 2) and females (Table 3). In the 90-day study, mean kidney-to-body mass ratios were increased, compared to controls, in males in the 20 and 80 mg/kg/d groups (18% and 35%, respectively; Table 4) and females in the 80 mg/kg/d group (19%; Table 5). Additionally, mean absolute kidney mass was increased in females in the 20 and 80 mg/kg/d groups (13% and 14%, respectively) in the 90-day study.
Absolute and Relative Organ Masses of Male Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 14 Days.
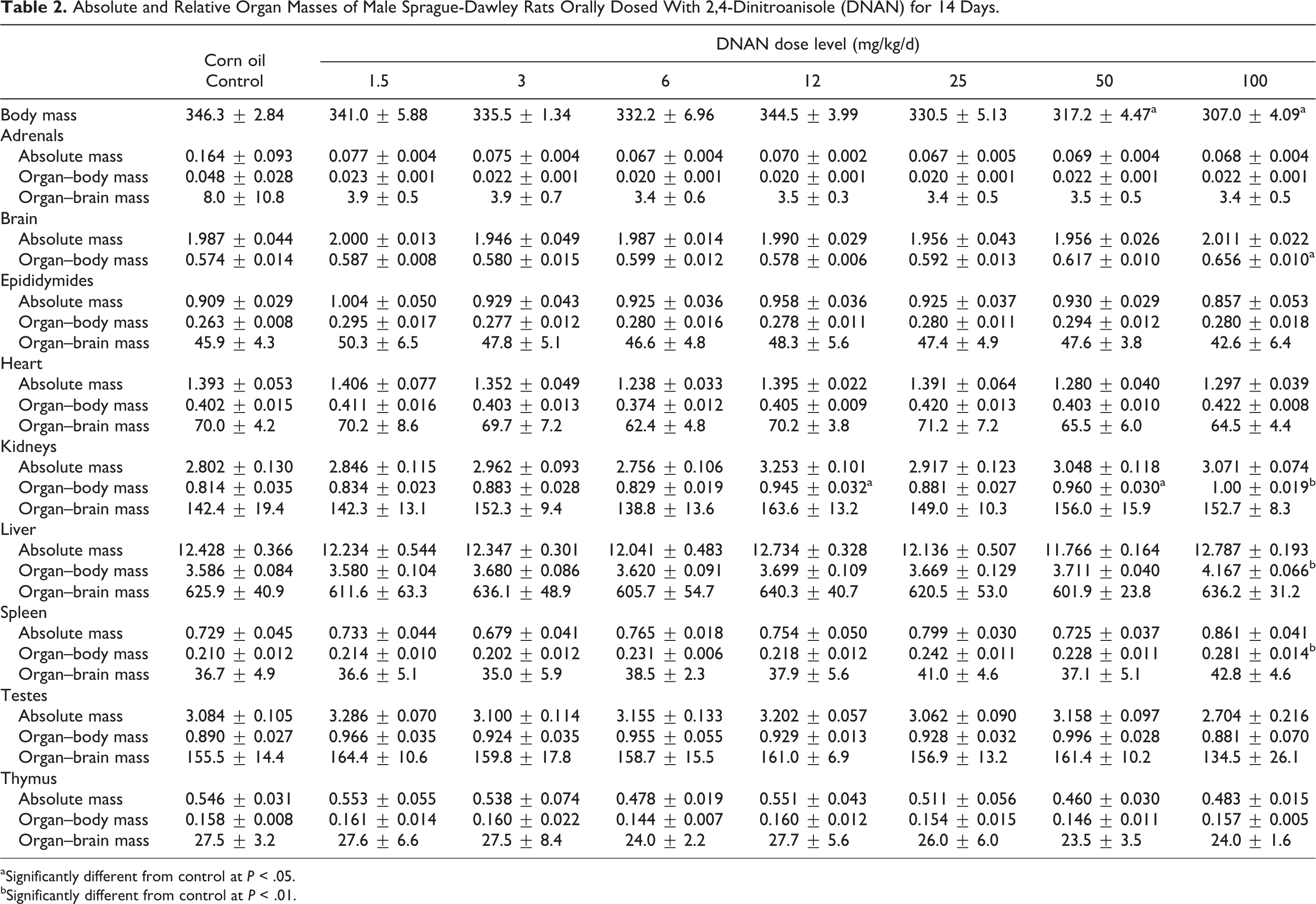
aSignificantly different from control at
bSignificantly different from control at
Absolute and Relative Organ Masses of Female Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 14 Days.
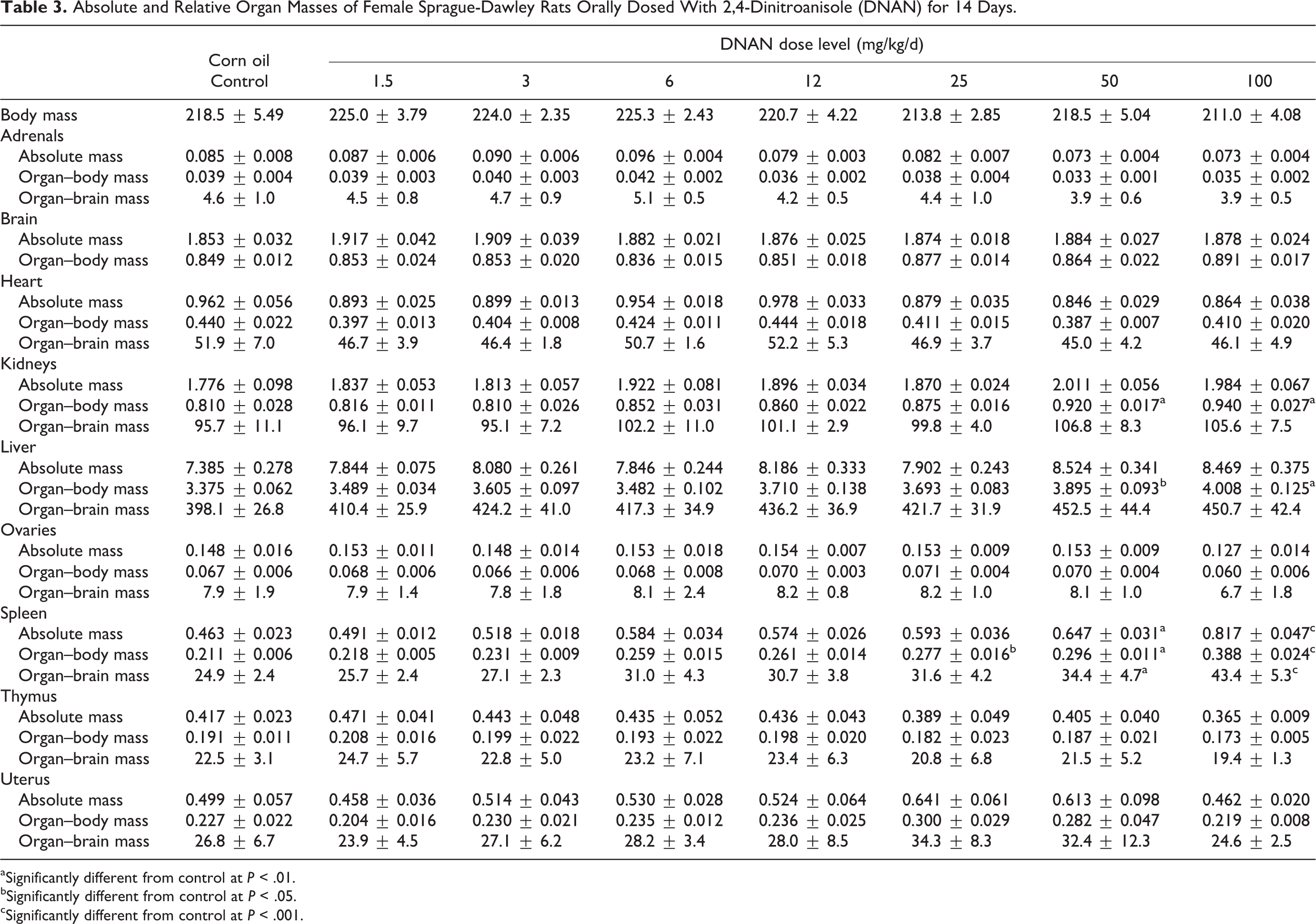
aSignificantly different from control at
bSignificantly different from control at
cSignificantly different from control at
Absolute and Relative Organ Masses of Male Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 90 Days.
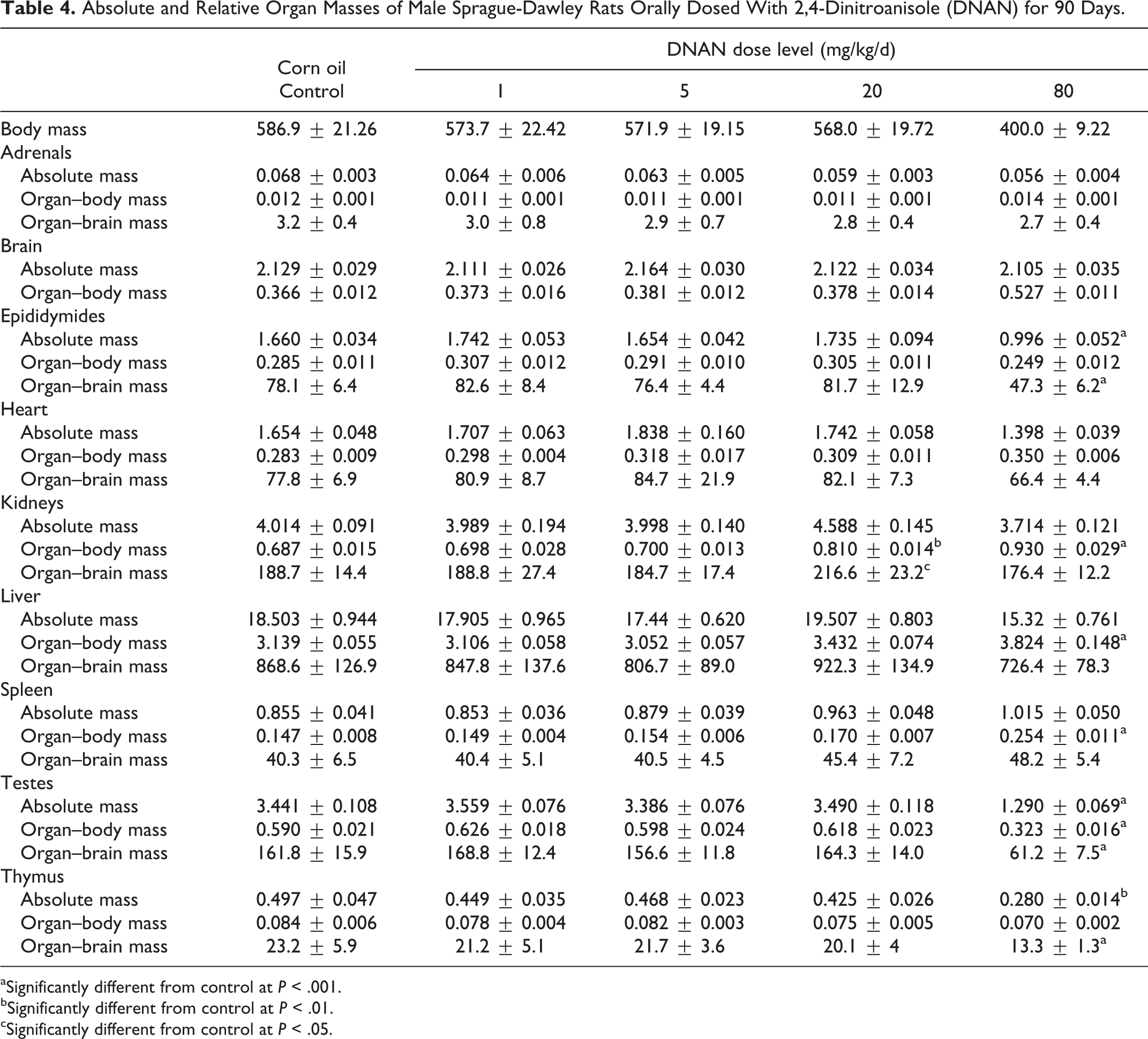
aSignificantly different from control at
bSignificantly different from control at
cSignificantly different from control at
Absolute and Relative Organ Masses of Female Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 90 Days.
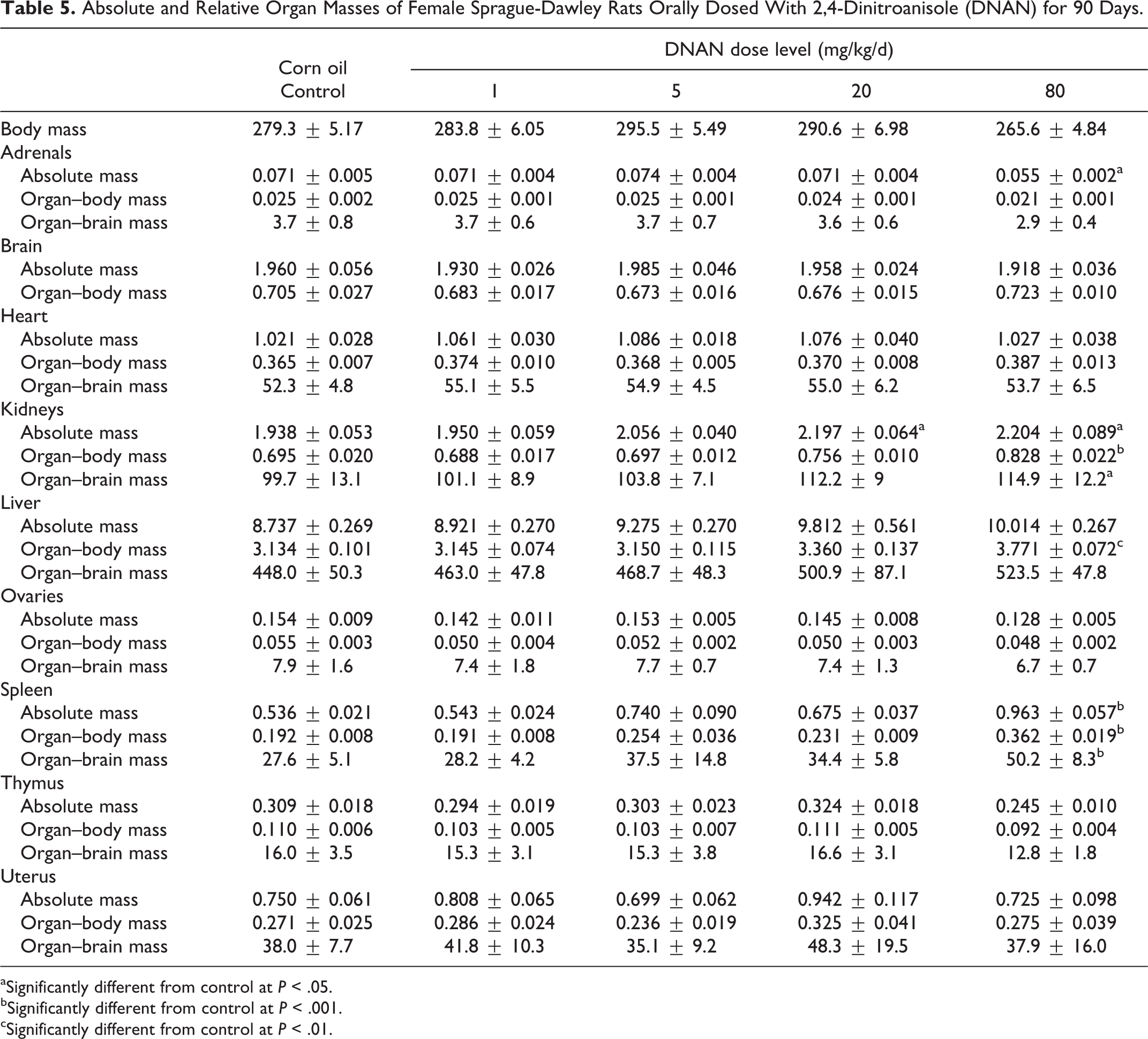
aSignificantly different from control at
bSignificantly different from control at
cSignificantly different from control at
Mean liver-to-body mass ratios were increased, relative to the control, in males in the 100 mg/kg/d dose group (11%) and females in the 50 and 100 mg/kg/d groups (15% and 19%, respectively) in the 14-day study (Tables 2 and 3). In the 90-day study, liver-to-body mass ratios were increased, relative to the control, in males (22%) and females (20%) in the 80 mg/kg/d dose group (Tables 4 and 5).
Mean spleen-to-body mass ratios were increased, relative to the control, in males in the 100 mg/kg/d dose group (33%) and females in the 25, 50, and 100 mg/kg/d groups (31%, 40%, and 84%, respectively) in the 14-day study (Tables 2 and 3). Female rats given 50 or 100 mg/kg/d DNAN also had increased mean absolute spleen mass (40% and 77%, respectively) and spleen-to-brain mass ratios (38% and 74%, respectively). In the 90-day study, spleen-to-body mass ratios were increased, relative to controls, in males and females given 80 mg/kg/d DNAN (73% and 88%, respectively; Tables 4 and 5). Absolute spleen mass (80%) and spleen-to-brain mass ratios (82%) were also increased, relative to controls, in females in the 80 mg/kg/d group in the 90-day study.
In males, mean testes mass, testes-to-body mass, and testes-to-brain mass ratios were reduced (63%, 45%, and 62%, respectively) in the 80 mg/kg/d group in the 90-day study (Table 4). Epididymides mass and epididymides-to-brain mass ratio were also reduced, relative to the control (40% and 39%, respectively), in males given 80 mg/kg/d DNAN in the 90-day study (Table 4).
Mean thymus mass and thymus-to-brain mass ratios were reduced (44% and 43%, respectively), relative to controls, in males in the 80 mg/kg/d group in the 90-day study (Table 4). Adrenal mass was reduced (22%), relative to controls, in females in the 80 mg/kg/d group in the 90-day study (Table 5).
Sperm Analysis
The mean number of sperm per gram in the cauda epididymis in male rats in the 80 mg/kg/d group was reduced by 96% compared to controls in the 90-day study (Figure 8). No motile sperm were found in any of the animals in the 80 mg/kg/d group. No significant reductions in sperm per gram, percent motile sperm, or percent progressively motile sperm were observed in the 1, 5, or 20 mg/kg/d dose groups.
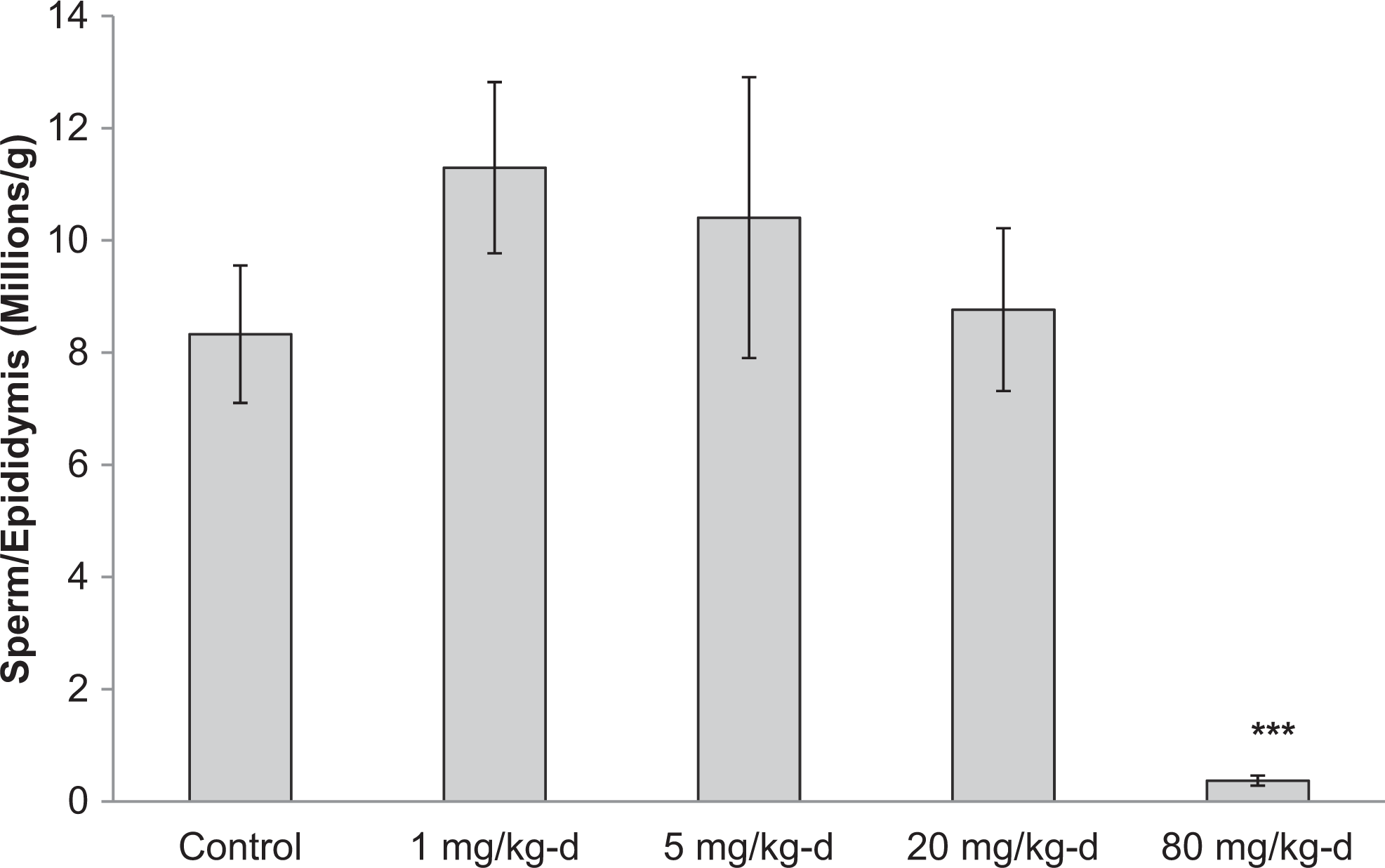
Caudal epididymal sperm counts for male Sprague-Dawley rats orally dosed with 2,4-dinitroanisole (DNAN) for 90 days. ***Significantly different from control at
Clinical Chemistry
Among the 17 clinical chemistry parameters examined in the 14- and 90-day studies, only CHOL, ALT, and BUN demonstrated dose-related differences from controls. The CHOL levels decreased in a dose-dependent manner in DNAN-treated males and were lower (1.4- and 1.5-fold, respectively) in the 20 and 80 mg/kg/d groups than in the control group (
The ALT levels were elevated (
Hematology
The WBC count increased in a dose-dependent manner in female rats in the 90-day study and was significantly higher (1.5- and 1.7-fold, respectively) in the 20 and 80 mg/kg/d groups relative to the control group (Table 6). This was due to an increase in monocytes in the 20 and 80 mg/kg/d groups (2.2- and 3.2-fold, respectively) relative to the control group. The WBC count and differentials did not differ between control and treated groups in the 14-day study (data not shown) or for males in the 90-day study.
Hematological Parameters of Male and Female Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 90 Days.
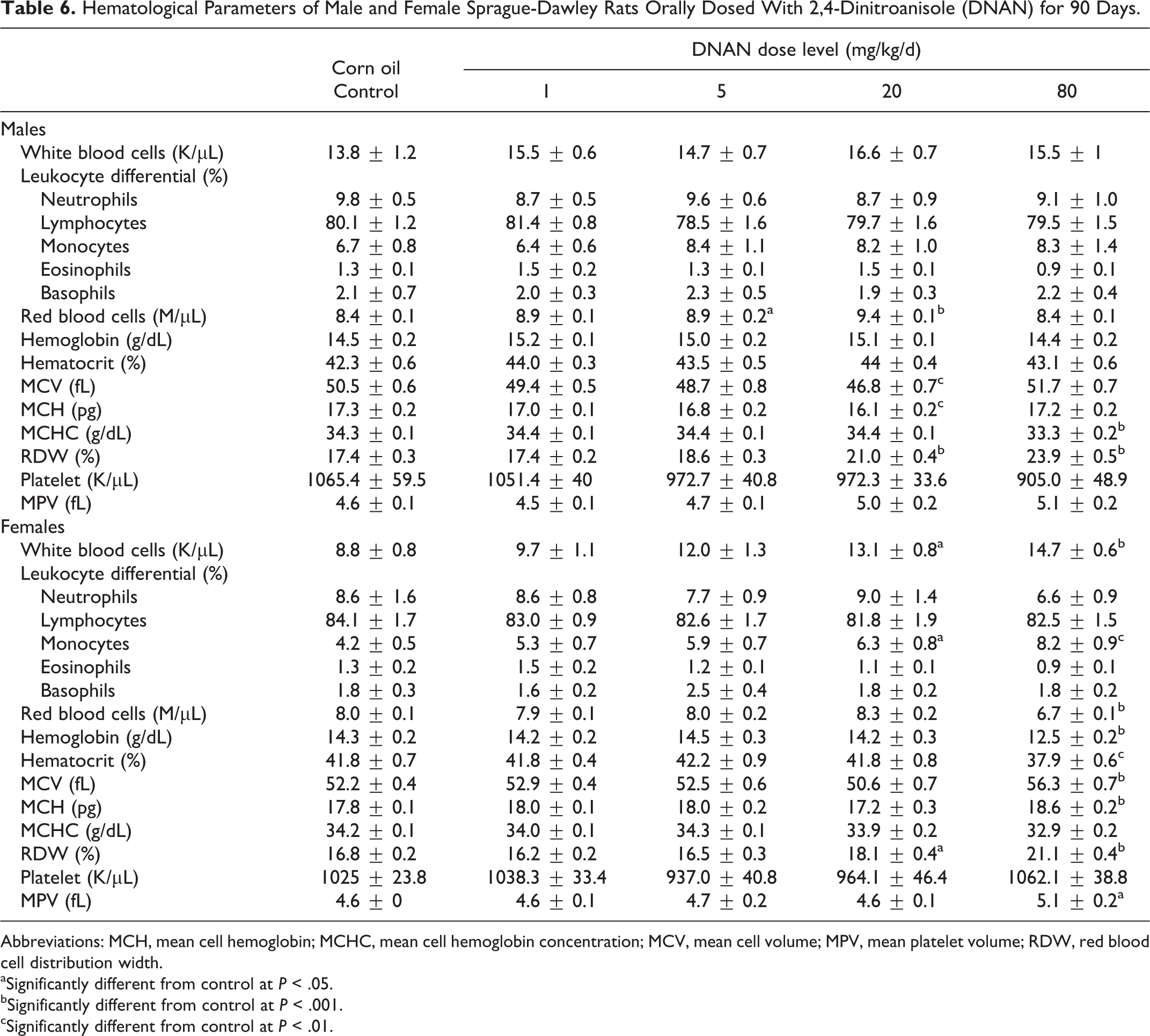
Abbreviations: MCH, mean cell hemoglobin; MCHC, mean cell hemoglobin concentration; MCV, mean cell volume; MPV, mean platelet volume; RDW, red blood cell distribution width.
aSignificantly different from control at
bSignificantly different from control at
cSignificantly different from control at
The RBC counts, HGB, and HCT were reduced (0.84-, 0.88-, and 0.91-fold, respectively) in females in the 90-day study in the 80 mg/kg/d group compared to controls (Table 6). The MCH did not differ among treated and control groups for female rats; however, MCHC was reduced (0.96-fold) in the 80 mg/kg/d group. The MCV was increased (1.1-fold) in females in the 80 mg/kg/d group. Mean RDW was increased in females in the 20 and 80 mg/kg/d dose groups (1.1- and 1.3-fold, respectively) in the 90-day study. Similarly, in the 14-day study, females in the 100 mg/kg/d group had reduced RBC (0.86-fold), HGB (0.86-fold), HCT (0.90-fold), and MCHC (0.95-fold) compared to controls (data not shown).
In male rats in the 90-day study, mean RBC counts were increased (1.1-fold) in the 5 and 20 mg/kg/d groups relative to controls (Table 6), whereas HCT and HGB did not differ among treated and control groups. The MCH was reduced (0.93-fold) in males in the 20 mg/kg/d group, and MCHC was reduced (0.97-fold) in the 80 mg/kg/d group relative to the control. The MCV was reduced (0.93-fold) in the 20 mg/kg/d group. The RDW was increased in the 20 and 80 mg/kg/d groups (1.2- and 1.4-fold, respectively) in the 90-day study. Hematology parameters were not affected by DNAN treatment for males in the 14-day study (data not shown).
The PLT count did not differ between treated and control groups for either male or female rats in either the 14- or 90-day studies. The MPV was slightly increased (1.1-fold) in the 80 mg/kg/d groups in the 90-day study; however, this increase was only significant in females (Table 6).
Histopathology
Mortality occurred in 3 males and 1 female dosed at 80 mg/kg/d in the 90-day study. Selected tissues were collected from these animals; no cause of death was identified based on microscopic examination. Test article-related microscopic findings were noted in the testes, epididymides, spleen, liver, and brain of animals sacrificed at study termination in both the 14- and 90-day studies (Tables 7 and 8).
Histopathological Findings of Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 14 Days.
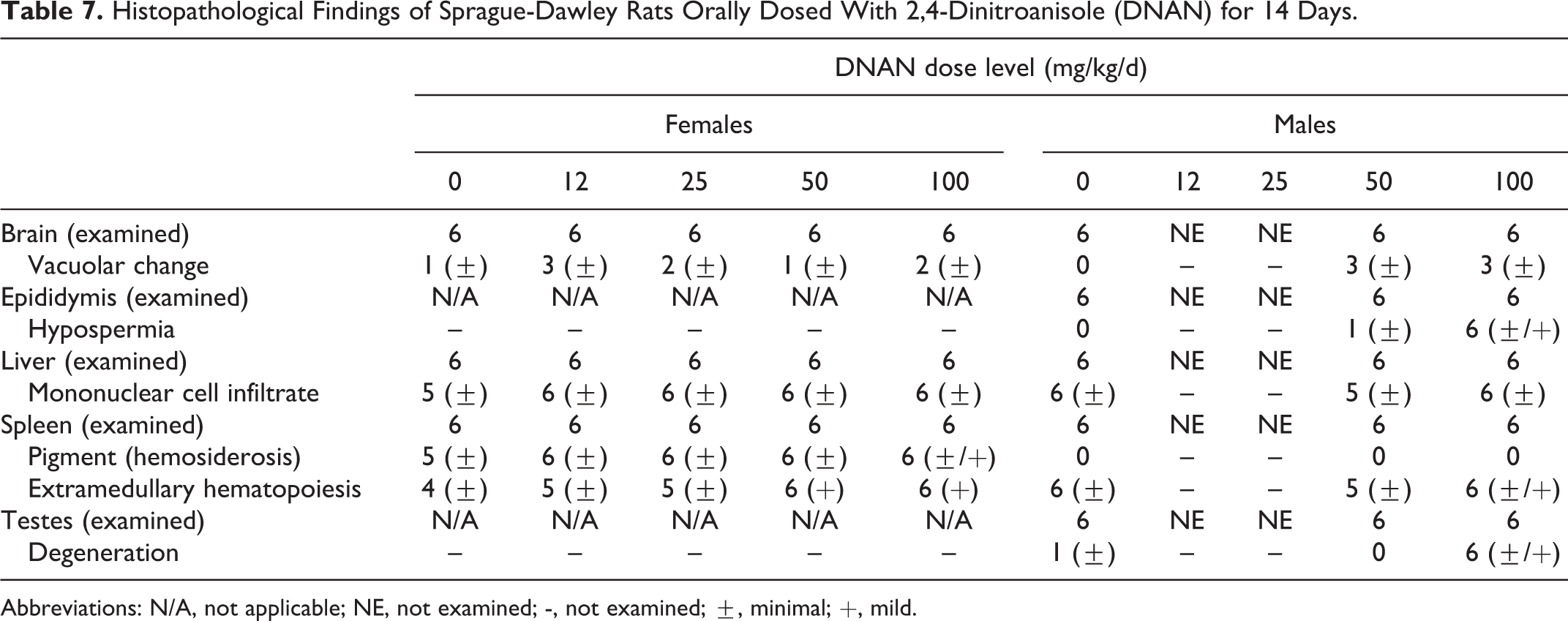
Abbreviations: N/A, not applicable; NE, not examined; -, not examined; ±, minimal; +, mild.
Histopathological Findings of Sprague-Dawley Rats Orally Dosed With 2,4-Dinitroanisole (DNAN) for 90 Days.
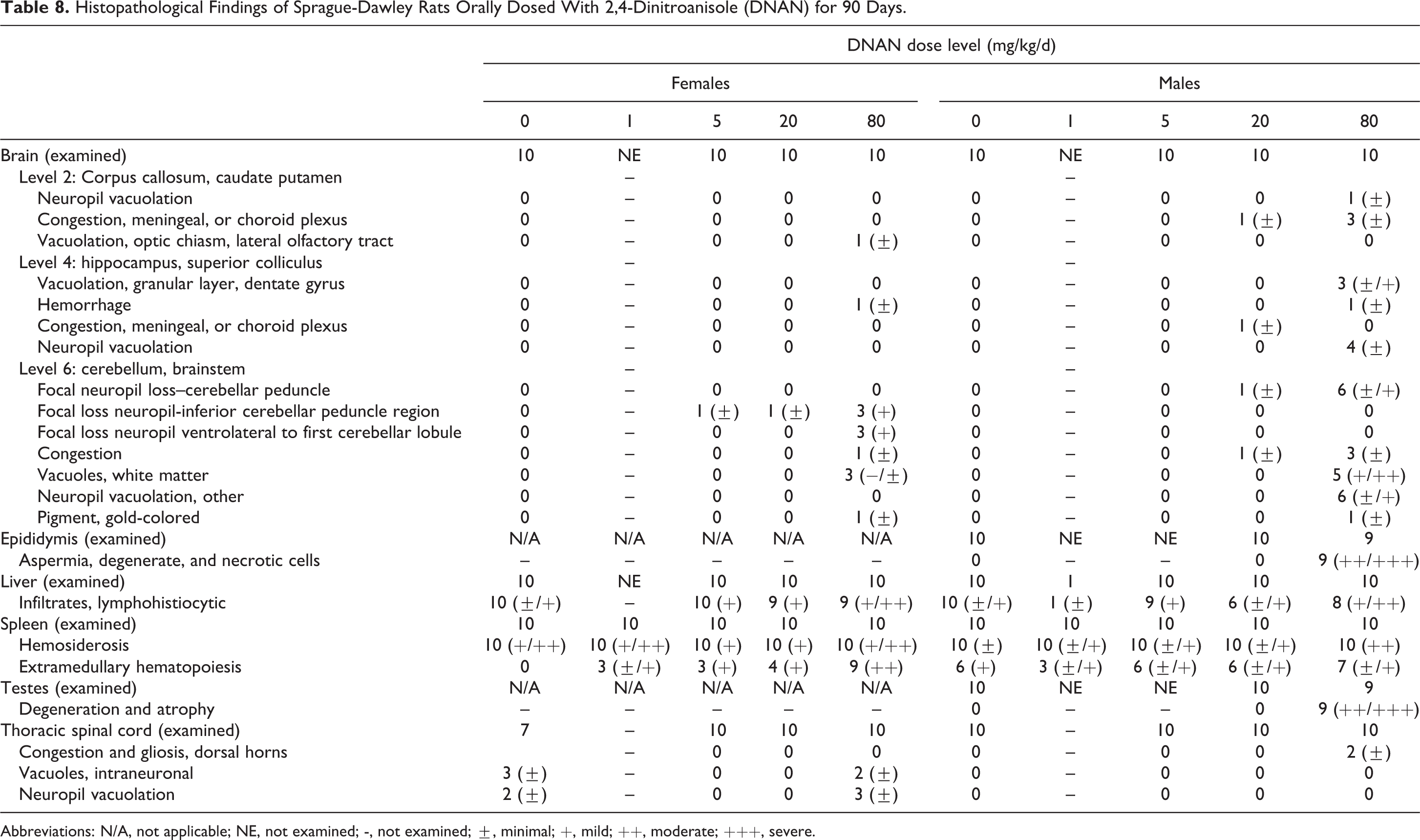
Abbreviations: N/A, not applicable; NE, not examined; -, not examined; ±, minimal; +, mild; ++, moderate; +++, severe.
Degeneration and atrophy of testicular seminiferous tubules (moderate to severe) was present in all males examined in the 80 mg/kg/d group in the 90-day study. No test article-related changes were noted in the testes in the control and 20 mg/kg/d groups in the 90-day study. Seminiferous tubules of the 80 mg/kg/d group retained only Sertoli cells, spermatogonia, and early spermatocytes. Absent germ cell layers included all spermatid and late spermatocyte stages resulting in the absence of mature sperm in seminiferous tubules. Testes additionally demonstrated moderate to numerous numbers of atrophic tubules. There was also evidence of seminiferous tubule degeneration in the 14-day study at 50 and 100 mg/kg/d groups. Degeneration and loss of seminiferous epithelium, with large numbers of multinucleated giant cells, were evident in many tubules in the 100 mg/kg/d group. At 50 mg/kg/d, these changes were more subtle and minimal, with only an increase in multinucleated giant cells easily discerned in some rats.
In the epididymides, hypospermia/aspermia with eosinophilic cellular tubular debris (moderate to severe) was present in all males examined in the 80 mg/kg/d group in the 90-day study. Hypospermia was also noted in the epididymides of all males examined in the 100 mg/kg/d group in the 14-day study. This change was a reflection of the testicular effect, not evidence of an additional toxic effect on the epithelium or smooth muscle of the epididymis proper.
In the spleen, increased extramedullary hematopoiesis (EMH), principally of red cell precursors, was noted in many rats. This is a common finding in laboratory rats and, by itself, is not an evidence of toxic effect. However, in this study, hematopoiesis was increased in severity in males in the 14-day study at 100 mg/kg/d and females at 50 and 100 mg/kg/d in the 14-day study and at 80 mg/kg/d in the 90-day study, when compared to their respective controls. Hemosiderin, excess iron deposited in the spleen in normal rats due to the breakdown of erythrocytes, was present in all rats on study. An increase in hemosiderin deposits can result from hemolytic crisis or hematotoxic insult. A dose-related increase in the severity of hemosiderosis was apparent in females in the 14-day study at 100 mg/kg/d and in males in the 90-day study at 80 mg/kg/d groups. The EMH and hemosiderosis are consistent with effects on RBCs such that iron pigments from increased red cell turnover are trapped within the spleen (hemosiderin), and red cell production is increased to maintain erythrocytic homeostasis. These anatomic changes are consistent with clinical chemistry data indicative of increased RBC turnover or anemia.
Male rats in the 80 mg/kg/d group in the 90-day study exhibited lesions in cerebellar peduncle, consisting of focal loss or attenuation of neuropil, increase in glial cell numbers, occasional hemorrhage, pigment, and variably degenerate neurons (Figure 9). Focal loss of neuropil also occurred in nuclei between cranial nerves V, VII, and VIII in the ventral brainstem in males in the 80 mg/kg/d group. In the white matter tracks of cranial nerves V or VIII and VII, vacuolation and dilation of myelin sheaths were observed in male rats in the 80 mg/kg/d group. Female rats in the 80 mg/kg/d group in the 90-day study exhibited similar but less frequent lesions in the same locations; however, the frequency was not statistically different from controls. The only other central nervous system findings consisted of minimal cerebral congestion in both genders of treated rats. Neuronal vacuolation and dorsal horn neuropil vacuolation were noted in the thoracic spinal cord of females in the 80 mg/kg/d group (Figure 10). No dose-related peripheral nervous system lesions were observed. The incidence of lesions in the 20 mg/kg/d group in the 90-day study did not differ from control. In the 14-day study, subtle increased perineuronal spaces, often associated with shrunken Purkinje cells, were noted in half of the males at both 50 and 100 mg/kg/d, and in 1 to 3 females in all dose groups. The morphologic appearance noted as vacuolar change was consistent with the presence of a slight degree of cerebellar edema, which would be an evidence of per acute neurotoxicity.
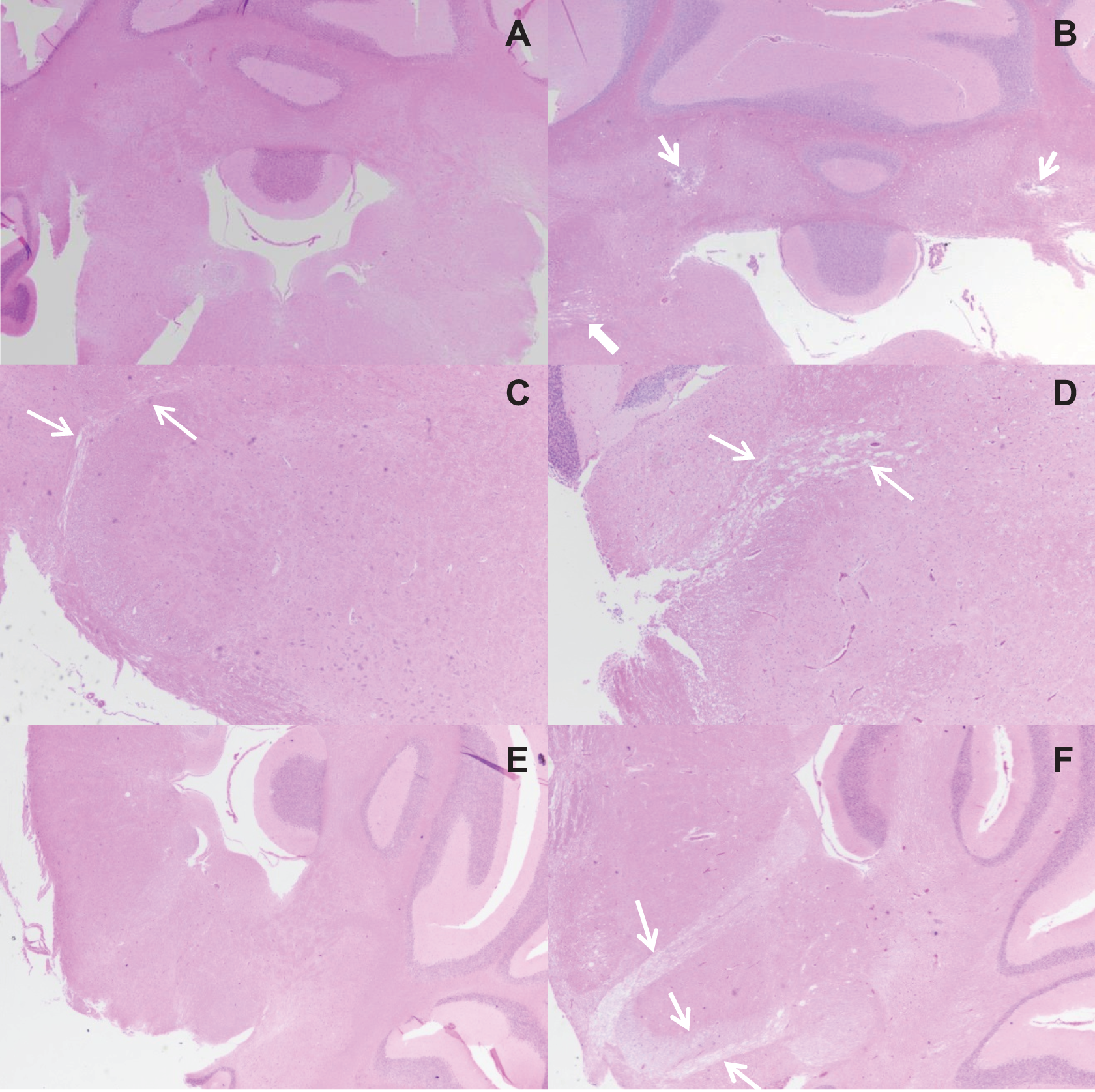
Photomicrographs of brain sections from male Sprague-Dawley rats orally dosed with corn oil (A, C, and E) or 80 mg/kg/d 2,4-dinitroanisole (DNAN) (B, D, and F) for 90-days. Lesions in male rats were characterized by bilateral neuropil loss with vacuolation of the cerebellar peduncle (B, thin arrows), vacuolation of cranial nerve VIII (cerebellar; B, thick arrow, and D—compare to same area between arrows in C), and vacuolation of cranial nerve VII, spinal trigeminal tract (Sp5), and cranial nerve VIII (F, arrows). Hematoxylin and eosin, magnification 2×.
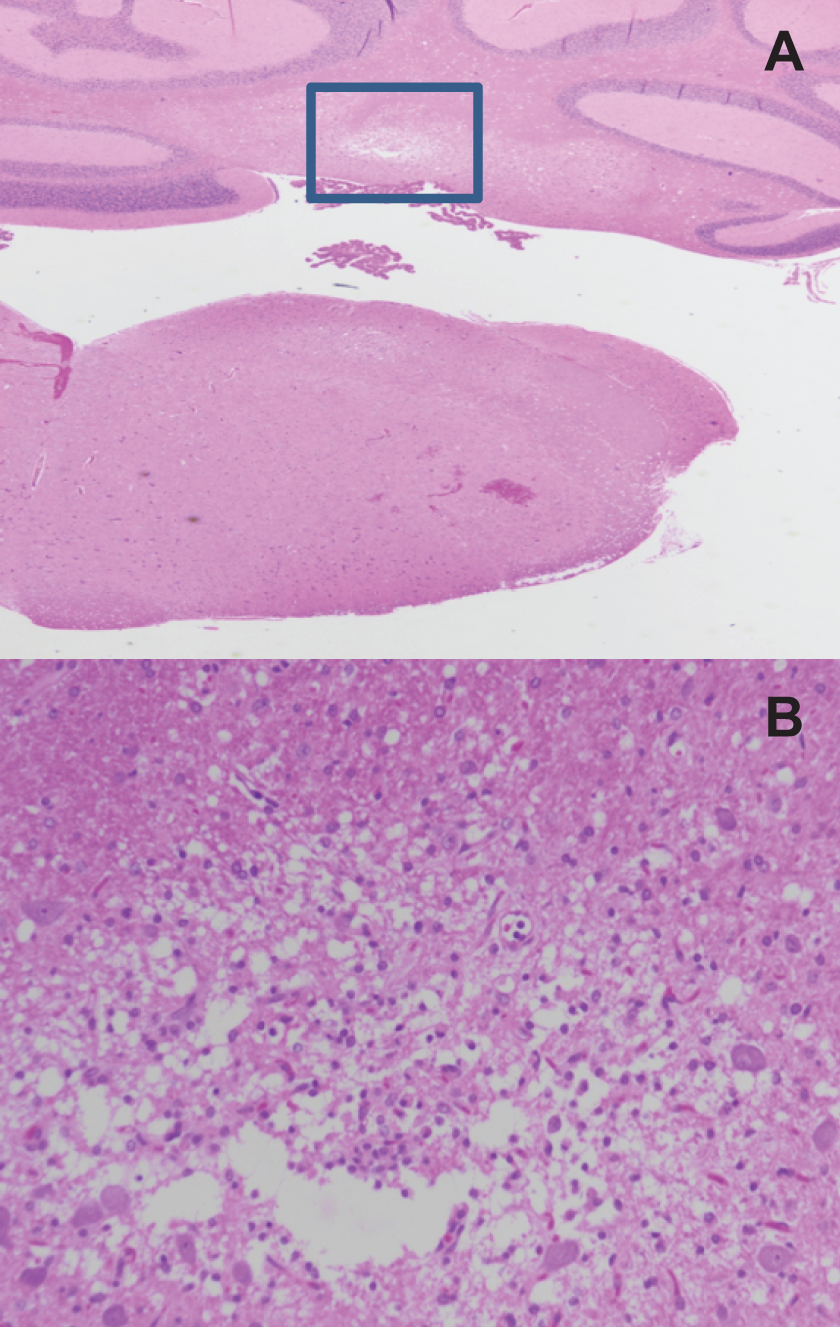
Photomicrographs of thoracic spinal cord sections from female Sprague-Dawley rats orally dosed with 80 mg/kg/d 2,4-dinitroanisole (DNAN) for 90 days. Lesions in female rats were characterized by neuronal cell loss with vacuolation of gray matter and increased glial cells (hematoxylin and eosin; A, 2× and B, 20×).
In the liver, lymphohistiocytic infiltrates were observed in both treated and control groups. Although these aggregates are often considered background lesions, the frequency may be increased by treatment. A slight increase in severity with increasing dose was noted in females in the 90-day study, which may have been treatment related.
Additional lesions noted in the 90-day study but determined to be background or incidental due to low frequency of occurrence or comparable occurrence in control and treated groups included: prostatic, epididymal and coagulating gland lymphocytic infiltrates, harderian gland lymphocytic infiltrates, rare lymphoid hyperplasia of submandibular or mesenteric lymph nodes, adrenal gland vacuolation, plasmacytosis of the submandibular lymph node, pancreatic acinar atrophy or degeneration, myocardial necrosis, cardiac lymphocytic infiltrates, basophilic tubules, pelvic dilatation (hydronephrosis), lymphocytic interstitial infiltrates of the kidney, focal hepatic biliary hyperplasia, and ultimobranchial cysts of the thyroid. Renal mineralization at the corticomedullary junction was noted in females in the 90-day study but did not demonstrate a dose response.
Discussion
Both subacute and subchronic exposure to DNAN resulted in reduced body mass in male rats in the highest dose groups (50 and 100 mg/kg/d and 80 mg/kg/d, respectively) but did not affect body mass in female rats. The reduced body mass was not attributable to a reduction in food intake as food intake did not differ among treatment groups for male rats. The absence of an effect on body mass in female rats may, however, have been due to the increase in food intake observed in female rats. Feed conversion efficiency was reduced in male, but not female rats, in the highest dose groups. These data suggest that the effects of DNAN on body mass may have been due to impacts on metabolism. The DNAN is metabolized to 2,4-dinitrophenol (2,4-DNP), 3 a compound which increases basal metabolic rate by uncoupling oxidative phosphorylation. 11 Thus, the effects of DNAN on metabolism may be attributable to 2,4-DNP. The disparity in effects of DNAN on body mass between males and females suggests a possible difference in the conversion of DNAN to DNP between the sexes. Preliminary data indicate that 4 to 8 hours after oral dosing with DNAN, blood levels of DNAN are higher in females (2.2-3.4 times) than in males, whereas 2,4-DNP levels are slightly lower (0.83 times) in females than in males. 5
Gender differences in the conversion of DNAN to DNP may also contribute to the differences in effects observed in organ systems. Female rats exhibited enlargement of the spleen, splenic hemosiderosis, and EMH associated with changes in hematology indicative of anemia, including decreased RBC count, HCT, HGB, and mean corpuscular HGB concentration and increased RDW and MCV. Although gross and microscopic changes of the spleen were observed in male rats, the prevalence and severity were lower than that seen in females. Overt anemia was not apparent in male rats; however, a compensated anemia may have been present. As previous studies with rats and dogs demonstrated no hematological abnormalities following subchronic exposure to DNP, 12 the hematological effects observed in the females likely resulted from exposure to the parent compound, DNAN. Again, the differences between the sexes may be attributable to a higher blood DNAN levels in female rats. 5
In contrast, the higher incidence and greater severity of neurological effects in male rats may have resulted from higher DNP exposure. Although both male and female rats developed gait abnormalities and exhibited signs of neurotoxicity in the neurobehavioral assessment, male rats additionally exhibited consistent ventral or lateral recumbency subsequent to DNAN administration and a higher incidence of brain lesions. Ventral or lateral recumbency was noted as a consistent indicator of development of brain lesions in rats exposed to 3-nitroproprionic acid (3-NPA). 13 Gait irregularities similar to those noted in the current study, including walking on toes, hunched back, and partial disuse of rear legs, have been noted in association with gliovascular lesions in the brain stem of rats exposed to 1,3,5-trinitrobenzene (TNB), 1,3-dinitrobenzene (DNB), and nitrobenzene (NB). 14 Like DNP, 3-NPA and 1,3-DNB disrupt energy metabolism through inhibition of succinate dehydrogenase (SDH) activity. 13,15 Although both appear to be mediated by SDH inhibition, lesions produced by NPA are characteristically striatal lesions, whereas NBs selectively damage the cerebellum. 13,15
Recent work has demonstrated that the onset of neuron-depleted striatal lesions typically associated with both acute and chronic administration of 3-NPA is preceded by reactive astrocytosis and mild behavioral/gait abnormalities. 16 Neuronal death that occurs only after rescue from the disease pathway is no longer possible. The focal loss of neuropil of the cerebellar peduncle associated with DNAN treatment may similarly represent early stages in progressive neurodegenerative pathology or may be a regenerative response to toxic insult. 17 We hypothesize that the absence of neuronal death may be attributable to low levels of DNP reaching the brain following metabolism of DNAN. As has been hypothesized with 3-NPA, this may lead to less severe metabolic impairment and the build-up of free radicals, nitric oxide, and lactic acid which can impair cell functioning. 18 Progressive NPA-induced cell death reportedly is a slow process, taking up to 7 days for completion. 19 Behavioral changes were, however, noted after 3 to 4 days and overt lesions after 5 days in rats given high doses of 3-NPA for 5 days. 20 In rats given low doses of 3-NPA for 2 days, behavioral alterations were not apparent until days 5 to 7, and overt lesions did not manifest. 18
Delayed onset of lesions has similarly been noted following administration of NBs. 14,21,22 In 1,3,5-TNB-exposed rats, 10 days of exposure to 71 mg/kg/d was required to produce brain lesions; rats exposed to 35.5 mg/kg/d for 4 or 10 days and 71 mg/kg/d for 4 days did not exhibit brain lesions. 21 Studies in which 1,3-DNB was given as a single bolus or infused over time demonstrated that a high concentration alone does not induce neurotoxicity; a concentration–time threshold exists. 22 Chandra et al21 reasoned that the delayed onset of neurotoxicity was attributable to a need for massive doses to accumulate in the brain before damage would ensue. Although this may well be true, the delay may also be the result of progressive neurodegenerative processes that have been noted in the case of 3-NPA to take 7 days to result in lesions. 19
A delay in onset of neurotoxicity of DNAN or its metabolite(s) was apparent in the onset of gait abnormalities observed during clinical observations as well as during the neurotoxicity evaluation. Significant gait differences were detected in the FOB in males at week 5 and females at week 9. The appearance of gait abnormalities during clinical observations was variable, with some rats making apparent recovery from 1 day to the next; however, the mean first day of appearance in the 80 mg/kg/d groups was day 32 ± 13 in males and day 26 ± 15 in females. This delay in onset may suggest the presence of concentration–time threshold for some aspects of DNAN-induced neurotoxicity as well. Ventral and lateral recumbency, however, a reliable indicator of 3-NPA-induced brain lesions, were present in some rats within the first day of dosing, suggesting either lesions were also present or recumbency is not associated with DNAN-induced brain lesions. The earliest evidence of DNAN-induced brain lesions was in the female euthanized at day 19. That some indications of neurotoxicity were apparent as early as the first day of dosing (recumbency), whereas the onset of others (gait irregularities) was considerably delayed, suggests a progressive neuropathology, or perhaps separate DNAN- and DNP-induced neurotoxicities.
Although DNP is neurotoxic at high doses, low doses have been shown to be neuroprotective, maintaining mitochondrial function and reducing oxidative neuronal damage induced by excitotoxic pathways. 11 Perhaps the concentrations of DNP present in the brains were such that a neuroprotective effect was established. This might explain the slow onset of gait abnormalities, as well as why brain lesions were limited to neuropil attenuation and did not progress to neuronal death. Although gait irregularities were reported in a subacute inhalation study, 7 this work represents the first documentation of DNAN-induced neurotoxicity associated with brain lesions. Much can be gained by comparison with other compounds that disrupt mitochondrial energy homeostasis; however, that DNAN-induced neurotoxicity is induced by this mechanism remains speculation at this time.
The mechanism by which DNAN-induced testicular toxicity is also not apparent from the current study. That it is a testicular toxicant is clear; however, this toxicity occurred after repetitive long-term dosing and, in the 90-day study, at exposures that also result in mortality. Males in the 80 mg/kg/d group had reduced testes mass, degeneration and atrophy of testicular seminiferous tubules, severe oligospermia with eosinophilic cellular tubular debris of the epididymis, and no detectable sperm in the cauda epididymal sperm analysis. Degeneration of seminiferous epithelium was also evident in the 50 and 100 mg/kg/d groups in the 14-day study. Because germ cells are dependent on the function and processes of other cell types within the testis, disruption of the germ cell supporting environment often results in their death. 23 The repetitive and prolonged dosing used in this study often results in progressive germ cell loss, regardless of the mechanism of toxicity. The end result often being seminiferous tubules lined only by Sertoli cells, which, though sensitive to alterations in function, are extremely resistant to cell death. 23 Testicular toxicity of this nature has been reported for the structurally similar nitroaromatic compounds TNB 24 –26 and TNT. 27,28 The TNT was determined to be a direct testicular toxicant, inducing damage to spermatozoa through oxidative DNA damage mediated by its metabolite. 29 The authors of the article further suggest that oxidative DNA damage likely plays a role in testicular toxicity for other nitroaromatic compounds. The role of this mechanism in DNAN-induced testicular toxicity is, however, unclear.
The DNAN-induced testicular toxicity is likely attributable to the parent compound rather than its metabolite, DNP. The 2,4-DNP was toxic to Sertoli-germ cell cocultures; however, this occurred only at 10−4 M concentrations (1.98 × 107 µg/mL) 30 that are far in excess of the 50 µg/mL 2,4-DNP blood levels observed in males orally dosed with 114.5 mg/kg/d DNAN. 5 Additionally, 2,4-DNP has shown no testicular toxicity in laboratory animals 31 and was negative in the rodent Hershberger bioassay, indicating that it is not an antiandrogenic compound. 32 A slight increase in the incidence of tailless sperm was noted after 14 days of administration of 30 mg/kg/d 2,4-DNP, suggesting a possible spermatotoxic effect; however, the effect was only observed at a near lethal dose. 33 At the same dose (30 mg/kg/d), 2,4-DNP demonstrated reproductive and developmental toxicity, reducing the number of live births, live birth index, and body weight of pups. 34 Menstrual irregularities have been reported in humans taking 2,4-DNP as a diet aid, indicating possible endocrine activity. 12 Embryo toxicity (45 mg/kg/d) and teratogenicity (15 and 45 mg/kg/d) were also observed following oral administration of DNAN during gestation (GD5-19). 29 Given the testicular and embryo toxicity of DNAN and in light of the reproductive and developmental effects of 2,4-DNP, further investigation of the reproductive, developmental, and endocrine disrupting effects of DNAN is warranted.
Possible nephrotoxicity occurred in both males and females. Kidney mass was increased in both the 14- and 90-day studies. In the 90-day study, the increase in kidney mass was accompanied by changes in several urinalysis parameters (ie, volume, specific gravity, and protein concentration) in the 20 and 80 mg/kg/d groups, suggesting an impaired concentrating ability of the kidneys. In the 14-day study, BUN was increased in the 100 mg/kg/d females, again suggesting impaired kidney function. These changes were not, however, accompanied by altered histopathology of the kidney.
Summary and Conclusions
This study, the first repetitive oral dosing study conducted with DNAN, demonstrated a steep dose-response curve, with most effects occurring only in the highest doses and occurring at or near lethal doses. Mortality occurred in 3 males and 1 female in the 80 mg/kg/d dose group. Although the cause of death was not evident based on microscopic examination of tissues from these animals, mortality may have resulted from the conversion to 2,4-DNP, an inhibitor of mitochondrial energy homeostasis via uncoupling of oxidative phosphorylation. In humans, deaths due to poisoning with 2,4-DNP have been attributed to hyperthermia and cardiac arrest. 35,36 Body temperatures were not monitored in this study; however, DNAN treatment resulted in an apparent increase in metabolic rate, leading to reduced feed conversion efficiency and ultimately reduced body mass gain in males. Other toxic effects of DNAN (ie, splenomegaly, EMH, anemia, seminiferous tubular degeneration, neurotoxicity, and possible nephrotoxicity) are generally similar to those of nitroaromatic compounds including TNT 27,28 and TNB. 26,37 Although most toxic effects occurred in the highest dose groups, indicators of anemia and changes in urine parameters occurred in lower dose groups. The EMH in female rats was selected as the critical effect, and a benchmark dose lower confidence limit for a 10% response (BMDL10) of 0.93 mg/kg/d and a workplace environmental exposure level of 0.1 mg/m3 (0.01 mg/kg/d; 8-hour time weighted average) were determined for DNAN. 38 In comparison, a critical effect Lowest-Observed-Adverse-Effect Level (LOAEL) of 0.5 mg/kg/d and an oral reference dose of 0.0005 mg/kg/d were determined for TNT. 39 Based on the available data, DNAN appears to be less toxic than TNT; however, the chronic and carcinogenicity studies that are the basis for the safe exposure levels for TNT have not been conducted for DNAN.
Footnotes
Authors’ Note
Lent, E. contributed to conception and design, contributed to acquisition, analysis and interpretation, drafted manuscript, and critically revised manuscript. Crouse, L. contributed to conception and design, contributed to acquisition, and critically revised manuscript. Wallace, S. contributed to acquisition, analysis and interpretation, drafted manuscript, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy. The views expressed in this article are the views of the author(s) and do not reflect the official policy of the Department of the Army, the Department of Defense, or the US government.
Acknowledgments
The authors gratefully thank the efforts of Theresa Hanna in performing the neurobehavioral tests, LTC Erica Carroll for evaluating the subchronic study brain histopathology, Allen Singer (Battelle, Columbus, Ohio) for evaluating the subacute study histopathology, Matthew Bazar and Mark Way for conducting clinical chemistry analyses, and Michael Hable for analyzing the dosing solutions/suspensions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the US Army Armament Research, Development, and Engineering Center (MIPR number MIPR8FGM009485, 2008).
