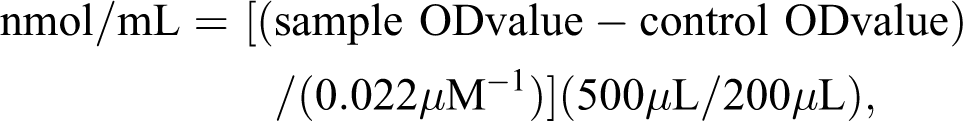Abstract
In the present study, we aimed to evaluate the hepatoprotective and antioxidant effects of Chunggan extract (CGX) in an animal model of hepatosteatosis. The C57BL/6N mice were fed either methionine- and choline-sufficient (MCS) diet (n = 10) or a methionine- and choline-deficient (MCD) diet (n = 50) for 4 weeks, and then they were treated orally with CGX (100 or 200 mg/kg), ursodeoxycholic acid (80 mg/kg, as a positive control), or distilled water (DW, MCS diet group, and MCD diet group) for the final 2 weeks (once per day). The MCD diet induced severe hepatic injury with the typical features of hepatosteatosis in both serum and hepatic tissues. CGX treatment significantly attenuated these alterations in the serum levels including triglyceride (TG), aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, and total bilirubin. Moreover, CGX also efficiently prevented from the hepatic TG accumulation in the hepatic tissue, evidenced by histopathological findings, compared with the MCD diet. In addition, CGX treatment significantly ameliorated the excessive oxidative stress and antioxidant markers in the serum as well as the hepatic levels of reactive oxygen species, the levels of malondialdehyde, the protein carbonyl, and total antioxidant capacity, and the activities of superoxide dismutase, catalase, glutathione peroxidase, and glutathione reductase. In conclusion, our results indicate the experimental relevance of CGX for potential clinical application in patients with hepatosteatotic disorders and a possible mechanism related to its antioxidant properties.
Keywords
Introduction
Along with the high prevalence of metabolic syndromes such as obesity, insulin resistance, and hyperlipidemia, nonalcoholic fatty liver disease (NAFLD) has become a health problem worldwide. 1 NAFLD can develop from simple steatosis to serious nonalcoholic steatohepatitis (NASH), liver cirrhosis due to the progression of fibrosis, or occasionally hepatocellular carcinoma. 2 NASH is characterized by fat accumulation, lobular inflammation, and ballooning degeneration in the hepatocytes in the absence of alcohol abuse. 3 Approximately 20% of NASH cases progress to cirrhosis. 4 The common factors that cause pathogenesis of NAFLD, including NASH, are known to be the excessive lipogenesis and disturbance of glucose homeostasis followed by oxidative stress in the liver tissue. 5 Hepatic accumulation of fat leads to increased generation of reactive oxygen species (ROS), causing membrane lipid peroxidation, inflammatory responses, and overproduction of extracellular matrix. 6,7 Clinical studies have shown that several characteristic markers of oxidative stress, such as malondialdehyde (MDA) and 4-hydroxynonenal (4-HNE), are significantly increased among the NASH patients. Meanwhile, the levels of antioxidants, including glutathione (GSH) and superoxide dismutase (SOD), are reduced. 8,9 So far, various complications, such as insulin resistance and obesity, have been investigated in patients with NASH, and antioxidant supplements have been used as important remedies in studies of patients with NAFLD, including those with NASH. 10,11
Chunggan extract (CGX) is a traditional hepatotherapeutic herbal medicine that has the name that indicates “cleaning the liver.” This drug has been used for patients suffering from various hepatic disorders such as chronic viral types B and C hepatitis, alcoholic liver diseases, and hepatic cirrhosis. CGX has been reported to show hepatotherapeutic effects in several animal models, and the antioxidant properties were shown to be the central activities of its pharmaceutical actions. 12 –14 In addition, CGX has shown positive effect on hyperlipidemia in high-fat diet animal experiments. 15 Moreover, the safety and toxicity study of CGX was already studied in beagle dog and rat models. 16,17 Accordingly, it is strongly assumed that CGX would be effective in NASH-related hepatic disorders.
The animal models that have been used for studying drugs against NASH are based on genetic defects (ob/ob mice or Fa/Fa rats) or feeding a methionine- and choline-deficient (MCD) diet in rodents. 18 The MCD diet model has been verified as more suitable experiment model to study the human disorder than the ob/ob mouse model, especially with respect to the histopathological features of severe pericentral steatosis and necroinflammation as well as severe oxidative stress, which eventually progresses to fibrosis. 19 –21
This study aimed to evaluate the hepatoprotective and antioxidant effects of CGX in an MCD diet-induced hepatosteatosis animal model.
Materials and methods
Reagents and chemicals
Ursodeoxycholic acid (UDCA) and other reagents, including p-dimethylaminobenzaldehyde, 1,1,3,3-tetraethoxypropane (TEP), chloramines-T, potassium chloride (KCl), n-butanol, N,N-diethyl-paraphenylenediamine sulfate (DEPPD), ferrous sulfate, trichloroacetic acid (TCA), 5,5-dithiobis-(2-nitrobenzoic acid) (DTNB), potassium phosphate, reduced glutathione, myoglobin, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS), GSH, glutathione reductase (GSH-Rd), glutathione peroxidase (GSH-Px),
Preparation of CGX
CGX was prepared according to the process described in over-the-counter Korean monographs by Kyoungbang Pharmacy (Incheon, Korea). CGX consists of 13 herbs, including 5 g each of Artemisiae capillaris herba, Trionycis carapax, and Raphani semen; 3 g each of Atractylodis rhizoma alba, Poria, Alismatis rhizoma, Atractylodis rhizoma, and Salviae miltiorrhizae radix; 2 g each of Polyporus, Amomi fructus, and Poncirus fructus; and 1 g of Glycyrrhizae radix and Aucklandiae radix. Briefly, 120 kg of the 13-herb mixture was boiled in 1200 L of distilled water for 4 h at 100°C, filtered using a 300-mesh filter (50 μm), condensed, and then lyophilized. All herbs used in the CGX formulation satisfied the Korean Pharmacopoeia criteria, and the CGX extract satisfied the criteria using a confirmation test for each herb, heavy metals, general bacteria, fungi, and specific pathogens. The final yield was 10.1% (w/w) from the original dried mixture. Lyophilized CGX extract (100 mg) was dissolved in 50% of methanol (20 mL) and mixed. The solution was filtered through an Acrodisc® LC 13 mm Syringe filter (0.45 μm pore size; Pall Life Science, Ann Arbor, Michigan, USA).
Quantitative analysis of CGX using high-performance liquid chromatography
The reference standard stock solutions of scopoletin, liquiritin, naringin, esculetin, rosmarinic acid, salvianolic acid B, poncirin, glycyrrhizin, and tanshinone IIA (200 μg/mL) were prepared in methanol and stored at <4°C. The standard solutions were prepared by six concentrations of diluted solutions (methanol). All calibration curves were attained by assessing the peak areas for six concentrations in the range of 3.13–100 μg/mL for all standard samples. The linearity of the peak area (y) versus concentration (x, μg/mL) curve for each component was used to calculate the contents of main components in CGX. The quantitative analysis was performed under the simultaneous conditions using a 1100 series high-performance liquid chromatography (HPLC; Agilent Technologies, Santa Clara, California, USA) equipped with an autosampler (G11313A), column oven (GA1316A), binary pump (G1312), diode-array detector, and degasser (GA1379A). The analytical column with a Gemini C18 (4.6 × 250 mm; particle size 5 μm; Phenomenex, Torrance, California, USA) was kept at 30°C during performance. The data were acquired and processed by chemstation software (Agilent Technologies). The mobile phase conditions contained 10% acetonitrile in water with 0.05% formic acid (A) and 90% acetonitrile in water (B). The gradient flow was as follows: 0–30 min, 0–20% B; 30–50 min, 20–75% B; 50–60 min, 75–100%. The analysis was operated at a flow rate of 1.0 mL/min and detected at 280 nm. The injection volume was 10 μL.
Animals and experimental design
Male 6-week-old C57BL/6N mice were purchased from a commercial animal breeder (Koatk, Gyeonggi-do, Korea). All animals were acclimated for 1 week and housed in an environmentally controlled room at 22 ± 2°C with a 12-h light–dark cycle. Commercial pellets (Gibio) and tap water were provided ad libitum. The animal experiments were conducted in accordance with protocols approved by the Institutional Animal Care and Use Committee of Daejeon University (DJUARB2011-031). The MCD diet and the methionine- and choline-sufficient (MCS) diet were purchased from Dyets Inc. (#518810 and #518754, respectively; Bethlehem, Pennsylvania, USA).
The mice (n = 60) were fed either the MCS diet or the MCD diet ad libitum for 4 weeks and divided into six groups (10 animals each): the MCS diet group, the MCD 2-week group (for confirmation of the development of hepatic injury), the MCD 4-week, the CGX (0, 100 or 200 mg/kg) group, and the UDCA (80 mg/kg, as a positive control) group. CGX (100 or 200 mg/kg), UDCA (80 mg/kg), or distilled water (DW, MCS diet, and MCD diet group) were orally administered daily for the final 2 weeks of the MCD diet feeding experiment. On the last experimental day, all mice were killed by collecting whole blood via the inferior vena cava under ether anesthesia after 5 h of fasting. The livers were removed and their weights were measured. Portions of the liver tissues were fixed with 10% formalin solution or individually frozen under −70°C for histopathological, biochemical, or molecular study.
Serum biochemical analysis
Serum was prepared following blood clotting. The serum levels of aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), total bilirubin, total protein, albumin, total cholesterol, triglyceride, and glucose were determined using an Auto Chemistry Analyzer (AU400; Olympus, Tokyo, Japan).
Histopathological and immunohistochemical analysis
For the histopathological evaluation, the liver tissues were fixed with 10% formalin solution and embedded in paraffin. The sections (4 μm thick) were stained with hematoxylin and eosin (H&E), and the routine frozen sections (7 μm thick) were stained with oil red O (Sigma). For immunohistochemistry against 4-HNE (1:200; Abcam, Cambridge, UK), the sections were incubated with 4-HNE primary antibody and biotinylated secondary antibody (Nichirei Biosciences, Tokyo, Japan) followed by the avidin–biotin–peroxidase complex. The immunoreactive signal was developed using their substrates, 3-amino-9-ethylcarbazole (Abcam). The slides were counterstained with hematoxylin (Sigma) and examined under an optical microscope (Leica Microsystems, Wetzlar, Germany).
Measurement of hepatic triglyceride
The hepatic lipid was extracted according to Folch et al. 22 Briefly, 50 mg of liver tissue was homogenized in 1 mL of a chloroform–methanol mixture (2:1, v/v). After centrifugation at 10,000g for 15 min, the supernatant was dried at room temperature. Then, 200 μL of distilled water was added to all the samples. Next, the triglyceride levels were measured by routine methods using the appropriate commercial kit (Asan Pharmaceutical Co., Ltd, Seoul, Korea), according to the manufacturer’s instructions.
Measurement of hepatic lipid peroxidation
Hepatic MDA levels were determined using the method of thiobarbituric acid reactive substances (TBARS), as described previously. 23 The concentration of TBARS was expressed as mol MDA/g tissue using TEP as a standard. Briefly, 0.1 g of liver tissue was homogenized in 1 mL of ice-cold 1.15% KCl, and 130 μL of the homogenate was mixed with 80 μL of 1% phosphoric acid and 260 μL of 0.67% TBA. After heating the mixture for 45 min in an oven at 100°C, 1.03 mL of n-butanol was added, vigorous vortexed, and centrifuged at 3000 rpm for 15 min. The absorbance of the upper organic layer was measured at 535 and 520 nm, with a spectrophotometer (Molecular Device Corp., Sunnyvale, California, USA).
Measurement of hepatic protein oxidation
Hepatic protein carbonyl content was determined by detecting protein oxidation using the DNPH reaction, according to the manufacturer’s protocol (Cayman Chemical, Hamburg, Germany). Briefly, the liver tissue homogenate was prepared with cold phosphate buffer (50 mM, pH 6.7, containing 1 mM EDTA), and 200 μL of the homogenate was mixed with 800 μL of DNPH (10 mM dissolved in 2.5 M HCl). After incubation in the dark at room temperature for 1 h with vortexing every 15 min followed by the sequential addition of 1 mL of 20% TCA and 10% TCA and incubation for 5 min at 4°C, a pellet was obtained by centrifugation at 10,000g for 10 min at 4°C. After resuspension in 1 mL of an ethanol/ethyl acetate mixture (1:1, v/v) and centrifugation, the protein pellets were resuspended in 500 μL of guanidine hydrochloride by vortexing and centrifuged at 10,000g for 10 min at 4°C. Then, 220 μL of the supernatants were transferred to a 96-well plate. The absorbance was measured at 370 nm with a spectrophotometer (Molecular Device Corp). The carbonyl content was defined as
where OD is the optical density.
Determination of the level of ROS
The total ROS level in the serum and liver tissue was determined according to the modified method of Hayashi et al. 24 Briefly, H2O2 was used as a standard for generating a calibration curve. DEPPD solution and ferrous sulfate solution were prepared beforehand. For preparing a sample from liver tissue, 100 mg of hepatic tissue was homogenized in radio immunoprecipitation assay buffer and centrifuged at 10,000g for 10 min at 4°C, and then the supernatant was mixed with 2.8% TCA (1:1, v/v). Ten microliters of standard solution, serum, or hepatic sample were added to 600 μL of ferrous sulfate (4.37 μM) and 10 μL of DEPPD (100 μg/mL) dissolved in 0.1 M sodium acetate buffer (pH 4.8), and the samples were centrifuged at 3000g for 1 min at 37°C. After incubation for 10 min at 37°C and reincubation for 10 min at room temperature, the 200 μL aliquots were transferred to a 96-well plate, and the absorbance was measured at 505 nm using a microplate reader (Molecular Device Corp.). The protein concentration was determined using a bicinchoninic acid protein assay kit (Sigma).
Determination of total antioxidant capacity
The total antioxidant capacity (TAC) level in the liver tissue was determined according to the method of Kambayashi. 25 Briefly, 90 μL of phosphate-buffered saline (pH 7.2, 10 mM), 50 μL of myoglobin solution (18 μM), 20 μL of 3 mM ABTS solution, 20 μL of diluted liver tissue homogenized supernatant, and various concentrations of gallic acid (internal standard) were added to a 96-well plate and mixed well at 25°C for 3 min. Then, 20 μL of 250 μM H2O2 were added to each well and incubated for 5 min. The absorbance was read using a plate reader at 600 nm (Molecular Device Corp). The level of TAC was expressed as gallic acid-equivalent antioxidant capacity.
Determination of SOD and catalase
SOD activity in the liver tissue was determined using an assay kit (Dojindo Laboratories, Kumamoto, Japan), according to the manufacturer’s protocol. Bovine erythrocyte SOD (Sigma) was used as a standard.
Catalase activity in the liver tissues was determined using the method described by Johansson and Borg, 26 with H2O2 as the substrate. The 140 μL of phosphate buffer (250 mM, pH 7.0), 150 μL of 12 mM methanol and 30 μL of 44 mM H2O2 were in the 13 × 100 mm test tube. The reaction was initiated with 300 μL of samples or standard solutions. The reaction was allowed to proceed for 10 to 20 min and completed by the addition of 450 μL of Purpald solution (22.8 mM of Purpald in a 2 N potassium hydroxide). The incubation mixture was mixed gently in a vortex mixer and left for 20 min at 25°C. Added to 150 μL of potassium periodate (65.2 mM in 0.5 N potassium hydrate) at the same tube and the tube vortexes gently again. The absorbance of the purple formaldehyde adduct produced was measured at 550 nm using a spectrophotometer (Molecular Devices). The standard concentration was prepared using catalase ranging from 2000 to 0 activities.
Determination of the total GSH content and the GSH-Px and GSH-Rd activity
Total GSH in liver tissue was determined according to the method of Ellman. 27 Briefly, 50 μL aliquots of the supernatant or standard were combined with 80 μL of a previously prepared DTNB/NADPH mixture (10 μL of 4 mM DTNB and 70 μL of 0.3 mM NADPH) in a 96-well plate. Finally, 20 μL (0.06 U) of GSH-Rd solution was added to each well and incubated for 5 min, and the absorbance was measured at 405 nm after 5 min.
GSH-Px activity in the liver tissue was determined using a previously described method by Ursini et al. 28 Briefly, 50 μL of NADPH reagent (5 mM NADPH, 42 mM GSH, and 10 U/mL GSH-Rd in 1.25 mL of distilled water) was added to 890 μL of GSH-Px buffer (50 mM Tris–HCl, pH 8.0, 0.5 mM EDTA). Then, 50 μL of the lysate and 10 μL of 30 mM tert-butyl hydroperoxide solution were added, and the absorbance was measured at 505 nm using a UV/visible spectrophotometer (Varian; Agilent Technologies). Enzyme activity was defined as 1 U/mL = ΔA340 × dilution factor/(6.22 × sample volume in mL).
The GSH-Rd activity in the liver tissue was determined according to a previously described method by Smith et al. 29 Briefly, 150 μL of GSSG and 30 μL of GSH-Rd assay buffer (100 mM potassium phosphate, pH 7.5, 1 mM EDTA) were added to 30 μL of lysate in each well of a 96-well plate and diluted with 30 μL of GSH-Rd dilution buffer (GSH-Rd assay buffer supplemented with 1 mg/mL bovine serum albumin). Then, 75 μL of DTNB and 15 μL of 2 mM NADPH were added, and the absorbance was measured at 412 nm. Enzyme activity was defined as 1 U/mL = (ΔA sample − ΔA blank) × dilution factor/(εmM × sample volume in mL), where εmM = 14.15 mM−1 cm−1.
Statistical analysis
The results were expressed as the mean ± SD (n = 10). The statistical significance of differences among groups was analyzed by one-way analysis of variance followed by Student’s unpaired t test. Differences at the levels of p < 0.05 and p < 0.01 were regarded as statistically significant.
Results
Fingerprinting analysis of CGX
The standard curves for the nine components containing scopoletin, liquiritin, naringin, esculetin, rosmarinic acid, salvianolic acid B, poncirin, glycyrrhizin, and tanshinone IIA were y = 11.887x − 8.247 (R 2 = 0.999), y = 16.447x − 11.451 (R2= 0.999), y = 15.211x − 11.069 (R 2 = 0.999), y = 11.713x − 9.079 (R 2 = 0.999), y = 17.497x − 16.149 (R 2 = 0.999), y = 7.937x − 16.028 (R 2 = 0.998), y = 17.155x − 8.843 (R 2 = 0.999), y = 0.728x − 0.245 (R 2 = 0.991), and y = 29.448x − 21.802 (R 2 = 0.999), respectively. HPLC analysis of CGX and standard mixtures was carried out at 280 nm (Figure 1(a) and (b)). The retention time of each compound was 22.13 min (scopoletin), 23.16 min (liquiritin), 28.30 min (naringin), 31.21 min (esculetin), 32.05 min (rosmarinic acid), 37.63 min (salvianolic acid B), 38.22 min (poncirin), 46.38 min (glycyrrhizin), and 59.12 min (tanshinone IIA). The contents of each component were in the range of 1.823–228.79 μg/g (Figure 1(c)).

Fingerprinting analysis of CGX. The CGX and its nine main compounds were subjected to HPLC. All chromatograms were obtained at a wavelength of 280 nm (details in the text). Chromatogram of CGX (a), chromatogram of standard mixtures (b) and the contents of each reference compounds in CGX (c). CGX: Chunggan extract; HPLC: high-performance liquid chromatography.
Body and organ weights
The 4-week MCD diet remarkably decreased body weight (p < 0.01) and liver weight (p > 0.05), while the absolute spleen weight increased (p > 0.05) compared with the MCS diet. The CGX treatment did not show a significant effect on the body weight or the absolute liver and spleen weight, while the changes in the relative liver weight and the relative spleen weight were significantly ameliorated by CGX treatment (200 mg/kg) compared with the MCD 4-week diet (p < 0.05). UDCA treatment showed a similar effect on the relative liver and spleen weights with CGX (p < 0.05). However, no significant change was observed for body weight after treatment with CGX or UDCA (Table 1).
Change in body, liver, and spleen weights and serum biochemistry.a
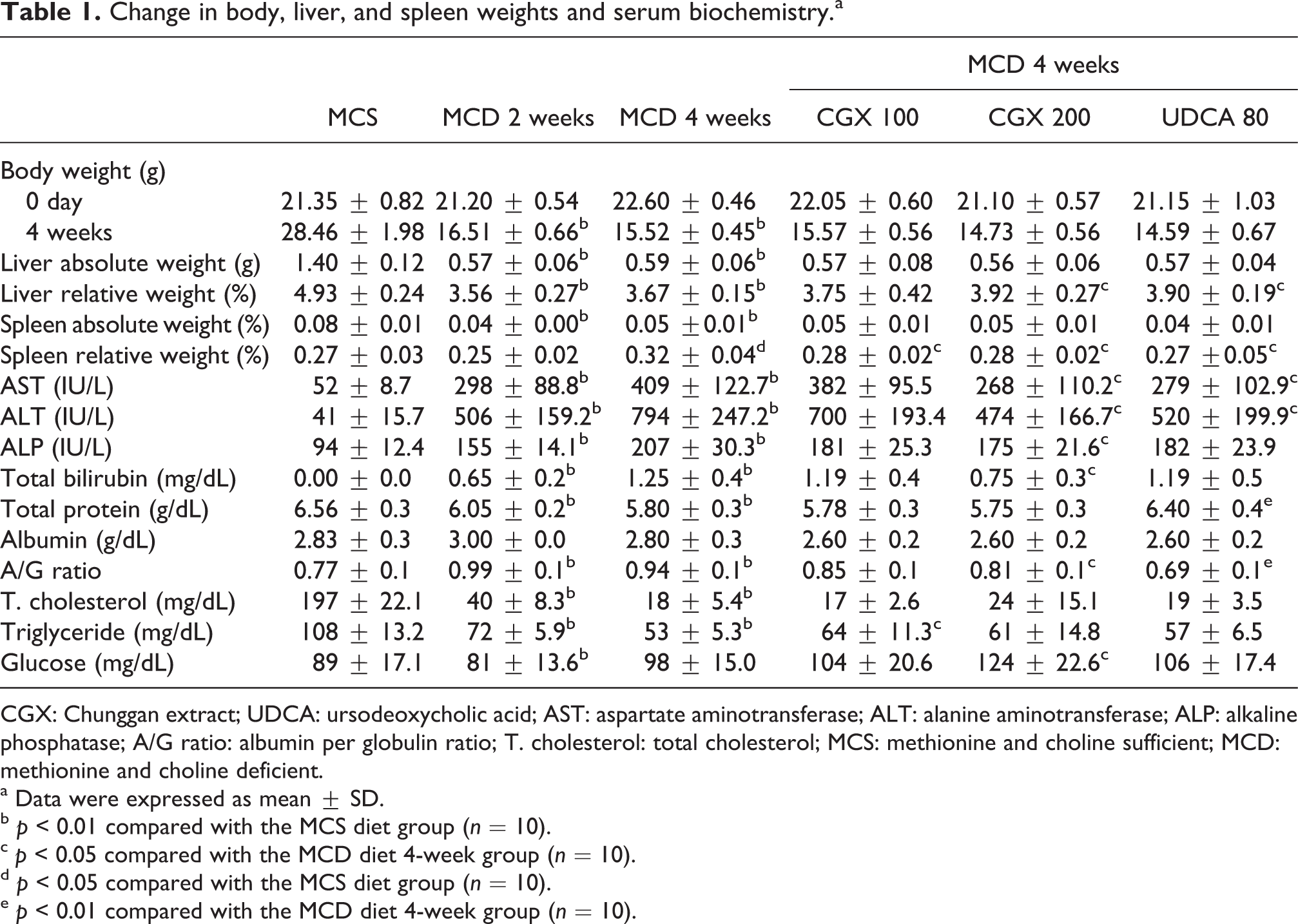
CGX: Chunggan extract; UDCA: ursodeoxycholic acid; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; A/G ratio: albumin per globulin ratio; T. cholesterol: total cholesterol; MCS: methionine and choline sufficient; MCD: methionine and choline deficient.
a Data were expressed as mean ± SD.
b p < 0.01 compared with the MCS diet group (n = 10).
c p < 0.05 compared with the MCD diet 4-week group (n = 10).
d p < 0.05 compared with the MCS diet group (n = 10).
e p < 0.01 compared with the MCD diet 4-week group (n = 10).
Serum biochemistry
The 4-week MCD diet markedly increased the serum levels of AST, ALT, ALP, and total bilirubin by 2- to 10-fold compared with the MCS diet. The CGX treatment significantly attenuated the alterations in AST, ALT, ALP, and total bilirubin compared with the MCD 4-week diet in a dose-dependent manner (p < 0.05, CGX 200 mg/kg). The serum levels of total protein, total cholesterol, triglyceride, and glucose were notably lowered by the 4-week MCD diet, whereas the CGX treatment significantly ameliorated the changes in the triglyceride (p < 0.05, CGX 100 mg/kg) and glucose levels (p < 0.05, CGX 200 mg/kg). The increase in the A/G ratio as a result of the MCD diet was significantly reduced by CGX treatment compared with the MCD 4-week diet (p < 0.05). UDCA treatment also significantly attenuated the changes in AST, ALT, total protein, and the A/G ratio (p < 0.05 or 0.01, Table 1).
Histopathological and immunohistochemical findings
The 4-week MCD diet led to apparent vacuolization of hepatocytes, focal necrosis, and inflammatory cell accumulation in liver tissue under H&E staining. In addition, oil red O staining and immunostaining indicated an increased fat accumulation and a robust increase in the 4-HNE signal in the liver tissue as a result of the 4-week MCD diet. These pathological changes were considerably ameliorated by CGX treatment (especially by 200 mg/kg) compared with the MCD 4-week diet (Figure 2(a) to (c)). UDCA treatment also showed a slight improvement in the histopathological findings, especially in the 4-HNE immunostaining.

Histopathological examination. C57BL/6N mice (10 per group) were fed with the MCS or MCD diet for 4 weeks with CGX (100, 200 mg/kg), UDCA (80 mg/kg), or distilled water for the final 2 weeks. On the final day of the experiment, all of the livers in each group were fixed with 10% formalin solution. After staining with hematoxylin and eosin (a), oil red O (b), and immunohistochemistry for 4-hydroxynonenal (c), the histological findings were examined under an optical microscope (magnification ×200). Black arrows indicate the balloon shapes (a), the white arrows indicate the oil red O staining (b) and 4-HNE (b). MCS: methionine and choline sufficient; MCD: methionine and choline deficient; CGX: Chunggan extract; UDCA: ursodeoxycholic acid; 4-HNE: 4-hydroxynonenal.
Triglyceride, MDA, and protein carbonyl contents in liver tissue
The 4-week MCD diet dramatically increased the hepatic triglyceride, MDA, and protein carbonyl contents by 1.7- to 25-fold compared with the MCS diet. However, the CGX treatment (200 mg/kg) significantly attenuated the changes in the levels of triglyceride (p < 0.01), MDA (p < 0.05), and protein carbonyl (p < 0.05) compared with the MCD 4-week diet. UDCA treatment also significantly attenuated the alterations in the MDA and protein carbonyl levels (p < 0.05 or 0.01, Figure 3(a) to (c)).

Measurement of the levels of triglyceride, MDA, protein carbonyl, and ROS. C57BL/6N mice (10 per group) were fed the MCS or MCD diet for 4 weeks with CGX (100, 200 mg/kg), UDCA (80 mg/kg), or distilled water for the final 2 weeks. The hepatic triglyceride (a), MDA (b), and protein carbonyl levels (c), and the ROS (d) in serum and liver tissue were determined. # p < 0.05 and ## p < 0.01 compared with the MCS diet group. * p < 0.05 and ** p < 0.01 compared with the MCD diet 4-week group. MCS: methionine and choline sufficient; MCD: methionine and choline deficient; CGX: Chunggan extract; UDCA: ursodeoxycholic acid; ROS: reactive oxygen species; MDA: malondialdehyde.
ROS levels in the serum and liver tissue
The 4-week MCD diet significantly increased the serum ROS level by approximately 1.5-fold as well as the hepatic ROS level by approximately 3-fold compared with the MCS diet. However, CGX treatment significantly attenuated the increase in the ROS level (p < 0.05) in the serum and hepatic tissue (p < 0.01 or p < 0.05). UDCA treatment showed positive effects on the only serum ROS level (p < 0.05, Figure 3(d)).
TAC, SOD, and catalase activity in liver tissue
The 4-week MCD diet significantly depleted the antioxidant parameters, such as TAC, SOD, and catalase activity in the liver tissue, compared with the MCS diet. The CGX treatment significantly ameliorated the alteration of TAC (p < 0.01, CGX 200 mg/kg), SOD activity (p < 0.05, CGX 200 mg/kg), and catalase activity (p < 0.01) compared with the MCD 4-week diet. UDCA treatment showed a significant effect only on catalase activity (p < 0.01, Table 2).
Antioxidant parameters in liver tissue.a

CGX: Chunggan extract; UDCA: ursodeoxycholic acid; TAC: total antioxidant capacity; SOD: superoxide dismutase; GSH: glutathione; GSH-Px: glutathione peroxidase; GSH-Rd: glutathione reductase; MCS: methionine- and choline-sufficient; MCD: methionine- and choline-deficient.
a Data are expressed as mean ± SD.
b p < 0.05 compared with the MCS diet group (n = 10).
c p < 0.01 compared with the MCS diet group (n = 10).
d p < 0.01 compared with the MCD diet 4-week group (n = 10).
e p < 0.05 compared with the MCD diet 4-week group (n = 10).
Total GSH content and GSH-Px and GSH-Rd activity in the liver tissue
The 4-week MCD diet markedly depleted the hepatic GSH content and GSH-Rd activity in the liver tissue compared with the MCS diet. The CGX treatment (100 mg/kg) significantly ameliorated the alteration in the GSH-Rd activity but not the GSH content (p > 0.05). The GSH-Px activity slightly decreased on the 4-week of MCD diet (p > 0.05), while the CGX treatment (200 mg/kg) significantly increased the activity of GSH-Px compared with the MCD 4-week diet (p < 0.05). UDCA treatment did not show any significant effect on those parameters (Table 2).
Discussion
Due to changes in the modern lifestyle such as overnutrition, reduced physical exercise and frequent physiological stress, the population with metabolic syndrome is increasing worldwide. 1 NAFLD and NASH are considered as a liver-dominant pathologic status of metabolic syndrome. 30 Therefore, NAFLD and NASH have become important medical issues because of their epidemic proportions in the developed countries. 31 No effective therapeutics has yet been developed. However, there are many pharmaceutical studies of NAFLD and NASH using antidiabetic and hypolipidemic therapies and antioxidants in addition to lifestyle modification. 32 Furthermore, the use of herb-derived candidates or food supplements has been a major strategy for NAFLD and NASH. 33
CGX is a hepatotherapeutic herbal drug that has been used to treat patients with mainly alcoholic and viral hepatic disorders. 34 Our previous studies have demonstrated the hepatotherapeutic effects of CGX in several animal models with acute or chronic liver injuries caused by toxic chemicals 12,13 or immunological agents. 14 CGX also showed a positive effect on hyperglycemia in animal experiments, and it has been applied in patients with NASH. 15 Previous studies provide sufficient empirical evidence that CGX has proved its safeness and nontoxicological properties in beagle dong and rodent models. 16,17 To support the clinical application of CGX in the treatment of NASH-like disorders, this study investigated the hepatotherapeutic effect of CGX in an animal model of MCD diet-induced hepatic injury.
The MCD diet, which is deficient in two essential amino acids, methionine and choline, impairs the synthesis of phosphatidylcholine, which plays an important role as the phospholipid component for hepatic β-oxidation and very-low-density lipoprotein production in rodent models. 35 The MCD diet leads to hepatic lipid accumulation and peroxidation followed by inflammation and fibrosis, which shows histological features similar to those of human NASH. 7,20 Consistent with our expectation, the MCD diet led to apparent hepatic injury with inflammatory cell infiltration as well as fat accumulation in the liver tissue under H&E and oil red O staining, and these histological alterations became worse along the duration of the MCD diet. The severity was more notable in the 4-week MCD group than in the 2-week group. These histopathological findings were considerably ameliorated by CGX treatment compared with the MCD 4-week diet, and this effect was supported by our findings regarding the hepatic enzymes, including AST, ALT, ALP, and total bilirubin, especially in the group that received 200 mg of CGX (Figure 2(a), (b) and Table 1). The deterioration of liver tissue and liver function led to a small liver size and weight loss, which were improved by CGX treatment.
The obvious difference between human NASH and the MCD diet-induced model is that the MCD diet does not produce the typical human metabolic syndrome involving insulin resistance, hyperlipidemia, and obesity. 36 The MCD diet significantly induced the accumulation of triglycerides; however, it drastically decreased the body and liver weight and lowered the serum levels of total cholesterol and triglycerides compared with the MCS diet. One clinical study reported that 51% of patients with NASH have hyperlipidemia and 84% are obese. 37 Furthermore, 20% of NASH patients may progress into liver cirrhosis. 4 Our animal model did not progress based on the hepatic cirrhotic changes, limiting its comparability with human NASH. However, CGX produced pharmaceutical effects on hepatic triglyceride levels and alterations in the relative liver and spleen weight (Table 1 and Figure 3(a)).
It is well known that oxidative stress is produced under steatosis conditions and plays a key role in the development and progression of NASH. 6,7 Excessive ROS production can lead to biological damage of lipids, proteins, and nucleic acids. 38 Our study showed the drastic elevation of a typical oxidative stressor (ROS in hepatic tissue) and its peroxidation products, MDA as a lipid peroxidation, and protein carbonyl as a protein peroxidation (Figure 3(b) to (d)). The hepatic oxidative stress status was also evidenced by immunohistochemistry against 4-HNE (Figure 2(c)). 4-HNE is a lipid peroxidation product in the cell, and its levels were elevated in human studies of NASH. 39 The hepatic alterations described above were significantly attenuated by CGX treatment as evidenced by the results for oxidative stress-related parameters, such as ROS, MDA, and protein carbonyl as well as by the 4-HNE staining (Figures 2 and 3(b) to (d)). MDA and HNE can induce mitochondrial dysfunction by inhibiting mitochondrial respiration and activating hepatic stellate cells, along with promoting collagen synthesis and fibrosis among individuals with NASH. 40 Antioxidants, such as vitamin E and C, improved hepatic injury and lipid peroxidation in an MCD diet-induced NASH model. 41 The antioxidant effects of CGX have been proved in several animal models and in vitro experiments. CGX illustrated protective properties against strong chemotoxins-induced oxidative stress in both hepatocytes and hepatic tissues. 12,13 Moreover, CGX significantly inhibited the production of nitro oxide and suppressed the inducible nitro-oxide synthase gene in hepatic tissue as well as hepG2 cell line. 42
Oxidative stress occurs when oxidative stressors overwhelm the defense system over antioxidant. The antioxidant defense mechanism includes enzyme-based antioxidant systems and nonenzyme antioxidant molecules, such as SOD, catalase, and the GSH redox system, as well as vitamin C and E. 43 SOD is an essential enzyme for an effective defense against reactive oxygen species, and it accelerates the formation of H2O2 using superoxide radicals. 44 Furthermore, the catalase and the GSH redox systems play a central role in accelerating the degradation of unstable H2O2 to water and oxygen. 45 It has been reported that the levels of these antioxidant parameters are decreased in the livers of patients with NASH. 46 Consistent with our expectation, the MCD diet significantly diminished the antioxidant-associated parameter levels, including TAC, SOD, catalase, and GSH-redox enzymes dependent on the MCD feeding period, whereas these depletions were significantly improved by GSH treatment.
Several groups have reported the positive effects on NAFLD or NASH in clinical trials using herbal drugs such as the Qianggan capsule, 47 the tiaozhi yanggan decoction, 48 and Yo Jyo Hen Shi Ko. 49 CGX also presented antihepatosteatosis and antioxidative stress effects in the present study. However, the major active compounds in the CGX were not identified. Most of the herbal medicines usually contain complex ingredients, so it is important to figure out the toxicological studies before clinical trials. Before experiment, we already obtained the safeness and nontoxicological study of CGX. 16,17 In the present study, we assumed that the ingredients of CGX may act on multiple targets that may help cure NAFLD or NASH, just like how multidrug therapies are used for multitarget pathways in metabolic syndrome. 50,51
Taken together, this study demonstrated the hepatotherapeutic effect of CGX in an MCD diet-induced NASH animal model, and the antioxidant action may be a possible mechanism that explains the effect.
Footnotes
Conflict of Interest
The authors declared no conflicts of interests.
Funding
This research was supported by the Oriental Medicine Research and Development Project, Ministry of Health and Welfare, Republic of Korea (No. B120047).