Abstract
Visual system toxicity may manifest anywhere in the visual system, from the eye proper to the visual brain. Therefore, effective screening for visual system toxicity must evaluate not only ocular structures (ie, eye and optic nerve) but also multiple key brain regions involved in vision (eg, optic tract, subcortical relay nuclei, and primary and secondary visual cortices). Despite a generally comparable pattern across species, the neuroanatomic organization and function of the visual brain in rodents and rabbits exhibit appreciable differences relative to nonrodents. Currently recognized sampling practices for general toxicity studies in animals, which are based on easily discerned external neuroanatomic landmarks and guided by extant stereotaxic brain atlases, typically will permit histopathologic evaluation of many brain centers involved in visual sensation (eg, optic chiasm, optic tract, dorsal lateral geniculate nucleus, primary and secondary visual cortices) and often some subcortical brain nuclei involved in light-modulated nonvisual activities needed for visual attention and orientation (eg, rostral colliculus in quadrupeds, termed the superior colliculus in bipeds; several cranial nerve nuclei). Pathologic findings induced by toxicants in the visual brain centers are similar to those that are produced in other brain regions.
Introduction
Traditionally, evaluations of visual system toxicity during general toxicity studies in laboratory animals have emphasized assessment of ocular structures (ie, eye and intraorbital optic nerve) along with examination of the visual fields found in the occipital pole of the cerebral cortex in routinely sampled brain sections. 1 Details of neuroanatomy, neurochemistry, neurophysiology, and evaluation of neuropathologic responses to toxicant exposure in animals have been described elsewhere for both the neural eye (retina and optic nerve) 2- 6 and visual cortex. 7- 10
Although the receiver function of the retina is critical to vision, visual perception requires delivery and integration of retinal impulses within the central nervous system (CNS) before interpretation of light-related information can occur. For example, diurnally fluctuating light levels are essential signals that regulate multiple light-related nonvisual activities within other brain centers. For this reason, the evaluation of ocular neurotoxicity involves not only a thorough examination of the retina and optic nerve but also a survey of major central (brain) pathways to which retinal input is projected. This review will (1) propose a pragmatic strategy for assessing the integrity of central visual pathways in rodents and nonrodents appropriate for use in microscopic evaluation during general toxicity studies in mammalian test species and (2) briefly describe the neuroanatomy and neuropathology of the primary brain regions and circuits that participate in light-based visual and nonvisual information processing in mammals.
Practical Screening of Extraocular Light-Related Organs
Conventional Good Laboratory Practice (GLP)-compliant animal toxicity studies typically include evaluation of the eyes (including retinas), optic nerves, and occipital (visual) cortex but rarely incorporate specific evaluation of other key CNS centers involved in visual perception and light-related, nonvisual (ie, nonimage-forming) functions. 1 Nonetheless, even routine brain sampling schemes employed in common test species will include many visual and light-related, nonvisual centers. 1,11,12 The difficulty for most pathologists is that these brain centers cannot be identified without frequent reference to well-annotated, species-specific neuroanatomy atlases, 13- 19 which may slow the pace at which brain sections can be evaluated. This section briefly reviews brain trimming schemes for effective sampling of the most important central visual and light-related nonvisual brain centers in rodents and nonrodents.
In general, the authors believe that screening for potential extraocular visual neurotoxicity will be effective if sections show the features of the major visual sensory and light-related nonvisual brain centers already evident within currently recommended levels for brain sampling. 1,20 This approach should be suitable for screening visual and some larger light-related nonvisual brain centers during GLP-compliant and, where brain is sampled, non-GLP-type animal studies. Fixed brains in all test animal species are sliced using external landmarks on the ventral brain surface to correctly position the trimming planes. To a first order of approximation, similar neuroanatomic regions are visible in coronal sections of rat brain (Figure 1) and coronal hemisections of nonrodent brain (Figure 2). In rats, conventional brain sections, depending on the level, may demonstrate (from rostral to caudal) the optic tract, dorsal lateral geniculate nucleus (LGN), several pretectal nuclei, rostral colliculus (RC) or superior colliculus (SC; RC in quadrupeds, SC in bipeds; NOTE 1 ), somatic and autonomic nuclei associated with the oculomotor nerve (cranial nerve [cn] 3), and the primary and secondary visual cortices (Figure 3). 1 In nonrodents, recommended brain sections may show the optic chiasm, optic tract, dorsal LGN, several pretectal nuclei (not shown), and the primary and secondary visual cortices (Figure 4). 1,11 Other visual structures may require acquisition and processing of additional (typically 1-2) brain sections, especially in nonrodent brains. The optic chiasm is readily identified on the ventral surface of the brain and thus may be harvested easily when necessary. This same section also may include 2 light-related nonvisual nuclei—the suprachiasmatic nucleus (SCN) and ventrolateral preoptic nucleus (VLPO, Figures 3 and 4)—located just dorsal to the optic chiasm; however, their presence is not certain due to their small volume, so additional sectioning might be necessary to ensure that these 2 nuclei are available for analysis. Careful trimming based on external and internal landmarks can be used to find the pineal gland (PIN). The various motor nuclei of other cranial (trochlear [cn4] and abducens [cn6]) nerves that regulate visual acuity by innervating the remaining extraocular muscles are small and typically will not be available for review in standard screening sections. These nuclei may be sought by taking brain sections that contain internal landmarks visible in coronal sections of species-specific stereotaxic brain atlases for rats, 15,21 rabbits, 12,13 dogs, 19,22 pigs, 14 and nonhuman primates. 16,17,23 Although ferrets (Mustela putorius) are not a common test species for nonclinical toxicity studies, their frequent use to explore anatomical and physiological characteristics of visual system development warrants mention as an additional animal model for ocular biology research and very specialized toxicity testing. 18,24,25
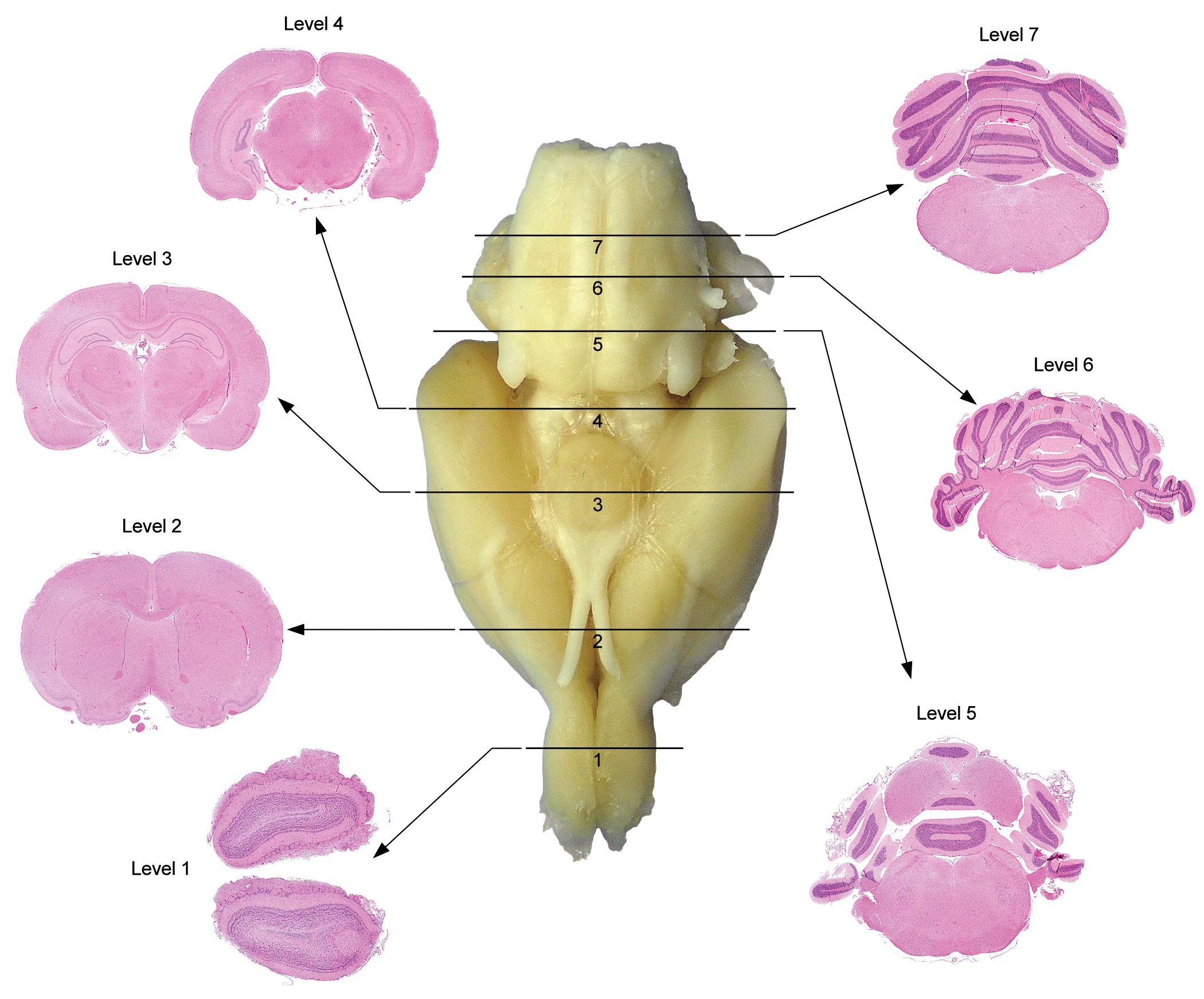
Best practice brain sampling scheme to consistently demonstrate major domains in the adult rat brain during general toxicity studies. [Image reproduced from Bolon et al, 1 by permission of Sage Publications.]
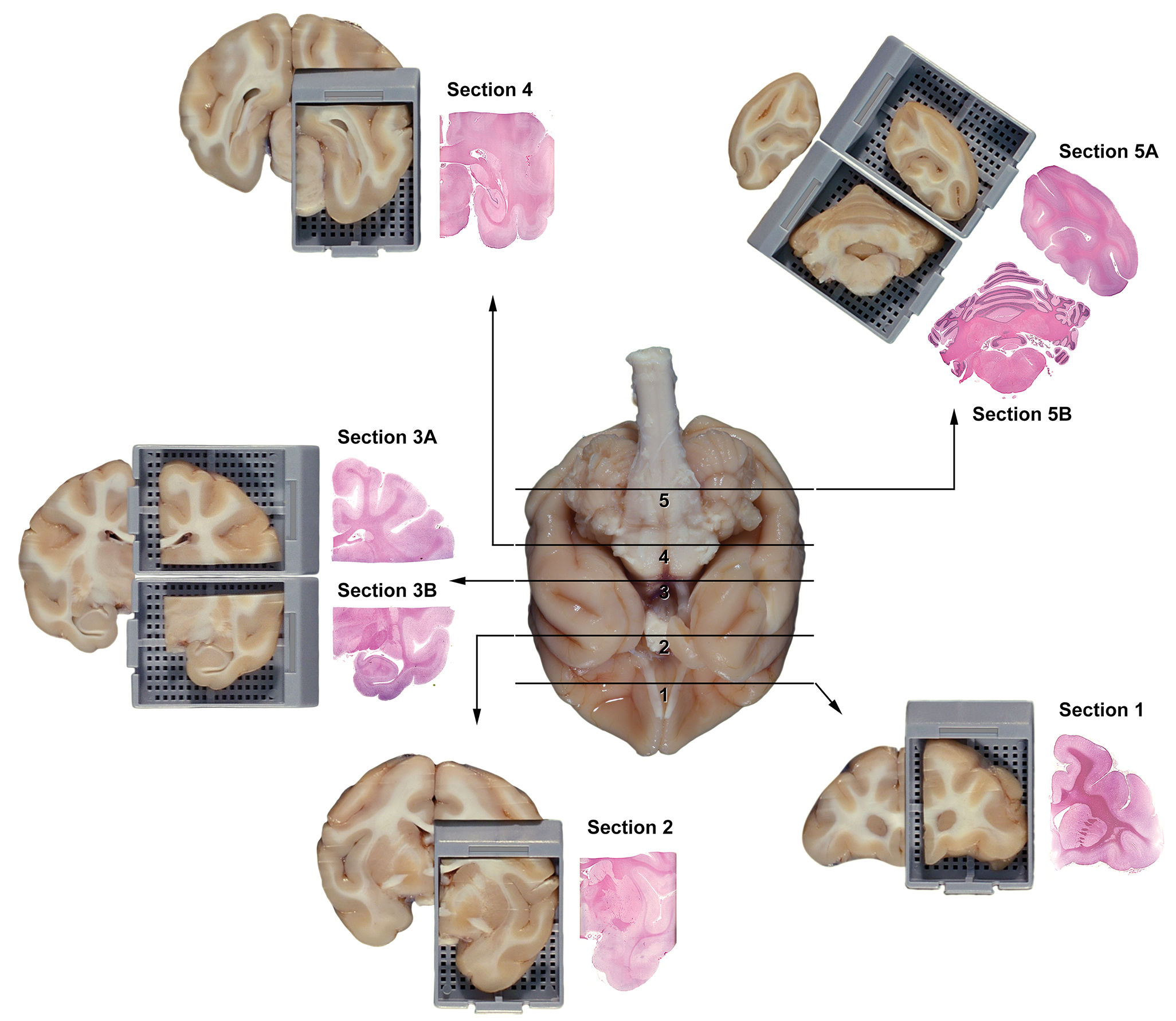
Best practice brain sampling scheme to reliably trim the nonhuman primate (cynomolgus macaque) brain during general toxicity studies. [Image reproduced from Bolon et al, 1 by permission of Sage Publications.]
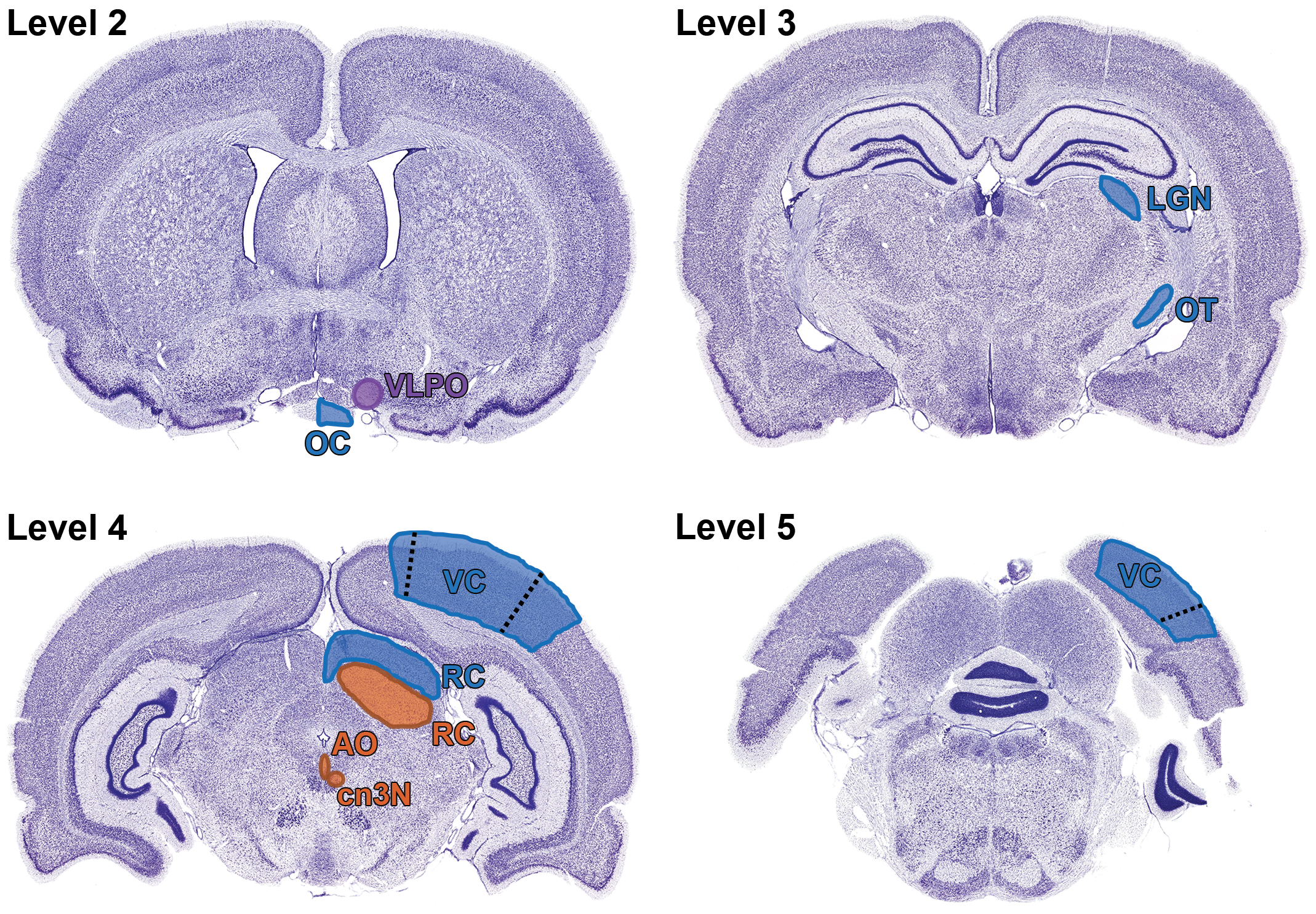
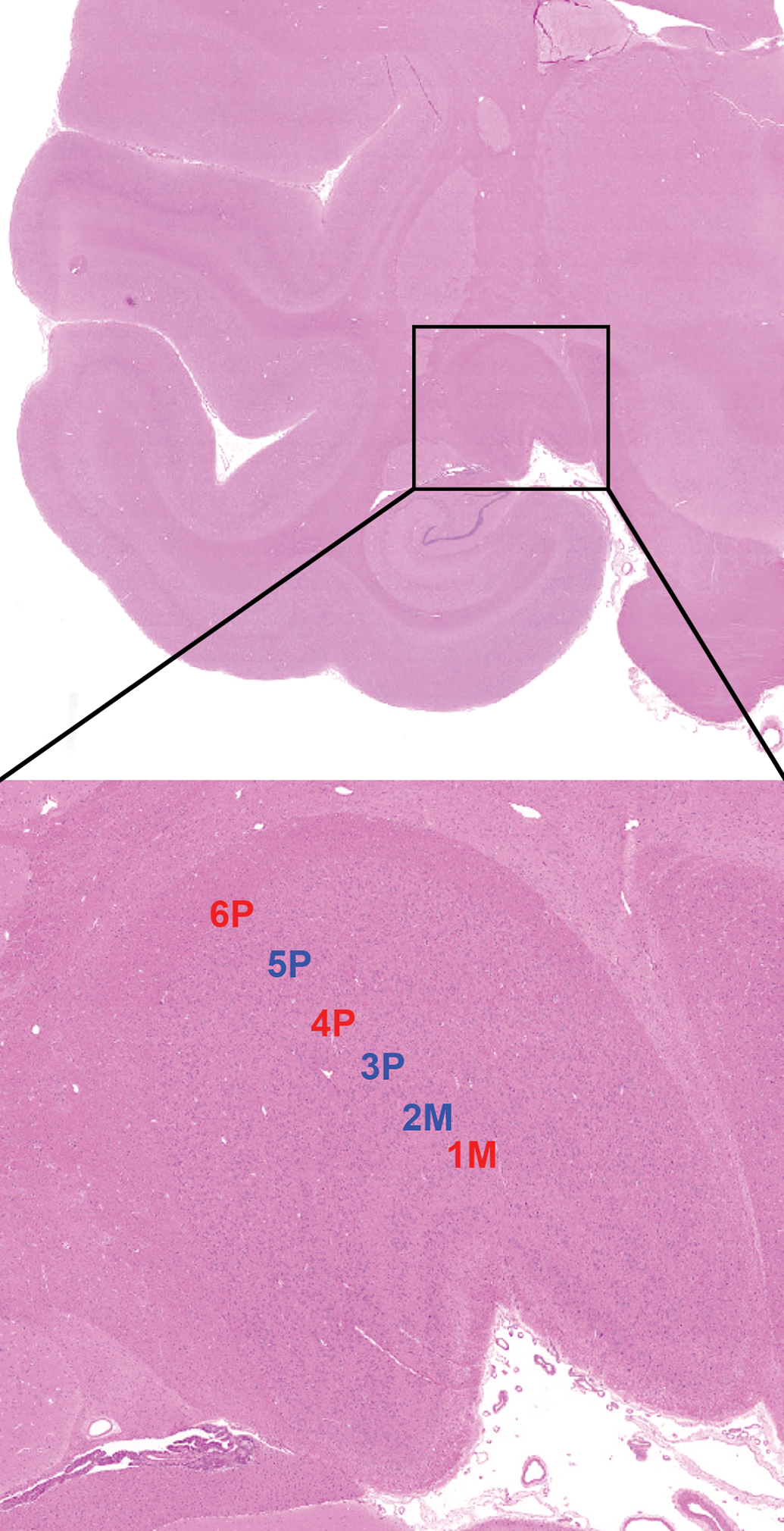
Relevant neuroanatomic areas involved in visual sensory (blue), visual motor (orange), and visual circadian/sleep (purple) pathways in the rat as seen in standard sections acquired by current “best practice” recommendations for sampling the nonrodent brain during general toxicity studies (detailed in Bolon et al 1 ). Level 2 is a section taken through the rostral commissure showing the optic chiasm (OC) and approximate location of the ventrolateral preoptic nucleus (VLPO); the suprachiasmatic nucleus (SCN) is not shown because it usually is slightly caudal to the illustrated trimming plane and so will not be present in all sections taken at this level. Level 3 shows a section through the diencephalon showing the optic tract (OT) as it courses toward the lateral geniculate nucleus (LGN). Level 4 is a section through the midbrain showing the locations of the sensory (dorsal) and motor (ventral) portions of the rostral colliculus (RC) relative to the small oculomotor nucleus (cn3N) and accessory oculomotor (or Edinger-Westphal) nucleus (AO); these 2 very small nuclei also may be absent depending on the trimming plane. The visual cortex is evident in the overlying cerebral cortex, where the primary visual field is annotated (as VC) and the blue-shaded areas demarcated by dashed lines represent the secondary visual centers. Level 5 is a section of the rostral hindbrain showing the locations of the primary visual field (labeled as VC) and the secondary visual zones (separated from the label by a dashed line). Nissl stain (selected since this better serves to highlight margins between adjacent populations of neurons). Adult rat. Nissl stain (used to highlight discrete neuron populations).
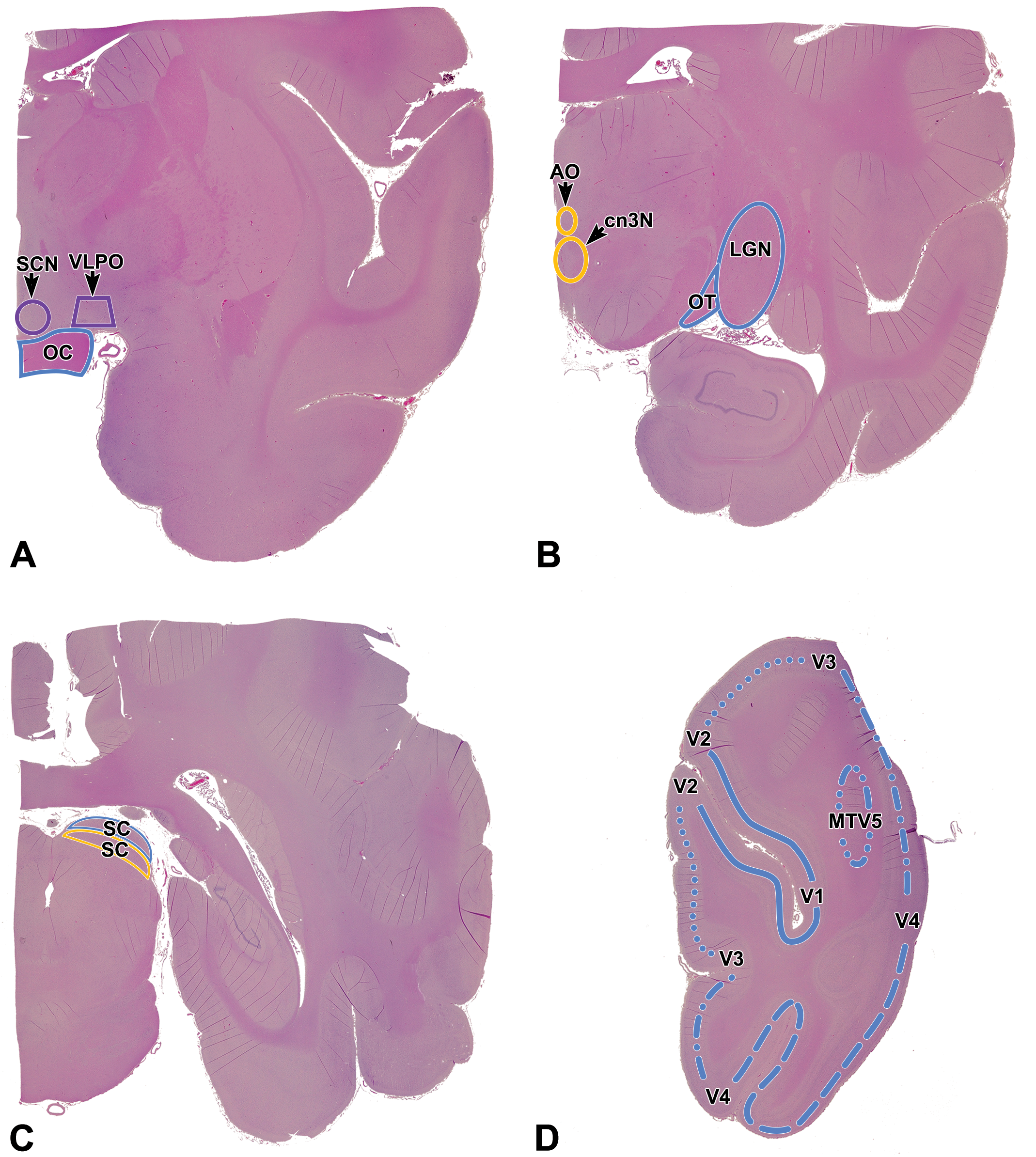
Relevant neuroanatomic areas involved in visual sensory (blue), visual motor (yellow), and visual circadian/sleep (purple) pathways in the cynomolgus macaque as seen in standard sections acquired by current “best practice” recommendations for sampling the nonrodent brain during general toxicity studies. 1 A, Level 2 = section through the anterior commissure showing the optic chiasm (OC) and approximate locations of the suprachiasmatic nucleus (SCN) and ventrolateral preoptic nucleus (VLPO); the latter 2 nuclei will not be present in all sections taken at this level since they are quite small. B, Level 3B = section through the anterior thalamus showing the optic tract (OT) as it courses toward the lateral geniculate nucleus (LGN). The small oculomotor nucleus (cn3N, which harbors somatic motor neurons for cranial nerve 3 [cn3]) and accessory oculomotor (or Edinger-Westphal) nucleus (AO, which has autonomic [parasympathetic] neurons that innervate the iris sphincter and the ciliary muscles inside the eye) are located at the midline ventral to the third ventricle; these 2 very small nuclei also may be absent depending on the trimming plane. C, Level 4 = section through the midbrain showing the locations of the sensory (dorsal) and motor (ventral) portions of the superior colliculus (SC). D, Level 5A = section of the occipital cortex showing the approximate locations of the primary visual sensory cortex (V1; solid line) and the several accessory visual cortical fields (V2, V3, V4, MTV5).
As stated above, currently accepted best practices for nervous system sampling in rodents and nonrodents during general toxicity studies 1 consistently provide a suitable screen for key extraocular neural organs (brain and optic nerve) involved in visual sensation. The recommended brain sampling strategy also may permit evaluation of certain brain regions that support light-related, nonvisual functions, although availability of such small domains (eg, SCN, VLPO) typically will exhibit modest variability among animals and across studies for a given species as well as across animal species. Rarely, in-life clinical signs and/or prior knowledge of test article-related findings may suggest a need to specifically evaluate small light-related nuclei (eg, SCN or VLPO). In such cases, additional sampling and some degree of serial sectioning may be necessary to reveal the suspected target structure(s). A well-annotated, species-specific brain atlas will be an indispensable resource for such investigations.
Extraocular Visual Sensory Pathways—Normal Neurobiology
Visual sensation starts with light stimuli impinging on paired peripheral receptor organs (the retinas) housed within the eyes. However, the bulk of the visual sensory pathways exists outside the globes. The extraocular pathways that support visual perception include both extracranial and intracranial components.
Extracranial Constituents of the Extraocular Visual Sensory Pathways
The visual cascade begins when light reaching the photoreceptor layer of the retina initiates a complex series of chemical reactions that transduce light photons into electrical signals. The key photoreceptor cell types, cones and rods, form synapses with interneurons in the outer and inner nuclear layers of the retina, which in turn process the signals and transmit them to the retinal ganglion cells (RGCs). 3,26 The RGC axons course centrally to form the bulk of the axons that will comprise the optic nerves.
Optic nerve exits the caudal pole of each globe (Figure 5), 27 appearing as a thick white nerve trunk surrounded (prior to dissection) by bellies of the extraocular skeletal muscles and (in rodents) by lobules of the Harderian (lacrimal) gland. Each nerve has 4 anatomically defined portions. 28 The first segment, which is located within the eye, is the optic nerve head (optic disc). This division serves as the collection point for axons that will penetrate the wall of the globe. The caudal boundary of this segment is the lamina cribrosa (LC, Figure 5), a transverse plane of porous connective tissue (made by fibroblasts and glial cells); in mammalian test species, the optic nerves of most nonrodents (eg, dogs, pigs, primates) have a well-developed LC; rats and rabbits (and chickens) possess a barely perceptible LC; and mice lack an LC. 29,30 Nerve fibers in the optic nerve first become myelinated immediately caudal to the LC in most nonrodent mammals. In contrast, intraretinal myelination of nerve fibers in the optic disc occurs in some species with barely visible LC (eg, chickens, rabbits), although it is not seen in other mammals with a faintly perceptible LC (rats) or that lack an LC (mice). 30 The second optic nerve segment is the intraorbital trunk, which is curved to some degree to permit lengthening of the nerve as the globe shifts orientation within the orbit (thereby avoiding traumatic stretching of the optic nerve fibers). The third optic nerve segment is the intracanalicular trunk, which is the short expanse of optic nerve within the optic canal (ie, optic foramen) at the base of the skull. The fourth optic nerve segment is the intracranial trunk, which terminates after a very short distance as the nerve reaches the optic chiasm. The microscopic appearance and responsiveness to toxicants is similar for the intraorbital, intracanalicular, and intracranial optic nerve segments. Therefore, routine microscopic evaluation of the optic nerve typically is conducted by sampling the first and second segments only. 1
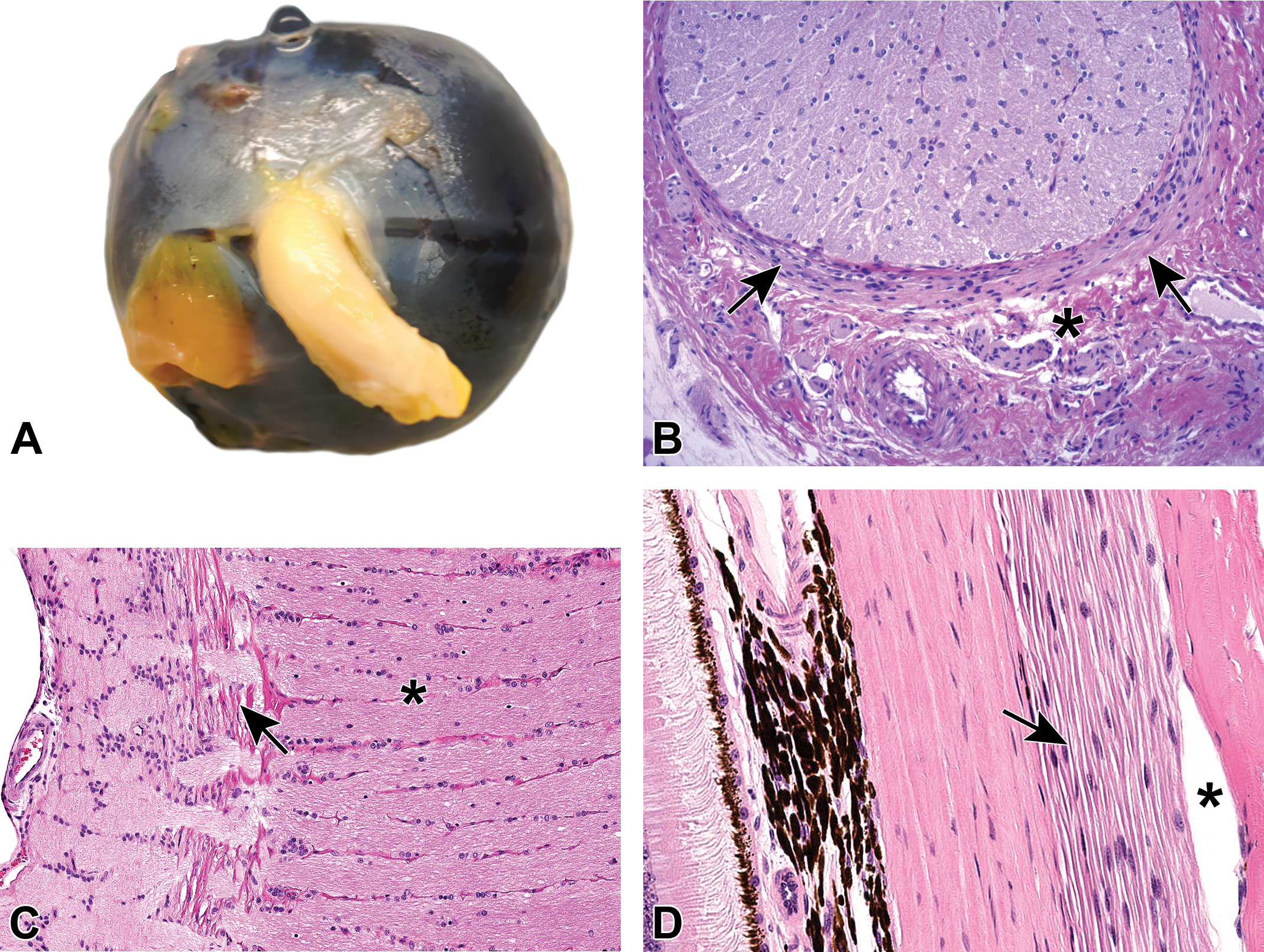
A, Posterior view of the eye from a cynomolgus macaque showing the optic nerve (cranial nerve 2 [cn2]) trunk exiting the globe. B, Transverse section of the large optic nerve trunk from a Wistar rat showing densely myelinated nerve fibers, scattered oligodendrocyte nuclei, and the bordering meninges (arrows). Nearby, a small tangential section of an autonomic nerve (asterisk) likely represents parasympathetic innervation from postganglionic fibers originating from neurons in the ciliary and pterygopalatine ganglia. C, The lamina cribrosa (arrow) in a cynomolgus macaque is a stack of horizontally oriented, fenestrated connective tissue plates at the level of the sclera through which retinal axons leave the caudal pole of the eye to form the optic nerve (asterisk). D, Autonomic (parasympathetic) nerve fibers (arrow) of a cynomolgus macaque arise from the posterior ciliary ganglion that innervates the posterior aspect of the eye. The asterisk denotes an artifact of histologic processing.
The paired optic nerves (cn2) are the retinofugal (eye-to-brain) conduits by which light information received by the retinas is transmitted to the brain. Axons of the RGCs travel centrally to the optic nerve head and then are directed caudally as the optic nerve. Axons in the optic nerves may carry either afferent (arising from the retina and pupil) or efferent (traveling to the retina) signals, and they support either somatic or autonomic activities in the eye. 31 The number of axons in the optic nerve can vary by 2-fold among adults. 32 Optic nerve axons all exhibit a similar appearance in routine optic nerve sections. The unbranched axons from RGCs leave the eye in an approximately retinotopic fashion (ie, with axons from specific retinal fields clustered together in organized arrays), but this quasi-ordered arrangement is lost after the axons enter the optic nerve. 10,33 In rodents, numbers of RGC axons in the optic nerve for animals of the same size vary among strains. 10,34
The optic nerves are considered to be components of the CNS, 28 for several reasons. First, the eye primordia arise as extensions from the rostroventral wall of the telencephalon (primitive forebrain) during development. Second, the retina and optic nerves both contain astrocytes and microglia, 2 of the major glial cell types found in the CNS. 34 Third, myelin sheaths around optic nerve fibers are produced by oligodendrocytes, a CNS glial cell type that functions to ensheath CNS nerve fibers. Finally, the optic nerve trunks are encased in meninges (Figure 5) and bathed in cerebrospinal fluid. 35 The optic nerve meninges in the orbit (extracranial) fuse with the sclera, while the optic nerve meninges inside the optic canal are continuous with the meninges that cover the outer surface of the brain. In summary, the optic nerves are most closely related in origin and structure to white matter tracts in the spinal cord rather than to other peripheral (extracranial) nerves. 32
Intracranial Constituents of the Visual Sensory Pathways
The intracranial visual sensory pathways are longer and more intricate than the extracranial elements (retina and optic nerve). 36 The primary intracranial visual sensory circuitry consists of tracts, formed by RGC-derived axons, that course toward a series of subcortical relay nuclei and ultimately the visual fields within the cerebral cortex. 37 Retinal axons project to these brain domains in retinotopic fashion, exhibiting a point-for-point correspondence from a particular field of RGCs to specific areas of the CNS structures involved in visual sensation. 7,8,10 The regions of the optic brain are reviewed briefly below, including important structural variations among key mammalian species utilized in toxicity testing.
Optic chiasm
The optic nerves from each eye meet at the optic chiasm. The chiasm is located at the base of the diencephalon (ie, ventral forebrain) just rostral to the pituitary gland. Within the optic chiasm, some nerve fibers enter the ipsilateral optic tract while others decussate (cross over) to reach the contralateral optic tract. 27 In general, each optic tract contains nerve fibers from the lateral (temporal) retina of the ipsilateral eye and the medial (nasal) retina of the contralateral eye (Figure 6). 38 Projection of nerve fibers from both eyes to both sides of the brain permits stereopsis (ie, depth perception made possible by binocular vision).
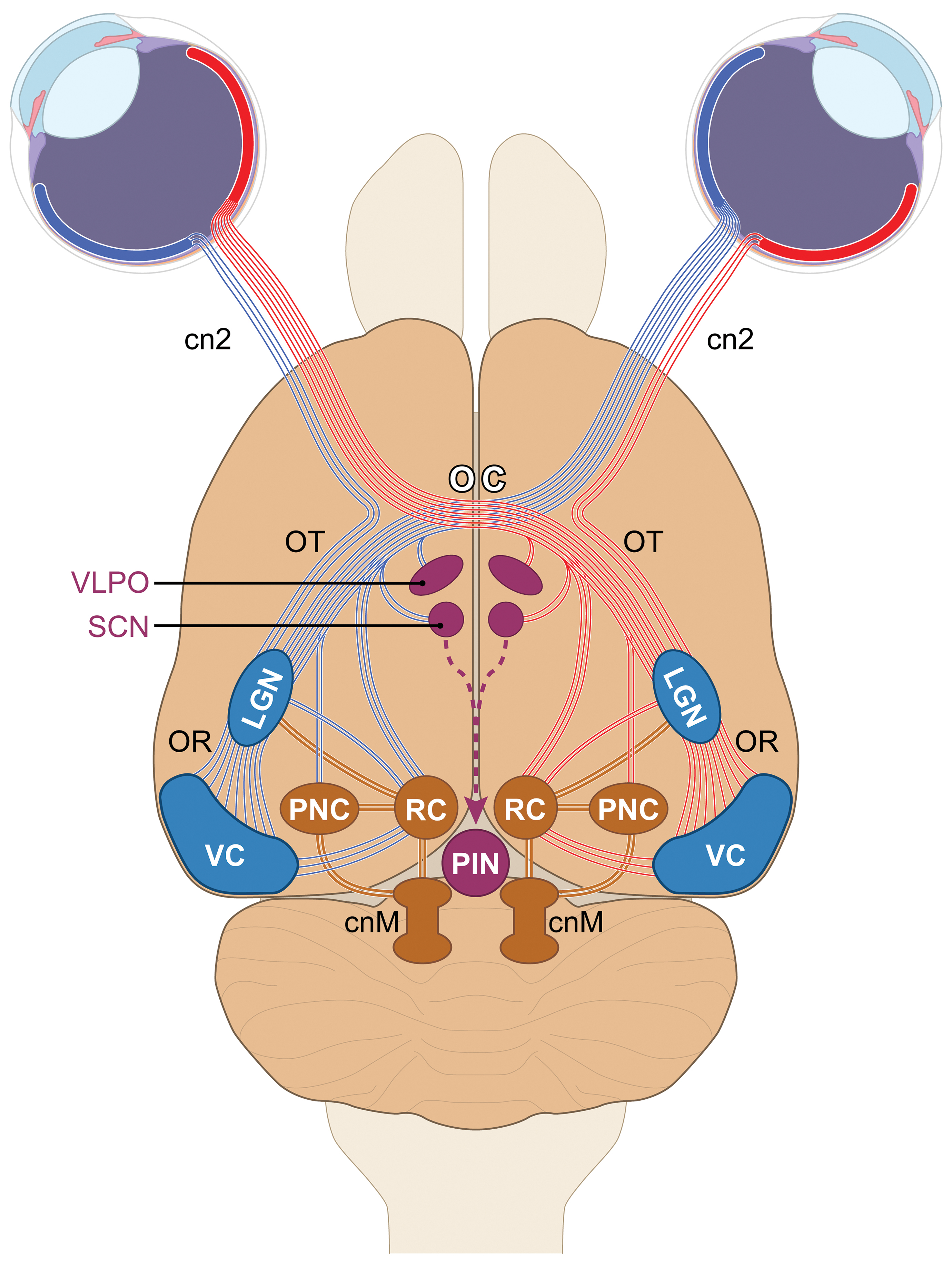
Schematic diagram (dorsoventral view in the rat) of key central visual sensory and light-mediated nonvisual centers. Axons from retinal ganglion cells (RGCs) leave the eyes via the optic nerves (cranial nerve 2 [cn2]) and travel to the optic chiasm (OC). For visual perception, most axons from the lateral (temporal) retina continue in the ipsilateral optic tract (OT), while those from the medial (nasal) retina decussate (cross) to reach the contralateral OT. Most axons in the OT enter the lateral geniculate nucleus (LGN). Axons from second-order relay neurons in the LGN project via the optic radiations (OR) to reach the visual cortex (VC). For light-related nonvisual functions, axons from a subset of RGCs extend directly to multiple diencephalic nuclei, the chief of which are the suprachiasmatic nucleus (SCN, which regulates circadian rhythms via axons sent to the pineal gland [PIN]); ventrolateral preoptic nucleus (VLPO, a sleep control center); or the rostral (superior) colliculus (RC, which controls the direction of visual attention). Integration of light-related nonvisual and visual sensory centers is provided by LGN axons extending to the pretectal nuclear complex (PNC), which controls intraocular and extraocular muscles involved in pupillary constriction and lens accommodation. Axons from the PNC, and lesser numbers from the RC, project to the cranial nerve motor nuclei (cnM) whose neurons travel in the oculomotor, trochlear, and abducens cranial nerves that innervate the extraocular muscles. [Diagram crafted by Mr Timothy Vojt.]
The degree of optic nerve fiber decussation varies among species. 39 As a general rule, more lateral placement of the eyes on the head leads to a greater degree of decussation. 40 In species with no binocular overlap in the visual fields (eg, fish, most birds), 100% of RGC axons cross into the contralateral optic tract. 41,42 In rodents and rabbits, where only a modest degree of binocular overlap occurs, the majority of RGC axons (90% or more, depending on the species and stock/strain) decussate to enter the contralateral optic tract. 10,42 -44 In nonrodents, where binocular overlap is substantial due to the more frontal placement of the eyes, large fractions of RGC fibers instead project to the ipsilateral optic tract—from 25% to 35% in carnivores and 45% to 50% in primates. 41,44,45
Optic tract
The optic tract carries RGC axons from the optic chiasm to the subcortical relay centers of the visual sensory pathway (chiefly the LGN, pretectal nuclear complex (PNC), and RC [SC in primates]). In essence, the optic tracts constitute a continuation of the optic nerves into the brain (Figures 6 and 7). Each optic tract actually consists of 2 separate but parallel axon bundles: a left subcomponent harboring RGC axons from the lateral (temporal) retinal fields of the left eye and medial (nasal) retinal fields of right eye, and a right subcomponent that holds RGC axons from the lateral retinal fields of the right eye and the medial retinal fields of the left eye (Figure 6). The numbers of RGC axons in each optic tract exceed the axon numbers in the optic nerve since some RGC axons bifurcate in the optic chiasm. 10
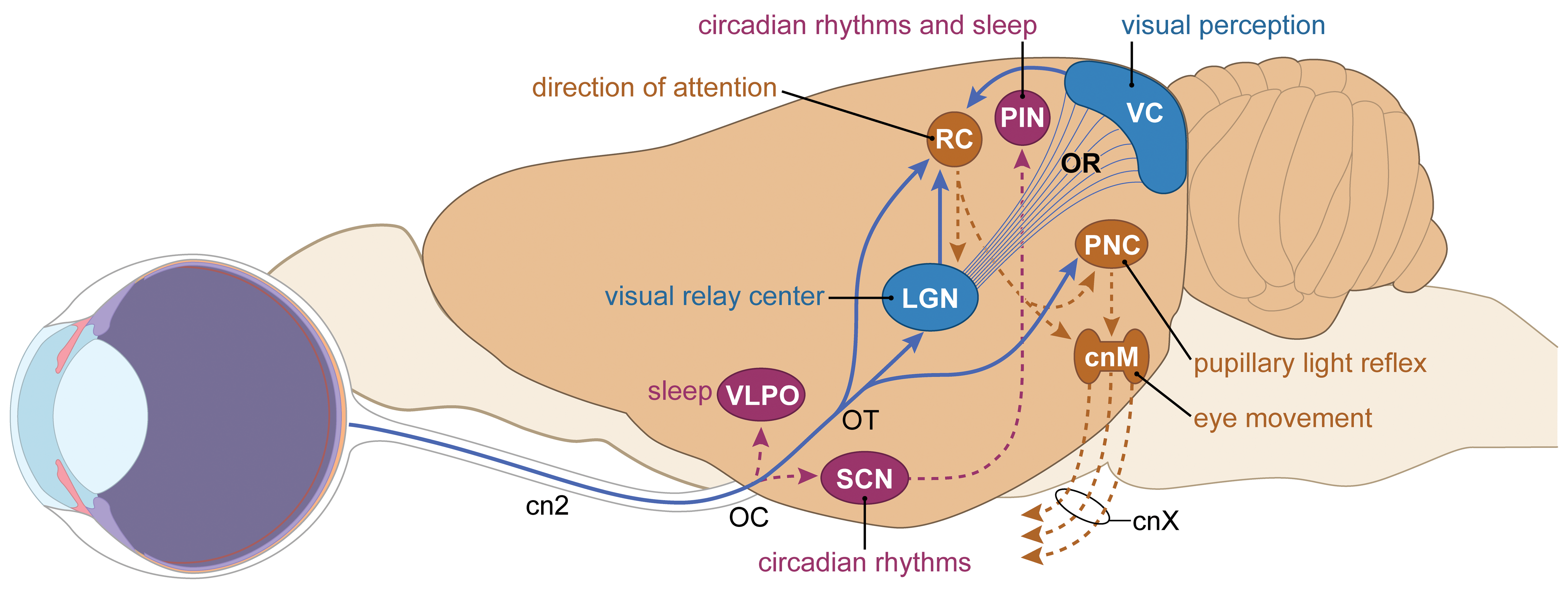
Schematic diagram (lateral view in the rat) of key central visual sensory and light-mediated nonvisual centers. Axons from retinal ganglion cells (RGCs) leave the eyes via the optic nerves (cranial nerve 2 [cn2]) and travel to the optic chiasm (OC). For visual perception (blue objects and lines), most axons enter the optic tract (OT) to reach the lateral geniculate nucleus (LGN). Axons from second-order relay neurons in the LGN project primarily to the primary visual cortex (VC) via the optic radiations (OR). For light-related regulation of circadian rhythms and sleep (purple objects and lines), RGC axons directly synapse in several diencephalic nuclei, the chief of which are the suprachiasmatic nucleus (SCN), ventrolateral preoptic nucleus (VLPO), and rostral (superior) colliculus (RC). Additional fiber tracts extend from the LGN to the RC; from the RC back to the LGN; from the RC to the pretectal nuclear complex (PNC) and thence on to the cranial nerve motor nuclei (cnM) for the 3 cranial nerves (cnXs collectively, comprised of the oculomotor, trochlear, and abducens nerves) that innervate intraocular and extraocular muscles; and from the SCN to the pineal gland (PIN). [Diagram crafted by Mr Timothy Vojt.]
Lateral geniculate nucleus (LGN)
Axons from the RGCs traveling in the optic tract synapse in several discrete brain nuclei in the diencephalon and dorsal midbrain (Figures 6 and 7). In terms of visual perception, the major recipient region in the diencephalon is the LGN, a visual relay center that is located in the caudal thalamus. Visual perception is precise because each RGC axon projects to only a small number of LGN neurons.
Considerable variation exists in LGN structure among species. 46 For example, in rodents, the LGN is organized as a caudodorsal “outer shell” and a ventromedial “inner core.” 10 In contrast, in primates, the LGN is located more ventrally and consists of 3 subcomponents: the dorsal nucleus, the pregeniculate complex, and the intergeniculate leaflet. The dorsal nucleus of the LGN is the largest portion as well as the region that receives retinal input. 28 The RGC axons from the ipsilateral and contralateral eyes terminate in different regions within the LGN, although some overlap in innervation occurs in rodents. 44
The LGN neurons are arranged in 6 layers, 44,47 which can be distinguished readily in carnivores and primates (Figure 8) but are relatively indistinct in rodents. The lack of visible neuronal layers in the rodent LGN reflects the near absence of RGC axons from the ipsilateral retina. 34 In primates, the 2 ventral (inferior) layers harbor large magnocellular (M) neurons, while the 4 dorsal (superior) layers contain parvocellular (P) neurons; very small koniocellular (K) neurons are located in the 6 parenchymal fields ventral to each of the M-cell and P-cell layers. M-cells are color insensitive but well-suited to detecting contrast and movement, P-cells are color sensitive, and K-cells are even more color sensitive and are devoted to color discrimination in shape recognition. 47,48 Incoming optic nerve axons from the medial (nasal) retina of the contralateral eye run to LGN layers 1, 4, and 6, while axons from the lateral (temporal) retina of the ipsilateral eye travel to LGN layers 2, 3, and 5. 49 In rodents, the LGN is organized as a set of anatomically indistinct overlapping territories called “eye-specific domains,” which fulfill the same purpose as the discrete neuronal layers characteristic of the primates LGN. 50 Axons arising from LGN neurons pass through the optic radiation, a white matter tract comprising part of the internal capsule, before terminating in the primary visual (or striate) cortex. This bridge between the LGN and visual cortex comprises the geniculocortical (or geniculostriate) pathway. The dorsal LGN receives reciprocal feedback via corticogeniculate axons arising predominantly in the ipsilateral visual cortex.
Normal morphology of the primate lateral geniculate nucleus (LGN) showing 6 distinct neuronal layers. Laminae 1 and 2 hold large magnocellular (M) neurons, laminae 3 to 6 contain smaller parvocellular (P) neurons, and the intervening pale zones contain very small koniocellular neurons. Incoming retinal axons enter particular layers in retinotopic fashion (ie, with point-to-point concordance). Alignment of the laminar labels shows the location of congruent receptive fields for both eyes, while the colors of the labels denote that incoming fibers arise from the ipsilateral (blue) or contralateral (red) eye. Cynomolgus macaque. Hematoxylin and eosin.
Some general principles of LGN functional organization can be identified. The dorsal LGN engages in processing retinal input for visual perception and then forwarding the integrated input to the primary visual cortex, while the ventral LGN projects to various elements of the PNC to control pupillary contraction and modulate eye movements. 7,44,51 The LGN neurons act either as second-order relay cells that send geniculocortical axons to specific regions of the visual cortex (the case for approximately 75%-80% of LGN neurons) or as interneurons that engage in crosstalk with other LGN regions (about 20%-25% of cells). 10,52 The number of RGC axons that synapse on LGN neurons varies among species and is higher in carnivores and primates (about 90% in humans) 47 than in rodents (about 35%). 10,34 Connections with RGC axons are the primary drivers of LGN neuron activity, although retinal input to LGN neurons represents only 5% to 10% of synapses in carnivores 53 and 30% to 40% of synapses in primates. 52 The sources of the nonretinal axons to the LGN have yet to be defined in full, but current thinking is that these nerve fibers provide feedback from other subcortical nuclei (including but not limited to other visual centers) and the visual cortex itself. 44 Neurons in the LGN may fire in the absence of visual input from the retina, and such spontaneous tonic oscillations are thought to be vital in modulating the visual sensory information received from the retina. 52
Optic radiation
The LGN on each side of the brain is connected to the ipsilateral primary visual cortex by the optic radiation. These radiations appear bilaterally as sheet-like fiber tracts which follow the contours of the lateral ventricles to reach the occipital pole of the cerebral cortex. Each radiation contains projecting axons from LGN neurons that are traveling toward the primary visual cortex as well as reciprocal axons from neurons in the visual cortex providing feedback to LGN cells. The retinotopic organization of the LGN is maintained as the optic radiation reaches the visual cortex. 47
Visual cortex
Visual cortex is parsed into multiple domains in all species, all of which are localized to the dorsocaudal region of the occipital pole of the cerebral cortex (Figure 4). 8,44 The primary visual cortex (designated as zone V1) is the first cortical area to which partially integrated visual sensory information is sent after the initial separate processing undertaken in the retina and LGN. 7 Thus, V1 is essential for initial interpretation of visual input and also for passing information to secondary cortical visual centers for further interpretation. 7 Recent work suggests that in addition to V1, a second pathway for processing primary visual information may exist in mammals in the postrhinal cerebral cortex, which is located rostral and ventral to the recognized visual sensory fields in the occipital cortex. 54 Secondary (or associative) visual cortex consists of several regions (designated as zones V2, V3, V4, and V5) that receive visual sensory information from zone V1 and then subject it to further processing and integration to allow the brain to determine and implement appropriate motor responses to visual sensory information. 35,55 Integration of information by the various secondary visual cortex zones is conducted through massively parallel processing. 7,56 The primary visual and secondary visual cortices are connected to the corresponding brain areas in the contralateral hemisphere by white matter tracts in the caudal portion of the corpus callosum. 35
Primary visual cortex (V1) or “striate cortex” (so called due to the prominence of cortical neuron layer 4 in this area relative to other portions of the occipital cerebral cortex 44 ) is localized primarily on the lateral side in rodents and carnivores but is concentrated on the medial side with some spillover to the lateral side in primates. 7,34 In primates, V1 is divided by the calcarine fissure into upper (cuneus gyrus) and lower (lingual gyrus) parts. No such external landmark is visible in rodent brains. The cuneus and lingual gyri receive retinotopic projections from the superior and inferior retina, respectively. Similarly, the cuneus and lingual gyri receive projections from the macula (ie, the central portion of the retina that is involved in sharp visual definition). The representation of the macula is more extensive in the caudal (posterior) aspect of the visual cortex, while peripheral retina dominates in the rostral (anterior) visual cortex. 57
The V1 zone represents the cytoarchitecturally distinct brain region known in humans as Brodmann’s area 17. 42,58 Neurons in this domain form 6 main layers that are subdivided variously into 9 to 11 sublayers (labeled 1, 2, 3, 4A, 4B, 4C alpha, 4C beta, 5A, 5B, 6A, and 6B) 7,59 ; neuronal layer 4 in V1 is especially prominent, particularly in primates, and gives this area its striated appearance in tissue sections. 44 Activation of neurons in V1 depends completely on input from LGN neurons, the majority of which synapse in layer 4. 7,57 Even so, midbrain projections to V1 form only a small portion (approximately 5%) of synaptic input to V1. 47 About 80% of V1 cells are pyramidal (excitatory) neurons concentrated in sublayers 2/3 and 5/6. 7 In macaques, LGN axons from M-cells and P-cells synapse in sublayer 4C of the visual cortex, while K-cell axons project to cortical layers 1, 3, and 4A; interestingly, LGN axons of humans and apes do not synapse in layer 4A but do synapse in other portions of layer 4. As in other parts of the cerebral cortex, V1 neurons are aggregated into narrow “cortical columns” of cells that share a common function—in this location, a visual receptive field. 7,55 Each eye is provided with its own set of cortical columns. 44 Input to V1 from the dorsal LGN is organized in retinotopic fashion. The retinal projection onto V1 forms a single unified pattern, but the representation is not equal throughout the region. 44 Instead, the cone-rich central retina (ie, the fovea, which is the area of highest visual acuity in diurnal species) comprises less than 1% of the retinal surface but innervates approximately 50% of V1 in primates. 44,60
The secondary (or associative) visual cortex (zones V2, V3, V4, and V5) collectively is designated the “extrastriate cortex,” since this region lacks the prominent cortical neuron layer 4 that is characteristic of the striate cortex (V1). 44 These zones form an arc surrounding V1 on all sides and thus are found on both the lateral and medial sides of the occipital region of the cerebral cortex. The secondary visual cortex encompasses Brodmann areas 18 (zone V2) and 19 (chiefly V3 but also V4 and V5) in humans. 42,58 Visual information received in V2, V3, V4, and V5 arrives almost entirely from V1, with little or none arising from the LGN. 44 As with V1, input to V2, V3, V4, and V5 exhibits a retinotopic organization, although the arrangement is much more complex relative to V1. Instead, multiple retinotopic patterns exist concurrently in the secondary visual cortex, ranging from 13 to 14 in rodents and carnivores to several dozen (24 or more) in various primate species. 44
The visual cortex projects widely to other visual centers in the brain (Figure 3). The sum of V1 activity is conveyed mostly to the secondary visual association areas V2, V3, V4, and V5 for further processing. 35,55 The V1 region also projects to the LGN, RC (SC in primates), and other diencephalic and midbrain nuclei that are involved in visual attention and eye movements. 44 The V2 zone sends axons to V1 (for feedback) as well as to V3, V4, and V5 (for further processing); the other areas of the secondary visual cortex supply axons to other cortical regions and subcortical nuclei as a means of integrating visual cues with other behavioral processes. The cascading links from V1 to various secondary visual cortical domains form 2 distinct circuits (or “streams”), with separate functions. 44 The dorsal or occipitoparietal stream (V1 to V2 to V3 to middle temporal cortex to parietal cortex) is involved in defining where objects of visual interest are located, while the ventral or occipitotemporal stream (V1 to V2 to V4 to ventral [inferior] temporal cortex) participates in identifying what objects are. 7,44 Both streams ultimately connect to the prefrontal cortex.
The complexity of the visual cortex circuitry varies considerably among species in relation to the importance of vision as a sensory modality. In general, vision is more critical for microsmatic species (eg, primates), where the sense of smell is relatively underdeveloped, and less important for macrosmatic species (eg, rodents > dogs), where the sense of smell is vital to survival (in the wild). 61 The sizes of both primary and secondary visual cortices are substantially greater in diurnal species (like primates) in which vision is a major sensory modality. 44 Brain cortical areas devoted entirely or mainly to vision comprise 23% of the adult modern human neocortex 62 and 55% of the adult macaque neocortex. 63 In contrast, in nocturnal animals such as rats, only 7% to 8% of the adult dorsal cortex is devoted to the visual fields, an amount that is dwarfed by the percentage of the cortex devoted to olfactory, auditory, and other sensory modalities of greater importance to night dwellers. 64 Diurnal rats have relatively more visual cortex dedicated to areas 17 (primary visual cortex) and 18 (secondary visual cortex) compared to nocturnal rats. Locations and cytoarchitectural complexity of V1 and V2, V3, V4, and V5 vary across species 65 and among animals with differing genetic backgrounds of the same species. 66
Light-Related, Nonvisual (Nonimage-Forming) Brain Centers
Light-related information carried in RGC axons is also distributed to several diencephalon and midbrain nuclei involved in nonvisual functions (Figures 3 and 4). 67,68 The subcortical centers that comprise these light-modulated, nonvisual pathways are located in the diencephalon, midbrain, and pons. These nonvisual circuits serve one of 2 major roles. The first is to control muscle groups inside the eye or in the orbit to regulate eye orientation and the amount of light projected on the retina, thus permitting visual cues in the environment to be assessed more effectively. This motor function incorporates a mixture of somatic (voluntary) and autonomic (involuntary) responses. For this motor role, retinal signals are routed to subcortical relay nuclei like the LGN or RC (SC in primates) before being redirected to subcortical motor nuclei that are responsible for altering head and eye movements. The second light-related, nonvisual function is maintenance of circadian rhythms, which fluctuate in response to shifting ambient light levels and thereby modify the state of consciousness. For this role, retinal signals are sent to subcortical nuclei that act to either promote sleep or inhibit arousal. All these nonvisual functions are controlled centrally, but the motor functions are implemented by neural connections that project from the brain to the eye or periocular structures.
Circadian Rhythms and Sleep Control
Control of these functions is accomplished by RGC axons projecting to key diencephalic nuclei located in the hypothalamus (ventral diencephalon) via the retinohypothalamic tract (RHT). This tract is a monosynaptic circuit that synchronizes diurnal light–dark fluctuations with the sleep–wake cycle. The RHT originates from a distinct RGC subset, known as intrinsically photosensitive RGCs (ipRGCs), which contain the vitamin A-dependent photoreceptor molecule melanopsin. 69 These ipRGCs are directly sensitive to light and thus constitute a third class of mammalian retinal photoreceptors, distinct from rods and cones. 70 Axons from ipRGC synapse on many small, bilaterally symmetrical nuclei in this region, the chief of which are the SCN and VLPO (Figures 3, 4, 5, and 6), although a number of other minor nuclei in this region also participate in such functions. 71 The SCN is involved in regulating circadian rhythms, 70 while the VLPO is a sleep control center. 72 In mammals, the SCN directly regulates PIN production of melatonin, a sleep-inducing hormone. In the absence of light, SCN signaling is decreased, which permits the PIN to escape SCN inhibition and increase melatonin secretion to initiate the onset of sleep. 73 In contrast, the VLPO promotes sleep by inhibiting the brainstem-based arousal systems that maintain consciousness. 74
Saccadic Eye Movements and Pupillary Muscle Control
Retinal axons also interact directly with a number of mesencephalic (midbrain) nuclei via the retinotectal tract. The chief of these in terms of functional relevance in both rodents and primates, including humans, are the rostral colliculus (RC in quadrupeds, known as the superior colliculus [SC] in biped primates), pretecal nuclear complex (PNC, sometimes referred to as the pretectum), and multiple cranial nerve motor nuclei in the midbrain and brainstem (Figures 3 and 4). 71 The RC and PNC serve as relay nuclei to control eye and head movements needed for vision, while the cranial nerves implement such movements (cn3, cn4, cn6) or provide sensory information from the ocular region (ophthalmic part of the trigeminal nerve [cn5]).
The RC (the amniote equivalent to the optic tectum in phylogenetically lower vertebrates) is a dorsal midbrain nucleus that modifies eye movements related to focusing on environmental objects within the range of view. 35,44 This multilayered nucleus has superficial, intermediate (central), and periventricular zones comprised of 7 layers (when viewed using special stains). The superficial zone is comprised of 3 neuronal laminae that are innervated in retinotopic fashion, being targeted by approximately 10% of RGCs from rod-rich regions of the peripheral retina (with axons from the ipsilateral retina arriving rostrally while those from the contralateral eye are distributed caudally). 7,34,44 The incoming axons are small in caliber, originate from RGCs with small dendritic fields, and do not project to other retinal targets in the brain. 75 The superficial RC also receives feedback from the primary visual cortex (V1) and, interestingly, the deep RC receives input from auditory (ear), somatosensory (whisker barrels), and vestibular (labyrinth) receptor organs. 34 Collectively, this feedback from other brain centers (including nonvisual sensory modalities) integrates head and saccadic (ie, rapid) eye movements to orient the visual axis toward objects in the visual field. 7,44,76 In rodents, only about a third of RGC axons project to the LGN, while nearly all project to the RC. 10,34 The axons of deep RC neurons project to the midbrain and brainstem nuclei that are concerned with generating eye movements. 7 The efferent projections from the superficial RC zone travel to other thalamic nuclei (especially to visual sensory centers like the dorsal LGN and pulvinar nucleus) and midbrain nuclei. The intermediate and periventricular RC zones receive incoming axons mainly from the visual and somatosensory cortices as well as other midbrain nuclei involved in eye movements. 44 Efferent signals from the intermediate and periventricular RC zones are segregated functionally, with uncrossed projections controlling avoidance and defense responses while crossed projections regulate visual orientation. 10 The various RC zones connect reciprocally to the comparable regions of the contralateral RC. 44
The PNC is a series of clustered nuclei that comprise a nonimage-forming, subcortical relay center to control pupillary constriction. 7 The pretectum is located at the most rostral pole of the midbrain bordering the thalamus. Among the 5 distinct nuclei that comprise the PNC, 77,78 the most prominent PNC nucleus is designated the olivary pretectal nucleus, which participates in the pupillary light reflex and also helps to coordinate eye and head movements. 10 The PNC receives axons that project directly from a subset of ipRGCs in a retinotopic fashion (Figures 6 and 7), although these cells receive some communication from cones and rods. 67 Importantly, axons entering the PNC from the retina do not pass through the LGN and thus are not linked directly to the CNS visual sensory system. Neuronal subsets of the PNC project to the oculomotor nucleus (the origin of cn3), 79 accessory oculomotor nucleus, 80 and trochlear nucleus (the origin of cn4), all of which reside in the periaqueductal gray matter of the midbrain. These PNC targets are the entities which act as the proximate regulatory centers for PNC-mediated motor events associated with ocular and periocular structures.
The nearby accessory oculomotor (Edinger-Westphal) nucleus is an unpaired autonomic parasympathetic nucleus located rostral and dorsal to the oculomotor nucleus. This structure harbors preganglionic neurons and send axons via cn3 to the ipsilateral ciliary ganglion, a small autonomic parasympathetic ganglion located deep in the orbit caudal (posterior) to the eye. Postganglionic axons from the ciliary ganglion innervate 2 major intraocular muscles; both of these autonomic functions are not under voluntary neural control. 32,81,82 The first muscle is the pupillary sphincter, an iris muscle that modulates pupil diameter when the pupillary light reflex is activated by enhanced or diminished ambient light levels in the external environment. The second muscle is the ciliary muscle, a ciliary body muscle that alters lens curvature to provide accommodation needed to see objects at variable distances from the eye. In contrast to the ciliary and pupillary sphincter muscles, the iris dilator muscle is controlled by autonomic postganglionic sympathetic nerve fibers originating from the cranial (superior) cervical ganglia. The preganglionic input to the cranial cervical ganglia stems from the lateral (or intermediolateral) cell column in the spinal cord segments C8-T1.
Extraocular Muscle Control
The paired oculomotor nuclei (OMN) regulate somatic (voluntary) movements of 4 extraocular muscles. This nuclear complex is located ventral (inferior) to the mesencephalic aqueduct (of Sylvius) at the same level as the RC. The OMN consists of many paired and bilaterally symmetrical as well as a few unpaired and midline subnuclei; each OMN component innervates a single extraocular muscle. Axons leaving various paired OMN subnuclei travel in cn3 to innervate the ipsilateral medial rectus, ventral (inferior) rectus, and ventral (inferior) oblique muscles or the contralateral dorsal (superior) rectus. 83 An unpaired, caudally positioned OMN subnucleus supplies axons to the dorsal (superior) levator palpebrae muscle that elevates the upper eyelid. All of these roles are somatic functions subject to some degree of voluntary control. The paired cn3 exit the ventral surface of the rostral midbrain near the cerebral peduncles and may be seen on the base of the skull on either side of the sella turcica rims, just caudal to the optic chiasm (Figure 9).
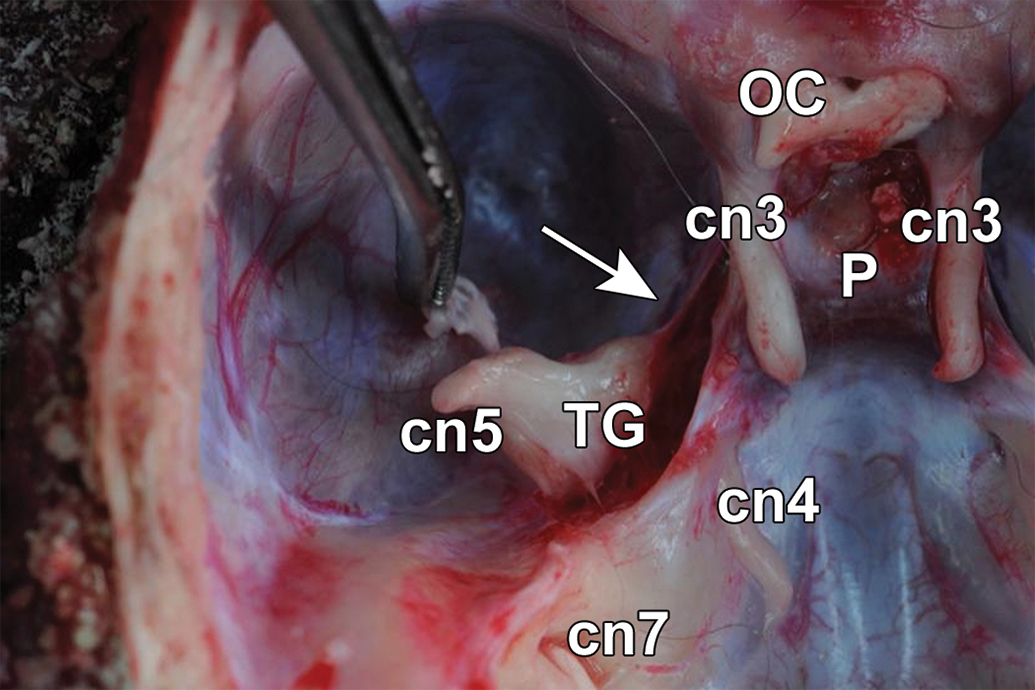
Location of major cranial nerves with visual system-related functions on the floor of the calvarium of a cynomolgus macaque. The optic chiasm (OC), formed by the intersection of the paired optic nerves (ie, cranial nerve [cn] 2, the trunks of which are hidden by the zygomatic bones in this image), is immediately rostral to the pituitary gland (P), which rests within the sella turcica. Paired oculomotor nerves (cn3) follow the lateral margins of the sella turcica. A trigeminal nerve (cn5) trunk, its ophthalmic branch (white arrow), and a trigeminal ganglion (TG) are located lateral to cn3 on each side of the skull. The trochlear nerve (cn4) and facial nerve (cn7) are visible caudal to the TG.
Paired trochlear nuclei (TN) control somatic (voluntary) movement of the contralateral dorsal (superior) oblique muscle. 83 The TN on each side of the brain reside in the midbrain ventral (inferior) to the mesencephalic aqueduct, where it is found just caudal to the oculomotor nucleus. Axons leaving the TN nuclei travel in cn4, which is unique in that the nerve trunk runs dorsally and then crosses the midline before emerging from the dorsal surface of the caudal midbrain to course ventrally along the lateral midbrain surface prior to beginning its rostral course. If sampling this structure is judged to be necessary, the narrow cn4 trunks may be seen caudal to the sella turcica, located between the trunk of cn3 medially and the trigeminal ganglion (cn5) laterally (Figure 9).
Paired abducens nuclei control somatic (voluntary) movement of the ipsilateral lateral rectus muscle. 83 These nuclei are located near the midline in the rostral brainstem, specifically in the pons just beneath the surface of the fourth ventricle. The abducens nerve (cn6) exits the ventral brainstem near the pontomedullary junction.
Sensory Innervation of the Ocular Structures
The trigeminal nerve (cn5) is a mixed sensorimotor nerve with several branches, of which the ophthalmic division (Figure 9) serves the orbit (among other head regions). The ophthalmic branch (cn51) is the smallest cn5 branch and fulfills a purely sensory function. 84 With respect to the eye, cn51 innervates the cornea, iris, ciliary body, conjunctiva, and lacrimal glands via several small branches. The 3 sensory nuclei for cn5 form a continuous column that spans the midbrain, brainstem, and cranial spinal cord. The nucleus that receives axons through cn51 resides in the dorsal pons and processes sensory signals from the ipsilateral eye.
Toxicology of Extraocular Light-Related Organs
The published literature contains relatively few reports of toxicity where the descriptions of oculotoxic and neurotoxic findings were evaluated specifically in the central visual pathways. Several classes of toxicants have been shown to impact the extraocular light-related nervous system (optic nerve and brain), including small molecule drugs and/or drug candidates, some chemicals, and a few metals (Table 1). For the majority of these toxicants, findings in extraocular light-related neural structures have been reported only in 3 visual-related domains: cn2, LGN, and/or visual (occipital) cortex. These 3 key regions often (for LGN) or always (cn2 and visual cortex) will be available for histopathologic evaluation in sections from both rodents (Figures 1 and 3) and nonrodents (Figures 2 and 4) acquired using current best practice recommendations for nervous system sampling during general toxicity studies. 1 The authors believe that the paucity of ocular toxicity literature for other light-related CNS centers (both visual and nonvisual) reflects the limited toxicological research for these regions rather than lesser susceptibility of these structures to toxicants. Further details regarding visual system toxicants, the findings they induce in selected extraocular tissues, and their mechanisms may be obtained in more comprehensive reviews of ocular toxicologic pathology. 3,85
Visual System Toxicity (Direct or Indirect) Affecting Components of the Extraocular Visual System.
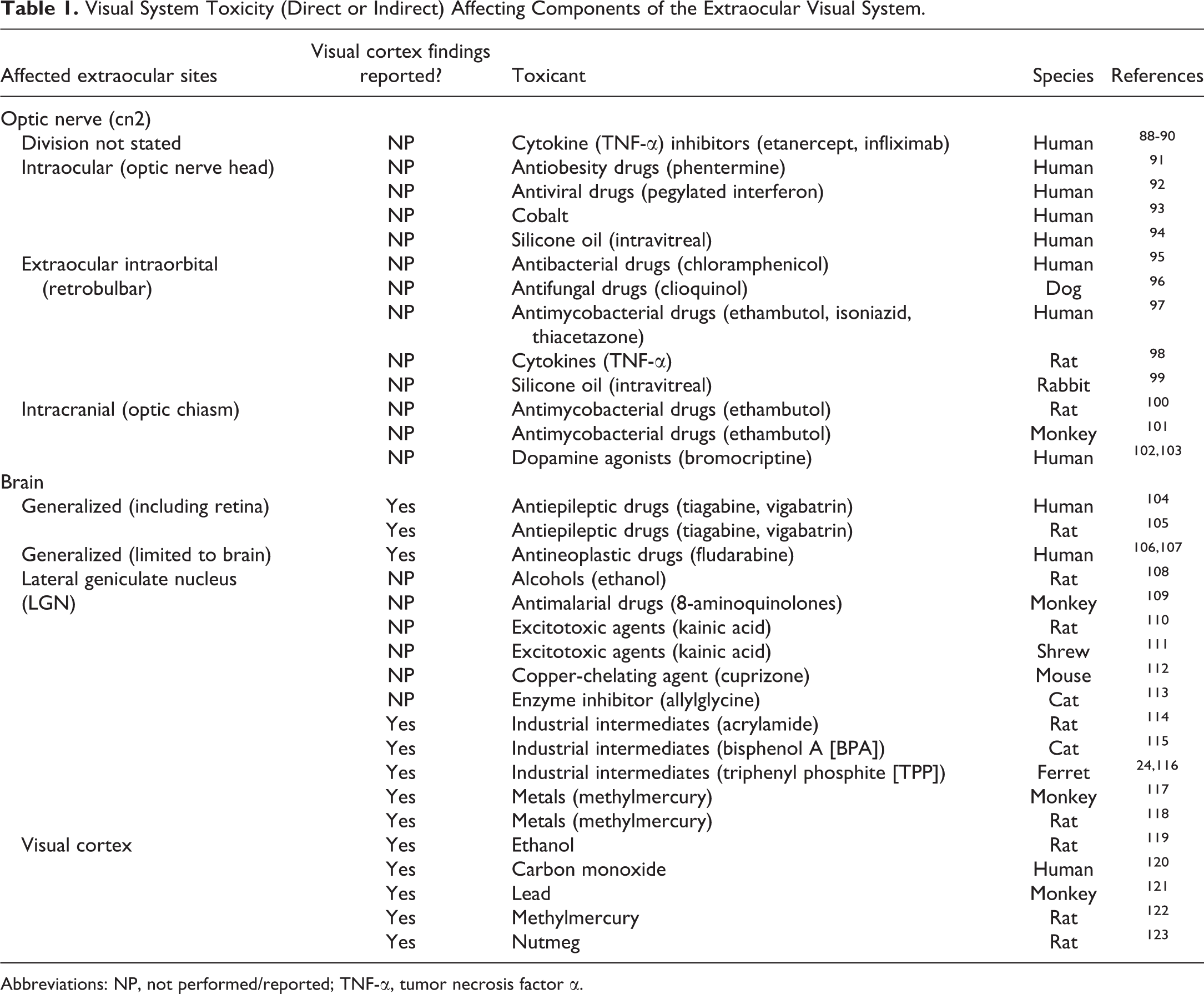
Abbreviations: NP, not performed/reported; TNF-α, tumor necrosis factor α.
In general, toxicant-induced changes in the brain centers involved in the visual pathway will be similar to those observed in nonvisual brain centers. Harmonized diagnostic terminology has been defined for nonproliferative and proliferative findings that might be seen in the optic nerve 86 and brain. 87 This lesion nomenclature is applicable not only to toxicant-induced lesions but also to changes produced by other etiologies, including conditions such as optic nerve atrophy (a secondary consequence of retinal atrophy or enucleation), optic nerve degeneration (an age-related change in some rodent strains), and optic nerve necrosis (which may follow blood sampling from the retro-orbital sinus in rodents).
Conclusions
For the past 25 years, technology has turned the world upside down and inside out. But much is still left to understand about the extent and complexity of biological systems particularly the visual system. The continually expanding toolbox for interrogating the various pathways has persistently led to unraveling the diversity of neuronal circuits from retina to subcortical to cortical brain networks that contribute to image processing and formation. Despite the evolutionary separation that nonhuman primates, rats and mice have from humans, these animal species have been used extensively and successfully as models to study the visual system of humans and also as models of various ocular diseases. However, owing to its extreme complexity, visual system toxicity may manifest anywhere in the visual system, from the eye proper to the visual brain. Therefore, effective screening for visual system toxicity must evaluate not only ocular structures (ie, eye and optic nerve) but also multiple key brain regions involved in vision (eg, optic tract, subcortical relay nuclei, and primary and secondary visual cortices). In this manuscript, we have covered the salient features of the primary brain regions and circuits that participate in light-based visual and nonvisual information processing and have also proposed a pragmatic strategy for assessing the integrity of central visual pathways in rodents and nonrodents appropriate for use in microscopic evaluation during general toxicity studies.
Footnotes
Authors’ Note
All procedures used to prepare macroscopic and microscopic images of animal specimens for this article were performed in accordance with regulations and established guidelines for humane treatment of research animals and were reviewed and approved in advance by an Institutional Animal Care and Use Committee.
Acknowledgments
The authors thank Mr Timothy Vojt for his excellence in crafting the schematic diagrams of key structures in the central visual system as well as Mr Walter Bobrowski (Lakeshore Digital Art LLC) and Ms Beth Mahler for their invaluable assistance in optimizing the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
