Abstract
Nanosized titanium dioxide (nano-TiO2) is widely used in the chemical, electrical, and electronic industries. Nanosized TiO2 has been reported to be an efficient photocatalyst, which is able to produce reactive oxygen species (ROS) under UVA irradiation. In the present work, we evaluate the effect of mitochondrial respiratory chain on the generation of ROS and cytotoxicity in keratinocyte (HaCaT) cells induced by nano-TiO2 under UVA irradiation. HaCaT cells were pretreated with different inhibitors of mitochondrial respiratory chain and followed by treatment with 200 µg/mL nano-TiO2, then exposed to UVA (365 nm) for 1 hour and cultured for 24 hours. Our results demonstrated that the complexes I and III of the mitochondrial respiratory chain are the major site in the ROS generation induced by nano-TiO2. Our results also demonstrated that the uncouplers of mitochondrial oxidative phosphorylation resulted in obvious changes in the production of intracellular ROS induced by nano-TiO2. The ROS sources of lipoxygenase, cyclooxygenase, and nicotinamide adenine dinucleotide phosphate oxidase had no significant effect on the ROS production. To some extent, nitric oxide synthase had effect on the ROS production. These results indicated that mitochondrial respiratory chain may be the main source of intracellular ROS production induced by nano-TiO2.
Keywords
Introduction
Among the various nanomaterials, nanosized titanium dioxide (nano-TiO2) is chemically inert, noncorrosive, and photocatalytic, thus, nano-TiO2 has been widely applied in consumer products, pigments, 1 photocatalysts, 2 and cosmetic products. 3 For this reason, toxicological properties of nano-TiO2 have been studied with several routes of exposure, including dermal, oral, and pulmonary exposures. Nano-TiO2 showed toxicity in many animal organ types, including lung, 4,5 kidney, 6 liver, 7 and brain. 8 It has been demonstrated that reactive oxygen species (ROS) generation is responsible, at least in part, for the toxicity of nano-TiO2, which resulted in an inflammatory response. 9 –11 The role of cellular ROS induced by nano-TiO2 has gained much attention, but the exact mechanism was not fully described.
Mitochondria are a source of ROS from superoxide anion formed along electron transfer in the respiratory chain. It is converted into hydrogen peroxide (H2O2) by superoxide dismutase (SOD), and consecutively, H2O2 turns into water by glutathione peroxidase and catalase. It has been demonstrated that nano-TiO2 is able to induce an increase in ROS production and cause damage to DNA in human lung cells 12 and to induce micronucleus formation in human epidermal cells. 13 Nano-TiO2 reduces the glutathione content, 13 increases lipid peroxidation, and decreases SOD levels. 14 Even though nano-TiO2 is not internalized into mitochondria, there is an increasing evidence for the alterations induced in mitochondria, including cytochrome c release from mitochondria to cytosol, changes in mitochondrial membrane permeability, and a decrease in mitochondrial membrane potential. 14
Thus, based on the hypothesis that exposure of cells to nano-TiO2 may impair mitochondria, the present study was designed to investigate the effect of the role of mitochondrial respiratory chain in the generation of ROS induced by nano-TiO2. Furthermore, the sites in the ROS generation induced by nano-TiO2 was also examined.
Materials and Methods
Nano-TiO2 and Reagents
Nano-TiO2 (P25, 25% rutile, and 75% anatase) with 21 nm was purchased from Degussa GmbH (Germany). The particle sizes and distribution of nano-TiO2 were measured by Tecnai G2 20 transmission electron microscope (FEI, Holland). Crystal structure was characterized by X’Pert Prox X-ray diffraction instrument (FEI). HaCaT cells were purchased from China Center for Type Culture Collection (China). Rhodamine 123, 2′,7′-dichlorofluorescein diacetate (DCFH-DA) and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma (St. Louis, MO). Modified Eagle medium (MEM), penicillin–streptomycin, and fetal bovine serum (FBS) were obtained from Hyclone Laboratories Inc. All other chemicals were of the highest grade that could be obtained commercially.
Preparation Suspension of Nano-TiO2
Nano-TiO2 was suspended in the cell culture medium and dispersed using a sonifier cell disruptor equipped with a microprobe for 10 to 15 minutes to produce the suspension. In each study, the suspension was freshly prepared and then immediately applied to human keratinocyte (HaCaT) cells.
Cell Culture and Exposure to Nano-TiO2
HaCaT cells were grown in MEM supplemented with 10% FBS in the presence of 5% CO2 in air at 37°C. Cells were incubated up to approximately 24 hours and grown to about 80% confluence before experiments. Rotenone (ROT, 10 μmol/L), thenoyltrifluoroacetone (TTFA, 25 μmol/L), antimycin A (AA, 10 μmol/L), and sodium azide (NaN3, 1 mmol/L) were used as inhibitors for complexes I, II, III, and IV, respectively. Diphenyleneiodonium (DPI) chloride (10 μmol/L) and Nω-nitro-
UVA Irradiation
UVA light was provided by a UV lamp (Shanghai Jinguang Instrument, China) with continuous emission spectrum with a peak at 365 nm. The UV intensity at 365 nm was determined using a UVA radiometer (Beijing Normal University, China). The UVA lamp had a fluence rate of 3.5 mW/cm2 and is an appropriate substitute for UVA occurring in natural sunlight. 15 As reference, 1 hour irradiation corresponds to an incident dose of UVA of 12.6 J/cm2.
Measurement of Intracellular ROS
2′,7′-Dichlorofluorescein diacetate is a cell-permeable nonfluorescent probe that is cleaved by intracellular esterases and turns into the fluorescent dichlorofluorescein upon reaction with ROS. The level of ROS generation was determined by the concomitant increase in dichlorofluorescein fluorescence. After treatments, cells were treated with 10 μmol/L DCFH-DA for 30 minutes at 37°C. The cells were washed again with PBS and then sonicated in 300 μL 0.1% Triton X-100 (PBS, pH 7.4) on ice for 10 seconds. After incubation at 4°C for 10 minutes, the homogenates were centrifuged at 12,000 rpm for 30 minutes, and the supernatants were used for assay with excitation wavelength at 488 nm and emission wavelength at 525 nm.
Cell Viability Assay
The cytotoxicity of particles was determined using the MTT assay. Briefly, cells were seeded in 96-well culture plates at a density of 5 × 103 cells/well; after treatment, cells were then washed and wells refilled with fresh culture medium containing 0.5 mg/mL MTT (M2003; Sigma-Aldrich). After 4 hours, the insoluble crystals were solubilized by dimethyl sulfoxide 150 µL/well, and the absorbance was read at 490 nm.
Measurement of Intracellular MDA
HaCaT cells (1 × 105 mL−1) were transferred to 24-well plates in 1 mL MEM and preincubated overnight. After preincubation, the cells were incubated at 37°C for 24 hours without or with 200 µg/mL nano-TiO2. At the end of the treatment, cells were washed with PBS and homogenized in 300 µL 0.1% Triton X-100 (PBS, pH 7.4) through sonication on ice for 10 seconds. After incubation at 4°C for 10 minutes, the homogenates were centrifuged at 10,000 rpm for 10 minutes, and the supernatants were used for the assay. Malondialdehyde (MDA) content was measured with the MDA kit by a spectrophotometer (WFZ, UV-2102).
Measurement of Cell Death
Cell death was quantified by Evan blue. For 100% cell death, the culture was heated at 100°C for 5 minutes. The percentage of cell death in selected treatments was also verified by counting several hundred cells under a microscope.
Western Blot Analysis
Proteins from mitochondrial as well as cytosolic fraction (50 µg) were separated on 12.5% (wt/vol) sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The proteins were transferred on to nitrocellulose, blocked in Tris-buffered saline, 0.3% (wt/vol) Tween 20, and 3% (wt/vol) dried skimmed milk, and they were labeled with antibody against cytochrome c (1/1,000; Zymed Laboratories, South San Francisco) at 4°C overnight. After washing (3 times for 15 minutes) in block buffer, the membrane was incubated in rabbit antimouse horseradish peroxidase conjugate (1/5,000) in Tris-buffered saline/Tween 20 for 3 hours at room temperature and washed again. Labeling was detected by chemiluminescence.
Statistical Analysis
Statistical analyses were performed using Origin software (version 6.1; OriginLab). Experimental results were expressed as mean ± standard deviation (SD) for 3 different replicates at each test concentration. Statistical significance was established at P < 0.01.
Results
Characterization of Nano-TiO2 Suspension
The morphology and size of nano-TiO2 are shown in Figure 1A. There was no surface coating on nano-TiO2. The size of nano-TiO2 particles that were ultrasonicated ranged from 20 to 50 nm. The crystal structure of nano-TiO2 is shown in Figure 1B, which was a mixture of anatase and rutile.
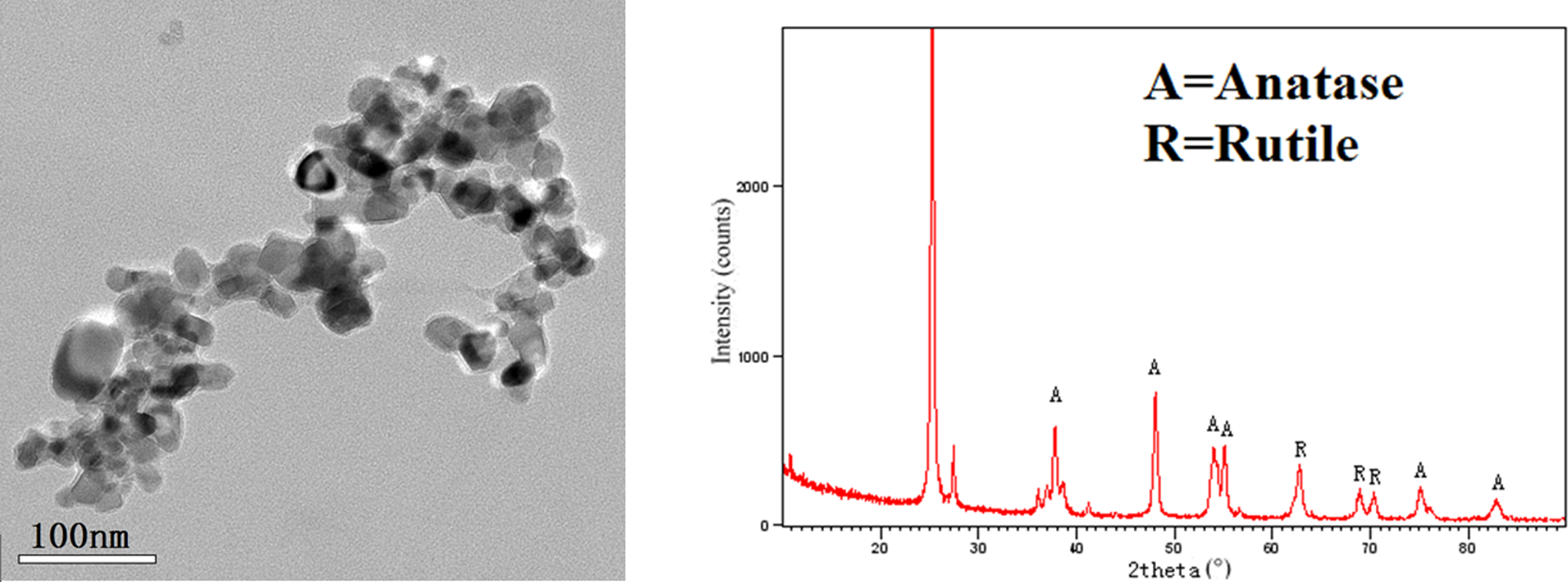
Transmission electron micrographs and X-ray diffraction of nano-TiO2 suspension. nano-TiO2 indicates nanosized titanium dioxide.
Effects of Mitochondrial Respiratory Chain on the ROS Production Induced by Nano-TiO2
The mitochondrial respiratory chain is a major source of ROS under pathological conditions. Oxidative stress is therefore implicated in cell injury, thereby causing inflammatory processes. As shown in Figure 2, the fluorescence in cells with nano-TiO2 treatment significantly increased as compared with the control group. Pretreatment with ROT (Figure 2A) and AA (Figure 2B) increased the fluorescence by 32.26% and 22.69%, respectively, compared with the nano-TiO2 group. On the contrary, our results also demonstrated that the fluorescence in cells pretreated with TTFA (Figure 2C) and NaN3 (Figure 2D) alone was not altered.
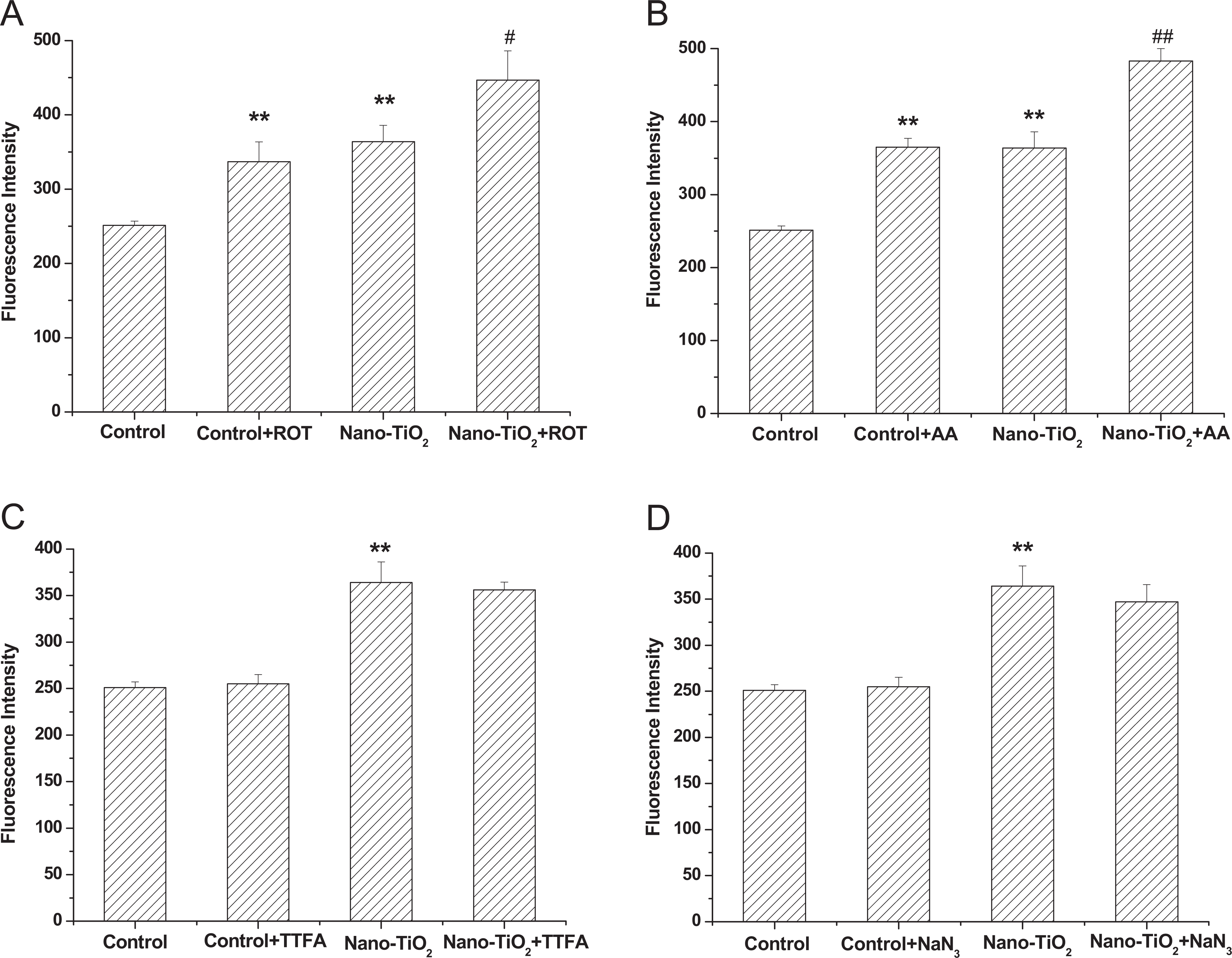
Effects of ROT (A), AA (B), TTFA (C), and NaN3 (D) on intracellular ROS production induced by nano-TiO2. Cells were treated with 200 µg/mL nano-TiO2 only, or pretreated with ROT (10 μmol/L), AA (10 μmol/L), TTFA (25 μmol/L), and NaN3 (1 mmol/L) for 30 minutes, respectively, followed by treatment with 200 µg/mL nano-TiO2. Control received the culture medium only. All samples were irradiated with the UVA light for 1 hour and then cultured for 24 hours. Results are expressed as mean ± SEM of at least 4 different experiments. **P < 0.01 represents the comparison with the control group; # P < 0.05 and ## P < 0.01 represent the comparison with the nano-TiO2 group. AA indicates antimycin A; nano-TiO2, nano titanium oxide; ROS, reactive oxygen species; ROT, rotenone; SEM, standard error of the mean; TTFA, thenoyltrifluoroacetone.
Effects of Uncouplers of Mitochondrial Oxidative Phosphorylation on the ROS Production Induced by Nano-TiO2
To further study the effects of mitochondrial respiratory chain on intracellular ROS production induced by nano-TiO2, CCCP was used as an inhibitor for uncouplers of mitochondrial oxidative phosphorylation. As shown in Figure 3, the fluorescence in cells was not altered significantly as compared with the control group. However, the fluorescence in cells of nano-TiO2 pretreated with CCCP decreased by 32.26% compared with the nano-TiO2 group.
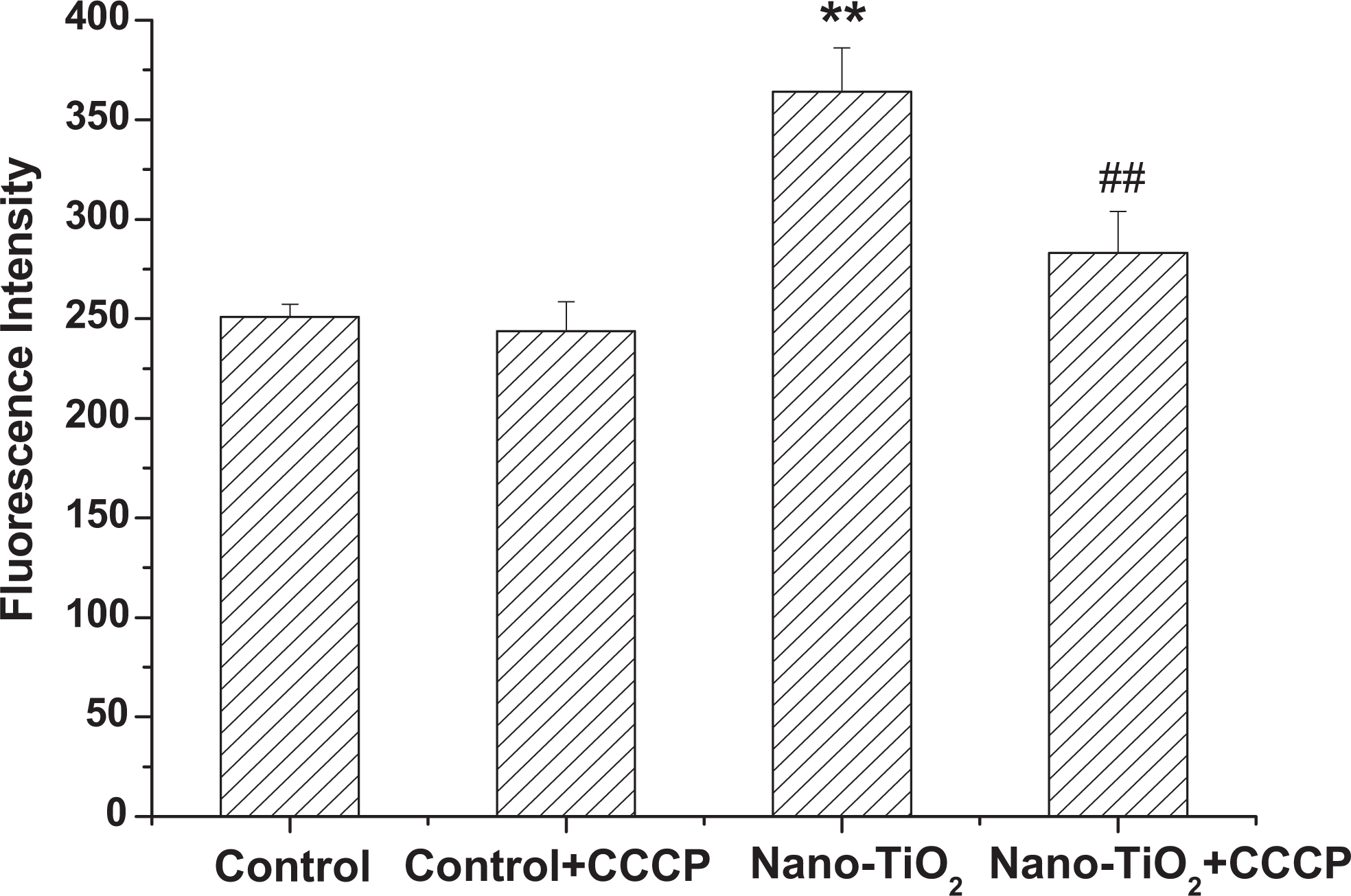
Effects of CCCP on intracellular ROS production induced by nano-TiO2. Cells were treated with 200 µg/mL nano-TiO2 only, or pretreated with CCCP (2.0 μmol/L) for 30 minutes, followed by treatment with 200 µg/mL nano-TiO2. Control received the culture medium only. All samples were irradiated with the UVA light for 1 hour and then cultured for 24 hours. Results are expressed as mean ± SEM of at least 4 different experiments. **P < 0.01 represents the comparison with the control group; # P < 0.05 and ## P < 0.01 represent the comparison with the nano-TiO2 group. CCCP, carbonyl cyanide m-chlorophenylhydrazone; ROS, reactive oxygen species; nano-TiO2, nanosized titanium dioxide; SEM, standard error of the mean.
Effects of NADPH Oxidase and NOS on the ROS Production Induced by Nano-TiO2
Several intracellular sources contribute to the production of ROS, including NADPH oxidase, NOS, LOX, and xanthine oxidase.
16
To test whether the NADPH oxidase and NOS were involved in the production of ROS in HaCaT cells induced by nano-TiO2, HaCaT cells were preincubated with specific inhibitors of NADPH oxidase and NOS. During the addition of NADPH oxidase inhibitor, DPI chloride, the fluorescence in cells was not altered compared with the nano-TiO2 group (Figure 4A). While addition of NOS inhibitor, Nω-nitro-
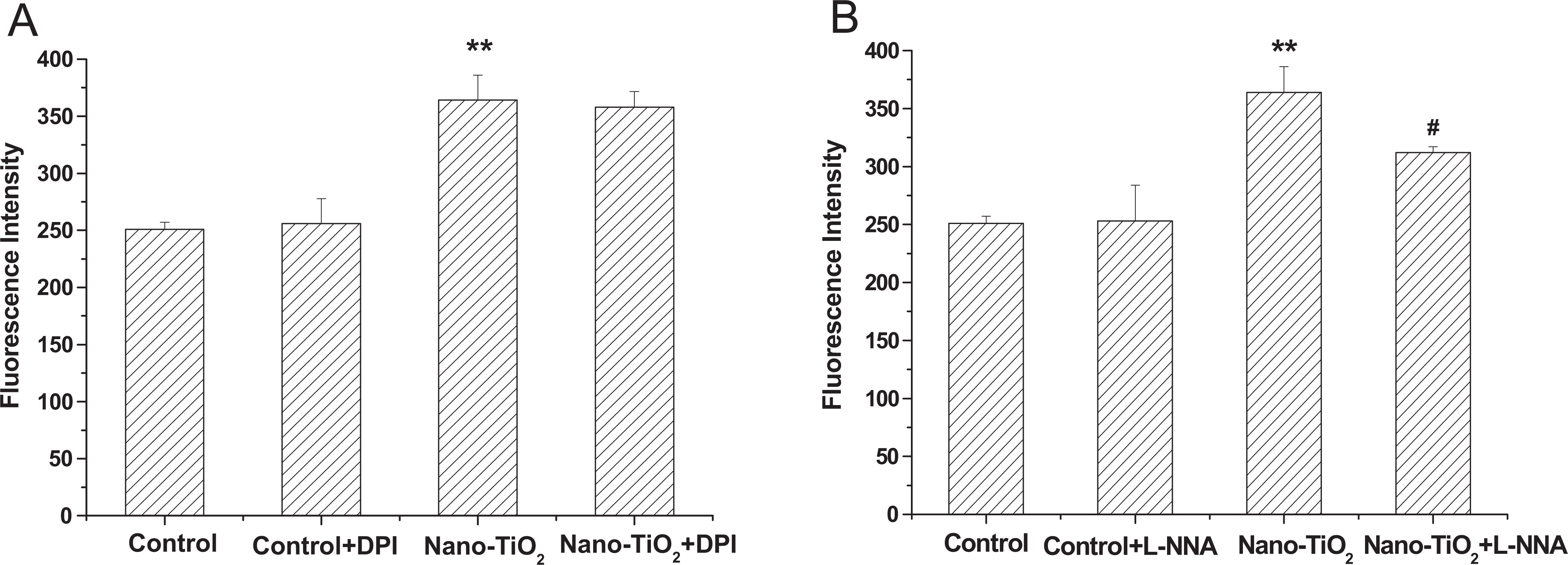
Effects of DPI (A) and
Effects of Arachidonic Acid Metabolism on the ROS Production Induced by Nano-TiO2
Reactive oxygen species are also generated by arachidonic acid metabolites, which are released from membrane phospholipids via the activity of cytosolic phospholipase A2. 17 In this study, we describe recent studies concerning the generation of ROS by arachidonic acid metabolites. In particular, we have focused on the manner in which arachidonic acid metabolism via LOX and COX contributes to ROS generation. However, during the addition of LOX inhibitor, NDGA (Figure 5A) and COX inhibitor, INDO (Figure 5B), the fluorescence in cells was not altered compared with the nano-TiO2 group.
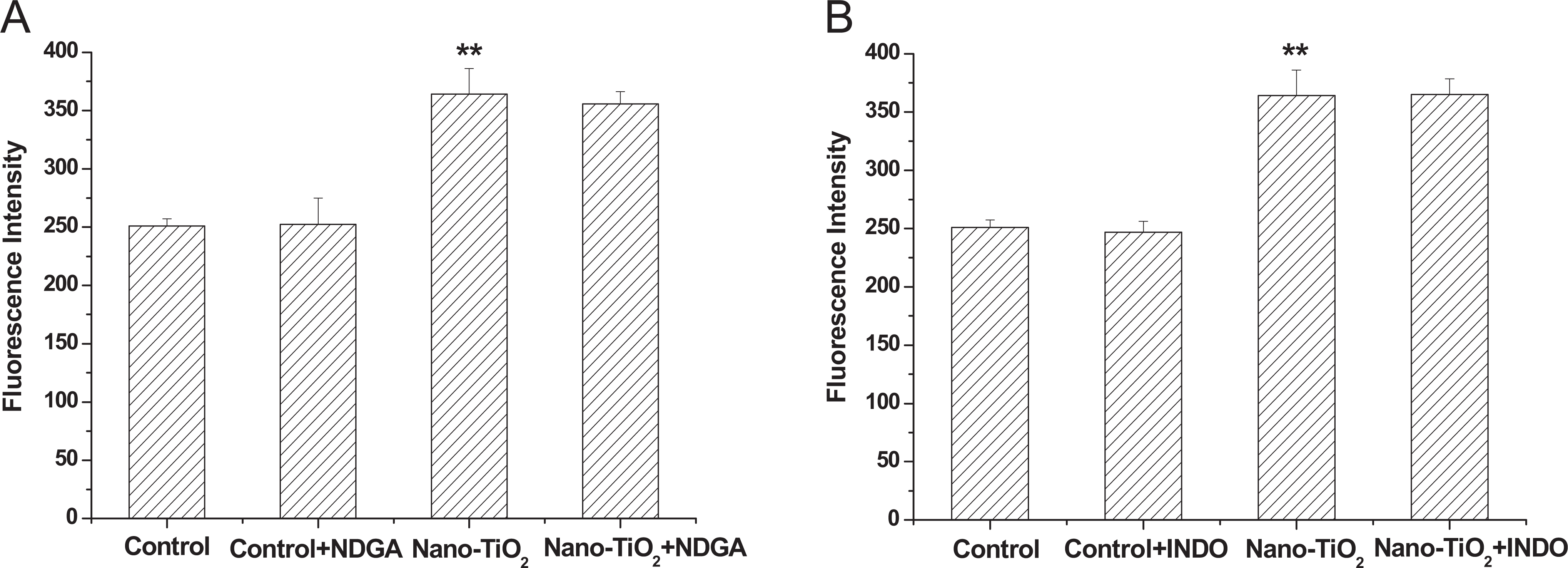
Effects of NDGA (A) and INDO (B) on intracellular ROS production induced by nano-TiO2. Cells were treated with 200 µg/mL nano-TiO2 only, or pretreated with NDGA (10 μmol/L), INDO (10 μmol/L) for 30 minutes, respectively, followed by treatment with 200 µg/mL nano-TiO2. Control was received culture medium only. All samples were irradiated with the UVA light for 1 hour and then cultured for 24 hours. Results are expressed as mean ± SEM of at least 4 different experiments. **P < 0.01 represents the comparison with the control group; # P < 0.05 and ## P < 0.01 represent the comparison with the nano-TiO2 group. INDO, indomethacin; nano-TiO2, nanosized titanium oxide; NDGA, nordihydroguaiaretic acid; ROS, reactive oxygen species; SEM, standard error of the mean.
Effects of ROT and AA on Cell Viability and MDA Content
Pretreatment with ROT (10 μmol/L) and AA (10 μmol/L) led to a significant (P < 0.01) loss of cell viability from 49.74% to 43.81%, 40.51% compared with cells treated with nano-TiO2 alone (Figure 6A), whereas the MDA content was significantly increased from 0.9 to 1.44, 1.56, respectively (Figure 6B). The results also demonstrated that cell viability and MDA content were not significantly affected after treatment with ROT (10 μmol/L) or AA (10 μmol/L) alone under the conditions tested in this study.
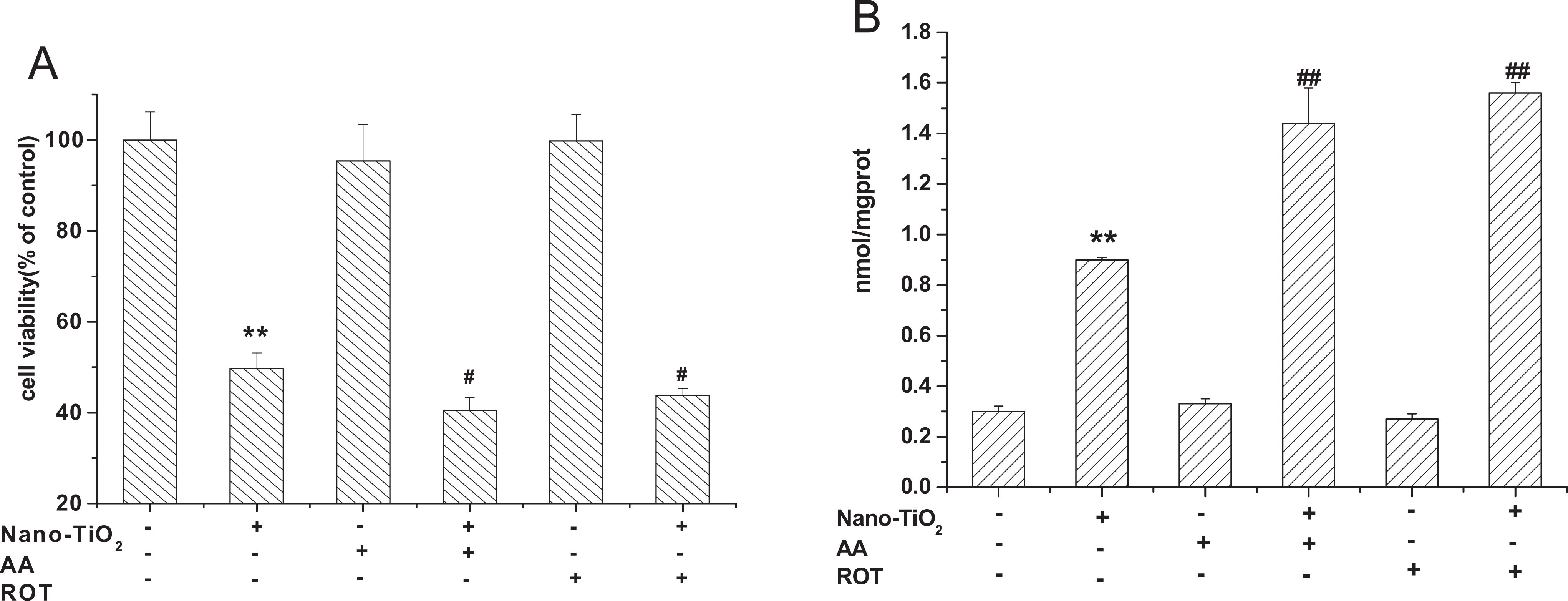
Effects of ROT and AA on cell viability (A) and MDA content (B) induced by nano-TiO2. Cells were treated with 200 µg/mL nano-TiO2 only, or pretreated with ROT (10 μmol/L), AA (10 μmol/L) for 30 minutes, respectively, followed by treatment with 200 µg/mL nano-TiO2. Control received the culture medium only. All samples were irradiated with the UVA light for 1 hour and then cultured for 24 hours. Results are expressed as mean ± SEM of at least 4 different experiments. **P < 0.01 represents the comparison with the control group; # P < 0.05 and ## P < 0.01 represent the comparison with the nano-TiO2 group. AA indicates antimycin A; MDA, malondialdehyde; nano-TiO2, nanosized titanium dioxide; ROT, rotenone; SEM, standard error of the mean.
Effects of CCCP and l -NNA on Cell Viability and MDA Content
Carbonyl cyanide m-chlorophenylhydrazone and
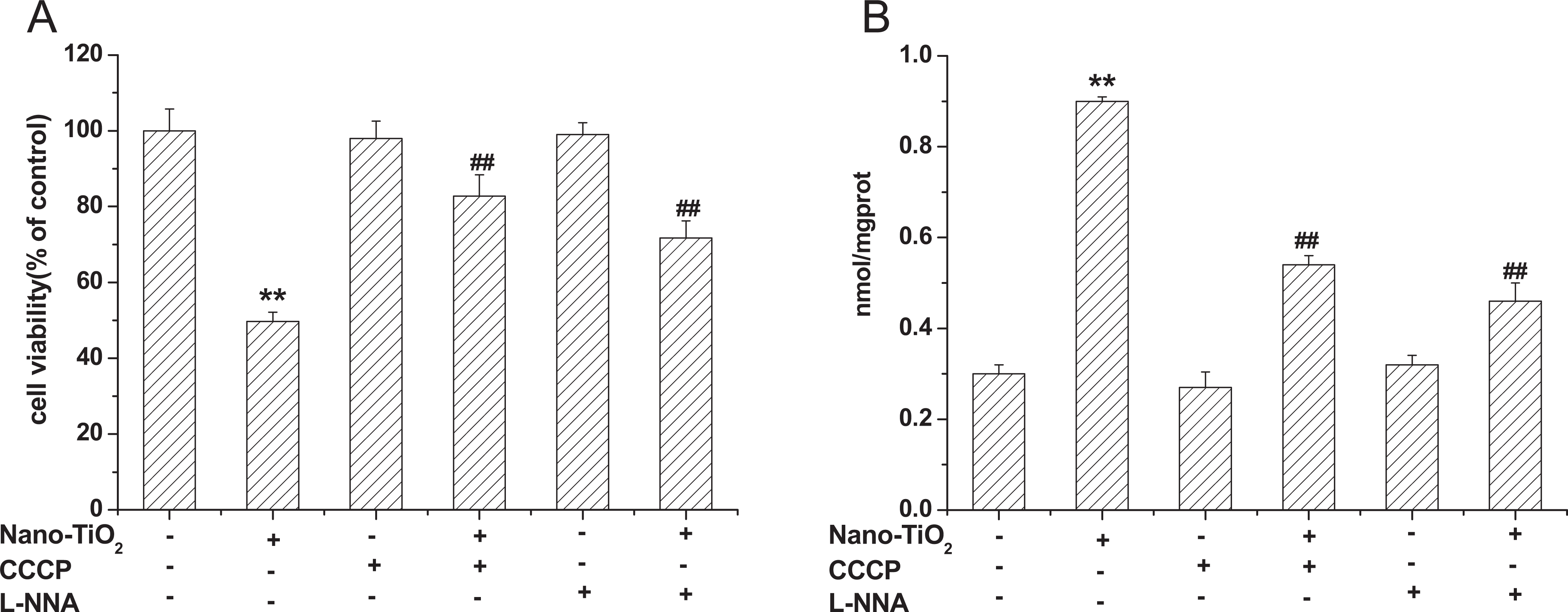
Effects of CCCP and
Opening of Permeability Transition Pore and Release of Cytochrome c
One of the major molecular mechanisms that lead to profound changes in mitochondrial functioning is caused by an alteration in MPT, which protects the mitochondria from the loss of electrochemical potential for H+ by preventing nonspecific transfer of solutes of 1,500 D. 18 To test the role of MPT in the oxidative stress induced by nano-TiO2, we took advantage of the demonstrated inhibition of MPT by CsA. Staining the cells with Evan blue showed that CsA effectively reduced the amount of cell death from 64% to 33% (Figure 8A). The localization of cytochrome c in cells was studied by Western blotting. Cytosolic proteins from cells treated with nano-TiO2 in the presence or absence of CsA were separated on 12.5% (wt/vol) SDS-PAGE and were probed with antibody against cytochrome c. 19 Cyclosporin A effectively reduced the release of cytochrome c from mitochondria. However, CsA did not completely block the release of cytochrome c into the cytosol, and a small amount of cytochrome c was found also in the cytosol (Figure 8B).
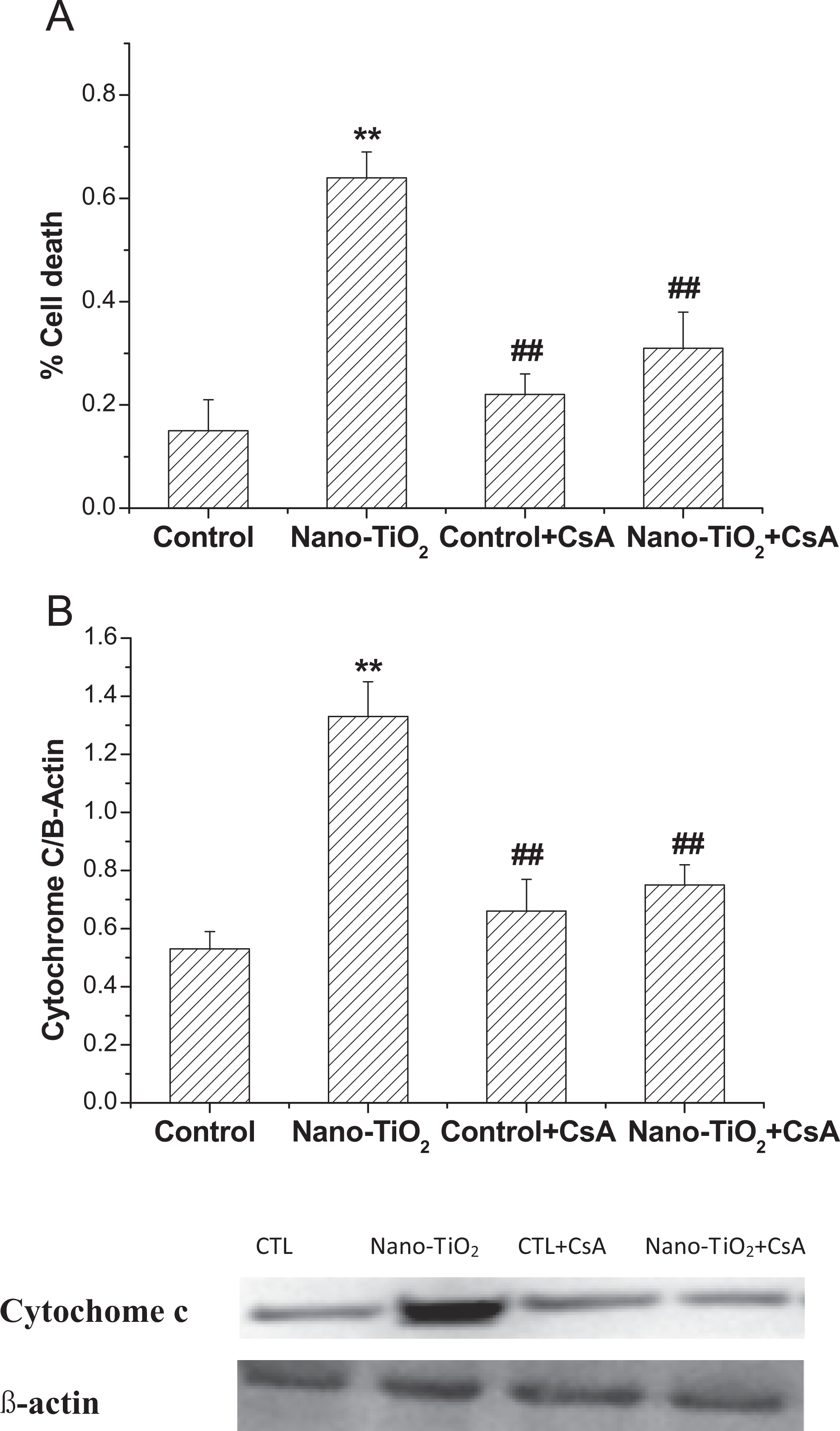
Effects of MPT pore in nano-TiO2 induced cell death (A) and cytochrome c releasing (B) in HaCaT cells. Results are expressed as mean ± SEM of at least 4 different experiments. **P < 0.01 represents the comparison with the control group; ## P < 0.01 represents the comparison with the nano-TiO2 group. MPT, mitochondrial permeability transition pore; nano-TiO2 nanosized titanium dioxide; SEM, standard error of the mean.
Discussion
Mitochondria are the main source for the generation of ROS. 1,20 More than 90% of the oxygen consumed by mammalian cells is utilized in mitochondria, and up to 4% of this oxygen is transformed into ROS. 21 Dysfunction in electron transfer through the mitochondrial respiratory chain may result in the generation of ROS species such as superoxide anion radical (O2 •), hydrogen peroxide (H2O2), and the hydroxyl radical (OH•). 22,23 In most organisms, the mitochondrial respiratory chain contains the 4 classical components of the electron transport chain (complexes I, II, III, and IV), where the electron transport couples with translocation of protons from the mitochondrial matrix to the intermembrane space. 24,25 Complexes I and III in the electron transport chain are thought to be responsible for ROS production because O2 • is formed from molecular oxygen (O2) and then dismutated to H2O2 as electrons pass through the mitochondrial respiratory chain (ie, complexes I and III). 26 Mitochondrial electron transport is inhibited by the disappearance of metallothionein in human bronchial epithelial cells following exposure to silver nitrate. Nano-TiO2 is usually present under 2 crystalline forms, anatase and rutile, that have a different photocatalytic power. The anatase crystal form is more photoreactive than the rutile one. The anatase–rutile mixture (consists of 75% anatase and 25% rutile, P25) is a widely used nano-TiO2 in the world, which is applied in water treatment, self-cleaning windows, antimicrobial coatings and paints, and chemical industry. Owing to the existence of P25 in environment, skin is inevitable to become a potential target site. In our previous study, P25-induced cytotoxicity was evaluated using HaCaT cells. It was found that nano-TiO2 (50-200 μg/mL) caused cytotoxicity through increasing ROS formation and matrix metalloproteinase collapse under UV irradiation. Ultraviolet irradiation, particularly UVA, has a capacity to generate ROS in cells; this may be due to the formation of ROS when TiO2 is exposed to UV light. 14 Notably, nano-TiO2-induced cytotoxicity was prevented by antioxidants and ROS scavengers such as N-acetylcysteine, a sulfhydryl-containing antioxidant. 27 These findings suggest that mitochondria are the main target of nano-TiO2-induced oxidative stress, and the mitochondrial respiratory chain plays an important role in the ROS generation induced by nano-TiO2. Therefore, evaluating the direct effects of mitochondrial respiratory chain in the ROS generation induced by nano-TiO2 seems to be important to better understand its mechanisms of cytotoxicity. Here, we observed the mitochondrial respiratory chain complexes I, II, III, and IV in the ROS generation by the addition of their inhibitors. Our results showed that cells pretreated with ROT (complexes I inhibitor) and AA (complexes III inhibitor) increased the ROS generation, whereas pretreatment with TTFA (complexes II inhibitor) and NaN3 (complexes IV inhibitor) did not alter the ROS generation. Addition of CCCP, an inhibitor of uncouplers of mitochondrial oxidative phosphorylation, caused significant changes in ROS production. This reconfirms the role of the mitochondrial respiratory chain in the ROS generation induced by nano-TiO2. Also, our findings about different effects of inhibitors on ROS production suggested that complexes I and III are main sites for superoxide production than complexes II and IV, which have been reported by others. 28,29 These may be that the inhibitor complexes I and III may have a role to induce an inflammatory response in primary cultured cells and complex II or IV may be not directly act on cells to induce inflammation, and mitochondrial-derived ROS production may be involved in inflammation response. In the murine microglial BV-2 cells model, the inhibition of mitochondrial respiratory chain Complex I and III indeed induced activation of p38, and NF-κB, which stimulatory effects were slightly more than complex II and the complex IV. 29
Besides mitochondrial respiratory chain, other possible sources contribute to the production of ROS, including endothelial arachidonic acid metabolism, NADPH oxidase, and NOS. 16 Thus, the results of arachidonic acid metabolism, NADPH oxidase, and NOS on ROS generation induced by nano-TiO2 were evaluated.
Arachidonic acid exerts a deleterious effect on mitochondria and ROS production. 17 The manner in which arachidonic acid metabolism via LOX and COX has no effect on ROS generation. Furthermore, the addition of DPI, a specific inhibitor of NADPH oxidase, did not show any fall in ROS generation compared with that observed with sole pretreatment with nano-TiO2, indicating that NADPH oxidase caused no significant changes in ROS generation.
Mitochondria have also been credited as a source of reactive nitrogen species derived from NO. Nitric oxide is generated enzymatically by a family of NOS, which includes neuronal NOS, eNOS, and inducible NOS.
30
All 3 NOS isoforms can also contribute to ROS production, since they have been shown to be susceptible to the “uncoupling” that leads to the formation of O2
− (rather than NO).
31
For eNOS, this process is triggered in vitro through the absence of the cofactors
In animal systems, it was shown that mitochondria are directly involved in the activation of cytosolic aspartate-specific Cys proteases (caspases) via release of cytochrome c. 37 The loss of cytochrome c from the inner mitochondrial membrane during apoptosis has been reported in many systems and is considered as a crucial regulatory step in apoptosis. At the early phases of apoptosis, the readdition of exogenous cytochrome c can markedly restore respiratory functions. 38 In Petunia (Petunia hybrida), the release of cytochrome c was found to be dispensable for the induction of a programmed cell death (PCD). 39 The increased cytochrome c following nano-TiO2 treatment in the present study could promote caspase activation and apoptotic cell death. 20 Moreover, pretreatment with CsA attenuated both the rapid increase in apoptosis and membrane potential collapse following nano-TiO2 administration. Therefore, it is possible to postulate that one or all of these events may be responsible for the toxicity and apoptosis of treatment with nano-TiO2. Our result is in agreement with other reports suggesting that nano-TiO2–induced ROS production leads to not only lipid peroxidation but also glutathione oxidation that could damage the mitochondrial membrane integrity and open the MPT pores. 27
Conclusion
In summary, we studied the effects of mitochondrial respiratory chain on the generation of ROS and cytotoxicity in HaCaT cells to extend our insights to the mechanisms of nano-TiO2 toxicity. The experimental results suggest that mitochondrial respiratory chain may be the main source of intracellular ROS production induced by nano-TiO2, the main sites were complexes I and III. The experimental results suggest that nano-TiO2 induced mitochondrial oxidative stress and subsequently caused a gradual opening of MPT pore leading to the translocation of cytochrome c to the cytosol, terminating in PCD.
Footnotes
Authors’ Note
Guozhen Liu contributed equally to this work.
Author Contributions
Chengbin Xue contributed for the conception and design and for drafting the manuscript. Xiaonan Li and Guozhen Liu contributed for the conception and for critically revising the manuscript. Wei Liu contributed for analysis and for critically revising the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Hubei Province (No. 2011CDB372), Wuhan Planning Project of Science and Technology (No. 2014070404010203). Project supported by the National Natural Science Foundation of China (Grant No. NSFC31470968) and Wuhan Middle-Aged and Young Backbone Personnel Training Project of Medicine.
