Abstract
Nitrotriazolone (3-nitro-1,2,4-triazol-5-one; NTO) and dinitroanisole (2,4-dinitroanisole; DNAN), insensitive energetic materials used in explosive formulations, have induced testicular toxicity and oligospermia in repeated-dose oral toxicity tests. To identify the target site of testicular toxicity of NTO and DNAN, Sprague Dawley rats were orally dosed with NTO (500 mg/kg/d) or DNAN (50 or 100 mg/kg/d) in corn oil for 1, 3, 7, or 14 days. Degeneration of germinal epithelium occurred in multiple tubule stages on days 7 and 14 in treated rats. Degeneration increased in severity with time and was characterized by degeneration/apoptosis of pachytene spermatocytes and round and elongating spermatids, depletion of step 19 spermatids, luminal spermatogenic cell sloughing, multinucleate cells, and pronounced Sertoli cell vacuolation. Serum luteinizing hormone and follicle-stimulating hormone did not differ between NTO- and DNAN-treated and control rats on any sampling day. Serum testosterone levels reduced only in rats given 50 mg/kg/d DNAN for 7 days. These results suggest that the initial site of testicular injury for both NTO and DNAN is the Sertoli cell.
Introduction
Nitrotriazolone (3-nitro-1,2,4-triazol-5-one; NTO) and dinitroanisole (2,4-dinitroanisole; DNAN) are components of insensitive munition (IM) mixtures such as IMX 101 and IMX 104. Insensitive munitions are viewed as safer replacements for traditional explosives (eg, 2,4,5-trinitrotoluene and hexahydro-1,3,5-trinitro-1,3,5-triazine) because they are designed to maintain chemical stability when subjected to unplanned, environmental stimuli, thus minimizing inadvertent and potentially catastrophic explosions. 1,2 As alternatives to traditional munition formulations, NTO and DNAN must meet performance requirements while demonstrating an acceptable safety level with regard to human health and the environment. Although IMs improve safety for warfighters, the production and use of IMs and their components raise concerns about potential health effects associated with occupational and environmental exposures. The presence of unexploded ordnance has led to contamination at training ranges. 3 Highly water-soluble components (eg, NTO) have the potential to migrate off-site and become a source of public soil and water contamination. Munition constituents may also be released to the environment in treated wastewater discharge from ammunition manufacturing facilities. 4,5
Nitrotriazolone and dinitroanisole demonstrate very different toxicity profiles. Nitrotriazolone has low acute toxicity, with a median lethal dose (LD50) greater than 5,000 mg/kg in rats and mice,
6
is negative in
Many components of the toxicity of DNAN can be attributed to its primary metabolite, 2,4-dinitrophenol.
11
Testicular toxicity has not, however, been demonstrated for 2,4-dinitrophenol,
14
suggesting that this effect is due to the parent compound. The toxicity of NTO is similarly due to the parent compound because, although 2 metabolites of NTO (5-hydroxy-1,2,4-trizol-3-one and 5-amino-1,2,4-triazol-3-one) were identified in
Subchronic studies with both DNAN and NTO demonstrated testicular toxicity; however, the prolonged duration of the exposures resulted in maturation depletion and complete or nearly complete loss of germ cell populations. 10,11 This precluded identification of the earliest change, a critical point in differentiating primary and secondary effects and elucidating the mode of action of testicular toxicity. The present study (WRAIR protocol 14-VET-20) was conducted to identify the target site of NTO and DNAN testicular toxicity in rats. To identify the earliest changes in testis morphology and serum reproductive hormones (luteinizing hormone [LH], follicle-stimulating hormone [FSH], and testosterone), a time course study was conducted in which rats were treated with NTO or DNAN for 3, 7, or 14 days.
Materials and Methods
Test Compounds
Neat NTO (CAS # 932-64-9; lot 11L375-061; purity 100%) and DNAN (CAS # 119-27-7; lot#BAE10H281-008; purity: 100%) were obtained from BAE Ordnance Systems (Kingsport, Tennessee). One NTO dosing suspension and 2 DNAN dosing suspensions were prepared for the main study by mixing test compounds with a measured volume of corn oil (Mazola May182015D). The concentration of each dosing solution preparation was verified analytically. Dinitroanisole suspensions were diluted in acetone and isoamyl acetate and analyzed using a gas chromatograph equipped with an electron capture detector and a DB-210 column. The NTO suspension was extracted using acetonitrile/water, and the concentration was verified via high-performance liquid chromatography with ultraviolet detection. Dosing suspensions were stored at room temperature prior to use and were stirred continuously during administration. Nitrotriazolone and DNAN dosing suspensions were previously determined to be stable for at least 1 month under these conditions. 11,13
Animals
Male (12 weeks old; 543.9 ± 55.5 g) Sprague Dawley rats (Crl: CD (SD) CD IGS; n = 80) were obtained from Charles River Laboratories (Wilmington, Massachusetts) and acclimatized for at least 7 days prior to study initiation. The animal room was maintained at 21.1°C to 23.3°C, with a relative humidity of 30% to 70%, 12 to 15 air changes hourly, and a 12:12-hour light:dark cycle. Rats were singly housed in suspended, polycarbonate cages on Beta Chip bedding (Northeastern Products Corp, Warrensburg, New York) and were provided a polycarbonate tunnel for enrichment. Rats were fed a pelleted rodent food (RMH3000; Lab Diet, St Louis, Missouri) and provided drinking water ad libitum. Sentinel rats used to monitor the health status of the experimental animals tested negative for all monitored pathogens. All procedures were approved by the Institutional Animal Care and Use Committee. The facility where this research was conducted is fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care, International. Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals and adhered to principles stated in the
Test Compound Administration
To examine the time course of testicular toxicity in rats treated with DNAN or NTO, a 4 × 4 factorial design with time course and dose as factors was used. Levels for the time course were day 1, 3, 7, and 14. The dose factor levels were set at 50 and 100 mg/kg/d DNAN, 500 mg/kg/d NTO, and corn oil vehicle control, based on results from previous studies conducted in rats. 10 -13 Following an acclimatization period, rats were stratified by body mass and randomly distributed to 1 of 16 treatment groups (5 rats/group). Dosing suspensions were administered via oral gavage using a stainless steel 16 G × 2 in gavage needle at approximately the same time daily for 1 to 14 days based on time course grouping. Vehicle control animals were dosed with corn oil at the same volume per body mass (5 mL/kg) as the test compound exposed animals. Animals were weighed daily and observed for morbidity and signs of toxicity at least twice daily.
Necropsy
Surviving rats were anesthetized with carbon dioxide (CO2), blood for hormone assays was collected by intracardiac puncture, and rats were euthanized by CO2 asphyxiation and thoracotomy approximately 24 hours after the last treatment. Necropsy order was randomized across treatment groups. The left epididymis and testis were separated, trimmed, and weighed. The right testis and epididymis were fixed in modified Davidson fixative for 24 to 48 hours and submitted for histopathology.
Measurement of Serum Hormones
Blood samples were centrifuged and stored at −80°C prior to analysis. Follicle-stimulating hormone and LH were measured in serum using rat enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s protocols (TSZ ELISA, Waltham, Massachusetts). Serum testosterone was determined using a rat testosterone ELISA kit according to the manufacturer’s protocol (MyBiosource, San Diego, California). Samples and standards were assayed in duplicate. An external quality control standard for testosterone (Rat Control 1 Lot 001) was within 93% of the reference point.
Histology and Light Microscopy
The testis and epididymis were paraffin embedded, sectioned at 5 µm, and routinely processed to slides. Slides were stained with periodic acid-Schiff reagent and hematoxylin or hematoxylin and eosin. A board-certified pathologist examined slides, via light microscopy, for compound-induced histopathology changes. The prevalence and severity of findings was compared to controls, and a stage-aware evaluation of spermatogenesis was conducted to determine the target cell(s) of toxicity. Testis injury was semiquantitatively graded using a 5-point scale based on the percentage of affected tubules: grade 1 (minimal), <5% of the tubules; grade 2 (slight), 6% to 25% of tubules affected; grade 3 (moderate), 26% to 50% of tubules affected; grade 4 (marked), 51% to 75% of the tubules affected; and grade 5 (severe), >75% of the tubules affected. A severity grade of 0 was assigned to a parameter if the parameter was within normal limits. A similar 1 to 5 (minimal–severe) semiquantitative grading scale was used in the evaluation of the epididymis. Notable stage- and cell-specific findings were described as applicable.
Statistical Analysis
Body weight, organ weight, and serum hormone levels were expressed as mean (standard deviation). The mean severity grade was calculated as the sum of severity grades (for each respective testis or epididymal finding) divided by the number of rats exhibiting the respective finding. Data were analyzed using SPSS 21.0. Continuous data were analyzed using a 2-factor analysis of variance (ANOVA) with dose and time course as the main effects. If the interaction between the main effects was significant, the effect of dose on the variable was determined within each level of time course using a 1-factor ANOVA. When significant main effects were observed (
Results
Analytical Chemistry
Analytical concentrations were 93% to 96% and 89% of nominal concentrations for the DNAN and NTO suspensions, respectively. All results were within the 70% to 130% recovery limits for the analysis. As such, all results were reported using the nominal concentrations.
Clinical Observations and Mortality
One rat in the control group developed respiratory difficulty, likely due to dosing difficulty, and was removed from the study on day 1. All rats in the 100 mg/kg/d DNAN group either died or were determined to be moribund and euthanized on day 1. One rat in the day 7, 50 mg/kg/d DNAN group died on day 2. All other rats survived to scheduled necropsy.
Body and Organ Weights
Final body weight did not differ between control and treated rats on any sampling day (Table 1). In NTO-treated rats, testis weight was reduced (29%), relative to controls, only on day 14 (
Body and Organ Weights in Rats Orally Dosed With DNAN or NTO for 1, 3, 7, or 14 Days.
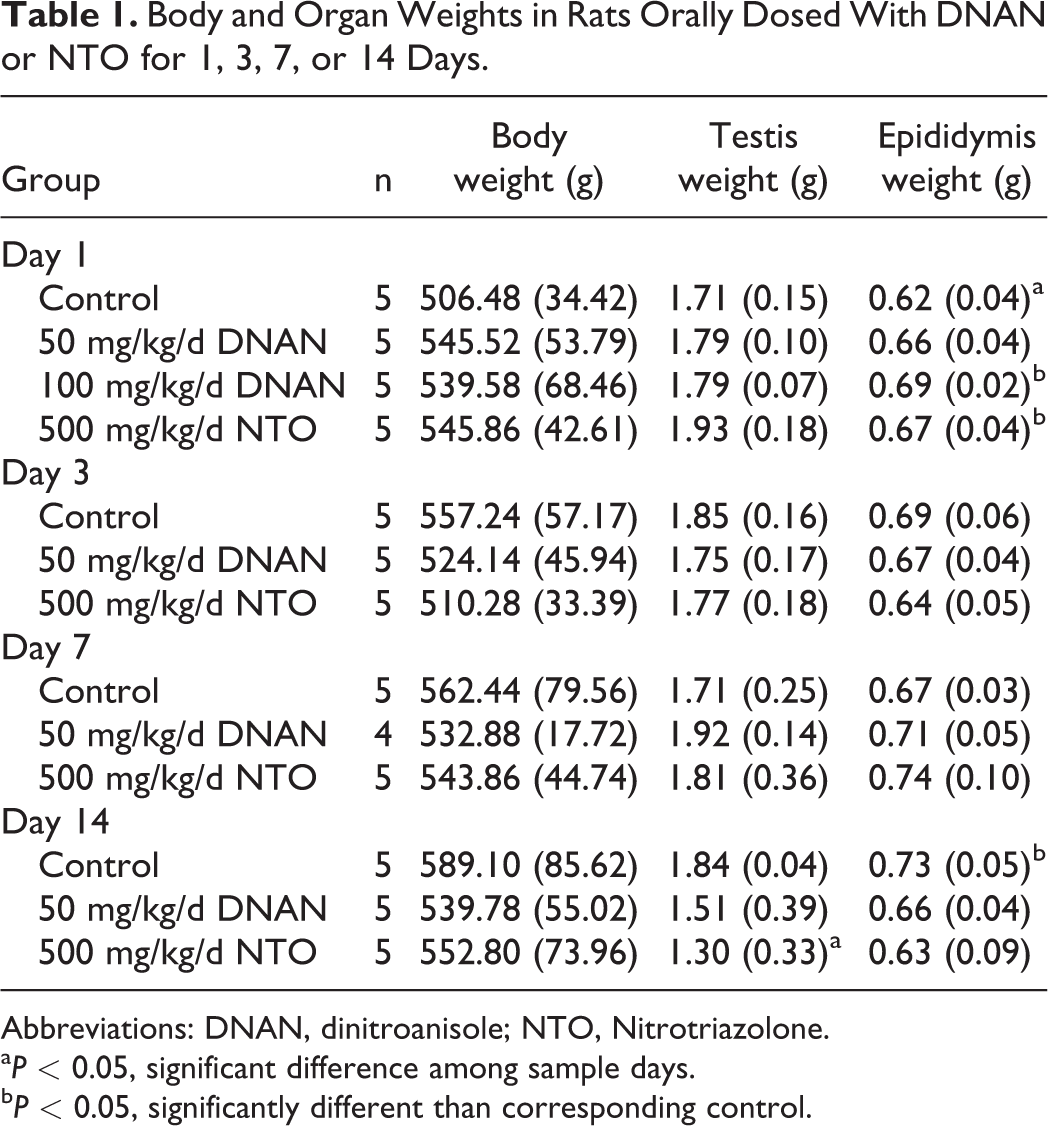
Abbreviations: DNAN, dinitroanisole; NTO, Nitrotriazolone.
a
b
Serum Hormone Levels
Compared to corresponding controls, serum testosterone levels were decreased by 90% in 50 mg/kg/d DNAN rats on day 7 only. No biologically significant changes were observed in serum LH, FSH, or testosterone levels. This may be due to the limited number of animals, robustness of response, single time point collection, and method of collection (data not shown).
Histology and Light Microscopy
Degeneration/atrophy of germinal epithelium was noted in rats exposed to 50 mg/kg/d DNAN or 500 mg/kg/d NTO on days 7 and 14, but not on days 1 and 3. The degeneration was characterized by degeneration/apoptosis of predominantly pachytene spermatocytes (noted in multiple stages; coded in Table 2 for stage 7/8 tubules), degeneration/sloughing of round and elongating spermatids (noted in multiple stages; coded in Table 2 for stage 7/8 tubules), depletion of step 19 spermatids, and luminal spermatogenic cell sloughing (Figure 1). Multinucleate cell formation (luminal and intraepithelial) and pronounced Sertoli cell degeneration (noted as cytoplasm vacuolar change) were significantly increased only in rats exposed to 500 mg/kg/d NTO. Severity was generally greater in NTO- (moderate to severe) than in DNAN- (slight to moderate) exposed rats and increased from day 7 to day 14 in both the groups. Epididymal hypospermia and sloughed luminal multinucleate/germ cells (including residual bodies) occurred on days 7 and 14 in rats exposed to 500 mg/kg/d NTO and on day 14 in rats exposed to 50 mg/kg/d DNAN. Similarly, though mild, testis degeneration/atrophy was also noted in 1 rat exposed to 100 mg/kg/d DNAN for 1 day. All control tissues were within normal limits.
Testicular and Epididymal Histopathology in Rats Orally Dosed With DNAN and NTO for 1, 3, 7, or 14 Days.a
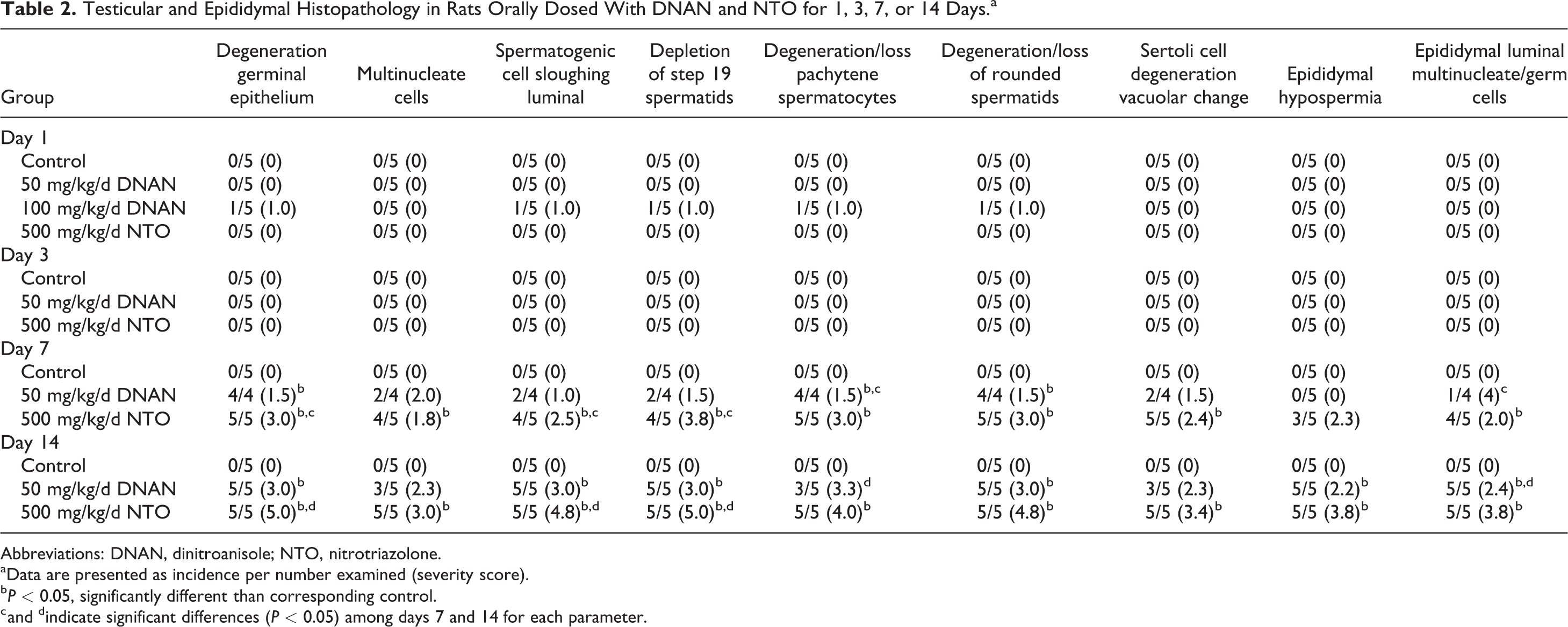
Abbreviations: DNAN, dinitroanisole; NTO, nitrotriazolone.
aData are presented as incidence per number examined (severity score).
b
c and dindicate significant differences (
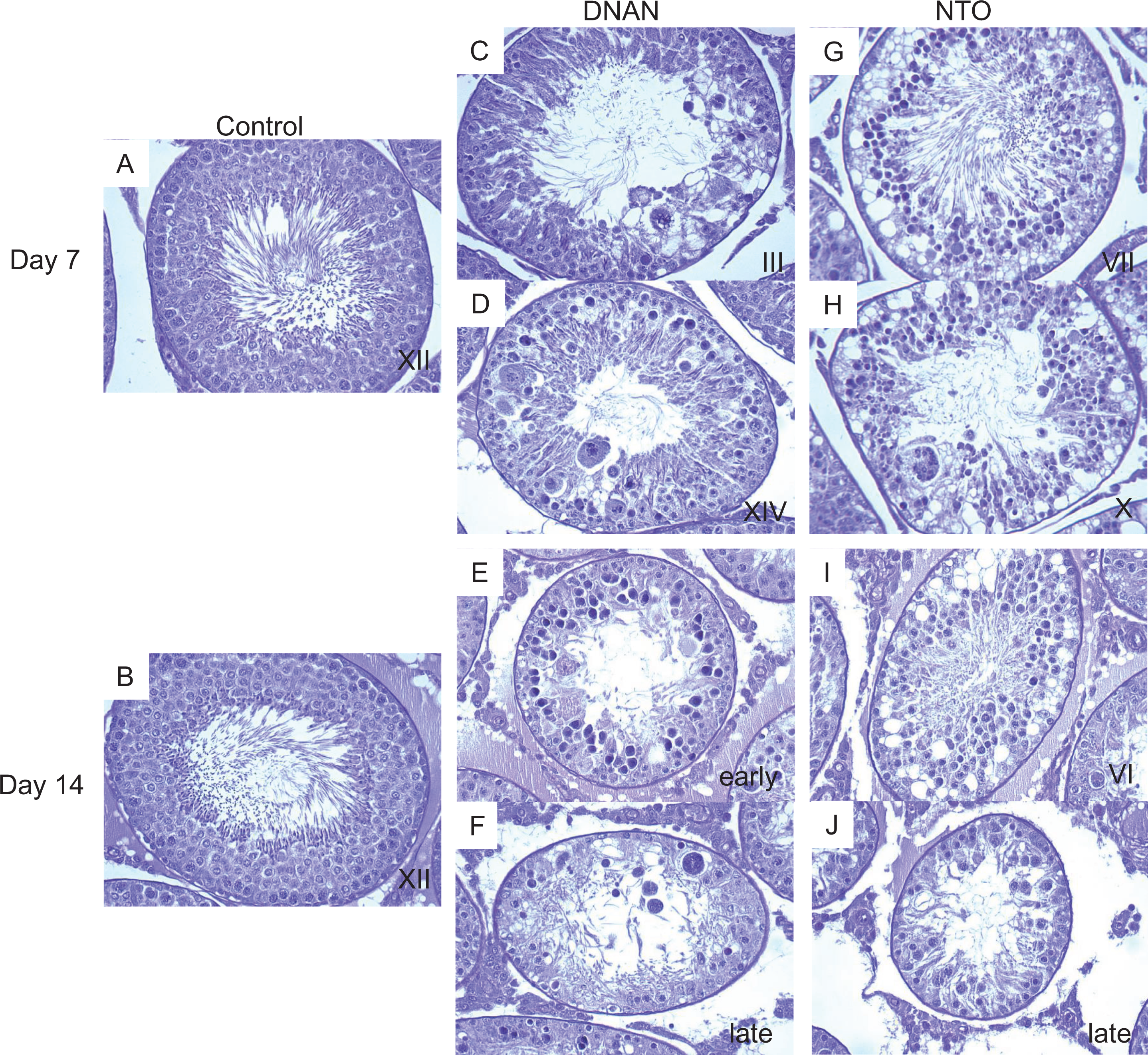
Light micrographs of seminiferous tubules from rats orally dosed with corn oil, 50 mg/kg/d DNAN, or 500 mg/kg/d NTO for 7 or 14 days. Stage 7 tubules from control rats on day 7 (A) and day 14 (B). C, An early stage tubule (stage 3) on DNAN day 7 exhibiting degeneration of elongating spermatids to include multifocal apoptotic spermatocytes (and loss) in association with pronounced Sertoli cell vacuolization. D, Stage 14 tubule on DNAN day 7 exhibiting Sertoli cell vacuolization associated with apoptosis of pachytene spermatocytes and degeneration/loss of step 14 spermatids. E, An early-stage tubule on DNAN day 14 exhibiting degeneration/loss of pachytene spermatocytes and elongating spermatids. Sertoli cells appear finely vacuolated with lacey cytoplasm (evidence of Sertoli cell degeneration). Numerous apoptotic spermatocytes (pachytene) are evident. Note the preservation of spermatogonia along the basement membrane. F, Suspected late-stage tubule on DNAN day 14 exhibiting diffuse degeneration/loss of pachytene/zygotene spermatocytes and elongating spermatids. Note the paucity of spermatocytes/elongating spermatids. Pronounced Sertoli cell degeneration is marked by vacuolization and lacey cytoplasm. Apoptotic pachytene spermatocytes (and 1 multinucleate cell) reside within Sertoli cell vacuoles. Note the preservation of spermatogonia along the basement membrane. G, Stage 7 tubule on NTO day 7 exhibiting diffuse apoptosis of pachytene spermatocytes and elongating spermatids. Note the pronounced Sertoli cell vacuolization, consistent with degeneration. H, Late-stage tubule (suspected stage 10) on NTO day 7 exhibiting numerous apoptotic pachytene spermatocytes. Note the multinucleate cell formation, cell sloughing, and paucity of spermatocytes/elongating spermatids. Sertoli cells contain several large and small cytoplasmic vacuoles. In rats exposed to NTO for 14 days, degeneration/loss of pachytene spermatocytes and elongating spermatids is evident; Sertoli cells contain numerous large and small vacuoles (degeneration), and numerous apoptotic spermatocytes (suspected pachytene) are evident in stage 6 (I) and late stage (J) tubules. Note the preservation of spermatogonia along the basement membrane. Periodic acid-Schiff reagent (PAS; ×40). DNAN indicates dinitroanisole; NTO, nitrotriazolone.
Discussion
In developmental and reproductive studies in rats, NTO demonstrated effects consistent with reduced testosterone levels. 12,13 In peripubertal rats given 250 mg/kg/d NTO and F1 generation rats given 3,600 mg/L NTO, the testes exhibited degenerative changes characteristic of low testosterone levels, including degeneration of pachytene spermatocytes and depletion or absence of elongating spermatids. 12,13 Additionally, the weight of accessory sex organs (ie, seminal vesicles and lateral prostate), which are the most sensitive indicator of reduced testosterone as they integrate changes in androgen levels over time, 19 was reduced in both studies. These studies suggest that NTO-induced reproductive toxicity is mediated by reduced androgenic tone, the most common hormonal cause of altered male reproductive morphology. 19
What is not apparent from these studies is whether the reduced androgenic tone is secondary to central action on gonadotropin releasing hormone (GnRH) and/or LH due to reduced steroidogenesis by Leydig cells, altered androgen receptor interactions, altered metabolism and excretion of testosterone, or a consequence of altered germ cell populations and/or Sertoli cell function.
20
Intratesticular testosterone levels can be 40-fold to 250-fold higher than serum testosterone levels in rats and man, respectively, but the relationship between serum and intratesticular levels is not constant. 22 -24 Intratesticular testosterone supports development of pachytene spermatocytes and germ cells through stage VII by controlling secretion of proteins from Sertoli cells 25 ; however, spermatogenesis can be maintained by 5% to 10% of normal intratesticular testosterone levels. 22 Despite this, testosterone depletion results in characteristic effects on spermatogenesis that take a finite period to develop, with initial degeneration of pachytene spermatocytes and round spermatids being observed in stage VII tubules 4 days after withdrawal of testosterone. 20 With time, the number of degenerating cells in stages VII and VIII increases, and cells fatally damaged by the absence of testosterone at stages VII and VIII appear as degenerating cells in subsequent stages (X and XI). Effects on Sertoli or germ cells seen prior to day 4 are likely a consequence of direct toxicity rather than a consequence of hormonal changes. In the current study, spermatogenesis was not affected in rats exposed to 50 mg/kg/d DNAN or 500 mg/kg/d NTO for 1 or 3 days; the earliest effects on spermatogenesis occurred on day 7 in both the groups. This timing might suggest an endocrine-mediated effect. However, degeneration was observed in all stages on day 7, which would not be expected for testosterone depletion based on cell-cycle progression of effects beginning on day 4 in stages VII and VIII. Additionally, the limited data available from the 100 mg/kg/d DNAN group indicates that, at higher doses, effects occur as early as day 1 for DNAN, earlier than when endocrine-mediated effects would occur. Of course, different mechanisms could be operational at high doses. Although the pattern of morphological changes previously observed following exposure to NTO was consistent with low testosterone levels, 12,13 that pattern was not observed in the current study. Here, diffuse loss of pachytene/zygotene spermatocytes and step 1 to 19 spermatids was noted in tubules at all stages (1-14) of the cycle for both NTO- and DNAN-exposed rats. This was accompanied by Sertoli cell degeneration marked by cytoplasm vacuolization and rarefaction (lacey cytoplasm). The testicular effects were associated with hypospermia and sloughed luminal multinucleate/germ cells in the epididymides, common secondary responses to testicular toxicity. The epididymal effects associated with testosterone deficiency, including decreased weight and atrophy, apoptosis of epididymal epithelium, cribriform changes, and decreased tubular diameter, 26 were not observed in the present study.
Germinal epithelial degeneration observed in mice exposed to NTO was also accompanied by indications of Sertoli cell damage. 27 In mice exposed to 500 or 1,000 mg/kg/d NTO for 1, 3, 7, or 14 days, degeneration of spermatids and spermatocytes was first observed at 7 days in the 500- and 1,000 mg/kg/d groups. In contrast to rats in which degeneration was noted in all stages, in mice, degeneration of spermatids was first noted in stage II–III tubules in the 1,000 mg/kg/d NTO group and in stage IX tubules in the 500 mg/kg/d NTO group. By day 14, spermatocytes in stage XII tubules were also affected in the 1,000 mg/kg/d group. This degeneration was accompanied by spermatid retention in stage XII and stage I tubules on day 14 in the 1,000 mg/kg/d group. 27 As the release of mature spermatids is a function of Sertoli cells, their retention may also be indicative of NTO-induced Sertoli cell damage. 28
Because Sertoli cells provide support and nutrition to germ cells and are necessary for proper spermatogenesis, toxicant-induced impairment of Sertoli cell function can increase testicular germ cell apoptosis. Germ cell apoptosis can also be induced by direct injury to the germ cell, making determination of the primary site of toxicity difficult. 29,30 However, apoptosis of the spermatocyte germ cell and persistence of spermatogonia is a hallmark of Sertoli cell injury, while apoptosis of spermatogonia is primarily seen with direct germ cell toxicity. 31 Thus, in the current study, the apoptosis observed in spermatocytes and preservation of the spermatogonia population suggests that both NTO and DNAN are primary Sertoli cell toxicants. Additionally, vacuolation of Sertoli cells was observed starting on day 7 of exposure to 50 mg/kg/d DNAN and 500 mg/kg/d NTO and on day 1 in a single rat exposed to 100 mg/kg/d DNAN. Sertoli cell vacuolation is a common, early morphological response to Sertoli cell injury that has been observed following exposure to 2,5-hexanedione, 32,33 tri-o-cresyl phosphate, 34 1,3-dinitrobenzene, 35 cyclohexylamine, 36 perfluorooctanesulfonic acid (PFOS), 37 and phthalate esters. 38 Although Sertoli cell vacuolation is generally an early event that precedes germ cell degeneration, in the present study, Sertoli cell vacuolation and germ cell degeneration co-occurred. This may have been an artifact of the experimental design; Sertoli cell vacuolation may have first occurred between sampling days 3 and 7. This time course study suggests the Sertoli cell as the target site of action for both NTO and DNAN. This is in contrast to results of earlier NTO studies that were consistent with reduced testosterone-mediated effects. This difference may be due to the timing of the histopathological assessment or differential effects of NTO on developing rats, as the earlier NTO studies suggesting testosterone-mediated effects were conducted in prepubertal and neonatal rats.
The mechanisms of Sertoli cell toxicity have proven difficult to determine despite extensive investigations. 39 The cytoskeleton, however, appears to be a primary target of Sertoli cell toxicants. 40 Carbendazim, 41,42 2,5-hexanedione, 43 -46 tri-o-cresylphosphate, monoethylhex-yl phthalate, 47 and PFOS 48 disrupt microtubule assembly that can alter Sertoli–germ cell adhesion, Sertoli cell–cell adhesion, structural support of the seminiferous epithelium, movement of spermatids, and spermiation. 49 Sertoli cell toxicants that target the cytoskeleton can cause a variety of morphological changes including sloughing of the germinal epithelium, abnormal location of elongate spermatids, residual body retention, retained spermatids, Sertoli cell vacuolation, seminiferous tubule atrophy, and malorientation of round spermatids. 40,50 Although the exact mode of action cannot be determined based on histopathological evaluation alone, observation of the signature lesion of cytoskeletal toxicants and sloughing of the seminiferous epithelium following exposure to DNAN and NTO indicates that the Sertoli cell cytoskeleton should be considered a target for both DNAN and NTO. 40
In conclusion, the results of the present study suggest that DNAN and NTO are Sertoli cell toxicants. Observation of signature histopathological lesions suggests disruption of the Sertoli cell cytoskeleton as a possible mode of action. However, mechanistic studies are required to confirm this. Evidence of altered androgen tone observed in previous studies with NTO is likely secondary to Sertoli cell toxicity.
Footnotes
Authors’ Note
The views expressed in this article are the views of the author(s) and do not reflect the official policy of the Department of the Army, the Department of Defense, or the US Government.
Acknowledgments
The authors thank the WRAIR technicians and animal husbandry staff, especially Tesafaye Mekonnen. The authors thank Anthony Skowronek (Battelle, Columbus, Ohio) for expert completion of the histopathological analyses and Karl P. Kroeck, APHC, Laboratory Sciences Directorate for performing the analytical chemistry.
Author Contributions
Emily May Lent, Anna B. Mullins, and Kenneth E. Despain designed the experiments; Emily May Lent, Anna B. Mullins, Anthony D. May, Cary L. Honnold, and Kenneth E. Despain performed the experiments; Emily May Lent analyzed the data; Emily May Lent and Anna B. Mullins drafted the manuscript; Emily May Lent, Anna B. Mullins, Anthony D. May, Cary L. Honnold, and Kenneth E. Despain critically revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Strategic Environmental Research and Development Program (SERDP; Project ER-2223).
