Abstract
Multiwalled carbon nanotubes (MWCNTs) have been explored in pharmaceutical applications such as tumor targeting and delivery of drugs, in which MWCNTs are given through intravenous injection. However, the biosafety of MWCNTs is of concern for such application. Therefore, in the current study, we used a fatty liver model to investigate the possible toxicity of MWCNTs to the liver, as MWCNTs were retained mainly in the liver of mice after intravenous injection. Male Sprague Dawley rats were used to generate the fatty liver model, and the effects of intravenous administration of MWCNTs on fatty liver were studied. Hematoxylin and eosin staining for hepatocellular anatomy and Masson trichrome staining for hepatic fibrosis were conducted. Histologically, MWCNTs aggravated steatohepatitis with higher nonalcoholic fatty liver disease scores. Analysis of liver injury markers indicated that MWCNTs administration resulted in chronic hepatitis, along with increased liver fat and altered liver oxidation, including the increase of P6 protein and the depletion of glutathione. In conclusion, our results suggest that MWCNTs can aggravate nonalcoholic steatohepatitis in Sprague Dawley rats, and oxidative injury may be involved in this process.
Keywords
Introduction
Because of the unique structure and physicochemical properties, multiwalled carbon nanotubes (MWCNTs) have been currently used in electrical, mechanical, and optical industries, as well as in biomedical and pharmaceutical area. 1 However, the biosafety of MWCNTs remains an issue, and to date, there have been many studies evaluating the toxic effects of MWCNTs, especially on the respiratory system, as MWCNTs can be absorbed through the airway. Nonetheless, in recent years, MWCNTs have been used in biological applications such as substrates for neural growth, bone cement, or biosensors 2 ; they have also been used as vehicle for tumor targeting 3 and delivery of drugs, proteins, peptides, or nucleic acids. 4,5 For such applications, different exposure approaches other than respiratory inhalation, that is, intravenous injection, are preferred. Therefore, it is important to evaluate the potential toxic effects of MWCNTs by intravenous exposure. Some preliminary studies have shown that MWCNTs can be trapped by the reticuloendothelial system and then retained mainly in the liver of mice after intravenous injection. 6 Thus, the liver seems to be the target organ for MWCNTs through intravenous exposure. In the study by Ji et al, the hepatic toxicity of MWCNTs to Kunming mice through intravenous injection was also evaluated. The results showed that a high dose of Tween 80-dispersed MWCNTs could induce hepatic toxicity in mice, whereas acid-oxidized MWCNTs exhibited less toxicity.
Nonalcoholic fatty liver disease is a chronic clinical disorder related to metabolic syndrome. The clinicopathologic spectrum of nonalcoholic fatty liver disease ranges from simple steatosis to nonalcoholic steatohepatitis (NASH), fibrosis, cirrhosis, and even hepatocellular carcinoma. 7,8 High-fat diet can lead to obesity and insulin resistance, both of which are well-known risk factors for nonalcoholic fatty liver disease. Triglycerides are the major storing form of hepatocellular lipids, whereas other lipid metabolites such as cholesterol (CHO), phospholipids, and free fatty acids (FFAs) also play certain roles in the progression of nonalcoholic fatty liver disease. It is estimated that nonalcoholic fatty liver disease occurs in 14% to 30% of the total human population. 9 Consequently, if MWCNTs were used intravenously, the toxic effects of MWCNTs on patients with nonalcoholic fatty liver disease are of great concern, as livers in these patients have already had certain damage. Therefore, in the current study, we used a high-fat diet-induced nonalcoholic fatty liver disease Sprague Dawley (SD) rat model to examine the effects of MWCNTs exposure by intravenous injection on the development of nonalcoholic fatty liver disease. As reported here, it was found that intravenous administration of MWCNTs aggravated high-fat diet-induced nonalcoholic fatty liver disease.
Materials and Methods
Preparation of MWCNTs
Multiwalled carbon nanotubes were provided by Dr F. Chen from the Lawrence Berkeley National Laboratory, Berkeley, California. The characteristics and ultrasonic processing of MWCNTs have been described by Guo et al. 10 In short, synthesized with the chemical vapor deposition method, the physicochemical characteristics of MWCNTs are: purity ≥95%; average diameter, 30 nm; length ≥1 µm; metal impurities, nickel 3.38%, yttrium 0.67%, and iron 0.13%. With respect to the condition of ultrasonicating the MWCNTs before each injection, MWCNTs were suspended in normal saline containing 1% Tween 80 before sonicating for 16 cycles of 5-second ultrasonication using an ultrasonic disruptor (200 W, JY 92-IIN; Scientz, Ningbo, China) and a 6-second pause in the ice bath. Transmission electron microscopy was used to identify the diameters of MWCNTs in the suspension (Figure 1).
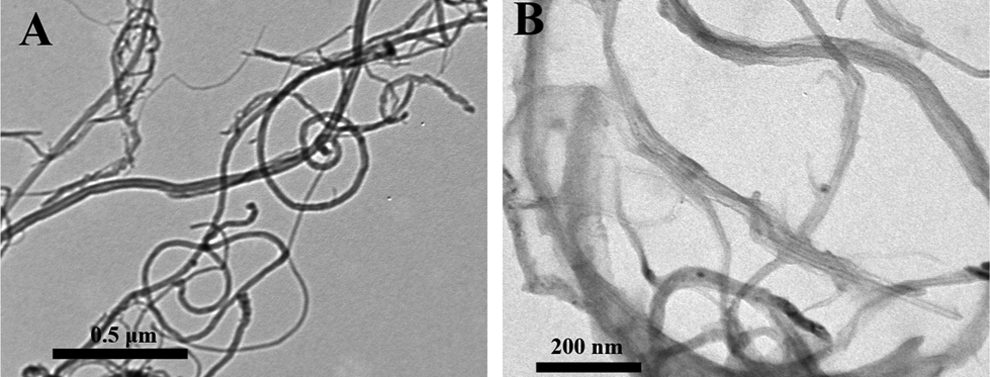
Transmission electron microscopy images of MWCNTs used in this experiment. A, Scale bar 0.5 µm and (B) scale bar 200 nm. MWCNTs indicates multiwalled carbon nanotubes.
Establishment of the Fatty Liver Model and MWCNT Exposure
The procedure for feeding high-lipid diet and MWCNTs administration has been described previously. 11 Fatty liver was developed in SD rats by feeding high-lipid chow for 4 months following administration of vitamin D3 (VD3; 700,000 U/kg) via gavage for 3 days. The high-lipid chow was a mixture of 2% cholesterol, 0.5% bile salt, 7% lard (all purchased from Sigma), Sigma-Aldrich (St.Louis, MO) and 90.5% basic diet. We checked the gross pathological specimen of the liver 4 months after the beginning of feeding to determine whether the fatty liver model has been established.
Sixty male SD rats (weighted 180-200 g) were obtained from the Experimental Animal Center in Zhejiang University, China. The animals were allowed to acclimatize to the laboratory environment for 4 days prior to the study. Rats were randomly assigned to 6 groups: group 1 (normal untreated group), group 2 (model group), and groups 3 and 4 (MWCNTs 0-, 50-, 100-, and 200-µg/kg group). Animals in group 1 were gavaged with normal saline in the first 3 days and received a regular chow for the whole study period, meanwhile, the other groups were gavaged with VD3 in the first 3 days and then high-lipid chow as described previously. Multiwalled carbon nanotube groups were administrated with a dose of 0-, 50-, 100-, and 200-µg/kg MWCNT suspension in normal saline containing 1% Tween 80 body weight, intravenously, respectively, twice per week. Four months later, all animals were fasted overnight before being anesthetized with pentobarbital. Blood samples were drawn from the femoral artery and refrigerated at −80°C. Liver was harvested, the left lobe was fixed with 10% neutral-buffered formalin (NBF), and the right lobe was rapidly frozen in liquid nitrogen. All animal experiments were conducted in compliance with the standards of the ethics committee of Zhejiang Province for the Care and Use of Laboratory Animals.
Measurement of Serum Aspartate Aminotransferase, γ-Glutamyl Transferase, and Glutathione peroxidase
Aspartate aminotransferase (AST), γ-glutamyl transferase (GGT), and glutathione peroxidase (GSH-Px) were measured by colorimetric methods according to the manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Jiangsu, China). The liver index was calculated as follows: liver weight (g)/body weight (g) × 100.
Measurement of Total Superoxide Dismutase, Malondialdehyde, Triglyceride, and FFA in Liver Tissue
The hepatic lipid was extracted from the tissue homogenate following the procedure provided by the manufacturer (Applygen Technologies Incorporation, China). The prepared sample was then centrifuged at 3,000 rpm for 15 minutes, and the obtained substratum was used for the measurement of triglyceride (TG) and FFA with the colorimetric methods. For the measurement of total superoxide dismutase (T-SOD) and malondialdehyde (MDA), liver tissue was put in ice-cold saline (10%, wt/vol) and homogenated. After centrifuging at 3,000 rpm for 15 minutes, the supernatant was measured with corresponding assay kits (Nanjing Jiancheng Bioengineering Institute, Jiangsu, China).
Microscopic Examination of Hematoxylin and Eosin–Stained Liver Sections
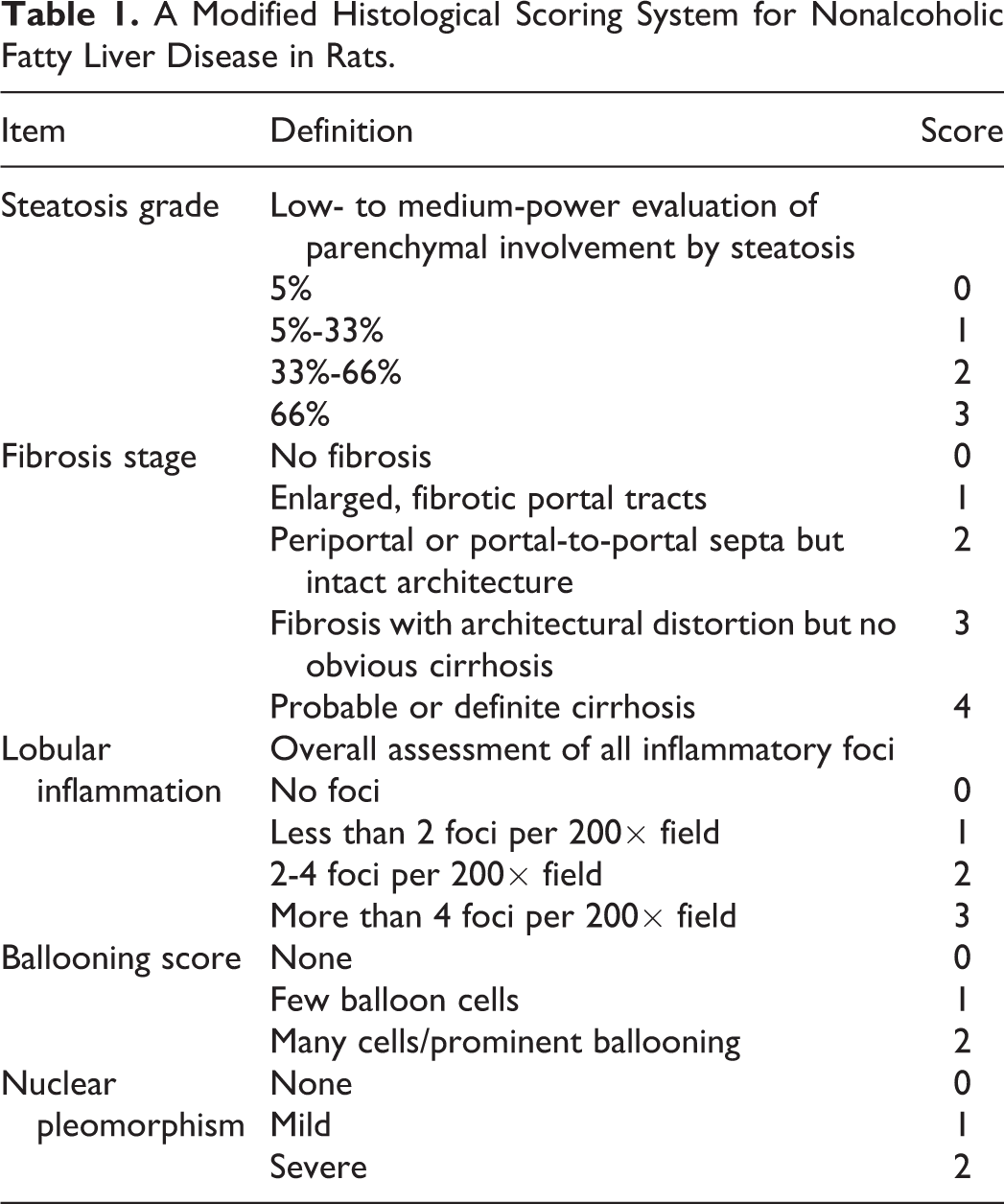
For histopathological studies, rat liver samples were fixed in 10% NBF and embedded in paraffin after proper fixation. The paraffin-embedded tissue sample was sectioned at 5-µm thickness, and the sections were stained with hematoxylin and eosin. In order to classify the level of steatohepatitis, we designed a histological scoring system for nonalcoholic fatty liver disease in rats (Table 1), based on the Roenigk classification (Roenigk, 1998) 12 and the nonalcoholic fatty liver disease activity score criteria proposed by the Pathology Committee of the NASH (Psoriasis Task Force, 2005). 13 The conventional nonalcoholic fatty liver disease scoring systems are mainly applied to human, while no standard criteria for mice exist. Thus, we combined the existing nonalcoholic fatty liver disease grading standard 14 –16 with pathological classification criteria for rodents 17 to establish a new scoring system, which is shown in Table 1.
A Modified Histological Scoring System for Nonalcoholic Fatty Liver Disease in Rats.
Immunohistochemistry
Paraffin-embedded liver tissues were cut into 5-µm-thick sections. Representative sections were deparaffinized, rehydrated, and treated with 3 mol/L urea in a pressure cooker for 10 minutes for antigen retrieval. Sections were incubated with antibodies to peroxiredoxin 6 (P6, 1:1,500, Cat# ab59543; Abcam), LC3 (Microtubule-associated protein 1 light chain 3, 1:1,000, Cat# L7543; Sigma), or p62 Sequestosome 1 (1:2,000, Cat# P0067; Sigma) overnight at 4°C, before being washed with 0.01 mol/L phosphate-buffered saline for 3 times, each 3 minutes. After incubating with corresponding secondary antibody, the immunoreactivity was visualized with the Vectastain ABC kit using 3-amino-9-ethylcarbazole or 3,3-diaminobenzidine tetrahydrochloride substrate as the final chromogen (Vector Laboratories, CA, USA). Sections were counterstained with Vector hematoxylin. Negative controls included sections stained after omission of the first antibody or after incubation with mouse immunoglobulin as the primary antibody. Finally, they were observed and photographed with a light microscope (BX53; Olympus, Tokyo, Japan) with a computer-assisted video camera (DP80; Olympus). Pictures were analyzed with integrated optical density, which was calculated with the software Image Pro Plus 6.0.
Statistical Analysis
The data were expressed as mean ± standard deviation. The 1-way analysis of variance followed by a post hoc least significant difference test was used for comparisons between groups. The statistical analysis was conducted using SPSS 16.0; P < 0.05 was considered statistically significant. GraphPad Prism 5.01 was used for statistical graph preparation.
Results
Multiwalled Carbon Nanotubes Treatment Aggravates Steatohepatitis: Macroscopic and Histologic Analysis
First, we wanted to determine whether fatty liver model had been successfully established. For the normal diet group, the livers were soft and red in color with a smooth capsule. In contrast, the livers from the model group were firm, swollen, and had rounded edges (Figure S1A). Histologically, rats in the model group had fatty livers, with indistinct lobules, hepatocellular vacuolar degeneration, and pyknosis, whereas rats in the normal group had a liver parenchyma that was well structured with distinguishable lobules and hepatic cords extending radially around the central veins (Figure S1B and C). In addition, the liver index from the model group was higher than the normal diet group (Figure S1D). Both the 2 fatty liver indexes, TG and CHO, from the model group were increased compared with the normal group (Figure S1E and F). Thus, it was concluded that the fatty liver model had been well established. Next, we examined liver indexes and histologic condition including liver fibrosis to determine the effect of MWCNTs on fatty liver.
For the liver index, no significant difference was observed between the MWCNTs-treated groups (50-200 µg/kg) and the solvent control group (Figure 2B). Still, compared with the solvent control group, livers from MWCNTs-treated groups were macroscopically darker in color, especially the 200-µg/kg MWCNTs group (Figure 2A).
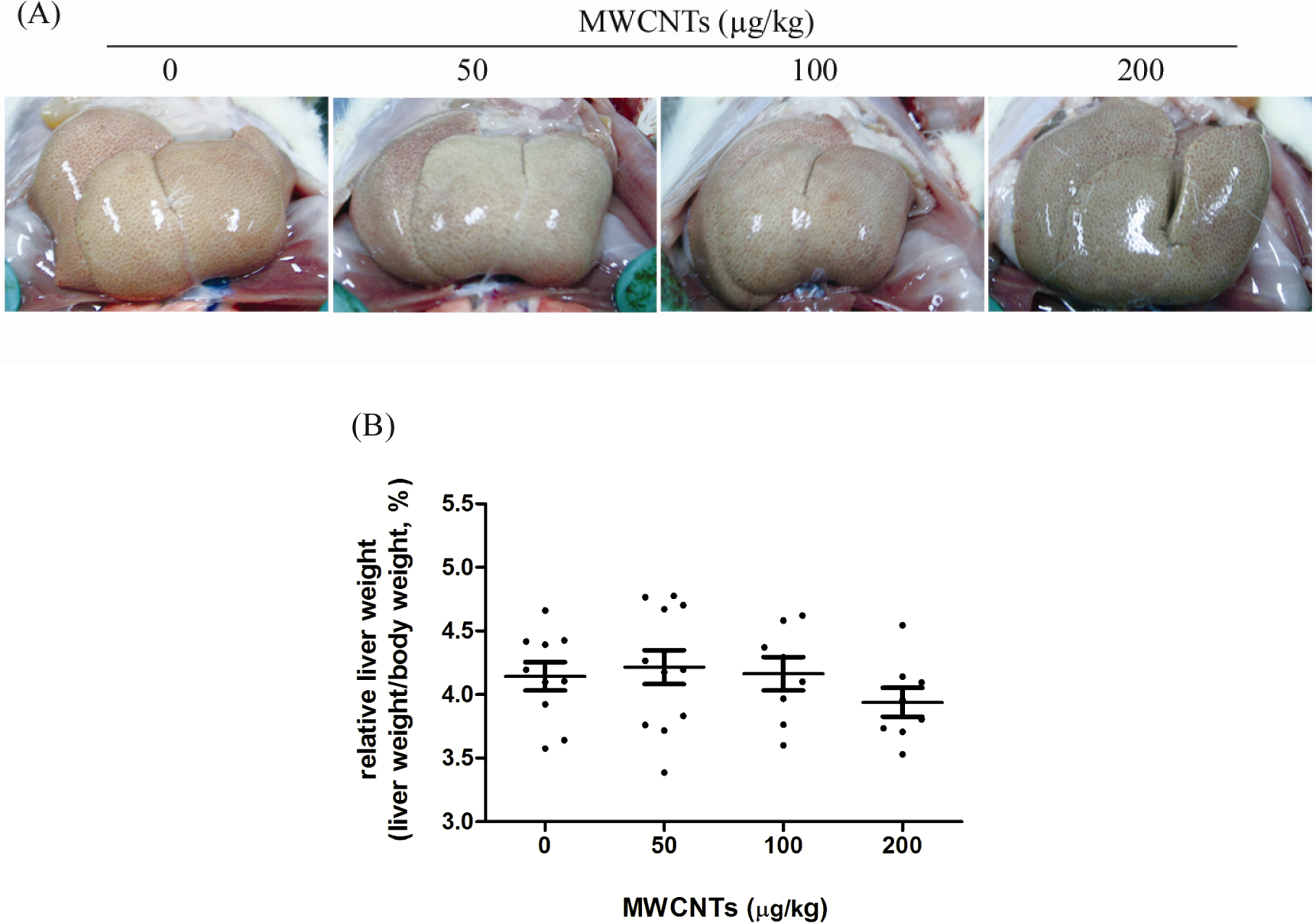
The effects of MWCNTs on the appearance of the liver and liver indexes. A, Representative images of the livers after various treatments. Compared with the solvent control group, livers from MWCNTs-treated groups were darker in color and blunt in edge, especially for the 200-µg/kg MWCNTs group. B, Liver indexes of rats after 4-month exposure. Values were represented as means ± SD, n = 10. MWCNTs indicates multiwalled carbon nanotubes; SD, standard deviation.
Furthermore, with 50 to 200 µg/kg MWCNTs administration, the lobules were indistinct and hepatocytes were swollen (Figure 3B). Steatohepatitis was present in the 100- to 200-µg/kg MWCNTs group, in which individual hepatocyte necrosis (red arrows in Figure 3B) and inflammatory cells (green arrows in Figure 3B) were observed, especially in the portal and centrilobular regions. In addition, we noticed that after the administration, MWCNTs were retained in the liver parenchyma, especially in the 100- to 200-µg/kg MWCNTs group, as shown with yellow arrows in Figure 3C. Specifically, its (describe appearance, color, etc) nanoparticles were engulfed by Kupffer cells (yellow arrows in Figure 3B).
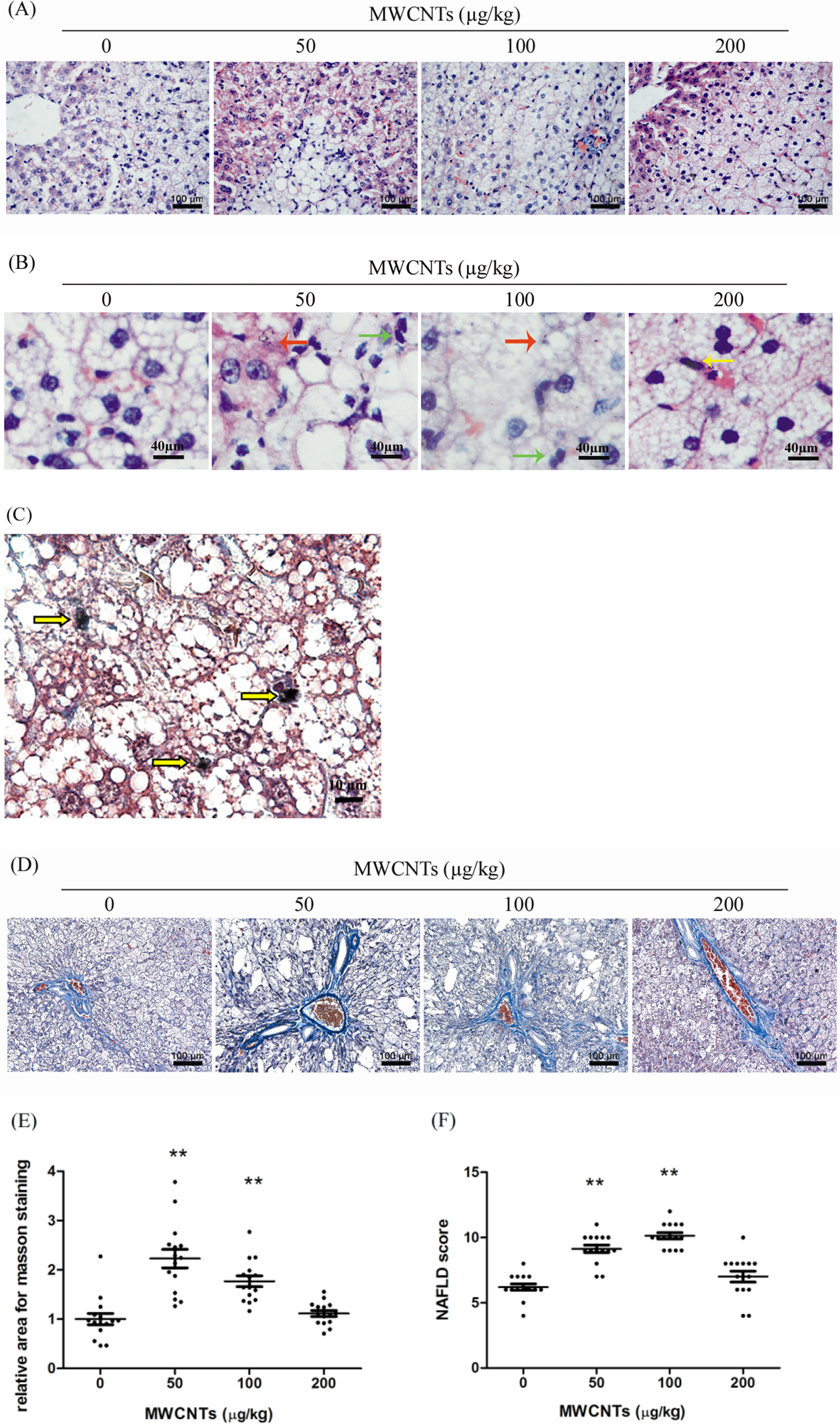
Histopathological changes in rat liver induced by MWCNTs. A, Hematoxylin and eosin staining, scale bar 100 µm; with 50 to 200 µg/kg MWCNTs administration, the lobules were indistinct and hepatocytes were swollen. B, Hematoxylin and eosin staining, scale bar 40 µm. Red arrows identify necrotic hepatocytes; green arrows identify inflammatory cells; yellow arrows identify Kupffer cells engulfing MWCNTs. C, Masson trichrome staining, scale bar 10 µm. Yellow arrows identify aggregates of MWCNTs within Kupffer cells in the liver from 200-µg/kg MWCNTs group. D, Representative images of Masson trichrome staining, scale bar 100 µm and (E) statistical analysis for fiber identification (blue) relative to normal diet group (n = 3; 5 pictures were randomly taken for each liver slice). F, Nonalcoholic fatty liver disease scores for the various treatment groups (n = 3). Image Pro Plus 6.0 was used for the image analysis and SPSS 16.0 for statistical analysis. **P < 0.01 versus solvent control group. MWCNTs indicates multiwalled carbon nanotubes; SD, standard deviation.
Masson trichrome staining was then used to identify hepatic fibrosis. Model group or solvent control group already showed more fibrosis in the liver than those fed with normal diet. Fibrosis was even more severe in rats fed with 50- and 100-µg/kg MWCNTs compared to the solvent control group. However, no significant difference was observed between the solvent control group and 200-µg/kg MWCNTs group (Figure 3D and E).
A new scoring system for nonalcoholic fatty liver disease was established through combining the existing nonalcoholic fatty liver disease grading standard with pathological classification criteria for rodents. Based on this system, 50- and 100-µg/kg MWCNTs groups showed significantly more severe steatohepatitis than the solvent control group, whereas 200-µg/kg MWCNTs group only showed mild increase but without significant difference compared to the solvent control group (Figure 3F).
Multiwalled Carbon Nanotubes Treatment Aggravates Steatohepatitis: Biochemical Analysis
In order to further evaluate the effects of MWCNTs on fatty liver, biochemical markers for liver fat composition and liver damage were then examined.
Increased TG and FFA are hepatic indicators for lipid metabolism disorder. After 4 months of experiment period, compared with solvent control, FFA in the 100-µg/kg MWCNTs group and TG in the 50-µg/kg MWCNTs group were increased 34% and 41%, respectively (Figure 4), suggesting that MWCNTs might interfere with lipid metabolism. However, no significant difference was observed for the 200-µg/kg group compared with the control group.
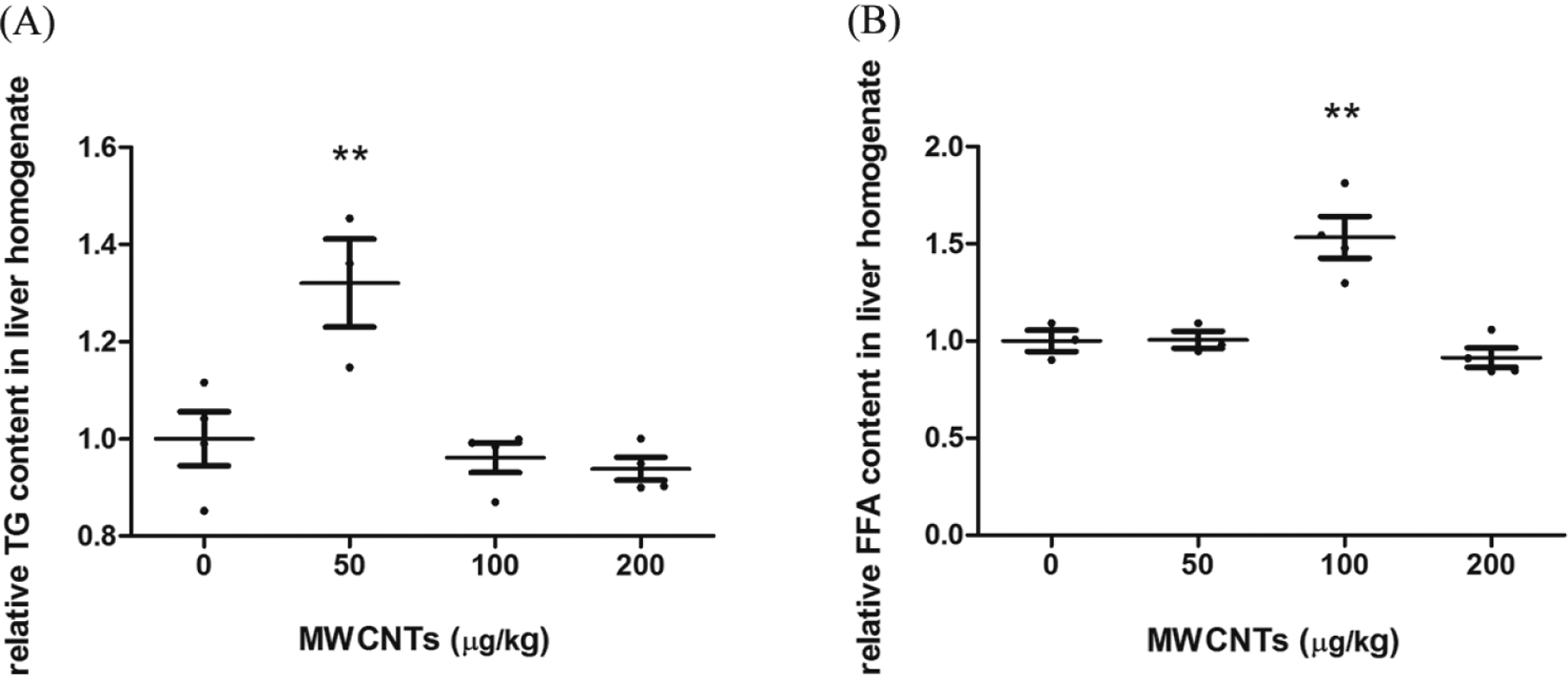
Relative TG and FFA concentrations in the liver homogenate of MWCNTs treatment groups. SPSS 16.0 was used for statistical analysis. **P < 0.01 versus solvent control group, n = 4, except for n = 3 in the 50-µg/kg MWCNTs group. FFA indicates free fatty acid; MWCNTs, multiwalled carbon nanotubes; TG, triglyceride.
Aspartate aminotransferase is a liver aminotransferase that is mainly located in the cytoplasm. 6 The increased AST indicates hepatocellular or myocyte leakage, usually caused by acute or chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma. γ-Glutamyl transferase is a biliary marker, serum GGT is rather low under normal condition, and biliary lesion/injury or cholestasis would cause an increase in GGT. In our experiment, compared with the solvent control group, AST in 50 and 100 µg/kg MWCNTs and GGT in 50 µg/kg MWCNTs increased 37%, 64%, and 361%, respectively (Figure 5). Taken together, such serum indexes for liver injury markers indicated chronic hepatitis in MWCNTs-treated rats.
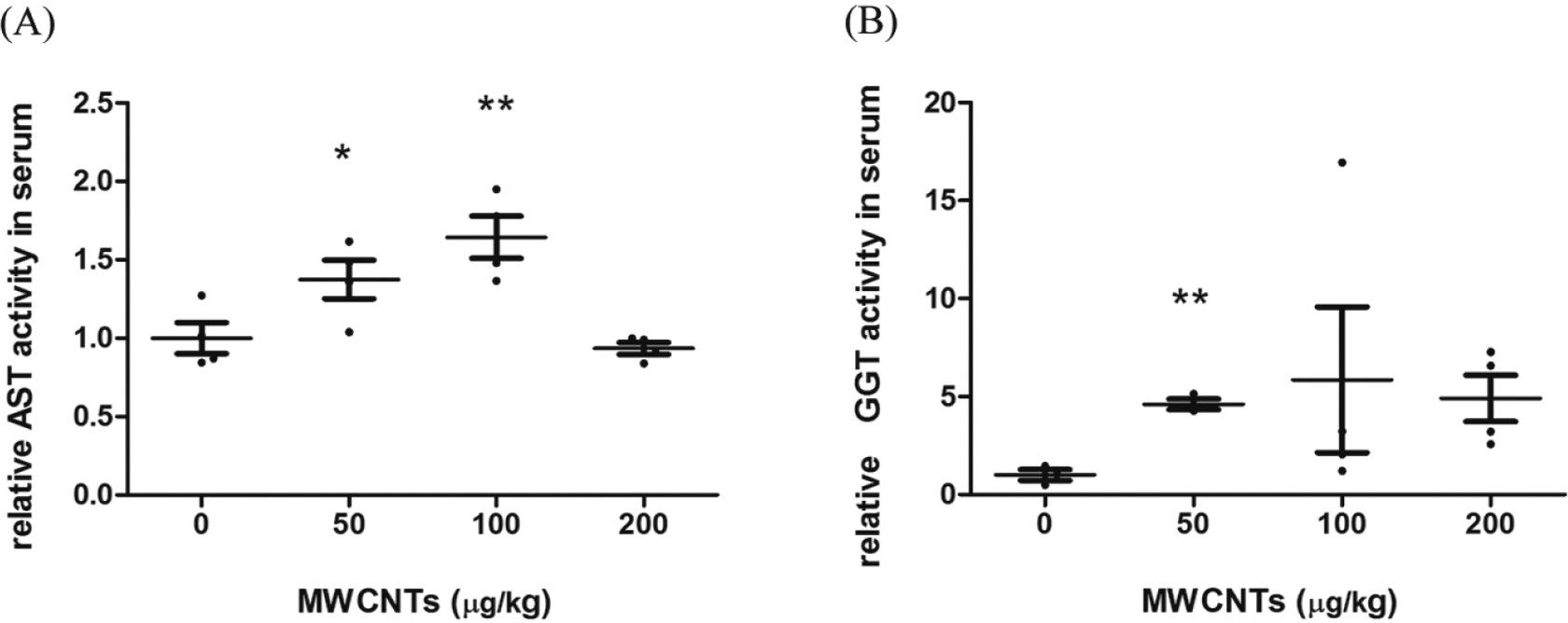
Relative serum AST and GGT activities for liver function. SPSS 16.0 was used for statistical analysis. *P < 0.05 versus solvent control group. **P < 0.01 versus solvent control group, n = 4, except for n = 3 in the 50-µg/kg MWCNTs group for GGT. AST indicates aspartate aminotransferase; GGT, γ-glutamyl transferase; MWCNTs, multiwalled carbon nanotubes.
Multiwalled Carbon Nanotubes Treatment Induces Oxidative Stress in Rats
Since it has been suggested that oxidative stress could be involved in the toxic effects of MWCNTs, we examined the expression of P6, which can be induced by oxidative stress in the liver. In our experiment, it was shown that high-lipid diet alone could increase the expression of P6 in rats than in those with normal diet. More importantly, compared with the solvent control, administration of MWCNTs further increased the expression of P6 at all 3 concentrations tested, although a clear dose–response was not observed, as there was no significant difference between the 200-µg/kg MWCNTs and 100-µg/kg MWCNTs groups (Figure 6A and B).
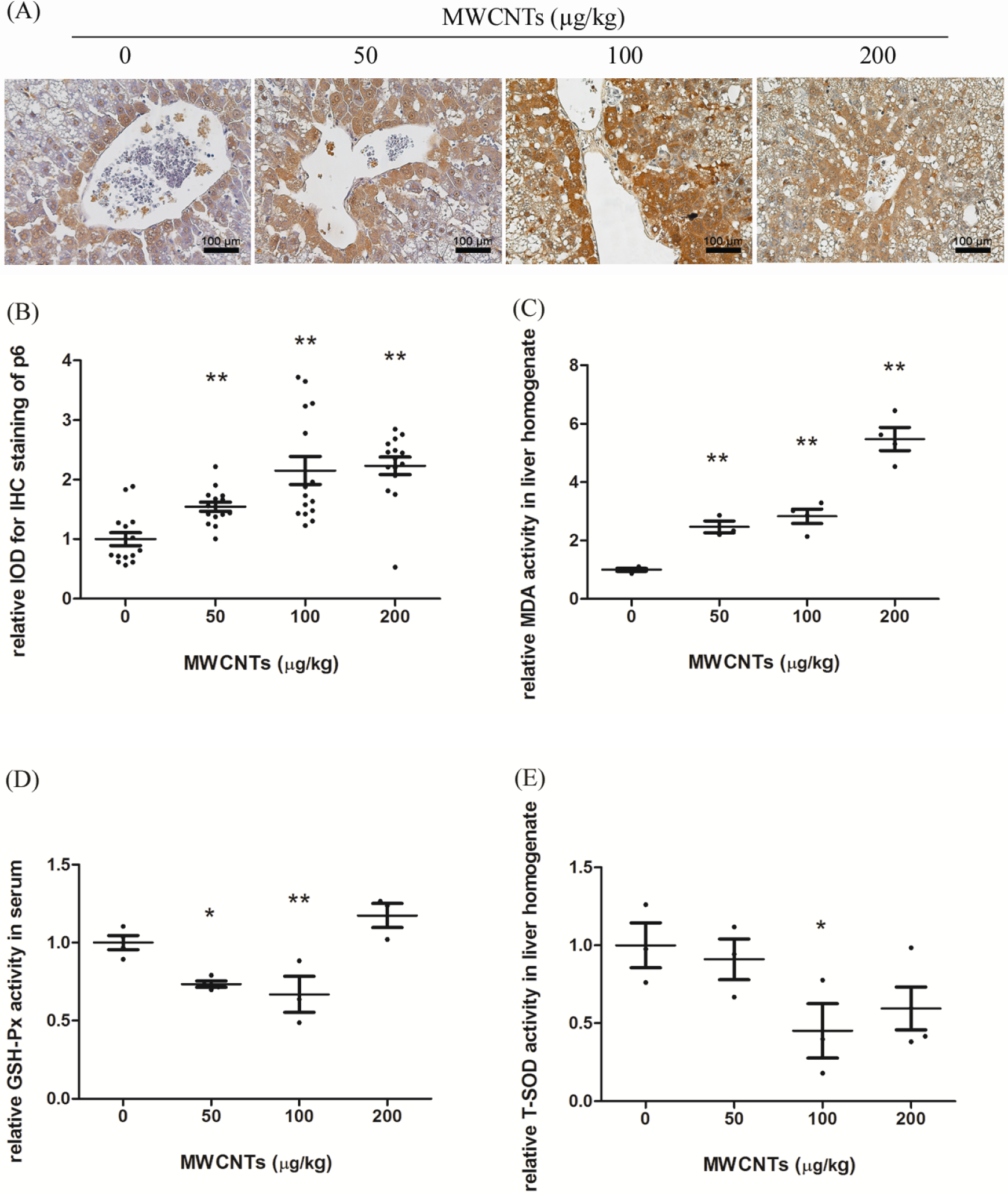
The effects of MWCNTs on oxidative stress-related molecules. Indexes were detected with corresponding kits. A, Representative images (scale bar 100 µm) of immunohistochemistry for P6 (brown), and compared with the solvent control, administration of MWCNTs further increased the expression of P6 at all 3 concentrations tested; and (B) statistical analysis results, n = 3. Relative activities of MDA (C) and T-SOD (E) in liver homogenate and GSH-Px in serum (D) were also shown. Image Pro Plus 6.0 was used for the IOD analysis and SPSS 16.0 for statistical analysis. *P < 0.05 versus solvent control group. **P < 0.01 versus solvent control group. For MDA, n = 4, except for n = 3 in the 50-µg/kg MWCNTs group. For GSH-Px, n = 3 in the 100- and 200-µg/kg MWCNTs groups, whereas n = 4 in the 0- and 50-µg/kg MWCNTs groups. For T-SOD, n = 3, except for n = 4 in the 200-µg/kg MWCNTs group. GSH-PX, glutathione peroxidase; IOD, integrated optical density; MDA, malondialdehyde; MWCNTs, multiwalled carbon nanotubes; P6, peroxiredoxin 6; T-SOD, total superoxide dismutase.
Furthermore, MDA is the final product of liperoxidation of polyunsaturated fatty acids and a marker of oxidative stress and liver damage. 18 With MWCNTs administration, MDA in the 50- to 200-µg/kg MWCNTs groups increased 147%, 183%, and 448%, respectively (Figure 6C). In contrast, GSH-Px is one of the most abundant molecules among endogenous antioxidants, which is always present in a relatively high concentration (1-10 mmol/L) that allows the rapid scavenge of reactive oxygen species either directly or indirectly. 19 In our experiment, GSH-Px in the 50- and 100-µg/kg MWCNTs groups decreased 27% and 33%, respectively (Figure 6D). Similarly, SOD is another essential cellular defense system against reactive oxygen species. 20 With MWCNTs administration, T-SOD in the 100-µg/kg MWCNTs group was reduced 55% (Figure 6E).
Discussion
With the ever increasing application of MWCNTs, human exposure to such substance and related health effects is becoming an important issue for the society, which led to many detailed studies with interesting results. Previous studies have reported that carbon nanotubes tended to accumulate in the liver, spleen, and lungs. More interestingly, it was shown that after intravenous injection, they were mainly retained in the liver of mice for a long time. 21 Still, most published in vivo studies applied the inhalation method and focused on the effects of MWCNTs on the respiratory system. Herein, we turned our focus to the toxicity of MWCNTs on the liver after intravenous injection.
First, we used high-fat diet to establish a fatty liver rat model. It was found that liver index from the model group was higher than the normal diet group (Figure S1D). Further macroscopic (Figure S1A) and histologic (Figure S1B and C) analyses revealed significant differences between the normal diet group and the model group. Last but not least, compared with the normal group, 2 biochemical indexes for fatty liver, TG and CHO, were increased in the model group. All those indicated that the fatty liver model had been successfully established.
The liver toxicity of MWCNTs was then evaluated. A pathological classification criteria system for nonalcoholic fatty liver disease was used to assess the liver condition based on hematoxylin and eosin and Masson trichrome staining. It was clear that with 50 to 100 µg/kg MWCNTs administration, histologically, liver parenchyma showed steatohepatitis (Figure 3). Among the serum markers, TG and FFA levels were increased, suggesting that the administration of MWCNTs could aggravate the abnormal lipid metabolism already induced by the high-fat diet. Aspartate aminotransferase is located mainly in the cytoplasm. High level of AST indicates hepatocellular injury or cytoplasmic leakage. In our experiment, the levels of AST and GGT, both liver injury markers, were increased, indicating chronic hepatitis in MWCNTs-treated rats. Similarly, Ji et al 6 also found that AST was increased in MWCNTs-treated mice. Furthermore, Caldwell et al had used electron microscopy to show that mitochondrial injury happened more often during NASH. 22 Taken together, these results indicated that MWCNTs aggravated hepatitis.
Many previous studies have pointed out that reactive oxygen species is a key player, which mediates many of the effects of nanomaterials. In vitro study also showed that the toxicity of MWCNTs to the hepatocyte cell line was at least in part mediated by reactive oxygen species/oxidative stress. 23 Therefore, we examined the oxidative status in the fatty liver rat model. Peroxiredoxin 6, a bifunctional 25-kDa protein, uses GSH-Px as the physiological reductant and has the ability to bind phospholipid hydroperoxides to remove cellular hydroperoxides, thus protecting cells from reactive oxygen species–mediated cell damage and death. 24 Eismann et al reported that p6-null mice showed increased injury in the liver when subjected to the oxidative stress of ischemia followed by reperfusion. 25,26 It has also been reported that P6 can bind to glycerophospholipids to produce FFAs and lysophospholipids. 27 Still, there are studies showing that P6 promotes the activity of NADPH (nicotinamide adenine dinucleotide phosphate) oxidase activity, which is one of the major sources of reactive oxygen species and also increases cellular toxicity paradoxically in consideration of the role of the cytosolic GSH-Px activity of P6. 24,28 In our experiment, MWCNTs administration led to significantly increased levels of P6, accompanied with increased liver damage, suggesting that MWCNTs might cause liver damage through the induction of oxidative stress.
Malondialdehyde, the final product of liperoxidation of polyunsaturated fatty acids and a marker of oxidative stress and liver damage, was also increased in all MWCNTs-treated groups. Besides, the cellular level of GSH-Px and T-SOD activity, both of which are essential parts of cellular defense mechanism against reactive oxygen species, was reduced. This is in consistent with the finding of Ji et al, in which GSH-Px level and SOD activity were reduced in mouse liver exposed to different doses of MWCNTs. 6 Similarly, Lee et al also reported that when Japanese rice fish (Oryzias latipes) were exposed to MWCNTs dispersed in water, gene expression levels of catalase and glutathione-S-transferase were both reduced at the fourth day. 29 Together, these data pointed out the importance of reactive oxygen species in mediating the bioeffects of MWCNTs.
Recent studies have suggested a possible role for autophagy during the development of nonalcoholic fatty liver disease. Autophagy has been shown to regulate intracellular lipid storage through autolipophagosomal translocation of lipid droplets, lysosomal degradation, and the release of fatty acids into the cytoplasm in rapid response to starvation. 30 –33 Thus, disruption of autophagy might contribute to the pathogenesis of NASH. 33,34 Interestingly, it has been reported that MWCNTs could induce autophagy dysfunction. 35,36 Thus, we examined the autophagy markers p62 and lc3 with immunohistochemical method, in an effort to determine whether MWCNTs can lead to nonalcoholic fatty liver disease through the induction of autophagy dysfunction. However, no significant changes in the expression levels of these 2 proteins were found (data not shown) in our experiments. Nonetheless, more detailed studies are warranted to evaluate whether MWCNTs affect autophagy in the nonalcoholic fatty liver disease model.
Still, although MWCNTs aggravated NASH, no obvious dose-dependent response was found, as the effects of 200 µg/kg MWCNTs were usually mild. This may be the result of the characteristic of nanoparticles, which tend to aggregate at high concentrations. Indeed, we have found such microscopic aggregates of MWCNTs in Kupffer cells (Figure 3C). Under such conditions, the aggregated MWCNTs may not act the same as dispersed MWCNTs. This is an area worth further exploring.
In conclusion, for the first time, we investigated the in vivo toxicity of MWCNTs to fatty liver in SD rats. In this study, we have shown that MWCNTs were able to aggravate NASH in SD rats, and oxidative stress may be involved in this process. Therefore, the liver toxicity of MWCNTs should be taken into consideration when it is applied in tumor targeting and delivery of drugs by intravenous injection.
Footnotes
Author Contributions
Yu-Ying Xu and Juan Ge contributed equally to this work. Yu-Ying Xu contributed to conception and design, contributed to acquisition, drafted the manuscript, and critically revised the manuscript. Juan Ge contributed to conception, contributed to analysis and interpretation, and drafted the manuscript. Mo-Han Zhang contributed to acquisition and drafted the manuscript. Wen-Jie Sun contributed to acquisition and analysis and drafted the manuscript. Jun Zhang contributed to acquisition and critically revised the manuscript. Pei-Lin Yu contributed to interpretation and critically revised the manuscript. Yi-Fan Zheng contributed to acquisition and drafted the manuscript. Jun Yang contributed to conception and critically revised the manuscript. Xin-Qiang Zhu contributed to design and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was in part supported by research grant from National Natural Science Foundation of China (30771824 and 30800923 to Xin-Qiang Zhu and to Jun Zhang).
