Abstract
Aim:
Concerns about the possible toxicity to environment and human health of titanium dioxide nanoparticles (TiO2 NPs) are increasing. The aim of this study was to investigate the relationship between toxicology and autophage in vitro.
Methods:
RAW 264.7 cells were exposed to five concentrations (50, 100, 200, 300, and 400 μg/mL) and two particle size of TiO2 NPs (30 and 100 nm) for 24 h.
Results:
The results showed that TiO2 NPs decreased cell viability, phagocytic rate, and phagocytic index in a concentration-dependent manner, thereby inducing autophagy. TiO2 NPs-induced autophagy was indicated by monodansyl cadaverine staining and transmission electron microscopy. TiO2 NPs-induced messenger RNA expression of autophagy-related proteins LC3 and Beclin-1 was also significantly increased compared with those of the unexposed control cells. LC3 and Beclin-1 protein expression levels were markedly increased with the increase of TiO2 NPs concentrations.
Conclusion:
These results suggest the possibility that TiO2 NPs-induced toxicology probably plays a key role in autophagy in RAW 264.7 cells, and further exhaustive research on the harmful effects of these NPs in relevant organisms is needed for their safe application.
Introduction
Nanomaterials are engineered structures with at least one dimension of 100 nm or less. 1 There has been an enormous growth in the application of titanium dioxide nanoparticles (TiO2 NPs) for photocatalyst, 2 cosmetics, 3 sterilization, pharmaceuticals, food colorants, 4 –6 and recently biomaterials. 7,8 Therefore, potential widespread exposure may occur during both manufacturing and using. However, with the increasing application of TiO2 NPs, possible toxicity to environment and human has to be addressed. TiO2 has been classified by the International Agency for Research on Cancer (IARC) as an IARC Group 2B carcinogen. 9 The toxic effects of TiO2 NPs are due to their distinct properties, greater specific surface area, and smaller size with high number per given mass which can cause their higher chemical reactivity, penetrate in the living cells, and increase production of reactive oxygen species (ROS). 10
Lungs are the main target organ of NPs. Studies suggested that TiO2 NPs could cause pulmonary inflammation and cancer in rats and mice. 11,12 A number of studies reported that TiO2 NPs can induce oxidative stress and DNA damage. 13,14 Moreover, owing to the generation of ROS, the anatase TiO2 NPs could induce mitochondrial injury in A549 cells. 15 In human and rat alveolar macrophages, the level of ROS was increased after exposure to nanosized TiO2. 16 A significant decrease in the level of glutathione was observed in rat alveolar macrophage following exposure to ultrafine TiO2. 17 Potential multiple effects of TiO2 NPs and the possible adverse effects of TiO2 NPs exposure need further clarification.
Both autophagy and apoptosis are programmed cell death, and the two are closely related. 18 The occurrence of autophagy is associated with oxidative stress, and TiO2 NPs can cause cell oxidative stress. However, there have been very few reports on the role of autophagy in the damage caused by TiO2 NPs. This work aims to explore the mechanism of TiO2 NP-related autophagy by employing murine macrophage cell line RAW 264.7.
Materials and methods
Characterization of TiO2 NPs
Pure rutile TiO2 NPs at 30 and 100 nm in diameter were purchased from Hangzhou Wan Jing New Material Co., Ltd (Hangzhou, China). The structure and morphology of the TiO2 NPs were characterized by scanning electron microscopy (SEM) with an accelerating voltage of 20 kV. The X-ray diffraction (XRD) measurement was conducted using a Rigaku D/max 2550 PC diffractometer (Rigaku Inc., Tokyo, Japan) to confirm the crystal structure of the TiO2 NPs.
TiO2 particles were sterilized by heating to 120°C for 2 h and then were suspended in sterilized phosphate-buffered saline (PBS) at 5 mg/mL and sonicated for 30 min. After sonication, to prepare the end point concentrations, Dulbecco’s modified Eagle’s medium (DMEM)-F12 medium (Hyclone, GA, USA) was transferred to test tubes and diluted with the TiO2 NP stock solution.
Cell culture and viability assay
The murine macrophage cell line RAW 264.7 was obtained from the Cell Bank of Chinese Academy of Sciences (Shanghai, China). The cells were incubated in high glucose DMEM supplemented with 10% fetal bovine serum (Hyclone, GA, USA), 100 U/mL penicillin–streptomycin in a humidified atmosphere of 5% carbon dioxide (CO2) at 37°C. The cell viability was measured by MTT (3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide) assay at 6, 12, 18, and 24 h. 19 The exponentially growing cells (about 5 × 104 cells/well) were seeded into 96-well culture plates and incubated with various concentrations of nano-TiO2 (50, 100, 200, 300, and 400 μg/mL) for 24 h. The supernatants were substituted with 90 μl fresh medium and 10 μl of MTT (1 mg/mL) solution. After 4 h of incubation at 37°C, the medium was aspirated and the formazan crystals were solubilized in 200 μl dimethyl sulfoxide. The optical density was measured spectrophotometrically (Bio-Rad 840, Hercules, California, USA) at 570 nm. Assays were performed in triplicate. The relative cell viability (%) related to control wells containing cells without NPs was calculated by [A]exposure/[A]control × 100, where [A]exposure is the absorbance of the treated sample and [A]control is the absorbance of the control sample.
Phagocytosis assays
Cultures of RAW 264.7 cells at a concentration of 3 × 105 cells/mL were seeded into 6-well plates and treated or not treated with various concentrations (50, 100, 200, 300, and 400 µg/mL) of nano-TiO2, then incubated at 37°C with an atmosphere of 5% CO2. At the end of 24 h of incubation, the adherent cell monolayer was washed with PBS, and sorted fluorescent microsphere was added. After 2 h of incubation in dark place, the plate was washed 5× with PBS and fixed with 4% paraformaldehyde. Fluorescent phagocytosis was observed by confocal microscopy (TCSSP2; Leica Microsystems, Wetzlar, Germany) to acquire the phagocytic rate (% of macrophages containing at least one fluorescent microsphere) and the phagocytic index (the mean number of phagocytosed fluorescent microsphere).
TEM analysis of cellular organelles
The TiO2 NP-treated cells were fixed in 2.5% glutaraldehyde with 1% osmium tetroxide (OsO4) buffer at 4°C for 6 h. The fixed cells were dehydrated in serially diluted ethanol (30, 50, 70, 80, 90, and 100%). After dehydration, the samples were infiltrated with a 1:1 mixture of propylene oxide and epon at 70°C overnight. Ultrathin sections (about 50 mm) were sliced, mounted on a copper grid, stained with uranyl acetate and lead citrate, and monitored using a JEM 1010 Transmission electron microscope (TEM; JEOL, Tokyo, Japan).
Detection of autophagic vacuoles by staining of cells with MDC
The development of autophagic vacuoles was studied at five concentrations (50, 100, 200, 300, and 400 µg/mL) by staining cells with 50 μmol/L monodansyl cadaverine (MDC). Treated cells were washed with PBS (pH 7.2) to remove the culture medium. Then, cells were incubated with 50 μmol/L MDC in PBS at 37°C for 30 min and were washed four times with PBS again, observed by fluorescence microscopy immediately (excitation wavelength = 338 nm; emission wavelength = 500 nm).
Quantitative analysis of MAP-LC3 and Beclin-1 expression
RAW 264.7 cells were exposed to 50, 100, 200, 300, and 400 µg/mL of nano-TiO2 suspension for 24 h. Then, RNA was extracted from cells using MiniBEST Universal RNA Extraction Kit (Takara, Kyoto, Japan). For each sample, RNA (100 ng) was reverse-transcribed using Prime Script TMRT reagent Kit with gDNA Eraser (Code no. RR047A, Takara), according to the manufacturer’s instructions. β-Actin (control) was used as normalization for total messenger RNA (mRNA) input and to confirm efficiency of complementary DNA (cDNA) synthesis. A relative quantitative real-time polymerase chain reaction analysis was performed on aliquots of the cDNA production, using SYBR Premix EX Taq™ II (Code no. RR820A, Takara) in the Bio-Rad-IQ5 Multicolor Real-time Detection System, following the manufacturer’s instructions. The primers were designed on the basis of published mRNA sequences of MAP1-LC3, Beclin-1, and β-actin. The primers were purchased from Sangon Biotech (Shanghai, China) (Table 1).
RT-PCR primers used in the gene expression analysis.

MAP-LC3 and Beclin-1 protein expression
Incubated cells at various concentrations (50, 100, 200, 300, and 400 µg/mL) of nano-TiO2 for 24 h were harvested for repeated freezing and thawing. The expression of MAP-LC3 and Beclin-1 was detected by an enzyme-linked immunosorbent assay (ELISA) kit (Andygene, Richardson). The absorbance was read at 450 nm using a microtiter plate reader (Bio-Rad). The levels of MAP-LC3 and Beclin-1 were calculated with reference to standard curves prepared with purified MAP-LC3 and Beclin-1.
Statistical analysis
Data were analyzed with SPSS 17.0 for Windows (SPSS Inc., Chicago, IL, USA). Statistical analyses were performed by either one-way analysis of variance or Student’s t-test when applicable to evaluate differences between the control and the test sample. Each experiment was repeated at least three times. Significance level was set at p < 0.05, unless otherwise mentioned.
Results
Characterization of TiO2 NPs
Rutile NPs displayed a typical rod-like crystal structure. TiO2 NPs rutile at 30 nm in diameter displayed a size approximately between 30 nm and 50 nm. TiO2 NPs rutile at 100 nm in diameter displayed a size approximately between 50 nm and 100 nm in SEM analysis (Figure 1). In addition, the XRD pattern showed that the TiO2 NPs contained a pure rutile form (Figure 2). No significant difference was observed with the manufacturer’s specifications and previous reports on the characterization of the shape of the NPs. 20

SEM image of TiO2 NPs shape and size: (a) 30 nm particle size and (b) 100 nm particle size. SEM: scanning electron microscopy; TiO2 NPs: titanium dioxide nanoparticles.

XRD data of titanium dioxide nanoparticles (TiO2 NPs). XRD data of rutile form of TiO2 NPs.XRD: X-ray diffraction; TiO2 NPs: titanium dioxide nanoparticles. (a): 30nm; (b): 100nm.
Effects of TiO2 NPs on RAW 264.7 cells viability and phagocytosis function
Cell viability has been inhibited strongly in a dose- and time-dependent manner with 30 nm or 100 nm TiO2 NPs (Figure 3(a) and (b)). Cell viability was significantly decreased (p < 0.05), with decreases of 61.8% and 55.26% for TiO2 rutile at 30 nm in diameter and TiO2 rutile at 100 nm in diameter in the treatment concentration of 400 µg/mL, respectively, when compared with the control. TiO2 NPs at 30 nm in diameter treatments at 50, 100, 200, 300, and 400 µg/mL for 24 h was selected to conduct the subsequent experiments.

Cell viability monitored by the MTT assay following 6, 12, 18, and 24 h exposure of TiO2 NPs at different concentrations (50, 100, 200, 300, and 400 μg/mL). Data are shown as % viability compared with the untreated control and are presented as means ± SD of three separate experiments. *p < 0.05: vs. control. (a) 30 nm TiO2 NPs and (b) 100 nm TiO2 NPs. TiO2 NPs: titanium dioxide nanoparticles.
Both 30 and 100 nm TiO2 NPs significantly decreased the phagocytic rate and phagocytic index compared with the control group (Table 2; p < 0.05) in a dose-dependent manner.
The results of phagocytic ability in RAW264.7 cells after 24 h of exposure to PM2.5.a

PM: particulate matter with diameter <2.5 µm; SEM: standard error of mean.
a Data represent the means ± SEM from three separate experiments.
bp < 0.05: treatments significantly different from the untreated control are presented.
cp < 0.01: treatments significantly different from the untreated control are presented.
TiO2 NPs induce autophagy in murine macrophage cells
To determine whether autophagy was induced in TiO2 NP-treated cells, the morphology of RAW 264.7 cells treated with or without TiO2 NPs by TEM was observed. Untreated cells showed normal cytoplasm, organelles, and nucleus shape (Figure 4). The exposure of cells to 400 µg/mL TiO2 NPs for 24 h resulted in the appearance of autophagocytic vacuoles containing extensively degraded cellular material or membranous structures. TiO2 NPs-treated cells exhibited more MDC-labeled particles compared with untreated cells (Figure 5). The results demonstrated that the TiO2 NPs induce autophage in murine macrophage cells.
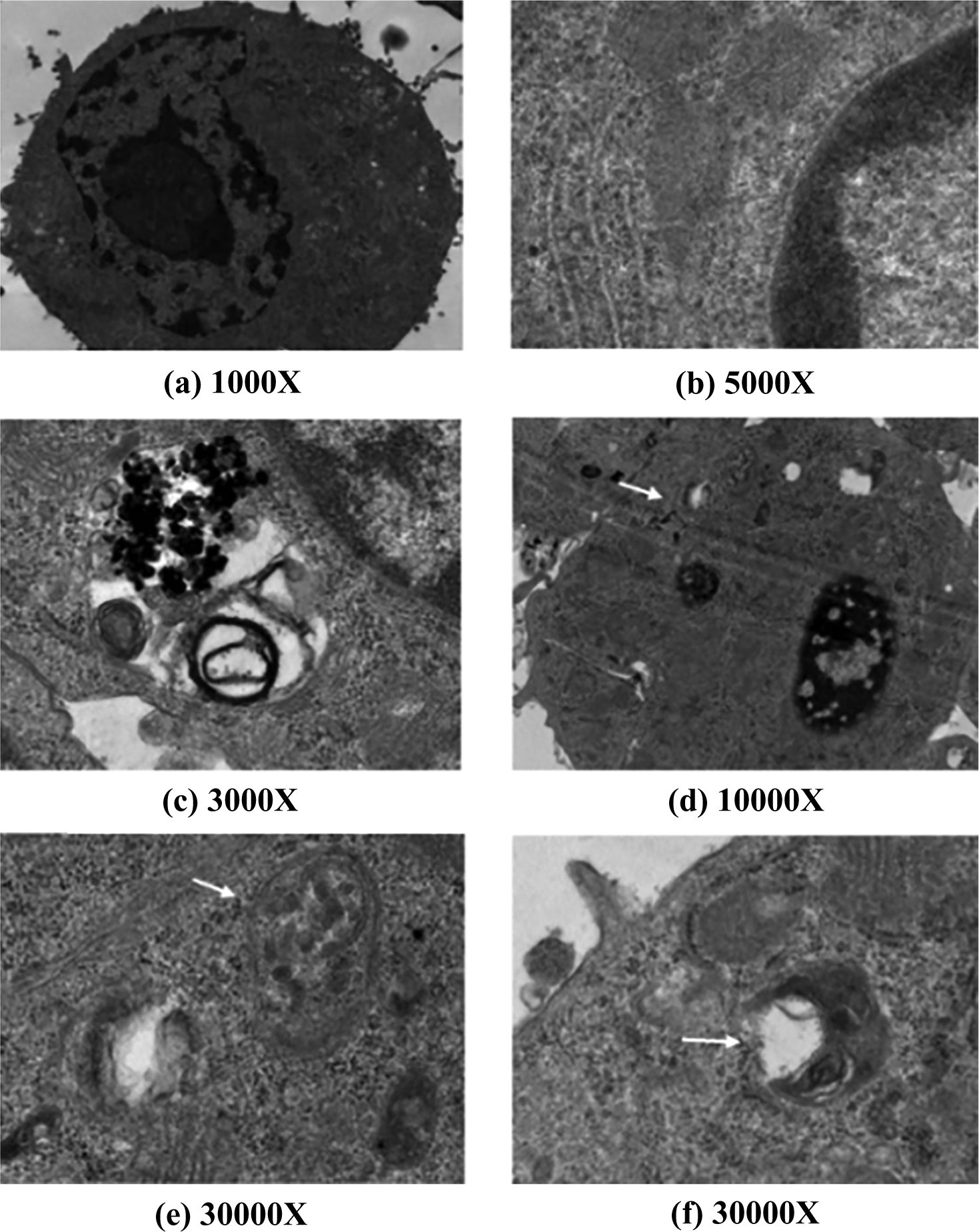
TEM images of autophagosomes in RAW 264.7 cells. Ultrastructure confirms autophagic vacuole (white arrow) generation in RAW 264.7 cells. Magnification of ultra-micrograph has been mentioned in each corresponding figure. (a and b) untreated RAW 264.7 cells. (c to f 264.7 cells treated with 100 μg/mL 30 nm TiO2 NPs for 24 h. TEM: transmission electron microscope; TiO2 NPs: titanium dioxide nanoparticles.
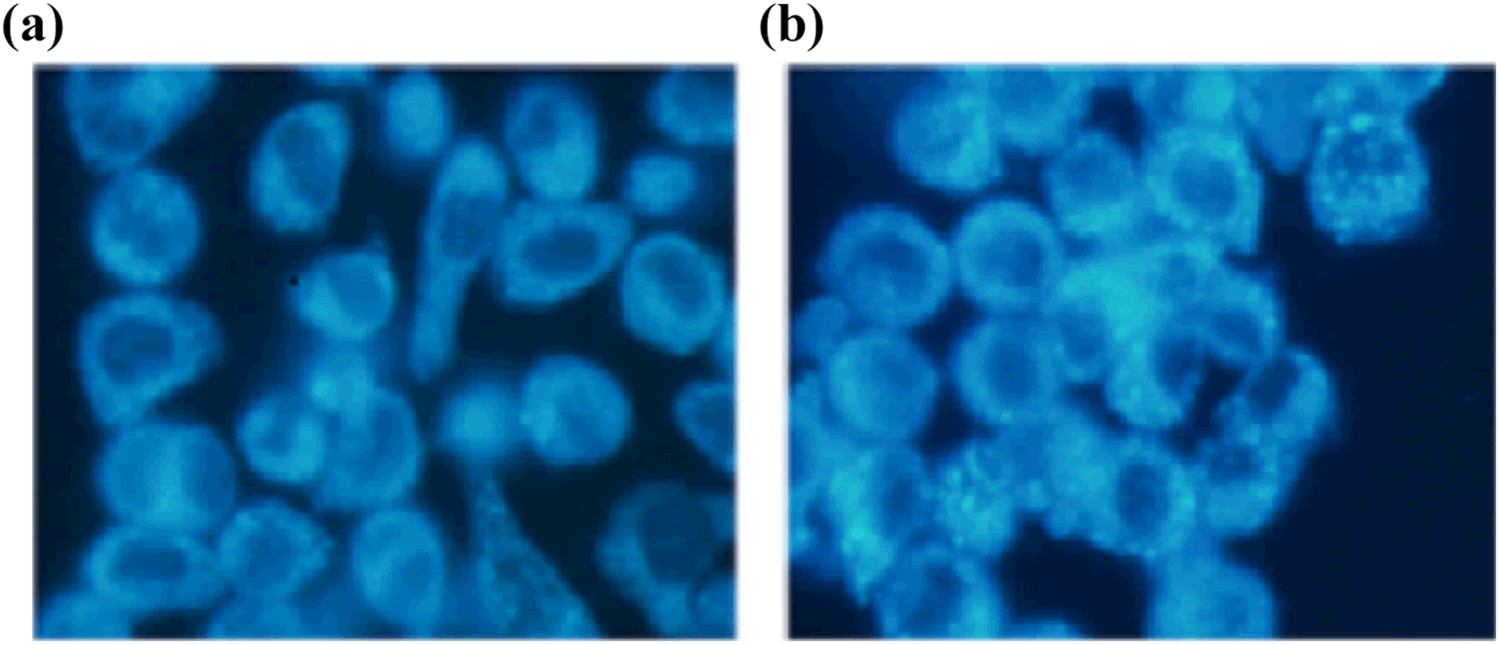
Identification of autophagic vacuoles using MDC staining. RAW 264.7 cells were stained with MDC (0.05 mM) which specifically stains autophagic vacuoles, confirms autophagic vacuolation at 24 h of exposure to 30 nm TiO2 NPs. (a) Control cells. (b) cells exposed to 100 μg/mL 30 nm TiO2 NPs for 24 h. MDC: monodansyl cadaverine; TiO2 NPs: titanium dioxide nanoparticles.
Increased expression of autophagic markers Beclin-1 and MAP-LC3 in RAW 264.7 cells
The expression levels of Beclin-1 and MAP-LC3 mRNA in exposed cells and unexposed cells are shown in Figure 6. The mRNA expression levels of MAP-LC3 were rising gradually with the increase of concentration. However, there was significance only in 300 and 400 µg/mL concentrations of TiO2 NPs (p < 0.05) (Figure 6(a)). Beclin-1 mRNA in cells exposed to 100, 200, 300, and 400 µg/mL concentrations of TiO2 NPs was significantly higher than in unexposed cells (p < 0.05) (Figure 6(b)).

The mRNA expression of autophagy-related genes. Cell lysates were used to perform real-time polymerase chain reaction experiments and evaluate the mRNA expression levels of (a) MAP-LC3 and (b) Beclin-1 after 24 h exposure to TiO2 NPs at the concentration of 50, 100, 200, 300, and 400 µg/mL. Data are means ± SD of five independent experiments. *p < 0.05: vs. control. mRNA: messenger RNA; TiO2 NPs: titanium dioxide nanoparticles; SD: standard deviation
In the study on concentration-dependent effects, all of the doses but 50 µg/mL were significantly enhanced in MAP-LC3 production (Figure 7(a)). In the experiment of Beclin-1 expression, lower doses were not significantly enhanced. However, 200, 300, and 400 µg/mL concentrations were increased significantly (Figure 7(b)).

The protein levels of (a) MAP-LC3 and (b) Beclin-1 in RAW264.7 cells after exposed to TiO2 NPs for 24 h at the concentration of 50, 100, 200, 300, and 400 µg/mL of TiO2 NPs. *p < 0.05: vs. control. TiO2 NPs: titanium dioxide nanoparticles.
Discussion
With the increasing applications of TiO2 NPs in various commercial products, concerns about their risk to human health and the unfriendly environment are growing. 7 There were reports of pulmonary inflammation and cancer in rats and mice. 11,12 However, the underlying mechanisms of these adverse effects have not been extensively characterized. 21,22 The current study showed that TiO2 NPs inhibited cell viability and decreased cell phagocytosis in RAW 264.7 cells. TiO2 NPs triggered autophage and increased the expression levels of autophage-related gene Beclin-1 and MAP-LC3.
The study demonstrated also that the phagocytic ability of RAW 264.7 macrophages was significantly impaired in 30 or 100 nm TiO2 NPs with increasing concentration. Of greater interest was the potential for 30 nm more than 100 nm TiO2 NPs. The impairment of rat alveolar macrophage phagocytosis and prolonged particle clearance after exposure to increasing doses (5, 50, and 250 mg/m3) of TiO2 has been reported, that is, the potential for 29-nm TiO2 NPs to impair phagocytosis more than 250 nm TiO2 NPs. 23,24 The study has confirmed with the previous reports, indicating the decrease of phagocytic ability of RAW 264.7 macrophages after treatment with TiO2 NPs. The structure and integrity of the cells has consequently resulted in the impairment of the cell phagocytosis. Furthermore, this study showed that, at an equal mass, the smaller particles caused more serious damage to the cells than the larger particles. Although the real reason has not been studied, the smaller particle size of TiO2 NPs may adhere or penetrate the cell membrane more easily.
Autophagy is a catabolic process involving the degradation of cytoplasmic material, playing an important role in cellular homeostasis by the elimination of damaged organelle and aggregated proteins. 25,26 It is an important response mechanism under different kinds of cellular stress, 27 generally promoting counteracting apoptosis cell death and cell survival. 28 The levels of cellular LC3-II correspond to the number of autophagosomes, and thus, it is a suitable and well-characterized marker to assess autophagy. The levels of LC3 have showed a significant difference in 300 and 400 µg/mL concentrations of TiO2 NPs. This could be due to the increased autophagic initiation and decreased autophagic degradation. 29,30 Beclin-1 levels appear to be one of the critical factors that affect the induction of autophagy. 31 The Beclin-1 protein is also an important convergence point of autophagy and apoptosis; it interacts with antiapoptotic Bcl-2-like proteins. 32 The study of Beclin-1 has showed that the 200, 300, and 400 µg/mL concentrations affected autophagy significantly.
Many studies have reported that TiO2 NPs can induce toxic response by increasing the generation of malondialdehyde and ROS; downregulating Bcl-2; triggering mitochondrial alteration, inflammatory response and DNA damage, and subsequent apoptotic cell death. 33,34 Lopes et al. 35 indicated that exposure to TiO2 NPs led to a dose-dependent increase in the autophagic effect under noncytotoxic conditions in human HaCaT cells, but the high-dose treatment appeared to impair the degradation of autophagic substrates more than the low-dose did over time. Autophagy can act in both cytoprotective and cell death functions. It plays a vital role in maintaining cell structure, functional integrity, and metabolism. Although more in-depth studies are needed to clarify the mechanism, our study demonstrated that the TiO2 NPs induce autophage in murine macrophage cells by TEM and MDC staining.
Conclusions
In summary, the study has demonstrated that TiO2 NPs reduced cellular ability in RAW 264.7 macrophage cell line. It also showed that TiO2 NP decreased cell viability, phagocytic rate, and phagocytic index in a concentration-dependent manner, thereby inducing autophagy. The results have supported strongly that TiO2 NPs can induce RAW 264.7 cell toxicology and autophage.
Footnotes
Authors' contributions
All authors contributed to the writing of the manuscript and gave approval to the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
