Abstract
Nonalcoholic fatty liver disease (NAFLD) is caused by fat accumulation and is related with obesity and oxidative stress. In this study, we investigated the effect of cuminaldehyde on NAFLD in rats fed a high fat diet (HFD). Male Wistar rats were fed a HFD for 42 days to induce NAFLD. The progression of NAFLD was evaluated by histology and measuring liver enzymes (alanine transaminase and aspartate transaminase), serum and hepatic lipids (total triglycerides and total cholesterol), and oxidative stress markers (thiobarbituric acid reactive substances, glutathione, superoxide dismutase, and catalase). The HFD feeding increased the liver weight and caused NAFLD, liver steatosis, hyperlipidemia, oxidative stress, and elevated liver enzymes. Administration of cuminaldehyde ameliorated the changes in hepatic morphology and liver weight, decreased levels of liver enzymes, and inhibited lipogenesis. Our findings suggest that cuminaldehyde could improve HFD-induced NAFLD via abolishment of hepatic oxidative damage and hyperlipidemia. Cuminaldehyde might be considered as a potential aromatic compound in the treatment of NAFLD and obesity through the modulation of lipid metabolism.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is one of the major global health issues, strongly correlated with obesity and oxidative stress. Nonalcoholic fatty liver disease is a common disorder, and its prevalence has increased globally. It is considered as an asymptomatic disease (having no symptoms of disease) that is typically identified when the liver enzymes alanine aminotransferase (ALT) and aspartate aminotransferase (AST) are raised. The United States National Health and Nutrition Examination Survey have recently conducted a population-based analysis revealing that the percentage of the United States population with NAFLD has steadily increased over the past 20 years. 1
HFD-induced dyslipidemia and lipid accumulation initiate the development of hepatic steatosis and may progress to nonalcoholic steatohepatitis (NASH), fibrosis, cirrhosis and, ultimately, hepatocellular carcinoma, 2 which comprises the NAFLD. Excessive triglyceride accumulation in hepatocytes is the hallmark of NAFLD. Based on the results of animal studies and epidemiological investigations, a two-hit hypothesis has been proposed for the pathogenesis of NAFLD: the first hit is excessive fat accumulation in the liver, and the second hit is oxidative stress that initiates hepatic steatosis to develop NASH. 3 Although hepatic steatosis is often self-limited, it is necessary to treat it to avoid its progression to more serious diseases. Much of the increase in prevalence of NAFLD is driven by its epidemiologic and pathophysiologic links to oxidative stress-mediated non-communicable diseases including type 2diabetes mellitus and obesity. 4 Oxidative stress develops when there is an imbalance between reactive oxygen species (ROS) and antioxidants that scavenge oxidative insults. 5 ROS are involved in the etiology of NAFLD by stimulating glutathione (GSH) depletion, accumulation of lipid peroxides, and oxidative damage of different organelles in liver tissue. 6 GSH is the major intracellular antioxidant in hepatocytes and plays an important role in maintaining the reduced cellular homeostasis in hepatocytes, and this is essential for optimum activities of several antioxidant enzymes including superoxide dismutase (SOD) and catalase (CAT). Currently, no treatments have been established for NAFLD beyond management of comorbidities and weight loss. Lifestyle intervention and pharmacotherapy to treat hepatic steatosis are limited because of poor compliance and side effects. As a result, new approaches to improve hepatic steatosis are urgently necessary. Limited evidence exists, which suggests that antioxidant supplements may have a role in preventing or treating NAFLD in patients with diabetes. 7 Recently, a considerable number of studies have investigated natural phytochemical as anti-NAFLD agents. 8 –10
Cuminaldehyde (4-isopropylbenzaldehyde) is a natural aldehyde organic compound with the molecular formula C10H12O. It is a biologically active constituent of the essential oils of Cuminum cyminum, Carum caraway, Cinnamomum cassia and Cinnamomum verum, and so on. 11,12 Cuminaldehyde has strong antidiabetic action against streptozotocin-induced diabetic rats. 13 It has also been found that cuminaldehyde acts as a good lipoxygenase inhibitor. 14 It has been demonstrated that cuminaldehyde has a good anticancer activity. 15 In the present article, the effect of cuminaldehyde against NAFLD in rats fed a HFD is explored.
Materials and methods
Chemicals and kits
Cuminaldehyde (purity; 98%, molecular weight; 148.20) was purchased from Sigma–Aldrich (St. Louis, Missouri, USA). Total cholesterol (TC) (code no. 25924, new code no. 71MB100-64), total triglyceride (TG) (code no. 77034, new code no. 72LS100-40), ALT (code no. 25706, new code no. 77MB101-50), and AST kits (code no. 25707, new code no. 76MB101-50) were purchased from Span Diagnostics Ltd (new name—ARKRAY Healthcare Pvt. Ltd), Surat, Gujarat (India). Rat leptin ELISA kit (BioVendor, Czech Republic, Cat no. ELR-Leptin-001), rat insulin ELISA kit (Alpco Diagnostics, Salem, USA, Cat no. 90060), and orlistat (purity > 99.5%) were obtained from Labex corporation, (Vasant kunj, New Delhi, India). All other chemicals used were of analytical grade.
Animals
Wistar male albino rats, weighing 150–200 g, were procured from the Central Animal House Facility (registration no. 173/GO/Re/S/2000/CPCSEA, dated—28 January 2000), Hamdard University, New Delhi (India), for this valuable study and acclimatized under standard laboratory conditions at 25 ± 2°C and relative humidity (50 ± 15%). The animals were kept six per cage under standard laboratory conditions (12 h light and 12 h dark) and had a free access to tap water ad libitum. The study protocol was approved (approval no. 607/CPCSEA, dated—13 June 2011, proposal no. 707) by the Institutional Animal Ethics Committee (IAEC) of Hamdard University, New Delhi, which is registered with Committee for the Purpose of Control & Supervision of Experiments on Animals (CPCSEA), Government of India.
Animal experiments
After 7 days of acclimation, animals were randomly separated into 5 groups (each containing six rats): one normal control group, one HFD pathogenic group (toxic group), and remaining groups as treatment groups.
Normal control group rats received normal pellet diet and high fat diet (HFD) pathogenic group rats fed with HFD for 6 weeks. The normal pellet diet (12.5% fats, 62.3% carbohydrate, and 24.3% protein) was procured from Pranav Agro Industries Ltd (Maharashtra, India). HFD (42% lipids, 36% carbohydrate, and 22% protein) was procured from National Centre for Laboratory Animal Sciences, National Institute of Nutrition, Hyderabad (Andhra Pradesh, India). Treatment groups (HFDC1, HFDC2, and HFDO) animals were fed with HFD for 2 weeks, after that respective drug were given simultaneously along with HFD for 4 weeks; HFDC1: HFD + cuminaldehyde; HFDC2: HFD + cuminaldehyde; HFDO: HFD + orlistat. 16 HFDC1 and HFDC2 were given cuminaldehyde respectively at 7 mg/kg and 14 mg/kg twice per day and HFDO were given orlistat at 30 mg/kg once per day by oral gavage (treatment schedules are shown in Table 1). Orlistat and cuminaldehyde drugs were suspended in 0.5% carboxymethyl cellulose sodium aqueous solution for administration. Blood was collected from the retro-orbital plexus of all rats following overnight fasting using microcapillary tubes containing heparin on 43th day. Serum was separated from blood by centrifugation at 1610 × g for 10 min (model no. Remi R-8C) and transferred to Eppendorf tubes.TC, TG, ALT, AST, leptin, and insulin in serum were measured with commercial kits. Double distilled water was used for all biochemical assays. After blood collection, overnight fasted rats were killed using urethane anesthesia. The livers from each group were dissected out, rinsed with phosphate buffer saline, and weighed. The collected livers were used for histopathology and lipid peroxidation, SOD, GSH, and CAT assay. A portion of the liver was minced and homogenized (10% w/v) for determination of lipid peroxidation by thiobarbituric acid reactive substances (TBARS), 17 reduced GSH, 18 SOD, 19 and CAT activities. 20,21 The hepatic tissue samples were fixed in 10% neutral buffered formalin (10% NBF), cut into 5μm sections (from liver right lobe), and stained with H and E (hematoxylin and eosin). The stained slides for each rat tissue samples were observed at 100× and 400× magnification using a Nikon’s Brightfield Compound Nikon microscope (model YS100). The sections of hepatic tissues were studied to find out the level of tissue damage by HFD.
Treatment schedule (showing experimental design).

HFD: high fat diet; HFDC1: HFD + cuminaldehyde; HFDC2: HFD + cuminaldehyde; HFDO: HFD + orlistat.
Statistical analysis
Data are expressed as the mean ± standard error of the mean (SEM).The statistical significance of difference between the mean values for the treatment groups was analyzed by analysis of variance followed by Dunnett's t-test using Graph-Pad InStat® version 3.06 (GraphPad Software). Values of p < 0.05 were considered significant.
Results
Effect of cuminaldehyde on serum TC and TG
Serum concentrations of TC and TG levels in HFD group were significantly (p < 0.001) increased as compared to the normal control group. HFDC1, HFDC2, and HFDO significantly (p < 0.001) reduced the serum concentration of TC and TG levels as compared with HFD group. HFDC2 showed more significant reduction in elevated level of TC and TG as comparison to HFDC1. Results are shown in Figures 1 and 2.
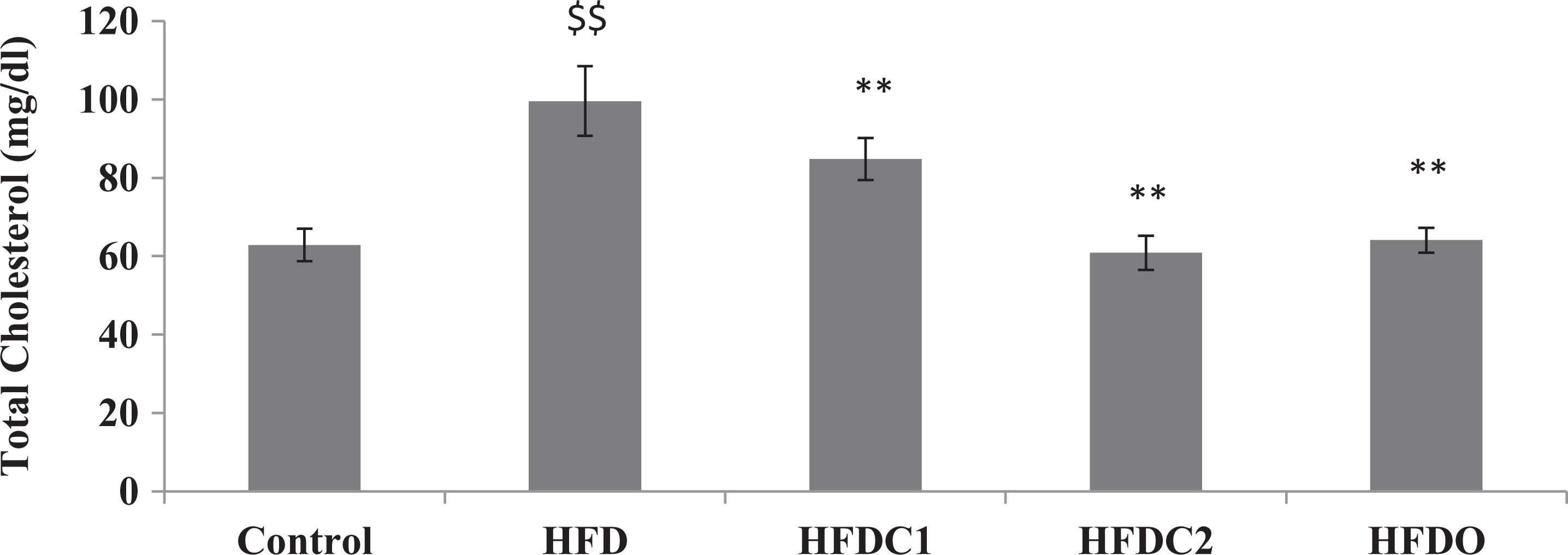
All values were expressed as mean ± SEM (n = 6). $$p < 0.001 as compared to control group. **p < 0.001 as compared to HFD group. HFD: high fat diet.

All values were expressed as mean ± SEM (n = 6). $$p < 0.001 as compared to control group. **p < 0.001 as compared to HFD group. HFD: high fat diet.
Effect of cuminaldehyde on liver weight
Liver weight was about 2.5 times greater in the HFD group than that in the normal control group. Cuminaldehyde and orlistat treatment significantly (p < 0.001) reduced the liver weight as compared with HFD group. HFDC2 showed more significant reduction in liver weight as compared to HFDC1. Results are shown in Figure 3.
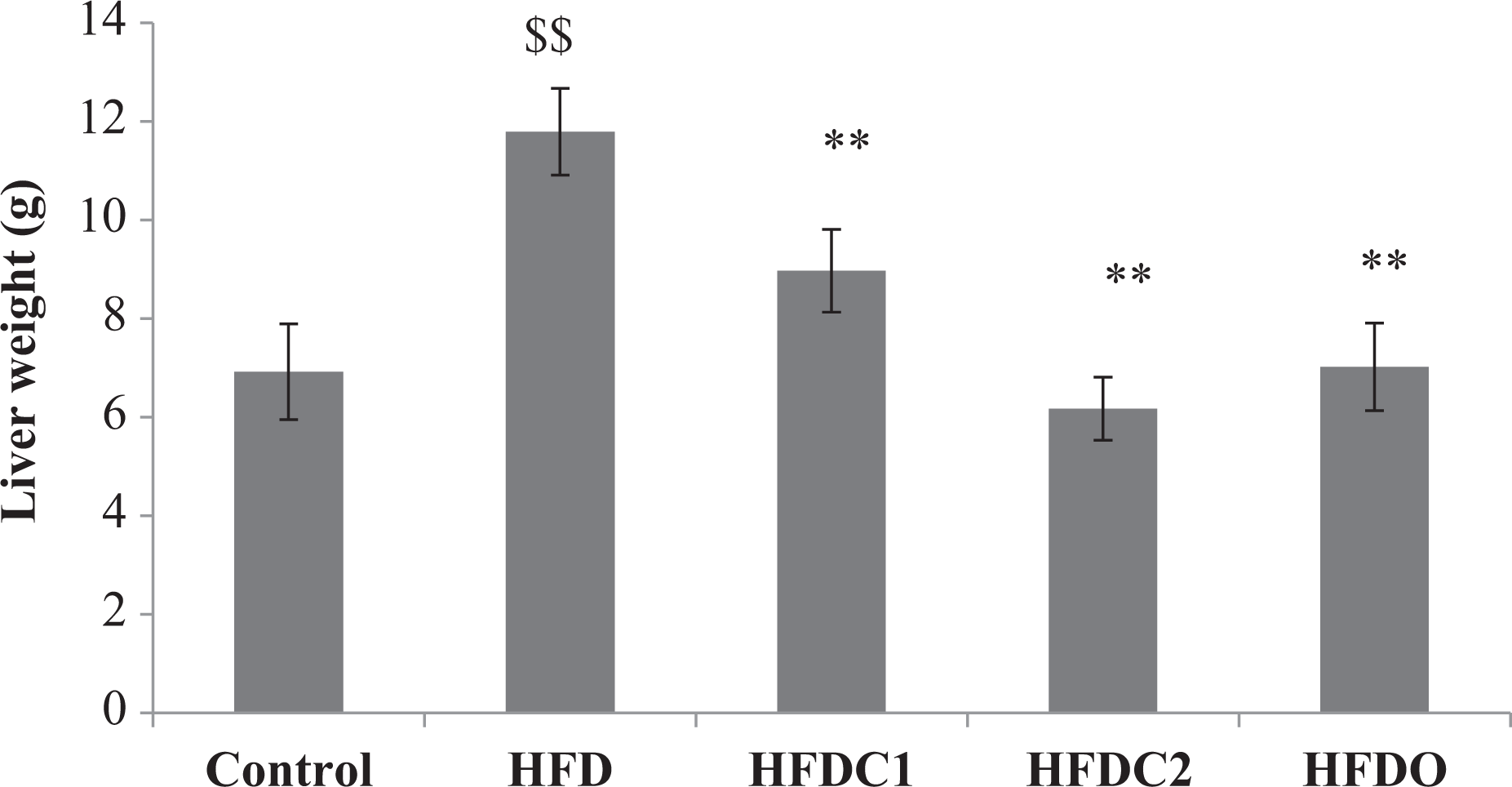
All values were expressed as mean ± SEM (n = 6). $$p < 0.001 as compared to control group. **p < 0.001 as compared to HFD group. HFD: high fat diet.
Effect of cuminaldehyde on hepatic lipid peroxidation and antioxidant level
HFD-fed rats showed significant (p < 0.001) decreases in hepatic GSH, SOD, and CAT and significant (p < 0.001) augmentation in malondialdehyde (MDA) as compared to the normal control group. In contrast, HFDC1, HFDC2, and HFDO treatment significantly augmented hepatic GSH, SOD, and CAT level and significantly reduced MDA level as compared to HFD group. HFDC2 treatment showed more significant augmentation in hepatic GSH, SOD, and CAT level and significant (p < 0.001) reduction in MDA level as compared to HFDC1 group. Results are shown in Table 2.
Effect of cuminaldehyde on MDA, GSH, SOD and CAT Level.a
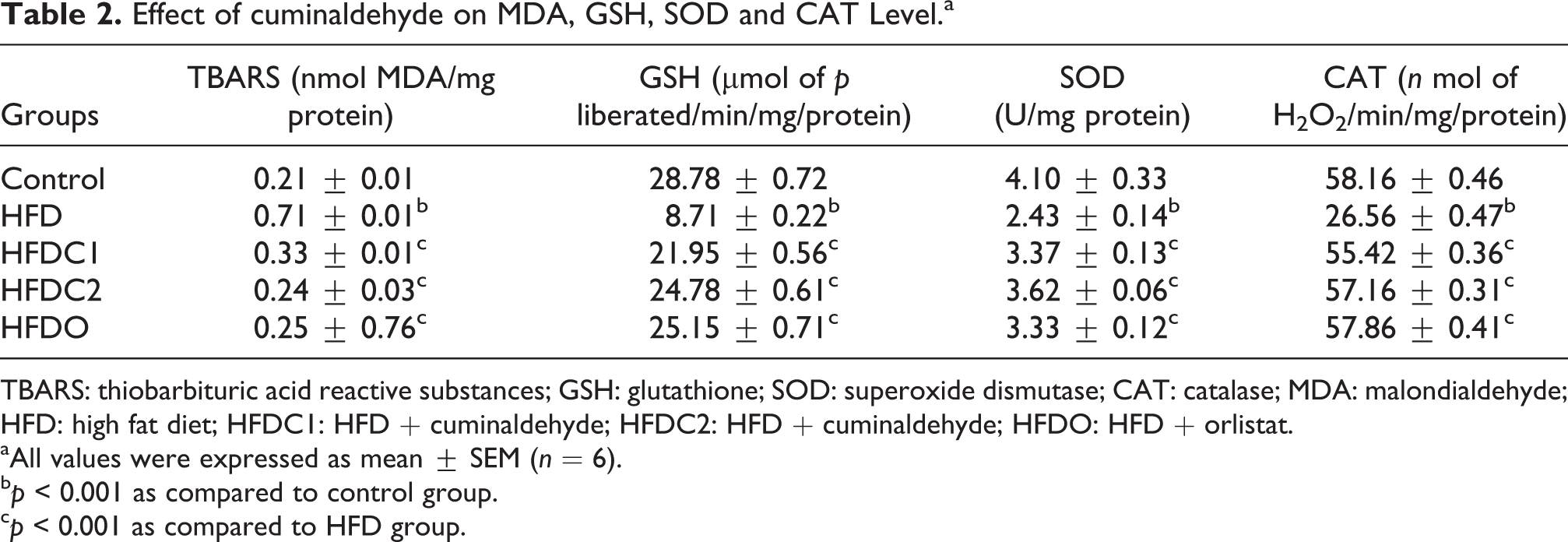
TBARS: thiobarbituric acid reactive substances; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; MDA: malondialdehyde; HFD: high fat diet; HFDC1: HFD + cuminaldehyde; HFDC2: HFD + cuminaldehyde; HFDO: HFD + orlistat.
aAll values were expressed as mean ± SEM (n = 6).
bp < 0.001 as compared to control group.
cp < 0.001 as compared to HFD group.
Effect of cuminaldehyde on liver enzyme
Significant (p < 0.001) elevated level of AST and ALT in HFD groups was found when compared with the normal control group. HFDC1 and HFDC2 were significantly lowered the increased level of AST and ALT as compared to HFD group. And HFDO were showed significant (p < 0.001) reduction of increased ALT level but did not alter in increased AST level. HFDC2 showed more significantly (p < 0.001) lowered the increased level of AST and ALT as compared to HFDC1 group. Results are shown in Table 3.
Effect of cuminaldehyde on liver enzymes and hepatic lipids (TG and TC) Level.a

AST: and aspartate aminotransferase; ALT: alanine aminotransferase; TG: total triglyceride; TC: total cholesterol; HFD: high fat diet; HFDC1: HFD + cuminaldehyde; HFDC2: HFD + cuminaldehyde; HFDO: HFD + orlistat.
aAll values were expressed as mean ± SEM (n = 6).
bp < 0.001 as compared to control group.
cp < 0.05 as compared to HFD group.
dp < 0.001 as compared to HFD group.
Effect of cuminaldehyde on hepatic lipids level (TG and TC)
HFD-fed rats showed significant (p < 0.001) increases in hepatic TG and TC levels as compared to the normal control group. HFDC1 and HFDC2 or orlistat treatment significantly (p < 0.001) ameliorated hepatic TG and TC levels as compared to HFD group. Results are shown in Table 3.
Effect of cuminaldehyde on serum leptin and insulin
Serum concentrations of leptin and insulin levels in HFD group were significantly (p < 0.001) increased as compared to the normal control group. HFDC1, HFDC2, and HFDO significantly (p < 0.001) reduced the serum concentration of leptin and insulin levels as compared with HFD group. HFDC2 showed more significant reduction in leptin and insulin levels as comparison to HFDC1. Results are shown in Figures 4 and 5.

All values were expressed as mean ± SEM (n = 6). $$p < 0.001 as compared to control group. **p < 0.001 as compared to HFD group. HFD: high fat diet.
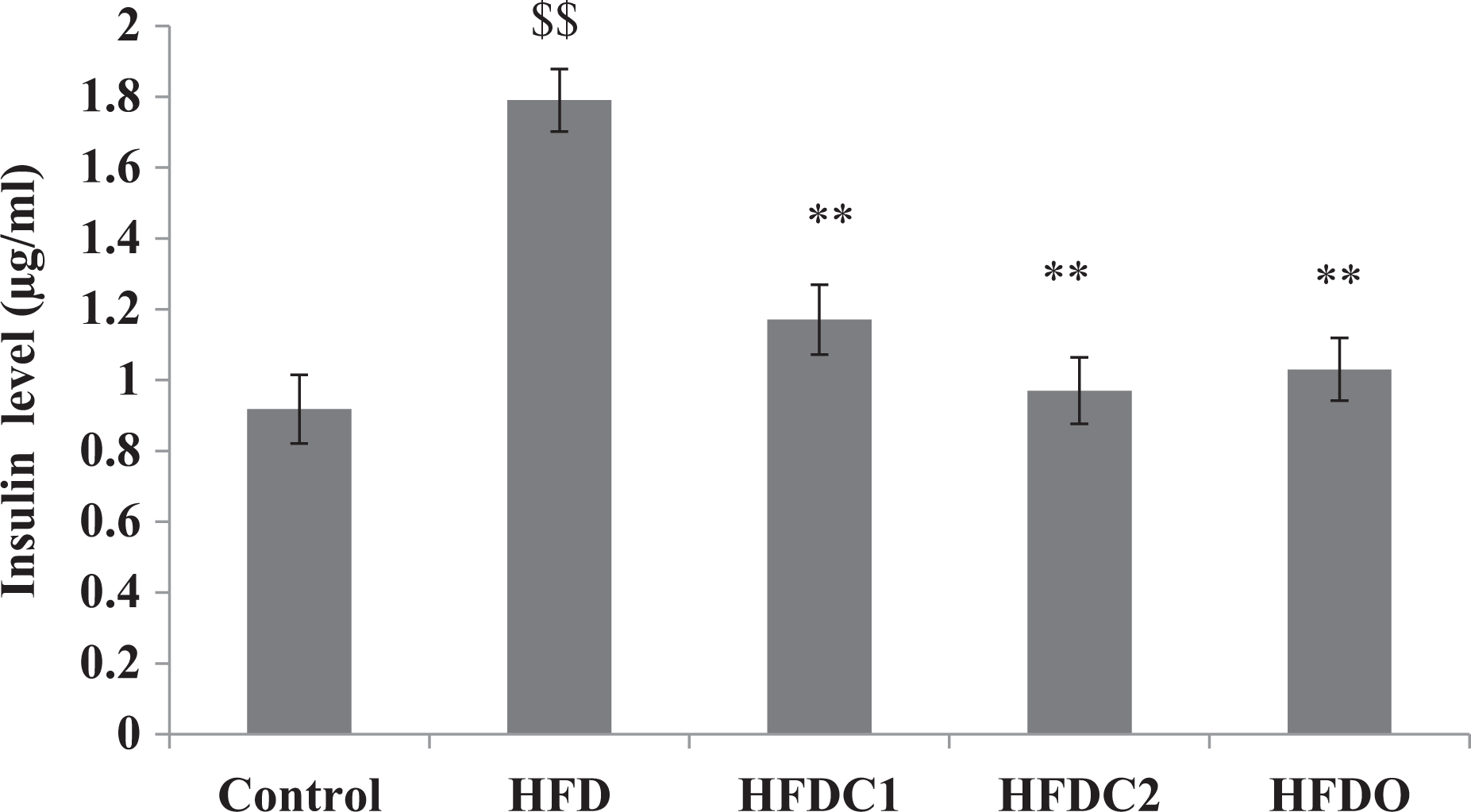
All values were expressed as mean ± SEM (n = 6). $$p < 0.001 as compared to control group. **p < 0.001 as compared to HFD group. HFD: high fat diet.
Effect of cuminaldehyde on liver tissue
Our histological examination of liver revealed the necrosis and inflammation in liver tissues of the HFD group as comparison to the normal control group. HFDC1 and HFDC2 or orlistat treatment noticeably prevented the extent of inflammation in hepatic tissues as comparison HFD group. Results are shown in Figures 6 and 7.
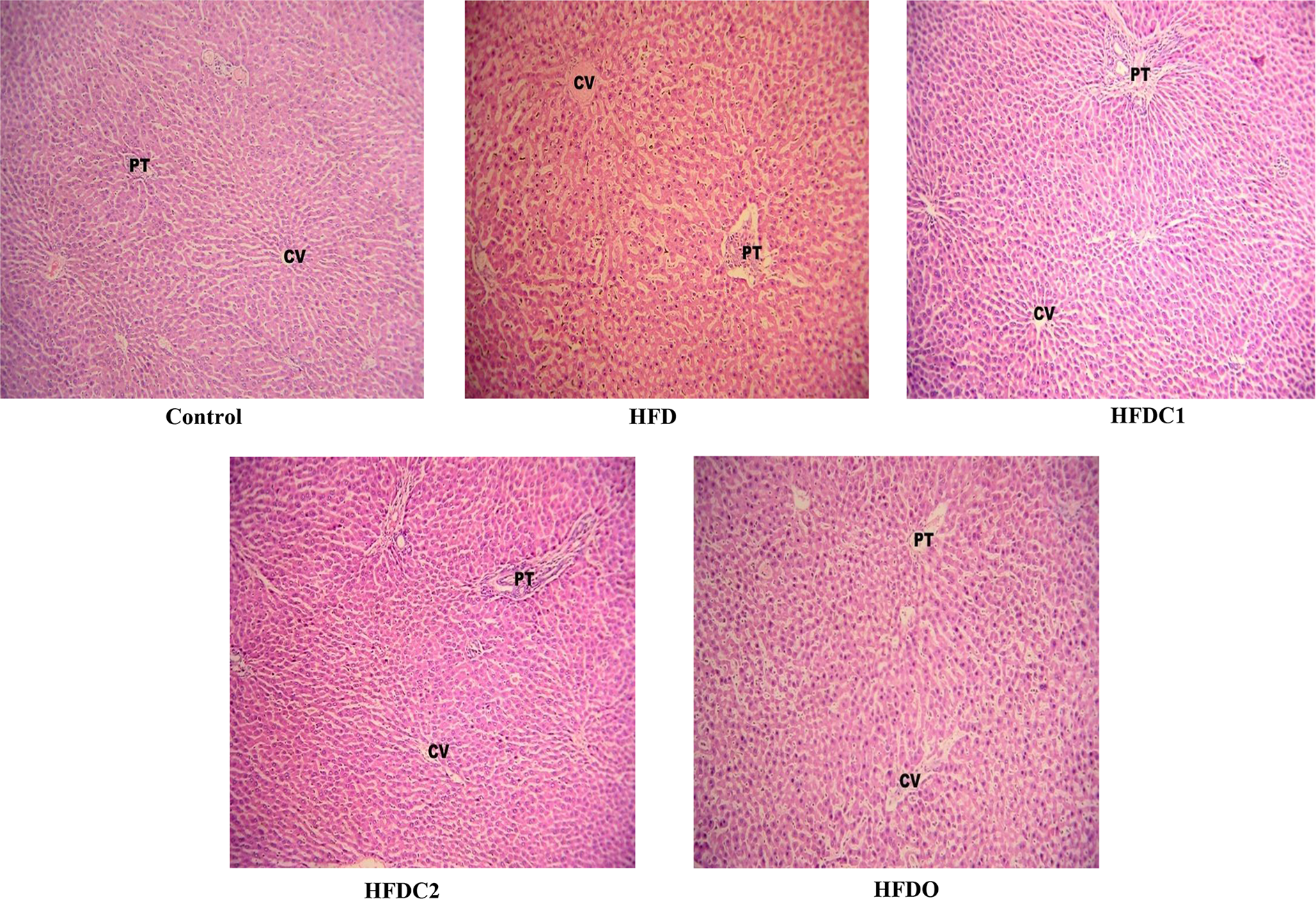
Observation of histopathological changes of liver in different groups (dye: H&E) at ×100 magnification. CV: central vein; PT: portal triad; H&E: hematoxylin and eosin.

Observation of histopathological changes of liver in different groups (dye: H&E) at ×400 magnification. CV: central vein; KC: Kupffer cells; Pig: lipofuscin pigment; Apo: apoptotic hepatocytes; Vac: vacuolated hepatocytes; N: hepatocyte nuclei; St: steatosis; H&E: hematoxylin and eosin.
Discussion
The present study was a preliminary research work on the protective effect of cuminaldehyde against NAFLD rats. A rodent model based on the high intake of HFD is advantageous in studying obesity-related significant disease such as oxidative stress in NAFLD. 22 Extensive consumption of dietary fat is considered a major factor in NAFLD and associated metabolic diseases in humans as well as in animal models. HFD is one of the most important environmental factors associated with the incidence of NAFLD. The epidemiological studies have showed a significant positive correlation between mean dietary fat intake and the incidence of NAFLD and its related complications and risk factors. 23 In the present study, the protective effect of cuminaldehyde in NAFLD rats was examined by measuring serum lipid level, oxidative stress marker, and hepatic lipid profile. HFD are extensively used in dietary experiments as a strategy to induce NAFLD conditions and fat accumulation in liver. 24
HFD-induced hyperlipidemia in serum and hepatic tissue are well reported. 25 The previous data also showed that rats in the HFD group exhibited significantly higher serum TG and TC. 26 However, the administration of cuminaldehyde or orlistat significantly lowered the TG and TC in serum. TGs are involved in the accumulation of lipid stores in the liver and are associated with a number of diseases such as metabolic syndrome. High TC is a risk factor for coronary heart disease. 27
Hyperlipidemia and NAFLD synergistically promote systemic oxidative stress imbalance between tissue free radicals, ROS, and antioxidants. ROS could react with polyunsaturated fatty acids, which lead to lipid peroxidation. 28 Possible mechanisms that generate oxidative stress in NAFLD include elevated lipid levels and inadequate antioxidant defenses. 29 Accumulation of liver fat is correlated with an increase in the lipid peroxidation product, MDA. MDA is a by-product of lipid peroxidation and reflects the degree of oxidation in the body. 30 The MDA level in hepatic tissue decreased significantly in the group treated with cuminaldehyde or orlistat. The reduction in the lipid peroxidation could be related to the antioxidant and free radical scavenging properties of cuminaldehyde. In the present study, cuminaldehyde or orlistat treatment showed significantly increased the SOD and CAT (free radical scavengers) in HFD-fed rats. Thus, this result suggests that the antioxidant activity of cuminaldehyde may, at least partly, contribute to the reduction of lipid level. It is therefore reasonable to assume that cuminaldehyde treatment improves oxidative stress balance in HFD-fed obese rats, because cuminaldehyde was able to reduce the level of TBARS and free radical generation. Thus, this result suggested that cuminaldehyde would be helpful in the protection against oxidative stress in NAFLD.
AST levels are predictive of damage to the hepatic and other organs with high metabolic activity (brain, heart, and lungs) and ALT is a sensitive marker of hepatic damage. The results of our study revealed that obese rats were more prone to hepatotoxicity as evidenced by increased level of serum AST, ALT, and hepatic lipid levels. Cuminaldehyde or orlistat treatment significantly reduced the elevated AST, ALT, and hepatic TC and TG levels in obese rats, which could be attributed to the protective effect on hepatic tissue. Numerous studies have demonstrated that resistance to both insulin and leptin actions have been implicated in accumulation of hepatic TG. In obese condition, leptin and insulin concentrations are elevated resulting in leptin and insulin resistance either centrally or locally at the level of the liver. 31,32 Our data also showed that rats in the HFD group exhibited significantly increases leptin and insulin levels in serum. However, the administration of cuminaldehyde or orlistat significantly decreased leptin and insulin levels. The results from the present studies suggest that cuminaldehyde caused considerable reduction of hepatic TG accumulation, indicated by reduced leptin and insulin levels. Thus, this result suggested that cuminaldehyde would be helpful in the protection against NAFLD through improvement in leptin and insulin sensitivity.
HFD is known to boost the synthesis of fatty acids or TG accumulation in the liver resulting in macrovesicular hepatic steatosis. 33 It was previously reported that a higher percentage of fat in the animals diet (30% W/W) led to severe hepatic microvesicular and macrovesicular steatosis which indicates severe NAFLD. 34 The histopathological analysis was done in this study to examine hepatocytes and liver tissues for any abnormalities. Fat accumulates in the liver in two forms: macrovesicular steatosis and microvascular steatosis. 35 Macrovesicular steatosis results in large fat droplets that replace hepatocyte nuclei, whereas microvascular steatosis results in small fat droplets that accumulate in the hepatocyte cytoplasm. Lipofuscin pigment and apoptotic hepatocyte are the clear feature of the more severe form of NAFLD/NASH. Microscopically, lipofuscin is a fine yellow brown granular pigment evident in H&E stained sections. 36 –38
Macrovesicular steatosis (hepatocyte nuclei), lipofuscin pigment and apoptotic hepatocytes were also observed in the HFD group of current study. The hallmark of NAFLD is TG accumulation in the cytoplasm of hepatocytes. 38 Cuminaldehyde or orlistat treatment significantly reduced the hepatic TG and TC levels as compared to HFD group. Moreover, cuminaldehyde or orlistat administration noticeably prevented the inflammation, steatosis, and damage in hepatic tissue, suggesting that cuminaldehyde may be a potential promising candidate for the protection of NASH. Based on the results of this study, cuminaldehyde act as a powerful antioxidant against the rat liver effect of high fat dietary induced-NAFLD. The results of the present study highlight the potential health benefits of cuminaldehyde against NAFLD. This health promoting effect of cuminaldehyde is suggested to present an economic potential of cuminaldehyde as a food supplement. The current study suggests that cuminaldehyde would be helpful in protection against HFD-induced NAFLD, because it exerts a strong preventative effect against oxidative stress in NAFLD.
In conclusion, this study showed feeding HFD to rats induced oxidative stress in liver tissue as compared to control group. Cuminaldehyde or orlistat treatment showed a protective effect against oxidative stress in NFALD. In this work, by means of serum and hepatic biochemical assays and histopathology, we demonstrate that cuminaldehyde has protective effect against NAFLD for the first time, which is achieved through regulating serum and hepatic lipids via improvement in leptin and insulin sensitivity, promoting antioxidant defense and decreasing enzyme activities (AST and ALT). HFDC2 showed more protective effect as compared to HFDC1. That means cuminaldehyde showed the dose-dependent action in rats fed a HFD. Results (HFDC2) were comparable with orlistat, a standard drug. Thus cuminaldehyde (a natural bioactive compound) may be an excellent alternative strategy for developing effective and safe protective agent against NAFLD in the future.
Footnotes
Acknowledgements
The authors are thankful to Department of Pharmacognosy and Phytochemistry, Faculty of School of Pharmaceutical Education and Research for providing all basic facilities for this research work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present research study was supported by a grant (grant letter no. 3-17/2008-CCRUM/UPC, dated—23/04/2009) from Central Council for Research in Unani Medicine (CCRUM), Department of Ayurvedic, Yoga and Naturopathy, Unani, Siddha and Homeopathy (AYUSH), Ministry of Health & Family Welfare, Government of India.
