Abstract
C-peptide is formed in the biosynthesis of insulin and is therefore deficient in patients with type 1 diabetes mellitus. A pegylated form of human synthetic C-peptide (CBX129801) has been developed to extend the half-life of the native peptide and is undergoing clinical investigation as replacement therapy to treat diabetic peripheral neuropathy. This monkey study was conducted to evaluate the toxicity of CBX129801 with weekly subcutaneous dosing for 39 weeks at dose levels of 0 (vehicle), 0.4, 1.33, and 4.0 mg/kg/wk. No systemic adverse effects were observed at any dose with maximal CBX129801 plasma concentrations of 735 to 1050 nmol/L during the dosing period (physiological range is 1-3 nmol/L). CBX129801-related effects were limited to minimal macrophagic vacuolization at the injection sites and in the associated draining (axillary) lymph nodes; these local effects largely resolved by the end of a 7-week recovery period. No systemic macrophagic vacuolization was observed. Additionally, there was no histological evidence for plaque formation in the major arteries of these nondiabetic animals.
Keywords
Introduction
In type 1 diabetes mellitus, autoimmune destruction of the pancreatic β cells results in deficient production of proinsulin and thus both insulin and C-peptide. Patients with type 1 diabetes mellitus are dependent on insulin injections to compensate for the lack of endogenous insulin production, but no replacement of C-peptide is currently available. Data from patients with type 1 diabetes mellitus and animal models exhibiting diabetic complications demonstrate that C-peptide is biologically active with beneficial effects on functional and structural abnormalities of both the kidneys 1 –3 and the peripheral nerves. 4 –6 Due to the short half-life (T1/2) of C-peptide, approximately 1 hour, it was necessary to administer the C-peptide subcutaneously (SC) multiple times daily to mimic physiological exposures. With the goal to have an acceptable commercial product for chronic use, Cebix developed a long-acting form of C-peptide via the conjugation of the 31 amino acid synthetic human peptide to a branched 40-kDa polyethylene glycol (PEG) at its nonactive terminus. The patented new chemical entity, CBX129801, retains biological activity of comparable potency to native (ie, nonpegylated) C-peptide with an extended circulating residence time in the plasma.
CBX129801 entered clinical testing based on results from 28-day toxicology studies in Sprague-Dawley rats and cynomolgus monkeys with weekly SC dosing of CBX129801. In the 28-day monkey study, there was a safety finding at the mid and high doses making the low dose the no observed adverse effect level (NOAEL). This NOAEL dose provided 27-fold and 29-fold safety margins for the human starting dose based on body surface area and plasma exposure expressed as area under the curve (AUC), respectively. Comparing the NOAELs between rat and monkey in the 28-day studies, the monkey was the more sensitive species. The monkey is also likely the most appropriate species for predicting the toxicity of CBX129801 in humans, since there is a high degree of homology between the cynomolgus monkey and human C-peptide amino acid sequences, which differ by only 1 amino acid substitution. Results from a phase 1 clinical study with CBX129801 in patients with type 1 diabetes mellitus showed that replacement levels of C-peptide were achieved with a T1/2 of 6 to 8 days as is desirable for a once-weekly product.
The objective of the present report is to describe the in-life observations, histopathologic findings, toxicokinetics (TK), and immunogenicity assessment from the chronic toxicity study conducted in cynomolgus monkeys with weekly SC dosing of CBX129801 for 26 and 39 weeks with a recovery period of 7 weeks. The results provided supportive safety information for the initiation of two phase 2 clinical studies to evaluate the efficacy and safety of CBX129801 for up to 1 year of treatment in patients with type 1 diabetes mellitus having diabetic peripheral neuropathy, a common complication of diabetes. Additionally, in the monkeys, an ancillary assessment was done to microscopically examine the major arteries to determine whether C-peptide is atherogenic after 9 months of exposure to supraphysiologic levels of CBX129801. This assessment addressed published reports in the literature that elevated C-peptide levels are associated with atherosclerotic plaque formation in type 2 diabetes. 7,8
Materials and Methods
Test Article and Vehicle
CBX129801 (pegylated synthetic human C-peptide) was manufactured by PolyPeptide Laboratories (Torrance, California) and formulated by Althea Technologies (San Diego, California; now Ajinomoto Althea); both are fully accredited good manufacturing practice sites. CBX129801 drug product (lot 1-FIN-0988) was a clear aqueous solution (10 mmol/L sodium phosphate buffer in 4.7% sorbitol at pH 6.0) that was filled under sterile conditions into 2-mL vials at 20 mg/mL for phase 1 clinical supply. The test article was diluted at the toxicology contract research laboratory with 1× phosphate-buffered saline (PBS) that was diluted from 10× stock (Sigma-Aldrich, Inc, St Louis, Missouri) with sterile water for injection, USP (Baxter Healthcare Corporation, Deerfield, Illinois), to achieve the desired dose with a 0.5-mL/kg injection volume. The vehicle for the control group was PBS.
Animals and Housing
The study was conducted in compliance with good laboratory practices (GLPs) at an AAALAC International-accredited contract research laboratory (WIL Research, Ashland, Ohio). All animals used in the study were treated humanely, with due consideration for alleviation of distress and discomfort. Male and female cynomolgus monkeys (Macaca fascicularis, of Chinese origin; approximately 1.8-4.9 years old) were obtained from Primate Products (Miami, Florida). Each animal was uniquely identified with a chest tattoo by the supplier prior to shipment, and SC microchips were implanted in each hindlimb on site during the acclimation period. Animals were socially housed in groups of 2 or 3 with access to an enriched environment; they were separated only on days of dosing to facilitate pre- and postdosing observations. Twice daily the animals were offered biscuits (PMI Nutrition International, LLC, Certified HI-Fiber Primate Diet 5K91), and this diet was supplemented with other nutrients (eg, fresh fruit). Water was provided ad libitum throughout the study period. The room temperature was 19°C to 25°C and the humidity was 30% to 70% throughout the study. The fluorescent light cycle was 12 hours/d, and there were at least 10 room fresh air changes per hour. All animals were randomly assigned to treatment groups. Body weights ranged from 2.1 to 3.7 kg for the males and 2.2 to 3.4 kg for the females at the initiation of dosing.
Experimental Design
Groups of 3 monkeys/sex received 26 or 39 weekly SC injections via 23-gauge needle of vehicle or CBX129801 at dosage levels of 0 (vehicle), 0.4, 1.33, or 4.0 mg/kg/wk at 2 alternating sites in the scapular area prior to necropsy (primary necropsy). An additional 4 monkeys/sex in the vehicle control and 1.33 and 4.0 mg/kg/wk groups were dosed on the same regimens, and of these, 2/sex/group entered a 7-week (no dosing) recovery period upon completion of the 26- or 39-week dosing periods prior to necropsy (recovery necropsies). Blood samples were collected in K3EDTA tubes for TK and antidrug antibody (immunogenicity) assessments at selected intervals throughout the study. Analyses using enzyme-linked immunosorbent assays were conducted in compliance with GLPs at MicroConstants, Inc (San Diego, California).
The monkeys were observed for morbidity and mortality twice daily. Clinical observations were recorded at the time of dosing, approximately 1 to 2 hours after dosing, and once daily on nondosing days including during the recovery period. Detailed physical examinations were performed once weekly as were macroscopic injection site observations at approximately 24 hours after each dose administration. Body weights were recorded once weekly. Ophthalmologic examinations were performed by a board-certified veterinary ophthalmologist prior to the start of dosing and at the end of the dosing and recovery periods. Hematology, coagulation parameters, clinical chemistry, thyroid hormone analysis, and urinalysis were performed before the initiation of dosing and just prior to the scheduled primary and recovery necropsies. Electrocardiograms (ECGs) were recorded for all animals once prior to the start of dosing and at approximately 48 hours (time of maximum CBX129801 plasma concentration [Tmax]) after the first dosing and following dosing in weeks 25, 31, and 38 and near the end of the recovery periods. All monkeys received a complete gross necropsy and weights were collected for a standard list of 16 organs. A complete standard list of tissues was collected at each necropsy. Additionally from all groups in the 9-month primary and recovery animals, along with the routine cross section of thoracic aorta, sections of major arteries (aortic arch, abdominal aorta [at junction of the renal arteries], carotid artery, femoral artery, and coronary artery [within routine sections of the ventricles]) were collected. A board-certified veterinary pathologist blinded to treatment examined all tissues microscopically.
Statistical Analysis
Analyses were conducted using 2-tailed tests for minimum significance levels of 1% and 5%, comparing each CBX129801-treated group to the control group by sex. Statistical analyses were not conducted if the number of animals was 2 or less. Body weight, ECG measurements, clinical pathology, and organ weight data were subjected to a parametric 1-way analysis of variance (ANOVA) 9 to determine intergroup differences. If the ANOVA revealed statistically significant (P < 0.05) intergroup variance, Dunnett test 10 was used to compare the CBX129801-treated groups with the control group.
Results
Test Article
CBX129801 remained within specifications for all attributes tested (including appearance, pH, and purity) through 12 months at the recommended storage condition of 2°C to 8°C indicating that the test article was stable for the duration of the study. The results of dose formulation analyses indicated that the CBX129801 concentrations ranged from 101% to 106% of nominal target concentrations. No CBX129801 was detected in the vehicle.
In-Life Observations
One control group male was euthanized after 6 months in the study with an undetermined cause of death. All remaining animals survived until the scheduled necropsies. All observations in the CBX129801-treated groups were noted with similar incidence in the control group were limited to single animals, were not noted in a dose-related manner, and/or were common findings for laboratory monkeys. Therefore, there were no CBX129801-related effects on clinical status, macroscopic injection site observations, body weights, hematology, clinical chemistry, coagulation parameters, thyroid hormones, urinalysis, ophthalmologic examinations, ECGs, gross necropsy observations, or organ weights.
Histopathology
After 9 months of dosing, CBX129801-related histologic effects were limited to the minimal presence of vacuolated macrophages at the SC injection sites and draining axillary lymph nodes. This was a subtle microscopic finding, not evident grossly, and not considered to be adverse. Minimal infiltrates of vacuolated macrophages were noted in both injection sites of animals administered ≥0.4 mg/kg/wk (ie, all CBX129801 doses) and in the axillary lymph nodes of some animals administered ≥1.33 mg/kg/wk. Macrophages in the injection sites were characterized as random cell clusters or single vacuolated macrophages in the subcutis or deep dermis. Not present at all injection sites, the numbers of vacuolated macrophages were typically very few with the example shown in Figure 1 for one of the larger cell clusters in the entire study. For some animals, the axillary lymph nodes exhibited clusters of vacuolated macrophages within the subcapsular or medullary sinuses. The overall incidence of vacuolated macrophage infiltration was diminished at the end of the recovery period.
Histological sections stained with hematoxylin and eosin from subcutaneous injection sites in cynomolgus monkeys following dosing (0.5 mL/kg) of CBX129801 or vehicle. A, Photomicrograph of male animal injected with high dose of CBX129801 for 26 weeks showing cluster of vacuolated macrophages (denoted by arrow; one of the larger ones in the study). Most of the time, such macrophage infiltrates were random and often contained only 1 to several vacuolated macrophages. The finding of infiltrated macrophages largely resolved during the recovery period. B, Photomicrograph of female animal injected with vehicle for 26 weeks showing normal collagen and adipose tissue with no clusters of macrophages.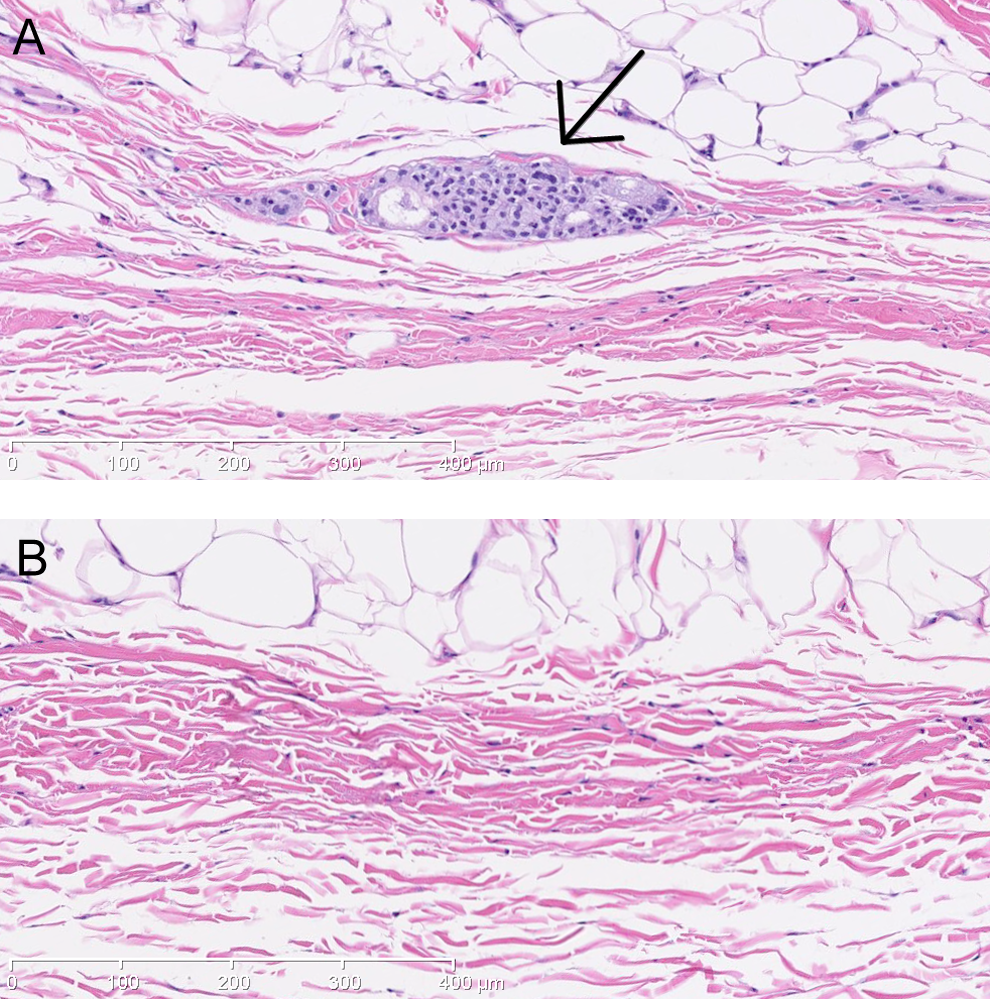
CBX129801-related microscopic changes were not observed in the major artery sections (thoracic aorta, aortic arch, abdominal aorta, carotid artery, femoral artery, and coronary artery) that were examined. Focal intimal hyperplasia was seen in the aortic arch of males and females from all groups, which was characterized microscopically as localized thickening of the intima. This finding was considered a normal response to the high blood flow (turbulence) in that region.
Toxicokinetics
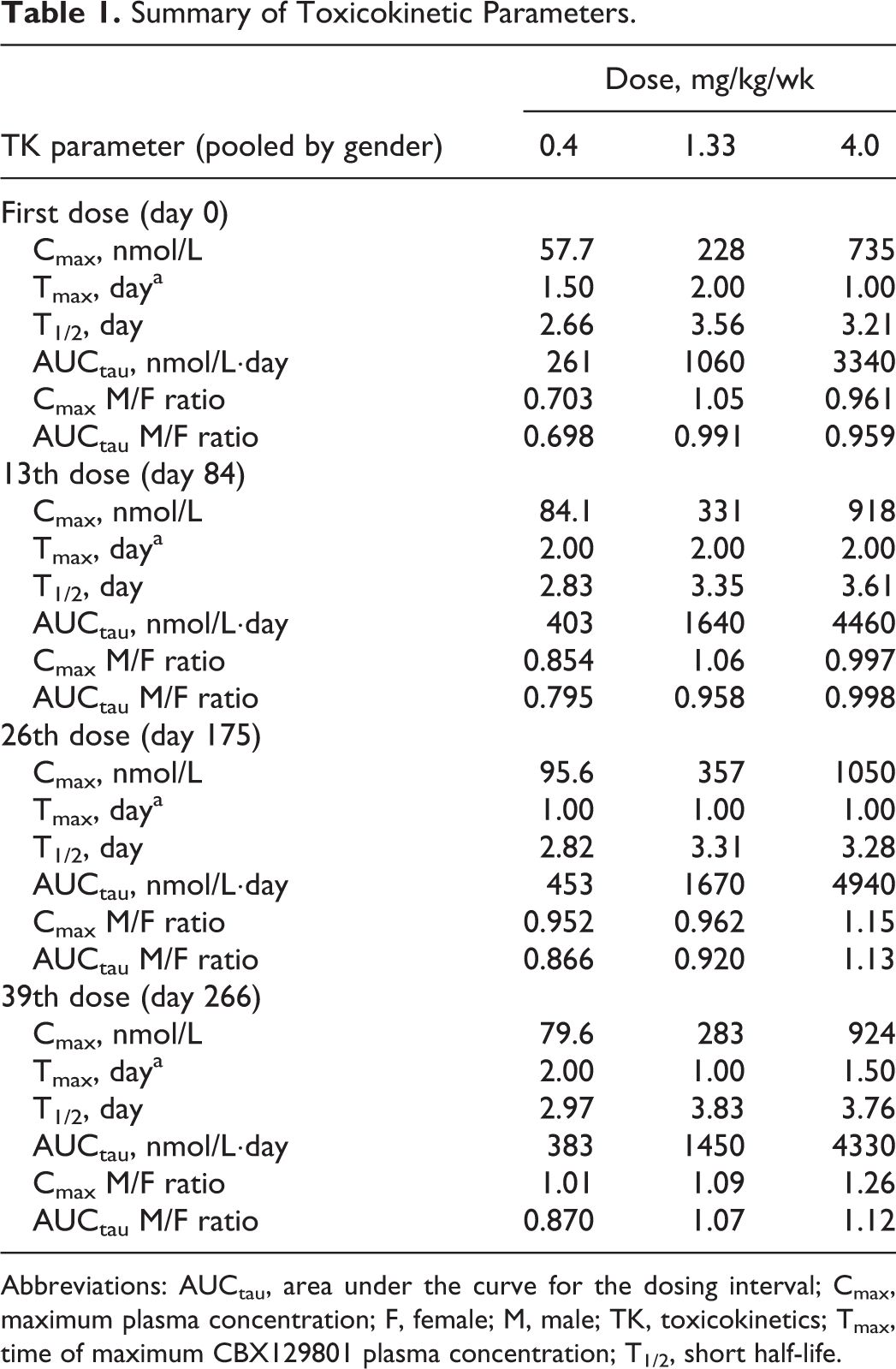
As shown in Figure 2, there were no remarkable gender differences for CBX129801 exposure, therefore, TK data are presented pooled for both sexes. The mean maximum plasma concentration (Cmax) following the first dose was 58, 228, and 735 nmol/L for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively (Table 1). Following the 26th dose (day 175), mean Cmax was 96, 357, and 1050 nmol/L for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively. Following the 39th dose (day 265), mean Cmax was 80, 283, and 924 nmol/L for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively. The Tmax occurred at 1 to 2 days in all groups at all intervals. The mean AUC for the dosing interval (AUCtau) following the first dose was 261, 1060, and 3340 nmol/L day for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively. Following the 26th dose, mean AUCtau was 453, 1670, and 4940 nmol/L day for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively. Following the 39th dose, mean AUCtau was 383, 1450, and 4330 nmol/L day for the 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively. Exposure was dose proportional at both intervals. Exposures were slightly higher following the 26th dose as compared to the first dose, but no additional increase was seen following the 39th dose. The T1/2 was 2.7 to 3.8 days. Steady state was achieved within 28 days. Tmax and T1/2 did not change with dose or with repeated dosing. Upon cessation of dosing, the plasma concentrations of CBX129801 markedly decreased over time and were substantially low during the recovery periods for all doses.
Mean plasma concentration–time profiles of CBX129801 in male and female monkeys following subcutaneously administration of CBX129801 over 39 weeks. There are no apparent gender differences in exposure response. CBX129801 concentrations increased as a function of CBX129801 dose and appeared to achieve a steady state on or before 28 days after start of dosing. At a given dose, there were no statistical significant differences (P > 0.05) among CBX129801 concentrations on days 28, 84, 175, and 266. The plasma concentrations of CBX129801 markedly decreased over time and were substantially low during the entire recovery period for all doses (last dose administered on day 266). Summary of Toxicokinetic Parameters. Abbreviations: AUCtau, area under the curve for the dosing interval; Cmax, maximum plasma concentration; F, female; M, male; TK, toxicokinetics; Tmax, time of maximum CBX129801 plasma concentration; T1/2, short half-life.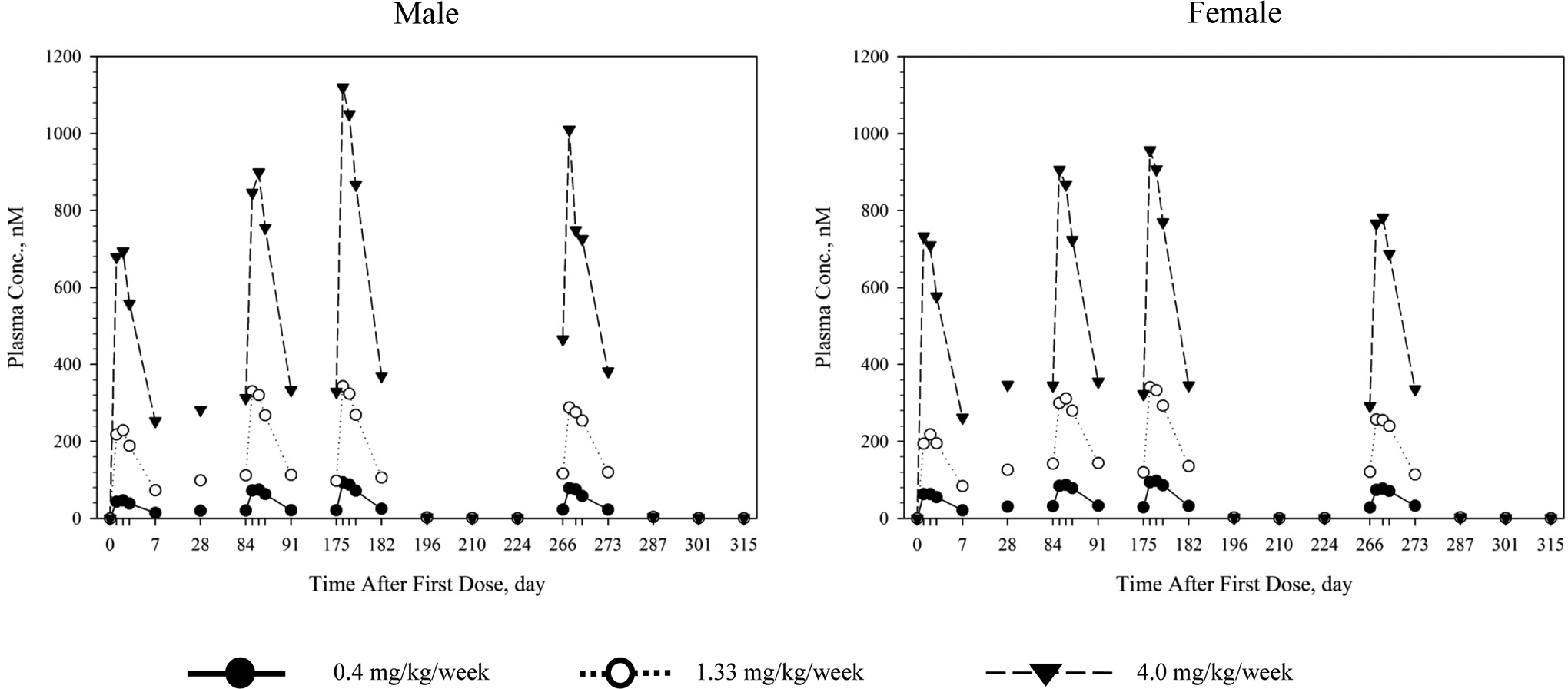
Antidrug Antibodies
At the end of 6 and 9 months of dosing, a total of 10 (of 71) samples were positive for antidrug antibodies: 4, 3, 0, and 3 samples from the vehicle, 0.4, 1.33, and 4.0 mg/kg/wk groups, respectively, for mixed genders and at various time points. The percentage of inhibitions that caused these samples to be considered positive were only marginally elevated, and no dose response was apparent despite marked and dose-proportional differences in exposures among the groups. Further, very high drug exposures were observed throughout the study. Therefore, no meaningful formation of antibodies against CBX129801 was considered to have occurred.
Discussion
The salient findings of this study with monkeys administered CBX129801 weekly via the clinical SC route for 9 months are the absence of systemic toxicity and lack of immunogenicity. This is not unexpected since C-peptide is an endogenous peptide (with close sequence homology between human and monkey), the PEG used to modify the peptide has a large therapeutic window when comparing the amounts used in approved products to the doses associated with toxicity, and PEG is often used to reduce antibody production to its conjugated protein. 11 Even the highest dose was very well tolerated and the CBX129801 exposures achieved in the monkeys were adequate for an evaluation of the potential toxicity of CBX129801 when considering the anticipated human exposure. Specifically, a target dose of 0.8 mg of CBX129801 has been identified from the phase 2a study 12 conducted with CBX129801 in patients with type 1 diabetes mellitus as one that restores the levels of C-peptide to the physiological range of 1 to 3 nmol/L. (Note that each 1 nmol/L of CBX129801 contains 1 nmol/L of C-peptide.) An exposure margin can be estimated from the highest CBX129801 dose in monkeys after 39 weeks of dosing (AUC = 4330 nmol/L day) and using a conservative assumption of a constant CBX129801 level in the plasma of 3 nmol/L (AUC of 21 nmol/L day). The resulting exposure margin is >200-fold, which is in excess of the 50-fold exposure margin allowed to define an acceptable study “high dose” per guidelines of the International Conference on Harmonization. 13 The pharmacokinetics of CBX129801, as shown in Figure 2, implies that a 3 times higher CBX129801 dose (ie, 2.4 mg) is expected to yield a likewise increase in CBX129801 plasma levels. In this case, the corresponding safety multiple is still quite large (ie, >66-fold). Both the 0.8 and 2.4 mg doses of CBX129801 are currently being evaluated for a 1-year treatment period in an international phase 2b study in 250 patients with type 1 diabetes mellitus having diabetic peripheral neuropathy, which is a common complication affecting up to 50% of patients with diabetes. 14
The CBX129801 doses used in this chronic monkey study were based on the results from the 28-day monkey study in which there were histologic findings in the thyroid (decreased organ weight and decreased follicle size) at the mid and high doses (4.0 and 13.3 mg/kg/wk, respectively, 5 total weekly SC doses). Because of these findings, which were reversible and had no associated change in function as assessed by thyroid hormones, the high dose in the chronic monkey study was selected as 4.0 mg/kg/wk. However, at this dose with weekly SC administration for 39 weeks, which represents about 8 times longer exposure than the 28-day study, there were no adverse findings in the thyroid. No definitive explanation was identified for why the thyroid findings were not replicated in the chronic toxicity study, but the batch of CBX129801 used in the 28-day monkey study was research grade with approximately 30% free PEG (in comparison, the clinical material has <3% free PEG), which may have been a contributing factor for the observed thyroid findings on the 28-day study. Importantly, no effects on thyroid function as assessed by abnormal changes in hormone levels were observed in the patients with type 1 diabetes mellitus enrolled in the phase 1 and phase 2a studies conducted with weekly SC dosing of CBX129801 up to 12 weeks.
The only noteworthy positive finding in the present study was the cytoplasmic infiltration of vacuolated macrophages in the tissues at the injection sites and draining axillary lymph nodes following SC administration of CBX129801 (Figure 1). The process of macrophagic vacuolization is presumably a normal physiologic phagocytosis and clearance of the pegylated C-peptide; it has been documented for other pegylated products as discussed subsequently. The injection site and lymph node findings were considered to be a local effect of CBX129801 and, absent any inflammatory response, not adverse or indicative of systemic toxicity. Further, the findings were similar to those previously observed with CBX129801 in the 28-day monkey study, although with a lesser severity due to the lower doses of CBX129801 administered and injection site alternation (the same injection site was used in the 28-day study). In the clinical testing of CBX129801, patients have been instructed to inject CBX129801 separate from insulin and at rotating sites. Additionally, the dose volume in patients is 0.5 mL as compared to 0.5 mL/kg in the monkey toxicity studies. There have been no reports of clinically meaningful injection site reactions in patients dosed with CBX129801 to date.
Significantly, in the present study, systemic tissues showed no vacuole formation with animals administered CBX129801 SC weekly for either 6 or 9 months. This is in contrast to a number of approved pegylated drugs that have reported in animals the presence of vacuoles in various tissues following repeated parenteral administration. The distribution of a pegylated protein into tissues is determined by characteristics of the PEG (ie, molecular weight, structure, and dose) and the tissue distribution of the native protein. 15 Low-molecular-weight PEGs are able to move easier from the circulation to extravascular tissues and then back, and the formation of vacuolated macrophages or vacuoles in tissues due to a prolonged accumulation of a pegylated compound takes time. This has been demonstrated in toxicology studies conducted with some of the approved pegylated drugs (Somavert, 16 Krystexxa, 17 and Cimzia 18 ) in which the vacuolization of macrophages and/or tissues was not observed until there had been repeated dosing for at least 6 months. Regulatory authorities approved these drugs because there were no apparent functional deficits associated with the vacuoles; however, based on feedback from regulatory authorities, there is concern with at least 1 new pegylated compound for the potential to cause vacuolization in critical organs such as the brain (ie, the ependymal cells in the choroid plexus), particularly in vulnerable populations. Case in point, a European Medicines Agency (EMA) published memo in 2012 discusses the clinical testing of pegylated drugs in the pediatric population. 19 After a review of the pegylated drug products approved for the European market, one of the EMA recommendations is to first analyze the biodistribution of the pegylated drug prior to pediatric dosing, unless the monthly PEG exposure is significantly lower than the cases where vacuolization of ependymal cells in the choroid plexus have been observed. This exposure was specified as ≥0.4 μmol/kg/month, which is 100-fold more than the PEG exposure corresponding to a CBX129801 dose of 2.4 mg (highest dose in the phase 2b study with CBX129801, assuming a 55-kg participants). Because there was no observed systemic tissue vacuolization after 6 to 9 months of CBX129801 dosing in animals and given the relatively small PEG load in CBX129801 present in the anticipated marketed dose of 0.8 to 1.0 mg, it is considered low risk that CBX129801 will have PEG-related toxicities with chronic dosing in adults and children.
CBX129801 is being developed as a treatment to replace C-peptide in patients with type 1 diabetes mellitus who have microvascular complications such as peripheral neuropathy, nephropathy, and retinopathy. This patient population along with patients with type 2 diabetes mellitus also has an increased incidence of macrovascular disease, manifested primarily as atherosclerosis of the coronary blood vessels, the cerebral vascular system, and arteries in the legs. The biochemical mechanisms underlying the atherogenic process are not fully understood but involve tissue insulin resistance, glucose-mediated formation of reactive oxygen species, and low-grade vascular inflammation with endothelial cell dysfunction. 20 Insulin and C-peptide are absent in type 1 diabetes mellitus but are elevated above normal levels early in the progression of type 2 diabetes mellitus; this feature of the disease has implicated both hormones as having potential roles in the pathogenesis of atherosclerosis. Recent studies have asserted that C-peptide directly contributes to the atherogenic process in type 2 diabetes mellitus due in part to a chemotactic effect of C-peptide on inflammatory cells within atherosclerotic plaque. 21 In a model with atherosclerosis-prone mice on a high fat diet, there was increased inflammatory cell infiltration and lesion development when C-peptide was administered for 12 weeks at C-peptide concentrations 4- to 5-fold above normal physiological levels. 22 In the present study, with C-peptide administered for 9 months to nondiseased young adult monkeys, there was no histological evidence of plaque formation in major arteries known to exhibit atherosclerotic lesions at C-peptide exposures >200-fold above physiological levels. Thus, C-peptide even at supraphysiologic levels does not appear to initiate plaque formation, although at high concentrations it may contribute to the sequelae of a lesion that results from other factors in type 2 diabetes mellitus. The point is moot when considering CBX129801 treatment of type 1 diabetes mellitus because the goal is to replace C-peptide at or near normal physiological levels.
Conclusion
The NOAEL for systemic toxicity with weekly SC administration of CBX129801 (pegylated human C-peptide) in this 39-week study was 4.0 mg/kg/wk, which was the highest evaluated dose. CBX129801-related effects were limited to minimal macrophagic vacuolization at the injection sites and in the associated draining axillary lymph nodes, for which the overall incidence was diminished at the end of the recovery period. There was no cellular vacuolization in tissues at PEG exposures markedly higher than the PEG load in CBX129801 at the anticipated efficacious clinical dose. Additionally, the supraphysiologic exposures of C-peptide (in pegylated form) did not induce plaque formation in these healthy (nondiseased) young adult monkeys.
Footnotes
Acknowledgment
The authors gratefully acknowledge the contributions of study pathologist James B. Nold, DVM, PhD, DACVP of WIL Research Laboratories, LLC in the evaluation and interpretation of this study.
Author Contributions
Dennis Naas contributed to conception and design, interpretation, and critically revised the article. Michelle Mazzoni contributed to conception and design, interpretation, and drafted the article. Ahmed Kousba contributed to analysis and critically revised the article. Teresa Morris contributed to acquisition and interpretation and critically revised the article. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Corresponding author Dennis Naas is a member of the IJT editorial Board and is recused from editing this manuscript. No other potential conflicts of interest are known to exist.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
