Abstract
Hepatic fibrosis is a leading cause of morbidity and mortality worldwide. Attenuation of fibrogenic process can significantly lower the mortality rate. However, pharmaceutical intervention at fibrogenesis stage remains a major task in medicine. So there is a need for a natural compound to treat hepatic fibrosis. This study was outlined to investigate the anti-fibrotic effect of β-amyrin in dimethylnitrosamine (DMN)-induced hepatic fibrosis male rats. Serum liver function markers (aspartate transaminase, alanine transaminase, alkaline phosphatase and lactate dehydrogenase), oxidative stress markers (malondialdehyde, superoxide dismutase, catalase, glutathione peroxidase, glutathione reduced content and vitamin C), tissue inflammatory marker (tumor necrosis factor α (TNF-α)), apoptosis marker (caspase 3) and fibrolytic marker (tissue inhibitor of metalloproteinase 1 (TIMP-1)) were evaluated before and after β-amyrin treatment in DMN-induced rat. β-Amyrin treatment attenuated the altered levels of the serum enzyme markers produced by DMN and caused a subsequent recovery toward normalization. Oxidative stress markers and TNF-α levels were reduced significantly (p < 0.001) as well as proteins’ (caspase-3 and TIMP-1) expression was reduced in β-amyrin–treated DMN rats. By virtue of β-amyrin properties of inhibiting oxidative stress, apoptosis, inflammation, and fibrogenesis, it might act as an ideal anti-inflammatory and anti-fibrogenic agent to halt the progression of liver fibrosis to chronicity.
Introduction
Triterpenoid derived from plants used for many therapeutic purposes in Asian countries have various pharmacological properties. β-Amyrin (3β-hydroxy-olean-12-en-3-ol) is the major pentacyclic triterpenoid compound found in medicinal plants and oleoresin obtained by bark incision of several species of Bursera or Protium of the Burseraceae family. Both in vitro and in vivo studies have proven the biological role of β-amyrin including antimicrobial, antidepressant, anti-inflammatory, antinociceptive, and gastroprotective activities. 1 –3 The mixture of triterpenoid like ursane and oleanane, which have chemical resemblance of β-amyrin possess hepatoprotection against a variety of hepatotoxicants. 4
Liver fibrosis is a leading cause of morbidity and mortality worldwide. More than 50% of all patients with complicated liver cirrhosis die within the first 17 years. In more than 90% of all cases, cirrhotic liver leads to hepatocellular carcinoma. Therefore, attenuation of the fibrogenic process can significantly lower the morbidity. 5
Early stage of fibrogenesis is a chronic inflammatory condition. It occurs due to the activation of immunocompetent cells (e.g. Kupffer cells) and the release of proinflammatory cytokines, such as tumor necrosis factor α (TNF-α), interleukin (IL)-6 and IL-12. These mediators and accumulation of potentially toxic free fatty acids cause high reactive oxygen species (ROS) production that exposes hepatocyte to an oxidative stress, primarily via peroxidation of membrane lipids and DNA damage, which leads to hepatocyte apoptosis. 6
Hepatocyte apoptosis results in the formation of apoptotic bodies and other cellular debris, which are phagocytosed by hepatic stellate cells (HSCs), resulting in their activation, proliferation, differentiation and matrix deposition. HSCs’ activation represents a critical event in fibrosis because these cells become the primary source of extracellular matrix (ECM) in liver injury. 7
Matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinase (TIMP) play an important role in inducing and preventing the degradation of the ECM, respectively. 8 Many studies have shown that MMPs and TIMPs play a pivotal role in matrix remodeling during hepatic injury and repair. 9,10 Among them, TIMP-1 is a widely expressed and secreted protein that plays a critical role in tissue remodeling via inhibiting members of a large family of MMPs. 11 The profibrogenic effects of TIMP-1 are thought to be mediated via preventing collagen degradation through inhibition of MMPs and protecting against activated HSC death. 12 It is believed that activated HSCs and Kupfer cells are the major sources for TIMP-1 production during liver injury. 13 Upregulation of collagen synthesis during activation is among the most striking molecular responses of HSCs to fibrosis. TIMP-1 pathway is a potential target for therapeutic intervention of fibrotic liver diseases. 14
Dimethylnitrosamine (DMN) is a potent hepatotoxin, carcinogen and mutagen, which is used to induce liver fibrosis in rat. Exposure of rat to low doses of DMN leads to nodular regeneration, portal hypertension and fibrosis. DMN is a well-established model of liver fibrosis with a pathophysiology closely resembling that of cirrhosis in man and therefore widely used for screening anti-fibrotic agent. 15
As chronic liver diseases involve multiple risks, such as portal hypertension, ascites, hepatic encephalopathy and, of course, HCC, pharmaceutical intervention at the stage of fibrogenesis remains a major task in medicine. The development of drugs based on natural compounds is necessary to treat liver fibrosis. Thus, the present study was designed to establish the hepatoprotective potential of β-amyrin in DMN-intoxicated rats as an animal model. Here, we aimed to demonstrate the effect of β-amyrin on oxidative stress markers, inflammatory marker, apoptotic marker and TIMP-1 in DMN-induced liver fibrosis male rats.
Materials and methods
Animals
Twenty-four Wistar strain male albino rats weighing 180–200 g were used in this study. The animals were housed individually in polypropylene cages under hygienic, standard environmental conditions (28 ± 2°C, humidity 60–70%, 12-h light/12-h dark cycle). They were provided with nutritionally adequate standard laboratory diet and water ad libitum. They were acclimatized to the environment for 1 week prior to experimental use. The study was duly approved by the Institutional Animal Ethics Committee.
Dosage fixation/chronic toxicity
β-Amyrin was purchased from Sigma-Aldrich (St Louis, Missouri, USA). β-Amyrin was administered to the trial groups in graded doses ranging up to 50 mg/kg body weight/day and the rats were observed for any signs of mortality and behavioral infirmities for 4 weeks afterwards. The median lethal dose was then calculated based on the mortality and behavioral of rats. The minimum dose level of β-amyrin, viz. 30 mg/kg body weight/day, was used for oral administration to rats during experiment.
Experimental protocol
Rats were divided into four groups of six animals in each group. Group I served as normal control. Group II served as positive control in which normal rats were administered orally a dose of β-amyrin (30 mg/kg) for 4 weeks. Group III were intoxicated with DMN (Sigma, St. Louis) in saline (10μl/kg, intraperitoneal) on three consecutive days per week for 4 weeks and group IV DMN rats were treated orally with β-amyrin (30 mg/kg) every day for 4 weeks. After 4 weeks, all rats were killed and the liver tissues were sectioned and fixed in phosphate-buffered 10% formaldehyde for histological analysis. The remaining portions and serum were collected for biochemical and molecular analysis.
Transmission electron microscopy
Specimens were fixed in 2.5% glutaraldehyde for 24 h, subsequently washed in phosphate buffer (pH 7.4), post-fixed in 1% osmium tetroxide in phosphate buffer (pH 7.4) and dehydrated in increasing concentrations of alcohol. Then, the tissues were washed with propylene oxide and embedded in epoxy resin-embedding media. Semi-thin sections of about 3 mm thick were cut with a glass knife on a LKB Nova Ultramicrotome (Sweden). These sections were stained with methylene blue and examined by a Nikon Optiphot light microscope (Japan). Ultrathin sections were collected on copper grids, stained with uranyl acetate and lead citrate and then examined under a Jeol JEM 1200 Ex transmission electron microscope (Japan).
Assessment of liver function
The functional state of the liver was determined by estimating the biochemical parameters such as AST and alanine transaminase (ALT), 16 alkaline phosphatase (ALP) and lactate dehydrogenase (LDH) 17 spectrophotometrically.
Lipid peroxidation and antioxidant enzyme assay
Lipid peroxidation assay was determined by Beuge and Aust method 18 and antioxidant enzyme activities such as superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), glutathione reduced content (GSH) and vitamin C level. 19 –23 All analyses were performed spectrophotometrically.
Determination of TNF-α level by enzyme-linked immunosorbent assay
Serum TNF-α was measured using TNF-α rat enzyme-linked immunosorbent assay kit (catalogue no: ab46070; Abcam, UK) according to manufacturer’s instructions.
Determination of caspase-3 by Western blot
Liver tissues were washed with phosphate-buffered saline (PBS) and homogenized using 200 µl ice-cold radioimmunoprecipitation assay (RIPA) lysis buffer supplemented with protease inhibitor cocktail and protein concentration was measured by the bicinchoninic acid protein assay kit. The caspase 3 expression was determined using anti-caspase 3 (1:1000 dilution; Santa Cruz Biotechnology, California, USA). 24 Band intensity was analyzed by using ImageJ software (http://rsb.info.nih.gov/ij/).
Immunofluorescence assay of caspase 3
Formaldehyde-fixed liver tissue samples were embedded in paraffin and thin sections of 5 µm thickness were cut and stained with hematoxylin–eosin (HE). The thin sections were made into permanent slides for immunofluorescence assay. The immunofluorescence assay protocol described by Johansson et al. 1998 25 was followed using anti-caspase 3.
RNA isolation, cDNA synthesis and reverse transcription PCR for caspase 3 messenger RNA
Total RNA was extracted from the liver of control and experimental rats using Trizol (Invitrogen, USA). Complementary DNA (cDNA) were synthesized using a Rever Tra Ace qPCR RT kit (Toyobo Co., Osaka, Japan) according to the manufacturer’s instructions. The resulting cDNA was subjected to polymerase chain reaction (PCR) using a TaqDNA polymerase kit (QIAGEN, Hilden, Germany). PCR was performed in a 25 µl reaction mixture and consisted of one cycle of 70 s at 95°C, 25 cycles of 55 s at 93°C, 45 s at 61°C and 40 s at 72°C, and one cycle of 100 s at 72°C, according to the manufacturer’s instructions for the thermal cycler (Gene Amp PCR System 2700, Applied Biosystems, Foster City, California, USA). The primers (Invitrogen, Carlsbad, California, USA) used were as follows: caspase 3 (243 bp; forward 5′-F: TGGTTCATCCAGTCGCTTTG-3′ and reverse 5′-CATTCTGTTGCCACCTTTCG-3′) and β-actin (349 bp; forward 5′-GCAGAAGGAGATTACTGCCCT-3′ and reverse 5′-GCTGATCCACATCTGCTGGAA-3′). The β-actin primer was used as an internal control. PCR products were separated by electrophoresis in 2% agarose gels containing ethidium bromide. Digital images were captured by LAS-3000 (Fujifilm, Tokyo, Japan) and Multi-Gauge v3.0 software (Fujifilm). Bands corresponding to caspase 3 of the different groups were analyzed densitometrically with respect to the normal control group using ImageJ software. Data were normalized to the corresponding internal control β-actin mRNA bands.
Determination of TIMP-1 expression in HSCs by Western blot
The liver tissues were washed with PBS, and HSCs were isolated as described previously. 26 HSCs were homogenized using 200 µl ice-cold RIPA lysis buffer supplemented with the protease inhibitor cocktail. The blotting procedure was followed as described above using the primary antibody anti-TIMP-1 (1:2000 dilutions) (Santa Cruz Biotechnology).
Statistical analysis
All values were expressed as mean ± SEM. Statistically significant differences between mean values were determined by one-way analysis of variance followed by Tukey’s test for multiple comparisons. p < 0.05 was considered to be significant.
Results
Chronic toxicity
There were no visible signs of toxicity and death in animals treated with β-amyrin at doses of 10 and 50 mg/kg for 4 weeks. The animals that received various doses of β-amyrin did not exhibit a significant decrease in food and water consumption in 4 weeks following the treatment.
Noticeable signs of toxicity were observed when β-amyrin was administered orally at doses greater than 30 mg/kg. The toxicity signs included tremor, ataxia, increased respiration and decreased activity.
Transmission electron microscopy
Ultrastructural analysis of the liver sections revealed significantly lower organelle injury scores in DMN- and β-amyrin-treated group when compared to rats intoxicated with DMN alone (Figure 1). Disintegrated chromatin distribution and large dilation were observed in DMN-intoxicated rats, whereas in β-amyrin-treated rats, less mitochondrial dilation was observed.

TEM analysis of liver tissue. (a) indicates Negative control rats contain normal ucleus (N) and mitochondria with a prominent edema (small arrow); (b) indicates positive control rats treated with β-amyrin alone contain normal nucleus (N) and mitochondria with a prominent edema (small arrow); (c) indicates DMN-intoxicated rats contain disintegrated nuclei (DN) and irregular lamellar organization and large dilations with focal breaks in ERs of hepatocytes; (d)D indicates β-amyrin–treated DMN-intoxicated rats contain vocalization (V) in the ER, and normal nucleus (N). TEM: transmission electron microscopy; DMN: dimethylnitrosamine; ER: endoplasmic reticulum.
Liver enzyme markers
The purpose of the study was to evaluate the therapeutic efficacy of β-amyrin in liver injury induced by DMN. Table 1 shows the effect of β-amyrin at a dose of 30 mg/kg on serum marker enzymes in DMN-induced hepatic injury. In animals with hepatic injury induced by DMN increase in the marker levels of AST, ALT, ALP and LDH compared to control (p < 0.001) was observed. Administration of β-amyrin at a 30 mg/kg dose attenuated the altered levels of the serum enzyme markers produced by DMN and caused a subsequent recovery toward normalization. This shows that β-amyrin restores the liver function.
Effects of β-amyrin on serum liver marker enzymes in DMN-induced hepatic fibrosis.a

DMN: dimethylnitrosamine; AST: aspartate transaminase; ALT: alanine transaminase; ALP: alkaline phosphatase; LDH: lactate dehydrogenase.
aAll values were expressed as mean ± SEM.
bComparison of group III with groups I and II.
c p < 0.001.
dComparison of group IV with group III.
Oxidative stress marker and antioxidant enzymes
Oxidative stress markers in liver-intoxicated rats were evaluated in β-amyrin-supplemented and control groups. In liver-intoxicated rats, malondialdehyde (MDA) level was significantly (p < 0.001) increased as compared to normal and vehicle control animals, as shown in Table 2. β-Amyrin supplementation significantly reduced the MDA level in DMN-induced rats. A significant (p < 0.001) decline in the antioxidant enzyme level (SOD, CAT, GPx and GSH) was recorded in DMN-administered rats compared with normal controls. After β-amyrin treatment in DMN-intoxicated rats, these enzymes attained a near-normalcy at a significant level of p < 0.001.
Effects of β-amyrin on oxidative stress marker and antioxidant marker in DMN-induced hepatic fibrosis.

DMN: dimethylnitrosamine; MDA: malondialdehyde; SOD: superoxide dismutase; CAT: catalase; GPX: glutathione peroxidase; GSH: glutathione reduced content.
aAll values were expressed as mean ± SEM.
bComparison of group III with groups I and II.
c p < 0.001.
dComparison of group IV with group III.
Serum TNF-α level
Figure 2 shows the serum TNF-α level in DMN-intoxicated rats before and after β-amyrin treatment. Serum TNF-α concentration in DMN-intoxicated rats was highly significant (p < 0.001) compared to controls. β-Amyrin treatment decreased the levels of TNF-α in DMN-intoxicated rats significantly (p < 0.001).

Effect of β-amyrin on serum TNF-α levels in DMN intoxicated rats. All values were expressed as mean ± SEM, where, (a) denotes comparison of group III with groups I and II; (b) denotes comparison of group IV with group III; $ denotes p < 0.001. TNF-α: tumor necrosis factor α; DMN: dimethylnitrosamine.
Protein expression and gene expression of apoptotic marker of liver
Figure 3(a) shows the protein expression of caspase 3 before and after β-amyrin supplementation in DMN-intoxicated rats. Expression of caspase 3 decreased in β-amyrin–treated and control rats compared to DMN-intoxicated rats. This was confirmed by Western blot and densitometry analysis (Figure 3(b)). Detection of cleaved caspase 3 in DMN-intoxicated and β-amyrin–treated rats were further analyzed by immunofluorescence assay (Figure 4).

Protein expression of apoptotic protein caspase 3 before and after β-amyrin treatment in DMN-induced rat (a). Representative Western blot image for the different groups of rats. (b). Densitiometric analysis of Western blot bands for caspase 3 was normalized to β-actin. All values were expressed as mean ± SEM, where, (a′) denotes comparison of group III with groups I and II; (b′) denotes comparison of group IV with group III ; $ denotes p < 0.001. DMN: dimethylnitrosamine.
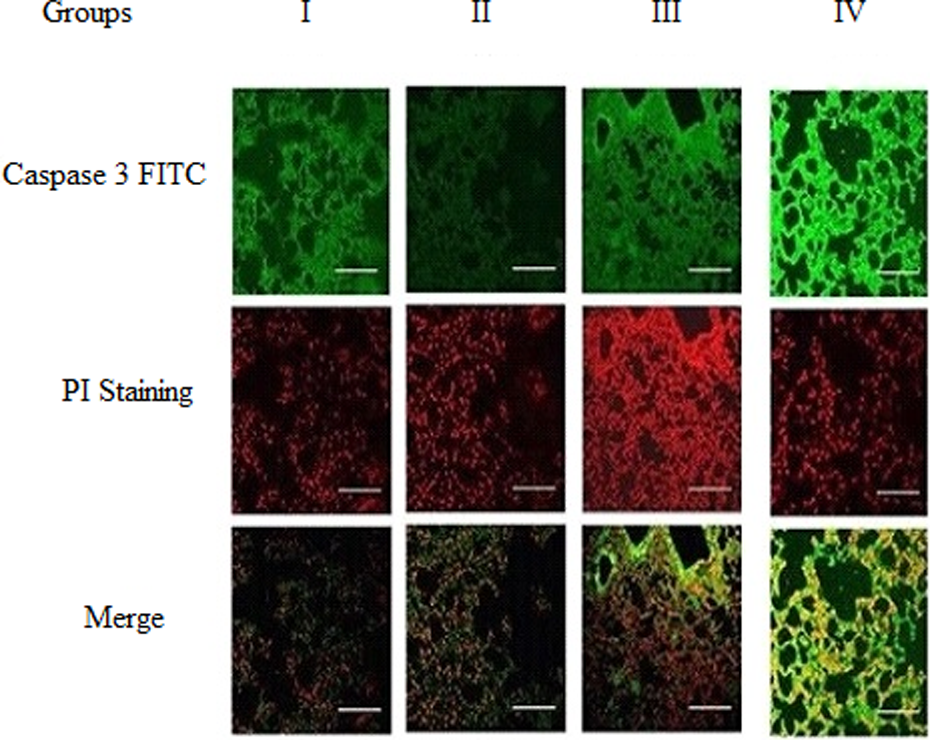
Localization of casapase 3 in liver tissue. Localization of caspase 3 was determined in liver tissue slides using double staining immunofluorescence microscopy. Slides were labeled with antibody caspase 3 followed by staining with secondary antibodies labeled with FITC (row 1). Slides were costained with PI to visualize dead cells (row 2). Merged images were evaluated to determine the localization of caspase 3 relative to dead cells in (row 3). (Scale bars, 5 μm, ×20.) FITC: fluorescein isothiocyanate; PI: propidium iodide.
Liver tissues from rats of different groups were tested for caspase 3 mRNA expression by reverse transcription (RT) PCR (Figure 5(a)). When caspase 3 bands of the different groups were densitometrically analyzed relative to the normal control group and normalized to β-actin levels, caspase 3 mRNA expression in the DMN-intoxicated group was found to be significantly (p < 0.001) elevated. β-Amyrin treatment of DMN-induced rats significantly (p < 0.001) reduced caspase 3 mRNA expression compared to DMN-intoxicated group, as shown in Figure 5(b).

mRNA expression of apoptotic protein caspase 3 before and after β-amyrin treatment in DMN-induced rat (a). Representative RT-PCR image of the different groups of rats. (b) Relative expression of caspase 3 mRNA of the different groups is expressed densitometrically as a percentage. Expression was normalized to corresponding internal control β-actin mRNA. Bars mean ± SEM, n = 6. Significant differences between groups were analyzed by one-way ANOVA, where, (a′) denotes comparison of group III with groups I and II; (b′) denotes comparison of group IV with group III, $ denotes p < 0.001. mRNA: messenger RNA; DMN: dimethylnitrosamine; RT-PCR: reverse transcription polymerase chain reaction; ANOVA: analysis of variance.
Protein expression of TIMP-1 in isolated HSCs
Protein expression of TIMP-1 before and after β-amyrin supplementation in DMN-intoxicated rats is shown in Figure 6(a). Western blot analysis of TIMP-1 showed increased expression in DMN-treated rat as compared to control. Densitometric analysis shows that treatment with β-amyrin significantly decreased TIMP-1 expression (p < 0.001) in DMN-intoxicated rats, as shown in Figure 6(b).

Protein expression of TIMP-1 in HSCs isolated from liver before and after β-amyrin treatment in DMN-induced rat. Western blot analysis of TIMP-1 (a). Representative Western blot image for the different groups of rats. (b) Densitometric analysis of the Western blot bands for TIMP-1 normalized to β-actin. All values were expressed as mean ± SEM, where, (a′) denotes comparison of group III with groups I and II and (b′) denotes comparison of group IV with group III; $ denotes p < 0.001. TIMP-1: tissue inhibitor of metalloproteinase 1; HSCs: hepatic stellate cells; DMN: dimethylnitrosamine.
Discussion
Protective effect of β-amyrin against DMN intoxication was assessed using animal model. Liver damage induced by DMN has been broadly studied for hepatoprotective drug screening. 27 Analysis of organ damage is aided by the measurement of a wide range of nonfunctional serum enzymes characteristic for that tissue or organ, but the amount of enzymes released depends on the degree of cellular damage, the intracellular concentration of the enzymes, and the mass of affected tissue. AST and ALT are enzymes normally present in the liver, heart, muscles, and blood cells. They are basically secreted by hepatocytes, and the concentration of the enzymes released reflects the severity of the damage.
β-Amyrin is known to decrease the leakage of serum marker enzymes AST, ALT, and ALP. 3 In this study, levels of AST, ALT, ALP, and LDH increased significantly in DMN-intoxicated rats compared to control, which indicates hepatic cell damage. Treatment with β-amyrin decreased the serum levels of AST, ALT, LDH, and ALP toward their normal value, which indicates the stabilization of plasma membrane as well as repair of hepatic tissue damage caused by DMN. This effect is in agreement with the commonly accepted view that serum levels of transaminases would return to normal after the healing of hepatic parenchyma and regeneration of hepatocytes. 28
The majority of the diseases or disorder is mainly due to oxidative stress, which is a consequence of an imbalance between the production of ROS and the available antioxidant defense against them. Hepatotoxicity, which is caused by drugs and chemicals, leads to impaired liver function. DMN, a well-known hepatotoxin, induces oxidative stress. 29 MDA is a commonly used biomarker of lipid peroxidation, which arises from the breakdown of lipid peroxyl radicals and is one of the important indicators of oxidative stress. Measured levels of MDA can be considered a direct index of oxidative injuries associated with lipid peroxidation. In this context, a marked increase in the concentration of MDA indicates oxidative stress in DMN-intoxicated rats when compared to control rats. There was reduction in antioxidant enzyme (SOD, GPx, and GST) and nonenzyme (GSH and vitamin C) activities and increased MDA in the liver by DMN. β-Amyrin replenish depleted hepatic GSH, reduce liver damage and suppress cytochrome P450 leading to hepatoprotection. β-Amyrin treatment significantly decreased the level of MDA and increased the antioxidant enzymes, which demonstrate that β-amyrin detoxicate the DMN-induced liver injury by reducing the oxidative stress. Thereby, β-amyrin role in hepatoprotection is by inhibiting the free radical–mediated damage, which elucidates that it acted as an antioxidant agent.
In the liver, proinflammatory cytokines like IL-6 and TNF-α produced by hepatocytes participate in hepatotoxic responses. 30 In the present study, expression of TNF-α level was high in DMN-intoxicated rats. This may be due to the impaired electron flow caused by DMN, which produces ROS in oxidative stress. Oxidative stress causes damage to hepatic macrophages, which in turn generates various inflammatory mediators and cytokines that modulate the phenotype of neighboring hepatocytes and other immune cells that travel through the liver. 31 Similar to the infiltration of lipoprotein particles into the arterial wall, fat accumulation in the liver stimulates hepatic cytokine production, which could further contribute to the increased TNF-α level. This study demonstrated the role of β-amyrin in reducing inflammatory markers of liver damage. β-Amyrin significantly decreased the level of TNF-α. Reduction in oxidative stress markers after β-amyrin treatment may be one of the reasons for the decrease in TNF-α levels. Study by da Silva et al. (2011) 32 showed that activation of cannabinoid receptor by β-amyrin causes the inhibition of TNF-α and prevention of nuclear factor κB (NFκB) activation. A recent study has found that the anti-fibrotic efficacy of liver fibrosis depends on the reduction of TNF-α levels and prevention of NFκB activation. 33 Based on this, our results evidenced the anti-inflammatory and antifibrotic efficacy of β-amyrin. DMN-induced inflammatory mediators in liver is normalized by β-amyrin.
The most direct and intuitive hypothesis is that hepatocyte death provides a signal for the development of liver fibrosis. The connection between hepatocyte death and fibrosis can be evidenced in chronic hepatitis C infection where apoptosis and caspase activity correlate with the histological activity of the disease. The same has been shown in experimental models of liver fibrosis. 34 In the present study, apoptotic marker caspase 3 expression was high in DMN-intoxicated rats, which indicates severe liver fibrosis. Many studies have shown that TNF-α signaling induced the hepatocyte apoptosis. 35 TNF-α exerts a variety of effects in cell death, which are mediated mainly by TNF-receptor 1 (TNF-R1). Activation of TNF-R1 stimulates multiple apoptotic pathways involved in the activation of pro-death B-cell lymphoma 2 family proteins C-Jun NH2-terminal kinase, cathepsin B, acidic sphingomyelinase, and neutral sphingomyelinase. These pathways are closely interlinked and mainly act on mitochondria, which releases the apoptogenic factors such as caspase 9 and caspase 3, thus resulting in apoptosis. The present study demonstrated the role of β-amyrin in regulating the apoptotic markers by decreasing the expression of caspase 3 in DMN-intoxicated rats. Immunofluorescence and RT-PCR analysis of caspase 3 further confirmed the apoptotic property of β-amyrin.
Activation of HSCs, which is both proliferative and fibrogenic, is an important event in the pathogenesis of liver. It is believed that activated HSCs and Kupffer cells are the major sources for TIMP-1 production in liver injury. TIMP-1 expression was found to be increased in DMN-intoxicated rats. Previous studies have proven that expression of both TIMP-1 mRNA and proteins are upregulated by inflammatory cytokines in rat hepatocytes. The inducible TIMP-1 can modulate the expression of fibrosis-related genes in the liver. Upregulation of TIMP-1 and activated HSCs can inhibit the activity of interstitial collagenases, which favors the accumulation of fibrotic tissue.14 After β-amyrin treatment, the level of TIMP-1 expression decreased. This decrease may be due to two factors: reduction in hepatocyte apoptosis and reduction in HSC activation. All these factors lead to an increase in the degradation of ECM, a major mechanism of fibrosis resolution. This indicates the anti-fibrotic property of β-amyrin.
Since DMN results in hepatotoxicity through its enzymatic conversion to reactive free radicals, 36 antioxidant treatment is essential to be effective in reducing the degree of injury. As reported in several studies, β-amyrin treatment inhibits the production of ROS in experimental rats due to its antioxidant properties. 37 We found notable evidence under transmission electron microscopy that β-amyrin decreased the cellular damage, fibrosis, and inflammation, which gives a better measurement of ultrastructural injury.
Efficacy of β-amyrin against DMN-induced liver injury is proved by reducing TNF-α and caspase 3 levels. The underlying mechanism for this may be achieved by the antioxidant property of β-amyrin, which block the DMN-induced ROS-mediated oxidative stress pathway in liver. In conclusion, efficacy of β-amyrin may give a new insight into a major therapeutic approach against DMN-intoxicated liver in rat. By virtue of β-amyrin properties of inhibiting oxidative stress, apoptosis, inflammation, and fibrogenesis, it might be act as an ideal anti-inflammatory and anti fibrogenic agent to halt the progression of liver fibrosis to chronicity.
Footnotes
Acknowledgement
The authors are very grateful to Dr Tamilselvi Elango for her support in completing this research work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
