Abstract
Carbon tetrachloride (CCl4) is a widely used hepatotoxin for the studies of liver fibrosis and cirrhosis, and taurine has function to abate liver fibrosis induced by CCl4. But the interacting mechanisms between taurine and CCl4 in liver are still largely unknown. These made us to hypothesize that CCl4 may induce liver fibrosis by affecting the expressions of taurine biosynthetic enzymes and taurine synthesis. We thus assayed the expressions of hepatic cysteine dioxygenase (CDO), cysteine sulfonate acid decarboxylase (CSAD) and taurine transporter (TauT) in the progression of mouse liver fibrosis induced by CCl4. The results demonstrated that CCl4 treatment markedly decreased hepatic CSAD, CDO expressions, and taurine levels in hepatic tissue, although TauT expression did not exhibit significant decline. It was contrasting that hepatic α-SMA, serum AST, ALT, ALP kept increasing, which were accompanied by the pathological characters of liver, whereas taurine supplement attenuated the progression of liver fibrosis induced by CCl4. These results demonstrate that CCl4 may induce liver fibrosis by inhibiting hepatic CSAD and CDO expressions and taurine synthesis, which are crucial for our understanding the mechanisms of liver fibrosis induced by CCl4, and also potential for establishing therapeutic strategies of liver fibrosis and related diseases.
Introduction
Liver fibrosis is a wound healing response to various types of injury, which can progress into liver cirrhosis and/or hepatocellular carcinoma.1,2 Therefore, the considerable efforts have been devoted to elucidate the mechanisms driving liver fibrosis by using in vitro and in vivo models, among which carbon tetrachloride (CCl4) mouse model is included.3–5
Carbon tetrachloride is a widely used hepatotoxin in the study of liver fibrosis and cirrhosis in rodents. Hepatic biotransformation of CCl4 relies on cytochrome P450 2E1 into a very reactive trichloromethyl free radical (CCl3) inducing acute hepatic damage, in which several free radical reactions and lipid peroxidation processes are involved.4,6–9 The acute hepatic damage is characterized by the activation of Kupffer cells, the induction of an inflammatory response and excessive accumulation of extracellular matrix (ECM) proteins, such as smooth muscle actin alpha (α-SMA). 10 Whereas the damaged hepatocytes and activated Kupffer cells secrete reactive oxygen species and produce pro-inflammatory and profibrogenic cytokines, such as tumor necrosis factor-α, interleukins-6 and transforming growth factor-β1, all of which are involved in the development of liver fibrosis.11,12 The current potential therapeutic approaches have been focused on the elimination of liver injury mediators and reduction of inflammation by using the agents, such as taurine, to reduce oxidative stress and enhance matrix degradation.13,14 However, the further studies about the mechanisms of liver fibrosis are crucial for establishing the effective therapeutic strategies for liver fibrosis and the development of anti-fibrosis agents.
Taurine, a sulfur-containing amino acid, is a major free intracellular amino acid present in many tissues of human and animal, although it does not incorporate into proteins. The maintenance of body taurine level relies on diet uptake and the endogenous taurine synthesis. Liver is the major organ to synthesize taurine and regulate taurine homeostasis. 15 While taurine synthesis is through the sequential actions of taurine synthetases, including cysteine dioxygenase (CDO) and cysteine sulfinate acid decarboxylase (CSAD), which are highly expressed in liver, 16 although taurine is not considered to be an essential amino acid for most mammals.13,17 However, the biosynthetic capacity and turnover rate of taurine vary greatly in mammals, and mouse has higher capacity of synthesizing taurine.18,19 The functions of taurine include bile acid conjugation, neurotransmission, detoxification of certain xenobiotics, membrane stabilization, calcium homeostasis, osmoregulation and anti-oxidative stress.20–22 Taurine performs its hepatoprotective effects by reducing oxidative damage, preventing fibrosis and hepatocyte apoptosis induced by CCl4.23–26 But it still remains unclear about the protective mechanism of taurine against hepatic damage.
In addition, taurine levels both in the serum and hepatic tissue decrease after suffering from liver diseases and the reduced taurine content in taurine transporter (TauT) knockout liver hepatocytes leads to liver fibrosis.15,27 However, exogenous taurine supplementation can prevent liver injury, inhibit ECM deposition in the damaged liver and prevent liver fibrosis caused by different harmful substances, including CCl4.14,27,28 These made us to ask whether CCl4 affects the biosynthetic processes of taurine by inhibiting the expressions of hepatic CDO, CSAD and taurine uptake by TauT, which subsequently cause the taurine depletion and liver damage. The present study was thus proposed to study the relationships among liver damage induced by CCl4 and hepatic CDO, CSAD, TauT expressions and taurine synthesis in the progress of mouse liver fibrosis, and value the rescuing effects of taurine supplement on progression of liver fibrosis induced by CCl4.
Materials and methods
Reagents and antibodies
CCl4, taurine, 2-hydroxy-1-ethanethiol, methanol, glutamine, diaminobenzidine (DAB) were purchased from Sigma-Aldrich Chemical Company (Sigma, St. Louis, MO, USA). Antibodies included: Glyceraldehyde-3-phosphate dehydrogenase (GAPDH, 1:20000, ab8245, Abcam), CSAD (1:10000, ab91016, Abcam), TauT (1:5000, ab236898, Abcam), CDO (1:10000, ab53436, Abcam), α-SMA (1:200 for IHC and 1:5000 for Western blotting, sc-53142, Santa Cruz Biotechnology), horseradish peroxidase-conjugated secondary antibody (1:20000, Zhongshan Bio Corp.). Other chemicals were purchased from Sinopharm Chemical Reagent Co., Ltd (Beijing, PR China).
Animals and treatments
Institutional Animal Care and Use Committee (IACUC) at the Yangzhou University approved the experimental protocol of this study (No. 202103203). Healthy adult male C57BL/6 mice, weighted 25–30 g, were obtained from animal house facility at Yangzhou University. All mice were housed at 24 ± 2°C, relative humidity of 55 ± 15%, fed with rodent chow and clean water under 12 h/12 h light–dark cycle. They were allowed 1-week acclimatization period before the initiation of the experiment. Total 90 mice were used and randomly divided into 3 group (30 mice per group). The first group were used to identify the effects of CCl4 on the expressions of CSAD, CDO, TauT and taurine synthesis in the development of live fibrosis. Mice received intraperitoneal administration of CCl4 (0.5 mL/kg) in olive oil (1:3, vol/vol) twice a week. Although 1–2 mL/kg body weight of CCl4 was used to induce acute hepatitis, 24 a lower dose of CCl4 was administrated for a long time to learn its hepatotoxicity to avoid mice death. The second group were used to value whether taurine supplement has function to alleviate the progression of liver fibrosis induced by CCl4. The mice were supplied with taurine by providing drinking water containing 2% taurine and CCl4 were injected as above (CCl4+Taurine). The third group received equal olive oil (controls). After 0,2, 4, 6 and 8 weeks CCl4 treatments (6 mice for each treatment and per time point), the mice were anesthetized by intraperitoneal injection of 50 mg/kg Zoletil 50 (Virbac, Carros, France) and euthanized by exsanguination. The serum and liver samples were collected and stored in −80°C for biochemical parameters measurement. Additionally, some liver samples were fixed in neutral-buffered formalin for histopathological examination.
Determination of taurine by HPLC-UV
Taurine contents in liver and serum samples were measured by HPLC-UV. Firstly, samples were weighed, homogenized and deproteinized by 0.2 M sulfosalicylic acid. After being centrifuged at 14000 g for 20 min, supernatants were added into a dual-bed column containing cation and anion exchange resins to remove other amino acids and metabolic precursors of taurine. Secondly, 100 μM glutamine were added into samples as an internal standard. Then all samples were filtrated through a 0.22 μM polyvinylidene fluoride (PVDF) membrane and saved in −80°C freezer until use. The samples and the standard samples of taurine which were 100, 50, 25, 10, 5, 2 and 1 μM were derivate with phthaldialdehyde (OPA, Sigma) solution (20 mg OPA, 2 mL methanol, 80 μL 2-hydroxy-1-etanethiol, 18 mL 0.1 M borate buffer (pH 9.6)). 3 min later, 20 μL of sample was automatically injected into a six-port valve to analysis with 1 min delay. The HPLC conditions were: flow A: 100% methanol, flow B: sodium phosphate buffer pH 4.7 containing 50% methanol. Flow rate was 1.2 mL/min, and the detection wavelength was 340 nm. The duration times were 2.3 min and 4.95 min for the internal standard and taurine. The separation column was 150 mm × 5 mm, 4.6 μM, Waters® Symmetry C18.
Histologic analysis and α-SMA immunohistochemistry (IHC)
For histologic analysis, sections were taken from the anterior portion of the left lateral lobe of the liver. The tissues were then immersed in 10% neutral-buffered formalin, followed by paraffin embedding. Hematoxylin-eosin staining was performed on sections of approximately 5-μm thickness. Histologic changes were evaluated. For α-SMA IHC staining, the sections were dewaxed and rehydrated, and then the antigen retrieval was performed by microwaving for 20 min in 0.01 M sodium citrate buffer (pH 6.0). After washing with PBS (pH 7.2) for 30 min, non-specific endogenous peroxidase activity was blocked by 3% (vol/vol) H2O2 diluted in PBS. Non-specific binding sites were then blocked by 10% normal goat serum diluted in PBS at least for 3 h at room temperature (RT). The sections were then incubated with α-SMA antibody (1:200) diluted in PBS overnight at 4°C. After washing with PBS for 30 min, the sections were incubated with biotinylated goat anti-rabbit/mouse IgG (Zymed laboratories, 1:200) for 3 h at RT. After washing with PBS for 30 min, the sections were incubated with streptavidin peroxidase complex (Zymed laboratories, 1:200)) for 30 min at RT. Finally, the signals were visualized by incubating the sections with 0.05 mol/L Tris-HCL (pH 6.5) containing 0.06% (wt/vol) diaminobenzidine (DAB) and 0.03% (vol/vol) H2O2. In addition, α-SMA was used to quantify fibrosis using Image J software (NIH, Bethesda, MD, USA).
Western blotting
Samples were homogenized and centrifuged. The total proteins content was determined by a BCA Protein Assay Kit (Beyotime, Nanjing, PR China). 50 μg total proteins were resolved by 12% or 15% SDS-polyacrylamide gels (wt/vol) and transferred onto a PVDF membrane (Millipore, Billerica, MA, USA). The non-specific binding sites were blocked by 5% (wt/vol) non-fat milk diluted in tween/tris-buffered saline (TBST) for at least 2 h at RT. The membranes were then incubated with primary antibodies (CSAD 1:10000, CDO 1:10000, GAPDH 1:20000, α-SMA 1:5000, TauT 1:5000) diluted in TBST buffer overnight at 4°C. After washing for 30 min with 3 changes of TBST, the membranes were incubated with horseradish peroxidase-conjugated secondary antibody (1:20000) diluted in TBST for 2 h at RT. The signals were detected using Pierce ECL Kit (Pierce, Rockford, IL, USA) and visualized by Tannon gel imager (Tanon, Shanghai, PR China). The intensity values pertaining to each sample were normalized against the density of GAPDH.
RNA extraction and analysis
Total RNA was extracted from samples by RNAiso Plus (Takara, Dalian, PR China) and 2 μg of total RNA from samples were reverse transcribed using M-MLV reverse transcriptase (Promega, Madison, WI, USA). Primers used for real-time quantitative PCR (RT-qPCR) assay were purchased from Sangon (Shanghai, PR China), and the primer sequences were as follows: Cysteine dioxygenase (
Serum ALT, AST and ALP activities
The serum alanine amino-transferase (ALT), aspartate aminotransferase (AST) and Alkaline phosphatase (ALP) activities were respectively determined by commercial colorimetric assay kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, PR China).
Statistical analysis
Data were analyzed by one-way ANOVA for single factor independent samples and by two-way NVOVA for two factor independent samples. All values are expressed as means ± SEM.
Results
Effects of CCl4 on hepatic CDO, CSAD, TauT expressions and taurine synthesis
In order to identify the relations of hepatic CDO, CSAD, Taut expressions and taurine synthesis to the progression of liver fibrosis induced by CCl4, the mRNA and protein levels of hepatic CDO, CSAD and TauT were assayed by RT-qPCR and Western blotting. Taurine levels in hepatic tissues and serum were respectively measured using HPLC-UV after 2, 4,6 and 8 weeks of CCl4 treatments. The results showed that Effects of CCl4 on the expressions of hepatic CSAD, CDO, TauT, a-SMA and taurine levels in serum and hepatic tissues. (a-c), Effect of CCl4 treatment on hepatic 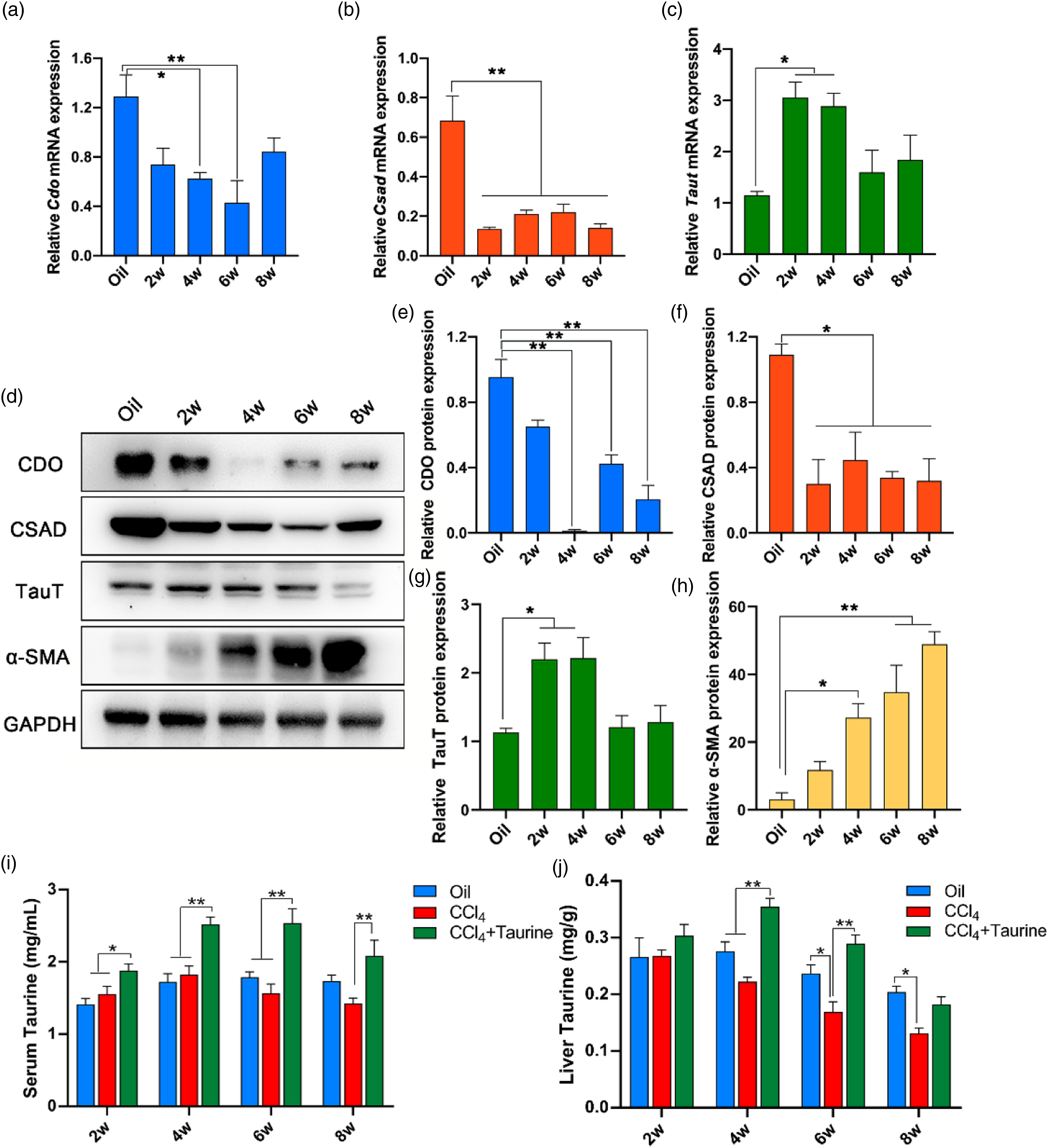
The progression of liver fibrosis induced by CCl4 is closely related to the decline of the hepatic CSAD expression and taurine concentration
In order to relate the changes of CSAD, CDO expressions and hepatic taurine levels to the progress of liver fibrosis induced by CCl4, α-SMA, an important extracellular matrix marker of liver fibrosis, was examined by Western blotting and IHC. The Western blotting results showed that α-SMA expression kept increasing in the progression of liver fibrosis, which reached the maximum at 8 weeks after CCl4 treatment (Figure 1(h)). However, α-SMA staining was totally absent at 2 weeks of CCl4 treatment (data not shown), which were similar to the controls (Figure 2(a), (e) and (i)), although weak staining was detected at 4 weeks (Figure 2(b)). By 6 weeks, intensive α-SMA positive stellate cells were observed (Figure 2(f)), the intensity of the staining is much higher and the number of positive stained stellate cells in the fibrotic area increased by 8 weeks after CCl4 treatment (Figure 2(j)). In addition, the quantified staining of α-SMA sharply increased from 4 to 8 weeks after CCl4 treatment (Figure 2(b), (d), (f), (h), (j), (l)). Effects of CCl4 and combined administration with taurine (CCl4+Taurine) on α-SMA expressions detected by IHC staining of liver sections. (a), (e) and (i): Controls; (b), (f), (j): IHC staining of α-SMA after 4, 6 and 8 weeks CCl4 treatments; (c), (g) and (k): IHC staining of α-SMA after 4, 6 and 8 weeks treatments by CCl4 and taurine (CCl4+Taurine); (d), (h) and (l): Quantitative analysis of α-SMA staining by Image J software. Data are presented as means ± SEM, 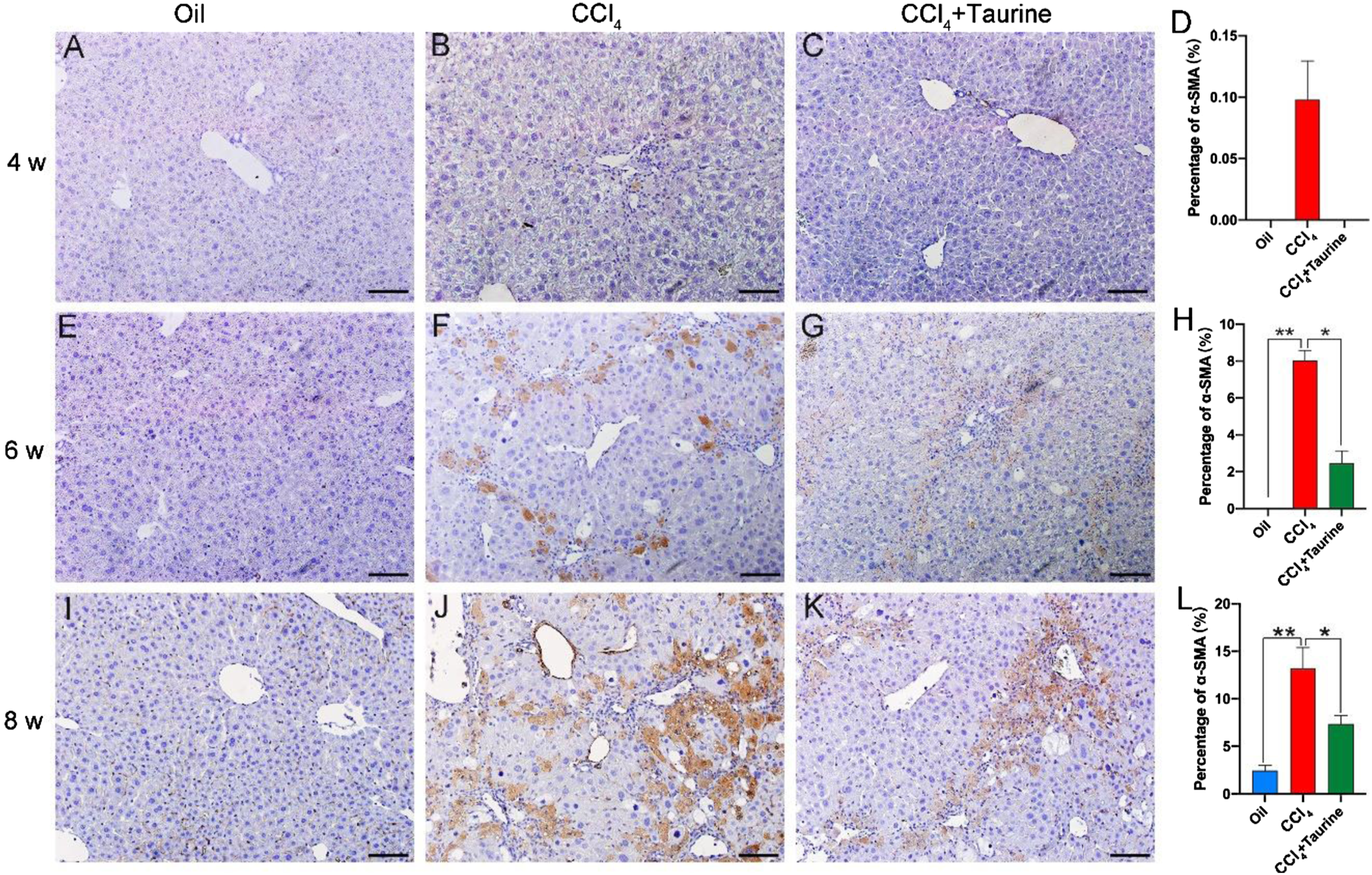
Furthermore, histopathological examination was performed and the results showed that mouse liver exhibited normal lobular architecture with hepatocytes arranged in hepatic cords radiating from a central vein and separated by obvious blood sinusoids in controls (Figure 3(a)–(d)). However, 2 and 4 weeks CCl4 treatments caused notable liver lesions including deformed cord arrangement, ballooning degeneration of hepatocytes, condensed nuclei, widespread inflammatory cell infiltration (Figures 3(e) and (f)). After 6 and 8 weeks CCl4 treatments, much severer hepatic damage, including widespread hepatic stellate cells and hepatocellular necrosis, were observed. (Figures 3(g) and (h)). Microscopic pictures of hematoxylin-eosin (HE) stained liver sections. (a), (b), (c) and (d): Control groups; (e), (f), (g) and (h): CCl4 treated groups; (i), (j), (k) and (l): CCl4+Taurine treated groups. w: weeks. Red arrows: inflammatory infiltrates, blue arrows: nuclear swelling, green arrows: nuclear enrichment, yellow arrows: hepatic fibrosis. Bars = 50 μm.
In addition, the hepatic damage was evaluated by measuring serum ALT, AST and ALP at 4, 6 and 8 weeks of CCl4 treatments (Figure 4(a), (b) and (c)), all of which sharply increased at every stage examined compared with controls, and there was no significant difference among 4, 6 and 8 weeks after CCl4 treatments. These collective data demonstrate that the progression of liver fibrosis induced by CCl4 is closely related to the decline of hepatic expressions of taurine biosynthetic enzymes and taurine synthesis in mouse liver. Effects of CCl4 and combined administration of CCl4 with taurine (CCl4+Taurine) on serum ALT, AST and ALP levels. (a), (b) and (c): Serum ALT, AST and ALP contents. Data are presented as means ± SEM, 
Taurine supplement attenuates the progression of liver fibrosis induced by CCl4
In order to identify whether the liver fibrosis induced by CCl4 could be attenuated by taurine, 0.2% (w/v) taurine was supplied to the mice treated with CCl4 by drinking water (CCl4+Taurine) throughout the experiment. After 2, 4, 6 and 8 weeks CCl4+taurine treatments, α-SMA expression, hepatic histology, serum ALT, AST and ALP were respectively examined. The results demonstrated that the intensity of α-SMA IHC staining got much weaker at 4, 6 and 8 weeks of CCl4+taurine treatment than CCl4 treatment alone (Figure 2(c), (g) and (k)), which were corresponding to the quantified results of α-SMA staining (Figure 2(d), (h) and (l)). The results of histological examination showed that taurine supplement markedly lessened the destruction in lobule structure, inflammatory cell infiltration and hepatocellular necrosis (Figure 3(i)–(l)). In addition, serum ALT, AST and ALP levels did not have significant differences with the controls at every stage examined (Figure 4(a)–(c)). These demonstrate that taurine supplement attenuates the progression of the liver fibrosis induced by CCl4.
Discussion
It is well known that taurine plays its hepatoprotective effects in CCl4-induced liver injury by reducing oxidative damage, preventing fibrosis and hepatocyte apoptosis.13,14,17,22,29 But the interacting mechanisms between taurine and CCl4 in liver are still largely unknown. The present study demonstrates that CCl4 induces liver fibrosis by disrupting the expressions of hepatic CDO/CSAD and taurine synthesis. The results are crucial for our understanding the mechanism of liver fibrosis induced by CCl4 and potential for the choice of the drug target to prevent and alleviate the occurrence of liver fibrosis.
Although it has been reported that the acute CCl4 treatment affects hepatic CSAD and CDO expressions,7,30 the results of present study firstly demonstrate the CSAD and CDO expressions and endogenous taurine synthesis were inhibited during the progression of liver fibrosis induced by CCl4, and suggest that the development of liver fibrosis induced by CCl4 is at least partly resulted from the taurine depletion caused by the decline of CSAD, CDO expressions in liver. In support, the decline of hepatic CSAD expression is accompanied with the increased α-SMA expression, serum AST, ALT, ALP concentration, and the histological destruction. However, there are reports that the hepatotoxicity of CCl4 is attributed to the reactive oxygen species and free radicals produced during its metabolism31,32 which subsequently lead chemical liver damage involving the aggravation of inflammatory processes and recruitment of inflammatory cells, ultimately result in hepatic fibrosis.4,33,34
In addition, the results presented here demonstrate that the hepatic taurine concentration decreases from 4 weeks after CCl4 administration, accompanied with the declines of the CSAD and CDO expressions from 2 to 8 weeks. This may be related to the general adaptation mechanism in response to liver injury at earlier stage of the CCl4 insult. In addition, the remarkable decreases of hepatic CSAD, CDO expressions and taurine level are closely related to the progression of hepatopathy, including the increased serum ALT, AST, ALP levels and hepatic α-SMA expression, which are in accordance with the reported studies.29,35,36 Therefore, the inhibiting effects of CCl4 on CDO, CSAD expressions and taurine synthesis are important mechanism causing liver injury and occurrence of liver fibrosis. This is supported by the results of the present study that taurine supplement reduced the loss of hepatic taurine concentration concomitant with reduced liver injury observed both histologically and biochemically, although the rescuing effects of the taurine supplement on CSAD, CDO expressions were not detected in this study.
Both CSAD and CDO are two key enzymes involved in the biosynthetic process of taurine, but the effect of CCl4 on hepatic CDO expression was not parallel to the CSAD expression and taurine level in the progression of liver fibrosis. Compared with CSAD, CDO is an upstream enzyme responsible for the transfer of cysteine to cysteine sulfinate, which is then converted to taurine by CSAD. 20 However, it is still needed to be elucidated about the signaling pathway and related molecular mechanism of CCl4 affecting the expressions of CSAD and CDO, and taurine synthesis in the future study.
However, it is out of our expectation that CCl4 did not have significant influence on the global taurine level, and this may be account for that the serum taurine is not only correlated with the taurine de novo synthesis in liver, but also interrelated with the taurine transport from other organs. In addition, liver is the largest organ to synthesize taurine, and also plays important roles in regulating taurine metabolism and homeostasis. 20 Furthermore, taurine and glutathione are two metabolic products of cysteine and methionine, and both of them are mainly synthesized in liver, 37 while the relations of the liver taurine and glutathione to global taurine level need to be elucidated in the future study.
In conclusion, the results of the present study firstly demonstrate that CCl4 induces liver fibrosis by inhibiting the expressions of hepatic taurine biosynthetic enzymes and taurine synthesis, and the taurine deficiency resulted from CCl4 administration is an etiology of liver fibrosis in mouse. While taurine supplement attenuates the toxic effects of CCl4 on liver. These results are crucial for understanding the mechanisms of liver fibrosis induced by CCl4, and also potential for establishing therapeutic strategies of liver fibrosis and related diseases, although the related molecular mechanisms need to be investigated further.
Footnotes
Author contribution
Di Zhang and Sheng Cui conceived and designed the research; Di Zhang, Jiaming Zheng, Guobin Qiu, Tongjuan Niu and Yuneng Gong performed experiments; Di Zhang and Jiaming Zheng analyzed results and processed data; Di Zhang, Jiaming Zheng and Guobin Qiu prepared figures; Di Zhang and Sheng Cui interpreted results of experiments and drafted the manuscript.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
Informed consent
All the authors have read and approved the final submission of this study.
Data availability
The data that support the findings of this study are available from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
