Abstract
2-Deoxy-
Introduction
The potential mechanisms associated with anticonvulsant and antiepileptic properties of 2-deoxy-
As an analogue of glucose, uptake of 2-DG into cells occurs through glucose transporters and is dependent on metabolic demand, which is higher in neural circuitry undergoing synchronization during seizures. After uptake via the glucose transporter, 2-DG is phosphorylated at the sixth position to 2-DG-6P. But, unlike glucose-6P, 2-DG-6P cannot be further metabolized by glucose-6P isomerase and is “trapped” in cells due to limited dephosphorylation by phosphatases, inhibiting subsequent steps of glycolysis. 6
Minor et al 7 have performed a long-term dietary toxicity study of synthetic 2-DG at doses ranging from 20 to 300 mg/kg (0.04%-0.6% 2-DG in the diet) in F344 and brown Norway rats, which identified cardiac toxicity associated with the use of 2-DG as a potential calorie restriction mimetic and for other therapeutic applications. The study demonstrated that 2-DG increases mortality of male Fischer 344 rats, increases incidence of pheochromocytoma in the adrenal medulla, reduces weight gain secondary to reduced food intake, and increases vacuolation of cardiac myocytes with an increase in autophagic flux.
The findings of Minor et al 7 prompted a need to better understand 2-DG-induced cardiac toxicity and to develop a cardiac safety biomarker for monitoring 2-DG therapy. Cardiac biomarkers, for example, cardiac troponin (cTnI and cTnT), atrial natriuretic peptide (ANP), brain natriuretic peptide (BNP), N-terminal pro-brain natriuretic peptide (NT-proBNP), and/or creatine kinase (CK), are proteins secreted into blood when myocardial injury occurs and play a crucial role in diagnosis, specific risk analysis, and personalized treatment of patients with various cardiac anomalies. 8 Atrial natriuretic peptide is secreted by stretching of the atria or with increase in blood or extracellular fluid volume. 9 Brain natriuretic peptide is another natriuretic hormone secreted by cardiac ventricles and functions locally to reduce ventricular fibrosis. 10 Brain natriuretic peptide is synthesized and stored as prohormone, and after secretion, it is cleaved into equimolar amounts of active BNP and NT-proBNP. Brain natriuretic peptide and NT-proBNP are validated biomarkers for diagnosis and exclusion of congestive heart failure. 11,12 Diagnosis of acute myocardial infarction can be achieved by the use of troponins 13 and CK. The troponin complex has 3 components, including cTnC, cTnI, and cTnT, which engage with components of tropomyosin and actin filaments to safeguard contraction of cardiac and striated muscles. 14 All isoforms of cTnI are expressed solely in cardiomyocytes. 15 Creatine kinase is an 86-kD dimeric enzyme found in heart muscle, brain, and skeletal muscle, which catalyzes the reaction of creatine and adenosine triphosphate to form phosphocreatine and adenosine diphosphate to generate energy at a cellular level. 16
It has been clinically established that troponin serves as a biomarker for drugs causing cardiac toxicity. 17 Troponin I and T secretion has been associated with neonatal rat cardiomyocyte damage. 18 Empirically, NT-proBNP alone in human patients 19 or troponin T and NT-proBNP both in rat models 20 have been observed to be early markers of anthracycline- and amitriptyline-induced cardiovascular toxicity, respectively.
Previous studies have indicated that other cardiac biomarkers such as BNP and NT-proBNP secreted by heart ventricles due to muscular stretching or cardiac damage may be useful prognostic biomarkers for cardiac abnormalities. 21,22 There are reports wherein NT-proBNP has been found to be of prognostic value as an early marker in patients with breast cancer on chemotherapy experiencing anthracycline-induced cardiotoxicity. 23
The goal of this study was to reproduce 2-DG-induced cardiac toxicity using a clinically relevant route/dosing schedule in a rat model and to identify potential cardiac biomarkers that can be useful to monitor cardiac toxicity during clinical evaluation of 2-DG.
Experimental Design
Twenty rats/sex were assigned to each dose group including control, low, mid, and high. Animals were gavaged with either vehicle or 2-DG at target doses of 50, 125, or 350 mg/kg twice daily (BID), for total daily doses of 100, 250, or 750 mg/kg 2-DG, respectively (Table 1). Animals received a dose volume of 10 mL/kg per administration approximately 6 hours apart. On the day of scheduled necropsy, animals received only the morning dose of vehicle or test article, and core groups were terminated approximately 4 hours postdose.
Experimental Design.
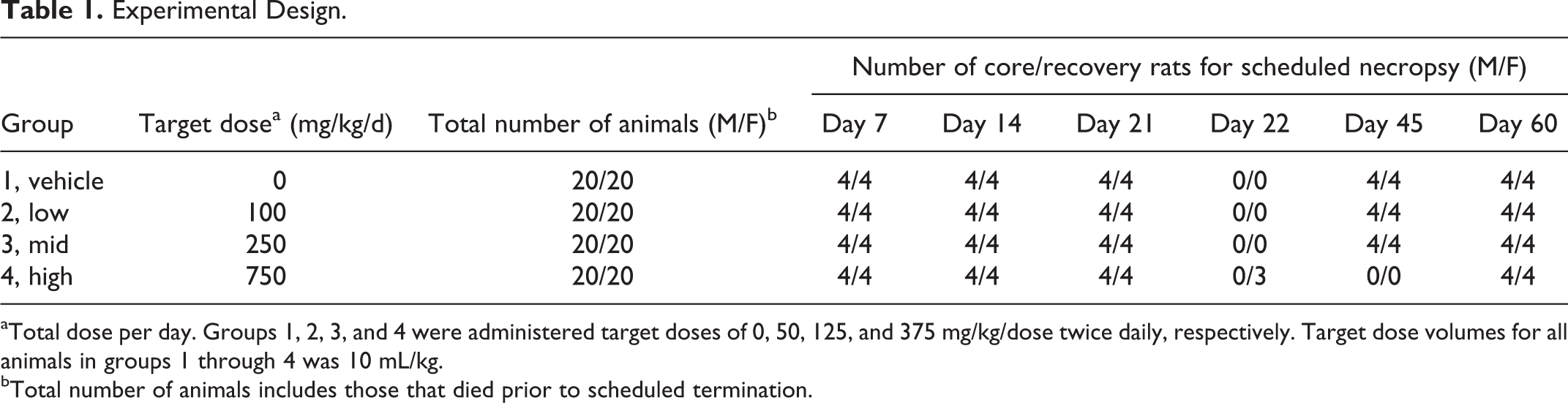
aTotal dose per day. Groups 1, 2, 3, and 4 were administered target doses of 0, 50, 125, and 375 mg/kg/dose twice daily, respectively. Target dose volumes for all animals in groups 1 through 4 was 10 mL/kg.
bTotal number of animals includes those that died prior to scheduled termination.
End points reported in this study included cardiac biomarkers and anatomical pathology of heart tissue. The 4 rats/sex/dose group/day were scheduled for terminal necropsy on study days 7, 14, 21, and 45, with 4/sex recovery animals per dose group scheduled for terminal necropsy on study day 60. Due to unscheduled deaths in the high-dose group (750 mg/kg/d), surviving animals were either maintained without dosing until the end of the recovery period on study day 60 (4/sex) or scheduled for early termination and necropsy on study day 22 (3 females).
Materials and Methods
Formulation Preparation
The test article formulations were prepared weekly at concentrations of 0, 5, 12.5, and 37.5 mg/mL of test article (purity ≥98%; Sigma-Aldrich, Saint Louis, Missouri) in deionized water. Dose formulation concentration analyses were performed for the first preparation using high-performance liquid chromatography/ultraviolet detection. In addition, 9-day formulation stability of 5, 25, and 125 mg/mL 2-DG concentration was determined when stored at room temperature and protected from light. Target doses were 0, 100, 250, and 750 mg/kg/d, and target formulation concentrations were 0, 5, 12.5, and 37.5 mg/mL.
Test System
A total of 80 Fischer 344 rats/sex of approximately 9 weeks of age with 127 to 232 g (males) and 104 to 156 g (females) weights were purchased from Charles River Laboratories, Inc (Stone Ridge, New York). Prior to use on the study, all animals were quarantined for 8 days to evaluate the health status.
Housing and Environmental Conditions
Polycarbonate cages with hardwood bedding were used to house individual animals according to current Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International standards and current requirements stated in the Guide for the Care and Use of Laboratory Animals (National Research Council [NRC]). Twelve hours of light/dark cycle and a minimum of 10 room air changes per hour with the room temperature and relative humidity of 64°F to 79°F and 30% to 70%, respectively, were used. Cages and feeders were changed and sanitized at least weekly.
Food and Water
A pelleted certified feed (Harlan Teklad 2018C; Harlan Teklad, Indianapolis, Indiana) was fed to all rats ad libitum. Water was provided ad libitum from the West Jefferson, Ohio, municipal water supply facility. The water conformed to the current US Environmental Protection Agency (EPA) drinking water standards. No known or reported contaminants in either the water or the feed affected the study results or interpretations.
Group Assignment and Animal Identification
Animals were identified by cage card with a pretest identification number and permanent tail tattoo. Cage cards were color coded by group. Body weights were obtained from individual animals, and group assignments were made using the Xybion Next Generation PATH/TOX System (Xybion, Bensalem, Pennsylvania), which ensured similar group mean body weights by sex.
Clinical Observations
During the study, BID observations for morbidity/moribundity and mortality were performed on all animals, with additional cage-side observations at least once each week.
Cardiac Biomarkers Analysis
Whole blood was collected under CO2/O2 anesthesia into tubes containing EDTA as an anticoagulant on study days 2, 7, 14, 21, 22, 45, and 60. Serum was collected in tubes containing serum separator gel. Specimens were collected and preserved as single aliquots and stored in a freezer set to maintain −70°C. The cardiac safety biomarkers analyzed were cTnI, cTnT, BNP, NT-proBNP, ANP, and CK. The BNP, NT-proBNP, cTnI, and cTnT were analyzed using biomarker kits from Meso Scale Diagnostics (catalog numbers of K153KFD, K153JKD, and K15161C, respectively; Meso Scale Diagnostics, Rockville, Maryland), whereas ANP was analyzed using biomarker kit from Shanghai BlueGene Biotech (Shanghai, China; catalog number of E02A0493). The BNP, NT-proBNP, cTnI, and cTnT assay plates were read by the Meso Scale Diagnostic’s Sector Imager 2400 (model 1250), and the ANP assay plates were read by the µQuant plate reader using KC4 software (BioTek Instruments, Inc., Winooski, Vermont). The data were then transferred to Softmax Pro 5 for the generation of the reference curves and sample analysis. All assay reference curves were qualified to determine whether the assay kits were performing as per the manufacturer’s instructions. Each reference curve was analyzed a minimum of 12 times in duplicate to demonstrate reproducibility.
Necropsy
Core study animals were necropsied at approximately 4 hours postdose on study days 7, 14, 21, 22, and 45, and the remaining 4 recovery animals/sex/group were necropsied on study day 60. All animals were individually weighed and anesthetized with CO2/O2 for blood collection prior to necropsy.
Every necropsy comprised an examination of the external body surface and all orifices including the cranial, thoracic, abdominal, and pelvic cavities and their contents. Heart tissues were collected and preserved in 10% neutral-buffered formalin. Heart tissues from all groups necropsied were processed to slides and stained with hematoxylin and eosin for histopathologic examination.
Statistical Analysis
Statistical differences for cTnI, cTnT, BNP, NT-proBNP, and ANP data were assessed using Student t test assuming equal variance (Microsoft Excel Program). Creatine kinase data were analyzed based on homogeneity/nonhomogeneity by Bartlett test. All data are expressed as mean ± standard deviation, and the statistical significance level was set at P ≤ 0.05.
Results
Dose Formulation Analysis
Formulations of 2-DG prepared in deionized water were within 10% of the targeted concentrations of 0, 5, 12.5, and 37.5 mg/mL, with a 9-day stability confirmed for 5, 25, and 125 mg/mL 2-DG concentration when stored at room temperature and protected from light.
Survival and Clinical Observations
In the high-dose group (750 mg/kg/d), 1 male (study day 19), 2 males (study day 21), and 1 male and 1 female (study day 22) were found dead. Due to severe toxicity, dose administration for the high-dose treatment was stopped on study day 21, and the remaining 3 females were killed on study day 22. Clinical observations of lethargy, rough coat, thin appearance, and reduced/absent feces were observed in animals in the high-dose (750 mg/kg/d) group during study day 18 through day 22.
Cardiac Biomarkers
Plasma or serum specimens were evaluated for CK, ANP, BNP, NT-proBNP, cTnI, and cTnT concentrations as potential cardiac toxicity biomarkers. There were no remarkable changes in ANP, cTnI, or cTnT concentrations. Plasma concentrations of NT-proBNP were significantly increased (P ≤ 0.05) on study days 7, 14, and 45 in all dose groups except for high-dose females (Figure 1). Although a dose-dependent increasing trend was also observed with BNP concentrations, significant difference compared to control was noticed only on day 45 (Figure 2). Elevations in NT-proBNP in individual subjects were, in general, accompanied by vacuolar degenerative (atrium) histological changes. On study day 14, this association was noticed in 3 of 4 males and 1 of 4 females in the high-dose (750 mg/kg/d) group and 1 of 4 females in the mid-dose (250 mg/kg/d) group; on study day 21, 2 of 4 males and 4 of 4 females in the high-dose (750 mg/kg/d) group; and on study day 45, 3 of 4 males and 1 of 4 females in the mid-dose (250 mg/kg/d) group. By study day 60, following 15 days of recovery period, significant increases in NT-proBNP and BNP concentration levels were not observed (Figures 1 and 2).
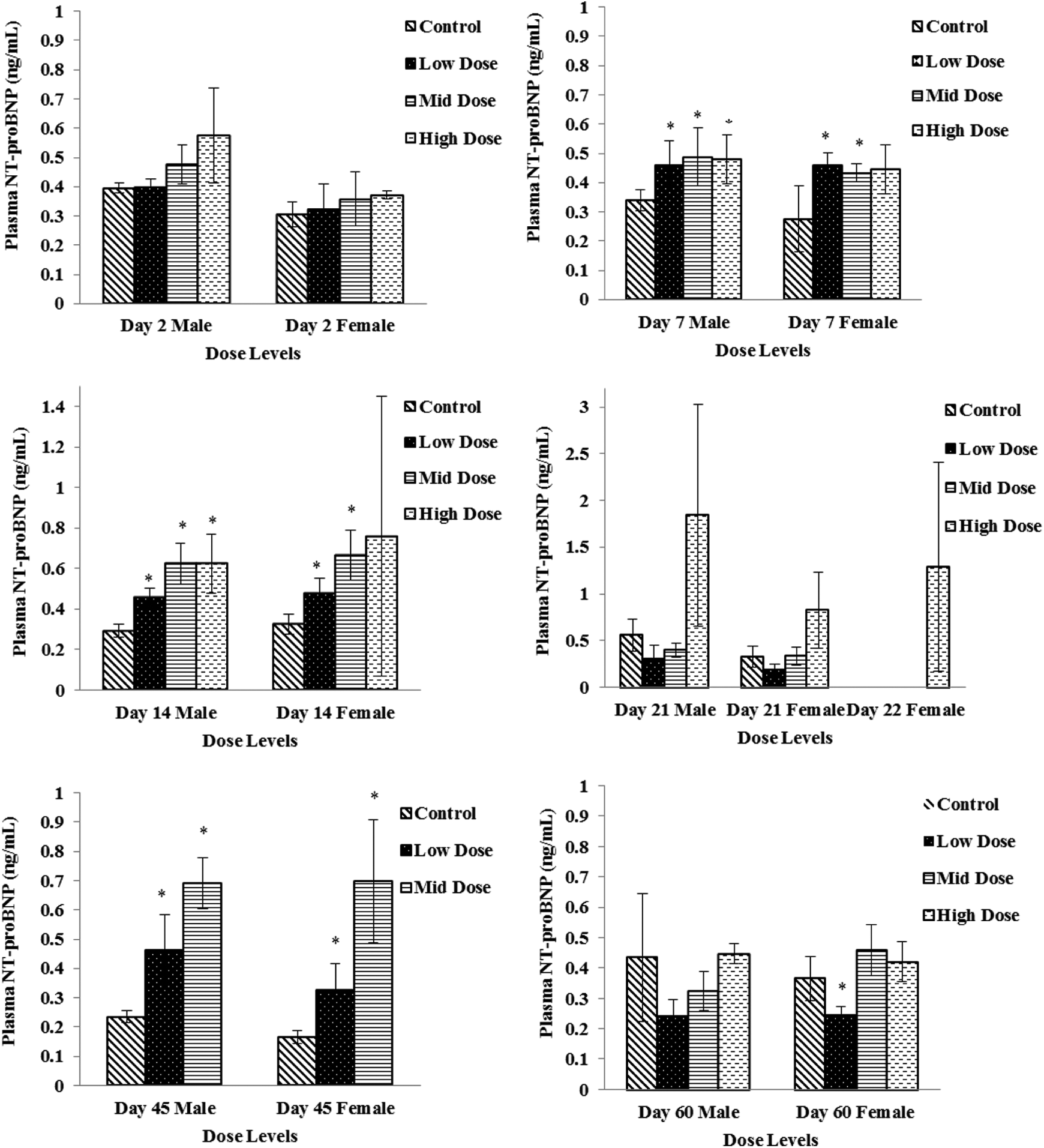
Plasma N-terminal pro-brain natriuretic peptide (NT-proBNP) levels in low-dose (100 mg/kg/d), mid-dose (250 mg/kg/d), and high-dose (750 mg/kg/d) rats. When N > 2, an asterisk (*) indicates a statistical (P ≤ 0.05) difference from the control group.
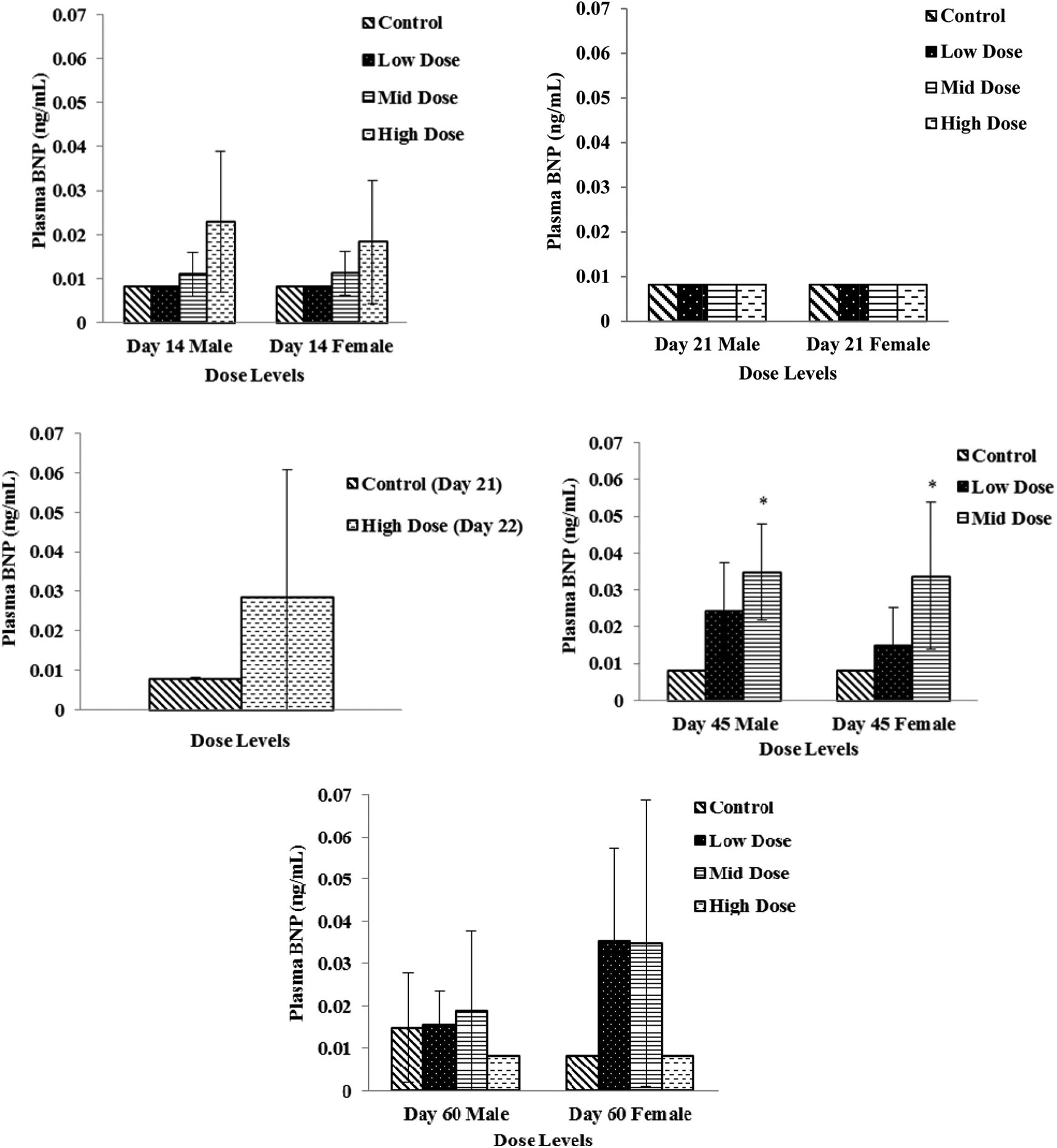
Plasma brain natriuretic peptide (BNP) levels in low-dose (100 mg/kg/d), mid-dose (250 mg/kg/d), and high-dose (750 mg/kg/d) rats. When N > 2, an asterisk (*) indicates a statistical (P ≤ 0.05) difference from the control group.
Creatine Kinase
Creatine kinase activity decreased sporadically in several dose groups (males and females) with return to baseline levels compared to the vehicle group on study days 45 and 60 (Figure 3). In males, the change was −37% in the low-dose (100 mg/kg/d) group on study day 2. In females, changes were noted on study day 2 in the mid- and high-dose groups (−47% and −59% for the 250 and 750 mg/kg/d groups, respectively) and on study day 14 in the low-dose and high-dose groups (−47% and −51%, for the 100 and 750 mg/kg/d groups, respectively).
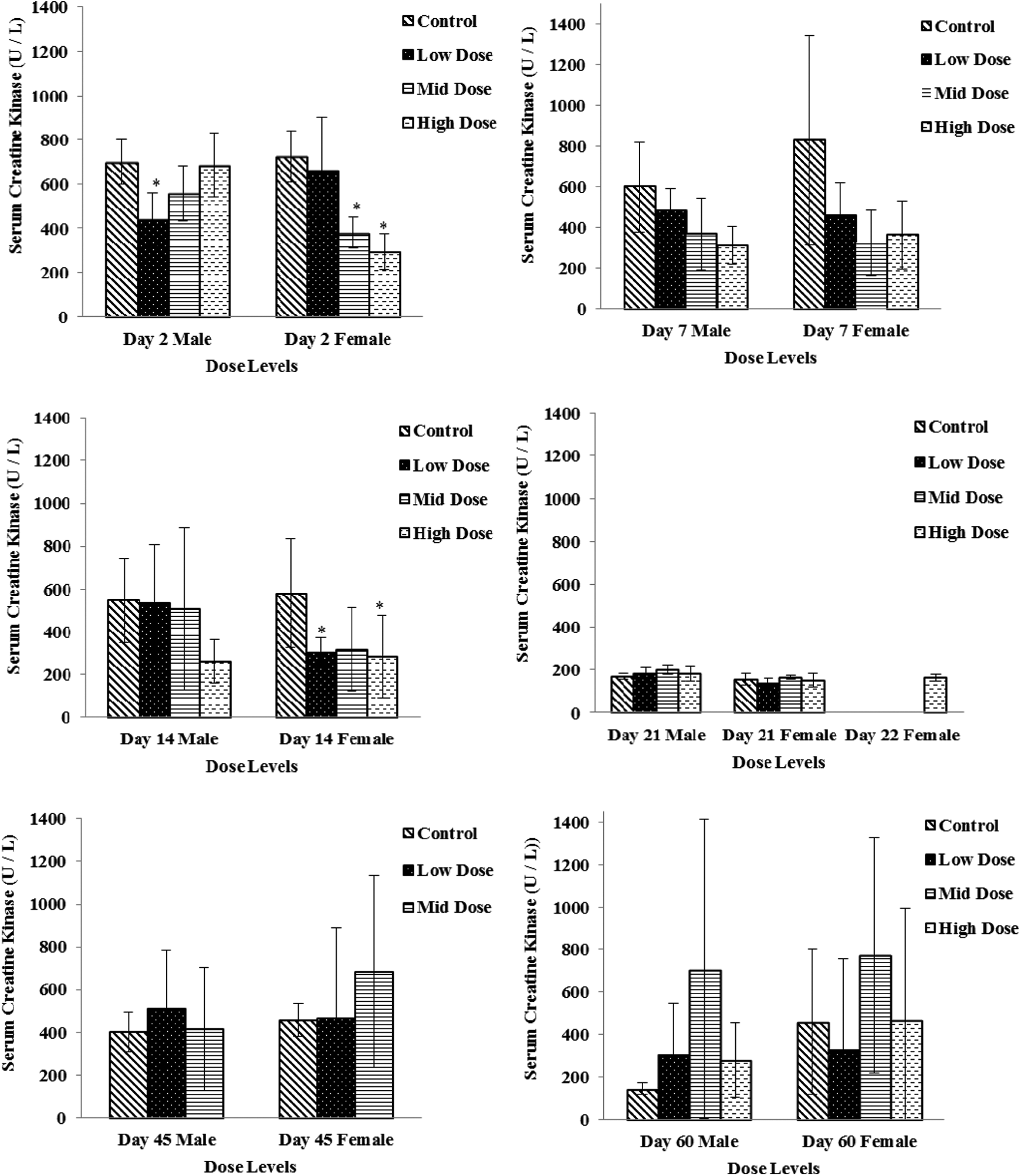
Serum creatine kinase levels in low-dose (100 mg/kg/d), mid-dose (250 mg/kg/d), and high-dose (750 mg/kg/d) rats. When N > 2, an asterisk (*) indicates a statistical (P ≤ .05) difference from the control group.
Necropsy
Gross observations and histopathology
Necropsies on study days 7, 14, and 21
No 2-DG-related gross observations were noted at necropsy. Hearts from animals administered 2-DG for 7, 14, and 21 days were examined. Cardiac lesions are summarized in Table 2. Beginning on study day 14 in males at 750 mg/kg/d and in females at 250 and 750 mg/kg/d, test article-associated microscopic findings of vacuolar degeneration (Figure 4) and hypertrophy of the endothelial cells of the endocardium were present in the heart of animals administered 2-DG. Vacuolar degeneration in the unilateral or bilateral atrial myocardium, and occasionally within the ventricular myocardium, was characterized by vacuoles within the myofibers, often replacing the entire fiber with displacement of the fiber nuclei. Sporadically, vacuoles within atrial tissue were associated with either atrophy or complete loss/replacement of the myofiber as was noticed in 2-DG administered animals in the mid- and high-dose (250 and 750 mg/kg/d) groups on study day 14 (3 of 4 males and 2 of 4 females in the high-dose group and 1 of 4 females in the mid-dose group) and study day 21 (4 of 4 males and females in the high-dose group with the highest incidence). Vacuolar degeneration was coded as minimal (grade 1) for involvement of up to 50% of 1 atrium (usually the left); mild (grade 2) for either involvement of 100% of 1 atrium or up to 50% of both atria; moderate (grade 3) for 100% involvement of 1 atrium and up to 50% of the other; and marked (grade 4) when the lesion approached maximal, involving 100% of both atria. Atrial vacuolar degeneration was often associated with endothelial cell hypertrophy of the atrial endocardium and less frequently within the ventricular endocardium.
Microscopic Cardiac Lesions in Males and Females Administered 2-DG for 14, and 21/22, and 45 Days.
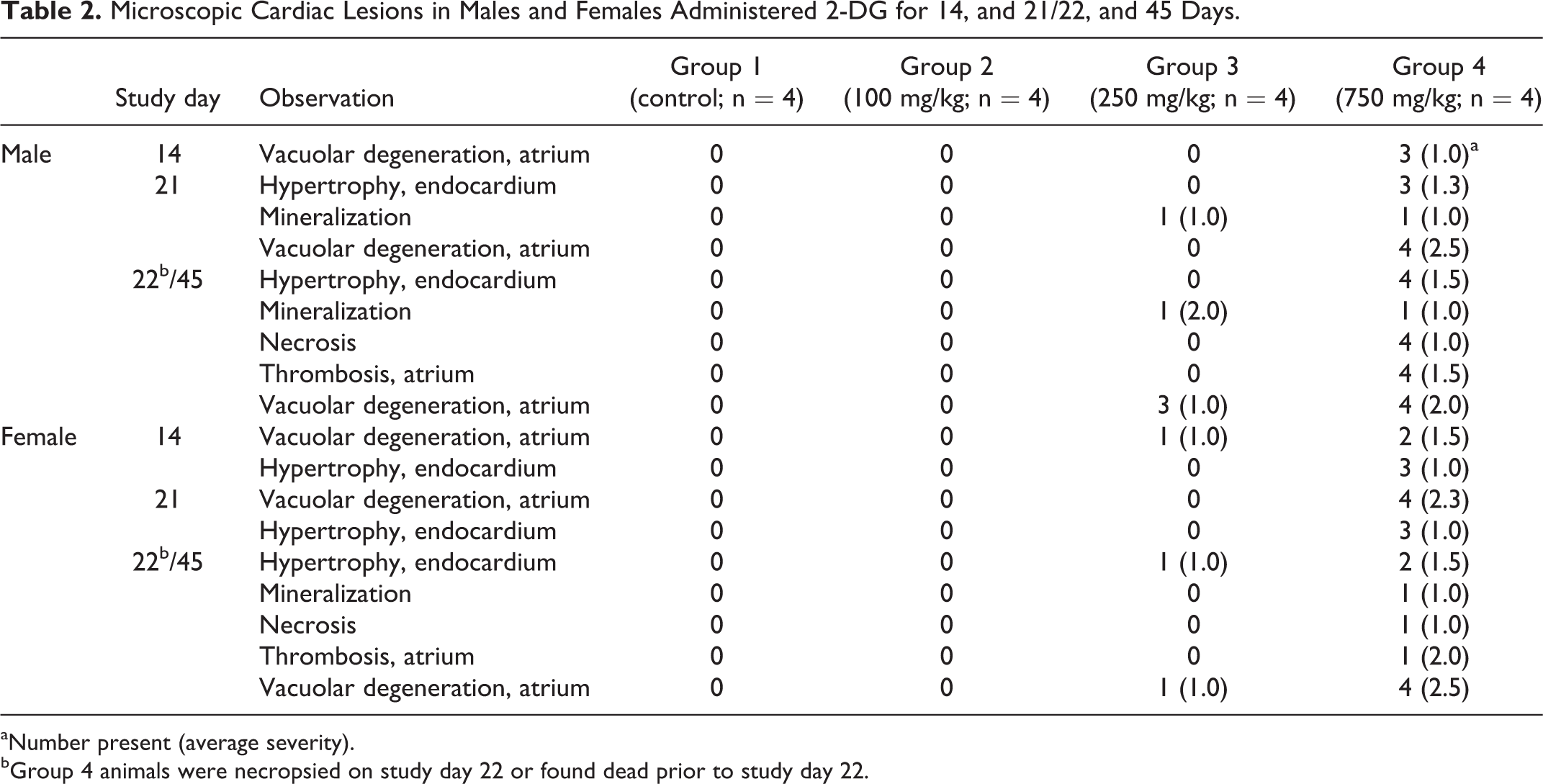
aNumber present (average severity).
bGroup 4 animals were necropsied on study day 22 or found dead prior to study day 22.
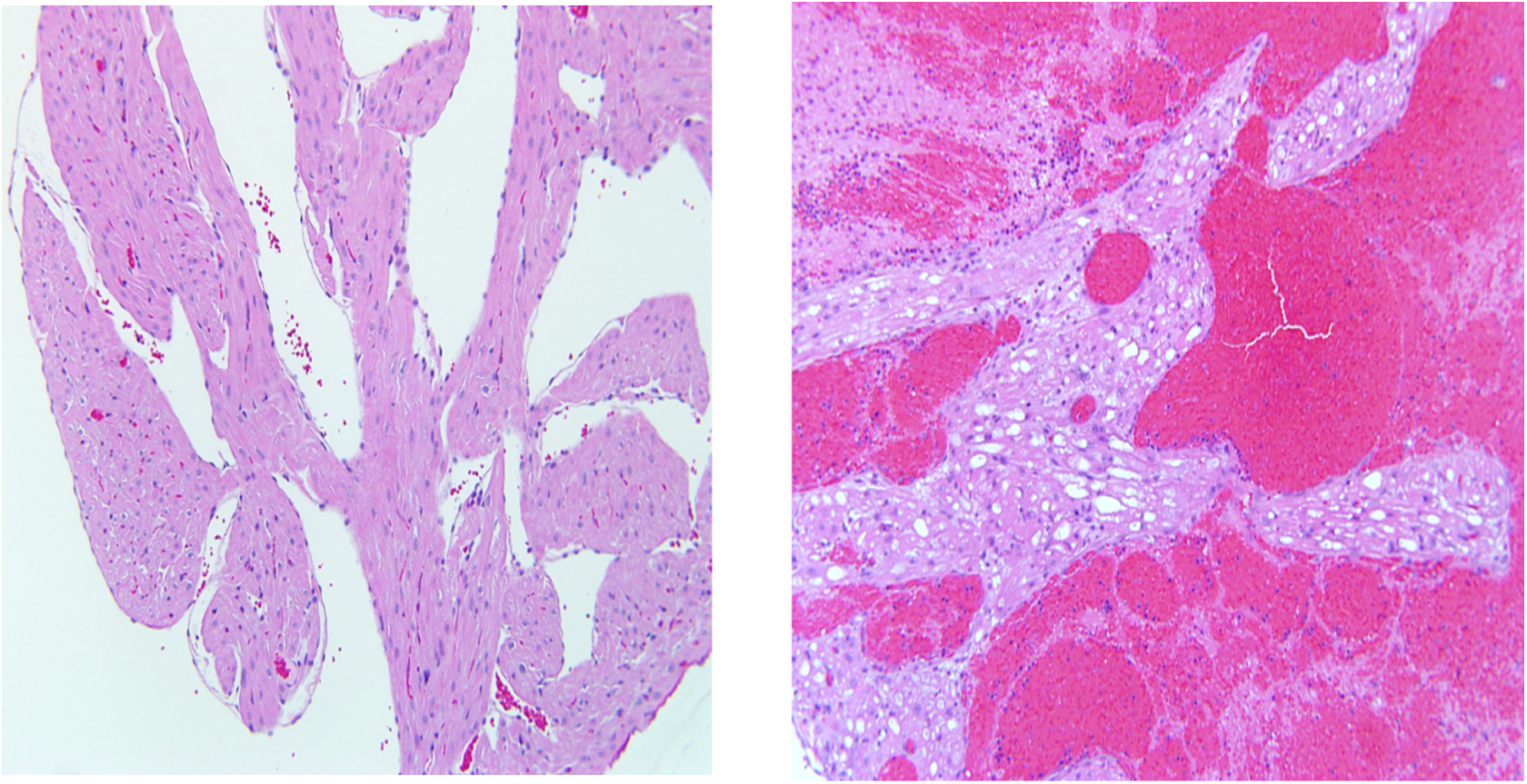
Photomicrographs of sections of the cardiac atrium with H&E stain, 10×; control (left) and high dose (right).
Cardiac myofiber inflammation was seen in some animals from all groups, including controls, and was composed of mixed inflammatory infiltrates associated with scattered, fragmented, or sometimes missing myofibers and was not considered to be test article related. This lesion was indistinguishable from spontaneous cardiomyopathy, which is considered a normal background finding in rats of this age.
Cardiac mineralization occurred in 1 mid-dose (250 mg/kg/d) and 1 high-dose (750 mg/kg/d) male rat. The mineralization consisted of replacement of either myofibers (when associated with degeneration) or mineralization of the elastic fibers at the base of the aorta. The mineralization was considered a secondary effect and not directly related to the administration of 2-DG. There were no test article-related changes noted in the low-dose, 100 mg/kg/d animals.
Forty-five-day interval necropsy (high dose necropsied on study day 22 and early deaths)
Heart tissue lesions are summarized in Table 2. Heart lesions in animals administered 2-DG for 45 days were similar but more marked than rats administered 2-DG for up to 21 days (surviving high dose terminated at 22 days and early death high-dose animals).
Heart lesions such as vacuolar degeneration of the atrium, endocardial (endothelial cell) hypertrophy, inflammation, and mineralization observed in high- and mid-dose animals were similar to that described in the 7-, 14-, and 21-day animals. Vacuolar degeneration and endocardial (endothelial cell) hypertrophy were considered test article related. In 1 mid-dose animal (250 mg/kg/d), vacuolar degeneration of the atrium of the heart and other cardiac changes were also noted. In high-dose animals, thrombosis with associated necrosis occurred in the left atrium in those animals found dead. In animals with minimal (grade 1) atrial thrombosis, up to 50% of the left atrial lumen was occluded; mild (grade 2) affected the entire left atrium; and grade 3 was coded to include the entire left atrium and up to 50% of the right atrium.
Study day 60 recovery necropsy (45-day dosing with 15-day recovery; high-dose group concluded dosing on study day 21 and necropsied on study day 60)
There were no vacuolar degenerative changes in myocardium noted in this group except for a single male rat in the high-dose group, suggesting that test article-associated cardiac alterations were reversible (Table 3).
Microscopic Cardiac Lesions in Recovery Males and Females.a

aDose administration in group 4 animals was terminated on study day 21. Groups 1, 2, and 3 were terminated on day 60.
bNumber present (average severity).
Discussion/Conclusion
The study was conducted to see whether 2-DG-induced cardiac toxicity could be reproduced using clinically relevant route and dosing schedule in a rat model to identify potential cardiac biomarkers that can be useful to monitor cardiac toxicity during its clinical evaluation. 2-Deoxy-
Minor et al 7 have performed a long-term dietary toxicity study of 2-DG in rats that showed 2-DG increases mortality, increases incidence of pheochromocytoma in the adrenal medulla, reduces weight gain secondary to reduced food intake, and increases vacuolation of cardiac myocytes with an increase in autophagic flux. Decreases in the mean arterial blood pressure followed by intravenous administration of 2-DG was observed in anesthetized rats. 24,25
Current subchronic study further characterizes 2-DG-induced cardiac toxicity and identifies potential cardiac biomarkers that might be helpful to monitor cardiac toxicity during clinical trials. Dosing formulations were analyzed for 2-DG concentrations and were found to be within ±10% of the targeted concentrations, confirming the accuracy of dosing formulations. Clinical signs were noticed only in high-dose group animals that included lethargy, rough coat, thin appearance, hunched posture, and reduced/absent feces. By day 22 of the treatment, 4 male and 1 female rat died from the high-dose group, indicating males are more sensitive to 2-DG toxicity. Increased sensitivity of male rats to 2-DG toxicity was also observed by Minor et al. 7 This sex difference in toxicity could be accounted for higher metabolic demand in male rats compared to females as 2-DG is a known inhibitor of glycolysis.
Plasma or serum specimens were evaluated for CK, ANP, BNP, NT-proBNP, cTnI, and cTnT concentrations as potential cardiac toxicity biomarkers. Plasma concentrations of NT-proBNP were significantly increased in a dose-dependent manner on study days 7, 14, and 45. On study day 21, although dose-dependent increases were observed, the changes were not statistically different compared to the concurrent control group possibly due to higher control levels and/or variability. Brain natriuretic peptide concentrations were also statistically increased on study day 45. The increases in NT-proBNP levels were often associated with microscopic myocardial vacuolar degeneration. It appears that NT-proBNP is more sensitive to early 2-DG-induced cardiac injury compared to BNP, as more robust changes in NT-proBNP were noticed, which may be accounted for a longer biological half-life of NT-proBNP than that of BNP (∼70 vs 20 minutes). 26 Elevated levels of NT-proBNP and BNP were decreased by the end of recovery period (day 60), which corresponded with the microscopic evidence of recovery in heart tissue. There were no remarkable changes observed in CK, cTnI, or cTnT concentrations, indicating 2-DG may be causing no or minimal necrotic changes such as myocardial infarction as these markers are more likely be indicative of cell death. There were no significant changes observed in ANP levels. More robust changes observed in NT-proBNP and BNP levels indicating 2-DG-induced cardiac toxicity could be related to cardiac hypertrophy/dilatation of the heart leading to stretching of cardiac tissues and releasing these markers into the blood. This is the first evidence of altered serum NT-proBNP and BNP levels in rats in response to 2-DG toxicity indicating their usefulness in clinical trials of 2-DG as potential cardiac safety biomarkers. If 2-DG-induced cardiac toxicity in rat is relevant to human, these biomarkers may need to be qualified/validated as per the Food and Drug Administration (FDA) guidance. It is quite possible that 2-DG-induced cardiac toxicity could be limited to rodents due to higher metabolic demand, hence, further evaluation of 2-DG toxicity in higher order animal species such as dog or monkey would be helpful.
There were no 2-DG-related gross lesions noted at necropsy. Test article-related microscopic findings in the heart were noted beginning on exposure study day 14 in the 250 and 750 mg/kg/d groups. Vacuolar degeneration of the atrial myocardium associated with endocardial (endothelial cell) hypertrophy was the most significant early lesion. By study day 22, 4 male rats and 1 female rat administered high dose (750 mg/kg/d) of 2-DG had died likely due to cardiac failure associated with atrial thrombosis. In the mid-dose (250 mg/kg/d) group at study day 45, 2-DG related findings consisted of vacuolar degeneration and endocardial (endothelial cell) hypertrophy of the heart. Cardiotoxicity noted in the present study corroborated with the findings of Minor et al 7 and Lane et al. 27
Treatment-related cardiac lesions were not evident in the low-dose (100 mg/kg/d) group. The onset of treatment-related lesions in heart tissue occurred in the mid-dose (250 mg/kg/d) group females following 14 consecutive days of exposure to 2-DG and in the mid-dose group males after 21 consecutive days of exposure. Onset of treatment-related cardiac lesions in both males and females of the high-dose (750 mg/kg/d) group occurred after 14 days of exposure. Notable progression of treatment-related lesions, in both incidence and severity, was observed in the heart tissue of mid- and high-dose group animals from study day 14 to the end of exposure on study days 22 (high-dose group) and 45 (mid-dose group). Following a recovery period of 15 days, treatment-related cardiac lesions were completely remodeled in the mid-dose group animals. After 39 days of recovery, a majority of treatment-related cardiac lesions were remodeled in the high-dose group animals, with one incidence of vacuolar degeneration (atrium) persisting among high-dose males.
2-Deoxy-
Footnotes
Acknowledgments
The authors thank Alan Koester and other Battelle staff for conducting the study.
Author Contributions
P. S. Terse contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. P. S. Joshi contributed to analysis and interpretation, drafted the manuscript, and critically revised the manuscript. N. R. Bordelon and A. M. Brys contributed to design, contributed to acquisition, analysis, and interpretation, and critically revised the manuscript. K. M. Patton and T. P. Arndt contributed to acquisition, analysis, and interpretation and critically revised the manuscript. T. P. Sutula contributed to conception and design, contributed to interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TPS holds a use patent for 2-DG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NCI-Leidos (Contract No. HHSN261200800001E) and NINDS under BrIDGS/NCATS Program.
