Abstract
Bood POZ containing gene type 2 (BPOZ2), a Broad-Complex, Tramtrack, and Bric a brac domain containing protein, is an adaptor protein for the E3 ubiquitin ligase scaffold protein CUL3. It plays an important role in acute carbon tetrachloride (CCl4)-induced liver injury and regeneration in mice. In this study, we investigated the role of BPOZ2 in the process of liver fibrosis induced by chronic CCl4 treatment. The results indicate that BPOZ2 deficiency decreases sustained activation of hepatic stellate cells, attenuates collagen αI(I) and tissue inhibitor of matrix metalloprotease 1 expression, and decreases liver fibrosis after repeated CCl4 administration. These findings suggest BPOZ2 as a new therapeutic target for the prevention and treatment of hepatic fibrosis in chronic liver disease.
Introduction
Liver fibrosis is the major consequence of repeated liver tissue damages such as viral infection, toxin- or drug-induced injury, and autoimmune or metabolic diseases. 1 These damages impair normal liver function and eventually lead to cirrhosis and organ failure. 2,3 Several models of liver fibrosis have been established. Among these, carbon tetrachloride (CCl4)-induced liver fibrosis in rodent is the most widely studied. 4 –6 Chronic CCl4 treatment causes hepatocyte necrosis and apoptosis with associated hepatic stellate cell (HSC) activation and liver fibrosis. 7 Liver fibrosis is characterized by an excessive deposition of extracellular matrix proteins such as collagen. 2,3 During progression of fibrosis, HSCs are activated by extracellular matrix deposition and migrate into the area of injury due to chemotaxis. 8,9 Activated HSCs have a myofibroblastic phenotype notably characterized by the expression of α-smooth muscle actin (α-SMA). These cells are responsible for excessive synthesis of extracellular matrix components, particularly collagen I, and of extracellular matrix remodeling enzymes such as tissue inhibitor of matrix metalloprotease (TIMP), which leads to the inhibition of collagenase activity and subsequent accumulation of matrix proteins in the extracellular space. 2,8,10 Bood POZ containing gene type 2 (BPOZ2), a Broad-Complex, Tramtrack, and Bric a brac (BTB) domain containing protein cloned from the human leukocyte cell complementary DNA library in 2000, is an evolutionarily conserved gene. 11 Mouse BPOZ2 gene localizes at the region of chromosome 6qD1, comprising 12 exons. Sequence pattern analysis shows that it contains N-terminal ankyrin (ANK) repeat, a bipartite nuclear localization signal and 2 BTB domains. It is well known that both ANK (ANK repeat domain) and BTB domain are protein–protein interaction domains and play important roles in cell proliferation and differentiation. 12 – 17 Recent studies show that BPOZ2 is an adaptor protein for the E3 ubiquitin ligase scaffold protein Cullin 3. Although there is some in vitro evidence indicating BPOZ2 is involved in cell cycle regulation, 18,19 as a relative new gene, the role of BPOZ2 in vivo is poorly understood.
Our previous study shows that BPOZ2 deficiency delays liver injury and impairs hepatocyte proliferation after acute CCl4 treatment, accompanied by altered kinetics of CYP2E1 protein expression, compromised cyclin D1 expression, and shortened duration of ERK activation, suggesting it is an important regulator involved in liver injury process. 20 Here, we use BPOZ2-deficient mice to investigate the role of BPOZ2 in liver fibrosis after repeated CCl4 administration.
The results indicate that BPOZ2 deficiency decreases sustained activation of HSCs, attenuates collagen αI(I) and TIMP-1 expression, and reduces liver fibrosis after chronic CCl4 treatment. Our findings extend the comprehension of BPOZ2 function in vivo once again, suggesting BPOZ2 plays essential roles in liver physiology, especially in the process of liver injury and repair.
Materials and Methods
Animals and Administration of CCl4
Female mice (8-10 weeks old) with a mixed genetic background of C57BL6 and 129SvJ and their age- and sex-matched wild-type littermates were used in this study. The BPOZ2 knockout mice were generated in our laboratory. 19,21 Exons 2 to 12 and partial exon 1 of BPOZ2 gene were deleted. Mice with BPOZ2 deletion of both alleles (−/−) and homozygous wild type (+/+) were used in this study. The mice were allowed free access to food and water and housed at 25°C with a 12-hour light–dark cycle in a specific pathogen-free environment. To genotype genetically modified mice, 0.5 cm of the tip of mouse tail was removed from anesthetized mouse. DNA isolated from tail biopsies was extracted and applied to polymerase chain reaction (PCR) using primers described in Xiang Y-G, et al. 21 In studies of acute CCl4 liver injury (indicated as 1, 2, and 3 days in figures), a single 1.0 mL/kg dose (CCl4 diluted 1:4 in mineral oil) of CCl4 was administered intraperitoneally to mice. Animals were killed by CO2 at 0, 24, 48, and 72 hours after CCl4 injection (at least 6 mice from each group for each time point). For chronic CCl4-induced liver fibrosis (indicated as 21 days), mice were injected intraperitoneally with 1.0 mL/kg body weight dose (CCl4 diluted 1:4 in mineral oil) of CCl4 on Tuesday and Friday of each week for 3 weeks (total of 6 doses) in treatment group, while a same dose of mineral oil (without CCl4) was delivered to mice in the control group (indicated as 0 day). Each dose was adjusted to animal body weight obtained at the day of injection. At least 6 mice of each genotype at any given time point were examined. Mice were killed by CO2 and liver samples were harvested, rapidly frozen in liquid nitrogen, or fixed in 4% paraformaldehyde solution in phosphate-buffered saline (pH 7.4) for histological analysis. Samples were fixed in paraformaldehyde overnight. All procedures of animal experiments were approved by the Institutional Animal Care and Use Committee of Shanghai Jiao Tong University School of Medicine and the euthanasia protocol was in accordance to American Veterinary Medical Association guideline.
Immunohistochemical Analysis
Tissue sections were prepared according to paraffin-embedding procedure. After being deparaffinized and rehydrated, sections were processed in 10 mmol/L citrate buffer (pH 6.0) and continuously heated at 92°C to 96°C for 20 minutes for antigen retrieval and treated with 3% hydrogen peroxidase for 10 minutes for blocking endogenous peroxidase activity. Sections were incubated with blocking serum (goat origin, included in ABC staining kit) for 30 minutes and then incubated with primary antibody overnight at 4°C. ABC staining system (Santa-Cruz Biotechnology, Dallas, Texas) was used to develop the slides. Mouse anti-α-SMA (sc32251; Santa-Cruz Biotechnology), rabbit polyclonal anticollagen I (Rockland Immunochemicals, Gilbertsville, Pennsylvania), and rabbit anti-TIMP-1 (sc-5538; Santa-Cruz Biotechnology) antibodies were used, respectively, for different section staining. All primary antibodies were used by dilution of 1:500. For quantification of α-SMA and TIMP-1 staining, 8 fields (200×) in periportal areas were randomly picked and measured for each mouse.
Measurement of Liver Collagen
Quantitative analysis of collagen in Picrosirius Red-stained liver sections was performed by morphometric analysis. Briefly, 5-μm thick sections of paraffin embedded liver tissue were stained with Picrosirius Red for 1 hour (Sigma-Aldrich, St Louis, Missouri), then washed twice by acidified water. The whole section of liver was measured. Image-Pro Plus 6.0 software (Media Cybernetics, Rockville, Maryland) was utilized to calculate the percentage of collagen staining. Fibrosis deposition is expressed as a percentage of Picrosirius Red-stained area (red) on the total area of the section. To prevent potential variability in the intensity of tissue sections from different staining batches, all image acquisition parameters were fixed. The consistent threshold of the areas specifically stained with Picrosirius Red is applied through all the sections.
Reverse Transcriptase-Polymerase Chain Reaction
RNA was isolated with Trizol reagent (Invitrogen, Carlsbad, California). Total liver RNA was reverse transcribed using AMV reverse transcriptase (Takara, Otsu, Japan). The specific primer sequences for collagen αI(I), TIMP-1, and β-actin were as follows: collagen αI(I) (forward: ATGTTCAGCTTTGTGGACCTC and reverse: TCCCTCGA CTCCTACATCTTC), TIMP-1 (forward: TCTTGGTTCCCTGGCGTACT and reverse: GTGGCAGGCAAGCAAAGTG), and β-actin (forward: CTGGCCGG GACCTGACAGACTACC and reverse: (ATCGGAACCGCTCGTTGCCAATAG). The primer sequences were obtained from PrimerBank (URL: http://pga.mgh.harvard.edu/primerbank). The PCR reaction condition is as follows: denaturation, 95°C, 20 seconds; annealing, 58°C, 20 seconds; extension, 58°C, 20 seconds; 20 to 22 cycles. The messenger RNA (mRNA) expression of genes was analyzed and normalized to the mRNA of expression β-actin. The PCR products were separated by agarose gel electrophoresis. The gel was scanned using Tanon imaging workstation (Tanon, Shanghai, China) and the intensity of bands visualized by ethidium bromide staining was quantitated using Bio-Rad QuantityOne 4.6.2 (Bio-Rad, Hercules, California). The bands were defined by identical region of interest and calculated as integrated pixel intensity for all samples. The background was determined by measuring the average pixel intensity of a user-defined area.
Western Blot Analysis
Western blotting was carried out as described previously. 20 Briefly, frozen tissues were homogenized in modified RIPA buffer (50 mmol/L Tris-HCl [pH 7.5], 2 mmol/L EDTA, 150 mmol/L NaCl, 0.1% sodium dodecyl sulfate, and 1% NP-40) containing Protease Inhibitor Cocktail (1:100, P8340; Sigma-Aldrich) and phenylmethanesulfonyl fluoride (PMSF, 2 mmol/L) and separated on an 8% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to a nitrocellulose transfer membrane (Millipore, Billerica, Massachusetts). Membranes were incubated with primary antibodies raised against mouse α-SMA (sc32251; Santa-Cruz Biotechnology) or β-actin (sc-47778; Santa-Cruz Biotechnology) for 2 hours at room temperature or at 4°C over night, and then incubated with a fluorescein-conjugated secondary antibody for 30 minutes at room temperature. The membranes were scanned with an Odyssey Infrared Imaging System (LI-COR Biotechnology, Lincoln, Nebraska). The intensity of bands was quantitated by Bio-Rad QuantityOne 4.6.2. The bands were defined by identical region of interest and calculated as integrated pixel intensity for all samples. The background was determined by measuring the average pixel intensity of a user-defined area.
Statistical Analysis
All parameters determined in this study are presented as means ± standard deviations. Statistical analyses were performed using
Results
BPOZ2 Deficiency Reduces Liver Fibrosis After Chronic CCl4 Administration
After 6 doses of repeated CCl4 administration, liver fibrosis in the mice was observed through hematoxylin and eosin and Picrosirius Red staining (used for selective staining of collagen). The results reveal that there was obvious fibrous periportal and bridging in wild-type livers, while BPOZ2-deficient mice showed significantly reduced hepatic fibrosis compared to wild-type mice (Figure 1A and B). The degree of liver collagen deposition was significantly lower in BPOZ2-deficient mice than that of wild-type livers at the end of the 3-week period (Figure 1B and C).
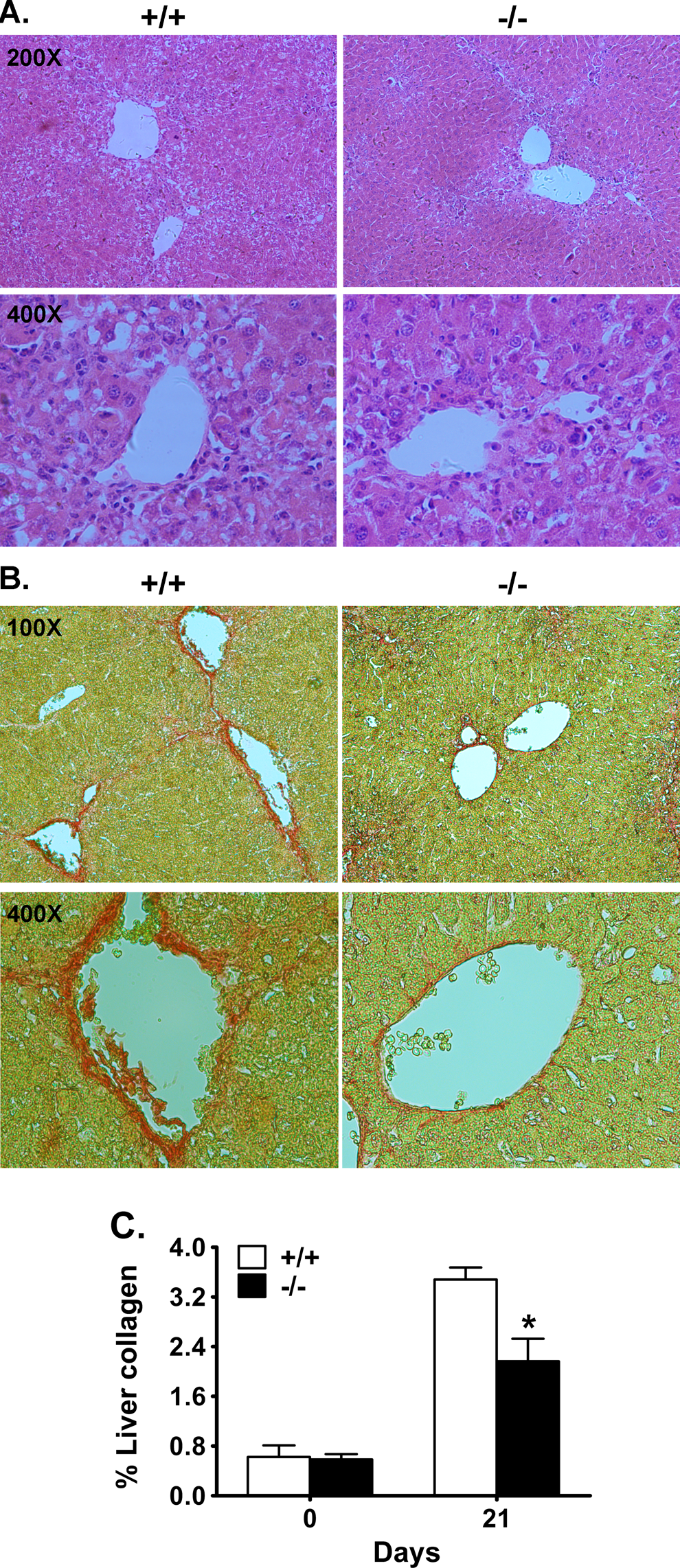
Reduced fibrosis in BPOZ2-deficient livers after chronic CCl4 administration. A and B, Decreased liver fibrosis in BPOZ2-deficient livers. Mice were subjected to 6 doses of CCl4 treatment within 21 days. At the end of the 3-week repeated CCl4 treatment period, liver samples were harvested, tissue sections were prepared and stained with hematoxylin and eosin (A) or Picrosirius Red (B). C, Quantitative analysis of collagen in Picrosirius Red-stained liver sections. Fibrosis deposition is expressed as a percentage of Picrosirius Red-stained area on the total area of the section. Values are the mean ± standard deviation (SD; n = 6-8). The amount of liver collagen in BPOZ2-deficient mice is significantly lower than that of wild-type littermates at the end of the 3-week repeated CCl4 treatment period. “*” indicates
Decreased Induction of Collagen αI(I) in BPOZ2-deficient Livers After CCl4 Administration
The expression of collagen αI(I) in the liver of the mice after CCl4 administration was evaluated by immunohistochemical staining and semiquantified reverse transcriptase PCR (RT-PCR) assay. The results show the expression of collagen αI(I) after CCl4 administration increased in both the liver of wild-type and that of BPOZ2-deficient mice, but the extent of collagen increment in BPOZ2 knockout mice was significantly lower than that in wild-type mice (Figure 2).
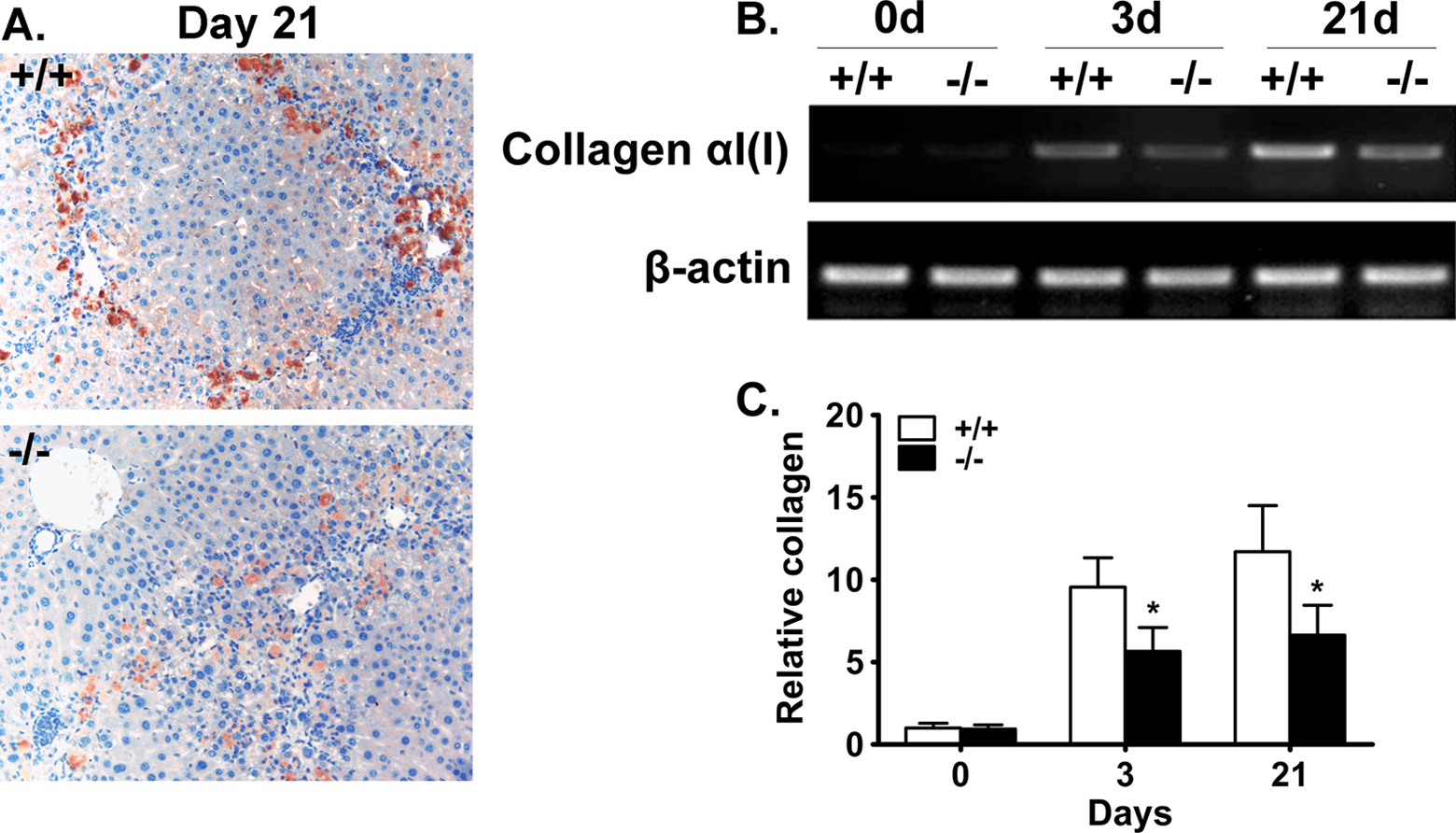
Decreased collagen αI(I) expression in BPOZ2-deficient liver. A, Representative photographs of collagen αI(I) expression in murine liver at the end of the 3-week repeated CCl4 treatment period by immunohistochemistry. Magnification: 200×. B, Representative photographs of the expression of collagen αI(I) in CCl4-treated murine liver by semiquantitative RT-PCR. C, Quantitative analysis of collagen αI(I) expression measured by semiquantitative RT-PCR. After densitometry of the relevant bands of the RT-PCR products, collagen αI(I) content was normalized for β-actin content. Data shown are the mean ± standard deviation (SD; n = 6 mice) expressed as relative to wild-type group at 0 hour. “*” indicates
BPOZ2 Deficiency Decreases Sustained Activation of HSCs After CCl4 Administration
We first detected the activated HSCs in the livers of wild-type and BPOZ2-deficient mice after CCl4 treatment by immunohistochemistry using α-SMA antibody. The results reveal that there were more activated HSCs in the injured areas in the livers of wild-type mice than those of BPOZ2-deficient mice at both 3 and 21 days after CCl4 treatment (Figure 3A and B). Meanwhile, we found that all the activated HSCs (α-SMA positive) were located in the injured areas in the livers of both wild-type and BPOZ2-deficient mice at different time points after CCl4 treatment (Figure 3A). We further investigated the expression of α-SMA (a well-known marker of activated HSCs) at different time points after CCl4 administration by Western blotting. The results show that HSC activation occurred following a single dose of CCl4. At 48 hours after acute CCl4 treatment, the expression of α-SMA dramatically increased in both wild-type and BPOZ2 null livers. However, at 72 hours post-CCl4, the amount of α-SMA in BPOZ2-deficient livers was markedly reduced, which remained elevated in wild-type livers (Figure 3C and D). At 21 days after 6 doses of repeated CCl4 treatment, the pattern of α-SMA expression in wild-type and BPOZ2-deficient mice was similar to that at 72 hours (Figure 3A). All these data suggest that absence of BPOZ2 has an impact on status of sustained activation of HSCs after CCl4 treatment.
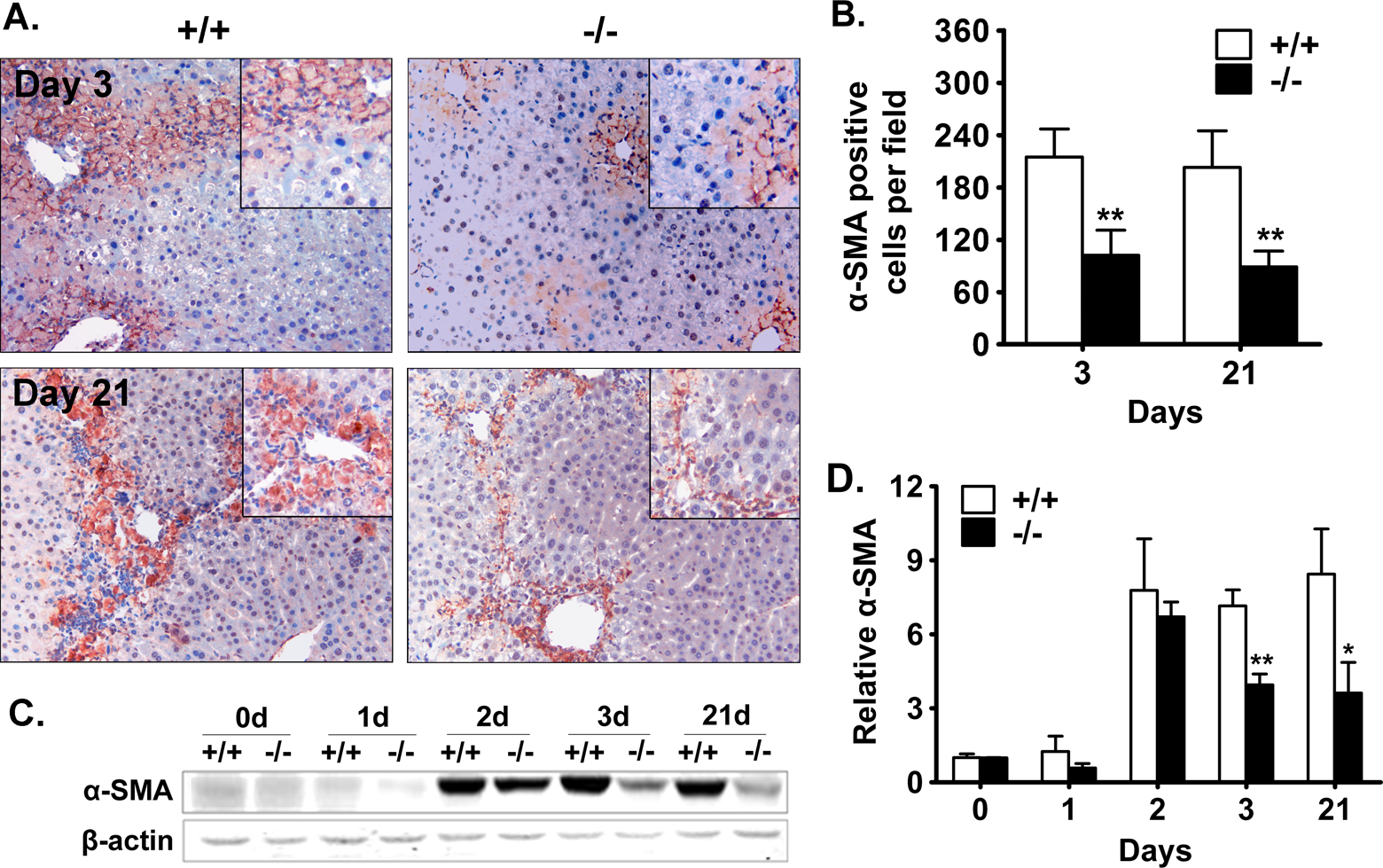
Decreased sustained activation of HSCs measured by α-SMA expression in BPOZ2 deficient murine liver after CCl4. A, Representative photographs of α-SMA expression in CCl4-treated murine liver by immunohistochemistry. Expression of α-SMA was detected in wild-type and BPOZ-2-deficient livers at 3 and 21 days after CCl4 treatment. Magnification: 200×. B, Quantitative analysis of α-SMA-positive HSCs in liver sections. Values are the mean ± SD (n = 6). C, Representative photographs of α-SMA expression in CCl4-treated murine liver by Western blot. D, Quantitative analysis of α-SMA expression measured by Western blot. After densitometry of the relevant bands, α-SMA content was normalized for β-actin content. Data shown are the mean ± SD (n = 6) expressed as relative to wild-type group at 0 hour. “*” indicates
BPOZ2 Deficiency Attenuates TIMP-1 Activation in the Fibrotic Liver
The expression of TIMP-1 in the fibrotic livers was evaluated using immunostaining and semiquantitative RT-PCR. The results showed that the expression of TIMP-1 significantly increased in both wild-type and BPOZ2 null livers (Figure 4A and B) after 3-week period of CCl4 administration. A similar pattern was observed by semiquantitative RT-PCR. Nevertheless, BPOZ2 deficiency significantly attenuated the upregulation of TIMP-1 transcription. There was approximately 38% reduction in hepatic TIMP-1 transcript in BPOZ2-deficient livers compared to wild-type livers after chronic CCl4 treatment for 21 days (Figure 4C and D).
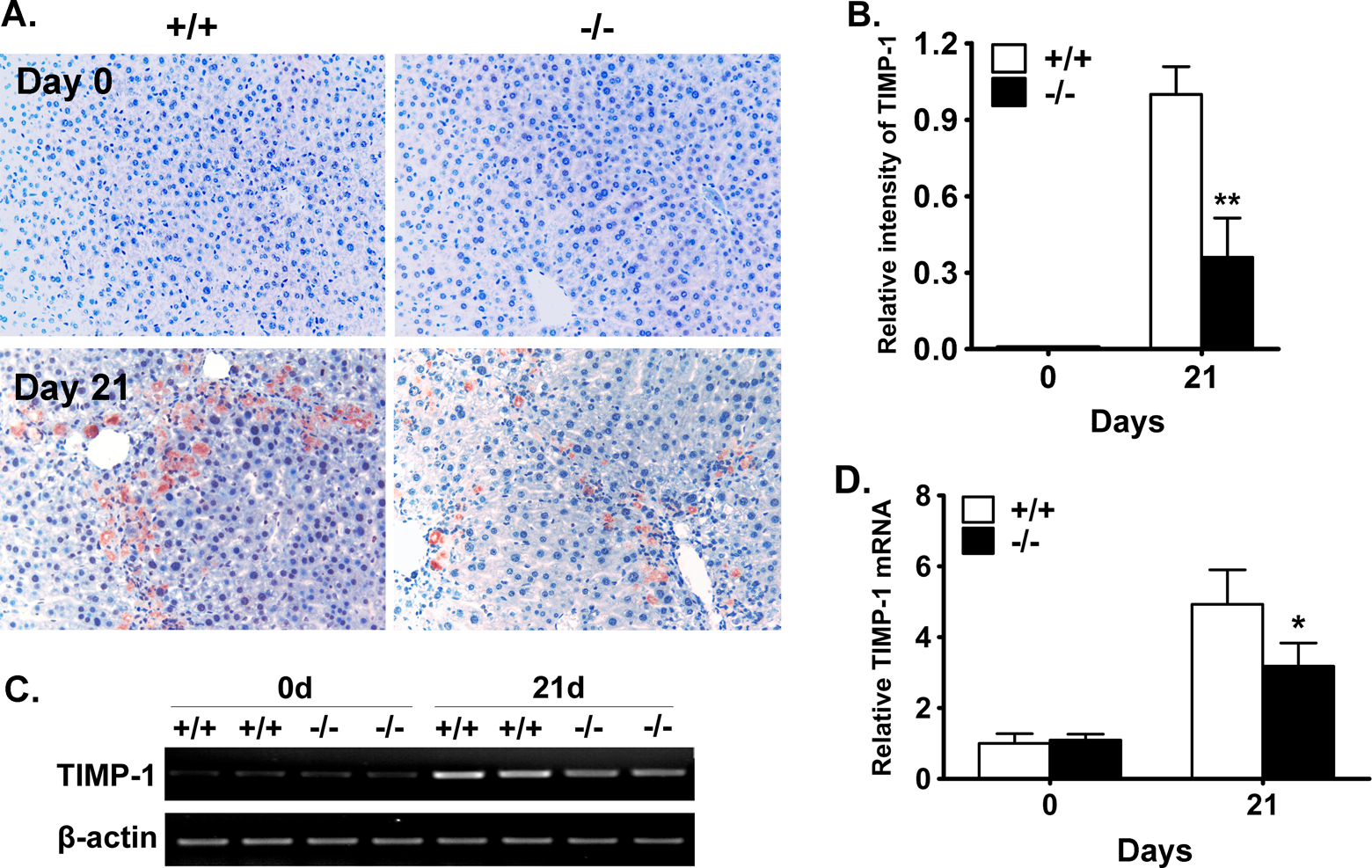
Attenuated TIMP-1 expression in BPOZ2-deficient liver. A, Representative photographs of TIMP-1 expression in CCl4-treated murine liver by immunohistochemistry. Expression of TIMP-1 was detected in wild-type and BPOZ-2-deficient livers before CCl4 treatment and 21 days after chronic CCl4 treatment. Magnification: 200×. B, Quantitative analysis of TIMP-1 amount in IHC-staining liver sections. Eight fields were calculated for each mouse. Data were normalized by the value of day 21. Values are the mean ± SD (n = 6). C, Representative photographs of TIMP-1 expression in CCl4-treated murine liver by semiquantitative RT-PCR. The TIMP-1 messenger RNA (mRNA) was significantly increased after CCl4 administration. The BPOZ2-deficiency decreased this upregulation. D, Quantitative analysis of TIMP-1 expression measured by semiquantitative RT-PCR. After densitometry of the relevant bands, TIMP-1 content was normalized to β-actin content. Data shown are the mean ± SD (n = 6 mice) expressed as relative to wild-type group at 0 hour. “*” indicates
Discussion
In this work, we investigated the effect of BPOZ2 deficiency on liver fibrosis in CCl4-induced mouse model. It is well known that liver fibrosis is characterized by excessive deposition of extracellular matrix in the liver such as collagen. 3,22 Here we show that BPOZ2 deficiency causes obvious reduction of liver fibrosis and collagen αI(I) expression after chronic toxic insult, indicating that BPOZ2 is involved in the development of liver fibrosis.
Hepatic fibrosis is a dynamic process resulting from liver tissue injury. It is an active, potentially reversible process originating from wound-healing responses to chronic liver injury. The principal effector of hepatic fibrogenesis is now widely recognized as HSC. 23 The HSCs are usually quiescent cells, but in response to liver injury they undergo an activation process in which they become highly proliferative and synthesize excessive fibrotic matrix, particular type I collagen. 8,9 Activation process of HSCs consists of 2 major phases, initiation and perpetuation. Initiation phase refers to early changes in gene expression and phenotype, rendering the cells responsive to other cytokines and stimuli, while perpetuation phase results from the effects of these stimuli on maintaining the activated phenotype and generating fibrogenesis. 2,24 On the other hand, clearance of activated HSCs, as the major mechanism of fibrosis regression, might counteract the process of fibrosis. During the process of fibrosis regression, activated HSCs are cleared mainly by apoptosis as well as returning to a quiescent phenotype. 25 In this study, we show that the amount of activated HSCs in BPOZ2 null livers significantly decreased compared to wild-type livers from 72 hours to 21 days during chronic CCl4 treatment indicated by α-SMA staining and collagen I mRNA expression. On the other hand, the distribution of activated HSCs has not been altered in BPOZ2 knockout mice compared to their wild-type counterparts during fibrosis progression. According to these findings, we conclude that BPOZ2 might be essential for the sustained activation of HSCs in vivo in the progress of chronic CCl4-induced liver fibrogenesis. Herein, BPOZ2 deficiency may have decreased liver fibrosis by facilitating the apoptosis or reversion to quiescence of activated HSCs in chronic liver injury.
The TIMP-1 plays a pivotal role in liver fibrogenesis by regulating matrix degradation. The TIMP-1 protein binds to and inhibits activated collagenases, subsequently protecting newly synthesized collagen from immediate degradation. 26 Overexpression of TIMP-1 in liver delays regression of liver fibrosis in experimental animals. 2,3 Conversely, using antibody or antisense oligonucleotides against TIMP-1 attenuates liver fibrosis by enhancing accumulating matrix degradation. 27,28 In this study, TIMP-1 mRNA and protein levels are shown to be significantly increased during liver fibrogenesis in wild-type liver, but less so in BPOZ2-deficient liver. It is possible that attenuated TIMP-1 upregulation is involved in the antifibrotic effects of BPOZ2 deficiency.
In conclusion, our study reveals that BPOZ2 knockout leads to a decrease in CCl4-induced liver fibrosis in mice. This is paralleled by a less sustained activation of HSCs and collagen αI(I) and TIMP-1 expression. These data indicate that BPOZ2 deficiency may provide protection against liver fibrosis.
Footnotes
Author Contributions
F. Zhang contributed to conception and design, acquisition, analysis, and interpretation, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. S. Dang contributed to conception, acquisition and analysis, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. R. Shu contributed to design, acquisition, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Y. Xiang contributed to conception and design, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Y. Kuang and J. Fei contributed to conception, critically revised the article, gave final approval, and agree to be accountable for all aspects of work ensuring integrity and accuracy. Z. Wang contributed to conception and design, drafted the article, critically revised the article, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Feng Zhang and Suying Dang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by National Natural Science Foundation of China (30370793 and 30530390), Ministry of Science and Technology of China (2011BAI15B02), the grants from Science and Technology Commission of Shanghai Municipality (10DZ2251500, 11DZ2292400, and 12ZR1421100), E-Institutes of Shanghai Municipal Education Commission (E03003), and the Doctoral Scientific Fund Project of the Ministry of Education of China (20110073110063).
